Aspergillus fumigatus Detection and Risk Factors in Patients with COPD–Bronchiectasis Overlap
Abstract
:1. Introduction
2. Results
2.1. Study Group Characteristics
2.2. A. fumigatus Antibodies
2.3. Fungal Sputum Culture
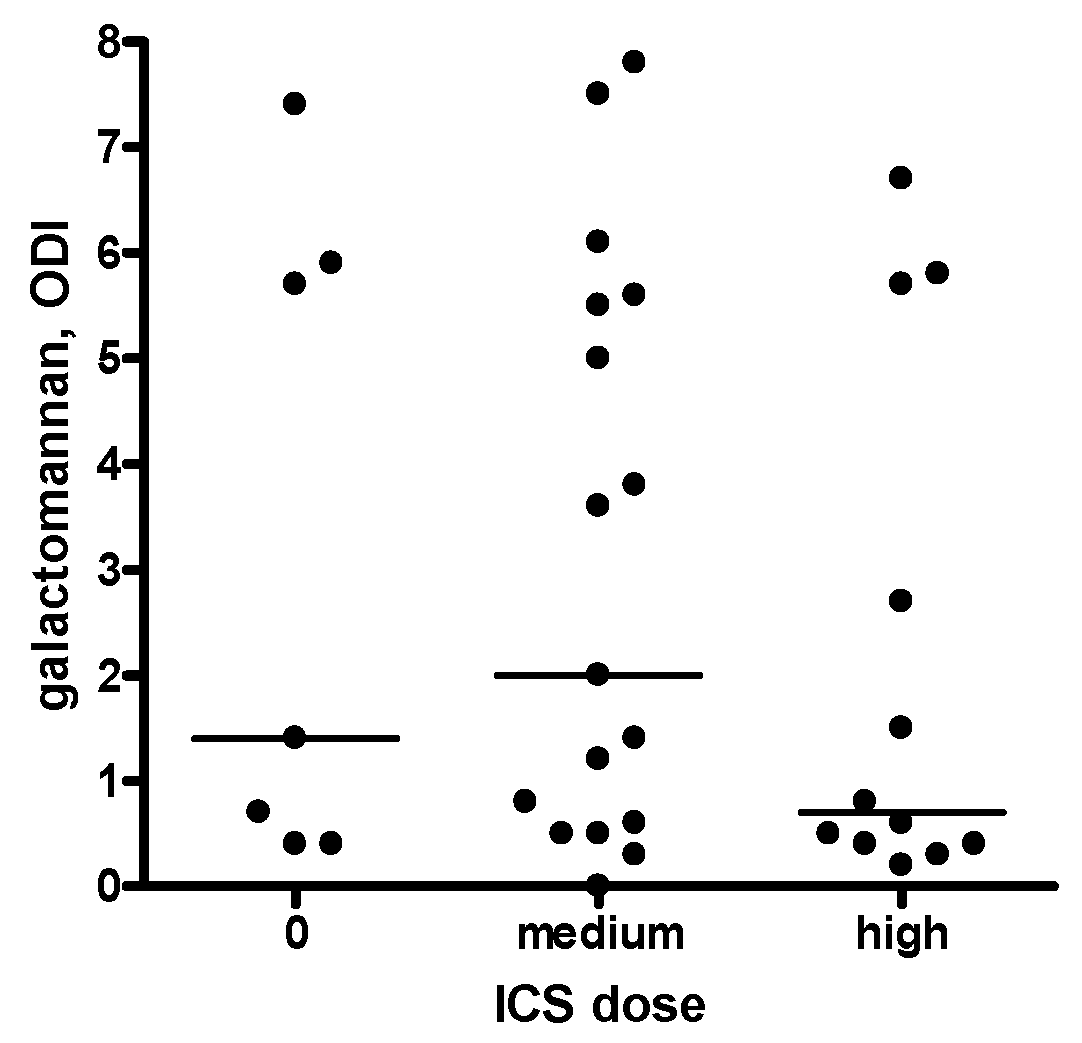
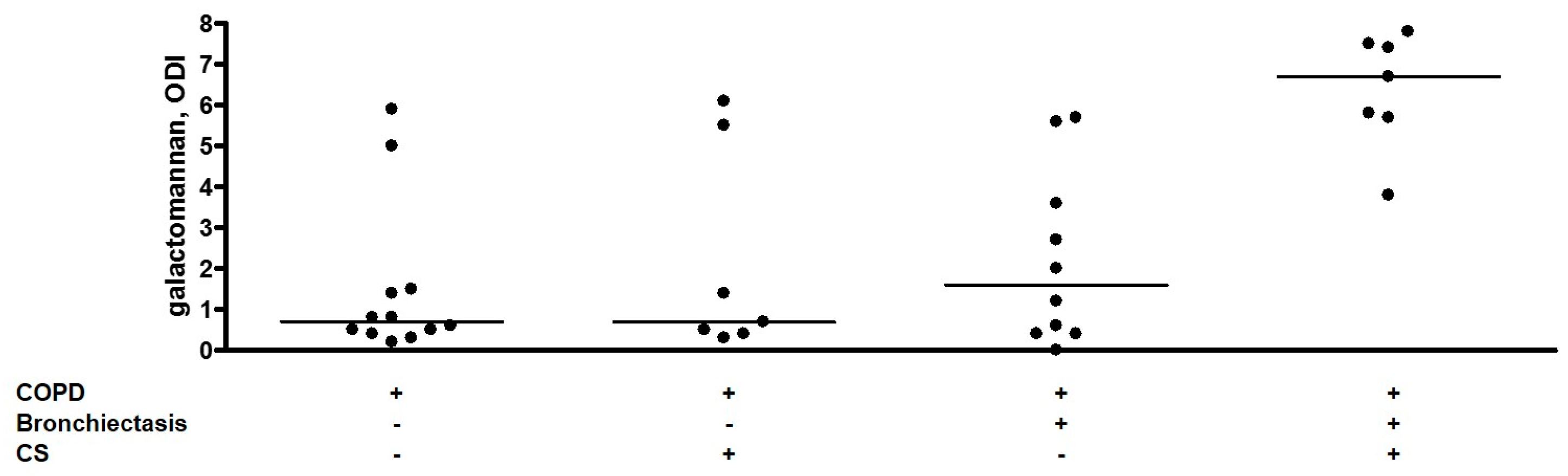
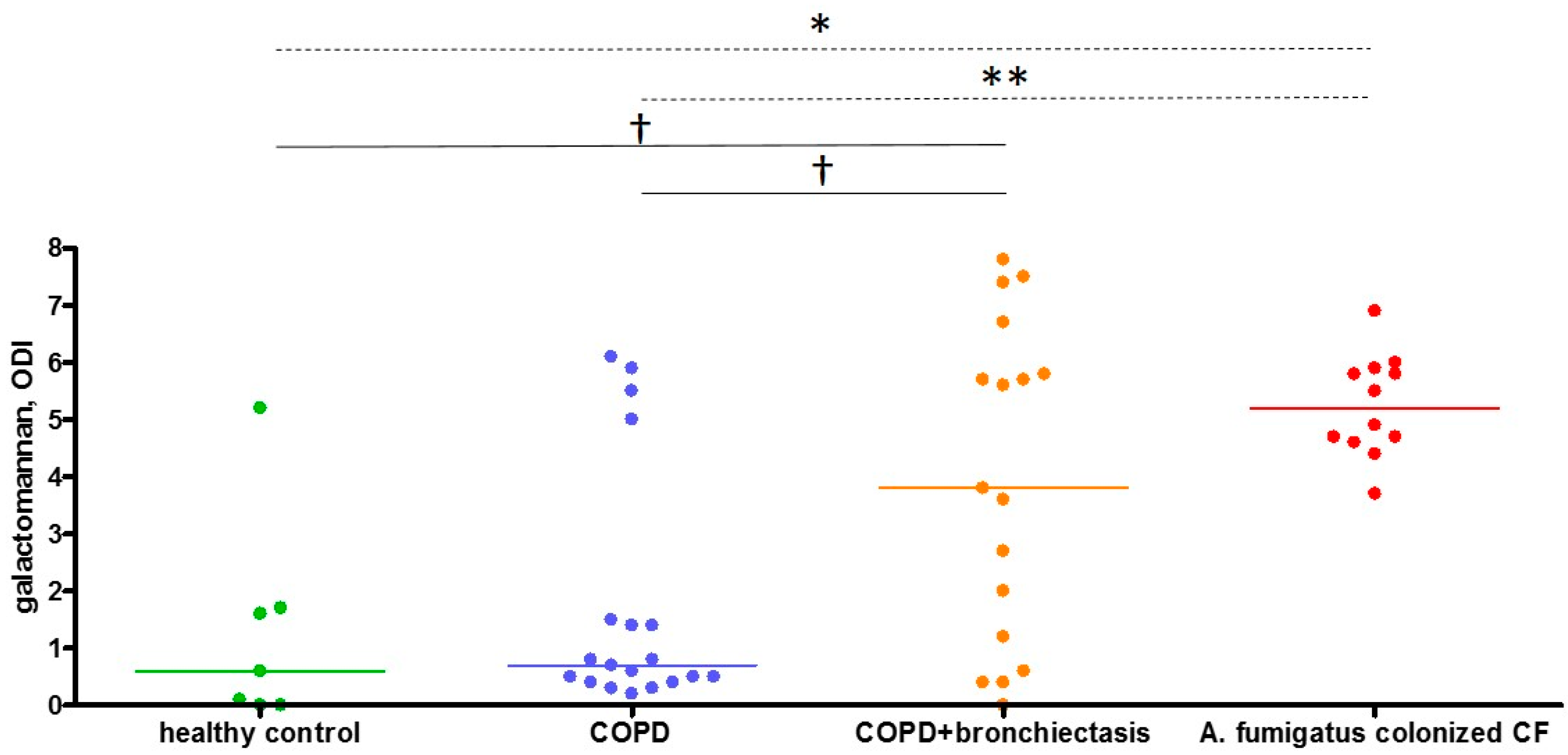
2.4. Sputum Galactomannan
2.5. Sputum A. fumigatus PCR
3. Discussion
4. Materials and Methods
4.1. Study Design and Subjects
4.2. Pulmonary Function and Questionnaires
4.3. CT Thorax
4.4. Eosinophils, Total IgE, A. fumigatus Sensitization, and A. fumigatus-Specific IgG
4.5. Sputum Collection, Homogenization, and Culture
4.6. Sputum Galactomannan Assay
4.7. Aspergillus PCR on Sputum
4.8. Statistical Analysis
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Vogelmeier, C.F.; Criner, G.J.; Martinez, F.J.; Anzueto, A.; Barnes, P.J.; Bourbeau, J.; Celli, B.R.; Chen, R.; Decramer, M.; Fabbri, L.M.; et al. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Lung Disease 2017 Report. GOLD Executive Summary. Am. J. Respir. Crit. Care Med. 2017, 195, 557–582. [Google Scholar] [CrossRef] [PubMed]
- Martínez-García, M.-A.; de la Rosa Carrillo, D.; Soler-Cataluña, J.-J.; Donat-Sanz, Y.; Serra, P.C.; Lerma, M.A.; Ballestín, J.; Sánchez, I.V.; Selma Ferrer, M.J.; Dalfo, A.R.; et al. Prognostic Value of Bronchiectasis in Patients with Moderate-to-Severe Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2013, 187, 823–831. [Google Scholar] [CrossRef] [PubMed]
- Jairam, P.M.; van der Graaf, Y.; Lammers, J.-W.J.; Mali, W.P.T.M.; de Jong, P.A. PROVIDI Study group Incidental findings on chest CT imaging are associated with increased COPD exacerbations and mortality. Thorax 2015, 70, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, S.M.D.; Paschoal, I.A.; de Capitani, E.M.; Moreira, M.M.; Palhares, L.C.; Pereira, M.C. COPD phenotypes on computed tomography and its correlation with selected lung function variables in severe patients. Int. J. Chronic Obstr. Pulm. Dis. 2016, 11, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Everaerts, S.; Lagrou, K.; Dubbeldam, A.; Lorent, N.; Vermeersch, K.; Van Hoeyveld, E.; Bossuyt, X.; Dupont, L.J.; Vanaudenaerde, B.M.; Janssens, W. Sensitization to Aspergillus fumigatus as a risk factor for bronchiectasis in COPD. Int. J. Chronic Obstr. Pulm. Dis. 2017, 12, 2629–2638. [Google Scholar] [CrossRef] [PubMed]
- Quint, J.K.; Millett, E.R.C.; Joshi, M.; Navaratnam, V.; Thomas, S.L.; Hurst, J.R.; Smeeth, L.; Brown, J.S. Changes in the incidence, prevalence and mortality of bronchiectasis in the UK from 2004 to 2013: A population-based cohort study. Eur. Respir. J. 2016, 47, 186–193. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, M.J.; Aliberti, S.; Goeminne, P.C.; Restrepo, M.I.; Finch, S.; Pesci, A.; Dupont, L.J.; Fardon, T.C.; Wilson, R.; Loebinger, M.R.; et al. Comorbidities and the risk of mortality in patients with bronchiectasis: An international multicentre cohort study. Lancet Respir. Med. 2016, 4, 969–979. [Google Scholar] [CrossRef]
- Du, Q.; Jin, J.; Liu, X.; Sun, Y. Bronchiectasis as a Comorbidity of Chronic Obstructive Pulmonary Disease: A Systematic Review and Meta-Analysis. PLoS ONE 2016, 11, e0150532. [Google Scholar] [CrossRef]
- Cole, P.J. Inflammation: A two-edged sword—The model of bronchiectasis. Eur. J. Respir. Dis. Suppl. 1986, 147, 6–15. [Google Scholar] [PubMed]
- Agbetile, J.; Fairs, A.; Desai, D.; Hargadon, B.; Bourne, M.; Mutalithas, K.; Edwards, R.; Morley, J.P.; Monteiro, W.R.; Kulkarni, N.S.; et al. Isolation of filamentous fungi from sputum in asthma is associated with reduced post-bronchodilator FEV1. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2012, 42, 782–791. [Google Scholar] [CrossRef] [PubMed]
- Amin, R.; Dupuis, A.; Aaron, S.D.; Ratjen, F. The effect of chronic infection with Aspergillus fumigatus on lung function and hospitalization in patients with cystic fibrosis. Chest 2010, 137, 171–176. [Google Scholar] [CrossRef] [PubMed]
- Fairs, A.; Agbetile, J.; Hargadon, B.; Bourne, M.; Monteiro, W.R.; Brightling, C.E.; Bradding, P.; Green, R.H.; Mutalithas, K.; Desai, D.; et al. IgE sensitization to Aspergillus fumigatus is associated with reduced lung function in asthma. Am. J. Respir. Crit. Care Med. 2010, 182, 1362–1368. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.; Panjabi, C. Allergic aspergillosis of the respiratory tract. Eur. Respir. Rev. Off. J. Eur. Respir. Soc. 2014, 23, 8–29. [Google Scholar] [CrossRef] [PubMed]
- Huerta, A.; Soler, N.; Esperatti, M.; Guerrero, M.; Menendez, R.; Gimeno, A.; Zalacaín, R.; Mir, N.; Aguado, J.M.; Torres, A. Importance of Aspergillus spp. isolation in Acute exacerbations of severe COPD: Prevalence, factors and follow-up: The FUNGI-COPD study. Respir. Res. 2014, 15, 17. [Google Scholar] [CrossRef] [PubMed]
- Guinea, J.; Torres-Narbona, M.; Gijón, P.; Muñoz, P.; Pozo, F.; Peláez, T.; de Miguel, J.; Bouza, E. Pulmonary aspergillosis in patients with chronic obstructive pulmonary disease: Incidence, risk factors, and outcome. Clin. Microbiol. Infect. 2010, 16, 870–877. [Google Scholar] [CrossRef] [PubMed]
- Barberán, J.; García-Pérez, F.-J.; Villena, V.; Fernández-Villar, A.; Malmierca, E.; Salas, C.; Giménez, M.-J.; Granizo, J.-J.; Aguilar, L. working group on Infectious Diseases from the Spanish Society of Internal Medicine Development of Aspergillosis in a cohort of non-neutropenic, non-transplant patients colonised by Aspergillus spp. BMC Infect. Dis. 2017, 17, 34. [Google Scholar] [CrossRef]
- Pashley, C.H. Fungal culture and sensitisation in asthma, cystic fibrosis and chronic obstructive pulmonary disorder: What does it tell us? Mycopathologia 2014, 178, 457–463. [Google Scholar] [CrossRef] [PubMed]
- Pashley, C.H.; Fairs, A.; Morley, J.P.; Tailor, S.; Agbetile, J.; Bafadhel, M.; Brightling, C.E.; Wardlaw, A.J. Routine processing procedures for isolating filamentous fungi from respiratory sputum samples may underestimate fungal prevalence. Med. Mycol. 2012, 50, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Bafadhel, M.; Mckenna, S.; Agbetile, J.; Fairs, A.; Desai, D.; Mistry, V.; Morley, J.P.; Pancholi, M.; Pavord, I.D.; Wardlaw, A.J.; et al. Aspergillus fumigatus during stable state and exacerbations of COPD. Eur. Respir. J. 2014, 43, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Fraczek, M.G.; Kirwan, M.B.; Moore, C.B.; Morris, J.; Denning, D.W.; Richardson, M.D. Volume dependency for culture of fungi from respiratory secretions and increased sensitivity of Aspergillus quantitative PCR. Mycoses 2014, 57, 69–78. [Google Scholar] [CrossRef] [PubMed]
- Kimura, S.; Odawara, J.; Aoki, T.; Yamakura, M.; Takeuchi, M.; Matsue, K. Detection of sputum Aspergillus galactomannan for diagnosis of invasive pulmonary aspergillosis in haematological patients. Int. J. Hematol. 2009, 90, 463–470. [Google Scholar] [CrossRef] [PubMed]
- Baxter, C.G.; Rautemaa, R.; Jones, A.M.; Webb, A.K.; Bull, M.; Mahenthiralingam, E.; Denning, D.W. Intravenous antibiotics reduce the presence of Aspergillus in adult cystic fibrosis sputum. Thorax 2013, 68, 652–657. [Google Scholar] [CrossRef] [PubMed]
- Fayemiwo, S.; Moore, C.B.; Foden, P.; Denning, D.W.; Richardson, M.D. Comparative performance of Aspergillus galactomannan ELISA and PCR in sputum from patients with ABPA and CPA. J. Microbiol. Methods 2017, 140, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Oguma, T.; Asano, K.; Tomomatsu, K.; Kodama, M.; Fukunaga, K.; Shiomi, T.; Ohmori, N.; Ueda, S.; Takihara, T.; Shiraishi, Y.; et al. Induction of mucin and MUC5AC expression by the protease activity of Aspergillus fumigatus in airway epithelial cells. J. Immunol. 2011, 187, 999–1005. [Google Scholar] [CrossRef] [PubMed]
- Namvar, S.; Warn, P.; Farnell, E.; Bromley, M.; Fraczek, M.; Bowyer, P.; Herrick, S. Aspergillus fumigatus proteases, Asp f 5 and Asp f 13, are essential for airway inflammation and remodelling in a murine inhalation model. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 982–993. [Google Scholar] [CrossRef] [PubMed]
- Miller, M.R.; Hankinson, J.; Brusasco, V.; Burgos, F.; Casaburi, R.; Coates, A.; Crapo, R.; Enright, P.; van der Grinten, C.P.M.; Gustafsson, P.; et al. ATS/ERS Task Force Standardisation of spirometry. Eur. Respir. J. 2005, 26, 319–338. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, E. The 1995 update of recommendations for a standard technique for measuring the single-breath carbon monoxide diffusing capacity (transfer factor). Am. J. Respir. Crit. Care Med. 1996, 154, 827–828. [Google Scholar] [CrossRef] [PubMed]
- Bestall, J.C.; Paul, E.A.; Garrod, R.; Garnham, R.; Jones, P.W.; Wedzicha, J.A. Usefulness of the Medical Research Council (MRC) dyspnoea scale as a measure of disability in patients with chronic obstructive pulmonary disease. Thorax 1999, 54, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Jones, P.W.; Harding, G.; Berry, P.; Wiklund, I.; Chen, W.-H.; Kline Leidy, N. Development and first validation of the COPD Assessment Test. Eur. Respir. J. 2009, 34, 648–654. [Google Scholar] [CrossRef] [PubMed]
- Naidich, D.P.; McCauley, D.I.; Khouri, N.F.; Stitik, F.P.; Siegelman, S.S. Computed tomography of bronchiectasis. J. Comput. Assist. Tomogr. 1982, 6, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, J.D.; Goeminne, P.; Aliberti, S.; McDonnell, M.J.; Lonni, S.; Davidson, J.; Poppelwell, L.; Salih, W.; Pesci, A.; Dupont, L.J.; et al. The Bronchiectasis Severity Index. An International Derivation and Validation Study. Am. J. Respir. Crit. Care Med. 2014, 189, 576–585. [Google Scholar] [CrossRef] [PubMed]
- Heinzerling, L.; Mari, A.; Bergmann, K.-C.; Bresciani, M.; Burbach, G.; Darsow, U.; Durham, S.; Fokkens, W.; Gjomarkaj, M.; Haahtela, T.; et al. The skin prick test—European standards. Clin. Transl. Allergy 2013, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- Paggiaro, P.L.; Chanez, P.; Holz, O.; Ind, P.W.; Djukanović, R.; Maestrelli, P.; Sterk, P.J. Sputum induction. Eur. Respir. J. Suppl. 2002, 37, 3s–8s. [Google Scholar] [PubMed]
- Mattei, D.; Rapezzi, D.; Mordini, N.; Cuda, F.; lo Nigro, C.; Musso, M.; Arnelli, A.; Cagnassi, S.; Gallamini, A. False-positive Aspergillus galactomannan enzyme-linked immunosorbent assay results in vivo during amoxicillin-clavulanic acid treatment. J. Clin. Microbiol. 2004, 42, 5362–5363. [Google Scholar] [CrossRef] [PubMed]
| characteristic | COPD without Bronchiectasis | COPD with Bronchiectasis |
|---|---|---|
| Subjects, n | 19 | 17 |
| Hospitalized, % | 42 | 65 |
| Age, y | 71 (60–75) | 68 (63–79) |
| Male, % | 84 | 88 |
| BMI, kg/m² | 25 (20–30) | 22 (19–27) |
| Pack-years * | 48 (40–55) | 40 (25–45) |
| FEV1, % pred | 51 (39–58) | 41 (37–52) |
| DLCO, % pred | 47 (40–57) | 44 (26–53) |
| GOLD | ||
| I/II/III/IV, % | 0/53/47/0 | 6/29/65/0 |
| A/B/C/D, % | 21/0/16/63 | 12/17/6/65 |
| Eosinophils, % | 1.4 (0.1–3.1) | 2.3 (0.8–4.6) |
| Eosinophils, μL | 200 (0–300) | 200 (100–350) |
| Total IgE, kU/L | 83 (16–592) | 43 (33–298) |
| mMRC | 2 (1–2) | 2 (1.5–3.5) |
| CAT | 14 (12–20) | 19 (13–24) |
| SGRQ | 41.9 (30.6–53.4) | 54.9 (37.3–66.7) |
| ≥2 exacerbations/y, % | 79 | 71 |
| ICS, % | 89 | 82 |
| Modified Reiff score | NA | 4 (2.5–6.5) |
| Test | COPD without Bronchiectasis | COPD with Bronchiectasis | p-Value |
|---|---|---|---|
| Subjects, n | 19 | 17 | |
| A. fumigatus IgG, mg/L | 17.4 (13.4–37.3) | 35.4 (25.5–51.6) | 0.14 |
| A. fumigatus sensitization, % | 15.7 | 29.4 | 0.37 |
| A. fumigatus sputum culture, % | 10.5 | 23.5 | 0.39 |
| Sputum galactomannan, ODI | 0.7 (0.4–1.5) | 3.7 (0.6–5.7) | 0.026 |
| Bivariate Models | ||
| Estimate | p-Value | |
| CS in COPD without bronchiectasis (n = 19) | 0.64 | 0.54 |
| CS in COPD with bronchiectasis (n = 17) | 4.17 | 0.0004 |
| Multivariate Model | ||
| Estimate | p-Value | |
| Bronchiectasis | 0.67 | 0.0028 |
| CS | 0.57 | 0.0044 |
| Antibiotics | 0.37 | 0.63 |
| Bronchiectasis–CS interaction | 3.43 | 0.022 |
| Subject | Sputum Galactomannan, ODI | PCR after EasyMAG Extraction, Ct | PCR after MycXtra® Extraction, Ct |
|---|---|---|---|
| CF1 | 4.4 | negative | positive, 36.7 |
| CF2 | 3.7 | negative | positive, 32.9 |
| CF3 | 4.7 | positive, 29.3 | positive, 26.2 |
| CF4 | 4.6 | positive, 30.7 | positive, 30.5 |
| CF5 | 4.7 | negative | positive, 33.2 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Everaerts, S.; Lagrou, K.; Vermeersch, K.; Dupont, L.J.; Vanaudenaerde, B.M.; Janssens, W. Aspergillus fumigatus Detection and Risk Factors in Patients with COPD–Bronchiectasis Overlap. Int. J. Mol. Sci. 2018, 19, 523. https://doi.org/10.3390/ijms19020523
Everaerts S, Lagrou K, Vermeersch K, Dupont LJ, Vanaudenaerde BM, Janssens W. Aspergillus fumigatus Detection and Risk Factors in Patients with COPD–Bronchiectasis Overlap. International Journal of Molecular Sciences. 2018; 19(2):523. https://doi.org/10.3390/ijms19020523
Chicago/Turabian StyleEveraerts, Stephanie, Katrien Lagrou, Kristina Vermeersch, Lieven J. Dupont, Bart M. Vanaudenaerde, and Wim Janssens. 2018. "Aspergillus fumigatus Detection and Risk Factors in Patients with COPD–Bronchiectasis Overlap" International Journal of Molecular Sciences 19, no. 2: 523. https://doi.org/10.3390/ijms19020523
APA StyleEveraerts, S., Lagrou, K., Vermeersch, K., Dupont, L. J., Vanaudenaerde, B. M., & Janssens, W. (2018). Aspergillus fumigatus Detection and Risk Factors in Patients with COPD–Bronchiectasis Overlap. International Journal of Molecular Sciences, 19(2), 523. https://doi.org/10.3390/ijms19020523






