Expanding the Clinical Spectrum of Sotos Syndrome in a Patient with the New “c.[5867T>A]+[=]”; “p.[Leu1956Gln]+[=]” NSD1 Missense Mutation and Complex Skin Hamartoma
Abstract
1. Introduction
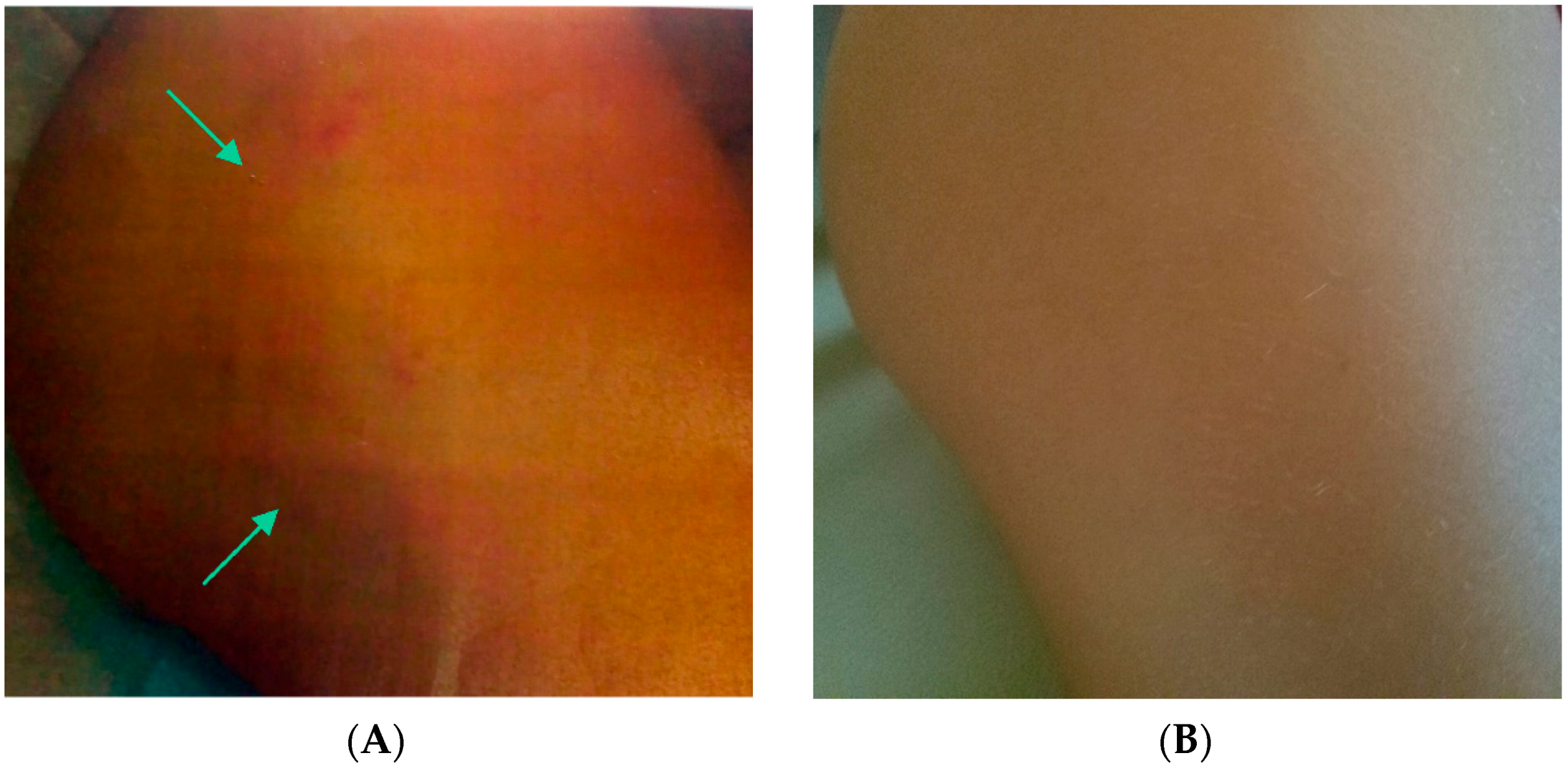
Case Report
2. Materials and Methods
2.1. Laboratory and Instrumental Analyses
2.2. Ethics Committee Approval
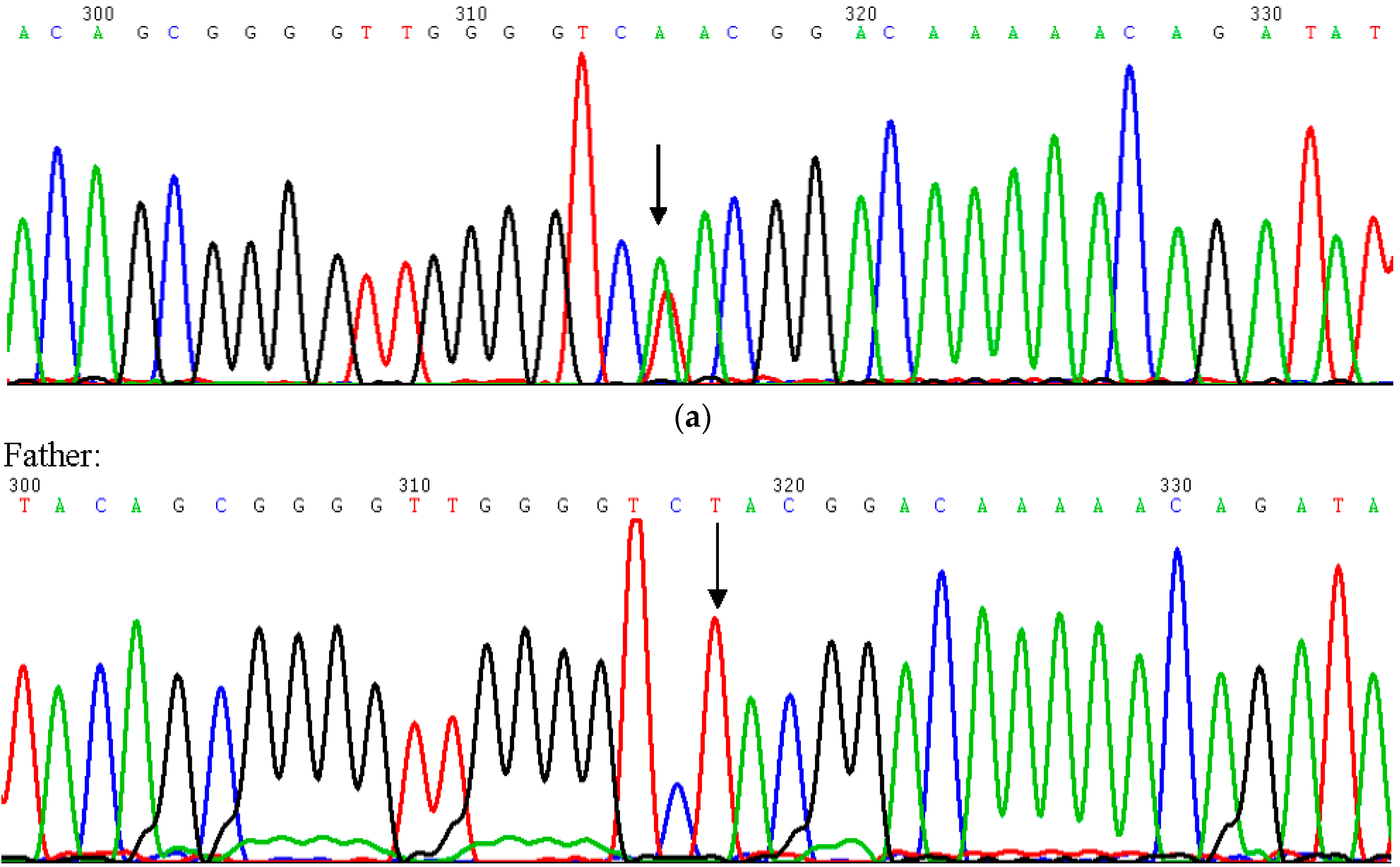
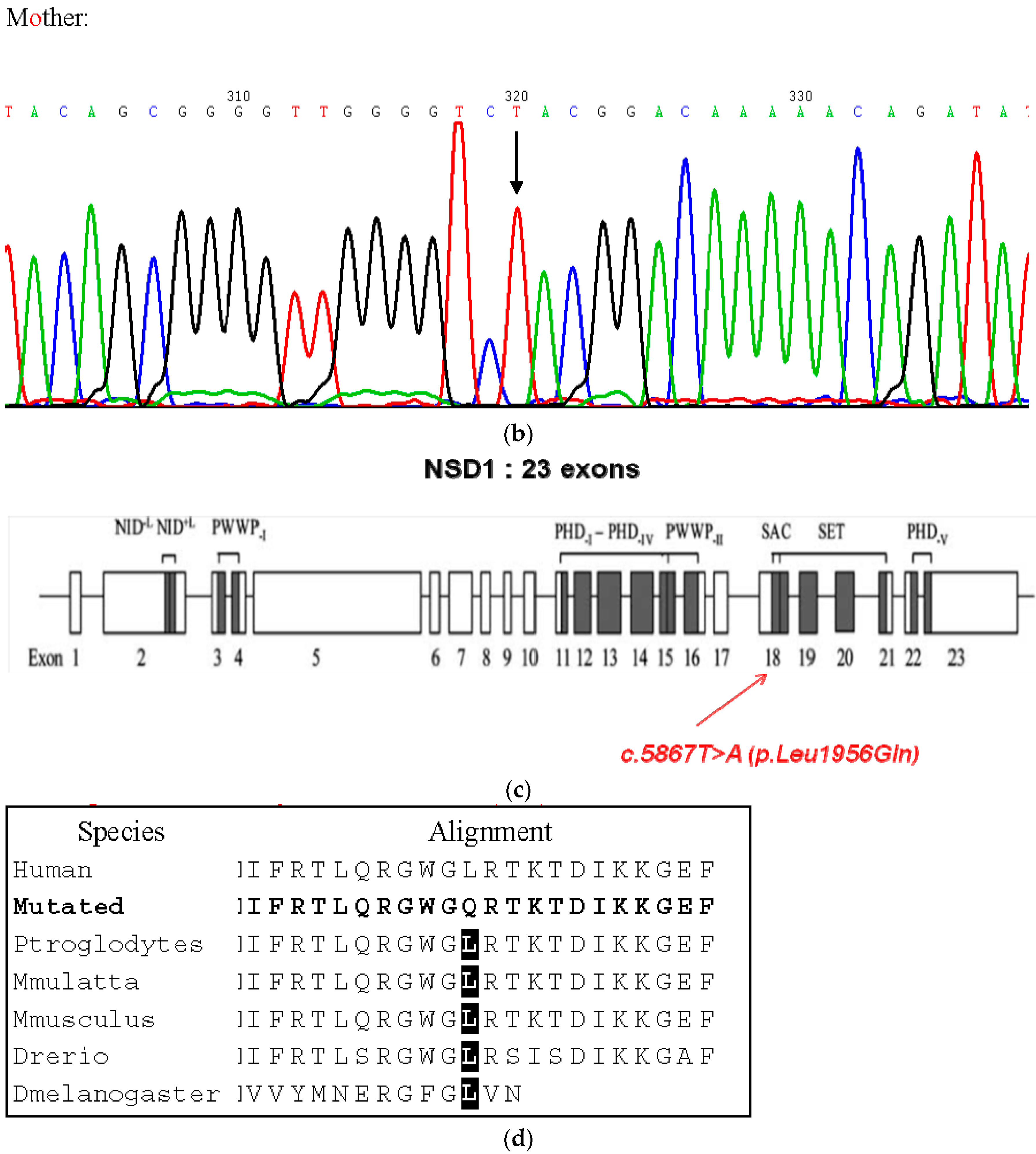
2.3. Genetic Analyses
3. Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Neylon, O.M.; Werther, G.A.; Sabin, G.A. Overgrowth syndromes. Curr. Opin. Pediatr. 2012, 24, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Visser, R.; Matsumoto, N. The genetics of Sotos syndrome. Curr. Opin. Pediatr. 2003, 15, 598–606. [Google Scholar] [CrossRef] [PubMed]
- Cole, T.R.; Hughes, H.E. Sotos syndrome: A study of the diagnostic criteria and natural history. J. Med. 1964, 271, 109–116. [Google Scholar] [CrossRef]
- Kurotaki, N.; Harada, N.; Yoshiura, K.; Sugano, S.; Niikawan, N.; Matsumoto, N. Molecular characterization of NSD1, a human homologue of the mouse NSD1 gene. Gene 2001, 279, 197–204. [Google Scholar] [CrossRef]
- Kurotaki, N.; Imaizumi, K.; Harada, N.; Masuno, M.; Kondoh, T.; Nagai, T.; Ohashi, H.; Naritomi, K.; Tsukahara, M.; Makita, Y.; et al. Haploinsufficiency of NSD1causes Sotos syndrome. Nat. Genet. 2002, 30, 365–366. [Google Scholar] [CrossRef] [PubMed]
- Hersh, J.H.; Cole, T.R.; Bloom, A.S.; Bertolone, S.J.; Hughes, H.E. Risk of malignancy in Sotos syndrome. J. Pediatr. 1992, 120 Pt 1, 572–574. [Google Scholar] [CrossRef]
- Villani, A.; Greer, M.C.; Kalish, J.M.; Nakagawara, A.; Nathanson, K.L.; Pajtler, K.W.; Pfister, S.M.; Walsh, M.F.; Wasserman, J.D.; Zelley, K.; et al. Recommendations for cancer surveillance in individuals with RASopathies and other rare genetic conditions with increased cancer risk. Clin. Cancer Res. 2017, 23, e83–e90. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, V.; Gaynon, P.S.; Poznanski, A.K. Cerebral gigantism associated with Wilms’ tumor. Am. J. Dis. Child. 1984, 138, 486–488. [Google Scholar] [CrossRef] [PubMed]
- Tatton-Brown, K.; Rahman, N. Sotos syndrome. Eur. J. Hum. Genet. 2007, 15, 264–271. [Google Scholar] [CrossRef] [PubMed]
- de Boer, L.; Van Duyvenvoorde, H.A.; Willemstein-Van Hove, E.C.; Hoogerbrugge, C.M.; Van Doorn, J.; Maassen, J.A.; Karperien, M.; Wit, J.M. Mutations in the NSD1 gene in patients with Sotos syndrome associate with endocrine and paracrine alterations in the IGF system. Eur. J. Endocrinol. 2004, 151, 333–341. [Google Scholar] [CrossRef] [PubMed]
- de Boer, L.; Roder, I.; Wit, J.M. Psychosocial, cognitive, and motor functioning in patients with suspected Sotos syndrome: A comparison between patients with or without NSD1 gene alterations. Dev. Med. Child. Neurol. 2006, 48, 582–588. [Google Scholar] [CrossRef] [PubMed]
- Tatton Brown, K.; Douglas, J.; Coleman, K.; Baujat, G.; Cole, T.; Das, S.; Horn, D.; Hughes, H.; Temple, I.K.; Faravelli, F.; et al. Genoype-phenotype associations in Sotos syndrome: An analysis of 266 individuals with NSD1 aberrations. Am. J. Hum. Genet. 2005, 77, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Rapaccini, V.; Esposito, S.; Strinati, F.; Allegretti, M.; Manfroi, E.; Miconi, F.; Pitzianti, M.; Prontera, P.; Principi, N.; Pasini, A. A child with a c.6923_6928dup (p.Arg2308_Met2309dup) SPTAN1 mutation associated with a severe early infantile epileptic encephalopathy. Int. J. Mol. Sci. 2018, 19, 1976. [Google Scholar] [CrossRef] [PubMed]
- Douglas, J.; Hanks, S.; Temple, I.K.; Davies, S.; Murray, A.; Upadhyaya, M.; Tomkins, S.; Hughes, H.E.; Cole, T.R.; Rahman, N. NSD1 mutations are the major cause of Sotos syndrome and occur in some cases of Weaver syndrome but are rare in other overgrowth phenotypes. Am. J. Hum. Genet. 2003, 72, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Jenuwein, T. Re-SET-ting heterochromatin by histone methyltransferases. Trends Cell Biol. 2001, 11, 266–273. [Google Scholar] [CrossRef]
- Van Rij, M.C.; Hollink, I.H.I.M.; Terhal, P.A.; Kant, S.G.; Ruivenkamp, C.; van Haeringen, A.; Kievit, J.A.; van Belzen, M.J. Two novel cases expanding the phenotype of SETD2-related overgrowth syndrome. Am. J. Med. Genet. A 2018, 176, 1212–1215. [Google Scholar] [CrossRef] [PubMed]
- Leventopoulos, G.; Kitsiou-Tzeli, S.; Kritikos, K.; Psoni, S.; Mavrou, A.; Kanavakis, E.; Fryssira, H. A clinic study of Sotos syndrome patients with review of the literature. Pediatr. Neurol. 2009, 40, 357–364. [Google Scholar] [CrossRef] [PubMed]
- Lapunzina, P. Risk of tumorigenesis in overgrowth syndromes: A comprehensive review. Am. J. Med. Genet. C 2005, 137C, 53–71. [Google Scholar] [CrossRef] [PubMed]
- Northrup, H.; Krueger, D.A. Tuberous sclerosis complex diagnostic criteria update: Recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatr. Neurol. 2013, 49, 243–254. [Google Scholar] [CrossRef] [PubMed]
- MacKeigan, J.P.; Krueger, D.A. Differentiating the mTor inhibitors everolimusand sirolimus in the treatment of tuberous sclerosis complex. Neuro Oncol. 2015, 17, 1550–1559. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi-Fakhari, D.; Meyer, S.; Vogt, T.; Pfohler, C.; Muller, C.S.L. Dermatological manifestations of tuberous sclerosis complex (TSC). J. Dtsch. Dermatol. Ges. 2017, 15, 695–700. [Google Scholar] [CrossRef] [PubMed]
- Hougland, P.; Kurotaki, N.; Kytola, S.; Miyake, N.; Somer, M.; Matsumoto, N. Familiar Sotos Syndrome is caused by a novel 1bp deletion of the NSD1 gene. J. Med. Genet. 2003, 40, 51–54. [Google Scholar] [CrossRef][Green Version]

| Features | Sotos Syndrome | Tuberous Sclerosis |
|---|---|---|
| Somatic overgrowth | +++ | − |
| Macrocephaly | ++ | − |
| Facial findings | ++ | − |
| Brain malformations | ++ | ++ |
| Seizures | +/− | +++ |
| Increased tumorigenesis | ++ | ++ |
| Multiple Hamartomas | − | ++ |
| Hypomelanotic macules | − | +++ |
| Learning disabilities | ++ | ++ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mencarelli, A.; Prontera, P.; Mencarelli, A.; Rogaia, D.; Stangoni, G.; Cecconi, M.; Esposito, S. Expanding the Clinical Spectrum of Sotos Syndrome in a Patient with the New “c.[5867T>A]+[=]”; “p.[Leu1956Gln]+[=]” NSD1 Missense Mutation and Complex Skin Hamartoma. Int. J. Mol. Sci. 2018, 19, 3189. https://doi.org/10.3390/ijms19103189
Mencarelli A, Prontera P, Mencarelli A, Rogaia D, Stangoni G, Cecconi M, Esposito S. Expanding the Clinical Spectrum of Sotos Syndrome in a Patient with the New “c.[5867T>A]+[=]”; “p.[Leu1956Gln]+[=]” NSD1 Missense Mutation and Complex Skin Hamartoma. International Journal of Molecular Sciences. 2018; 19(10):3189. https://doi.org/10.3390/ijms19103189
Chicago/Turabian StyleMencarelli, Annalisa, Paolo Prontera, Amedea Mencarelli, Daniela Rogaia, Gabriela Stangoni, Massimiliano Cecconi, and Susanna Esposito. 2018. "Expanding the Clinical Spectrum of Sotos Syndrome in a Patient with the New “c.[5867T>A]+[=]”; “p.[Leu1956Gln]+[=]” NSD1 Missense Mutation and Complex Skin Hamartoma" International Journal of Molecular Sciences 19, no. 10: 3189. https://doi.org/10.3390/ijms19103189
APA StyleMencarelli, A., Prontera, P., Mencarelli, A., Rogaia, D., Stangoni, G., Cecconi, M., & Esposito, S. (2018). Expanding the Clinical Spectrum of Sotos Syndrome in a Patient with the New “c.[5867T>A]+[=]”; “p.[Leu1956Gln]+[=]” NSD1 Missense Mutation and Complex Skin Hamartoma. International Journal of Molecular Sciences, 19(10), 3189. https://doi.org/10.3390/ijms19103189






