Neuroprotective and Nerve Regenerative Approaches for Treatment of Erectile Dysfunction after Cavernous Nerve Injury
Abstract
1. Introduction
2. Neurophysiology of Erection
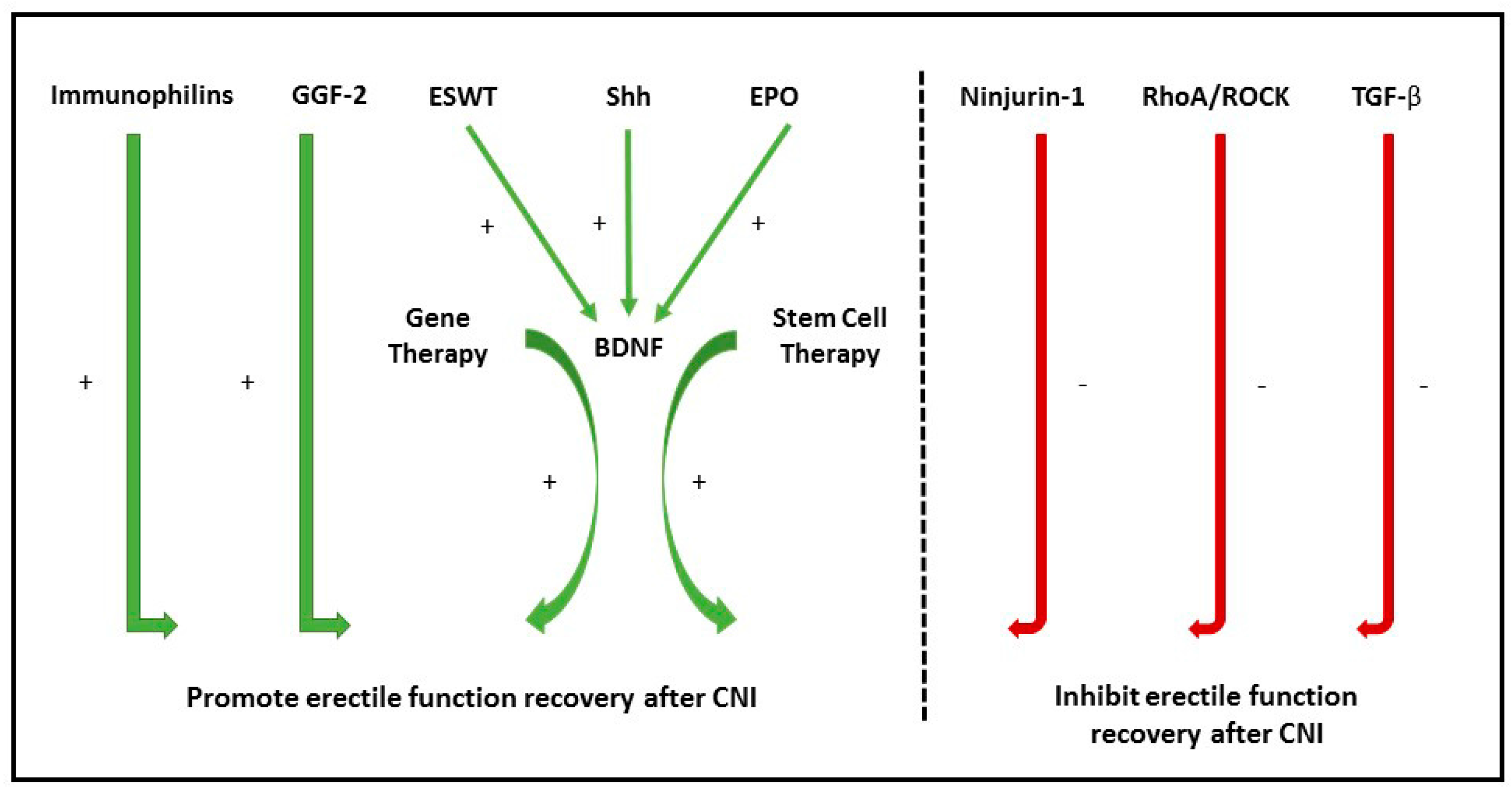
3. Nerve Regeneration Pathways in ED
3.1. Neurotrophins
3.2. Sonic Hegdehog Protein
3.3. Ninjurin-1
3.4. Neuregulins
3.5. Immunophilins
4. New Molecular Targets for Neurogenic ED
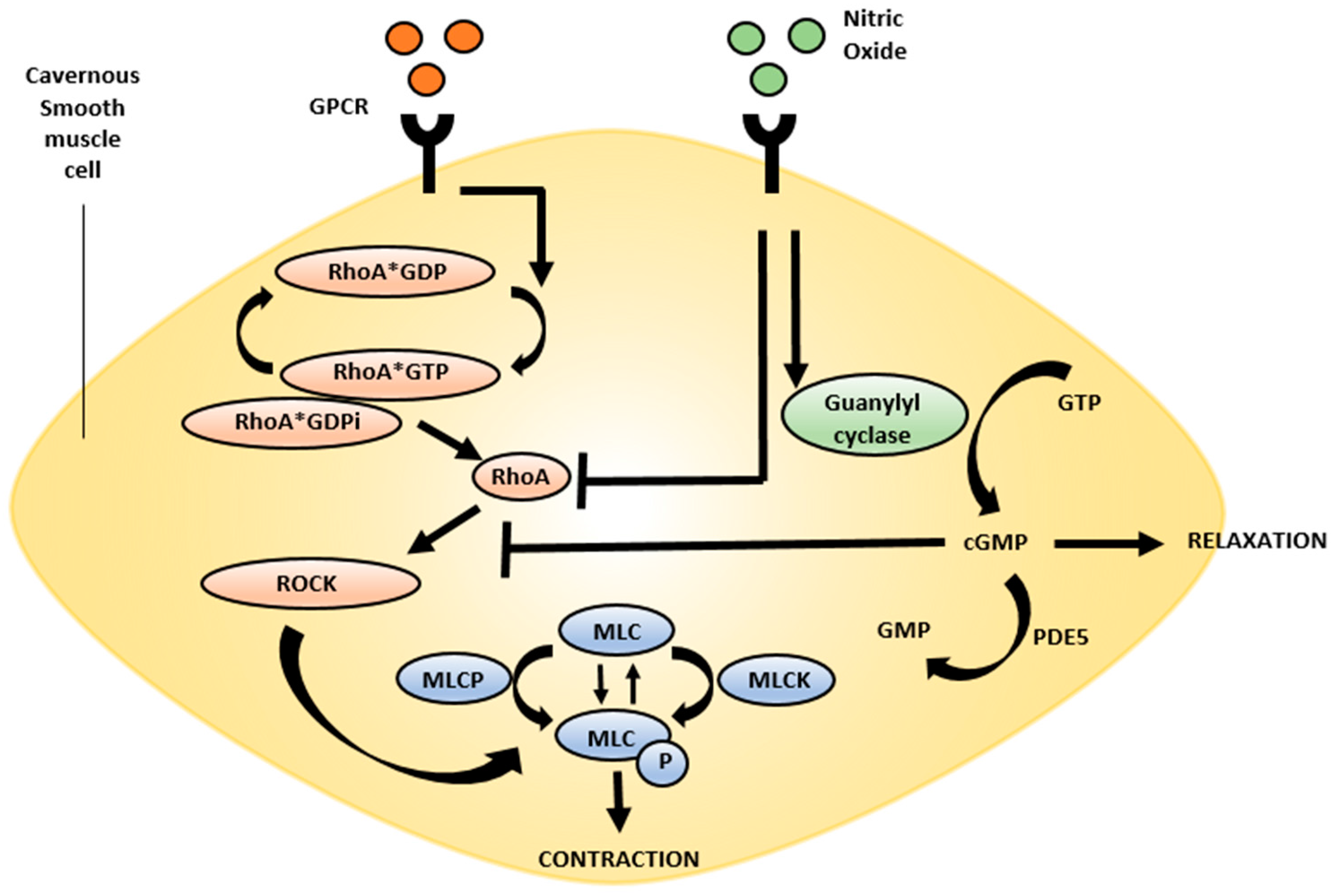
4.1. Rho Kinase Pathway
4.2. Transforming Growth Factor-β
4.3. Erythropoietin
5. Therapeutic Strategies for Neurogenic ED
5.1. Tacrolimus (FK506)
5.2. Rho-Kinase/ROCK Inhibitors
5.3. Valproic Acid
5.4. Erythropoietin
6. Regenerative Medicine for Neurogenic ED
6.1. Gene Therapy
6.2. Stem Cell Therapy
7. Surgical Strategies for Neuroprotection during Radical Prostatectomy
8. Conclusions
Author Contributions
Conflicts of Interest
References
- Bella, A.J.; Lee, J.C.; Carrier, S.; Bénard, F.; Brock, G.B. CUA practice guidelines for erectile dysfunction. Can. Urol. Assoc. J. 2015, 9, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Feldman, H.A.; Goldstein, I.; Hatzichristou, D.G.; Krane, R.J.; McKinlay, J.B. Impotence and its medical and psychosocial correlates: Results of the Massachusetts Male Aging Study. J. Urol. 1994, 151, 54–61. [Google Scholar] [CrossRef]
- Chew, K.K. Prevalence of erectile dysfunction in community-based studies. Int. J. Impot. Res. 2004, 16, 201–202. [Google Scholar] [CrossRef] [PubMed]
- McMahon, C.G. Erectile dysfunction. Intern. Med. J. 2014, 44, 18–26. [Google Scholar] [CrossRef] [PubMed]
- Corona, G.; Lee, D.M.; Forti, G.; O’Connor, D.B.; Maggi, M.; O’Neill, T.W.; Pendleton, N.; Bartfai, G.; Boonen, S.; Casanueva, F.F.; et al. Age-related changes in general and sexual health in middle-aged and older men: Results from the European Male Ageing Study (EMAS). J. Sex. Med. 2010, 7, 1362–1380. [Google Scholar] [CrossRef] [PubMed]
- Donovan, J.L.; Hamdy, F.C.; Lane, J.A.; Mason, M.; Metcalfe, C.; Walsh, E.; Blazeby, J.M.; Peters, T.J.; Holding, P.; Bonnington, S.; et al. Patient-reported outcomes after monitoring, surgery, or radiotherapy for prostate cancer. N. Engl. J. Med. 2016, 375, 1425–1437. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.; Metcalfe, C.; Young, G.J.; Peters, T.J.; Blazeby, J.; Avery, K.N.; Dedman, D.; Down, L.; Mason, M.D.; Neal, D.E.; et al. Patient-reported outcomes in the ProtecT randomized trial of clinically localized prostate cancer treatments: Study design, and baseline urinary, bowel and sexual function and quality of life. BJU Int. 2016, 118, 869–879. [Google Scholar] [CrossRef] [PubMed]
- Ryu, J.K.; Suh, J.K.; Burnett, A.L. Research in pharmacotherapy for erectile dysfunction. Transl. Androl. Urol. 2017, 6, 207–215. [Google Scholar] [CrossRef] [PubMed]
- Hawksworth, D.J.; Burnett, A.L. Pharmacotherapeutic management of erectile dysfunction. Clin. Pharmacol. Ther. 2015, 98, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.C.; Lue, T.F. Physiology of penile erection and pathophysiology of erectile dysfunction. Urol. Clin. N. Am. 2005, 32, 379–395. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.; Jo, H.W.; Kwon, H.; Jeong, N.Y. Clinical neuroanatomy and neurotransmitter-mediated regulation of penile erection. Int. Neurourol. J. 2014, 18, 58–62. [Google Scholar] [CrossRef] [PubMed]
- Haffner, M.C.; Landis, P.K.; Saigal, C.S.; Carter, H.B.; Freedland, S.J. Health-related quality-of-life outcomes after anatomic retropubic radical prostatectomy in the phosphodiesterase type 5 ERA: Impact of neurovascular bundle preservation. Urology 2005, 66, 371–376. [Google Scholar] [CrossRef] [PubMed]
- Burnett, A.L. Erectile function outcomes in the current era of anatomic nerve-sparing radical prostatectomy. Rev. Urol. 2006, 8, 47–53. [Google Scholar] [PubMed]
- Hakim, L.; van der Aa, F.; Bivalacqua, T.J.; Hedlund, P.; Albersen, M. Emerging tools for erectile dysfunction: A role for regenerative medicine. Nat. Rev. Urol. 2012, 9, 520–536. [Google Scholar] [CrossRef] [PubMed]
- Gur, S.; Sikka, S.C.; Kadowitz, P.J.; Silberstein, J.; Hellstrom, W.J. 2015 update of erectile dysfunction management following radical prostatectomy: From basic research to clinical management. Curr. Pharm. Des. 2015, 21, 1440–1454. [Google Scholar] [CrossRef] [PubMed]
- Shan, H.; Chen, F.; Zhang, T.; He, S.; Xu, L.; Wei, A. Stem cell therapy for erectile dysfunction of cavernous nerve injury rats: A systematic review and meta-analysis. PLoS ONE 2015, 10, e0121428. [Google Scholar] [CrossRef] [PubMed]
- Albersen, M.; Weyne, E.; Bivalacqua, T.J. Stem cell therapy for erectile dysfunction: Progress and future directions. Sex. Med. Rev. 2013, 1, 50–64. [Google Scholar] [CrossRef] [PubMed]
- Albersen, M.; Shindel, A.W.; Mwamukonda, K.B.; Lue, T.F. The future is today: Emerging drugs for the treatment of erectile dysfunction. Expert Opin. Emerg. Drugs 2010, 15, 467–480. [Google Scholar] [CrossRef] [PubMed]
- Bivalacqua, T.J.; Usta, M.F.; Champion, H.C.; Kadowitz, P.J.; Hellstrom, W.J. Endothelial dysfunction in erectile dysfunction: Role of the endothelium in erectile physiology and disease. J. Androl. 2003, 24, S17–S37. [Google Scholar] [CrossRef] [PubMed]
- Burnett, A.L.; Lowenstein, C.J.; Bredt, D.S.; Chang, T.S.; Snyder, S.H. Nitric oxide: A physiologic mediator of penile erection. Science 1992, 257, 401–403. [Google Scholar] [CrossRef] [PubMed]
- Burnett, A.L. Role of nitric oxide in the physiology of erection. Biol. Reprod. 1995, 52, 485–489. [Google Scholar] [CrossRef] [PubMed]
- Anele, U.A.; Morrison, B.F.; Burnett, A.L. Molecular pathophysiology of priapism: Emerging targets. Curr. Drug Targets 2015, 16, 474–483. [Google Scholar] [CrossRef] [PubMed]
- Corbin, J.D. Mechanisms of action of PDE5 inhibition in erectile dysfunction. Int. J. Impot. Res. 2004, 16, S4–S7. [Google Scholar] [CrossRef] [PubMed]
- Yafi, F.A.; Jenkins, L.; Albersen, M.; Corona, G.; Isidori, A.M.; Goldfarb, S.; Maggi, M.; Nelson, C.J.; Parish, S.; Salonia, A.; et al. Erectile dysfunction. Nat. Rev. Dis. Primers. 2016, 2, 16003. [Google Scholar] [CrossRef] [PubMed]
- Bennett, N.E.; Kim, J.H.; Wolfe, D.P.; Sasaki, K.; Yoshimura, N.; Goins, W.F.; Huang, S.; Nelson, J.B.; de Groat, W.C.; Glorioso, J.C.; et al. Improvement in erectile dysfunction after neurotrophic factor gene therapy in diabetic rats. J. Urol. 2005, 173, 1820–1824. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.C.; Minor, T.X.; Rahman, N.U.; Ho, H.C.; Nunes, L.; Lue, T.F. The additive erectile recovery effect of brain-derived neurotrophic factor combined with vascular endothelial growth factor in a rat model of neurogenic impotence. BJU Int. 2005, 95, 1077–1080. [Google Scholar] [CrossRef] [PubMed]
- Bakircioglu, M.E.; Lin, C.S.; Fan, P.; Sievert, K.D.; Kan, Y.W.; Lue, T.F. The effect of adeno-associated virus mediated brain derived neurotrophic factor in an animal model of neurogenic impotence. J. Urol. 2001, 165, 2103–2109. [Google Scholar] [CrossRef]
- Ng, B.K.; Chen, L.; Mandemakers, W.; Cosgaya, J.M.; Chan, J.R. Anterograde transport and secretion of brain-derived neurotrophic factor along sensory axons promote Schwann cell myelination. J. Neurosci. 2007, 27, 7597–7603. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.S.; Lue, T.F. Growth factor therapy and neuronal nitric oxide synthase. Int. J. Impot. Res. 2004, 16, S38–S39. [Google Scholar] [CrossRef] [PubMed]
- Hsieh, P.S.; Bochinski, D.J.; Lin, G.T.; Nunes, L.; Lin, C.S.; Lue, T.F. The effect of vascular endothelial growth factor and brain-derived neurotrophic factor on cavernosal nerve regeneration in a nerve-crush rat model. BJU Int. 2003, 92, 470–475. [Google Scholar] [CrossRef] [PubMed]
- Bella, A.J.; Lin, G.; Tantiwongse, K.; Garcia, M.; Lin, C.S.; Brant, W.; Lue, T.F. Brain-derived neurotrophic factor (BDNF) acts primarily via the JAK/STAT pathway to promote neurite growth in the major pelvic ganglion of the rat: Part I. J. Sex. Med. 2006, 3, 815–820. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Ning, H.; Reed-Maldonado, A.B.; Zhou, J.; Ruan, Y.; Zhou, T.; Wang, H.S.; Oh, B.S.; Banie, L.; Lin, G.; et al. Low-intensity extracorporeal shock wave therapy enhances brain-derived neurotrophic factor expression through PERK/ATF4 signaling pathway. Int. J. Mol. Sci. 2017, 18. [Google Scholar] [CrossRef] [PubMed]
- Choy, S.W.; Cheng, S.H. Hedgehog signaling. Vitam. Horm. 2012, 88, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Akazawa, C.; Tsuzuki, H.; Nakamura, Y.; Sasaki, Y.; Ohsaki, K.; Nakamura, S.; Arakawa, Y.; Kohsaka, S. The upregulated expression of sonic hedgehog in motor neurons after rat facial nerve axotomy. J. Neurosci. 2004, 24, 7923–7930. [Google Scholar] [CrossRef] [PubMed]
- Angeloni, N.; Bond, C.W.; Harrington, D.; Stupp, S.; Podlasek, C.A. Sonic hedgehog is neuroprotective in the cavernous nerve with crush injury. J. Sex. Med. 2013, 10, 1240–1250. [Google Scholar] [CrossRef] [PubMed]
- Bond, C.; Tang, Y.; Podlasek, C.A. Neural influences on sonic hedgehog and apoptosis in the rat penis. Biol. Reprod. 2008, 78, 947–956. [Google Scholar] [CrossRef] [PubMed]
- Angeloni, N.L.; Bond, C.W.; Tang, Y.; Harrington, D.A.; Zhang, S.; Stupp, S.I.; McKenna, K.E.; Podlasek, C.A. Regeneration of the cavernous nerve by Sonic hedgehog using aligned peptide amphiphile nanofibers. Biomaterials 2011, 32, 1091–1101. [Google Scholar] [CrossRef] [PubMed]
- Bond, C.W.; Angeloni, N.; Harrington, D.; Stupp, S.; Podlasek, C.A. Sonic hedgehog regulates brain-derived neurotrophic factor in normal and regenerating cavernous nerves. J. Sex. Med. 2013, 10, 730–737. [Google Scholar] [CrossRef] [PubMed]
- Angeloni, N.L.; Bond, C.W.; McVary, K.T.; Podlasek, C.A. Sonic hedgehog protein is decreased and penile morphology is altered in prostatectomy and diabetic patients. PLoS ONE 2013, 8, e70985. [Google Scholar] [CrossRef] [PubMed]
- Kusano, K.F.; Allendoerfer, K.L.; Munger, W.; Pola, R.; Bosch-Marce, M.; Kirchmair, R.; Yoon, Y.S.; Curry, C.; Silver, M.; Kearney, M.; et al. Sonic hedgehog induces arteriogenesis in diabetic vasa nervorum and restores function in diabetic neuropathy. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 2102–2107. [Google Scholar] [CrossRef] [PubMed]
- Podlasek, C.A. Sonic hedgehog, apoptosis, and the penis. J. Sex. Med. 2009, 6, 334–339. [Google Scholar] [CrossRef] [PubMed]
- Choe, S.; Veliceasa, D.; Bond, C.W.; Harrington, D.A.; Stupp, S.I.; McVary, K.T.; Podlasek, C.A. Sonic hedgehog delivery from self-assembled nanofiber hydrogels reduces the fibrotic response in models of erectile dysfunction. Acta Biomater. 2016, 32, 89–99. [Google Scholar] [CrossRef] [PubMed]
- Choe, S.; Bond, C.W.; Harrington, D.A.; Stupp, S.I.; McVary, K.T.; Podlasek, C.A. Peptide amphiphile nanofiber hydrogel delivery of sonic hedgehog protein to the cavernous nerve to promote regeneration and prevent erectile dysfunction. Nanomedicine 2017, 13, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Araki, T.; Milbrandt, J. Ninjurin, a novel adhesion molecule, is induced by nerve injury and promotes axonal growth. Neuron 1996, 17, 353–361. [Google Scholar] [CrossRef]
- Ifergan, I.; Kebir, H.; Terouz, S.; Alvarez, J.I.; Lécuyer, M.A.; Gendron, S.; Bourbonnière, L.; Dunay, I.R.; Bouthillier, A.; Moumdjian, R.; et al. Role of Ninjurin-1 in the migration of myeloid cells to central nervous system inflammatory lesions. Ann. Neurol. 2011, 70, 751–763. [Google Scholar] [CrossRef] [PubMed]
- Yin, G.N.; Kim, W.J.; Jin, H.R.; Kwon, M.H.; Song, K.M.; Choi, M.J.; Park, J.M.; Das, N.D.; Kwon, K.D.; Batbold, D.; et al. Nerve injury-induced protein 1 (Ninjurin-1) is a novel therapeutic target for cavernous nerve injury-induced erectile dysfunction in mice. J. Sex. Med. 2013, 10, 1488–1501. [Google Scholar] [CrossRef] [PubMed]
- Yin, G.N.; Choi, M.J.; Kim, W.J.; Kwon, M.H.; Song, K.M.; Park, J.M.; Das, N.D.; Kwon, K.D.; Batbold, D.; Oh, G.T.; et al. Inhibition of Ninjurin 1 restores erectile function through dual angiogenic and neurotrophic effects in the diabetic mouse. Proc. Natl. Acad. Sci. USA 2014, 111, E2731–E2740. [Google Scholar] [CrossRef] [PubMed]
- Falls, D.L. Neuregulins: Functions, forms, and signaling strategies. Exp. Cell. Res. 2003, 284, 14–30. [Google Scholar] [CrossRef]
- Fricker, F.R.; Lago, N.; Balarajah, S.; Tsantoulas, C.; Tanna, S.; Zhu, N.; Fageiry, S.K.; Jenkins, M.; Garratt, A.N.; Birchmeier, C.; et al. Axonally derived Neuregulin-1 is required for remyelination and regeneration after nerve injury in adulthood. J. Neurosci. 2011, 31, 3225–3233. [Google Scholar] [CrossRef] [PubMed]
- Burnett, A.L.; Sezen, S.F.; Hoke, A.; Caggiano, A.O.; Iaci, J.; Lagoda, G.; Musicki, B.; Bella, A.J. GGF2 is neuroprotective in a rat model of cavernous nerve injury-induced erectile dysfunction. J. Sex. Med. 2015, 12, 897–905. [Google Scholar] [CrossRef] [PubMed]
- Marks, A.R. Cellular functions of immunophilins. Physiol. Rev. 1996, 76, 631–649. [Google Scholar] [PubMed]
- Rulten, S.L.; Kinloch, R.A.; Tateossian, H.; Robinson, C.; Gettins, L.; Kay, J.E. The human FK506-binding proteins: Characterization of human FKBP19. Mamm. Genome 2006, 17, 322–331. [Google Scholar] [CrossRef] [PubMed]
- Sezen, S.F.; Lagoda, G.; Burnett, A.L. Role of immunophilins in recovery of erectile function after cavernous nerve injury. J. Sex. Med. 2009, 6, 340–346. [Google Scholar] [CrossRef] [PubMed]
- Steiner, J.P.; Dawson, T.M.; Fotuhi, M.; Glatt, C.E.; Snowman, A.M.; Cohen, N.; Snyder, S.H. High brain densities of the immunophilin FKBP colocalized with calcineurin. Nature 1992, 358, 584–587. [Google Scholar] [CrossRef] [PubMed]
- Gold, B.G.; Storm-Dickerson, T.; Austin, D.R. The immunosuppressant FK506 increases functional recovery and nerve regeneration following peripheral nerve injury. Restor. Neurol. Neurosci. 1994, 6, 287–296. [Google Scholar] [CrossRef] [PubMed]
- Gold, B.G.; Katoh, K.; Storm-Dickerson, T. The immunosuppressant FK506 increases the rate of axonal regeneration in rat sciatic nerve. J. Neurosci. 1995, 15, 7509–7516. [Google Scholar] [PubMed]
- Bavetta, S.; Hamlyn, P.J.; Burnstock, G.; Lieberman, A.R.; Anderson, P.N. The effects of FK506 on dorsal column axons following spinal cord injury in adult rats: Neuroprotection and local regeneration. Exp. Neurol 1999, 158, 382–393. [Google Scholar] [CrossRef] [PubMed]
- Sezen, S.F.; Hoke, A.; Burnett, A.L.; Snyder, S.H. Immunophilin ligand FK506 is neuroprotective for penile innervation. Nat. Med. 2001, 7, 1073–1074. [Google Scholar] [CrossRef] [PubMed]
- Burnett, A.L.; Becker, R.E. Immunophilin ligands promote penile neurogenesis and erection recovery after cavernous nerve injury. J. Urol. 2004, 171, 495–500. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Burnett, A.L. RhoA/Rho-kinase in erectile tissue: Mechanisms of disease and therapeutic insights. Clin. Sci. 2006, 110, 153–165. [Google Scholar] [CrossRef] [PubMed]
- Sopko, N.A.; Hannan, J.L.; Bivalacqua, T.J. Understanding and targeting the Rho kinase pathway in erectile dysfunction. Nat. Rev. Urol. 2014, 11, 622–628. [Google Scholar] [CrossRef] [PubMed]
- Hannan, J.L.; Albersen, M.; Kutlu, O.; Gratzke, C.; Stief, C.G.; Burnett, A.L.; Lysiak, J.J.; Hedlund, P.; Bivalacqua, T.J. Inhibition of Rho-kinase improves erectile function, increases nitric oxide signaling and decreases penile apoptosis in a rat model of cavernous nerve injury. J. Urol. 2013, 189, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.; Ito, M.; Amano, M.; Chihara, K.; Fukata, Y.; Nakafuku, M.; Yamamori, B.; Feng, J.; Nakano, T.; Okawa, K.; et al. Regulation of myosin phosphatase by Rho and Rho-associated kinase (Rho-kinase). Science 1996, 273, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Chitaley, K.; Webb, R.C. Nitric oxide induces dilation of rat aorta via inhibition of rho-kinase signaling. Hypertension 2002, 39, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Sauzeau, V.; Rolli-Derkinderen, M.; Marionneau, C.; Loirand, G.; Pacaud, P. RhoA expression is controlled by nitric oxide through cGMP-dependent protein kinase activation. J. Biol. Chem. 2003, 278, 9472–9480. [Google Scholar] [CrossRef] [PubMed]
- Chitaley, K.; Wingard, C.J.; Clinton Webb, R.; Branam, H.; Stopper, V.S.; Lewis, R.W.; Mills, T.M. Antagonism of Rho-kinase stimulates rat penile erection via a nitric oxide-independent pathway. Nat. Med. 2001, 7, 119–122. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Webber, C.A.; Wang, J.; Xu, Y.; Martinez, J.A.; Liu, W.Q.; McDonald, D.; Guo, G.F.; Nguyen, M.D.; Zochodne, D.W. Activated RHOA and peripheral axon regeneration. Exp. Neurol. 2008, 212, 358–369. [Google Scholar] [CrossRef] [PubMed]
- Gratzke, C.; Strong, T.D.; Gebska, M.A.; Champion, H.C.; Stief, C.G.; Burnett, A.L.; Bivalacqua, T.J. Activated RhoA/Rho kinase impairs erectile function after cavernous nerve injury in rats. J. Urol. 2010, 184, 2197–2204. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Wang, J.; Liu, X.; Cheng, Y.; Deng, L.; Zhong, Y. Y-39983 downregulates RhoA/Rho-associated kinase expression during its promotion of axonal regeneration. Oncol. Rep. 2013, 29, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Hiraga, A.; Kuwabara, S.; Doya, H.; Kanai, K.; Fujitani, M.; Taniguchi, J.; Arai, K.; Mori, M.; Hattori, T.; Yamashita, T. Rho-kinase inhibition enhances axonal regeneration after peripheral nerve injury. J. Peripher. Nerv. Syst. 2006, 11, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Cho, M.C.; Park, K.; Kim, S.W.; Paick, J.S. Restoration of erectile function by suppression of corporal apoptosis, fibrosis and corporal veno-occlusive dysfunction with rho-kinase inhibitors in a rat model of cavernous nerve injury. J. Urol. 2015, 193, 1716–1723. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, P.M.; Cole, K.E.; Dowling, D.P.; Christianson, D.W. Structure, mechanism, and inhibition of histone deacetylases and related metalloenzymes. Curr. Opin. Struct. Biol. 2011, 21, 735–743. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.U.; Krishnamurthy, S. Histone modifications as key regulators of transcription. Front. Biosci. 2005, 10, 866–872. [Google Scholar] [CrossRef] [PubMed]
- Barter, M.J.; Pybus, L.; Litherland, G.J.; Rowan, A.D.; Clark, I.M.; Edwards, D.R.; Cawston, T.E.; Young, D.A. HDAC-mediated control of ERK- and PI3K-dependent TGF-β-induced extracellular matrix-regulating genes. Matrix Biol. 2010, 29, 602–612. [Google Scholar] [CrossRef] [PubMed]
- Hannan, J.L.; Kutlu, O.; Stopak, B.L.; Liu, X.; Castiglione, F.; Hedlund, P.; Burnett, A.L.; Bivalacqua, T.J. Valproic acid prevents penile fibrosis and erectile dysfunction in cavernous nerve-injured rats. J. Sex. Med. 2014, 11, 1442–1451. [Google Scholar] [CrossRef] [PubMed]
- Erbayraktar, S.; Grasso, G.; Sfacteria, A.; Xie, Q.W.; Coleman, T.; Kreilgaard, M.; Torup, L.; Sager, T.; Erbayraktar, Z.; Gokmen, N.; et al. Asialoerythropoietin is a nonerythropoietic cytokine with broad neuroprotective activity in vivo. Proc. Natl. Acad. Sci. USA 2003, 100, 6741–6746. [Google Scholar] [CrossRef] [PubMed]
- Campana, W.M.; Myers, R.R. Erythropoietin and erythropoietin receptors in the peripheral nervous system: Changes after nerve injury. FASEB J. 2001, 15, 1804–1806. [Google Scholar] [CrossRef] [PubMed]
- Ehrenreich, H.; Weissenborn, K.; Prange, H.; Schneider, D.; Weimar, C.; Wartenberg, K.; Schellinger, P.D.; Bohn, M.; Becker, H.; Wegrzyn, M.; et al. Recombinant human erythropoietin in the treatment of acute ischemic stroke. Stroke 2009, 40, e647–e656. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.; Minnerup, J. Promoting recovery from ischemic stroke. Expert Rev. Neurother. 2016, 16, 173–186. [Google Scholar] [CrossRef] [PubMed]
- Bartesaghi, S.; Marinovich, M.; Corsini, E.; Galli, C.L.; Viviani, B. Erythropoietin: A novel neuroprotective cytokine. Neurotoxicology 2005, 26, 923–928. [Google Scholar] [CrossRef] [PubMed]
- Sobh, M.A.; Abd el Hamid, I.A.; Atta, M.G.; Refaie, A.F. Effect of erythropoietin on sexual potency in chronic haemodialysis patients. A preliminary study. Scand. J. Urol. Nephrol. 1992, 26, 181–185. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Allaf, M.E.; Lagoda, G.; Burnett, A.L. Erythropoietin receptor expression in the human urogenital tract: Immunolocalization in the prostate, neurovascular bundle and penis. BJU Int. 2007, 100, 1103–1106. [Google Scholar] [CrossRef] [PubMed]
- Allaf, M.E.; Hoke, A.; Burnett, A.L. Erythropoietin promotes the recovery of erectile function following cavernous nerve injury. J. Urol. 2005, 174, 2060–2064. [Google Scholar] [CrossRef] [PubMed]
- Valentine, H.; Chen, Y.; Guo, H.; McCormick, J.; Wu, Y.; Sezen, S.F.; Hoke, A.; Burnett, A.L.; Steiner, J.P. Neuroimmunophilin ligands protect cavernous nerves after crush injury in the rat: New experimental paradigms. Eur. Urol. 2007, 51, 1724–1731. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, N.; Minor, T.X.; Carrion, R.; Price, R.; Nunes, L.; Lue, T.F. The effect of FK1706 on erectile function following bilateral cavernous nerve crush injury in a rat model. J. Urol. 2006, 176, 824–829. [Google Scholar] [CrossRef] [PubMed]
- Evaluate the Effects of GPI 1485 on Erectile Function Following Bilateral Nerve-Sparing Prostatectomy. ClinicalTrials.gov. Available online: http://clinicaltrials.gov/ct/show/NCT00090376 (accessed on 1 July 2017).
- Mulhall, J.K.E.; Slawin, K.; Scardino, P.T. Tacrolimus (FK506) as a neuromodulator in the radical prostatectomy population; a multicenter, double-blind, randomized, placebo-controlled trial. J. Urol. 2010, 183, e530–e531. [Google Scholar] [CrossRef]
- Uvin, P.; Albersen, M.; Bollen, I.; Falter, M.; Weyne, E.; Linsen, L.; Tinel, H.; Sandner, P.; Bivalacqua, T.J.; de Ridder, D.J.; et al. Additive effects of the Rho kinase inhibitor Y-27632 and vardenafil on relaxation of the corpus cavernosum tissue of patients with erectile dysfunction and clinical phosphodiesterase type 5 inhibitor failure. BJU Int. 2017, 119, 325–332. [Google Scholar] [CrossRef] [PubMed]
- Löhn, M.; Plettenburg, O.; Ivashchenko, Y.; Kannt, A.; Hofmeister, A.; Kadereit, D.; Schaefer, M.; Linz, W.; Kohlmann, M.; Herbert, J.M.; et al. Pharmacological characterization of SAR407899, a novel rho-kinase inhibitor. Hypertension 2009, 54, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Lasker, G.F.; Pankey, E.A.; Allain, A.V.; Murthy, S.N.; Stasch, J.P.; Kadowitz, P.J. The selective Rho-kinase inhibitor azaindole-1 has long-lasting erectile activity in the rat. Urology 2013, 81. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.A.; Lee, D.Y.; Cho, H.M.; Kim, S.Y.; Iwasaki, Y.; Kim, I.K. Histone deacetylase inhibition attenuates transcriptional activity of mineralocorticoid receptor through its acetylation and prevents development of hypertension. Circ. Res. 2013, 112, 1004–1012. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.Z.; Gong, Y.Y.; Shi, Y.H.; Zhang, W.; Qin, X.H.; Wu, X.W. Valproate promotes survival of retinal ganglion cells in a rat model of optic nerve crush. Neuroscience 2012, 224, 282–293. [Google Scholar] [CrossRef] [PubMed]
- Burnett, A.L.; Allaf, M.E.; Bivalacqua, T.J. Erythropoietin promotes erection recovery after nerve-sparing radical retropubic prostatectomy: A retrospective analysis. J. Sex. Med. 2008, 5, 2392–2398. [Google Scholar] [CrossRef] [PubMed]
- Bivalacqua, T.J.; Strong, T.D. The use of gene transfer technology to study the pathophysiology of erectile dysfunction. J. Sex. Med. 2008, 5, 268–275. [Google Scholar] [CrossRef] [PubMed]
- Harraz, A.; Shindel, A.W.; Lue, T.F. Emerging gene and stem cell therapies for the treatment of erectile dysfunction. Nat. Rev. Urol. 2010, 7, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Melman, A. Gene therapy for male erectile dysfunction. Urol. Clin. N. Am. 2007, 34, 619–630. [Google Scholar] [CrossRef] [PubMed]
- Yoshimura, N.; Kato, R.; Chancellor, M.B.; Nelson, J.B.; Glorioso, J.C. Gene therapy as future treatment of erectile dysfunction. Expert Opin. Biol. Ther. 2010, 10, 1305–1314. [Google Scholar] [CrossRef] [PubMed]
- Kato, R.; Wolfe, D.; Coyle, C.H.; Huang, S.; Wechuck, J.B.; Goins, W.F.; Krisky, D.M.; Tsukamoto, T.; Nelson, J.B.; Glorioso, J.C.; et al. Herpes simplex virus vector-mediated delivery of glial cell line-derived neurotrophic factor rescues erectile dysfunction following cavernous nerve injury. Gene Ther. 2007, 14, 1344–1352. [Google Scholar] [CrossRef] [PubMed]
- Kato, R.; Wolfe, D.; Coyle, C.H.; Wechuck, J.B.; Tyagi, P.; Tsukamoto, T.; Nelson, J.B.; Glorioso, J.C.; Chancellor, M.B.; Yoshimura, N. Herpes simplex virus vector-mediated delivery of neurturin rescues erectile dysfunction of cavernous nerve injury. Gene Ther. 2009, 16, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Melman, A.; Bar-Chama, N.; McCullough, A.; Davies, K.; Christ, G. The first human trial for gene transfer therapy for the treatment of erectile dysfunction: Preliminary results. Eur. Urol. 2005, 48, 314–318. [Google Scholar] [CrossRef] [PubMed]
- Melman, A.; Bar-Chama, N.; McCullough, A.; Davies, K.; Christ, G. hMaxi-K gene transfer in males with erectile dysfunction: Results of the first human trial. Hum. Gene Ther. 2006, 17, 1165–1176. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.S.; Xin, Z.C.; Wang, Z.; Deng, C.; Huang, Y.C.; Lin, G.; Lue, T.F. Stem cell therapy for erectile dysfunction: A critical review. Stem Cells Dev. 2012, 21, 343–351. [Google Scholar] [CrossRef] [PubMed]
- Gokce, A.; Peak, T.C.; Abdel-Mageed, A.B.; Hellstrom, W.J. Adipose Tissue-Derived Stem Cells for the Treatment of Erectile Dysfunction. Curr. Urol. Rep. 2016, 17, 14. [Google Scholar] [CrossRef] [PubMed]
- Alwaal, A.; Zaid, U.B.; Lin, C.S.; Lue, T.F. Stem cell treatment of erectile dysfunction. Adv. Drug Deliv. Rev. 2015, 82–83, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Bochinski, D.; Lin, G.T.; Nunes, L.; Carrion, R.; Rahman, N.; Lin, C.S.; Lue, T.F. The effect of neural embryonic stem cell therapy in a rat model of cavernosal nerve injury. BJU Int. 2004, 94, 904–909. [Google Scholar] [CrossRef] [PubMed]
- Mangır, N.; Türkeri, L. Stem cell therapies in post-prostatectomy erectile dysfunction: A critical review. Can. J. Urol. 2017, 24, 8609–8619. [Google Scholar] [PubMed]
- Peak, T.C.; Anaissie, J.; Hellstrom, W.J. Current perspectives on stem cell therapy for erectile dysfunction. Sex. Med. Rev. 2016, 4, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Reed-Maldonado, A.B.; Lue, T.F. The current status of stem-cell therapy in erectile dysfunction: A review. World J. Mens. Health 2016, 34, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Haahr, M.K.; Jensen, C.H.; Toyserkani, N.M.; Andersen, D.C.; Damkier, P.; Sørensen, J.A.; Lund, L.; Sheikh, S.P. Safety and potential effect of a single intracavernous injection of autologous adipose-derived regenerative cells in patients with erectile dysfunction following radical prostatectomy: An open-label phase I clinical trial. E BioMed 2016, 5, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Yiou, R.; Hamidou, L.; Birebent, B.; Bitari, D.; Lecorvoisier, P.; Contremoulins, I.; Khodari, M.; Rodriguez, A.M.; Augustin, D.; Roudot-Thoraval, F.; et al. Safety of intracavernous bone marrow-mononuclear cells for postradical prostatectomy erectile dysfunction: An open dose-escalation pilot study. Eur. Urol. 2016, 69, 988–991. [Google Scholar] [CrossRef] [PubMed]
- Mulhall, J.P. Cavernous nerve stimulation and interposition grafting: A critical assessment and future perspectives. Rev. Urol. 2005, 7, S18–S25. [Google Scholar] [PubMed]
- Siddiqui, K.M.; Billia, M.; Mazzola, C.R.; Alzahrani, A.; Brock, G.B.; Scilley, C.; Chin, J.L. Three-year outcomes of recovery of erectile function after open radical prostatectomy with sural nerve grafting. J. Sex. Med. 2014, 11, 2119–2124. [Google Scholar] [CrossRef] [PubMed]
- Zorn, K.C.; Bernstein, A.J.; Gofrit, O.N.; Shikanov, S.A.; Mikhail, A.A.; Song, D.H.; Zagaja, G.P.; Shalhav, A.L. Long-term functional and oncological outcomes of patients undergoing sural nerve interposition grafting during robot-assisted laparoscopic radical prostatectomy. J. Endourol. 2008, 22, 1005–1012. [Google Scholar] [CrossRef] [PubMed]
- Joffe, R.; Klotz, L.H. Results of unilateral genitofemoral nerve grafts with contralateral nerve sparing during radical prostatectomy. Urology 2007, 69, 1161–1164. [Google Scholar] [CrossRef] [PubMed]
- Slawin, K.M.; Canto, E.I.; Shariat, S.F.; Gore, J.L.; Kim, E.; Kattan, M.W.; Wheeler, T.M.; Nath, R.K. Sural nerve interposition grafting during radical prostatectomy. Rev. Urol. 2002, 4, 17–23. [Google Scholar] [PubMed]
- White, W.M.; Kim, E.D. Interposition nerve grafting during radical prostatectomy: Cumulative review and critical appraisal of literature. Urology 2009, 74, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.R.; Samavedi, S.; Bates, A.S.; Kumar, A.; Coelho, R.; Rocco, B.; Palmer, K. Dehydrated human amnion/chorion membrane allograft nerve wrap around the prostatic neurovascular bundle accelerates early return to continence and potency following robot-assisted radical prostatectomy: Propensity score-matched analysis. Eur. Urol. 2015, 67, 977–980. [Google Scholar] [CrossRef] [PubMed]
- Raina, R.; Pahlajani, G.; Agarwal, A.; Zippe, C.D. Early penile rehabilitation following radical prostatectomy: Cleveland clinic experience. Int. J. Impot. Res. 2008, 20, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Ogaya-Pinies, G.; Palayapalam-Ganapathi, H.; Rogers, T.; Hernandez-Cardona, E.; Rocco, B.; Coelho, R.F.; Jenson, C.; Patel, V.R. Can dehydrated human amnion/chorion membrane accelerate the return to potency after a nerve-sparing robotic-assisted radical prostatectomy? Propensity score-matched analysis. J. Robot. Surg. 2017. [Google Scholar] [CrossRef] [PubMed]
- Finley, D.S.; Osann, K.; Skarecky, D.; Ahlering, T.E. Hypothermic nerve-sparing radical prostatectomy: Rationale, feasibility, and effect on early continence. Urology 2009, 73, 691–696. [Google Scholar] [CrossRef] [PubMed]
- Finley, D.S.; Osann, K.; Chang, A.; Santos, R.; Skarecky, D.; Ahlering, T.E. Hypothermic robotic radical prostatectomy: Impact on continence. J. Endourol. 2009, 23, 1443–1450. [Google Scholar] [CrossRef] [PubMed]
- Finley, D.S.; Chang, A.; Morales, B.; Osann, K.; Skarecky, D.; Ahlering, T. Impact of regional hypothermia on urinary continence and potency after robot-assisted radical prostatectomy. J. Endourol. 2010, 24, 1111–1116. [Google Scholar] [CrossRef] [PubMed]
- Liss, M.A.; Skarecky, D.; Morales, B.; Ahlering, T.E. The application of regional hypothermia using transrectal cooling during radical prostatectomy: Mitigation of surgical inflammatory damage to preserve continence. J. Endourol. 2012, 26, 1553–1557. [Google Scholar] [CrossRef] [PubMed]
- Fried, N.M.; Burnett, A.L. Novel methods for mapping the cavernous nerves during radical prostatectomy. Nat. Rev. Urol. 2015, 12, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Pan, H.; Kim, I.S.; Hwang, S.J.; Kim, S.J. Functional regeneration of severed peripheral nerve using an implantable electrical stimulator. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2010, 2010, 1511–1514. [Google Scholar] [CrossRef] [PubMed]
- Gamble, P.; Stephen, M.; MacEwan, M.; Ray, W.Z. Serial assessment of functional recovery following nerve injury using implantable thin-film wireless nerve stimulators. Muscle Nerve 2016, 54, 1114–1119. [Google Scholar] [CrossRef] [PubMed]
- Burnett, A.L.; Teloken, P.E.; Briganti, A.; Whitehurst, T.; Montorsi, F. Intraoperative assessment of an implantable electrode array for cavernous nerve stimulation. J. Sex. Med. 2008, 5, 1949–1954. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Zhang, R.; Yang, Z.; Lee, J.; Liu, Y.; Tian, J.; Qin, X.; Ren, Z.; Ding, H.; Chen, Q.; et al. Comparative effectiveness and safety of oral phosphodiesterase type 5 inhibitors for erectile dysfunction: A systematic review and network meta-analysis. Eur. Urol. 2013, 63, 902–912. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Campbell, J.D.; Burnett, A.L. Neuroprotective and Nerve Regenerative Approaches for Treatment of Erectile Dysfunction after Cavernous Nerve Injury. Int. J. Mol. Sci. 2017, 18, 1794. https://doi.org/10.3390/ijms18081794
Campbell JD, Burnett AL. Neuroprotective and Nerve Regenerative Approaches for Treatment of Erectile Dysfunction after Cavernous Nerve Injury. International Journal of Molecular Sciences. 2017; 18(8):1794. https://doi.org/10.3390/ijms18081794
Chicago/Turabian StyleCampbell, Jeffrey D., and Arthur L. Burnett. 2017. "Neuroprotective and Nerve Regenerative Approaches for Treatment of Erectile Dysfunction after Cavernous Nerve Injury" International Journal of Molecular Sciences 18, no. 8: 1794. https://doi.org/10.3390/ijms18081794
APA StyleCampbell, J. D., & Burnett, A. L. (2017). Neuroprotective and Nerve Regenerative Approaches for Treatment of Erectile Dysfunction after Cavernous Nerve Injury. International Journal of Molecular Sciences, 18(8), 1794. https://doi.org/10.3390/ijms18081794



