dIvergEnt: How IgE Axis Contributes to the Continuum of Allergic Asthma and Anti-IgE Therapies
Abstract
:1. Introduction
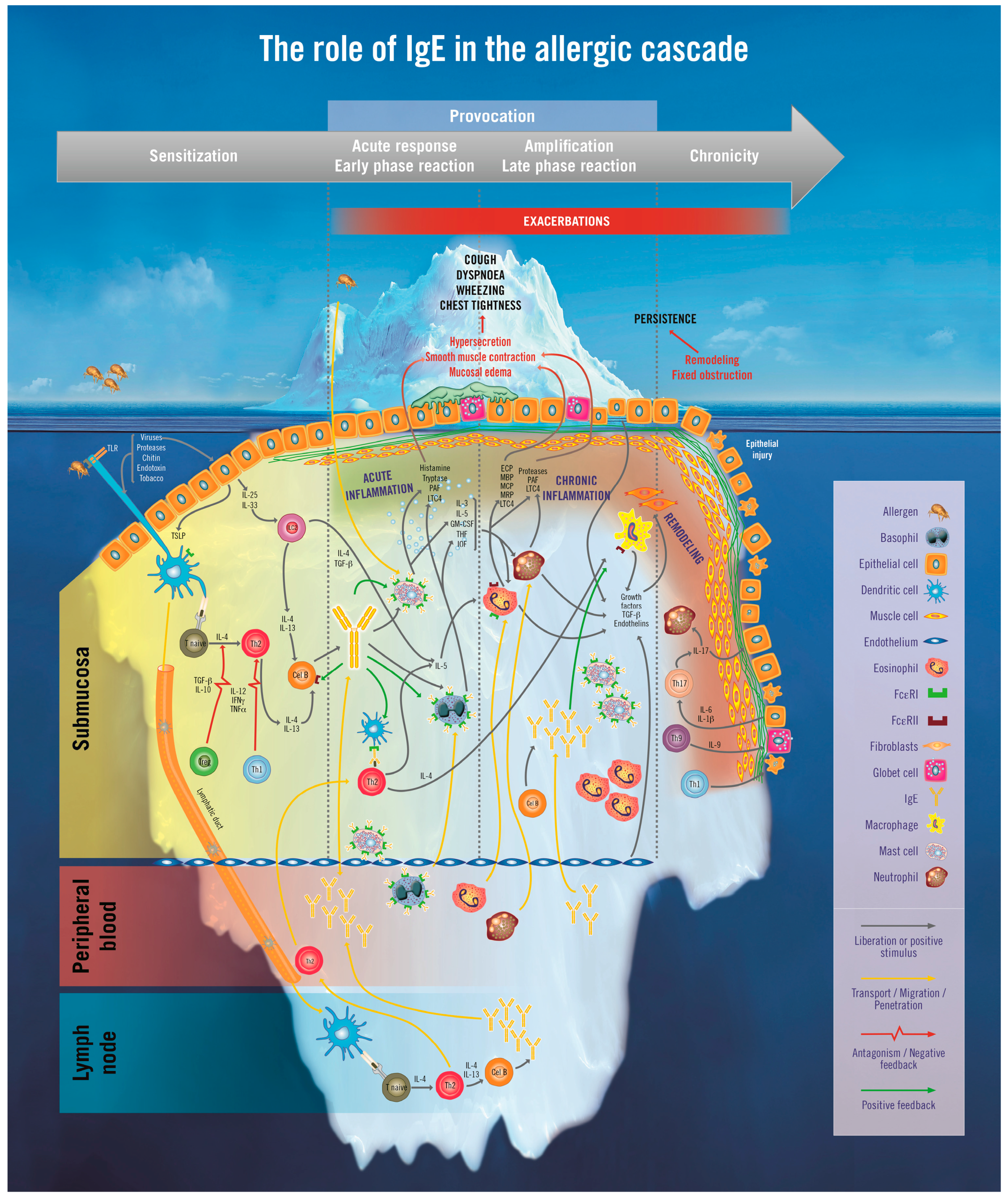
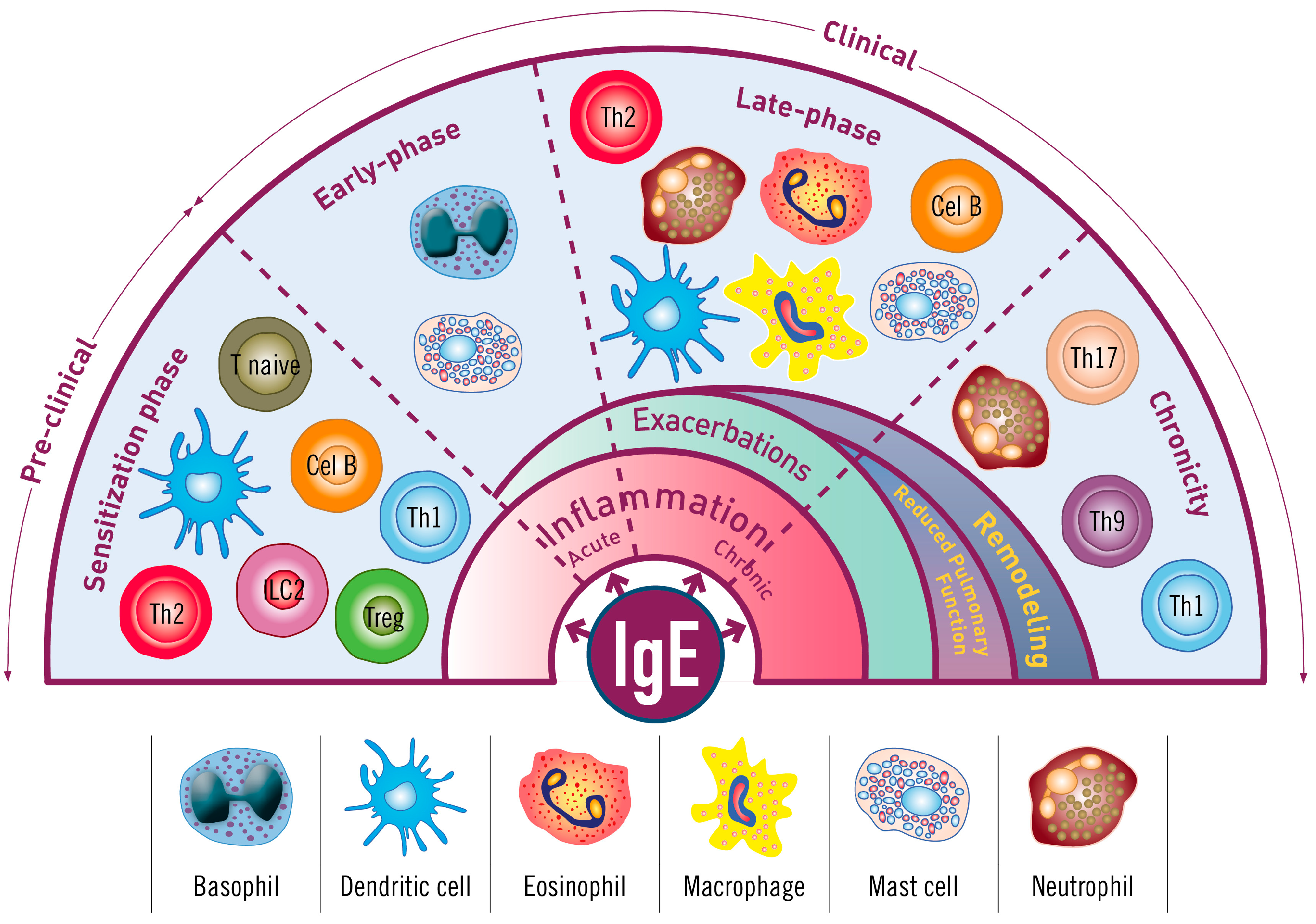
2. The Iceberg Model of Allergic Asthma: Immunological Pathways beyond Visible Clinical Symptoms
3. The Role of Immunoglobulin E (IgE) in Allergic Asthma
3.1. The Sensitization Phase
3.2. The Re-Exposure Phase
3.2.1. The Acute Reaction
3.2.2. The Late-Phase Response
3.2.3. The Chronic Phase
4. Immunopathophysiology of IgE in the Asthma Continuum
5. Should IgE Blocking Be a Key Therapeutic Target for Allergic Asthma?
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| APC | Allergen-presenting cells |
| BHR | Bronchial hyperresponsiveness |
| FcεRI | High affinity IgE receptor |
| FcεRII | Low affinity IgE receptor |
| FEV1 | Forced expiratory volume in 1 second |
| FVC | Forced vital capacity |
| IgE | Immunoglobulin E |
| IL | Interleukin |
| IS | Immune system |
| MC | Mast cells |
| SMC | Smooth muscle cells |
| Th2 | T helper type 2 lymphocytes |
References
- Martinez, F.D.; Vercelli, D. Asthma. Lancet 2013, 382, 1360–1372. [Google Scholar] [CrossRef]
- Bel, E.H. Clinical Practice. Mild asthma. N. Engl. J. Med. 2013, 369, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Xie, M.; Wenzel, S.E. A global perspective in asthma: From phenotype to endotype. Chin. Med. J. 2013, 126, 166–174. [Google Scholar] [PubMed]
- Wenzel, S.E. Asthma phenotypes: The evolution from clinical to molecular approaches. Nat. Med. 2012, 18, 716–725. [Google Scholar] [CrossRef] [PubMed]
- Comité Ejecutivo de la GEMA. Guía Española Para el Manejo del Asma. LUZÁN 5; GEMA: Berlin, Germany, 2015. [Google Scholar]
- Cisneros Serrano, C.; Melero Moreno, C.; Almonacid Sánchez, C.; Perpiñá Tordera, M.; Picado Valles, C.; Martínez Moragón, E.; de Llano, L.P.; Soto Campos, J.G.; Urrutia Landa, I.; García Hernández, G. Guidelines for severe uncontrolled asthma. Arch. Bronconeumol. 2015, 51, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, N.G.; Arakawa, H.; Carlsen, K.-H.; Custovic, A.; Gern, J.; Lemanske, R.; le Souef, P.; Makela, M.; Roberts, G.; Wong, G.; et al. International consensus on (ICON) pediatric asthma. Allergy 2012, 67, 976–997. [Google Scholar] [CrossRef] [PubMed]
- Lötvall, J.; Akdis, C.A.; Bacharier, L.B.; Bjermer, L.; Casale, T.B.; Custovic, A.; Lemanske, R.F., Jr.; Wardlaw, A.J.; Wenzel, S.E.; Greenberger, P.A. Asthma endotypes: A new approach to classification of disease entities within the asthma syndrome. J. Allergy Clin. Immunol. 2011, 127, 355–360. [Google Scholar] [CrossRef] [PubMed]
- Asher, M.I.; Montefort, S.; Björkstén, B.; Lai, C.K.W.; Strachan, D.P.; Weiland, S.K.; ISAAC Phase Three Study Group. Worldwide time trends in the prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and eczema in childhood: ISAAC phases one and three repeat multicountry cross-sectional surveys. Lancet 2006, 368, 733–743. [Google Scholar] [CrossRef]
- Björkstén, B.; Clayton, T.; Ellwood, P.; Stewart, A.; Strachan, D.; ISAAC Phase III Study Group. Worldwide time trends for symptoms of rhinitis and conjunctivitis: Phase III of the International Study of Asthma and Allergies in Childhood. Pediatr. Allergy Immunol. 2008, 19, 110–124. [Google Scholar] [CrossRef] [PubMed]
- Yuksel, H.; Dinc, G.; Sakar, A.; Yilmaz, O.; Yorgancioglu, A.; Celik, P.; Ozcan, C. Prevalence and comorbidity of allergic eczema, rhinitis, and asthma in a city in western Turkey. J. Investig. Allergol. Clin. Immunol. 2008, 18, 31–35. [Google Scholar] [PubMed]
- Almqvist, C.; Lundholm, C. Population-based data on asthma and allergic disease call for advanced epidemiologic methods. J. Allergy Clin. Immunol. 2015, 136, 656–657. [Google Scholar] [CrossRef] [PubMed]
- Isolauri, E.; Huurre, A.; Salminen, S.; Impivaara, O. The allergy epidemic extends beyond the past few decades. Clin. Exp. Allergy 2004, 34, 1007–1010. [Google Scholar] [CrossRef] [PubMed]
- Chang, C. Asthma in children and adolescents: A comprehensive approach to diagnosis and management. Clin. Rev. Allergy Immunol. 2012, 43, 98–137. [Google Scholar] [CrossRef] [PubMed]
- Mallol, J.; Crane, J.; von Mutius, E.; Odhiambo, J.; Keil, U.; Stewart, A.; ISAAC Phase Three Study Group. The International Study of Asthma and Allergies in Childhood (ISAAC) Phase Three: A global synthesis. Allergol. Immunopathol. 2013, 41, 73–85. [Google Scholar] [CrossRef] [PubMed]
- Pawankar, R.; Canonica, G.; Holgate, S.; Lockey, R. World Health Organization (WAO) White Book on Allergy; WAO: Milwaukee, WI, USA, 2011. [Google Scholar]
- Covar, R.A.; Strunk, R.; Zeiger, R.S.; Wilson, L.A.; Liu, A.H.; Weiss, S.; Tonascia, J.; Spahn, J.D.; Szefler, S.J.; Childhood Asthma Management Program Research Group. Predictors of remitting, periodic, and persistent childhood asthma. J. Allergy Clin. Immunol. 2010, 125, 359–366.e3. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.S.; Woodcock, A.; Langley, S.J.; Morris, J.; Custovic, A.; IFWIN Study Team. Secondary prevention of asthma by the use of Inhaled Fluticasone Propionate in Wheezy INfants (IFWIN): Double-blind, randomised, controlled study. Lancet 2006, 368, 754–762. [Google Scholar] [CrossRef]
- Guilbert, T.W.; Morgan, W.J.; Krawiec, M.; Lemanske, R.F.; Sorkness, C.; Szefler, S.J.; Larsen, G.; Spahn, J.D.; Zeiger, R.S.; Heldt, G.; et al. The Prevention of Early Asthma in Kids study: Design, rationale and methods for the Childhood Asthma Research and Education network. Control. Clin. Trials 2004, 25, 286–310. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.T.; Horner, A.; Shapiro, G.; Sternberg, A.L. Childhood Asthma Management Program (CAMP) Research Group. The prevalence of environmental exposure to perceived asthma triggers in children with mild-to-moderate asthma: Data from the Childhood Asthma Management Program (CAMP). J. Allergy Clin. Immunol. 2001, 107, 634–640. [Google Scholar] [CrossRef] [PubMed]
- Accordini, S.; Corsico, A.G.; Braggion, M.; Gerbase, M.W.; Gislason, D.; Gulsvik, A.; Heinrich, J.; Janson, C.; Jarvis, D.; Jõgi, R.; et al. The cost of persistent asthma in Europe: An international population-based study in adults. Int. Arch. Allergy Immunol. 2013, 160, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Calderon, M.A.; Demoly, P.; van Wijk, R.G.; Bousquet, J.; Sheikh, A.; Frew, A.; Scadding, G.; Bachert, C.; Malling, H.J.; Valenta, R.; et al. EAACI: A European Declaration on Immunotherapy. Designing the future of allergen specific immunotherapy. Clin. Transl. Allergy 2012, 2, 20. [Google Scholar] [CrossRef] [PubMed]
- Heineke, M.H.; van Egmond, M. Immunoglobulin A: Magic bullet or Trojan horse? Eur. J. Clin. Investig. 2017, 47, 184–192. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Cerutti, A. The function and regulation of immunoglobulin D. Curr. Opin. Immunol. 2011, 23, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Rigante, D. The truth on IgD in the ploy of immune surveillance and inflammation. Immunol. Res. 2016, 64, 632–635. [Google Scholar] [CrossRef] [PubMed]
- Kelly, B.T.; Grayson, M.H. Immunoglobulin E, what is it good for? Ann. Allergy Asthma Immunol. 2016, 116, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Aschermann, S.; Lux, A.; Baerenwaldt, A.; Biburger, M.; Nimmerjahn, F. The other side of immunoglobulin G: Suppressor of inflammation. Clin. Exp. Immunol. 2010, 160, 161–167. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.J.; Mauri, C.; Ehrenstein, M.R. Natural serum IgM maintains immunological homeostasis and prevents autoimmunity. Springer Semin. Immunopathol. 2005, 26, 425–432. [Google Scholar] [CrossRef] [PubMed]
- Matondang, C.S. Spectrum of asthma in children visiting the outpatient clinic of the subdivision of allergy and immunology. Paediatr. Indones. 1991, 31, 150–164. [Google Scholar] [PubMed]
- McNichol, K.N.; Williams, H.E. Spectrum of asthma in children. II. Allergic components. Br. Med. J. 1973, 4, 12–16. [Google Scholar] [CrossRef] [PubMed]
- Kotaniemi-Syrjänen, A.; Reijonen, T.M.; Romppanen, J.; Korhonen, K.; Savolainen, K.; Korppi, M. Allergen-specific immunoglobulin E antibodies in wheezing infants: The risk for asthma in later childhood. Pediatrics 2003, 111, e255–e261. [Google Scholar] [CrossRef] [PubMed]
- Resch, Y.; Michel, S.; Kabesch, M.; Lupinek, C.; Valenta, R.; Vrtala, S. Different IgE recognition of mite allergen components in asthmatic and nonasthmatic children. J. Allergy Clin. Immunol. 2015, 136, 1083–1091. [Google Scholar] [CrossRef] [PubMed]
- Buendía, E.; Zakzuk, J.; Mercado, D.; Alvarez, A.; Caraballo, L. The IgE response to Ascaris molecular components is associated with clinical indicators of asthma severity. World Allergy Organ. J. 2015, 8, 8. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, M.; Choudhry, S.; Tsai, H.-J.; Thyne, S.; Navarro, D.; Nazario, S.; Rodriguez-Santana, J.R.; Casal, J.; Torres, A.; Chapela, R.; et al. Association between IgE levels and asthma severity among African American, Mexican, and Puerto Rican patients with asthma. J. Allergy Clin. Immunol. 2007, 120, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Carroll, W.D.; Lenney, W.; Child, F.; Strange, R.C.; Jones, P.W.; Whyte, M.K.; Primhak, R.A.; Fryer, A.A. Asthma severity and atopy: How clear is the relationship? Arch. Dis. Child. 2006, 91, 405–409. [Google Scholar] [CrossRef] [PubMed]
- Warm, K.; Hedman, L.; Lindberg, A.; Lötvall, J.; Lundbäck, B.; Rönmark, E. Allergic sensitization is age-dependently associated with rhinitis, but less so with asthma. J. Allergy Clin. Immunol. 2015, 136, 1559–1565.e2. [Google Scholar] [CrossRef] [PubMed]
- Siroux, V.; Oryszczyn, M.-P.; Paty, E.; Kauffmann, F.; Pison, C.; Vervloet, D.; Pin, I. Relationships of allergic sensitization, total immunoglobulin E and blood eosinophils to asthma severity in children of the EGEA Study. Clin. Exp. Allergy 2003, 33, 746–751. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, A.; Jinno, M.; Hirai, K.; Miyata, Y.; Mizuma, H.; Yamaguchi, M.; Ohta, S.; Watanabe, Y.; Yamamoto, M.; Suzuki, S.; et al. Longitudinal increase in total IgE levels in patients with adult asthma: An association with poor asthma control. Respir. Res. 2014, 15, 144. [Google Scholar] [CrossRef] [PubMed]
- Maneechotesuwan, K.; Sujaritwongsanon, P.; Suthamsmai, T. IgE production in allergic asthmatic patients with different asthma control status. J. Med. Assoc. Thail. 2010, 93, S71–S78. [Google Scholar]
- Lowe, P.J.; Tannenbaum, S.; Gautier, A.; Jimenez, P. Relationship between omalizumab pharmacokinetics, IgE pharmacodynamics and symptoms in patients with severe persistent allergic (IgE-mediated) asthma. Br. J. Clin. Pharmacol. 2009, 68, 61–76. [Google Scholar] [CrossRef] [PubMed]
- Kulus, M.; Hébert, J.; Garcia, E.; Fowler Taylor, A.; Fernandez Vidaurre, C.; Blogg, M. Omalizumab in children with inadequately controlled severe allergic (IgE-mediated) asthma. Curr. Med. Res. Opin. 2010, 26, 1285–1293. [Google Scholar] [CrossRef] [PubMed]
- Pereira Santos, M.C.; Campos Melo, A.; Caetano, A.; Caiado, J.; Mendes, A.; Pereira Barbosa, M.; Branco Ferreira, M. Longitudinal study of the expression of FcεRI and IgE on basophils and dendritic cells in association with basophil function in two patients with severe allergic asthma treated with Omalizumab. Eur. Ann. Allergy Clin. Immunol. 2015, 47, 38–40. [Google Scholar] [PubMed]
- Swain, S.L.; Weinberg, A.D.; English, M.; Huston, G. IL-4 directs the development of Th2-like helper effectors. J. Immunol. 1990, 145, 3796–3806. [Google Scholar] [PubMed]
- Noben-Trauth, N.; Hu-Li, J.; Paul, W.E. Conventional, naive CD4+ T cells provide an initial source of IL-4 during Th2 differentiation. J. Immunol. 2000, 165, 3620–3625. [Google Scholar] [CrossRef] [PubMed]
- Paul, W.E.; Zhu, J. How are TH2-type immune responses initiated and amplified? Nat. Rev. Immunol. 2010, 10, 225–235. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.C.; Zarrin, A.A. The production and regulation of IgE by the immune system. Nat. Rev. Immunol. 2014, 14, 247–259. [Google Scholar] [CrossRef] [PubMed]
- Looney, T.J.; Lee, J.-Y.; Roskin, K.M.; Hoh, R.A.; King, J.; Glanville, J.; Liu, Y.; Pham, T.D.; Dekker, C.L.; Davis, M.M.; et al. Human B-cell isotype switching origins of IgE. J. Allergy Clin. Immunol. 2016, 137, 579–586.e7. [Google Scholar] [CrossRef] [PubMed]
- Robertson, D.G.; Kerigan, A.T.; Hargreave, F.E.; Chalmers, R.; Dolovich, J. Late asthmatic responses induced by ragweed pollen allergen. J. Allergy Clin. Immunol. 1974, 54, 244–254. [Google Scholar] [CrossRef]
- O’Byrne, P. Asthma pathogenesis and allergen-induced late responses. J. Allergy Clin. Immunol. 1998, 102, S85–S89. [Google Scholar] [CrossRef]
- Roth, M.; Zhong, J.; Zumkeller, C.; S’ng, C.T.; Goulet, S.; Tamm, M. The role of IgE-receptors in IgE-dependent airway smooth muscle cell remodelling. PLoS ONE 2013, 8, e56015. [Google Scholar] [CrossRef] [PubMed]
- Redhu, N.S.; Shan, L.; Al-Subait, D.; Ashdown, H.L.; Movassagh, H.; Lamkhioued, B.; Gounni, A.S. IgE induces proliferation in human airway smooth muscle cells: Role of MAPK and STAT3 pathways. Allergy Asthma Clin. Immunol. 2013, 9, 41. [Google Scholar] [CrossRef] [PubMed]
- Janeway, C.; Travers, P.; Walport, M.; Shlomchik, M. Immunobiology: The Immune System in Health and Disease; Garland Science: New York, NY, USA, 2001. [Google Scholar]
- Hart, P.H. Regulation of the inflammatory response in asthma by mast cell products. Immunol. Cell Biol. 2001, 79, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Amin, K. The role of mast cells in allergic inflammation. Respir. Med. 2012, 106, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C.; Alysandratos, K.-D.; Angelidou, A.; Delivanis, D.-A.; Sismanopoulos, N.; Zhang, B.; Asadi, S.; Vasiadi, M.; Weng, Z.; Miniati, A.; et al. Mast cells and inflammation. Biochim. Biophys. Acta 2012, 1822, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Skokos, D.; Botros, H.G.; Demeure, C.; Morin, J.; Peronet, R.; Birkenmeier, G.; Boudaly, S.; Mécheri, S. Mast cell-derived exosomes induce phenotypic and functional maturation of dendritic cells and elicit specific immune responses in vivo. J. Immunol. 2003, 170, 3037–3045. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Wilson, T.M.; Metcalfe, D.D. The mast cell and allergic diseases: Role in pathogenesis and implications for therapy. Clin. Exp. Allergy 2008, 38, 4–18. [Google Scholar] [CrossRef] [PubMed]
- Wills-Karp, M.; Luyimbazi, J.; Xu, X.; Schofield, B.; Neben, T.Y.; Karp, C.L.; Donaldson, D.D. Interleukin-13: Central mediator of allergic asthma. Science 1998, 282, 2258–2261. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Tsai, M. IgE and mast cells in allergic disease. Nat. Med. 2012, 18, 693–704. [Google Scholar] [CrossRef] [PubMed]
- D’Amato, G.; Piccolo, A.; Salzillo, A.; Noschese, P.; D’Amato, M.; Liccardi, G. A recombinant humanized anti-IgE monoclonal antibody (omalizumab) in the therapy of moderate-to-severe allergic asthma. Recent Pat. Inflamm. Allergy Drug Discov. 2007, 1, 225–231. [Google Scholar] [PubMed]
- D’Amato, G.; Liccardi, G.; Noschese, P.; Salzillo, A.; D’Amato, M.; Cazzola, M. Anti-IgE monoclonal antibody (omalizumab) in the treatment of atopic asthma and allergic respiratory diseases. Curr. Drug Targets Inflamm. Allergy 2004, 3, 227–229. [Google Scholar] [CrossRef] [PubMed]
- Licari, A.; Marseglia, G.; Castagnoli, R.; Marseglia, A.; Ciprandi, G. The discovery and development of omalizumab for the treatment of asthma. Expert Opin. Drug Discov. 2015, 10, 1033–1042. [Google Scholar] [CrossRef] [PubMed]
- Anonymous. Omalizumab: Anti-IgE monoclonal antibody E25, E25, humanised anti-IgE MAb, IGE 025, monoclonal antibody E25, Olizumab, Xolair, rhuMAb-E25. BioDrugs 2002, 16, 380–386. [Google Scholar]
- Boulet, L.P.; Chapman, K.R.; Côté, J.; Kalra, S.; Bhagat, R.; Swystun, V.A.; Laviolette, M.; Cleland, L.D.; Deschesnes, F.; Su, J.Q.; et al. Inhibitory effects of an anti-IgE antibody E25 on allergen-induced early asthmatic response. Am. J. Respir. Crit. Care Med. 1997, 155, 1835–1840. [Google Scholar] [CrossRef] [PubMed]
- Massanari, M.; Holgate, S.T.; Busse, W.W.; Jimenez, P.; Kianifard, F.; Zeldin, R. Effect of omalizumab on peripheral blood eosinophilia in allergic asthma. Respir. Med. 2010, 104, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Chipps, B.E.; Marshik, P.L. Targeted IgE therapy for patients with moderate to severe asthma. Biotechnol. Healthc. 2004, 1, 56–61. [Google Scholar] [PubMed]
- Boyman, O.; Kaegi, C.; Akdis, M.; Bavbek, S.; Bossios, A.; Chatzipetrou, A.; Eiwegger, T.; Firinu, D.; Harr, T.; Knol, E.; et al. EAACI IG Biologicals task force paper on the use of biologic agents in allergic disorders. Allergy 2015, 70, 727–754. [Google Scholar] [CrossRef] [PubMed]
- Humbert, M.; Busse, W.; Hanania, N.A.; Lowe, P.J.; Canvin, J.; Erpenbeck, V.J.; Holgate, S. Omalizumab in asthma: An update on recent developments. J. Allergy Clin. Immunol. Pract. 2014, 2, 525–536.e1. [Google Scholar] [CrossRef] [PubMed]
- Levine, S.J.; Wenzel, S.E. Narrative review: The role of Th2 immune pathway modulation in the treatment of severe asthma and its phenotypes. Ann. Intern. Med. 2010, 152, 232–237. [Google Scholar] [CrossRef] [PubMed]
- Fajt, M.L.; Wenzel, S.E. Asthma phenotypes and the use of biologic medications in asthma and allergic disease: The next steps toward personalized care. J. Allergy Clin. Immunol. 2015, 135, 299–310. [Google Scholar] [CrossRef] [PubMed]
- Rudulier, C.D.; Larché, M.; Moldaver, D. Treatment with anti-cytokine monoclonal antibodies can potentiate the target cytokine rather than neutralize its activity. Allergy 2016, 71, 283–285. [Google Scholar] [CrossRef] [PubMed]
- Roth, M.; Tamm, M. The effects of omalizumab on IgE-induced cytokine synthesis by asthmatic airway smooth muscle cells. Ann. Allergy Asthma Immunol. 2010, 104, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Takaku, Y.; Soma, T.; Nishihara, F.; Nakagome, K.; Kobayashi, T.; Hagiwara, K.; Kanazawa, M.; Nagata, M. Omalizumab attenuates airway inflammation and interleukin-5 production by mononuclear cells in patients with severe allergic asthma. Int. Arch. Allergy Immunol. 2013, 161, S107–S117. [Google Scholar] [CrossRef] [PubMed]
- Domingo, C. Omalizumab for severe asthma: Efficacy beyond the atopic patient? Drugs 2014, 74, 521–533. [Google Scholar] [CrossRef] [PubMed]
- Hill, D.A.; Siracusa, M.C.; Ruymann, K.R.; Tait Wojno, E.D.; Artis, D.; Spergel, J.M. Omalizumab therapy is associated with reduced circulating basophil populations in asthmatic children. Allergy 2014, 69, 674–677. [Google Scholar] [CrossRef] [PubMed]
- Sharquie, I.K.; Al-Ghouleh, A.; Fitton, P.; Clark, M.R.; Armour, K.L.; Sewell, H.F.; Shakib, F.; Ghaemmaghami, A.M. An investigation into IgE-facilitated allergen recognition and presentation by human dendritic cells. BMC Immunol. 2013, 14, 54. [Google Scholar] [CrossRef] [PubMed]
- Gill, M.A.; Bajwa, G.; George, T.A.; Dong, C.C.; Dougherty, I.I.; Jiang, N.; Gan, V.N.; Gruchalla, R.S. Counterregulation between the FcepsilonRI pathway and antiviral responses in human plasmacytoid dendritic cells. J. Immunol. 2010, 184, 5999–6006. [Google Scholar] [CrossRef] [PubMed]
- Lommatzsch, M.; Korn, S.; Buhl, R.; Virchow, J.C. Against all odds: Anti-IgE for intrinsic asthma? Thorax 2014, 69, 94–96. [Google Scholar] [CrossRef] [PubMed]
- Busse, W.W.; Morgan, W.J.; Gergen, P.J.; Mitchell, H.E.; Gern, J.E.; Liu, A.H.; Gruchalla, R.S.; Kattan, M.; Teach, S.J.; Pongracic, J.A.; et al. Randomized trial of omalizumab (anti-IgE) for asthma in inner-city children. N. Engl. J. Med. 2011, 364, 1005–1015. [Google Scholar] [CrossRef] [PubMed]
- Teach, S.J.; Gill, M.A.; Togias, A.; Sorkness, C.A.; Arbes, S.J.; Calatroni, A.; Wildfire, J.J.; Gergen, P.J.; Cohen, R.T.; Pongracic, J.A.; et al. Preseasonal treatment with either omalizumab or an inhaled corticosteroid boost to prevent fall asthma exacerbations. J. Allergy Clin. Immunol. 2015, 136, 1476–1485. [Google Scholar] [CrossRef] [PubMed]
- Ortega, H.G.; Liu, M.C.; Pavord, I.D.; Brusselle, G.G.; FitzGerald, J.M.; Chetta, A.; Humbert, M.; Katz, L.E.; Keene, O.N.; Yancey, S.W.; et al. Mepolizumab treatment in patients with severe eosinophilic asthma. N. Engl. J. Med. 2014, 371, 1198–1207. [Google Scholar] [CrossRef] [PubMed]
- Bel, E.H.; Wenzel, S.E.; Thompson, P.J.; Prazma, C.M.; Keene, O.N.; Yancey, S.W.; Ortega, H.G.; Pavord, I.D. Oral glucocorticoid-sparing effect of mepolizumab in eosinophilic asthma. N. Engl. J. Med. 2014, 371, 1189–1197. [Google Scholar] [CrossRef] [PubMed]
- Pavord, I.D.; Korn, S.; Howarth, P.; Bleecker, E.R.; Buhl, R.; Keene, O.N.; Ortega, H.; Chanez, P. Mepolizumab for severe eosinophilic asthma (DREAM): A multicentre, double-blind, placebo-controlled trial. Lancet 2012, 380, 651–659. [Google Scholar] [CrossRef]
- Castro, M.; Zangrilli, J.; Wechsler, M.E.; Bateman, E.D.; Brusselle, G.G.; Bardin, P.; Murphy, K.; Maspero, J.F.; O’Brien, C.; Korn, S. Reslizumab for inadequately controlled asthma with elevated blood eosinophil counts: Results from two multicentre, parallel, double-blind, randomised, placebo-controlled, phase 3 trials. Lancet Respir. Med. 2015, 3, 355–366. [Google Scholar] [CrossRef]
- Bleecker, E.R.; FitzGerald, J.M.; Chanez, P.; Papi, A.; Weinstein, S.F.; Barker, P.; Sproule, S.; Gilmartin, G.; Aurivillius, M.; Werkström, V.; et al. Efficacy and safety of benralizumab for patients with severe asthma uncontrolled with high-dosage inhaled corticosteroids and long-acting β2-agonists (SIROCCO): A randomised, multicentre, placebo-controlled phase 3 trial. Lancet 2016, 388, 2115–2127. [Google Scholar] [CrossRef]
- FitzGerald, J.M.; Bleecker, E.R.; Nair, P.; Korn, S.; Ohta, K.; Lommatzsch, M.; Ferguson, G.T.; Busse, W.W.; Barker, P.; Sproule, S.; et al. Benralizumab, an anti-interleukin-5 receptor α monoclonal antibody, as add-on treatment for patients with severe, uncontrolled, eosinophilic asthma (CALIMA): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2016, 388, 2128–2141. [Google Scholar] [CrossRef]
- Wenzel, S.; Castro, M.; Corren, J.; Maspero, J.; Wang, L.; Zhang, B.; Pirozzi, G.; Sutherland, E.R.; Evans, R.R.; Joish, V.N.; et al. Dupilumab efficacy and safety in adults with uncontrolled persistent asthma despite use of medium-to-high-dose inhaled corticosteroids plus a long-acting β2 agonist: A randomised double-blind placebo-controlled pivotal phase 2b dose-ranging trial. Lancet 2016, 388, 31–44. [Google Scholar] [CrossRef]
- Solèr, M.; Matz, J.; Townley, R.; Buhl, R.; O’Brien, J.; Fox, H.; Thirlwell, J.; Gupta, N.; della Cioppa, G. The anti-IgE antibody omalizumab reduces exacerbations and steroid requirement in allergic asthmatics. Eur. Respir. J. 2001, 18, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Buhl, R.; Hanf, G.; Solèr, M.; Bensch, G.; Wolfe, J.; Everhard, F.; Champain, K.; Fox, H.; Thirlwell, J. The anti-IgE antibody omalizumab improves asthma-related quality of life in patients with allergic asthma. Eur. Respir. J. 2002, 20, 1088–1094. [Google Scholar] [CrossRef] [PubMed]
- Finn, A.; Gross, G.; van Bavel, J.; Lee, T.; Windom, H.; Everhard, F.; Fowler-Taylor, A.; Liu, J.; Gupta, N. Omalizumab improves asthma-related quality of life in patients with severe allergic asthma. J. Allergy Clin. Immunol. 2003, 111, 278–284. [Google Scholar] [CrossRef] [PubMed]
- Corren, J.; Casale, T.; Deniz, Y.; Ashby, M. Omalizumab, a recombinant humanized anti-IgE antibody, reduces asthma-related emergency room visits and hospitalizations in patients with allergic asthma. J. Allergy Clin. Immunol. 2003, 111, 87–90. [Google Scholar] [CrossRef] [PubMed]
- Bousquet, J.; Cabrera, P.; Berkman, N.; Buhl, R.; Holgate, S.; Wenzel, S.; Fox, H.; Hedgecock, S.; Blogg, M.; Cioppa, G.D. The effect of treatment with omalizumab, an anti-IgE antibody, on asthma exacerbations and emergency medical visits in patients with severe persistent asthma. Allergy 2005, 60, 302–308. [Google Scholar] [CrossRef] [PubMed]
| Name | Subclasses | Form | Location | Main Function |
|---|---|---|---|---|
| IgA | 2 | Monomer, dimer and polymer | Mucosal tissue and blood | Opsonization and immune exclusion by binding to noxious antigens and preventing the adherence of microorganisms to the surface epithelium [23] |
| IgD | 1 | Monomer | Surface of mature B cells and blood | Transmembrane antigen receptor of unexposed antigen to complement the functions of IgM [24,25] |
| IgE | 1 | Monomer | Blood | To mediate the signalling response to pathogens [26] |
| IgG | 4 | Monomer | Blood | To bind to antigens to mediate the signalling response to antigens [27] |
| IgM | 1 | Monomer and pentamer | Surface of mature B cells and blood | Initial response to infections [28] |
| IgE | Th2 Cytokines |
|---|---|
| Type I (and also IV) hypersensitivity | Type IV hypersensitivity (IVb) |
| Recognition and specific memory for the involved allergens (e.g., venoms, environmental irritants) | No memory |
| Control in the effector arm of the allergy | Chemical messengers |
| Local and systemic effects | Local effects |
| Mean half-life 2.5 days (months when linked to its receptor) | Mean half-life, minutes |
| Central axis of Th2 response | Redundancy in asthma |
| Beneficial physiological role: response against helminthes | Beneficial physiological role: tissue repair and response against extracellular organisms |
| Examples of hypersensitivity reaction: allergic rhinitis, asthma, systemic anaphylaxis | Examples of hypersensitivity reaction: chronic asthma, chronic allergic rhinitis |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palomares, Ó.; Sánchez-Ramón, S.; Dávila, I.; Prieto, L.; Pérez de Llano, L.; Lleonart, M.; Domingo, C.; Nieto, A. dIvergEnt: How IgE Axis Contributes to the Continuum of Allergic Asthma and Anti-IgE Therapies. Int. J. Mol. Sci. 2017, 18, 1328. https://doi.org/10.3390/ijms18061328
Palomares Ó, Sánchez-Ramón S, Dávila I, Prieto L, Pérez de Llano L, Lleonart M, Domingo C, Nieto A. dIvergEnt: How IgE Axis Contributes to the Continuum of Allergic Asthma and Anti-IgE Therapies. International Journal of Molecular Sciences. 2017; 18(6):1328. https://doi.org/10.3390/ijms18061328
Chicago/Turabian StylePalomares, Óscar, Silvia Sánchez-Ramón, Ignacio Dávila, Luis Prieto, Luis Pérez de Llano, Marta Lleonart, Christian Domingo, and Antonio Nieto. 2017. "dIvergEnt: How IgE Axis Contributes to the Continuum of Allergic Asthma and Anti-IgE Therapies" International Journal of Molecular Sciences 18, no. 6: 1328. https://doi.org/10.3390/ijms18061328
APA StylePalomares, Ó., Sánchez-Ramón, S., Dávila, I., Prieto, L., Pérez de Llano, L., Lleonart, M., Domingo, C., & Nieto, A. (2017). dIvergEnt: How IgE Axis Contributes to the Continuum of Allergic Asthma and Anti-IgE Therapies. International Journal of Molecular Sciences, 18(6), 1328. https://doi.org/10.3390/ijms18061328





