Knockout of Murine Mamld1 Impairs Testicular Growth and Daily Sperm Production but Permits Normal Postnatal Androgen Production and Fertility
Abstract
:1. Introduction
2. Results
2.1. Anatomical, Histological, and Immunohistochemical Examinations of Reproductive Organs of Mamld1 Knockout (KO) Mice
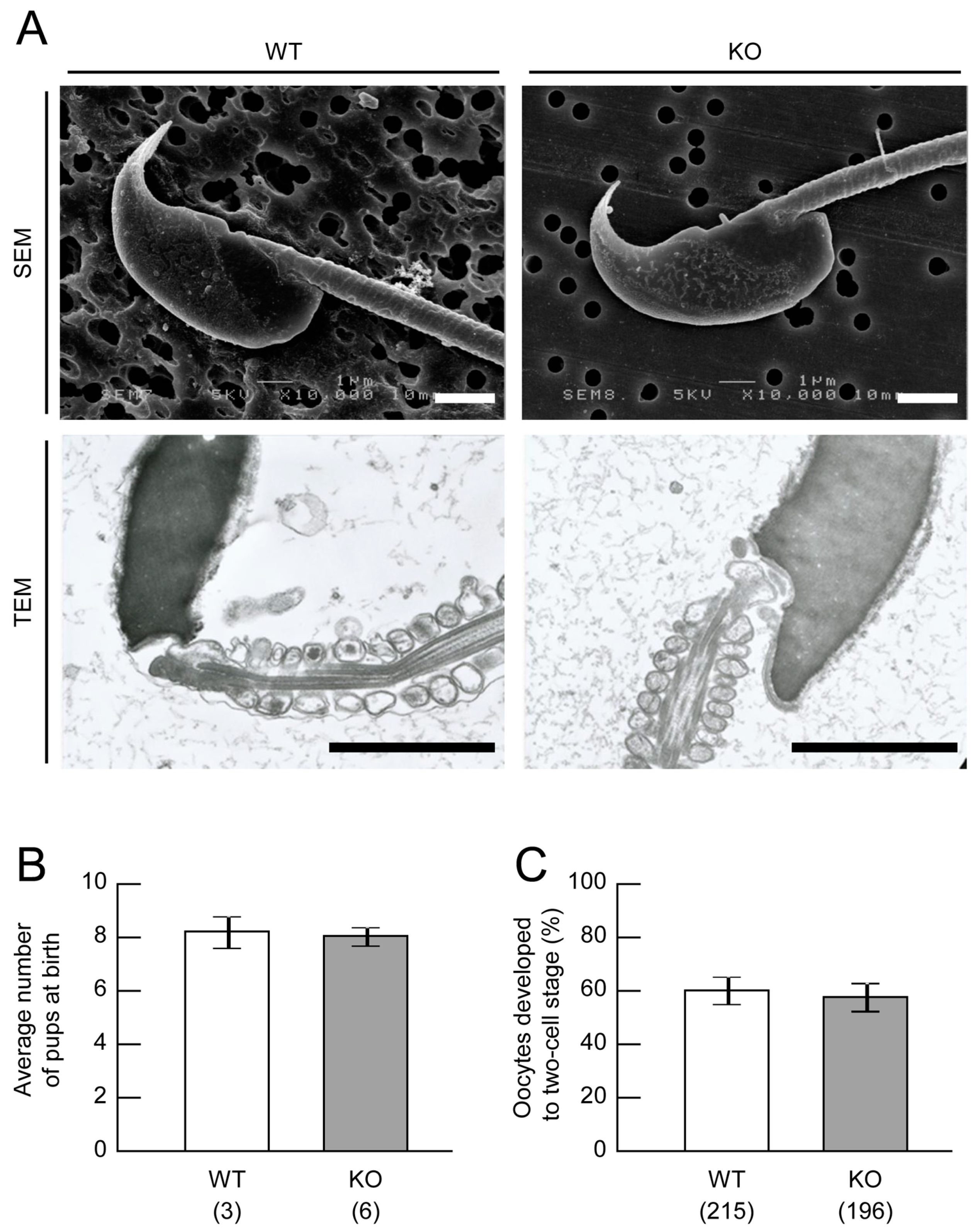
2.2. Sperm Analysis of Mamld1 KO Mice
2.3. Androgen Measurement of Mamld1 KO Mice
2.4. Fertility Assessment of Mamld1 KO Mice
3. Discussion
4. Materials and Methods
4.1. Animal Care
4.2. Morphological, Histological, and Immunohistochemical Examinations of Reproductive Organs of Mamld1 KO Mice
4.3. Sperm Analysis of Mamld1 KO Mice
4.4. Androgen Measurement of Mamld1 KO Mice
4.5. Fertility Assessment of Mamld1 KO Mice
4.6. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fukami, M.; Wada, Y.; Miyabayashi, K.; Nishino, I.; Hasegawa, T.; Nordenskjold, A.; Camerino, G.; Kretz, C.; Buj-Bello, A.; Laporte, J.; et al. CXorf6 is a causative gene for hypospadias. Nat. Genet. 2006, 38, 1369–1371. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, M.; Fukami, M.; Sugawa, F.; Miyado, M.; Nonomura, K.; Ogata, T. Mamld1 knockdown reduces testosterone production and Cyp17a1 expression in mouse leydig tumor cells. PLoS ONE 2011, 6, e19123. [Google Scholar] [CrossRef] [PubMed]
- Miyado, M.; Nakamura, M.; Miyado, K.; Morohashi, K.; Sano, S.; Nagata, E.; Fukami, M.; Ogata, T. Mamld1 deficiency significantly reduces mRNA expression levels of multiple genes expressed in mouse fetal leydig cells but permits normal genital and reproductive development. Endocrinology 2012, 153, 6033–6040. [Google Scholar] [CrossRef] [PubMed]
- Kalfa, N.; Liu, B.; Klein, O.; Audran, F.; Wang, M.H.; Mei, C.; Sultan, C.; Baskin, L.S. Mutations of CXorf6 are associated with a range of severities of hypospadias. Eur. J. Endocrinol. 2008, 159, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Kalfa, N.; Fukami, M.; Philibert, P.; Audran, F.; Pienkowski, C.; Weill, J.; Pinto, G.; Manouvrier, S.; Polak, M.; Ogata, T.; et al. Screening of MAMLD1 mutations in 70 children with 46,XY DSD: Identification and functional analysis of two new mutations. PLoS ONE 2012, 7, e32505. [Google Scholar] [CrossRef] [PubMed]
- Camats, N.; Fernandez-Cancio, M.; Audi, L.; Mullis, P.E.; Moreno, F.; Casado, I.G.; Lopez-Siguero, J.P.; Corripio, R.; de la Vega, J.A.B.; Blanco, J.A.; et al. Human MAMLD1 gene variations seem not sufficient to explain a 46,XY DSD phenotype. PLoS ONE 2015, 10, e0142831. [Google Scholar] [CrossRef] [PubMed]
- Igarashi, M.; Wada, Y.; Kojima, Y.; Miyado, M.; Nakamura, M.; Muroya, K.; Mizuno, K.; Hayashi, Y.; Nonomura, K.; Kohri, K.; et al. Novel splice site mutation in MAMLD1 in a patient with hypospadias. Sex. Dev. 2015, 9, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Fujisawa, Y.; Fukami, M.; Hasegawa, T.; Uematsu, A.; Muroya, K.; Ogata, T. Long-term clinical course in three patients with MAMLD1 mutations. Endocr. J. 2016, 63, 835–839. [Google Scholar] [CrossRef] [PubMed]
- Van Casteren, N.J.; Looijenga, L.H.; Dohle, G.R. Testicular microlithiasis and carcinoma in situ overview and proposed clinical guideline. Int. J. Androl. 2009, 32, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Miyado, M.; Miyado, K.; Katsumi, M.; Saito, K.; Nakamura, A.; Shihara, D.; Ogata, T.; Fukami, M. Parturition failure in mice lacking Mamld1. Sci. Rep. 2015, 5, 14705. [Google Scholar] [CrossRef] [PubMed]
- Jarvis, S.; Elliott, D.J.; Morgan, D.; Winston, R.; Readhead, C. Molecular markers for the assessment of postnatal male germ cell development in the mouse. Hum. Reprod. 2005, 20, 108–116. [Google Scholar] [CrossRef] [PubMed]
- He, Z.; Kokkinaki, M.; Jiang, J.; Dobrinski, I.; Dym, M. Isolation, characterization, and culture of human spermatogonia. Biol. Reprod. 2010, 82, 363–372. [Google Scholar] [CrossRef] [PubMed]
- Vom Saal, F.S.; Cooke, P.S.; Buchanan, D.L.; Palanza, P.; Thayer, K.A.; Nagel, S.C.; Parmigiani, S.; Welshons, W.V. A physiologically based approach to the study of bisphenol A and other estrogenic chemicals on the size of reproductive organs, daily sperm production, and behavior. Toxicol. Ind. Health 1998, 14, 239–260. [Google Scholar] [CrossRef] [PubMed]
- Kawano, N.; Kang, W.; Yamashita, M.; Koga, Y.; Yamazaki, T.; Hata, T.; Miyado, K.; Baba, T. Mice lacking two sperm serine proteases, ACR and PRSS21, are subfertile, but the mutant sperm are infertile in vitro. Biol. Reprod. 2010, 83, 359–369. [Google Scholar] [CrossRef] [PubMed]
- Ohnami, N.; Nakamura, A.; Miyado, M.; Sato, M.; Kawano, N.; Yoshida, K.; Harada, Y.; Takezawa, Y.; Kanai, S.; Ono, C.; et al. CD81 and CD9 work independently as extracellular components upon fusion of sperm and oocyte. Biol. Open 2012, 1, 640–647. [Google Scholar] [CrossRef] [PubMed]
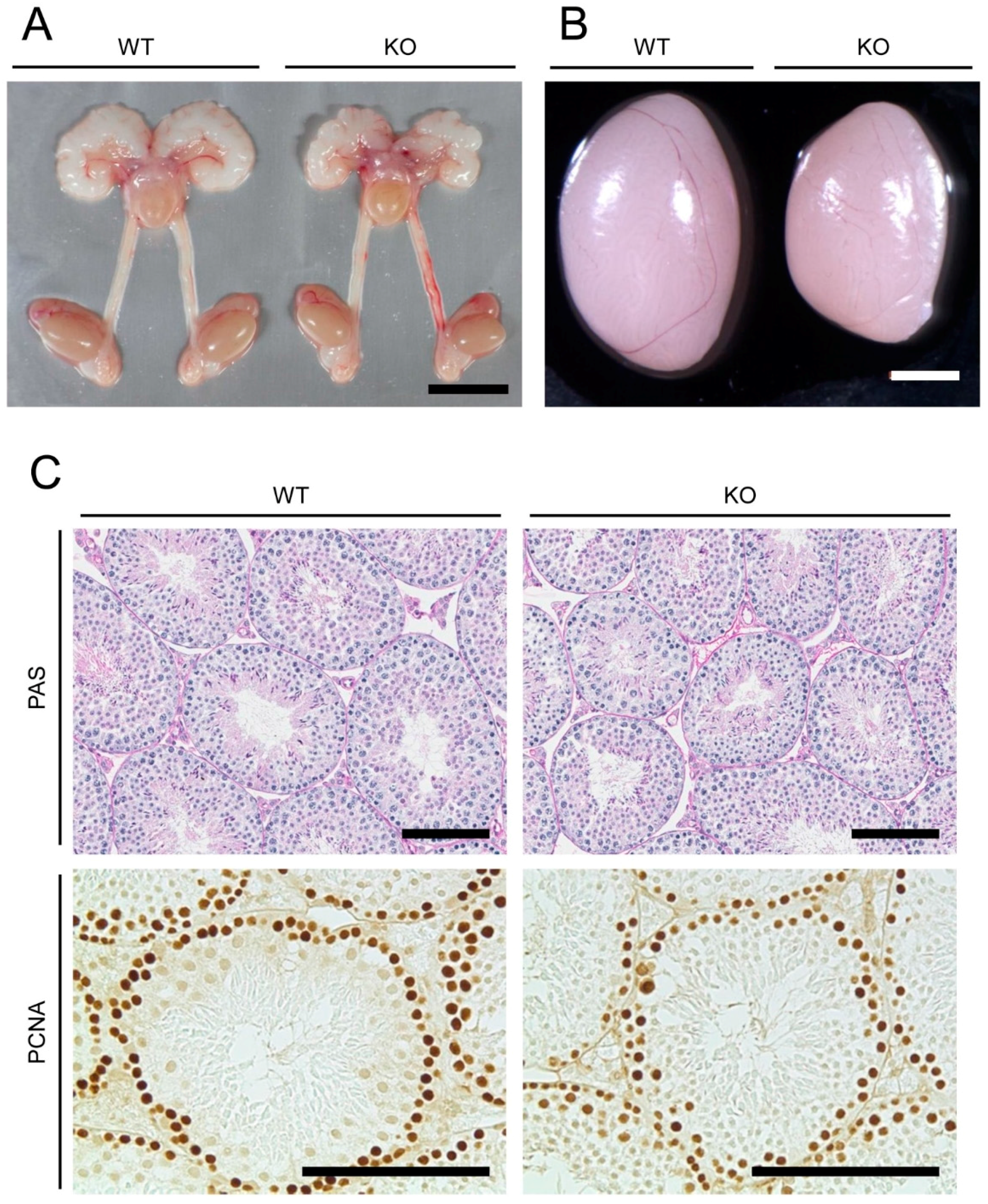
| Organs | Genotype | Statistical Significance (p Values) | |
|---|---|---|---|
| WT | Mamld1 KO | ||
| At 5 weeks of age (n) | 6 | 6 | |
| Body (g) | 21.0 ± 0.6 | 21.4 ± 0.3 | 0.75 |
| Testes (mg) | 134.5 ± 3.6 | 120.7 ± 1.3 | 0.0046 |
| Epididymides (mg) | 35.7 ± 1.6 | 35.7 ± 0.9 | 0.99 |
| At 8 weeks of age (n) | 9 | 8 | |
| Body (g) | 22.6 ± 0.4 | 25.2 ± 0.5 | 0.0015 |
| Testes (mg) | 169.8 ± 4.6 | 151.0 ± 2.7 | 0.0038 |
| Epididymides (mg) | 54.7 ± 1.3 | 55.2 ± 0.9 | 0.79 |
| At 20 weeks of age (n) | 6 | 7 | |
| Body (g) | 30.2 ± 0.6 | 34.8 ± 1.4 | 0.017 |
| Testes (mg) | 208.7 ± 7.6 | 187.2 ± 7.3 | 0.033 |
| Epididymides (mg) | 88.9 ± 2.9 | 86.7 ± 1.0 | 0.48 |
| Coagulating glands (mg) | 29.4 ± 1.4 | 30.2 ± 1.2 | 0.68 |
| Prostate glands (mg) | 39.6 ± 2.8 | 41.2 ± 2.3 | 0.67 |
| Seminal vesicles (mg) | 304.0 ± 11.5 | 320.5 ± 5.9 | 0.21 |
| Preputial glands (mg) | 108.0 ± 4.5 | 95.7 ± 10.6 | 0.34 |
| Parameters | WT | Mamld1 KO | Statistical Significance (p Values) |
|---|---|---|---|
| Daily sperm production per testis (×106) | 6.45 ± 0.24 (n = 6) | 4.67 ± 0.24 (n = 8) | 0.00026 |
| Daily sperm production per gram of testis (×107) | 7.85 ± 0.42 (n = 6) | 6.48 ± 0.24 (n = 8) | 0.011 |
| Epididymal sperm count (×107/mL) | 1.05 ± 0.06 (n = 7) | 1.04 ± 0.08 (n = 6) | 0.89 |
| Parameters | WT (n = 4) | Mamld1 KO (n = 3) | Statistical Significance (p Values) |
|---|---|---|---|
| Total motility (%) | 97.7 ± 0.3 | 94.3 ± 0.9 | 0.94 |
| Progressive motility (%) | 43.0 ± 5.4 | 49.7 ± 2.8 | 0.28 |
| Rapid motility (%) | 55.0 ± 5.1 | 57.7 ± 3.2 | 0.21 |
| Static cell (%) | 2.3 ± 0.3 | 4.0 ± 1.2 | 0.46 |
| Average path velocity (mm/s) | 127.6 ± 2.7 | 133.1 ± 2.3 | 0.37 |
| Amplitude of lateral head displacement (mm) | 8.3 ± 0.02 | 8.6 ± 0.3 | 0.17 |
| Hyperactivation (%) | 16.6 ± 1.7 | 23.0 ± 2.4 | 0.071 |
| Steroids | WT | Mamld1 KO | Statistical Significance (p Values) |
|---|---|---|---|
| Serum androstenedione (pg/mL) | 97.15 ± 41.67 (n = 8) | 243.80 ± 74.26 (n = 10) | 0.25 |
| Serum testosterone (ng/mL) | 2.06 ± 0.95 (n = 10) | 4.47 ± 1.69 (n = 10) | 0.50 |
| Serum dihydrotestosterone (pg/mL) | 105.30 ± 34.46 (n = 10) | 158.90 ± 51.58 (n = 10) | 0.50 |
| Intra-testicular testosterone (ng/testis) | 15.21 ± 5.66 (n = 3) | 30.34 ± 8.75 (n = 3) | 0.22 |
| Intra-testicular testosterone (ng/mg of testis) | 0.22 ± 0.11 (n = 3) | 0.44 ± 0.14 (n = 3) | 0.27 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miyado, M.; Yoshida, K.; Miyado, K.; Katsumi, M.; Saito, K.; Nakamura, S.; Ogata, T.; Fukami, M. Knockout of Murine Mamld1 Impairs Testicular Growth and Daily Sperm Production but Permits Normal Postnatal Androgen Production and Fertility. Int. J. Mol. Sci. 2017, 18, 1300. https://doi.org/10.3390/ijms18061300
Miyado M, Yoshida K, Miyado K, Katsumi M, Saito K, Nakamura S, Ogata T, Fukami M. Knockout of Murine Mamld1 Impairs Testicular Growth and Daily Sperm Production but Permits Normal Postnatal Androgen Production and Fertility. International Journal of Molecular Sciences. 2017; 18(6):1300. https://doi.org/10.3390/ijms18061300
Chicago/Turabian StyleMiyado, Mami, Kaoru Yoshida, Kenji Miyado, Momori Katsumi, Kazuki Saito, Shigeru Nakamura, Tsutomu Ogata, and Maki Fukami. 2017. "Knockout of Murine Mamld1 Impairs Testicular Growth and Daily Sperm Production but Permits Normal Postnatal Androgen Production and Fertility" International Journal of Molecular Sciences 18, no. 6: 1300. https://doi.org/10.3390/ijms18061300
APA StyleMiyado, M., Yoshida, K., Miyado, K., Katsumi, M., Saito, K., Nakamura, S., Ogata, T., & Fukami, M. (2017). Knockout of Murine Mamld1 Impairs Testicular Growth and Daily Sperm Production but Permits Normal Postnatal Androgen Production and Fertility. International Journal of Molecular Sciences, 18(6), 1300. https://doi.org/10.3390/ijms18061300






