Flavonoids, Thyroid Iodide Uptake and Thyroid Cancer—A Review
Abstract
:1. Introduction
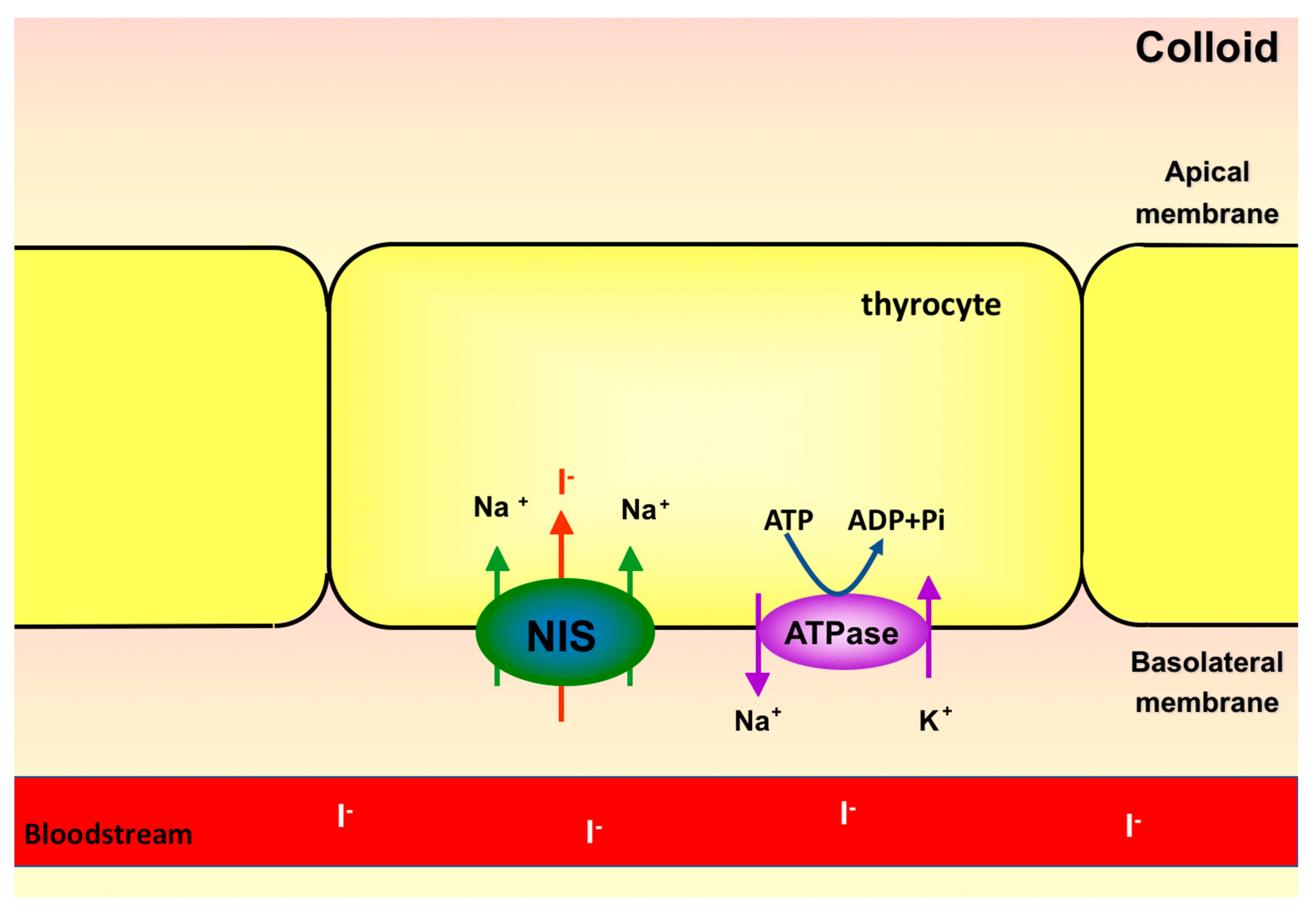
2. Sodium-Iodide Symporter
3. Thyroid Cancer
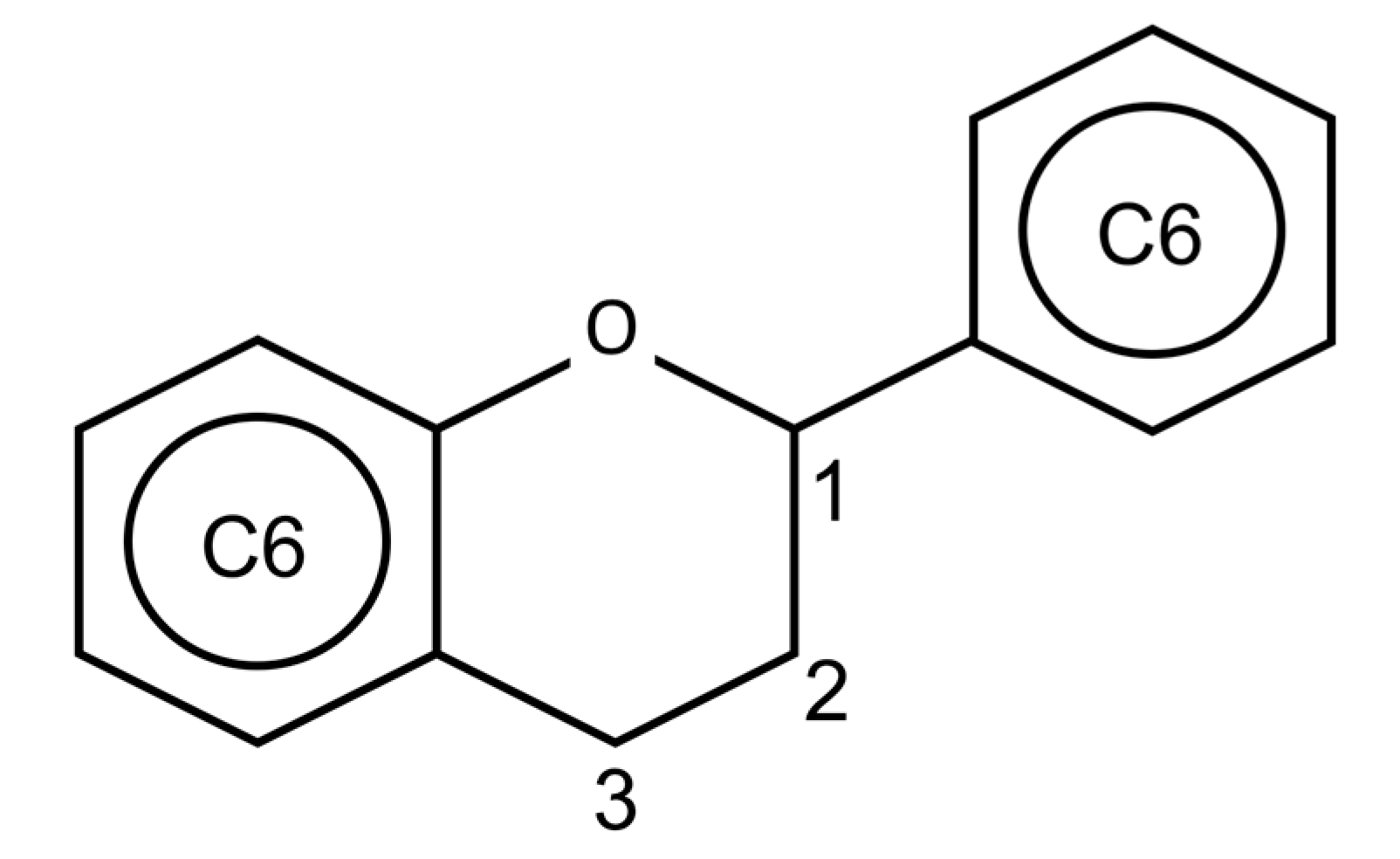
4. Flavonoids
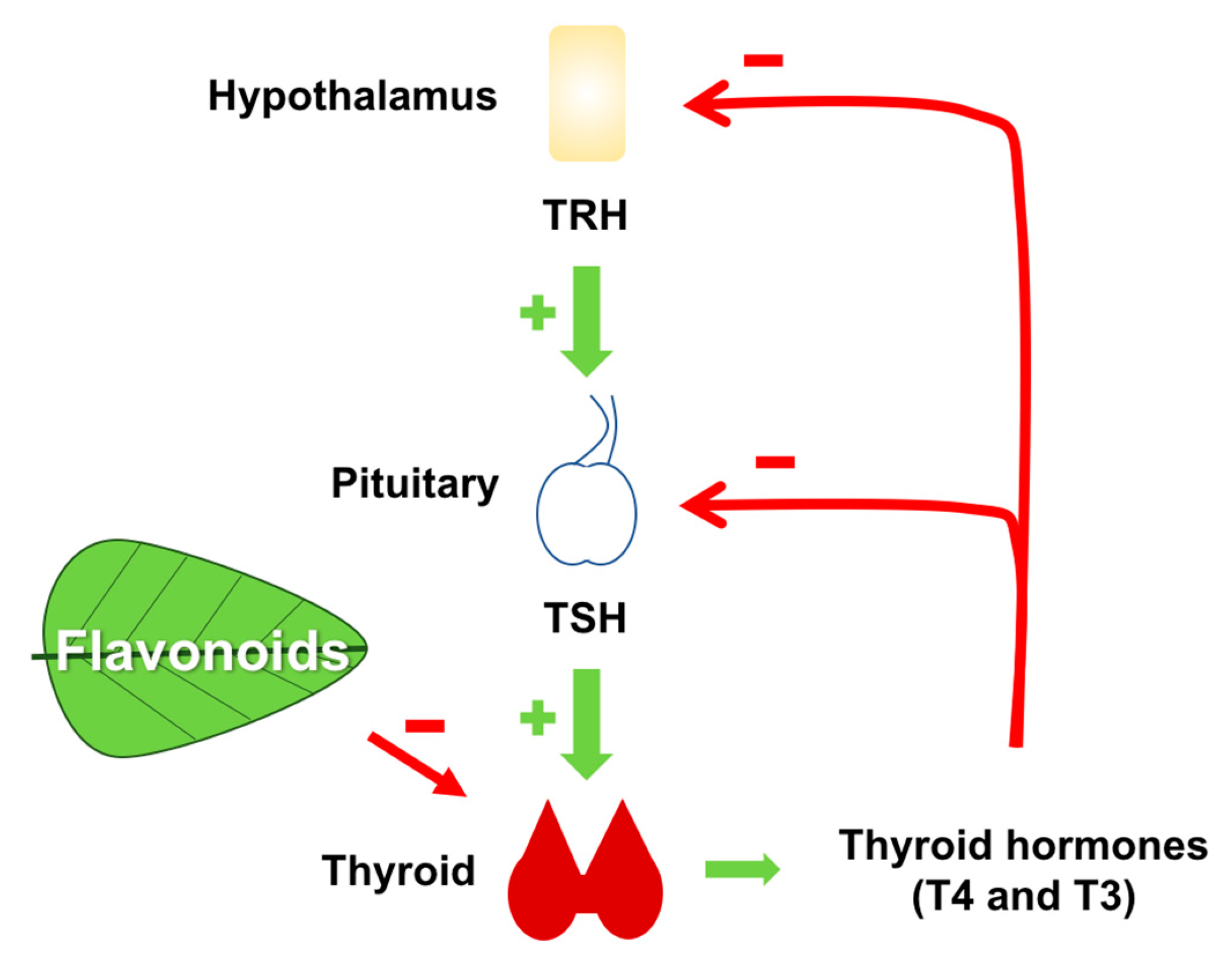
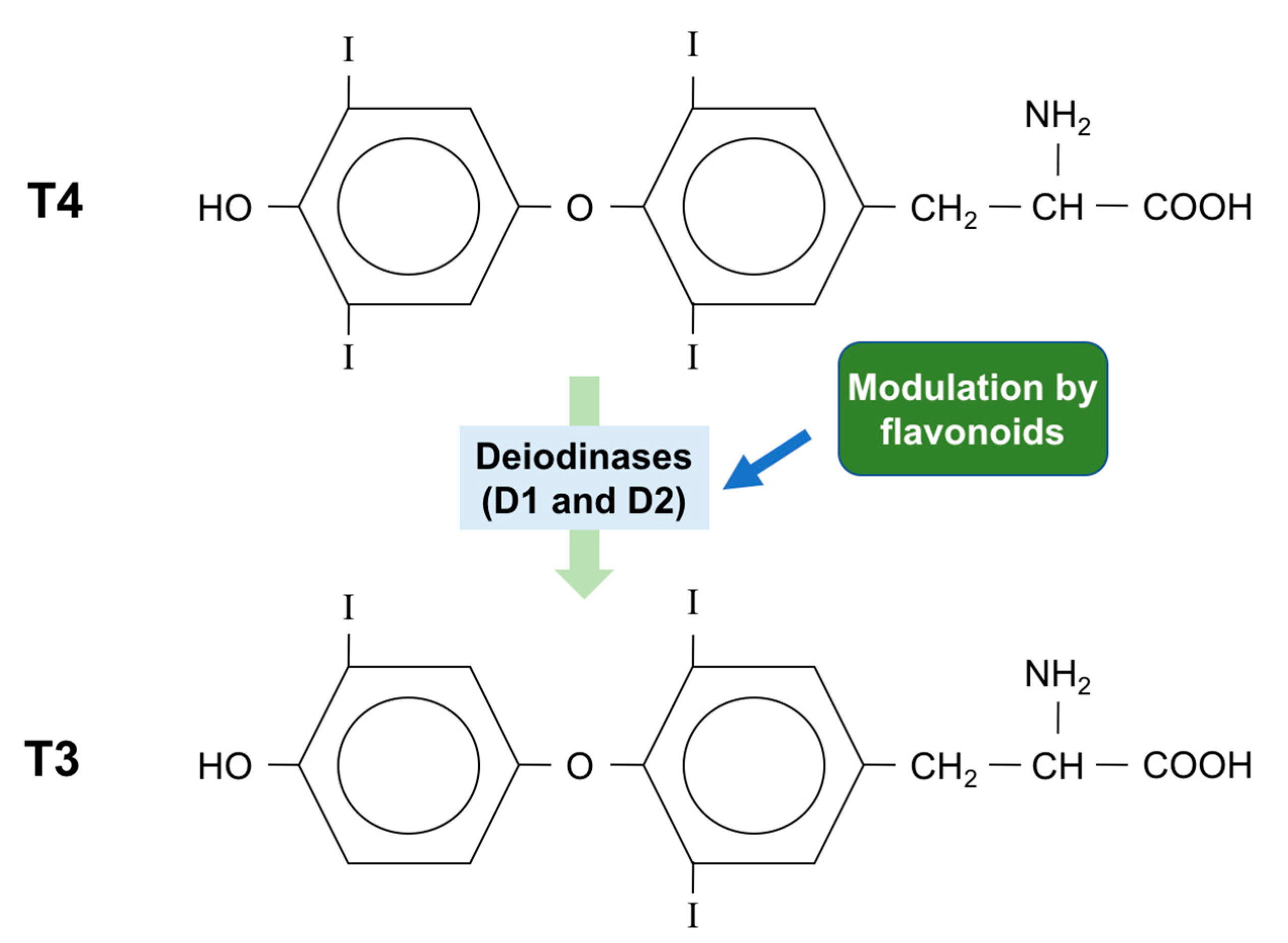
5. Effect of Flavonoids on the Synthesis and Metabolism of Thyroid Hormones
6. Flavonoids and Cancer
7. Effect of Flavonoids on Iodide Uptake and Thyroid Cancer
8. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De Souza Dos Santos, M.C.; Gonçalves, C.F.; Vaisman, M.; Ferreira, A.C.; de Carvalho, D.P. Impact of flavonoids on thyroid function. Food Chem. Toxicol. 2011, 49, 2495–2502. [Google Scholar] [CrossRef] [PubMed]
- De Vries, J.H.M.; Janssen, P.L.; Hollman, P.C.H.; van Staveren, W.A.; Katan, M.B. Consumption of quercetin and kaempferol in free-living subjects eating a variety of diets. Cancer Lett. 1997, 114, 141–144. [Google Scholar] [CrossRef]
- Hertog, M.G.; Kromhout, D.; Aravanis, C.; Blackburn, H.; Buzina, R.; Fidanza, F.; Giampaoli, S.; Jansen, A.; Menotti, A.; Nedeljkovic, S.; et al. Flavonoid intake and long-term risk of coronary heart disease and cancer in the seven countries study. Arch. Intern. Med. 1995, 155, 381–386. [Google Scholar] [CrossRef] [PubMed]
- Justesen, U.; Knuthsen, P.; Leth, T. Determination of plant polyphenols in Danish foodstuffs by HPLC-UV and LC-MS detection. Cancer Lett. 1997, 114, 165–167. [Google Scholar] [CrossRef]
- Middleton, E.J.R.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar] [PubMed]
- Lopes, H.F.; Martin, K.L.; Nashar, K.; Morrow, J.D.; Goodfriend, T.L.; Egan, B.M. Dash diet lowers blood pressure and lipid-induced oxidative stress in obesity. Hypertension 2003, 41, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Moudgal, N.R.; Raghupathy, E.; Sarma, O.S. Studies on goitrogenic agents in food. III. Goitrogenic action of some glycosides isolated from edible nuts. J. Nutr. 1958, 66, 291–300. [Google Scholar] [PubMed]
- Gaitan, E. Flavonoids and the thyroid. Nutrition 1996, 12, 127–129. [Google Scholar] [CrossRef]
- Gaitan, E.; Lindsay, R.H.; Reichert, R.D.; Ingbar, S.H.; Cooksey, R.C.; Legan, J.; Meydrech, E.F.; Hill, J.; Kubota, K. Antithyroid and goitrogenic effects of millet: Role of C-glycosylflavones. J. Clin. Endocrinol. Metab. 1989, 68, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Sartelet, H.; Serghat, S.; Lobstein, A.; Ingenbleek, A.R.; Petitfrère, A.A.G.; Martiny, L.; Haye, H. Flavonoids extracted from fonio millet (Digitaria exilis) reveal potent antithyroid properties. Nutrition 1996, 12, 100–106. [Google Scholar] [CrossRef]
- Divi, R.L.; Doerge, D.R. Inhibition of thyroid peroxidase by dietary flavonoids. Chem. Res. Toxicol. 1996, 9, 16–23. [Google Scholar] [CrossRef] [PubMed]
- Divi, R.L.; Chang, H.C.; Doerge, D.R. Anti-thyroid isoflavones from soybean: Isolation, characterization, and mechanisms of action. Biochem. Pharmacol. 1997, 54, 1087–1096. [Google Scholar] [CrossRef]
- Ferreira, A.C.F.; Rosenthal, D.; Carvalho, D.P. Thyroid peroxidase inhibition by Kalanchoe brasiliensis aqueous extract. Food Chem. Toxicol. 2000, 38, 417–421. [Google Scholar] [CrossRef]
- Ferreira, A.C.F.; Neto, J.C.; Da Silva, A.C.; Kuster, R.M.; Carvalho, D.P. Inhibition of thyroid peroxidase by Myrcia uniflora flavonoids. Chem. Res. Toxicol. 2006, 19, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, C.F.L.; Santos, M.C.; Ginabreda, M.G.; Fortunato, R.S.; Carvalho, D.P.; Freitas Ferreira, A.C. Flavonoid rutin increases thyroid iodide uptake in rats. PLoS ONE 2013, 8, e73908. [Google Scholar] [CrossRef] [PubMed]
- Spanka, M.; Hesch, R.D.; Irmscher, K.; Köhrle, J. 5′-Deiodination in rat hepatocytes: Effects of specific flavonoid inhibitors. Endocrinology 1990, 126, 1660–1667. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.C.F.; Lisboa, P.C.; Oliveira, K.J.; Lima, L.P.; Barros, I.A.; Carvalho, D.P. Inhibition of thyroid type 1 deiodinase activity by flavonoids. Food Chem. Toxicol. 2002, 40, 913–917. [Google Scholar] [CrossRef]
- Da-Silva, W.S.; Harney, J.W.; Kim, B.W.; Li, J.; Bianco, S.D.; Crescenzi, A.; Christoffolete, M.A.; Huang, S.A.; Bianco, A.C. The small polyphenolic molecule kaempferol increases cellular energy expenditure and thyroid hormone activation. Diabetes 2007, 56, 767–776. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.; Kar, A. Annonasquamosa seed extract in the regulation of hyperthyroidism and lipid-peroxidation in mice: Possible involvement of quercetin. Phytomedicine 2007, 14, 799–805. [Google Scholar] [CrossRef] [PubMed]
- Chandra, A.K.; De, N. Goitrogenic/antithyroidal potential of green tea extract in relation to catechin in rats. Food Chem. Toxicol. 2010, 48, 2304–2311. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.; Ha, T.K.; Han, S.H.; Kim, M.E.; Jung, I.; Lee, H.W.; Bae, S.K.; Lee, J.S. Myricetin induces apoptosis of human anaplastic thyroid cancer cells via mitochondria dysfunction. Anticancer Res. 2017, 37, 1705–1710. [Google Scholar] [PubMed]
- Mutlu Altundag, E.; Mine Yilmaz, A.; Kasaci, T.; Corek, C.; Taga, Y.; Suha Yalçin, A. The role of HSP90 in Quercetin-induced apoptosis in human papillary thyroid (B-CPAP) cancer cells. Free Radic. Biol. Med. 2014, 75 (Suppl. 1), S43. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Cheng, X.; Gao, Y.; Zheng, J.; Xu, Q.; Sun, Y.; Guan, H.; Yu, H.; Sun, Z. Apigenin induces autophagic cell death in human papillary thyroid carcinoma BCPAP cells. Food Funct. 2015, 6, 3464–3472. [Google Scholar] [CrossRef] [PubMed]
- Schröder-van der Elst, J.P.; van der Heide, D.; Romijn, J.A.; Smit, J.W. Differential effects of natural flavonoids on growth and iodide content in a human Na+/I− symporter-transfected follicular thyroid carcinoma cell line. Eur. J. Endocrinol. 2004, 150, 557–564. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, A.; Doseff, A.I.; Ringel, M.D.; Saji, M.; Rousset, B.; Zhang, X.; Jhiang, S.M. Apigenin in combination with Akt inhibition significantly enhances thyrotropin-stimulated radioiodide accumulation in thyroid cells. Thyroid 2014, 24, 878–887. [Google Scholar] [CrossRef] [PubMed]
- Carvalho, D.P.; Ferreira, A.C. The importance of sodium/iodide symporter (NIS) for thyroid cancer management. Arq. Bras. Endocrinol. Metabol. 2007, 51, 672–682. [Google Scholar] [CrossRef] [PubMed]
- Dohán, O.; De La Vieja, A.; Paroder, V.; Riedel, C.; Artani, M.; Reed, M.; Ginter, C.S.; Carrasco, N. The sodium/iodide Symporter (NIS): Characterization, regulation, and medical significance. Endocr. Rev. 2003, 24, 48–77. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.C.; Lima, L.P.; Araújo, R.L.; Müller, G.; Rocha, R.P.; Rosenthal, D.; Carvalho, D.P. Rapid regulation of thyroid sodium-iodide symporter activity by thyrotrophin and iodine. J. Endocrinol. 2005, 184, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Salvatore, D.; Davies, T.F.; Schlumberger, M.J.; Hay, I.D.; Larsen, P.R. Thyroid Physiology and Diagnostic Evaluation of Patients with Thyroid Disorders. In Williams Textbook of Endocrinology, 12th ed.; Melmed, S., Polonsky, K.S., Larsen, P.R., Kronemberg, H.M., Eds.; W.B. Saunders Company: Philadelphia, PA, USA, 2011; pp. 327–475. [Google Scholar]
- Schmutzler, C.; Winzer, R.; Meissner-Weigl, J.; Köhrle, J. Retinoic acid increases sodium/iodide symporter mRNA levels in human thyroid cancer cell lines and suppresses expression of functional symporter in nontransformed FRTL-5 rat thyroid cells. Biochem. Biophys. Res. Commun. 1997, 240, 832–838. [Google Scholar] [CrossRef] [PubMed]
- Kogai, T.; Sajid-Crockett, S.; Newmarch, L.S.; Liu, Y.Y.; Brent, G.A. Phosphoinositide-3-kinase inhibition induces sodium/iodide symporter expression in rat thyroid cells and human papillary thyroid cancer cells. J. Endocrinol. 2008, 199, 243–252. [Google Scholar] [CrossRef] [PubMed]
- Brown, R.L.; De Souza, J.A.; Cohen, E.E. Thyroid cancer: Burden of illness and management of disease. J. Cancer 2011, 4, 193–199. [Google Scholar] [CrossRef]
- Dai, G.; Levy, O.; Carrasco, N. Cloning and characterization of the thyroid iodide transporter. Nature 1996, 379, 458–460. [Google Scholar] [CrossRef] [PubMed]
- Smanik, P.A.; Liu, Q.; Furminger, T.L.; Ryu, K.; Xing, S.; Mazzaferri, E.L.; Jhiang, S.M. Cloning of the human sodium lodide symporter. Biochem. Biophys. Res. Commun. 1996, 226, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Wright, E.M.; Turk, E. The sodium/glucose cotransport family SLC5. Pflugers Arch. 2004, 447, 510–518. [Google Scholar] [CrossRef] [PubMed]
- Portulano, C.; Paroder-Belenitsky, M.; Carrasco, N. The Na+/I− symporter (NIS): Mechanism and medical impact. Endocr. Rev. 2014, 35, 106–149. [Google Scholar] [CrossRef] [PubMed]
- Levy, O.; De La Vieja, A.; Ginter, C.S.; Riedel, C.; Dai, G.; Carrasco, N. N-linked glycosylation of the thyroid Na+/I− symporter (NIS). Implications for its secondary structure model. J. Biol. Chem. 1998, 273, 22657–22663. [Google Scholar] [CrossRef] [PubMed]
- Dohán, O.; Portulano, C.; Basquin, C.; Reyna-Neyra, A.; Amzel, L.M.; Carrasco, N. The Na+/I− symporter (NIS) mediates electroneutral active transport of the environmental pollutant perchlorate. Proc. Natl. Acad. Sci. USA 2007, 104, 20250–20255. [Google Scholar] [CrossRef] [PubMed]
- Eskandari, S.; Loo, D.D.; Dai, G.; Levy, O.; Wright, E.M.; Carrasco, N. Thyroid Na+/I− symporter. Mechanism, stoichiometry, and specificity. J. Biol. Chem. 1997, 272, 27230–27238. [Google Scholar] [CrossRef] [PubMed]
- Vassart, G.; Dumont, J.E. The thyrotropin receptor and the regulation of thyrocyte function and growth. Endocr. Rev. 1992, 13, 596–611. [Google Scholar] [PubMed]
- Kogai, T.; Endo, T.; Saito, T.; Miyazaki, A.; Kawaguchi, A.; Onaya, T. Regulation by thyroid-stimulating hormone of sodium/iodide symporter gene expression and protein levels in FRTL-5 cells. Endocrinology 1997, 138, 2227–2232. [Google Scholar] [CrossRef] [PubMed]
- Ohno, M.; Zannini, M.; Levy, O.; Carrasco, N.; Di Lauro, R. The paired-domain transcription factor Pax8 binds to the upstream enhancer of the rat sodium/iodide symporter gene and participates in both thyroid-specific and cyclic-AMP-dependent transcription. Mol. Cell. Biol. 1999, 19, 2051–2060. [Google Scholar] [CrossRef] [PubMed]
- De La Vieja, A.; Dohan, O.; Levy, O.; Carrasco, N. Molecular analysis of the sodium/iodide symporter: Impact on thyroid and extrathyroid pathophysiology. Physiol. Rev. 2000, 80, 1083–1105. [Google Scholar] [PubMed]
- Riedel, C.; Levy, O.; Carrasco, N. Post-transcriptional regulation of the sodium/iodide symporter by thyrotropin. J. Biol. Chem. 2001, 276, 21458–21463. [Google Scholar] [CrossRef] [PubMed]
- Weiss, S.J.; Philp, N.J.; Ambesi-Impiombato, F.S.; Grollman, E.F. Thyrotropin-stimulated iodide transport mediated by adenosine 3′,5′-monophosphate and dependent on protein synthesis. Endocrinology 1984, 114, 1099–1107. [Google Scholar] [CrossRef] [PubMed]
- Kogai, T.; Taki, K.; Brent, G.A. Enhancement of sodium/iodide symporter expression in thyroid and breast cancer. Endocr. Relat. Cancer 2006, 13, 797–826. [Google Scholar] [CrossRef] [PubMed]
- Vadysirisack, D.D.; Venkateswaran, A.; Zhang, Z.; Jhiang, S.M. MEK signaling modulates sodium iodide symporter at multiple levels and in a paradoxical manner. Endocr. Relat. Cancer 2007, 14, 421–432. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Paik, J.Y.; Ko, B.H.; Lee, K.H. Mitogen-activated protein kinase signaling enhances sodium iodide symporter function and efficacy of radioiodide therapy in nonthyroidal cancer cells. J. Nucl. Med. 2008, 49, 1966–1972. [Google Scholar] [CrossRef] [PubMed]
- Saji, M.; Kohn, L.D. Insulin and insulin-like growth factor-I inhibit thyrotropin-increased iodide transport in serum-depleted FRTL-5 rat thyroid cells: Modulation of adenosine 3′,5′-monophosphate signal action. Endocrinology 1991, 128, 1136–1143. [Google Scholar] [CrossRef] [PubMed]
- García, B.; Santisteban, P. PI3K is involved in the IGF-I inhibition of TSH-induced sodium/iodide symporter gene expression. Mol. Endocrinol. 2002, 16, 342–352. [Google Scholar] [CrossRef] [PubMed]
- De Souza, E.C.; Padrón, A.S.; Braga, W.M.; de Andrade, B.M.; Vaisman, M.; Nasciutti, L.E.; Ferreira, A.C.; de Carvalho, D.P. MTOR downregulates iodide uptake in thyrocytes. J. Endocrinol. 2010, 206, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Andrade, B.M.; Araujo, R.L.; Perry, R.L.S.; Souza, E.C.L.; Cazarin, J.M.; Carvalho, D.P.; Ceddia, R.B. A novel role for AMP-kinase in the regulation of the Na+/I− symporter and iodide uptake in the rat thyroid gland. Am. J. Physiol. Cell Physiol. 2011, 300, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Abdulrahman, R.M.; Boon, M.R.; Sips, H.C.; Guigas, B.; Rensen, P.C.; Smit, J.W.; Hovens, G.C. Impact of metformin and compound c on nis expression and iodine Uptake in vitro and in vivo: A role for CRE in AMPK modulation of thyroid function. Thyroid 2014, 24, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Cazarin, J.M.; Andrade, B.M.; Carvalho, D.P. AMP-activated protein kinase activation leads to lysome-mediated Na(+)/I(−)-symporter protein degradation in rat thyroid cells. Horm. Metab. Res. 2014, 46, 313–317. [Google Scholar] [CrossRef] [PubMed]
- Pacifico, F.; Leonardi, A. Role of NF-κB in thyroid cancer. Mol. Cell. Endocrinol. 2010, 321, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA). Coordenação de Prevenção e Vigilância Estimativa 2016: Incidência de câncer No Brasil; Instituto Nacional de Câncer José Alencar Gomes da Silva (INCA): Rio de Janeiro, Brasil, 2015. [Google Scholar]
- Albi, E.; Cataldi, S.; Lazzarini, A.; Codini, M.; Beccari, T.; Ambesi-Impiombato, F.S.; Curcio, F. Radiation and thyroid cancer. Int. J. Mol. Sci. 2017, 18, 911. [Google Scholar] [CrossRef] [PubMed]
- Fortunato, R.S.; Ferreira, A.C.; Hecht, F.; Dupuy, C.; Carvalho, D.P. Sexual dimorphism and thyroid dysfunction: A matter of oxidative stress? J. Endocrinol. 2014, 221, R31–R40. [Google Scholar] [CrossRef] [PubMed]
- Iribarren, C.; Haselkorn, T.; Tekawa, I.S.; Friedman, G.D. Cohort study of thyroid cancer in a San Francisco Bay area population. Int. J. Cancer 2001, 93, 745–750. [Google Scholar] [CrossRef] [PubMed]
- Albi, E.; Krüger, M.; Hemmersbach, R.; Lazzarini, A.; Cataldi, S.; Codini, M.; Beccari, T.; Ambesi-Impiombato, F.S.; Curcio, F. Impact of gravity on thyroid cells. Int. J. Mol. Sci. 2017, 18, 972. [Google Scholar] [CrossRef] [PubMed]
- Kondo, T.; Ezzat, S.; Asa, S.L. Pathogenetic mechanisms in thyroid follicular-cell neoplasia. Nat. Rev. Cancer 2006, 6, 292–306. [Google Scholar] [CrossRef] [PubMed]
- Maciel, R. Carcinoma diferenciado da tiróide (Papilífero e Folicular): Diagnóstico e conduta. Arq. Bras. Endocrinol. Metabol. 1998, 42, 299–305. [Google Scholar] [CrossRef]
- Mete, O.; Asa, S.L. Pathological definition and clinical significance of vascular invasion in thyroid carcinomas of follicular epithelial derivation. Mod. Pathol. 2011, 24, 1545–1552. [Google Scholar] [CrossRef] [PubMed]
- Livolsi, V.A.; Asa, S.L. The demise of follicular carcinoma of the thyroid gland. Thyroid 1994, 4, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Xing, M. BRAF mutation in thyroid cancer. Endocr. Relat. Cancer 2005, 12, 245–262. [Google Scholar] [CrossRef] [PubMed]
- Wellbrock, C.; Karasarides, M.; Marais, R. The RAF proteins take center stage. Nat. Rev. Mol. Cell Biol. 2004, 5, 875–885. [Google Scholar] [CrossRef] [PubMed]
- Peyssonnaux, C.; Eychene, A. The Raf/MEK/ERK pathway: New concepts of activation. Biol. Cell 2011, 93, 53–62. [Google Scholar] [CrossRef]
- Kimura, E.T.; Nikiforova, M.N.; Zhu, Z.; Knauf, J.A.; Nikiforov, Y.E.; Fagin, J.A. High prevalence of BRAF mutations in thyroid cancer: Genetic evidence for constitutive activation of the RET/PTC-RAS-BRAF signaling pathway in papillary thyroid carcinoma. Cancer Res. 2003, 63, 1454–1457. [Google Scholar] [PubMed]
- Myers, S.M.; Eng, C.; Ponder, B.A.; Mulligan, L.M. Characterization of RET proto-oncogene 3′ splicing variants and polyadenylation sites: A novel C-terminus for RET. Oncogene 1995, 11, 2039–2045. [Google Scholar] [PubMed]
- Grieco, M.; Santoro, M.; Berlingieri, M.T.; Melillo, R.M.; Donghi, R.; Bongarzone, I.; Pierotti, M.A.; Della Porta, G.; Fusco, A.; Vecchio, G. PTC is a novel rearranged form of the RET proto-oncogene and is frequently detected in vivo in human thyroid papillary carcinomas. Cell 1990, 60, 557–563. [Google Scholar] [CrossRef]
- Santoro, M.; Melillo, R.M.; Fusco, A. RET/PTC activation in papillary thyroid carcinoma: European Journal of Endocrinology Prize Lecture. Eur. J. Endocrinol. 2006, 155, 645–653. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, N.; Akbani, R.; Aksoy, B.A.; Ally, A.; Arachchi, H.; Asa, S.L.; Auman, J.T.; Balasundaram, M.; Balu, S.; Baylin, S.B.; et al. Integrated genomic characterization of papillary thyroid carcinoma. Cell 2014, 159, 676–690. [Google Scholar] [CrossRef] [PubMed]
- Sabra, M.M.; Dominguez, J.M.; Grewal, R.K.; Larson, S.M.; Ghossein, R.A.; Tuttle, R.M.; Fagin, J.A. Clinical outcomes and molecular profile of differentiated thyroid cancers with radioiodine-avid distant metastases. J. Clin. Endocrinol. Metab. 2013, 98, E829–E836. [Google Scholar] [CrossRef] [PubMed]
- Chakravarty, D.; Santos, E.; Ryder, M.; Knauf, J.A.; Liao, X.H.; West, B.L.; Bollag, G.; Kolesnick, R.; Thin, T.H.; Rosen, N.; et al. Small-molecule MAPK inhibitors restore radioiodine incorporation in mouse thyroid cancers with conditional BRAF activation. J. Clin. Investig. 2011, 121, 4700–4711. [Google Scholar] [CrossRef] [PubMed]
- Durante, C.; Puxeddu, E.; Ferretti, E.; Morisi, R.; Moretti, S.; Bruno, R.; Barbi, F.; Avenia, N.; Scipioni, A.; Verrienti, A.; et al. BRAF mutations in papillary thyroid carcinomas inhibit genes involved in iodine metabolism. J. Clin. Endocrinol. Metab. 2007, 92, 2840–2843. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Hu, S.; Hou, P.; Jiang, D.; Condouris, S.; Xing, M. Suppression of BRAF/MEK/MAP kinase pathway restores expression of iodide-metabolizing genes in thyroid cells expressing the V600E BRAF mutant. Clin. Cancer Res. 2007, 13, 1341–1349. [Google Scholar] [CrossRef] [PubMed]
- Mitsutake, N.; Knauf, J.A.; Mitsutake, S.; Mesa, C., Jr.; Zhang, L.; Fagin, J.A. Conditional BRAFV600E expression induces DNA synthesis, apoptosis, dedifferentiation, and chromosomal instability in thyroid PCCL3 cells. Cancer Res. 2005, 65, 2465–2473. [Google Scholar] [CrossRef] [PubMed]
- Coelho, S.M.; Carvalho, D.P.; Vaisman, M. New perspectives on the treatment of differentiated thyroid cancer. Arq. Bras. Endocrinol. Metabol. 2007, 51, 612–624. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Neilson, E.G. Epithelial-mesenchymal transition and its implications for fibrosis. J. Clin. Invest. 2003, 112, 1776–1784. [Google Scholar] [CrossRef] [PubMed]
- Kalluri, R.; Weinberg, R.A. The basics of epithelial-mesenchymal transition. J. Clin. Invest. 2009, 119, 1420–1428. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, A.; Scarberry, D.; Shen, D.H.; Jhiang, S.M. Modulation of sodium iodide symporter in thyroid cancer. Horm. Cancer 2014, 5, 363–373. [Google Scholar] [CrossRef] [PubMed]
- Haugen, B.R. Management of the patient with progressive radioiodine non-responsive disease. Semin. Surg. Oncol. 1999, 16, 34–41. [Google Scholar] [CrossRef]
- Kandaswami, C.; Middleton, E.J.R. Free radical scavenging and antioxidant activity of plant flavonoids. Advant. Exp. Med. Biol. 1994, 366, 351–376. [Google Scholar]
- Ross, J.A.; Kasum, C.M. Dietary flavonoids: Bioavailability, metabolic effects, and safety. Ann. Rev. Nutr. 2002, 22, 19–34. [Google Scholar] [CrossRef] [PubMed]
- Zuanazzi, J.A.S.; Montanha, J.A. Flavonoides. In Farmacognosia: Da Planta ao Medicamento, 6th ed.; Simões, C.M.O., Schenkel, E.P., Gosmann, G., Mello, J.C.P., Mentz, L.A., Petrovick, P.R., Eds.; da UFSC: Porto Alegre, Brasil, 2010; pp. 489–516. [Google Scholar]
- Liggins, J.; Bluck, L.J.; Runswick, S.; Atkinson, C.; Coward, W.A.; Bingham, S.A. Daidzein and genistein contents of vegetables. Br. J. Nutr. 2000, 84, 717–725. [Google Scholar] [PubMed]
- Mueller, S.O.; Simon, S.; Chae, K.; Metzler, M.; Korach, K.S. Phytoestrogens and their human metabolites show distinct agonistic and antagonistic properties on estrogen receptor α (ERα) and ERβ in human cells. Toxicol. Sci. 2004, 80, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Kinjo, J.; Tsuchihashi, R.; Morito, K.; Hirose, T.; Aomori, T.; Nagao, T.; Okabe, H.; Nohara, T.; Masamune, Y. Interactions of phytoestrogens with estrogen receptors α and β(III). Estrogenic activities of soy isoflavoneaglycones and their metabolites isolated from human urine. Biol. Pharm. Bull. 2004, 27, 185–188. [Google Scholar] [CrossRef] [PubMed]
- Limer, J.L.; Speirs, V. Phyto-oestrogens and breast cancer chemoprevention. Breast Cancer Res. 2004, 6, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Setchell, K.D.; Brown, N.M.; Lydeking-Olsen, E. The clinical importance of the metabolite equol—A clue to the effectiveness of soy and its isoflavones. J. Nutr. 2002, 132, 3577–3584. [Google Scholar] [PubMed]
- Hampl, R.; Ostatnikova, D.; Celec, P.; Putz, Z.; Lapcík, O.; Matucha, P. Short-term effect of soy consumption on thyroid hormone levels and correlation with phytoestrogen level in healthy subjects. Endocr. Regul. 2008, 42, 53–61. [Google Scholar] [PubMed]
- Bianco, A.C.; Larsen, P.R. Cellular and structural biology of the deiodinases. Thyroid 2005, 15, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Meiyanto, E.; Hermawan, A.; Anindyajati. Natural products for cancer-targeted therapy: Citrus flavonoids as potent chemopreventive agents. Asian Pac. J. Cancer Prev. 2012, 13, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Jasim, S.; Ozsari, L.; Habra, M.A. Multikinase inhibitors use in differentiated thyroid carcinoma. Biol. Targ. Ther. 2014, 8, 281–291. [Google Scholar]
- Arons, I.; Freeman, J.; Sokoloff, B.; Eddy, W.H. Bio-flavonoids in radiation injury. II. Contact radiation in experimental cancer. Br. J. Radiol. 1954, 27, 642–644. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.S.; Wang, X. Green tea and cancer prevention. Nutr. Cancer 2010, 62, 931–937. [Google Scholar] [CrossRef] [PubMed]
- Horn-Ross, P.L.; Hoggatt, K.J.; Lee, M.M. Phytoestrogens and thyroid cancer risk: The San Francisco Bay Area thyroid cancer study. Cancer Epidemiol. Biomark. Prev. 2002, 11, 43–49. [Google Scholar]
- Verburg, F.A.; Reiners, C. The association between multinodular goiter and thyroid cancer. Minerva Endocrinol. 2010, 35, 187–192. [Google Scholar] [PubMed]
- Xiao, Q.; Park, Y.; Hollenbeck, A.R.; Kitahara, C.M. Dietary flavonoid intake and thyroid cancer risk in the NIH-AARP diet and health study. Cancer Epidemiol. Biomark. Prev. 2014, 23, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Yin, F.; Giuliano, A.E.; van Herle, A.J. Growth inhibitory effects of flavonoids in human thyroid cancer cell lines. Thyroid 1999, 9, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.J.; Youn, Y.K.; Hong, M.K.; Kim, L.S. Antiproliferation and redifferentiation in thyroid cancer cell lines by polyphenol phytochemicals. J. Korean Med. Sci. 2011, 26, 893–899. [Google Scholar] [CrossRef] [PubMed]
- Phan, T.; Yu, X.M.; Kunnimalaiyaan, M.; Chen, H. Antiproliferative effect of chrysin on anaplastic thyroid cancer. J. Surg. Res. 2011, 170, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Falola, J.; Zhu, X.; Gu, Y.; Kim, L.T.; Sarosi, G.A.; Anthony, T.; Nwariaku, F.E. Antiproliferative effects of Src inhibition on medullary thyroid cancer. J. Clin. Endocrinol. Metab. 2004, 89, 3503–3509. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.C.; Cha, Y.Y. Epigallocatechin-3-gallate induces growth inhibition and apoptosis of human anaplastic thyroid carcinoma cells through suppression of EGFR/ERK pathway and cyclin B1/CDK1 complex. J. Surg. Oncol. 2011, 104, 776–780. [Google Scholar] [CrossRef] [PubMed]
- Ahn, J.C.; Biswas, R.; Chung, P.S. Combination with genistein enhances the efficacy of photodynamic therapy against human anaplastic thyroid cancer cells. Lasers Surg. Med. 2012, 44, 840–849. [Google Scholar] [CrossRef] [PubMed]
- Mazumdar, M.; Adhikary, A.; Chakraborty, S.; Mukherjee, S.; Manna, A.; Saha, S.; Mohanty, S.; Dutta, A.; Bhattacharjee, P.; Ray, P.; et al. Targeting RET to induce medullary thyroid cancer cell apoptosis: An antagonistic interplay between PI3K/Akt and p38MAPK/caspase-8 pathways. Apoptosis 2013, 18, 589–604. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.M.; Phan, T.; Patel, P.N.; Jaskula-Sztul, R.; Chen, H. Chrysin activates Notch1 signaling and suppresses tumor growth of anaplastic thyroid carcinoma in vitro and in vivo. Cancer 2013, 119, 774–781. [Google Scholar] [CrossRef] [PubMed]
- De Amicis, F.; Perri, A.; Vizza, D.; Russo, A.; Panno, M.L.; Bonofiglio, D.; Giordano, C.; Mauro, L.; Aquila, S.; Tramontano, D.; et al. Epigallocatechin gallate inhibits growth and epithelial-to-mesenchymal transition in human thyroid carcinoma cell lines. J. Cell. Physiol. 2013, 228, 2054–2062. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Kang, J.G.; Kim, C.S.; Ihm, S.H.; Choi, M.G.; Yoo, H.J.; Lee, S.J. Apigenin induces c-Myc-mediated apoptosis in FRO anaplastic thyroid carcinoma cells. Mol. Cell. Endocrinol. 2013, 369, 130–139. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.N.; Yu, X.M.; Jaskula-Sztul, R.; Chen, H. Hesperetin activates the Notch1 signaling cascade, causes apoptosis, and induces cellular differentiation in anaplastic thyroid cancer. Ann. Surg. Oncol. 2014, 21 (Suppl. 4), S497–S504. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, C.; Noguchi, Y.; Harii, N.; Napolitano, G.; Tatone, D.; Bucci, I.; Piantelli, M.; Monaco, F.; Kohn, L.D. The flavonoid quercetin regulates growth and gene expression in rat FRTL-5 thyroid cells. Endocrinology 2008, 149, 84–92. [Google Scholar] [CrossRef] [PubMed]
- Giuliani, C.; Bucci, I.; Di Santo, S.; Rossi, C.; Grassadonia, A.; Piantelli, M.; Monaco, F.; Napolitano, G. The flavonoid quercetin inhibits thyroid-restricted genes expression and thyroid function. Food Chem. Toxicol. 2014, 66, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Lakshmanan, A.; Scarberry, D.; Green, J.A.; Zhang, X.; Selmi-Ruby, S.; Jhiang, S.M. Modulation of thyroidal radioiodide uptake by oncological pipeline inhibitors and apigenin. Oncotarget 2015, 6, 31792–31804. [Google Scholar] [PubMed]
- Celano, M.; Maggisano, V.; de Rose, R.F.; Bulotta, S.; Maiuolo, J.; Navarra, M.; Russo, D. Flavonoid fraction of citrus reticulata juice reduces proliferation and migration of anaplastic thyroid carcinoma cells. Nutr. Cancer 2015, 67, 1183–1190. [Google Scholar] [CrossRef] [PubMed]
- Quagliariello, V.; Armenia, E.; Aurilio, C.; Rosso, F.; Clemente, O.; de Sena, G.; Barbarisi, M.; Barbarisi, A. New treatment of medullary and papillary human thyroid cancer: Biological effects of hyaluronic acid hydrogel loaded with quercetin alone or in combination to an inhibitor of aurora kinase. J. Cell. Physiol. 2016, 231, 1784–1795. [Google Scholar] [CrossRef] [PubMed]
| Reference | Experimental Design | Summary of Results |
|---|---|---|
| Schröder-van der Elst et al., 2004 [24]. | Follicular thyroid cancer cell line (FTC-133) was treated with different flavonoids and 125I uptake, 125I efflux and DNA content of the cells were measured. | Most flavonoids inhibited cell growth. Myricetin was the only flavonoid studied that increased the influx and decreased the efflux of 125I. |
| Liu et al., 2004 [103]. | Human medullary carcinoma cell line (TT) was treated with a Src-specific tyrosine kinase inhibitor, PP2, or genistein and cell proliferation was examined. | Compared to control, genistein caused a modest decline in cell count and DNA synthesis, with minimal changes in apoptosis. |
| Phan et al., 2011 [102]. | HTH7 and KAT18 cells, derived from patients with anaplastic thyroid cancer (ATC), were treated with chrysin for up to 6 days. | Chrysin reduced ATC cell numbers by increasing apoptosis in vitro. |
| Kang et al., 2011 [101]. | Authors evaluated the effect of polyphenols (resveratrol, genistein, quercetin, kaempferol and resorcinol) on cell growth and NIS expression in thyroid cancer cell lines (TPC-1—papillary thyroid cancer; FTC-133—follicular thyroid cancer; NPA—poorly differentiated papillary thyroid cancer; FRO—undifferentiated/anaplastic thyroid cancer and ARO—undifferentiated/anaplastic thyroid cancer). | Growth of thyroid cancer cell lines was inhibited in response to genistein, resveratrol and quercetin. NIS mRNA increased in FTC-133 cells in response to genistein and resveratrol but there was no change in NPA, FRO and ARO cells. Quercetin induced NIS in FTC-133, NPA and FRO cells. |
| Lim and Cha, 2011 [104]. | Human ATC cell line, ARO, was treated with epigallocatechin-3-gallate (EGCG). | EGCG inhibited cell proliferation and induced apoptosis via suppression of the EGFR/ERK pathway and cyclin B1/CDK1 complex in ATC cells. |
| Ahn et al., 2012 [105]. | The effect of photodynamic therapy (PDT) and genistein was studied in a human anaplastic thyroid cancer cell line (SNU 80). | The individual treatment with PDT induced apoptosis in SNU 80 cells; however, the efficacy was greatly increased by association with genistein. |
| Mazumdar et al., 2013 [106]. | A human MTC cell line, TT, was incubated with theaflavins, the bioactive components of black tea. | Theaflavins induced apoptosis in human MTC cell line by downregulating both PI3K/Akt/Bad and Ras/Raf/ERK pathways. |
| Yu et al., 2013 [107]. | The effect of chrysin on tumor growth was evaluated using both in vitro model (ATC cell lines: HTh7 and KAT18) and in vivo using subcutaneous xenograft tumor model. | Chrysin inhibited tumor growth in ATC both in vitro and in vivo, which seems to be due to Notch1 signaling activation, leading to cancer cell apoptosis. |
| De Amicis et al., 2013 [108]. | Authors investigated the effect of EGCG, a major catechin in green tea, on the proliferation and motility of human thyroid papillary (FB-2) and follicular (WRO) carcinoma cell lines. | Epigallocatechin-3-gallate reduced proliferation of both thyroid cancer cell lines, besides decreasing cell motility and migration. Those effects seem to be mediated by loss of epithelial-to-mesenchymal transition markers. |
| Kim et al., 2013 [109]. | Authors studied the effect of apigenin on anaplastic thyroid carcinoma cell line (FRO) survival and c-Myc expression. | Apigenin induced apoptosis via c-Myc increment, along with increased phosphorylation of p53 and p38 in FRO cells. |
| Patel et al., 2014 [110]. | The effect of hesperetin on an ATC cell line (HTh7) proliferation and differentiation was evaluated. | Hesperetin reduced ATC cell proliferation and induced the expression of thyroid markers, including sodium-iodide symporter. |
| Zhang et al., 2015 [23]. | A papillary thyroid cancer cell line (BCPAP) was treated with apigenin and the effect on cell viability and the underlying mechanisms were studied. | Apigenin treatment reduced cell viability by inducing ROS generation, leading to DNA damage and a subsequent cell cycle arrest in G2/M phase. Autophagy was induced and eventually triggered human papillary thyroid cancer cell death. |
| Mutlu Altundag et al., 2016 [22]. | Human papillary thyroid cancer cells (BCPAP) were treated with quercetin and cell viability and apoptosis were evaluated. | Quercetin induced reduction of BCPAP cell viability by enhancing apoptosis. This effect seems to involve downregulation of HSP90, a heat shock protein important for stress response in cancer cells. |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gonçalves, C.F.L.; De Freitas, M.L.; Ferreira, A.C.F. Flavonoids, Thyroid Iodide Uptake and Thyroid Cancer—A Review. Int. J. Mol. Sci. 2017, 18, 1247. https://doi.org/10.3390/ijms18061247
Gonçalves CFL, De Freitas ML, Ferreira ACF. Flavonoids, Thyroid Iodide Uptake and Thyroid Cancer—A Review. International Journal of Molecular Sciences. 2017; 18(6):1247. https://doi.org/10.3390/ijms18061247
Chicago/Turabian StyleGonçalves, Carlos F. L., Mariana L. De Freitas, and Andrea C. F. Ferreira. 2017. "Flavonoids, Thyroid Iodide Uptake and Thyroid Cancer—A Review" International Journal of Molecular Sciences 18, no. 6: 1247. https://doi.org/10.3390/ijms18061247
APA StyleGonçalves, C. F. L., De Freitas, M. L., & Ferreira, A. C. F. (2017). Flavonoids, Thyroid Iodide Uptake and Thyroid Cancer—A Review. International Journal of Molecular Sciences, 18(6), 1247. https://doi.org/10.3390/ijms18061247





