Thymoquinone Defeats Diabetes-Induced Testicular Damage in Rats Targeting Antioxidant, Inflammatory and Aromatase Expression
Abstract
:1. Introduction
2. Results
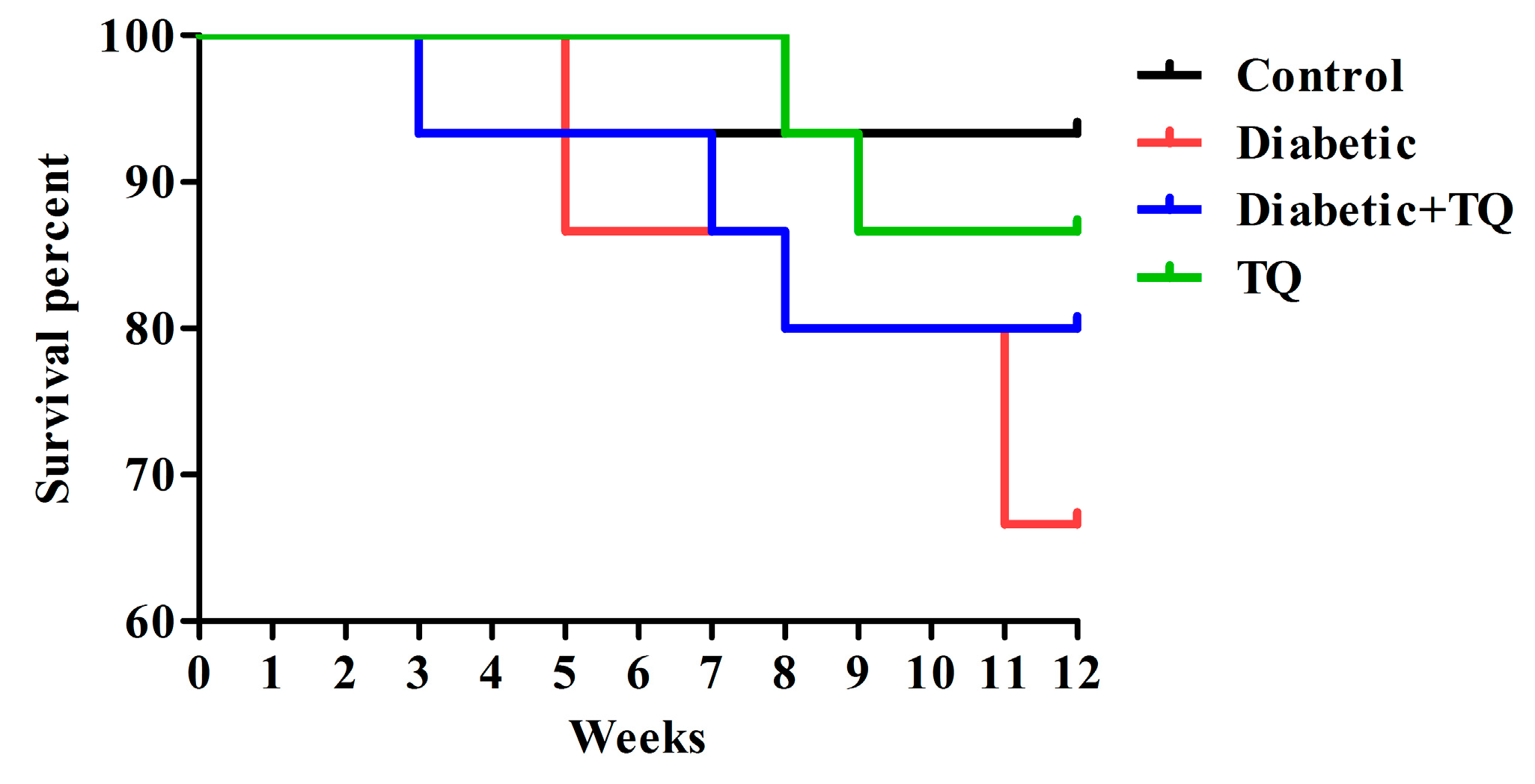
2.1. Survival Percentages
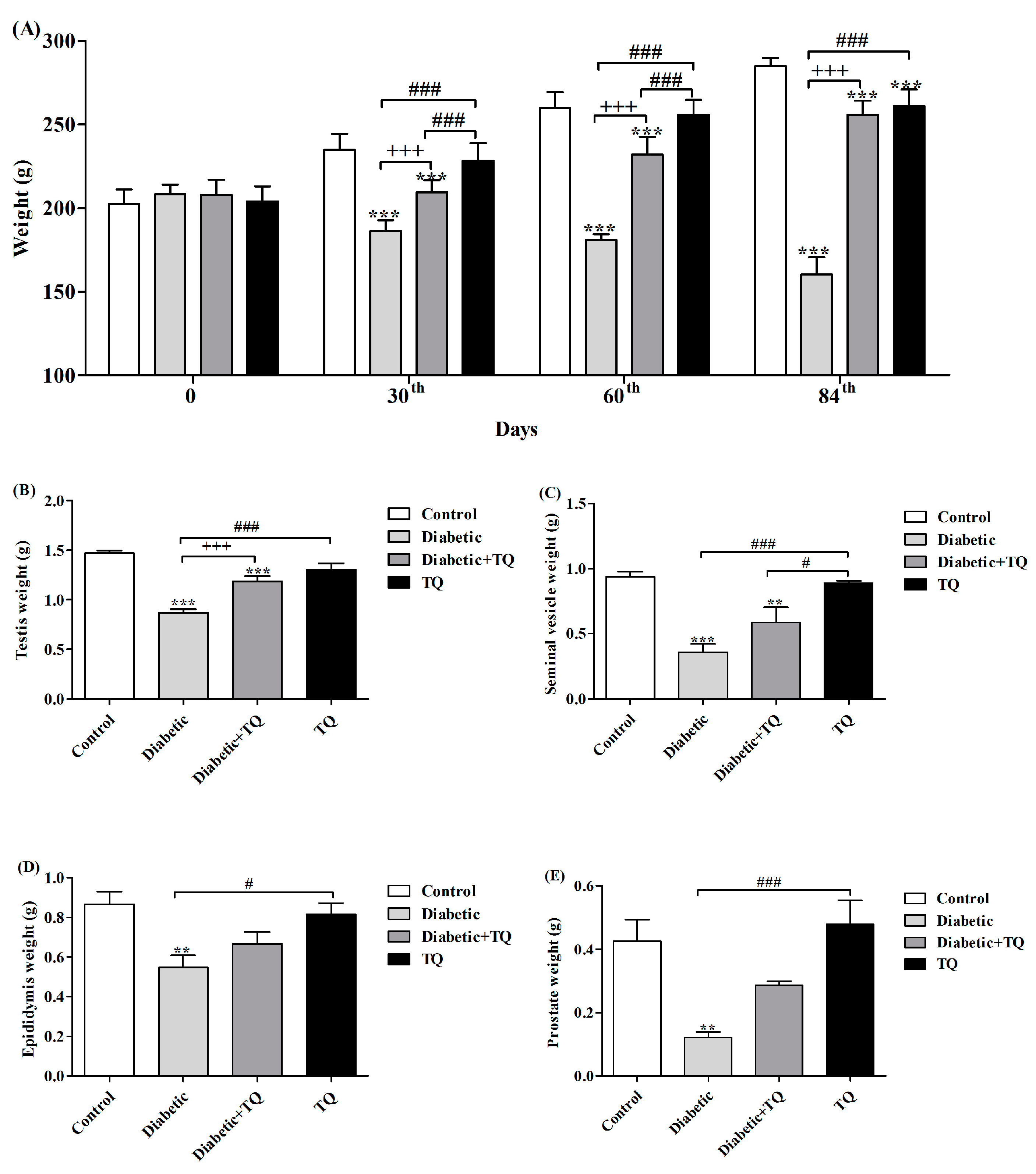
2.2. Body Weight and Accessory Glands Weight
2.3. Serum Glucose, Glycated Hemoglobin, and Insulin
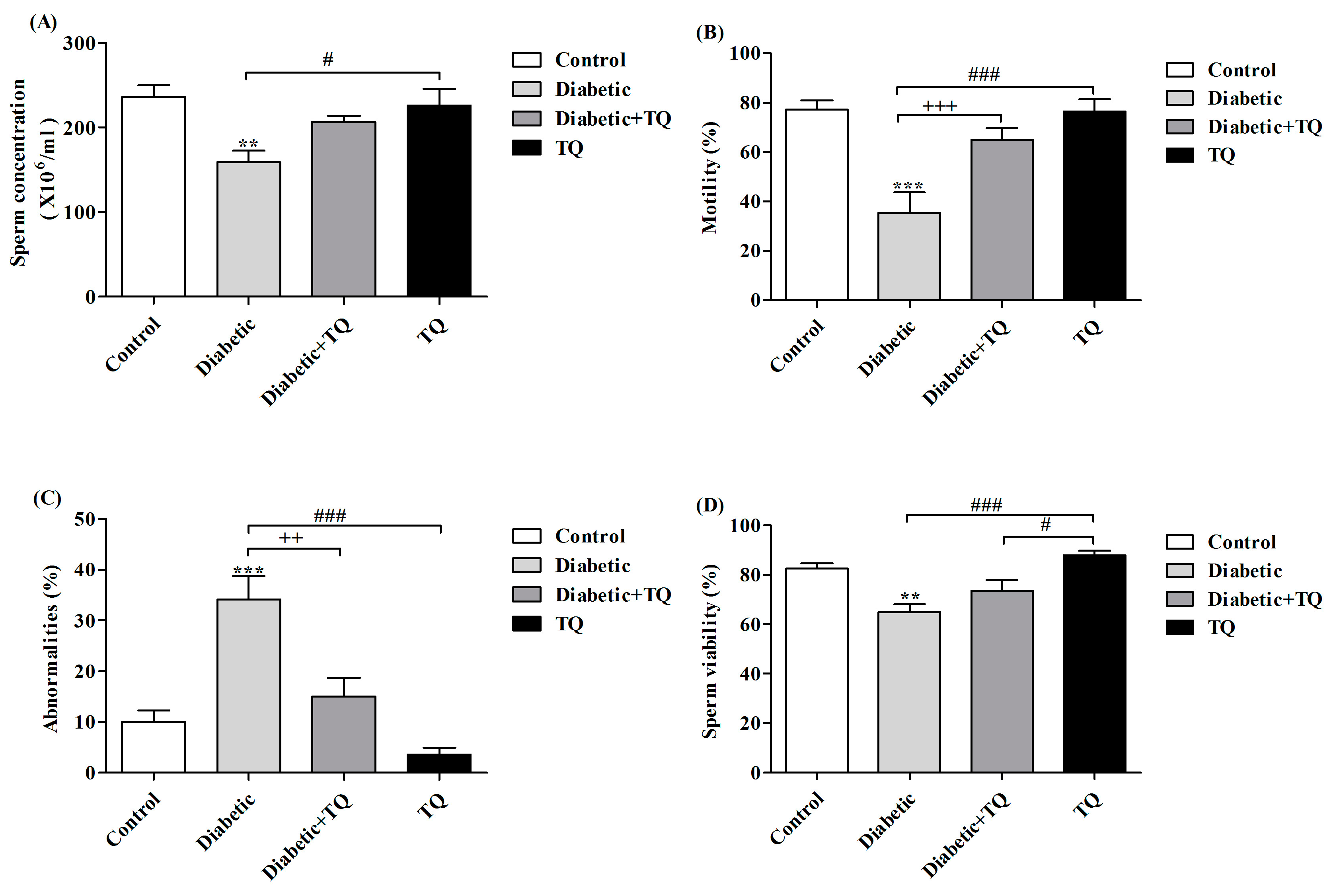
2.4. Sperm Parameters
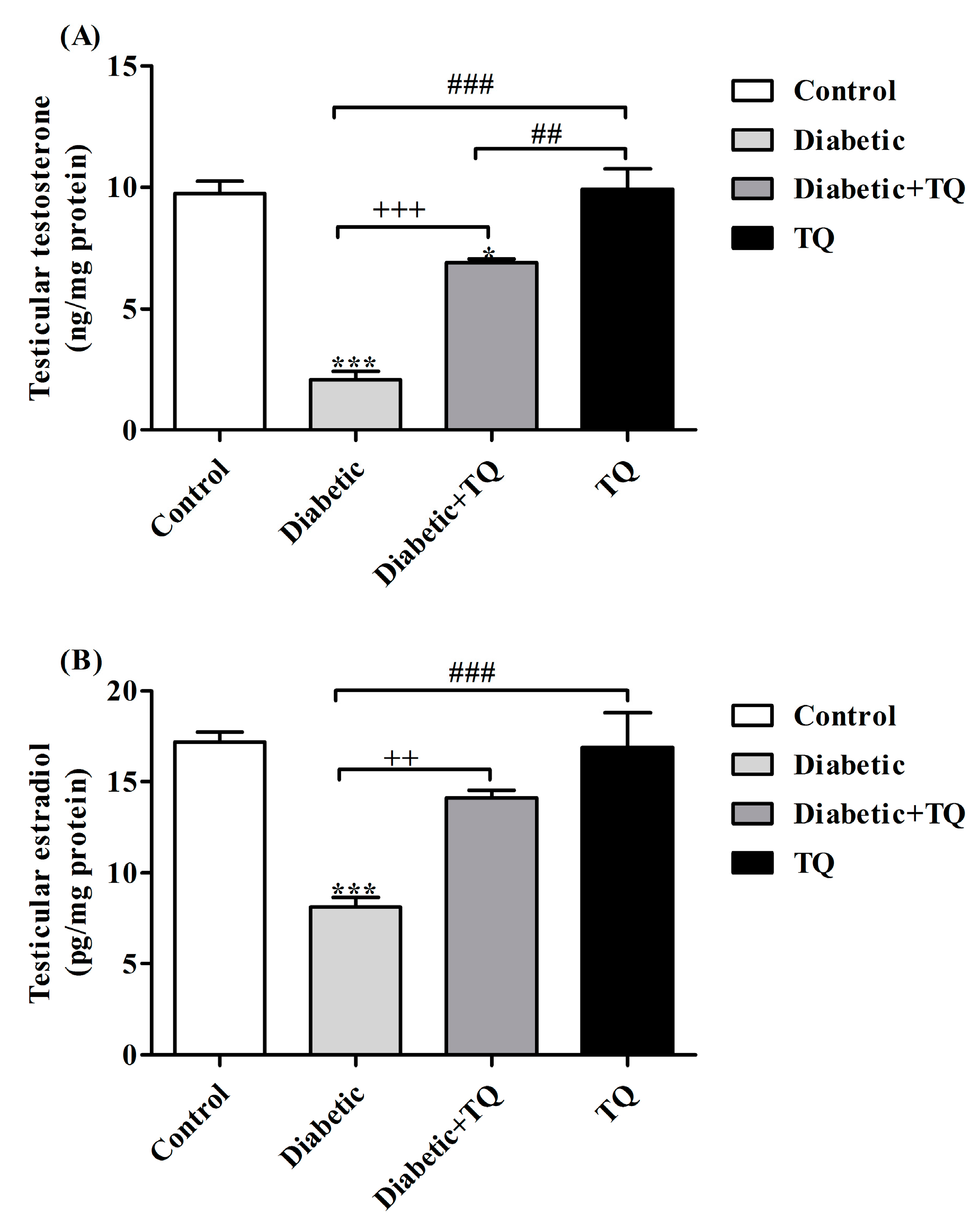
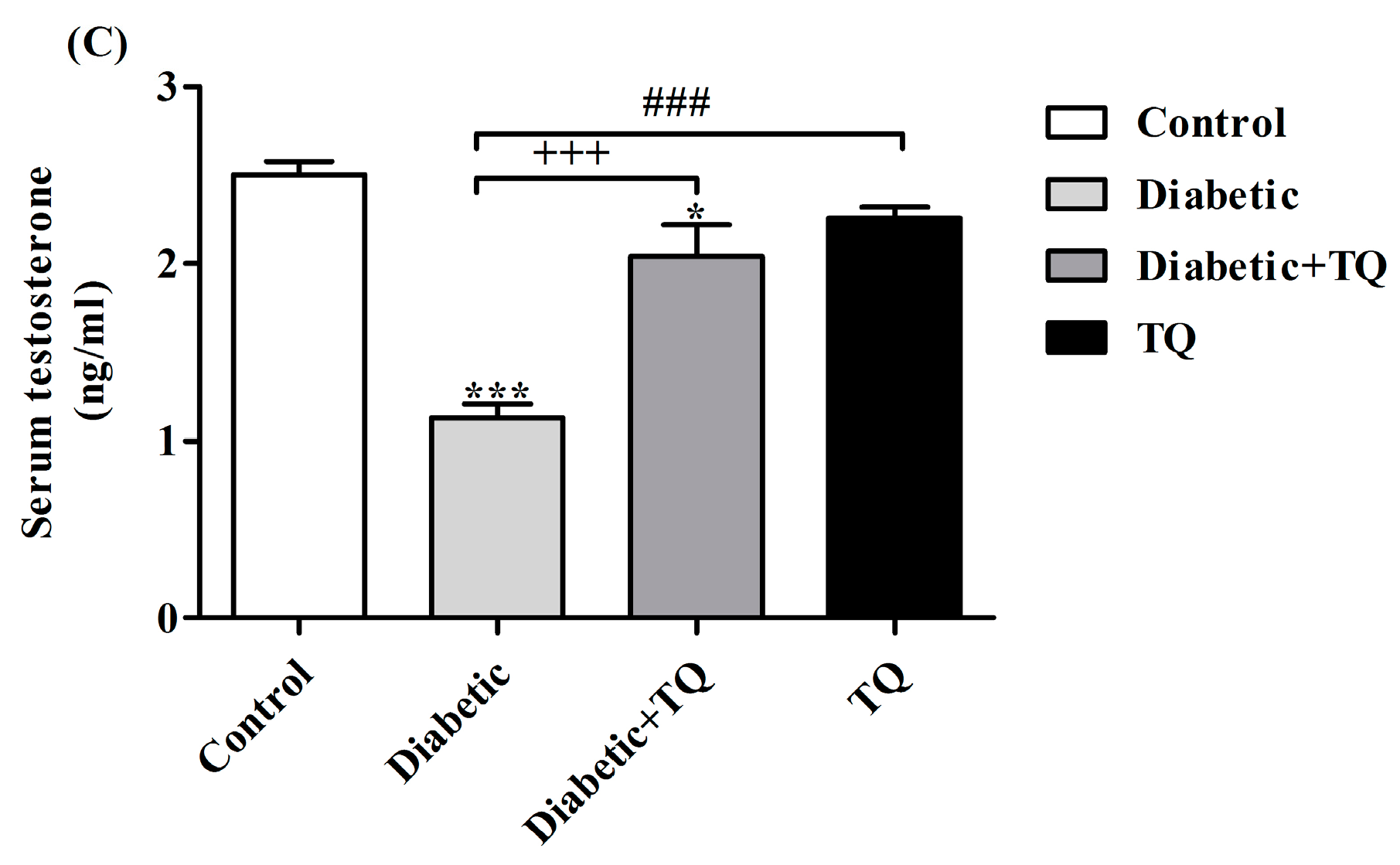
2.5. Testosterone and Estradiol
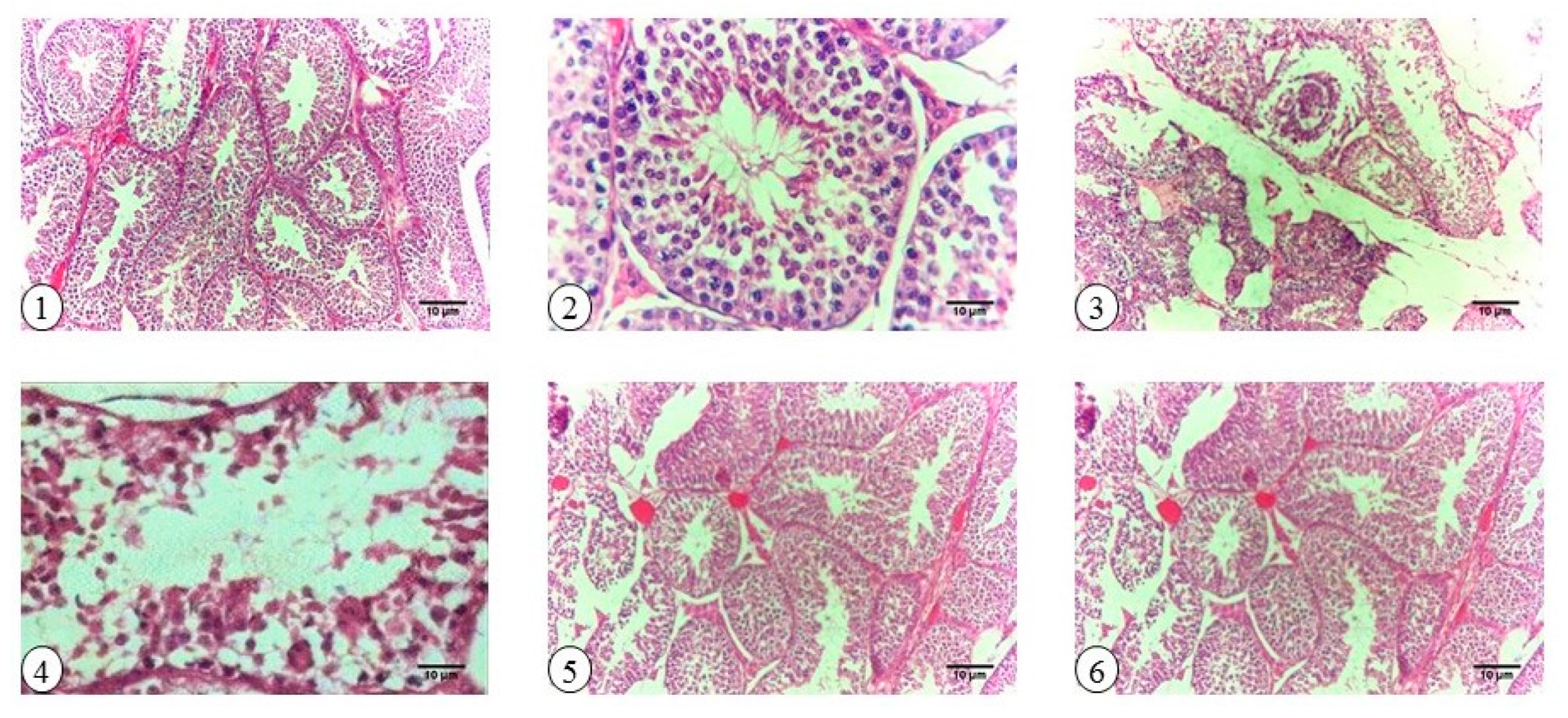
2.6. Testicular Histopathology and Spermatogenesis
2.7. Testicular Oxidative Stress and Antioxidant Status
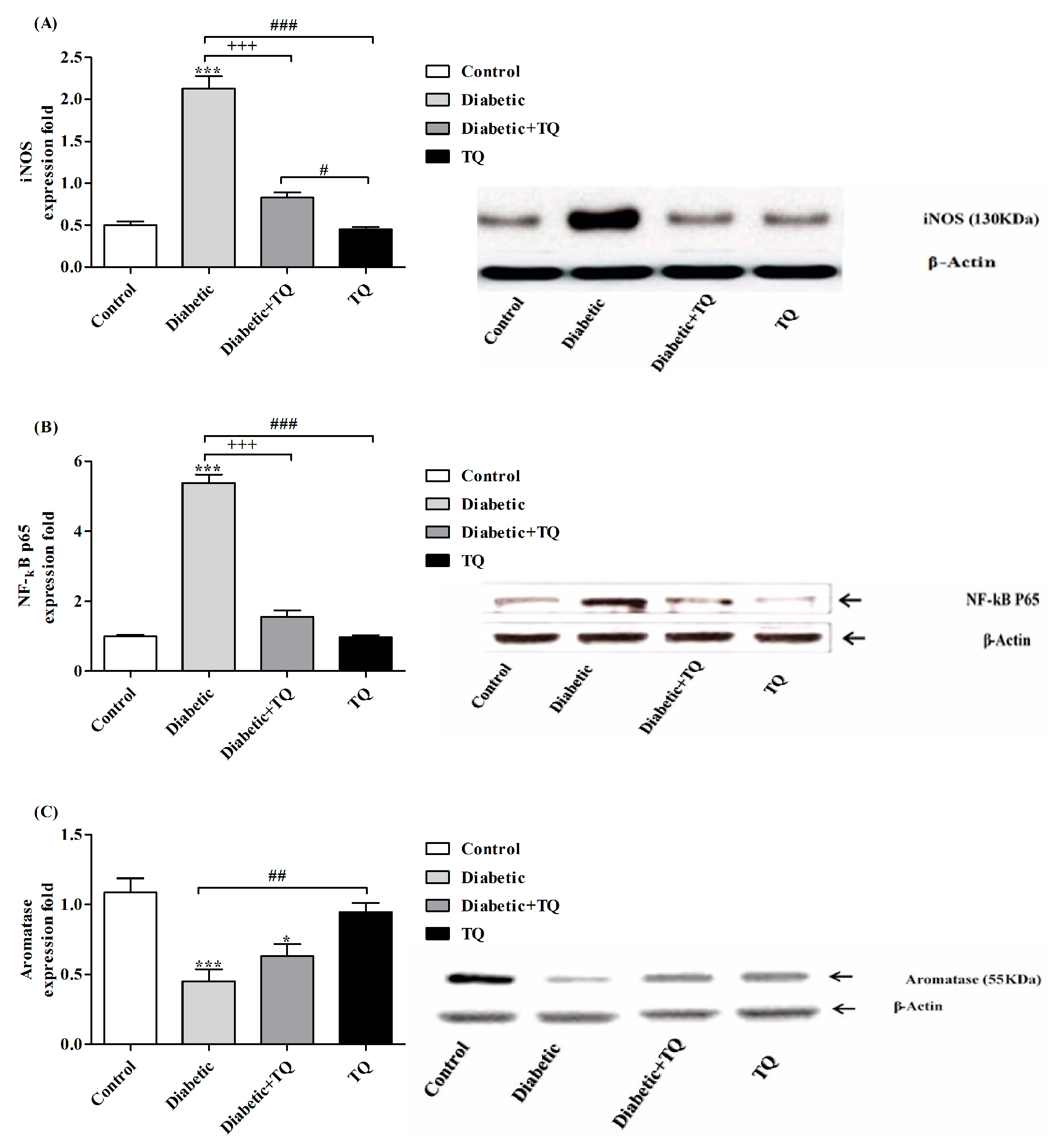
2.8. INOS, NF-κB-p65, and Aromatase Expressions in Testicular Homogenate
2.9. Histopathology
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Animals
4.3. Induction of Diabetes
4.4. Preparation of Thymoquinone
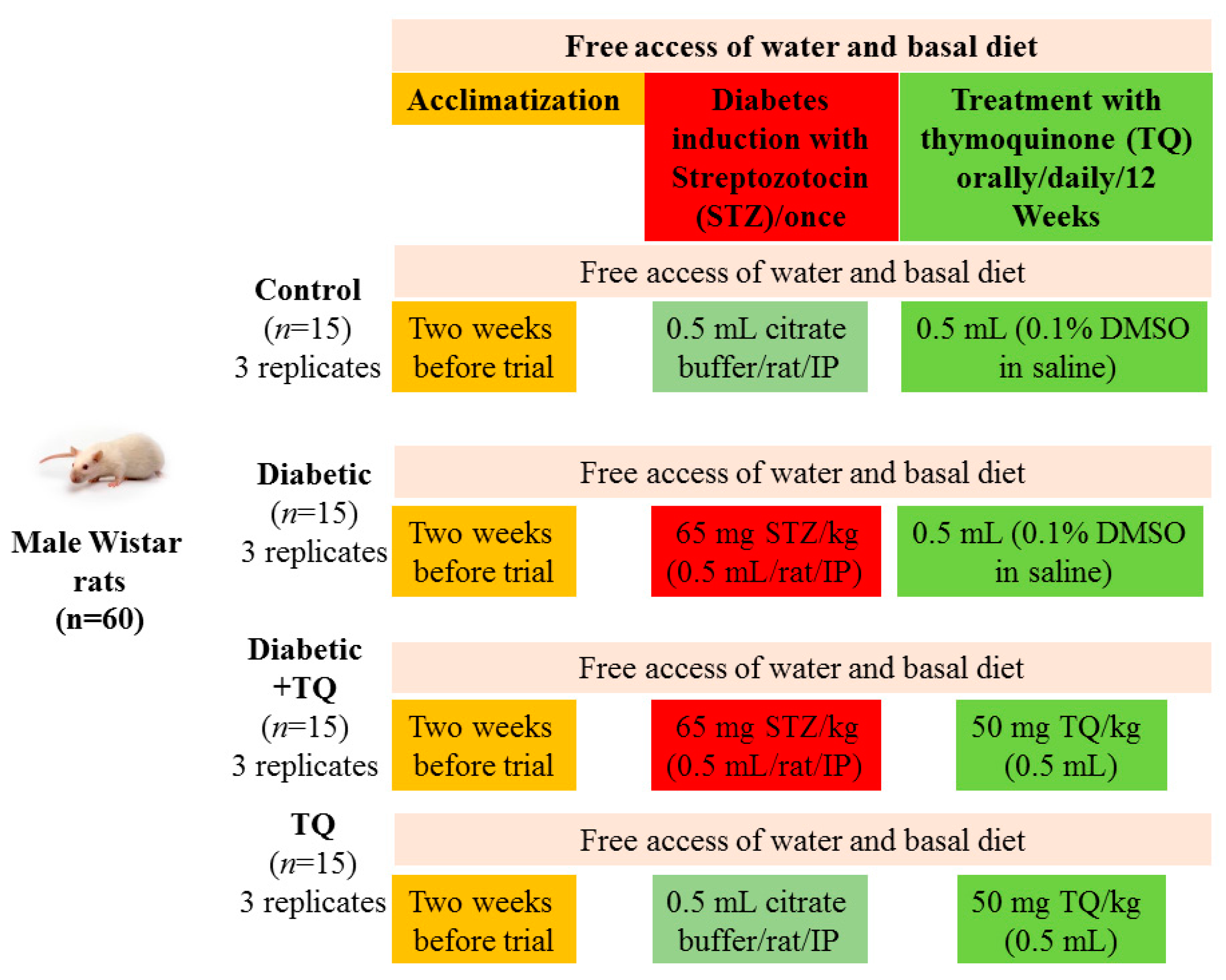
4.5. Experimental Design
4.6. Sampling
4.7. Semen Evaluation
4.8. Assessment of Spermatogenesis
4.9. Biochemical Assay
4.10. Analysis of Antioxidant Status in Testicular Tissues
4.11. Western Blotting
4.12. Histopathology
4.13. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| TQ | thymoquinone |
| MDA | malondialdehyde |
| ROS | reactive oxygen species |
| STZ | streptozotocin |
| ACP | acid phosphatase |
| ALP | alkaline phosphatase |
References
- Gavin, J.R.; James, R.; Alberti, K.G.M.M.; Davidson, M.B.; DeFronzo, R.A. Report of the expert committee on the diagnosis and classification of diabetes mellitus. Diabetes Care 1997, 20, 1183. [Google Scholar]
- Mosher, W.D.; Pratt, W.F. Fecundity and infertility in the United States: Incidence and trends. Fertil. Steril. 1991, 56, 192–193. [Google Scholar] [CrossRef]
- Kianifard, D.; Sadrkhanlou, R.-A.; Hasanzadeh, S. The histological, histomorphometrical and histochemical changes of testicular tissue in the metformin treated and untreated streptozotocin-induced adult diabetic rats. Vet. Res. Forum 2011, 2, 13–24. [Google Scholar]
- Vernet, P.; Aitken, R.J.; Drevet, J.R. Antioxidant strategies in the epididymis. Mol. Cell. Endocrinol. 2004, 216, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Amaral, S.; Oliveira, P.J.; Ramalho-Santos, J. Diabetes and the impairment of reproductive function: Possible role of mitochondria and reactive oxygen species. Curr. Diabetes Rev. 2008, 4, 46–54. [Google Scholar] [PubMed]
- Saalu, L.C. The incriminating role of reactive oxygen species in idiopathic male infertility: An evidence based evaluation. Pak. J. Biol. Sci. 2010, 13, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Abdelmeguid, N.E.; Fakhoury, R.; Kamal, S.M.; Al Wafai, R.J. Effects of Nigella sativa and thymoquinone on biochemical and subcellular changes in pancreatic beta-cells of streptozotocin-induced diabetic rats. J. Diabetes 2010, 2, 256–266. [Google Scholar] [CrossRef] [PubMed]
- El-Far, A.H. Thymoquinone anticancer discovery: Possible mechanisms. Curr. Drug Discov. Technol. 2015, 12, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Al-Zahrani, S.; Mohany, M.; Saleh, K.; Badr, G. Thymoquinone and vitamin E supplementation improve the reproductive characteristics of heat stressed male mice. J. Med. Plants Res. 2012, 6, 493–499. [Google Scholar] [CrossRef]
- Kanter, M. Thymoquinone reestablishes spermatogenesis after testicular injury caused by chronic toluene exposure in rats. Toxicol. Ind. Health 2011, 27, 155–166. [Google Scholar] [CrossRef] [PubMed]
- Gökçe, A.; Oktar, S.; Koc, A.; Yonden, Z. Protective effects of thymoquinone against methotrexate-induced testicular injury. Hum. Exp. Toxicol. 2010, 30, 897–903. [Google Scholar] [CrossRef] [PubMed]
- El-Sheikh, A.A.; Morsy, M.A.; Hamouda, A.H. Protective mechanisms of thymoquinone on methotrexate-induced intestinal toxicity in rats. Pharmacogn. Mag. 2016, 12 (Suppl. S1), 76–81. [Google Scholar]
- Fouad, A.A.; Jresat, I. Thymoquinone therapy abrogates toxic effect of cadmium on rat testes. Andrologia 2015, 47, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Ozdemir, O.; Akalin, P.P.; Baspinar, N.; Hatipoglu, F. Pathological changes in the acute phase of streptozotocin-induced diabetic rats. Bull. Vet. Inst. Pulawy 2009, 53, 783–790. [Google Scholar]
- Nair, K.S.; Ford, G.C.; Ekberg, K.; Fernqvist-Forbes, E.; Wahren, J. Protein dynamics in whole body and in splanchnic and leg tissues in type I diabetic patients. J. Clin. Investig. 1995, 95, 2926–2937. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, L.; McLachlan, R.; Wreford, N.; Robertson, D. Testosterone promotes the conversion of round spermatids between stages VII and VIII of the rat spermatogenic cycle. Endocrinology 1994, 135, 2608–2614. [Google Scholar] [PubMed]
- Soudamani, S.; Yuvaraj, S.; Malini, T.; Balasubramanian, K. Experimental diabetes has adverse effects on the differentiation of ventral prostate during sexual maturation of rats. Anat. Rec. A Discov. Mol. Cell. Evol. Biol. 2005, 287, 1281–1289. [Google Scholar] [CrossRef] [PubMed]
- Ballester, J.; Munoz, M.C.; Dominguez, J.; Rigau, T.; Guinovart, J.J.; Rodriguez-Gil, J.E. Insulin-dependent diabetes affects testicular function by FSH- and LH-linked mechanisms. J. Androl. 2004, 25, 706–719. [Google Scholar] [CrossRef] [PubMed]
- Bruening, J. Role of brain insulin receptor and control of body weight and reproduction. Science 2000, 289, 2122–2125. [Google Scholar] [CrossRef]
- Meral, I.; Yener, Z.; Kahraman, T.; Mert, N. Effect of Nigella sativa on glucose concentration, lipid peroxidation, anti-oxidant defence system and liver damage in experimentally-induced diabetic rabbits. J. Vet. Med. A Physiol. Pathol. Clin. Med. 2001, 48, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Kanter, M. Protective effects of thymoquinone on streptozotocin-induced diabetic nephropathy. J. Mol. Histol. 2009, 40, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Scarano, W.; Messias, A.; Oliva, S.; Klinefelter, G.; Kempinas, W. Sexual behaviour, sperm quantity and quality after short-term streptozotocin-induced hyperglycaemia in rats. Int. J. Androl. 2006, 29, 482–488. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, R.M.; Kerr, J.B.; McKinnell, C.; Millar, M. Temporal relationship between androgen-dependent changes in the volume of seminiferous tubule fluid, lumen size and seminiferous tubule protein secretion in rats. J. Reprod. Fertil. 1994, 101, 193–198. [Google Scholar] [CrossRef] [PubMed]
- Pentikainen, V.; Erkkila, K.; Suomalainen, L.; Parvinen, M.; Dunkel, L. Estradiol acts as a germ cell survival factor in the human testis in vitro. J. Clin. Endocrinol. Metab. 2000, 85, 2057–2067. [Google Scholar] [CrossRef] [PubMed]
- Farrell, J.B.; Deshmukh, A.; Baghaie, A.A. Low testosterone and the association with type 2 diabetes. Diabetes Educ. 2008, 34, 799–806. [Google Scholar] [CrossRef] [PubMed]
- Bauche, F.; Fouchard, M.H.; Jegou, B. Antioxidant system in rat testicular cells. FEBS Lett. 1994, 349, 392–396. [Google Scholar] [CrossRef]
- Ghosh, J.; Das, J.; Manna, P.; Sil, P.C. Taurine prevents arsenic-induced cardiac oxidative stress and apoptotic damage: Role of NF-κB, p38 and JNK MAPK pathway. Toxicol. Appl. Pharmacol. 2009, 240, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Mabrouk, A.; Ben Cheikh, H. Thymoquinone supplementation ameliorates lead-induced testis function impairment in adult rats. Toxicol. Ind. Health 2016, 32, 1114–1121. [Google Scholar] [CrossRef] [PubMed]
- Al Wafai, R.J. Nigella sativa and thymoquinone suppress cyclooxygenase-2 and oxidative stress in pancreatic tissue of streptozotocin-induced diabetic rats. Pancreas 2013, 42, 841–849. [Google Scholar] [CrossRef] [PubMed]
- Geng, X.; Shao, H.; Zhang, Z.; Ng, J.C.; Peng, C. Malathion-induced testicular toxicity is associated with spermatogenic apoptosis and alterations in testicular enzymes and hormone levels in male wistar rats. Environ. Toxicol. Pharmacol. 2015, 39, 659–667. [Google Scholar] [CrossRef] [PubMed]
- Kanimozhi, V.; Palanivel, K.; Akbarsha, M.A.; Kadalmani, B. Tributyltin-mediated hepatic, renal and testicular tissue damage in male syrian hamster (Mesocricetus auratus): A study on impact of oxidative stress. Springerplus 2016, 5, 1523. [Google Scholar] [CrossRef] [PubMed]
- Kushwaha, S.; Jena, G.B. Telmisartan ameliorates germ cell toxicity in the STZ-induced diabetic rat: Studies on possible molecular mechanisms. Mutat. Res. 2013, 755, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.K.; Lal, B. Pro-steroidogenic and pro-spermatogenic actions of nitric oxide (NO) on the catfish, clarias batrachus: An in vivo study. Gen. Comp. Endocrinol. 2017, 242, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.; Santani, D. Role of NF-κB in the pathogenesis of diabetes and its associated complications. Pharmacol. Rep. 2009, 61, 595–603. [Google Scholar] [CrossRef]
- Al-Malki, A.L.; Sayed, A.A.R. Thymoquinone attenuates cisplatin-induced hepatotoxicity via nuclear factor kappa-β. BMC Complement. Altern. Med. 2014, 14, 1. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Castelan, J.; Mendez-Tepepa, M.; Carrillo-Portillo, Y.; Anaya-Hernández, A.; Rodríguez-Antolín, J.; Zambrano, E.; Castelán, F.; Cuevas-Romero, E. Hypothyroidism reduces the size of ovarian follicles and promotes hypertrophy of periovarian fat with infiltration of macrophages in adult rabbits. BioMed Res. Int. 2017, 2017, 3795950. [Google Scholar] [CrossRef] [PubMed]
- Grumbach, M.M.; Auchus, R.J. Estrogen: Consequences and implications of human mutations in synthesis and action. J. Clin. Endocrinol. Metab. 1999, 84, 4677–4694. [Google Scholar] [CrossRef] [PubMed]
- Burul-Bozkurt, N.; Pekiner, C.; Kelicen, P. Diabetes alters aromatase enzyme levels in gonadal tissues of rats. Naunyn Schmiedebergs Arch. Pharmacol. 2010, 382, 33–41. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, L.; Robertson, K.M.; Jones, M.E.; Simpson, E.R. Estrogen and spermatogenesis. Endocr. Rev. 2001, 22, 289–318. [Google Scholar] [CrossRef] [PubMed]
- Carreau, S.; de Vienne, C.; Galeraud-Denis, I. Aromatase and estrogens in man reproduction: A review and latest advances. Adv. Med. Sci. 2008, 53, 139–144. [Google Scholar] [CrossRef] [PubMed]
- Budin, S.B.; Yusof, K.M.; Idris, M.H.M.; Hamid, Z.A.; Mohamed, J. Tocotrienol-rich fraction of palm oil reduced pancreatic damage and oxidative stress in streptozotocin-induced diabetic rats. Aust. J. Basic Appl. Sci. 2011, 5, 2367–2374. [Google Scholar]
- Kaplanoglu, G.T.; Bahcelioglu, M.; Gozil, R.; Helvacioglu, F.; Buru, E.; Tekindal, M.A.; Erdogan, D.; Calguner, E. Effects of green tea and vitamin E in the testicular tissue of streptozotocin-induced diabetic rats. Saudi Med. J. 2013, 34, 734–743. [Google Scholar] [PubMed]
- Al-Zahrani, S.; Kandeal, S.; Mohany, M.; Badr, G. Effects of vitamin E and thymoquinone on physiological and histological characteristics of heat-stressed male mice. Afr. J. Pharm. Pharmacol. 2011, 5, 2174–2183. [Google Scholar] [CrossRef]
- Kanter, M. Effects of Nigella sativa and its major constituent, thymoquinone on sciatic nerves in experimental diabetic neuropathy. Neurochem. Res. 2008, 33, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Sönmez, M.; Türk, G.; Yüce, A. The effect of ascorbic acid supplementation on sperm quality, lipid peroxidation and testosterone levels of male wistar rats. Theriogenology 2005, 63, 2063–2072. [Google Scholar] [CrossRef] [PubMed]
- Amann, R. Use of animal models for detecting specific alterations in reproduction. Fundam. Appl. Toxicol. 1982, 2, 13–26. [Google Scholar] [CrossRef]
- Johnsen, S. Testicular biopsy score count—A method for registration of spermatogenesis in human testes: Normal values and results in 335 hypogonadal males. Horm. Res. Paediatr. 1970, 1, 2–25. [Google Scholar] [CrossRef]
- Trinder, P. Determination of blood glucose using an oxidase-peroxidase system with a non-carcinogenic chromogen. J. Clin. Pathol. 1969, 22, 158–161. [Google Scholar] [CrossRef] [PubMed]
- Little, R.; Wiedmeyer, H.; England, J.; Wilke, A.; Rohlfing, C.; Wians, F.; Jacobson, J.; Zellmer, V.; Goldstein, D. Interlaboratory standardization of measurements of glycohemoglobins. Clin. Chem. 1992, 38, 2472–2478. [Google Scholar] [PubMed]
- Hillmann, G.Z. Prostatic and total acid phosphatase estimation. Klin. Chem. Klin. Biochem. 1971, 9, 273. [Google Scholar]
- Belfield, A.; Goldberg, D. Colorimetric determination of alkaline phosphatase activity. Enzyme 1971, 12, 561–566. [Google Scholar] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Beutler, E.; Duron, O.; Kelly, B.M. Improved method for the determination of blood glutathione. J. Lab. Clin. Med. 1963, 61, 882–888. [Google Scholar] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Nishikimi, M.; Rao, N.A.; Yagi, K. The occurrence of superoxide anion in the reaction of reduced phenazine methosulfate and molecular oxygen. Biochem. Biophys. Res. Commun. 1972, 46, 849–854. [Google Scholar] [CrossRef]
- Montgomery, H.; Dymock, J. The determination of nitrite in water. Analyst 1961, 86, 414–416. [Google Scholar]
- Bancroft, J.D.; Layton, C. The Hematoxylin and Eosin. In Theory Practice of Histological Techniques, 7th ed.; Suvarna, S.K., layton, C., bancroft, J.D., Eds.; Churchill Livingstone of El Sevier: Philadelphia, PA, USA, 2013. [Google Scholar]
| Experimental Group | Glucose (mg/dL) | HA1c (%) | Insulin (ng/mL) |
|---|---|---|---|
| Control | 101.4 ± 2.24 c | 5.887 ± 0.81 c | 2.167 ± 0.21 a |
| Diabetic | 379.4 ± 5.04 a | 9.767 ± 0.46 a | 0.8600 ± 0.08 c |
| Diabetic + TQ | 167.6 ± 3.34 b | 7.100 ± 0.61 b | 1.733 ± 0.12 b |
| TQ | 124.8 ± 2.88 bc | 5.700 ± 0.30 c | 2.000 ± 0.05 b |
| Group | Testicular Enzymes Markers | Enzymatic Status of Testicular Antioxidant | Johnsen Score | ||||
|---|---|---|---|---|---|---|---|
| ACP (U/mg Protein) | ALP (U/mg Protein) | MDA (nmol/mg Protein) | SOD (U/mg Protein) | GSH (nmol/mg Protein) | NO (nmol/mg Protein) | ||
| Control | 22.5 ± 2.12 a | 869.25 ± 6.21 a | 30.12 ± 4.37 bc | 29.2 ± 3.12 a | 9.12 ± 1.2 a | 49 ± 2.1 c | 9.86 ± 0.1 a |
| Diabetic | 7.8 ± 1.13 b | 562.12 ± 8.41 b | 68.15 ± 6.2 a | 8.9 ± 1.10 b | 3.22 ± 0.1 b | 99 ± 4.1 a | 4.97 ± 0.2 c |
| Diabetic + TQ | 18.2 ± 2.51 a | 701 ± 12.13 ab | 35.2 ± 5.2 b | 25.1 ± 3.21 ab | 7.25 ± 0.9 a | 60 ± 5.1 b | 8.71 ± 0.2 b |
| TQ | 23.5 ± 3.12 a | 889.2 ± 7.15 a | 25.24 ± 4.2 c | 30.2 ± 2.42 a | 9.65 ± 2.1 a | 48 ± 3.1 c | 9.77 ± 0.1 a |
| Ingredients | g/kg Diet |
|---|---|
| Corn flour | 529.5 |
| Casein | 200 |
| Sucrose | 100 |
| Soybean oil | 70 |
| Cellulose | 50 |
| Mineral mix | 35 |
| Vitamin mix | 10 |
| l-cystine | 3 |
| Choline | 2.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atta, M.S.; Almadaly, E.A.; El-Far, A.H.; Saleh, R.M.; Assar, D.H.; Al Jaouni, S.K.; Mousa, S.A. Thymoquinone Defeats Diabetes-Induced Testicular Damage in Rats Targeting Antioxidant, Inflammatory and Aromatase Expression. Int. J. Mol. Sci. 2017, 18, 919. https://doi.org/10.3390/ijms18050919
Atta MS, Almadaly EA, El-Far AH, Saleh RM, Assar DH, Al Jaouni SK, Mousa SA. Thymoquinone Defeats Diabetes-Induced Testicular Damage in Rats Targeting Antioxidant, Inflammatory and Aromatase Expression. International Journal of Molecular Sciences. 2017; 18(5):919. https://doi.org/10.3390/ijms18050919
Chicago/Turabian StyleAtta, Mustafa S., Essam A. Almadaly, Ali H. El-Far, Rasha M. Saleh, Doaa H. Assar, Soad K. Al Jaouni, and Shaker A. Mousa. 2017. "Thymoquinone Defeats Diabetes-Induced Testicular Damage in Rats Targeting Antioxidant, Inflammatory and Aromatase Expression" International Journal of Molecular Sciences 18, no. 5: 919. https://doi.org/10.3390/ijms18050919
APA StyleAtta, M. S., Almadaly, E. A., El-Far, A. H., Saleh, R. M., Assar, D. H., Al Jaouni, S. K., & Mousa, S. A. (2017). Thymoquinone Defeats Diabetes-Induced Testicular Damage in Rats Targeting Antioxidant, Inflammatory and Aromatase Expression. International Journal of Molecular Sciences, 18(5), 919. https://doi.org/10.3390/ijms18050919









