Effects of Melatonin and Its Analogues on Pancreatic Inflammation, Enzyme Secretion, and Tumorigenesis
Abstract
:1. Melatonin, Its Precursor and Derivatives
2. Melatonin and Its Receptors in the Pancreas
3. Role of Melatonin and Its Analogues in Pancreatic Enzyme Secretion
4. Melatonin System and Acute Pancreatitis
5. Melatonin System and Pancreatic Cancer
6. Conclusions
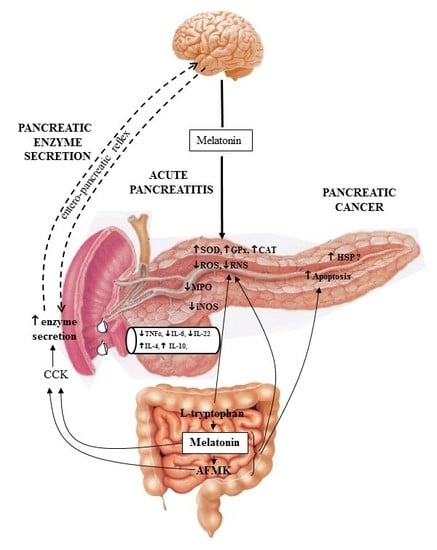
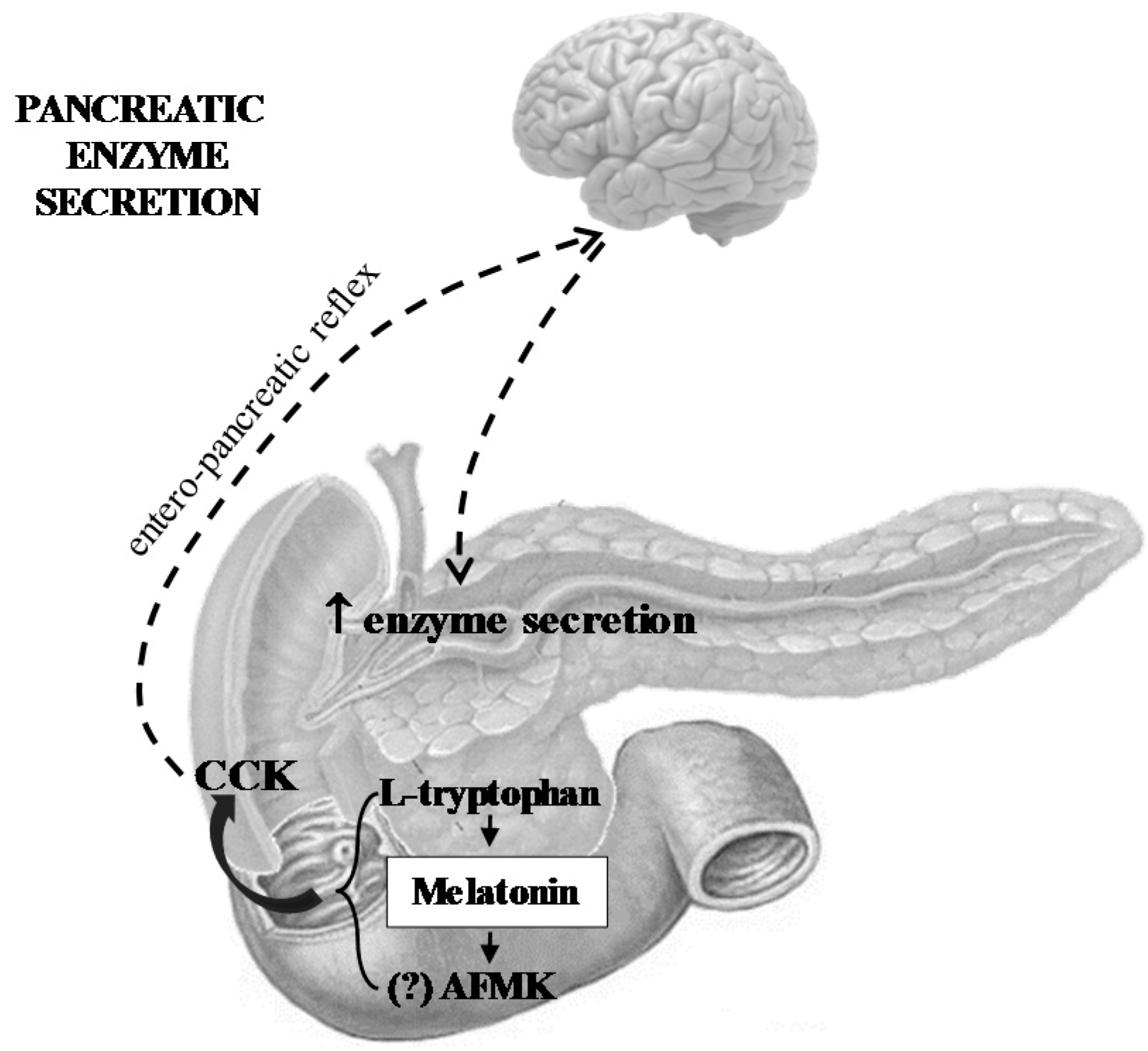
- Melatonin and its related molecules may be involved in the physiological stimulation of pancreatic exocrine secretion dependent on the stimulation of CCK release and activation of duodeno-pancreatic neural reflex (Figure 1).
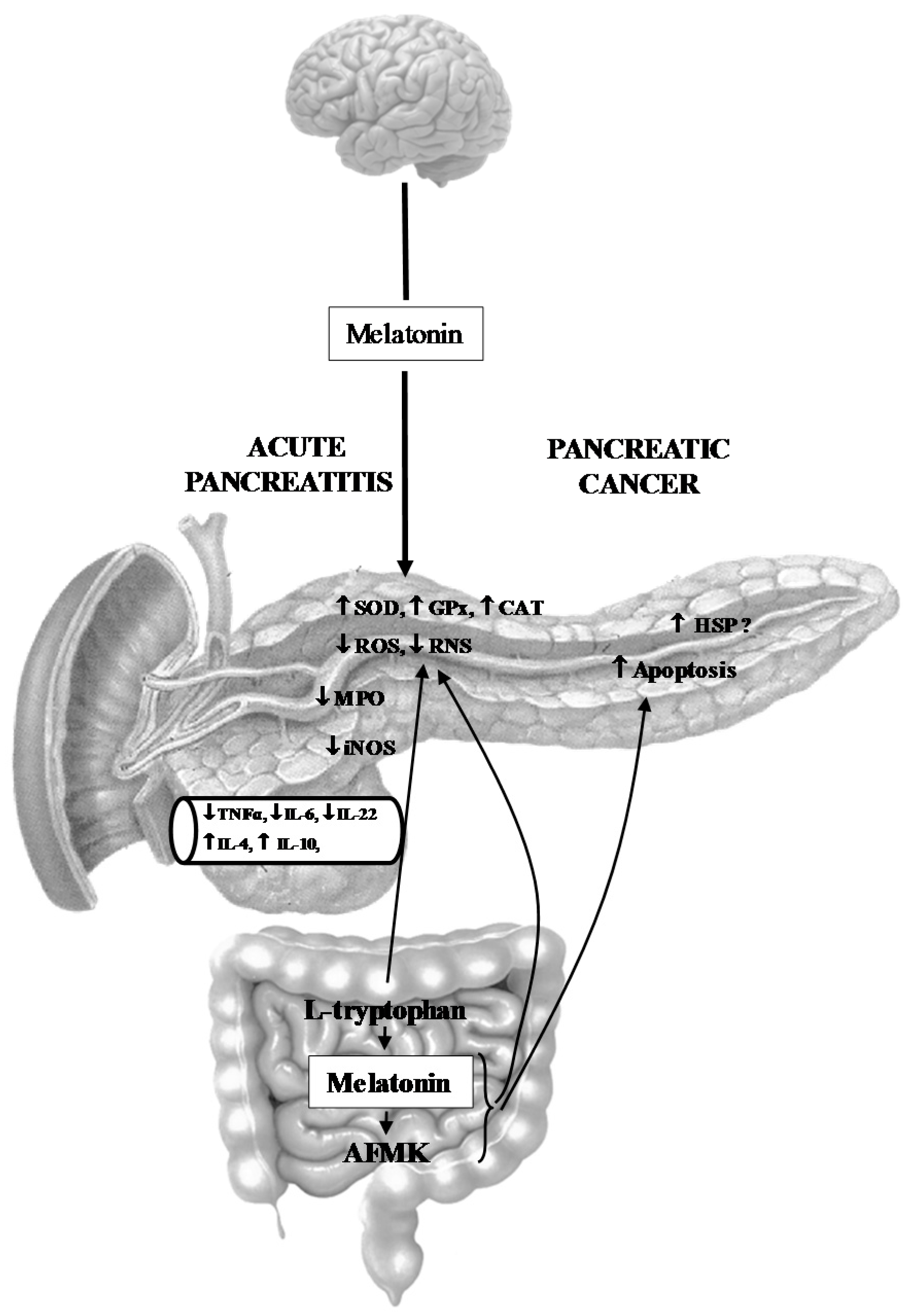
- Melatonin might be implicated in the activation of the innate defense system of pancreatic protection, and its derivatives are responsible for a considerable part of the protective action of melatonin on the pancreas as part of its scavenging cascade (Figure 2).
- Melatonin and its metabolites take part in the control of the increased production of heat shock proteins as well as in the signal transmission pathways modulating the process of apoptosis in pancreatic cancer cells, however its effect is not completely clear and requires further study (Figure 2).
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Reiter, R.J.; Tan, D.X.; Rosales-Corral, S.; Manchester, L.C. The universal nature, unequal distribution and antioxidant functions of melatonin and its derivatives. Mini Rev. Med. Chem. 2013, 13, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Calvo, J.R.; Gonzales-Yanes, C.; Maldonado, M.D. The role of melatonin in the cells of the innate immunity: A review. J. Pineal Res. 2013, 55, 103–120. [Google Scholar] [CrossRef] [PubMed]
- Zawilska, J.B.; Skene, D.J.; Arendt, J. Physiology and pharmacology of melatonin in relation to biological rhythms. Pharmacol. Rep. 2009, 61, 383–410. [Google Scholar] [CrossRef]
- Ren, W.; Liu, G.; Chen, S.; Yin, J.; Wang, J.; Tan, B.; Wu, G.; Bazer, F.W.; Peng, Y.; Li, T.; et al. Melatonin signaling in T cells: Functions and applications. J. Pineal Res. 2017, 62. [Google Scholar] [CrossRef] [PubMed]
- Mańka, S.; Majewska, E. Immunoregulatory action of melatonin. The mechanism of action and the effect on inflammatory cells. Postepy Hig. Med. Dosw. 2016, 70, 1059–1067. [Google Scholar] [CrossRef]
- Chojnacki, C.; Wisniewska-Jarosińska, M.; Walecka-Kapica, E.; Klupińska, G.; Jaworek, J.; Chojnacki, J. Evaluation of melatonin effectiveness in the adjuvant treatment of ulcerative colitis. J. Physiol. Pharmacol. 2011, 62, 327–334. [Google Scholar] [PubMed]
- Reiter, R.J.; Mayo, J.C.; Tan, D.X.; Sainz, R.M.; Alatorre-Jimenez, M.; Oin, L. Melatonin as an antioxidant: Under promises but over delivers. J. Pineal Res. 2016, 61, 253–278. [Google Scholar] [CrossRef] [PubMed]
- Goc, Z.; Szaroma, W.; Kapusta, E.; Dziubek, K. Protective effects of melatonin on the activity of SOD, CAT, GSH-Px and GSH content in organs of mice after administration of SNP. Chin. J. Physiol. 2017, 60, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Miller, E.; Mrowicka, M.; Malinowska, K.; Kędziora, J.; Majsterek, I. The effect of whole body cryotherapy and melatonin supplementation on total antioxidative status and some antioxidative enzyme in multiple sclerosis patients. Pol. Merkur. Lek. 2011, 31, 150–153. [Google Scholar]
- Colares, J.R.; Schemitt, E.G.; Hartmann, R.M.; Licks, F.; Soares, M.D.; Bosco, A.D.; Marroni, N.P. Antioxidant and anti-inflammatory action of melatonin in an experimental model of secondary biliary cirrosis induced by bile duct ligation. World J. Gastroenterol. 2016, 22, 8918–8928. [Google Scholar] [CrossRef] [PubMed]
- Cutando, A.; Montero, J.; Gomez-de Diego, R.; Ferrera, M.J.; Lopez-Valverde, A. Effect of topical melatonin on serum levels of C-rective protein (CRP), interleukin-6 (IL-6), and tumor necrosis factor-α (TNFα) in patients with type 1 or type 2 diabetes and periodontal disease. J. Clin. Exp. Dent. 2015, 7, e628–e633. [Google Scholar] [PubMed]
- Maldonado, M.D.; Garcia-Moreno, H.; Gonzales-Yanes, C.; Calvo, J.R. Possible involvement of the inhibition of NF-κB factor in anti-inflammatory actions that melatonin exerts on mast cells. J. Cell. Biochem. 2016, 117, 1926–1933. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.H.; Hong, S.W.; Zheng, H.M.; Lee, H.S.; Lee, H.; Lee, D.H.; Lee, S.Y.; Hong, S.S. Melatonin ameliorates caerulein-induced pancreatitis by the modulation of nuclear erythroid 2-related factor 2 and nuclear factor-kappa B in rats. J. Pineal Res. 2010, 48, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.J.; Huangh, S.H.; Chen, J.W.; Wang, K.C.; Yang, Y.R.; Liu, P.F.; Lin, G.J.; Sytwu, H.K. Melatonin enhances interleukin-10 expression and suppresses chemotaxis to inhibit inflammation in situ and reduce the severity of experimental autoimmune encephalomyelitis. Int. Immunopharmacol. 2016, 32, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Huai, J.P.; Sun, X.C.; Chen, M.J.; Jin, Y.; Ye, X.H.; Wu, J.S.; Huang, Z.M. Melatonin attenuates acute pancreatitis-associated lung injury inn rats by modulating interleukin 22. World J. Gastroenterol. 2012, 18, 5122–5128. [Google Scholar] [CrossRef] [PubMed]
- Ochoa, J.J.; Diaz-Castro, J.; Kajarabille, N.; Garcia, N.; Guisado, I.M.; De Teresa, C.; Guisado, R. Melatonin supplementation ameliorates oxidative stress and inflammatory signaling induced by strenuous exercise in adult human males. J. Pineal Res. 2011, 51, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Santofimia-Castano, P.; Clea Ruy, D.; Garcia-Sanchez, L.; Jimenez-Blasco, D.; Fernandez-Bermejo, M.; Bolanos, J.P.; Salido, G.M.; Gonzales, A. Melatonin induces the expression of Nrf2-regulated antioxidant enzymes via PKC and Ca2+ influx activation in mouse pancreatic acinar cells. Free Radic. Biol. Med. 2015, 87, 226–236. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Q.; Fichna, J.; Bashashati, M.; Li, Y.Y.; Storr, M. Distribution, function and physiological role of melatonin in the lower gut. World J. Gastroenterol. 2011, 17, 3888–3898. [Google Scholar] [CrossRef] [PubMed]
- Slominski, R.M.; Reiter, R.J.; Schlabritz-Loutsevitch, N.; Ostrom, R.S.; Slominski, A.T. Melatonin membrane receptors in peripheral tissues: Distribution and functions. Mol. Cell. Endocrinol. 2012, 35, 152–166. [Google Scholar] [CrossRef] [PubMed]
- Stebelova, K.; Anttila, K.; Manttari, S.; Saarela, S.I.; Zeman, M. Immunohistochemical definition of MT(2) receptors and melatonin in the gastrointestinal tissues of rat. Acta Histochem. 2010, 112, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, R.; Mahavadi, S.; Al-Shboul, O.; Bhattacharya, S.; Grider, J.R.; Muthry, K.S. Characterization of signaling pathways coupled to melatonin receptors in gastrointestinal smooth muscle. Regul. Pept. 2013, 184, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Costa, E.J.; Lopes, R.H.; Lamy-Freund, M.T. Permeability of pure lipid bilayers to melatonin. J. Pineal Res. 1995, 19, 123–126. [Google Scholar] [CrossRef] [PubMed]
- Reiter, R.J.; Tan, D.X.; Manchester, L.C.; Pilar-Terron, M.; Flores, L.J.; Koppisepi, S. Medical implications of melatonin: Receptor-mediated and receptor-independent actions. Adv. Med. Sci. 2007, 52, 11–28. [Google Scholar] [PubMed]
- Stefulj, J.; Hortner, M.; Ghosh, M.; Schauenstein, K.; Rinner, I.; Wolfler, A.; Sonner, J.; Liebman, P.M. Gene expression of the key enzymes in melatonin synthesis in extrapineal tissues in the rat. J. Pineal Res. 2001, 30, 243–247. [Google Scholar] [CrossRef] [PubMed]
- Shimozuma, M.; Tokuyama, R.; Tatehara, S.; Umeki, H.; Ide, S.; Mishima, K.; Saito, I.; Satomura, K. Expression and cellular localization of melatonin-synthesis enzyme in rat and human salivary glands. Histochem. Cell Biol. 2011, 135, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Leja-Szpak, A.; Bonior, J.; Nawrot, K.; Tomaszewska, R.; Stachura, J.; Sendur, R.; Konturek, S.J. Protective effect of melatonin and its precursor l-tryptophan on acute pancreatitis induced by caerulein overstimulation or ischemia/reperfusion. J. Pineal Res. 2003, 34, 40–52. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Nawrot-Porąbka, K.; Leja-Szpak, A.; Bonior, J.; Szklarczyk, J.; Kot, M.; Konturek, S.J.; Pawlik, W.W. Melatonin as modulator of pancreatic enzyme secretion and pancreatoprotector. J. Physiol. Pharmacol. 2007, 58, 65–80. [Google Scholar] [PubMed]
- Ciorba, M.A. Indoleamine 2,3 dioxygenase in intestinal disease. Curr. Opin. Gastroenterol. 2013, 29, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Terron, M.P.; Flores, L.J.; Reiter, R.J. One molecule, many derivatives: A never-ending interaction of melatonin with the reactive oxygen and nitrogen species. J. Pineal Res. 2007, 42, 28–42. [Google Scholar] [CrossRef] [PubMed]
- Harderland, R.; Tan, D.X.; Reiter, R.J. Kynuramines, metabolites of melatonin and other indoles: The resurrection of an almost forgotten class of biogenic amines. J. Pineal Res. 2009, 42, 109–126. [Google Scholar] [CrossRef] [PubMed]
- Tan, D.X.; Manchester, L.C.; Burkhardt, S.; Sainz, R.M.; Mayo, J.C.; Kohen, R.; Shohami, E.; Huo, Y.S.; Hardeland, R.; Reiter, R.J. N1-acetyl-N1-formyl-5-methoxy-kynuramine, a biogenic amine and melatonin metabolite, functions as a potent antioxidant. FASEB J. 2001, 15, 2294–2296. [Google Scholar] [PubMed]
- Janjetovic, Z.; Nahmias, Z.P.; Hanna, S.; Jarrett, S.G.; Kim, T.K.; Reiter, R.J.; Slominski, A.T. Melatonin and its metabolites ameliorate ultraviolet B-induced damage in human epidermal keratinocytes. J. Pineal Res. 2014, 57, 90–102. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, M.; Hardeland, R. The melatonin metabolite N1-acetyl-N1-formyl-5-methoxy- kynuramine is a potent singlet oxygen scavenger. J. Pineal Res. 2009, 46, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Mayo, J.C.; Sainz, R.M.; Tan, D.X.; Hardeland, R.; Leon, J.; Rodriguez, C.; Reiter, R.J. Antinflammatory action of melatonin and its metabolites N1-acetyl-N1-formyl-5-methoxykynuramine (AFMK), and N1-acetyl-5-methoxykynuramine (AMK) in macrophages. J. Neuroimmunol. 2005, 165, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Galano, A.; Tan, X.; Reiter, R.J. On the free radical scavenging activities of melatonin’s metabolites, AFMK and AMK. J. Pineal Res. 2013, 54, 245–257. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.O.; Ximenez, V.F.; Livramento, J.A.; Catalani, L.H.; Campa, A. High concentration of melatonin metabolite N1-acety-N1formyl-5-methoxykynuramine in cerebrospinal fluid of patients with meningitis; a possible immunomodulatory mechanism. J. Pineal Res. 2005, 39, 302–306. [Google Scholar] [CrossRef] [PubMed]
- Acuna-Castroviejo, D.; Escames, G.; Vernegas, C.; Diaz-Casado, M.E.; Lima-Cabello, E.; Lopez, L.C.; Rosales-Corral, S.; Tan, D.X.; Reiter, R.J. Extrapineal melatonin: Sources, regulation and potential functions. Cell Mol. Life Sci. 2014, 71, 2997–3025. [Google Scholar] [CrossRef] [PubMed]
- Hardeland, R.; Poeggeler, B. Melatonin and synthetic melatonergic agonists: Actions and metabolism in the central nervous system. Cent. Nerv. Syst. Agents Med. Chem. 2012, 12, 189–216. [Google Scholar] [CrossRef] [PubMed]
- Bubenik, G.A. Localization, physiological significance and possible clinical implication of gastrointestinal melatonin. Biol. Signals Recept. 2001, 10, 350–366. [Google Scholar] [CrossRef] [PubMed]
- Soderquist, F.; Hellstrom, P.M.; Cunningham, J.L. Human gastroenteropancreatic expression of melatonin and its receptors MT1 and MT2. PLoS ONE 2015, 10, e0120195. [Google Scholar] [CrossRef] [PubMed]
- Bubenik, G.A.; Brown, G.M. Pinealectomy reduces melatonin levels in the serum but not in the gastrointestinal tract of the rat. Biol. Signals 1997, 6, 40–44. [Google Scholar] [CrossRef] [PubMed]
- Peschke, E.; Bahr, I.; Muhlbauer, E. Melatonin and pancreatic islets: Interrelationships between melatonin, insulin and glucagon. Int. J. Mol. Sci. 2013, 14, 6981–7015. [Google Scholar] [CrossRef] [PubMed]
- Leja-Szpak, A.; Jaworek, J.; Pierzchalski, P.; Reiter, R.J. Melatonin Induces Pro-apoptotic Signaling Pathway in Human Pancreatic Carcinoma Cells (PANC-1). J. Pineal Res. 2010, 49, 248–255. [Google Scholar] [CrossRef] [PubMed]
- Gonzales, A.; del Castillo-Vaquero, A.; Miro-Moran, A.; Tapia, J.A.; Salido, G.M. Melatonin reduces pancreatic tumor cell viability by altering mitochondrial physiology. J. Pineal Res. 2011, 50, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Nawrot-Porabka, K.; Konturek, S.J.; Leja-Szpak, A.; Thor, P.; Pawlik, W.W. Melatonin and its precursor l-tryptophan: Influence on pancreatic amylase secretion in vivo and in vitro. J. Pineal Res. 2004, 36, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Leja-Szpak, A.; Jaworek, J.; Nawrot-Porabka, K.; Palonek, M.; Mitis-Musiol, M.; Dembinski, A.; Konturek, S.J.; Pawlik, W.W. Modulation of pancreatic enzyme secretion by melatonin and its precursor: l-tryptophan. Role of CCK and afferent nerves. J. Physiol. Pharmacol. 2004, 55, 33–46. [Google Scholar] [PubMed]
- Santofimia-Castaño, P.; Ruy, D.C.; Salido, G.M.; Gonzales, A. Melatonin modulates Ca2+ mobilization and amylase release in response to cholecystokinin octapeptide in mouse pancreatic acinar cells. J. Physiol. Biochem. 2013, 69, 897–908. [Google Scholar] [CrossRef] [PubMed]
- Bernard, P.P.; Polglaze, K.E.; Bernard, R.L.; Sandow, S.L.; Pozo, M.J. Detection of melatonin production from the intestinal epithelium using electochemical method. Curr. Pharm. Des. 2014, 20, 4802–4806. [Google Scholar]
- Hardeland, R. Melatonin in plants-diversity of levels and multiplicity of functions. Front. Plant Sci. 2016, 7, 198. [Google Scholar] [CrossRef] [PubMed]
- Bharwani, N.; Patel, S.; Prabhudesai, S.; Fotheringham, T.; Power, N. Acute pancreatitis: The role of imagining in diagnosis and management. Clin. Radiol. 2011, 66, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Gurusamy, K.S.; Belgaumkatr, A.P.; Haswell, A.; Pereira, S.P.; Davidson, B.R. Interventions for necrotizing pancreatitis. Cochrane Database Syst. Rev. 2016, 4, CD011383. [Google Scholar] [PubMed]
- Singh, P.; Garg, P.K. Pathophysiological mechanisms in acute pancreatitis: Current understanding. Indian J. Gastroenterol. 2016, 35, 153–166. [Google Scholar] [CrossRef] [PubMed]
- Sah, R.P.; Dawra, R.K.; Saluja, A.K. New insights into the pathogenesis in pancreatitis. Curr. Opin. Gastroenterol. 2013, 29, 523–530. [Google Scholar] [CrossRef] [PubMed]
- Manohar, M.; Verma, A.K.; Venkateshaiah, S.U.; Sanders, N.L.; Mishra, A. Pathogenetic mechanism of pancreatitis. World J. Gastrointest. Pharmacol. Ther. 2017, 8, 10–25. [Google Scholar] [CrossRef] [PubMed]
- Mylona, V.; Koussoulas, V.; Tzivras, D.; Makrygiannis, E.; Georgopoulou, P.; Koratzanis, G.; Giamarellos-Bourboulis, E.J.; Tzivras, M.D. Changes in adaptative and innate immunity in patients with acute pancreatitis and systemic inflammatory response syndrome. Pancreatology 2011, 11, 475–481. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, M. Acute pancreatitis as a model of SIRS. Front. Biosci. 2009, 14, 2042–2050. [Google Scholar] [CrossRef]
- Duan, L.; Ma, Y.; Chi, J.; Wang, X.; Wesely, A.J.; Chen, X. The regulatory role of immunosupressants on immune abnormalities in acute pancreatitis. Biomed. Rep. 2014, 2, 193–198. [Google Scholar] [PubMed]
- Watanabe, T.; Kudo, M.; Strober, W. Immunopathogenesis of pancreatitis. Mucosal Immunol. 2017, 10, 283–298. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Konturek, S.J. Hormonal protection in acute pancreatitis by ghrelin, leptin and melatonin. World J. Gastroenterol. 2014, 20, 16902–16912. [Google Scholar] [CrossRef] [PubMed]
- Szabolcs, A.; Reiter, R.J.; Letoha, T.; Hegy, P.; Papai, G.; Varga, I.; Jarmay, K.; Kaszaki, J.; Sari, R.; Rakonczay, Z., Jr.; et al. Effect of melatonin on the severity of l-arginine-induced experimental pancreatitis in rats. World J. Gastroenterol. 2006, 12, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J. Ghrelin and melatonin in the regulation of pancreatic exocrine secretion and maintaining of integrity. J. Physiol. Pharmacol. 2006, 57, 83–96. [Google Scholar] [PubMed]
- Qi, W.; Tan, R.X.; Reiter, R.J. Melatonin reduces lipid peroxidation and tissue edema in caerulein-induced acute pancreatitis in the rats. Dig. Dis. Sci. 1999, 44, 2257–2262. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Konturek, S.J.; Leja-Szpak, A. Role of endogenous melatonin and its MT2 receptor in the modulation of caerulein-induced pancreatitis in the rat. J. Physiol. Pharmacol. 2002, 53, 791–804. [Google Scholar] [PubMed]
- Esrefoglu, M.; Gul, M.; Ates, B.; Selimoglu, M.A. Ultrastructural clues for the protective effect of melatonin against oxidative damage in cerulein-induced pancreatitis. J. Pineal Res. 2006, 40, 92–97. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, C.; Marchena, A.M.; Holquin-Arevalo, M.S.; Martin-Partido, G.; Rodriquez, A.B.; Paredes, S.D.; Pariente, J.A. Anti-inflammatory effect of melatonin in a rat model of caerulein-induced acute pancreatitis. Cell Biochem. Funct. 2013, 31, 585–590. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.M.; Chen, J.C.; Ng, C.J.; Chiu, D.F.; Chen, M.F. Melatonin reduces pancreatic prostaglandins production and protects against caerulein-induced pancreatitis. J. Pineal Res. 2006, 40, 34–39. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Konturek, S.J.; Tomaszewska, R.; Leja-Szpak, A.; Bonior, J.; Nawrot, K.; Palonek, M.; Stachura, J.; Pawlik, W. The circadian rhythm of melatonin modulates the severity of caerulein-induced pancreatitis in the rat. J. Pineal Res. 2004, 37, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Col, C.; Dinler, K.; Hasdemir, A.O.; Bugdayci, G. The effect of intraperitoneal injection of melatonin on serum amylase levels in acute pancreatitis. JOP 2009, 10, 306–309. [Google Scholar] [PubMed]
- Baykal, A.; Iskit, A.B.; Hamaloglou, E.; Oguz, M.; Hascelik, G.; Sayek, Y. Melatonin modulates mesenteric blood flow and TNF alpha concentrations after lipopolysaccharide challenge. Eur. J. Surg. 2000, 166, 722–727. [Google Scholar] [PubMed]
- Sadek, A.S.; Khattab, R.S. The protective role of melatonin on l-arginine-induced acute pancreatitis. Folia Morphol. 2017, 76, 66–73. [Google Scholar] [CrossRef] [PubMed]
- Huai, J.P.; Shao, Y.; Sun, X.C.; Jin, Y.; Wu, J.S.; Huang, Z.M. Melatonin ameliorates necrotizing pancreatitis by the regulation of cytosolic Ca+2 homeostasis. Pancreatology 2012, 12, 257–263. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, J.; Zhao, Q.; Chen, Q.W.; Sun, Y.; Jin, Y.; Wu, J. Melatonin induces anti-inflammatory effects to play a protective role via endoplasmatic reticulum stress in acute pancreatitis. Cell. Physiol. Biochem. 2016, 40, 1094–1104. [Google Scholar] [CrossRef] [PubMed]
- Gulben, K.; Ozdemir, H.; Berberoglu, U.; Mersin, H.; Yrkin, F.; Cahyr, E.; Aksaray, S. Melatonin modulates the severity of taurocholate-induced acute pancreatitis. Dig. Dis. Sci. 2010, 55, 941–946. [Google Scholar] [CrossRef] [PubMed]
- Bonior, J.; Jaworek, J.; Konturek, S.J.; Pawlik, W.W. Increase of heat shock protein gene expression by melatonin in AR42J cells. J. Physiol. Pharmacol. 2005, 56, 471–481. [Google Scholar] [PubMed]
- Sun, X.; Shao, Y.; Jin, Y.; Huai, J.; Zhou, Q.; Huang, Z.; Wu, J. Melatonin reduces bacterial translocation by preventing damage to the intestinal mucosa in an experimental severe pancreatitis rat model. Exp. Ther. Med. 2013, 6, 1343–1349. [Google Scholar] [PubMed]
- Chou, T.Y.; Reiter, R.J.; Chen, K.H.; Leu, F.J.; Wang, D.; Yeh, D.Y. Pulmonary function changes in rats with taurocholate-induced pancreatitis are attenuated by pretreatment with melatonin. J. Pineal Res. 2014, 56, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, S.; Pandhi, P.; Malhortra, S.; Vaiphei, K.; Khanduya, K.L. Melatonin treatment is beneficial in pancreatic repair process after experimental acute pancreatitis. Eur. J. Pharmacol. 2010, 628, 282–289. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Szklarczyk, J.; Bonior, J.; Kot, M.; Goralska, M.; Pierzchalski, P.; Reiter, R.J.; Czech, U.; Tomaszewska, R. Melatonin metabolite, N1-acetyl-N1-formyl-5nmethoxykynuramine (AFMK), attenuates acute pancreatitis in the rat: In vivo and in vitro studies. J. Physiol. Pharmacol. 2016, 676, 411–421. [Google Scholar]
- Chen, G.Y.; Dai, R.W.; Luo, H.; Chen, T.; Lin, N.; Wang, T.; Luo, G.D.; Tang, L.J. Effect of percutaneous catheter drainage on pancreatic injury in rats with severe acute pancreatitis induced by sodium taurocholate. Pancreatology 2015, 15, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Booth, D.M.; Murphy, J.A.; Mukherjee, R.; Awais, M.; Neoptolemos, J.P.; Gerasimenko, O.V.; Tepikin, A.V.; Petersen, O.H.; Sutton, R.; Criddle, D.N. Reactive oxygen species induced by bile acid induce apoptosis and protect against necrosis in pancreatic acinar cells. Gastroenterology 2011, 140, 2116–2125. [Google Scholar] [CrossRef] [PubMed]
- Laothong, U.; Hiraku, Y.; Oikawa, S.; Intuyod, K.; Murata, M.; Pinlaor, S. Melatonin induces apoptosis in cholangiocarcinoma cell lines by activating the reactive oxygen species-mediated mitochondrial pathway. Oncol. Rep. 2015, 33, 1443–1449. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Wu, A.; Zhu, H.; Fang, H.; Xu, L.; Ye, J.; Shen, J. Melatonin is involved in the apoptosis and necrosis of pancreatic cancer cell line via modulating of Bcl-2/Bax balance. Biomed. Pharmacother. 2013, 67, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Wei, J.Y.; Li, M.W.; Zhou, L.L.; Lu, Q.N.; He, W. Melatonin induced apoptosis of colorectal cancer cells through HDAC4 nuclear import mediated by CaMKII inactivation. J. Pineal Res. 2015, 58, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Jaworek, J.; Zwirska-Korczala, K.; Szklarczyk, J.; Nawrot-Porąbka, K.; Leja-Szpak, A.; Jaworek, A.K.; Tomaszewska, R. Pinealectomy aggravates acute pancreatitis in the rat. Pharmacol. Rev. 2010, 62, 864–873. [Google Scholar] [CrossRef]
- Belayev, O.; Herzog, T.; Munding, J.; Bolik, B.; Vosschulte, A.; Uhl, W.; Muller, C.A. Protective role of endogenous melatonin in the early course of human acute pancreatitis. J. Pineal Res. 2011, 50, 71–77. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Lin, C.J.; Dong, L.M.; Chen, M.J.; Zhou, Q.; Wu, J.S. Clinical significance of melatonin concentrations in predicting the severity of acute pancreatitis. World J. Gastroenterol. 2013, 19, 4066–4071. [Google Scholar] [CrossRef] [PubMed]
- Leja-Szpak, A.; Jaworek, J.; Szklarczyk, J.; Konturek, S.J.; Pawlik, W.W. Melatonin stimulates HSP27 phosphorylation in human pancreatic carcinoma cells (PANC-1). J. Physiol. Pharmacol. 2007, 58, 177–188. [Google Scholar] [PubMed]
- Jaworek, J.; Leja-Szpak, A. Melatonin influences pancreatic cancerogenesis. Histol. Histopathol. 2014, 29, 423–431. [Google Scholar] [PubMed]
- Li, W.; Wu, J.; Li, Z.; Zhou, Z.; Zheng, C.; Lin, L.; Tan, B.; Huang, M.; Fan, M. Melatonin induces cell apoptosis in Mia PaCa-2 cells via the suppression of nuclear factor-κB and activation of ERK and JNK: A novel therapeutic implication for pancreatic cancer. Oncol. Rep. 2016, 36, 2861–2867. [Google Scholar] [CrossRef] [PubMed]
- Cucina, A.; Proietti, S.; D’Anselmi, F.; Coluccia, P.; Dinicola, S.; Frati, L.; Bizzarri, M. Evidence for a biphasic apoptotic pathway induced by melatonin in MCF-7 breast cancer cells. J. Pineal Res. 2009, 46, 172–180. [Google Scholar] [CrossRef] [PubMed]
- Fan, C.; Pan, Y.; Yang, Y.; Di, S.; Jiang, S.; Ma, Z.; Li, T.; Zhang, Z.; Li, W.; Li, X.; et al. HDAC1 inhibition by melatonin leads to suppression of lung adenocarcinoma cells via induction of oxidative stress and activation of apoptotic pathways. J. Pineal Res. 2015, 59, 321–333. [Google Scholar] [CrossRef] [PubMed]
- Hong, Y.; Won, J.; Lee, Y.; Lee, S.; Park, K.; Chang, K.T.; Hong, Y. Melatonin treatment induces interplay of apoptosis, autophagy, and senescence in human colorectal cancer cells. J. Pineal Res. 2014, 56, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.K.; Lin, Z.; Tidwell, W.J.; Li, W.; Slominski, A.T. Melatonin and its metabolites accumulate in the human epidermis in vivo and inhibit proliferation and tyrosinase activity in epidermal melanocytes in vitro. Mol. Cell. Endocrinol. 2015, 404, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Leja-Szpak, A.; Pierzchalski, P.; Goralska, M.; Nawrot-Porabka, K.; Bonior, J.; Link-Lenczowski, P.; Jastrzebska, M.; Jaworek, J. Kynuramines induce overexpression of heat shock proteins in pancreatic cancer cells via 5-hydroxytryptamine and MT1/MT2 receptors. J. Physiol. Pharmacol. 2015, 66, 711–718. [Google Scholar] [PubMed]
- Rozov, S.V.; Filatova, E.V.; Orlov, A.A.; Volkova, A.V.; Zhloba, A.R.; Blashko, E.L.; Pozdeyev, N.V. N1-acetyl-N2-formyl-5-methoxykynuramine is a product of melatonin oxidation in rats. J. Pineal Res. 2003, 35, 245–250. [Google Scholar] [CrossRef] [PubMed]
- Onuki, J.; Almeida, E.A.; Medeiros, M.H.; di Mascio, P. Inhibition of 5-aminolevulinic acid-induced DNA damage by melatonin, N1-acetyl-N2-formyl-5-methoxykynuramine, quercetin or resveratrol. J. Pineal Res. 2005, 38, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Boutin, J.A.; Marcheteau, E.; Hennig, P.; Moulharat, N.; Berger, S.; Delagrange, P.; Bouchet, J.P.; Ferry, G. MT3/QR2 melatonin binding site does not use melatonin as a substrate or a co-substrate. J. Pineal Res. 2008, 45, 524–531. [Google Scholar] [CrossRef] [PubMed]
- Budu, A.; Peres, R.; Bueno, V.B.; Catalani, L.H.; Garcia, C.R. N1-acetyl-N2-formyl-5-methoxykynuramine modulates the cell cycle of malaria parasites. J. Pineal Res. 2007, 42, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Dragicevic, N.; Copes, N.; O’Neal-Moffitt, G.; Jin, J.; Buzzeo, R.; Mamcarz, M.; Tan, J.; Cao, C.; Olcese, J.M.; Arendash, G.W.; et al. Melatonin treatment restores mitochondrial function in Alzheimer’s mice: A mitochondrial protective role of melatonin membrane receptor signaling. J. Pineal Res. 2011, 51, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Ju, HQ.; Li, H.; Tian, T.; Lu, Y.X.; Bai, L.; Chen, L.Z.; Sheng, H.; Mo, H.Y.; Zeng, J.B.; Deng, W.; et al. Melatonin overcomes gemcitabine resistance in pancreatic ductal adenocarcinoma by abrogating nuclear factor-κB activation. J. Pineal Res. 2016, 60, 27–38. [Google Scholar] [CrossRef] [PubMed]
- Uguz, A.C.; Cig, B.; Espino, J.; Bejarano, I.; Naziroglu, M.; Rodríguez, A.B.; Pariente, J.A. Melatonin potentiates chemotherapy-induced cytotoxicity and apoptosis in rat pancreatic tumor cells. J. Pineal Res. 2012, 53, 91–98. [Google Scholar] [CrossRef] [PubMed]
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jaworek, J.; Leja-Szpak, A.; Nawrot-Porąbka, K.; Szklarczyk, J.; Kot, M.; Pierzchalski, P.; Góralska, M.; Ceranowicz, P.; Warzecha, Z.; Dembinski, A.; et al. Effects of Melatonin and Its Analogues on Pancreatic Inflammation, Enzyme Secretion, and Tumorigenesis. Int. J. Mol. Sci. 2017, 18, 1014. https://doi.org/10.3390/ijms18051014
Jaworek J, Leja-Szpak A, Nawrot-Porąbka K, Szklarczyk J, Kot M, Pierzchalski P, Góralska M, Ceranowicz P, Warzecha Z, Dembinski A, et al. Effects of Melatonin and Its Analogues on Pancreatic Inflammation, Enzyme Secretion, and Tumorigenesis. International Journal of Molecular Sciences. 2017; 18(5):1014. https://doi.org/10.3390/ijms18051014
Chicago/Turabian StyleJaworek, Jolanta, Anna Leja-Szpak, Katarzyna Nawrot-Porąbka, Joanna Szklarczyk, Michalina Kot, Piotr Pierzchalski, Marta Góralska, Piotr Ceranowicz, Zygmunt Warzecha, Artur Dembinski, and et al. 2017. "Effects of Melatonin and Its Analogues on Pancreatic Inflammation, Enzyme Secretion, and Tumorigenesis" International Journal of Molecular Sciences 18, no. 5: 1014. https://doi.org/10.3390/ijms18051014
APA StyleJaworek, J., Leja-Szpak, A., Nawrot-Porąbka, K., Szklarczyk, J., Kot, M., Pierzchalski, P., Góralska, M., Ceranowicz, P., Warzecha, Z., Dembinski, A., & Bonior, J. (2017). Effects of Melatonin and Its Analogues on Pancreatic Inflammation, Enzyme Secretion, and Tumorigenesis. International Journal of Molecular Sciences, 18(5), 1014. https://doi.org/10.3390/ijms18051014