The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A Comprehensive Review and Future Therapeutic Perspectives
Abstract
1. Introduction
1.1. IBD Epidemiology and Dietary Intake of n-6/n-3 PUFAs
1.2. n-6/n-3 PUFAs and Inflammatory Processes in IBD
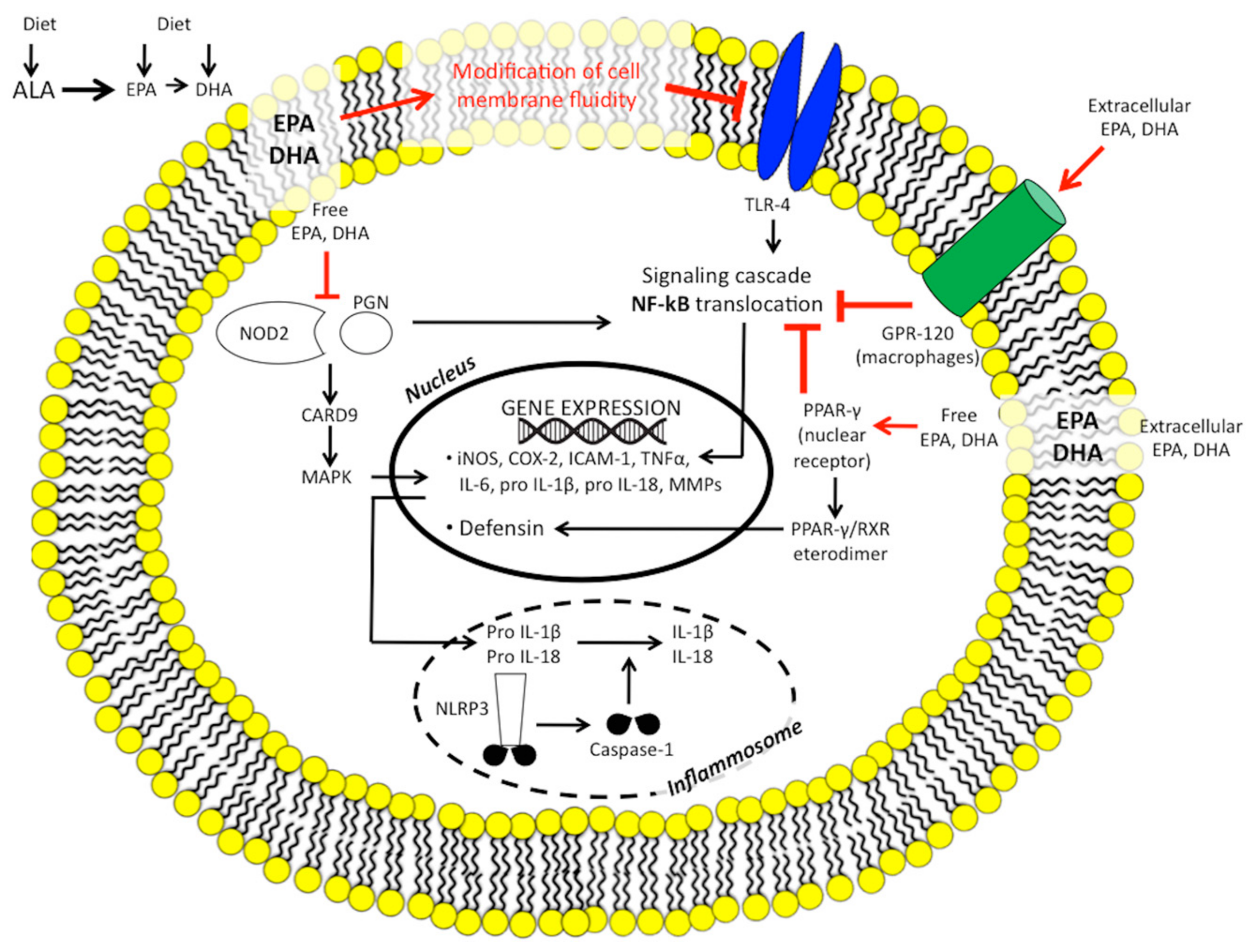
1.2.1. Eicosanoid Pathways and Inflammation
1.2.2. n-3 PUFAs and the Endocannabinoid Epoxides Pathway
1.2.3. n-3 PUFAs and Inflammatory Gene Expression
1.2.4. n-3 PUFAs and Adaptive Immunity
1.2.5. n-3 PUFAs, Innate Immunity, the Inflammasome, and Microbiome
1.2.6. n-3 PUFAs and Nitric Oxide
1.3. n-6 and n-3 PUFAs in IBD Clinical Trials
Cochrane Reviews of n-3 PUFAs Treatment in IBD
1.4. New Formulations and Bioavailability of n-3 PUFAs
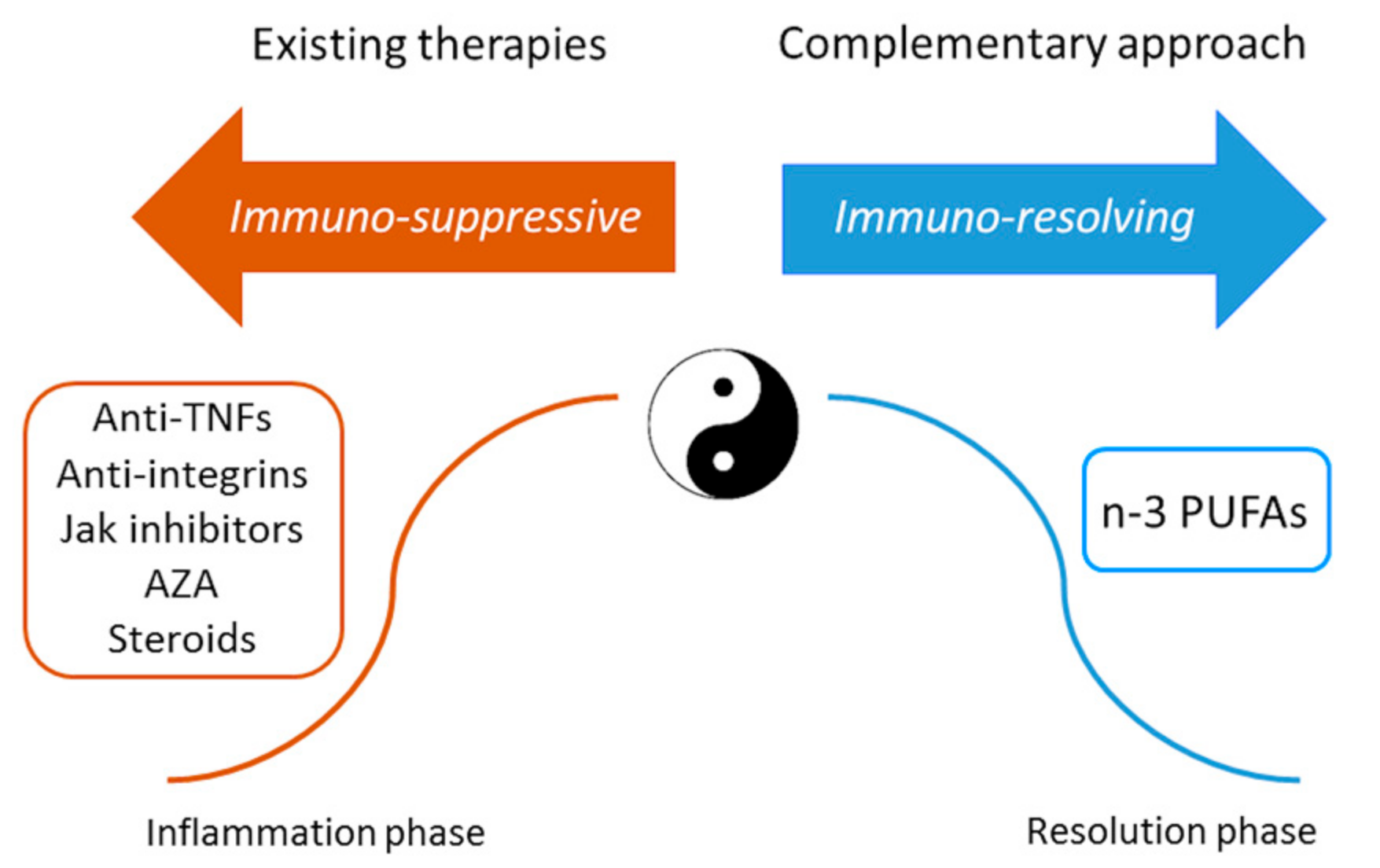
2. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 5-ASA | 5-aminosalicylic acid |
| AA | Arachidonic acid |
| AE | Adverse event |
| ALA | α-linolenic acid |
| CD | Crohn’s disease |
| CI | Confidence interval |
| COX | Cyclooxygenase |
| CDAI | Crohn’s Disease Activity Index |
| CRP | C reactive protein |
| DHA | Docosahexaenoic acid |
| DPA | Docosapentaenoic acid |
| EET | Epoxyeicosatrienoic acid |
| ELOVL | Elongase of very long chain fatty acids |
| EPA | Eicosapentaenoic acid |
| EPIC | European Prospective Investigation into Cancer and Nutrition |
| FADS | Fatty acid desaturase |
| FC | Faecal calprotectin |
| FFA | Free fatty acid |
| GLA | n-6 gamma linolenic acid |
| HETEs | Hydroxyeicosatetraenoic acid |
| HR | Hazard ratio |
| IBD | Inflammatory bowel disease |
| IL | Interleukin |
| LA | Linoleic acid |
| LOX | Lipoxygenase |
| LT | Leukotriene |
| LX | Lipoxin |
| MAPK | Mitogen-activated protein kinase |
| NFkβ | Nuclear factor kB |
| NLRs | NOD-like receptors |
| NLRP3 | NOD-like receptor protein 3 |
| NO | Nitric oxide |
| NOD2 | Nucleotide-binding oligomerisation domain 2 |
| NOS | Nitric oxide synthase |
| OA | Oleic acid |
| OR | Odds ratio |
| PG | Prostaglandin |
| PPAR-γ | Peroxisome proliferator-activated receptor γ |
| PUFAs | Polyunsaturated fatty acids |
| RBC | Red blood cell |
| SNPs | Single nucleotide polymorphisms |
| SPM | Specialised pro-resolving mediator |
| TLRs | Toll-like receptors |
| TNFα | Tumor necrosis factor α |
| TX | Thromboxane |
| UC | Ulcerative colitis |
| VCAM-1 | Vascular cell adhesion molecule 1 |
References
- Blasbalg, T.L.; Hibbeln, J.R.; Ramsden, C.E.; Majchrzak, S.F.; Rawlings, R.R. Changes in consumption of omega-3 and omega-6 fatty acids in the United States during the 20th century. Am. J. Clin. Nutr. 2011, 93, 950–962. [Google Scholar] [CrossRef] [PubMed]
- Molodecky, N.A.; Soon, I.S.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W.; et al. Increasing incidence and prevalence of the inflammatory bowel diseases with time, based on systematic review. Gastroenterology 2012, 142, 46–54. [Google Scholar] [CrossRef] [PubMed]
- De Silva, P.S.; Olsen, A.; Christensen, J.; Schmidt, E.B.; Overvaad, K.; Tjonneland, A.; Hart, A.R. An association between dietary arachidonic acid, measured in adipose tissue, and ulcerative colitis. Gastroenterology 2010, 139, 1912–1917. [Google Scholar] [CrossRef] [PubMed]
- IBD in EPIC Study Investigators; Tjonneland, A.; Overvad, K.; Bergmann, M.M.; Nagel, G.; Linseisen, J.; Hallmans, G.; Palmqvist, R.; Sjodin, H.; Hagglund, G.; et al. Linoleic acid, a dietary n-6 polyunsaturated fatty acid, and the aetiology of ulcerative colitis: A nested case-control study within a European prospective cohort study. Gut 2009, 58, 1606–1611. [Google Scholar] [PubMed]
- Ananthakrishnan, A.N.; Khalili, H.; Konijeti, G.G.; Higuchi, L.M.; de Silva, P.; Fuchs, C.S.; Willett, W.C.; Richter, J.M.; Chan, A.T. Long-term intake of dietary fat and risk of ulcerative colitis and Crohn’s disease. Gut 2014, 63, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Yates, C.M.; Calder, P.C.; Ed Rainger, G. Pharmacology and therapeutics of omega-3 polyunsaturated fatty acids in chronic inflammatory disease. Pharmacol. Ther. 2014, 141, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Fritsche, K. Fatty acids as modulators of the immune response. Annu. Rev. Nutr. 2006, 26, 45–73. [Google Scholar] [CrossRef] [PubMed]
- Novak, T.E.; Babcock, T.A.; Jho, D.H.; Helton, W.S.; Espat, N.J. NF-κB inhibition by omega −3 fatty acids modulates LPS-stimulated macrophage TNF-α transcription. Am. J. Physiol. Lung Cell Mol. Physiol. 2003, 284, L84–L89. [Google Scholar] [CrossRef] [PubMed]
- Yaqoob, P. Mechanisms underlying the immunomodulatory effects of n-3 PUFA. Proc. Nutr. Soc. 2010, 69, 311–315. [Google Scholar] [CrossRef] [PubMed]
- Goldman, D.W.; Pickett, W.C.; Goetzl, E.J. Human neutrophil chemotactic and degranulating activities of leukotriene B5 (LTB5) derived from eicosapentaenoic acid. Biochem. Biophys. Res. Commun. 1983, 117, 282–288. [Google Scholar] [CrossRef]
- Calder, P.C. Marine omega-3 fatty acids and inflammatory processes: Effects, mechanisms and clinical relevance. Biochim. Biophys. Acta 2015, 1851, 469–484. [Google Scholar] [CrossRef] [PubMed]
- Serhan, C.N. Pro-resolving lipid mediators are leads for resolution physiology. Nature 2014, 510, 92–101. [Google Scholar] [CrossRef] [PubMed]
- Lev-Tzion, R.; Griffiths, A.M.; Leder, O.; Turner, D. Omega 3 fatty acids (fish oil) for maintenance of remission in Crohn’s disease. Cochrane Database Syst. Rev. 2014, 2, CD006320. [Google Scholar]
- Turner, D.; Shah, P.S.; Steinhart, A.H.; Zlotkin, S.; Griffiths, A.M. Maintenance of remission in inflammatory bowel disease using omega-3 fatty acids (fish oil): A systematic review and meta-analyses. Inflamm. Bowel Dis. 2011, 17, 336–345. [Google Scholar] [CrossRef] [PubMed]
- Aslan, A.; Triadafilopoulos, G. Fish oil fatty acid supplementation in active ulcerative colitis: A double-blind, placebo-controlled, crossover study. Am. J. Gastroenterol. 1992, 87, 432–437. [Google Scholar] [PubMed]
- Stenson, W.F.; Cort, D.; Rodgers, J.; Burakoff, R.; DeSchryver-Kecskemeti, K.; Gramlich, T.L.; Beeken, W. Dietary supplementation with fish oil in ulcerative colitis. Ann. Intern. Med. 1992, 116, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, A.B.; Daneshmend, T.K.; Hawkey, C.J.; Belluzzi, A.; Everitt, S.J.; Holmes, G.K.; Malkinson, C.; Shaheen, M.Z.; Willars, J.E. Treatment of ulcerative colitis with fish oil supplementation: A prospective 12 month randomised controlled trial. Gut 1992, 33, 922–928. [Google Scholar] [CrossRef] [PubMed]
- McCall, T.B.; O’Leary, D.; Bloomfield, J.; O’Moráin, C.A. Therapeutic potential of fish oil in the treatment of ulcerative colitis. Aliment. Pharmacol. Ther. 1989, 3, 415–424. [Google Scholar] [CrossRef] [PubMed]
- Loeschke, K.; Ueberschaer, B.; Pietsch, A.; Gruber, E.; Ewe, K.; Wiebecke, B.; Heldwein, W.; Lorenz, R. n-3 fatty acids only delay early relapse of ulcerative colitis in remission. Dig. Dis. Sci. 1996, 41, 2087–2094. [Google Scholar] [CrossRef] [PubMed]
- Salomon, P.; Kornbluth, A.A.; Janowitz, H.D. Treatment of ulcerative colitis with fish oil n-3-omega-fatty acid: An open trial. J. Clin. Gastroenterol. 1990, 12, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Almallah, Y.Z.; Richardson, S.; O’Hanrahan, T.; Mowat, N.A.; Brunt, P.W.; Sinclair, T.S.; Ewen, S.; Heys, S.D.; Eremin, O. Distal procto-colitis, natural cytotoxicity, and essential fatty acids. Am. J. Gastroenterol. 1998, 93, 804–809. [Google Scholar] [CrossRef] [PubMed]
- Dichi, I.; Frenhane, P.; Dichi, J.B.; Correa, C.R.; Angeleli, A.Y.; Bicudo, M.H.; Rodrigues, M.A.; Victória, C.R.; Burini, R.C. Comparison of omega-3 fatty acids and sulfasalazine in ulcerative colitis. Nutrition 2000, 16, 87–90. [Google Scholar] [CrossRef]
- Lorenz, R.; Weber, P.C.; Szimnau, P.; Heldwein, W.; Strasser, T.; Loeschke, K. Supplementation with n-3 fatty acids from fish oil in chronic inflammatory bowel disease: A randomized placebo-controlled double-blind cross-over trial. J. Int. Med. 1989, 225, 225–232. [Google Scholar] [CrossRef]
- Mate, J.; Castanos, R.; Garcia-Samaniego, J.; Pajares, J.M. Does dietary fish oil maintain the remission of Crohn’s disease (CD): A study case control. Gastroenterology 1993, 100, 228. [Google Scholar]
- Lorenz-Meyer, H.; Bauer, P.; Nicolay, C.; Schulz, B.; Purrmann, J.; Fleig, W.E.; Scheurlen, C.; Koop, I.; Pudel, V.; Carr, L. Omega-3 fatty acids and carbohydrate diet for maintenance of remission in Crohn’s disease. Scan. J. Gastroenterol. 1996, 31, 778–785. [Google Scholar] [CrossRef]
- Cheifetz, A.S.; Gianotti, R.; Luber, R.; Gibson, P.R. Complementary and alternative medicines used by patients with inflammatory bowel diseases. Gastroenterology 2017, 152, 415–429. [Google Scholar] [CrossRef] [PubMed]
- Turner, D.; Steinhart, A.H.; Griffiths, A.M. Omega 3 fatty acids (fish oil) for maintenance of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2007, 3, CD006443. [Google Scholar]
- De Ley, M.; de Vos, R.; Hommes, D.W.; Stokkers, P. Fish oil for induction of remission in ulcerative colitis. Cochrane Database Syst. Rev. 2007, 4, CD005986. [Google Scholar]
- Appleyard, C.B.; Hernández, G.; Rios-Bedoya, C.F. Basic epidemiology of inflammatory bowel disease in Puerto Rico. Inflamm. Bowel. Dis. 2004, 10, 106–111. [Google Scholar] [CrossRef] [PubMed]
- Charlebois, A.; Rosenfeld, G.; Bressler, B. The Impact of Dietary Interventions on the Symptoms of Inflammatory Bowel Disease: A Systematic Review. Crit. Rev. Food. Sci. Nutr. 2016, 56, 1370–1378. [Google Scholar] [CrossRef] [PubMed]
- Damas, O.M.; Jahann, D.A.; Reznik, R.; McCauley, J.L.; Tamariz, L.; Deshpande, A.R.; Abreu, M.T.; Sussman, D.A. Phenotypic manifestations of inflammatory bowel disease differ between Hispanics and non-Hispanic whites: Results of a large cohort study. Am. J. Gastroenterol. 2013, 108, 231–239. [Google Scholar] [CrossRef] [PubMed]
- Eaton, S.B.; Konner, M. A consideration of its nature and current implications. N. Engl. J. Med. 1985, 312, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Konner, M.; Eaton, S.B. Paleolithic nutrition: Twenty-five years later. Nutr. Clin. Pract. 2010, 25, 594–602. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.L. Paleolithic diet, evolution, and carcinogens. Science 1987, 238, 1633–1634. [Google Scholar] [CrossRef] [PubMed]
- Jew, S.; AbuMweis, S.S.; Jones, P.J. Evolution of the human diet: Linking our ancestral diet to modern functional foods as a means of chronic disease prevention. J. Med. Food 2009, 12, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Whalen, K.A.; McCullough, M.L.; Flanders, W.D.; Hartman, T.J.; Judd, S.; Bostick, R.M. Paleolithic and mediterranean diet pattern scores are inversely associated with biomarkers of inflammation and oxidative balance in adults. J. Nutr. 2016, 146, 1217–1226. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A.P. The importance of the ratio of omega-6/omega-3 essential fatty acids. Biomed. Pharmacother. 2002, 56, 365–379. [Google Scholar] [CrossRef]
- Von Schacky, C.; Fischer, S.; Weber, P.C. Long-term effects of dietary marine omega-3 fatty acids upon plasma and cellular lipids, platelet function, and eicosanoid formation in humans. J. Clin. Investig. 1985, 76, 1231–1626. [Google Scholar] [CrossRef] [PubMed]
- Reif, S.; Klein, I.; Lubin, F.; Farbstein, M.; Hallak, A.; Gilat, T. Pre-illness dietary factors in inflammatory bowel disease. Gut 1997, 40, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Geerling, B.J.; Dagnelie, P.C.; Badart-Smook, A.; Russel, M.G.; Stockbrügger, R.W.; Brummer, R.J. Diet as a risk factor for the development of ulcerative colitis. Am. J. Gastroenterol. 2000, 95, 1008–1013. [Google Scholar] [CrossRef] [PubMed]
- Shoda, R.; Matsueda, K.; Yamato, S.; Umeda, N. Epidemiologic analysis of Crohn disease in Japan: Increased dietary intake of n-6 polyunsaturated fatty acids and animal protein relates to the increased incidence of Crohn disease in Japan. Am. J. Clin. Nutr. 1996, 63, 741–745. [Google Scholar] [PubMed]
- John, S.; Luben, R.; Shrestha, S.S.; Welch, A.; Khaw, K.T.; Hart, A.R. Dietary n-3 polyunsaturated fatty acids and the aetiology of ulcerative colitis: A UK prospective cohort study. Eur. J. Gastroenterol. Hepatol. 2010, 22, 602–606. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.S.; Luben, R.; Olsen, A.; Tjonneland, A.; Kaaks, R.; Lindgren, S.; Grip, O.; Bergmann, M.M.; Boeing, H.; Hallmans, G.; et al. Association between high dietary intake of the n-3 polyunsaturated fatty acid docosahexaenoic acid and reduced risk of Crohn’s disease. Aliment. Pharmacol. Ther. 2014, 39, 834–842. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.K.; Abraham, B.; El-Serag, H. Dietary intake and risk of developing inflammatory bowel disease: A systematic review of the literature. Am. J. Gastroenterol. 2011, 106, 563–573. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Polyunsaturated fatty acids, inflammatory processes and inflammatory bowel diseases. Mol. Nutr. Food Res. 2008, 52, 885–897. [Google Scholar] [CrossRef] [PubMed]
- Pearl, D.S.; Masoodi, M.; Eiden, M.; Brümmer, J.; Gullick, D.; McKeever, T.M.; Whittaker, M.A.; Nitch-Smith, H.; Brown, J.F.; Shute, J.K.; et al. Altered colonic mucosal availability of n-3 and n-6 polyunsaturated fatty acids in ulcerative colitis and the relationship to disease activity. J. Crohns Colitis 2014, 8, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.H.; Hoover, R.L.; Williams, J.D.; Sperling, R.I.; Ravalese, J., 3rd; Spur, B.W.; Robinson, D.R.; Corey, E.J.; Lewis, R.A.; Austen, K.F. Effect of dietary enrichment with eicosapentaenoic and docosahexaenoic acids on in vitro neutrophil and monocyte leukotriene generation and neutrophil function. N. Engl. J. Med. 1985, 312, 1217–1224. [Google Scholar] [CrossRef] [PubMed]
- Endres, S.; Ghorbani, R.; Kelley, V.E.; Georgilis, K.; Lonnemann, G.; van der Meer, J.W.; Cannon, J.G.; Rogers, T.S.; Klempner, M.S.; Weber, P.C.; et al. The effect of dietary supplementation with n-3 polyunsaturated fatty acids on the synthesis of interleukin-1 and tumor necrosis factor by mononuclear cells. N. Engl. J. Med. 1989, 320, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Rees, D.; Miles, E.A.; Banerjee, T.; Wells, S.J.; Roynette, C.E.; Wahle, K.W.; Calder, P.C. Dose-related effects of eicosapentaenoic acid on innate immune function in healthy humans: A comparison of young and older men. Am. J. Clin. Nutr. 2006, 83, 331–342. [Google Scholar] [PubMed]
- McDougle, D.R.; Watson, J.E.; Abdeen, A.A.; Adili, R.; Caputo, M.P.; Krapf, J.E.; Johnson, R.W.; Kilian, K.A.; Holinstat, M.; Das, A. Anti-inflammatory ω-3 endocannabinoid epoxides. Proc. Natl. Acad. Sci. USA 2017, 114, E6034–E6043. [Google Scholar] [CrossRef] [PubMed]
- Vanden Berghe, W.; Vermeulen, L.; Delerive, P.; De Bosscher, K.; Staels, B.; Haegeman, G. A paradigm for gene regulation: Inflammation, NF-êB and PPAR. Adv. Exp. Med. Biol. 2003, 544, 181–196. [Google Scholar] [PubMed]
- Moraes, L.A.; Piqueras, L.; Bishop-Bailey, D. Peroxisome proliferator-activated receptors and inflammation. Pharmacol. Ther. 2006, 110, 371–385. [Google Scholar] [CrossRef] [PubMed]
- Dubuquoy, L.; Jansson, E.A.; Deeb, S.; Rakotobe, S.; Karoui, M.; Colombel, J.F.; Auwerx, J.; Pettersson, S.; Desreumaux, P. Impaired expression of peroxisome proliferator-activated receptor γ in ulcerative colitis. Gastroenterology 2003, 124, 1265–1276. [Google Scholar] [CrossRef]
- Hassan, A.; Ibrahim, A.; Mbodji, K.; Coëffier, M.; Ziegler, F.; Bounoure, F.; Chardigny, J.M.; Skiba, M.; Savoye, G.; Déchelotte, P.; et al. An α-linolenic acid-rich formula reduces oxidative stress and inflammation by regulating NF-κB in rats with TNBS-induced colitis. J. Nutr. 2010, 140, 1714–1721. [Google Scholar] [CrossRef] [PubMed]
- Kliewer, S.A.; Sundseth, S.S.; Jones, S.A.; Brown, P.J.; Wisely, G.B.; Koble, C.S.; Devchand, P.; Wahli, W.; Willson, T.M.; Lenhard, J.M.; et al. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors α and γ. Proc. Natl. Acad. Sci. USA 1997, 94, 4318–4323. [Google Scholar] [CrossRef] [PubMed]
- Knoch, B.; Barnett, M.P.; Zhu, S.; Park, Z.A.; Nones, K.; Dommels, Y.E.; Knowles, S.O.; McNabb, W.C.; Roy, N.C. Genome-wide analysis of dietary eicosapentaenoic acid- and oleic acid-induced modulation of colon inflammation in interleukin-10 gene-deficient mice. J. Nutrigenet. Nutrigenomi. 2009, 2, 9–28. [Google Scholar] [CrossRef] [PubMed]
- Costea, I.; Mack, D.R.; Lemaitre, R.N.; Israel, D.; Marcil, V.; Ahmad, A.; Amre, D.K. Interactions between the dietary polyunsaturated fatty acid ratio and genetic factors determine susceptibility to pediatric Crohn’s disease. Gastroenterology 2014, 146, 929–931. [Google Scholar] [CrossRef] [PubMed]
- Corcos, L.; Lucas, D.; Le Jossic-Corcos, C.; Dréano, Y.; Simon, B.; Plée-Gautier, E.; Amet, Y.; Salaün, J.P. Human cytochrome P450 4F3: Structure, functions, and prospects. Drug Metabol. Drug Interact. 2012, 27, 63–71. [Google Scholar] [CrossRef] [PubMed]
- Kettunen, J.; Tukiainen, T.; Sarin, A.P.; Ortega-Alonso, A.; Tikkanen, E.; Lyytikäinen, L.P.; Kangas, A.J.; Soininen, P.; Würtz, P.; Silander, K.; et al. Genome-wide association study identifies multiple loci influencing human serum metabolite levels. Nat. Genet. 2012, 44, 269–276. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.Y.; Kothapalli, K.S.; Brenna, J.T. Desaturase and elongase-limiting endogenous long-chain polyunsaturated fatty acid biosynthesis. Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Grimble, R.F.; Howell, W.M.; O’Reilly, G.; Turner, S.J.; Markovic, O.; Hirrell, S.; East, J.M.; Calder, P.C. The ability of fish oil to suppress tumor necrosis factor α production by peripheral blood mononuclear cells in healthy men is associated with polymorphisms in genes that influence tumor necrosis factor α production. Am. J. Clin. Nutr. 2002, 76, 454–459. [Google Scholar] [PubMed]
- Sperling, R.I.; Benincaso, A.I.; Knoell, C.T.; Larkin, J.K.; Austen, K.F.; Robinson, D.R. Dietary omega-3 polyunsaturated fatty acids inhibit phosphoinositide formation and chemotaxis in neutrophils. J. Clin. Investig. 1993, 91, 651–660. [Google Scholar] [CrossRef] [PubMed]
- Calder, P.C. Omega-3 fatty acids and inflammatory processes. Nutrients 2010, 2, 355–374. [Google Scholar] [CrossRef] [PubMed]
- De Caterina, R.; Cybulsky, M.I.; Clinton, S.K.; Gimbrone, M.A., Jr.; Libby, P. The omega-3 fatty acid docosahexaenoate reduces cytokine-induced expression of proatherogenic and proinflammatory proteins in human endothelial cells. Arterioscler. Thromb. 1994, 14, 1829–1836. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, D.C.; Carding, S.R. Inflammatory bowel disease: Cause and immunobiology. Lancet 2007, 369, 1627–1640. [Google Scholar] [CrossRef]
- Kostic, A.D.; Xavier, R.J.; Gevers, D. The microbiome in inflammatory bowel disease: Current status and the future ahead. Gastroenterology 2014, 146, 1489–1499. [Google Scholar] [CrossRef] [PubMed]
- Cario, E. Toll-like receptors in inflammatory bowel diseases: A decade later. Inflamm. Bowel Dis. 2010, 16, 1583–1597. [Google Scholar] [CrossRef] [PubMed]
- Hugot, J.P.; Chamaillard, M.; Zouali, H.; Lesage, S.; Cézard, J.P.; Belaiche, J.; Almer, S.; Tysk, C.; O’Morain, C.A.; Gassull, M.; et al. Association of NOD2 leucine-rich repeat variants with susceptibility to Crohn’s disease. Nature 2001, 411, 599–603. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, S.; Ma, X. Role of Nod2 in the development of Crohn’s disease. Microbes Infect. 2009, 11, 912–918. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Plakidas, A.; Lee, W.H.; Heikkinen, A.; Chanmugam, P.; Bray, G.; Hwang, D.H. Differential modulation of Toll-like receptors by fatty acids: Preferential inhibition by n-3 polyunsaturated fatty acids. J. Lipid Res. 2003, 44, 479–486. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.; Mbodji, K.; Hassan, A.; Aziz, M.; Boukhettala, N.; Coëffier, M.; Savoye, G.; Déchelotte, P.; Marion-Letellier, R. Anti-inflammatory and anti-angiogenic effect of long chain n-3 polyunsaturated fatty acids in intestinal microvascular endothelium. Clin. Nutr. 2011, 30, 678–687. [Google Scholar] [CrossRef] [PubMed]
- Allam-Ndoul, B.; Guénard, F.; Barbier, O.; Vohl, M.C. Effect of n-3 fatty acids on the expression of inflammatory genes in THP-1 macrophages. Lipids Health Dis. 2016, 15, 69. [Google Scholar] [CrossRef] [PubMed]
- Martinon, F.; Mayor, A.; Tschopp, J. The inflammasomes: Guardians of the body. Annu. Rev. Immunol. 2009, 27, 229–265. [Google Scholar] [CrossRef] [PubMed]
- Corridoni, D.; Arseneau, K.O.; Cifone, M.G.; Cominelli, F. The dual role of Nod-like receptors in mucosal innate immunity and chronic intestinal inflammation. Front. Immunol. 2014, 5, 317. [Google Scholar] [CrossRef] [PubMed]
- Wen, H.; Gris, D.; Lei, Y.; Jha, S.; Zhang, L.; Huang, M.T.; Brickey, W.J.; Ting, J.P. Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 2011, 12, 408–415. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Jiang, W.; Spinetti, T.; Tardivel, A.; Castillo, R.; Bourquin, C.; Guarda, G.; Tian, Z.; Tschopp, J.; Zhou, R. Omega-3 fatty acids prevent inflammation and metabolic disorder through inhibition of NLRP3 inflammasome activation. Immunity 2013, 38, 1154–1163. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, C.; Cavalieri, D.; Di Paola, M.; Ramazzotti, M.; Poullet, J.B.; Massart, S.; Collini, S.; Pieraccini, G.; Lionetti, P. Impact of diet in shaping gut microbiota revealed by a comparative study in children from Europe and rural Africa. Proc. Natl. Acad. Sci. USA 2010, 107, 14691–14696. [Google Scholar] [CrossRef] [PubMed]
- Jansson, J.; Willing, B.; Lucio, M.; Fekete, A.; Dicksved, J.; Halfvarson, J.; Tysk, C.; Schmitt-Kopplin, P. Metabolomics reveals metabolic biomarkers of Crohn’s disease. PLoS ONE 2009, 4, e6386. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, S.; DeCoffe, D.; Brown, K.; Rajendiran, E.; Estaki, M.; Dai, C.; Yip, A.; Gibson, D.L. Fish oil attenuates omega-6 polyunsaturated fatty acid-induced dysbiosis and infectious colitis but impairs LPS dephosphorylation activity causing sepsis. PLoS ONE 2013, 8, e55468. [Google Scholar] [CrossRef] [PubMed]
- Piazzi, G.; D’Argenio, G.; Prossomariti, A.; Lembo, V.; Mazzone, G.; Candela, M.; Biagi, E.; Brigidi, P.; Vitaglione, P.; Fogliano, V.; et al. Eicosapentaenoic acid free fatty acid prevents and suppresses colonic neoplasia in colitis-associated colorectal cancer acting on Notch signaling and gut microbiota. Int. J. Cancer 2014, 135, 2004–2013. [Google Scholar] [CrossRef] [PubMed]
- Prossomariti, A.; Scaioli, E.; Piazzi, G.; Fazio, C.; Bellanova, M.; Biagi, E.; Candela, M.; Brigidi, P.; Consolandi, C.; Balbi, T.; et al. Short-term treatment with eicosapentaenoic acid improves inflammation and affects colonic differentiation markers and microbiota in patients with ulcerative colitis. Sci. Rep. 2017, 7, 7458. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Carrio, J.; Salazar, N.; Margolles, A.; González, S.; Gueimonde, M.; de Los Reyes-Gavilán, C.G.; Suárez, A. Free fatty acids profiles are related to gut microbiota signatures and short-chain fatty acids. Front. Immunol. 2017, 8, 823. [Google Scholar] [CrossRef] [PubMed]
- Watson, H.; Mitra, S.; Croden, F.C.; Taylor, M.; Wood, H.M.; Perry, S.L.; Spencer, J.A.; Quirke, P.; Toogood, G.J.; Lawton, C.L.; et al. A randomised trial of the effect of omega-3 polyunsaturated fatty acid supplements on the human intestinal microbiota. Gut 2017. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.K.; Maltz, B.E.; Coburn, L.A.; Slaughter, J.C.; Chaturvedi, R.; Schwartz, D.A.; Wilson, K.T. Increased serum levels of L-arginine in ulcerative colitis and correlation with disease severity. Inflamm. Bowel Dis. 2010, 16, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Beyazit, Y.; Purnak, T.; Kekilli, M. Role of nitric oxide in the treatment of non-alcoholic fatty liver by omega-3 fatty acids. Aliment. Pharmacol. Ther. 2010, 32, 303–304. [Google Scholar] [CrossRef] [PubMed]
- Middleton, S.J.; Naylor, S.; Woolner, J.; Hunter, J.O. A double-blind, randomized, placebo-controlled trial of essential fatty acid supplementation in the maintenance of remission of ulcerative colitis. Aliment. Pharmacol. Ther. 2002, 16, 1131–1135. [Google Scholar] [CrossRef] [PubMed]
- Best, W.R.; Becktel, J.M.; Singleton, J.W.; Kern, F. Development of a Crohn’s disease activity Index. Gastroenterolology 1976, 70, 439–444. [Google Scholar]
- Belluzzi, A. N-3 fatty acids for the treatment of inflammatory bowel diseases. Proc. Nutr. Soc. 2002, 61, 391–395. [Google Scholar] [CrossRef] [PubMed]
- Marion-Letellier, R.; Savoye, G.; Beck, P.L.; Panaccione, R.; Ghosh, S. Polyunsaturated fatty acids in inflammatory bowel diseases: A reappraisal of effects and therapeutic approaches. Inflamm. Bowel Dis. 2013, 19, 650–661. [Google Scholar] [CrossRef] [PubMed]
- Brignola, C.; Campieri, M.; Bazzocchi, G.; Farruggia, P.; Tragnone, A.; Lanfranchi, G.A. A laboratory index for predicting relapse in asymptomatic patients with Crohn’s disease. Gastroenterology 1986, 91, 1490–1494. [Google Scholar] [CrossRef]
- Belluzzi, A.; Brignola, C.; Campieri, M.; Pera, A.; Boschi, S.; Miglioli, M. Effect of an enteric—Coated fish oil preparation on relapses in Crohn’s disease. N. Engl. J. Med. 1996, 334, 1557–1560. [Google Scholar] [CrossRef] [PubMed]
- Feagan, B.G.; Sandborn, W.J.; Mittmann, U.; Bar-Meir, S.; D’Haens, G.; Bradette, M.; Cohen, A.; Dallaire, C.; Ponich, T.P.; McDonald, J.W.; et al. Omega-3 free fatty acids for the maintenance of remission in Crohn disease: The EPIC Randomized Controlled Trials. JAMA 2008, 299, 1690–1697. [Google Scholar] [CrossRef] [PubMed]
- Candy, S.; Wright, J.; Gerber, M.; Adams, G.; Gerig, M.; Goodman, R. A controlled double blind study of azathioprine in the management of Crohn’s disease. Gut 1995, 37, 674–678. [Google Scholar] [CrossRef] [PubMed]
- Romano, C.; Cucchiara, S.; Barabino, A.; Annese, V.; Sferlazzas, C. Usefulness of omega-3 fatty acid supplementation in addition to mesalazine in maintaining remission in pediatric Crohn’s disease: A double-blind; randomized; placebo-controlled study. World J. Gastroenterol. 2005, 11, 7118–7121. [Google Scholar] [CrossRef] [PubMed]
- Uchiyama, K.; Nakamura, M.; Odahara, S.; Koido, S.; Katahira, K.; Shiraishi, H.; Ohkusa, T.; Fujise, K.; Tajiri, H. N-3 polyunsaturated fatty acid diet therapy for patients with inflammatory bowel disease. Inflamm. Bowel Dis. 2010, 16, 1696–1707. [Google Scholar] [CrossRef] [PubMed]
- Scaioli, E.; Sartini, A.; Bellanova, M.; Campieri, M.; Festi, D.; Bazzoli, F.; Belluzzi, A. Eicosapentaenoic acid reduces fecal calprotectin and prevents relapse in ulcerative colitis patients: A placebo-controlled trial. Clin. Gastroenterol. Hepatol. 2017. under review. [Google Scholar]
- Sipponen, T.; Kolho, K.L. Fecal calprotectin in diagnosis and clinical assessment of inflammatory bowel disease. Scand. J. Gastroenterol. 2015, 50, 74–80. [Google Scholar] [CrossRef] [PubMed]
- Olsen, S.F.; Sørensen, J.D.; Secher, N.J.; Hedegaard, M.; Henriksen, T.B.; Hansen, H.S.; Grant, A. Randomised controlled trial of effect of fish-oil supplementation on pregnancy duration. Lancet 1992, 339, 1003–1007. [Google Scholar] [CrossRef]
- Schuchardt, J.P.; Hahn, A. Bioavailability of long-chain omega-3 fatty acids. PLEFA 2013, 89, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Lawson, L.D.; Hughes, B.G. Human absorption of fish oil fatty acids as triacylglycerols, free acids, or ethyl esters. Biochem. Biophys. Res. Commun. 1988, 152, 328–335. [Google Scholar] [CrossRef]
- Belluzzi, A.; Brignola, C.; Campieri, M.; Camporesi, E.P.; Gionchetti, P.; Rizzello, F.; Belloli, C.; De Simone, G.; Boschi, S.; Miglioli, M.; et al. Effects of new fish oil derivative on fatty acid phospholipid-membrane pattern in a group of Crohn’s disease patients. Dig. Dis. Sci. 1994, 39, 2589–2594. [Google Scholar] [CrossRef] [PubMed]
- Scaioli, E.; Cardamone, C.; Liverani, E.; Munarini, A.; Hull, M.A.; Belluzzi, A. The pharmacokinetic profile of a new gastroresistant capsule preparation of eicosapentaenoic acid as the free fatty acid. BioMed. Res. Int. 2015, 2015, 360825. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, M.; Hanley, M.P.; Mathias, G.; Sciavolino, F.; Hull, M.; Rosenberg, D.W. 652—A novel derivative of eicosapentaenoic acid reduces intestinal tumor burden in the APC Δ14/+ mouse model of familial adenomatous polyposis. Gastroenterology 2017, 152, S142. [Google Scholar] [CrossRef]
- Maki, K.C.; Bobotas, G.; Dicklin, M.R.; Huebner, M.; Keane, W.F. Effects of MAT9001 containing eicosapentaenoic acid and docosapentaenoic acid, compared to eicosapentaenoic acid ethyl esters, on triglycerides, lipoprotein cholesterol, and related variables. J. Clin. Lipidol. 2017, 11, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Vestland, T.L.; Asberg, A.; Klaveness, A.J.; Klaveness, J. Bioavailability of eicosapentaenoic acid and docosahexaenoic acid from omega-3 tablets. JSM Bioequiv. Bioavailab. 2017, 1, 1001. [Google Scholar]

| Study | Study Type—Duration | Number of Subjects-Inclusion Criteria | Age (years) | Supplementation | Placebo | Concurrent Medications | Outcomes | Results * |
|---|---|---|---|---|---|---|---|---|
| Hawthorne, 1992 | Double-blind, placebo-controlled, multicentre 1 year | 34 adults: 19 (n-3 PUFAs arm) 15 (placebo arm) In remission or while recovering from relapse No diet restrictions | 17–77 | n-3 PUFAs as 10 mL liquid form twice daily of HiEPA (5 g/day EPA + 1.2 g/day DHA, as triglyceride concentrate) | Olive oil, 10 mL twice daily | 5-ASA, steroids (≤20 mg/day) | Relapse rates (active symptoms and/or inflamed rectal mucosa) | 42% vs. 48%; (p = 0.54) |
| Time to relapse (days), median (IQR) | 365 (265–365) vs. 349 (240–365) (p = NS) | |||||||
| Loeschke, 1996 | Double-blind, placebo-controlled, multicenter2 years | 64 adults: 33 (n-3 PUFAs arm) 31 (placebo arm) In remission or low disease activity (Gomez score <8) | n-3 arm: 39 ± 11 # Placebo arm: 40 ± 13 # | Fish oil capsules, as long-chain ethyl esters n-3 PUFAs (5.1 g/day of total n-3 PUFAs; dose of EPA and DHA not reported) | Maize oil | 5-ASA (discontinued 3 months after randomisation) | Relapse rate (≥4-point increase in Gomez score) | 58% vs. 55%; (p = 0.81) |
| Mean clinical disease activity score at the third month of treatment | 2.2 vs. 4.4 (p < 0.05) | |||||||
| Mantzaris, 1996 | Double-blind, placebo-controlled,single-centre 1 year | 50 adults: 27 (n-3 PUFAs arm)23 (placebo arm) Clinical, endoscopic remissionNo diet restrictions | 17–65 | n-3 PUFAs as 10 mL liquid form twice daily of MaxEPA (3.2 g/day EPA + 2.1 g/day DHA, as triacylglycerol) | Olive oil, 10 mL twice daily5-ASA | 5-ASA (3.6 g/day) | Relapse rate (active symptoms or endoscopic signs of relapse) | 27% vs. 28% (p = 1.0) |
| Time to relapse (days), median (IQR) | 235 (100–365) vs. 218 (79–365) (p > 0.1) | |||||||
| Aslan, 1992 | Double-blind, placebo-controlled;crossover design 8 months (3 intervention period + 2 washout period) | 17 adultsMild to moderate disease activity | 31–74 | MaxEPA capsules (2.7 g/day EPA + 1.8 g/day DHA) | Corn oil (10.3 g OA + 2.1 g PA + 1.8 g LA) | Oral steroids (<20 mg/day) or SASP | Clinical response (decrease in DAI score after MaxEPA) | 56% vs. 4% p < 0.05 |
| Steroid-sparing effects during MaxEPA therapy | 72% reduced anti-inflammatory dosage and/or eliminated steroids (p value not reported) | |||||||
| Stenson, 1992 | Double-blind, placebo-controlled, randomised, multicentre; crossover design 5 months (4 intervention period + 1 washout period) | 24 adults Active disease | 25–62 | MaxEPA capsules (3.24 g/day EPA + 2.16 g/day DHA, as triacylglycerol) | Vegetable oil (12.36 g OA + 2.52 g PA + 2.16 g LA) | Prednisone, SASP | Endoscopic score improvement | n-3 PUFAs group: mean decrease of −2.09 (95% CI: −4.63 to 0.45; p = 0.06) Placebo group: mean decrease of −0.17 (95% CI: −1.75 to 1.41; p = 0.10) |
| Steroid-sparing effect | NS in both groups | |||||||
| Stack, 1997 | Double-blind, controlled, randomised; parallel design 4 months | 66 adults: 14 (n-3 PUFAs group) 13 (n-6 PUFAs group) 13 (n-3 and n-6 PUFAs group) 13 (placebo group) | n-3 PUFAs group: 1.5 g EPA | n-6 PUFAs group: 2.1 g GLA Placebo group: sunflower oil | Steroids | Steroid-sparing effect | No evidence with n-3 or n-6 PUFAs compared to placebo either alone or in combination (p value not reported) | |
| Almallah, 1998 | Double-blind, placebo-controlled, randomised, single-centre; parallel design 6 months | 18 adults: 9 (n-3 PUFAs arm) 9 (placebo arm) Low disease activity (Gomez score < 8) On Western diet | n-3 PUFAs arm: 29–64Placebo arm: 32–72 | n-3 PUFAs as 15 mL fish-oil extract (3.2 g/day EPA + 2.4 g/day DHA) | Sunflower oil, 15 ml (2.6 g/day oleic acid + 7.9 g/day linoleic acid) | SASP or 5-ASA | Clinical response | p < 0.05 |
| Endoscopic score improvement (RSS) | p = 0.013 (n-3 PUFAs arm) p = NS (placebo arm) | |||||||
| Histological score improvement | p = 0.016 | |||||||
| Dichi, 2000 | Randomised, single-centre; crossover design 6 months (2 intervention period + 2 washout period + 2 crossover intervention period) | 10 adults Mild to moderate disease activity | 33–65 | 5.4 g/day n-3 PUFAs fish-oil capsule, as fatty acids (180 mg EPA + 120 mg DHA in each capsule) | 2.2 g/day SASP | Laboratory blood parameters | ↑ of CRP, ERS, PLT during n-3 PUFAs treatment (p < 0.01) Changes during SASP therapy: p = NS | |
| Improvement in sigmoidoscopy score | At entry, mean 9.6 (SD ± 2.8) After n-3 PUFAs treatment, mean 5.0 (SD ± 5.7) p < 0.01 | |||||||
| Varghese, 2000 | Double-blind, randomised; parallel design 6 months | 51 adults: 21 (n-3 PUFAs arm) 30 (placebo arm) Active and extensive disease | Fish oil 5.6 mg/day | Sunflower oil | Clinical scores improvement | n-3 PUFAs arm: p = 0.001 (no quantitative data presented) | ||
| Endoscopic score improvement | n-3 PUFAs arm: p = 0.054 (no quantitative data presented) |
| Author | Study Type-Duration | Number of Subjects-Inclusion Criteria | Age (years) | Supplementation | Placebo | Concurrent Medications | Outcomes | Results (n-3 PUFAs Arm vs. Placebo arm) |
|---|---|---|---|---|---|---|---|---|
| Belluzzi, 1996 | Double-blind, placebo-controlled, single-centre 1 year | 78 adults: 39 (n-3 PUFAs arm) 39 (placebo arm) CDAI < 150 at baseline, but at high risk of relapse No diet restrictions | 18–67 | Enteric-coated, time-released, fish-oil capsule (1.8 g/day EPA + 0.9 g/day DHA, as FFAs) | Capsules 500 mg Miglyol 182 (caprylic acid + capric acid) | None | Relapse rate (CDAI >150 or ≥100 points increase in CDAI from baseline) | 11.28% vs. 27.69% (p < 0.001) |
| Time to first relapse | >1 year vs. 4 months (log rank test 0.006) | |||||||
| Adverse events | 10.3% vs. 2.6% (diarrhoea) | |||||||
| Lorenz-Meyer, 1996 | Double-blind, placebo-controlled, multicenter 1 year | 135 adults: 70 (n-3 PUFAs arm) 65 (placebo arm) CDAI < 150 at baseline (steroid-induced remission after an acute relapse) Diet low in AA and rich in fibre | 17–65 | Non-enteric-coated n-3 PUFAs capsules (3.3 g/day EPA + 1.8 g/day DHA, as ethyl ester) | Corn oil | Low-dose prednisolone in the first 8 weeks | Relapse rate (CDAI >200 or ≥60 points increase in CDAI from baseline, plus ≥2 SD above normal mean CRP increase | 57.00% vs. 36.55% (p = 0.84) |
| Time to first relapse | 159 days vs. 133 days (NS) | |||||||
| Adverse events | 1.4% vs. 1.5% (diarrhoea) 14.3% vs. 3.1% (halitosis) 14.3% vs. 3.1% (upper GI symptoms) | |||||||
| Belluzzi, 1997 | Double-blind, placebo-controlled, single-centre 1 year | 50 adults; 26 (n-3 PUFAs arm) 24 (placebo arm) CDAI <150 one month after ileal resection | Enteric-coated, time-released, fish-oil capsule (1.8 g/day EPA + 0.9 g/day DHA, as FFAs) | Capsules 500 mg Miglyol 182 (caprylic acid + capric acid) | None | Clinical relapse rate (CDAI >150 with an increase of >50 points from baseline) Histological relapse | 2.8% vs. 5.21% (p = 0.24) 9.34% vs. 15.62% (p = 0.09) | |
| Adverse events | None (personal communication) | |||||||
| Romano, 2005 | Double-blind, placebo-controlled, multicenter 1 year | 38 children: 18 (n-3 PUFAs arm) 20 (placebo arm) PCDAI <20 for at least 2 months at baseline | 5–16 | Enteric-coated n-3 PUFAs capsules (1.2 g/day EPA + 0.6 g/day DHA, as triglycerides) | Olive oil | Time-dependent 5-ASA (50 mg/kg/day) | Relapse rate (PCDAI > 20) | 61% vs. 95% (p < 0.001) |
| Time to first relapse | 8 months vs. 1 month | |||||||
| Adverse events | None | |||||||
| Feagan, 2008 (EPIC-1) | Double-blind, placebo-controlled, multicenter 52 weeks | 363 adults: 183 (n-3 PUFAs arm) 180 (placebo arm) CDAI <150 for at least 3 months but <12 months at baseline | n-3 PUFAs arm: 40.5 ±15.2 # Placebo arm: 38.2 ± 13.1 # | Enteric-coated, time-released n-3 PUFAs capsules (2.2 g/day EPA + 0.8 g/day DHA, as FFAs) * | 4 g/day medium-chain triglyceride oil | None | Relapse rate (CDAI >150 or >70 points increase from baseline) | 31.6% vs. 35.7% HR 0.82, 95% CI: 0.51–1.19 (p = 0.30) |
| Adverse events | 18.7% vs. 11.4% (diarrhoea)1% vs. 0.5% (dysgeusia) 9.1% vs. 2.2% (nausea) 14.4% vs. 6.5% (all upper GI symptoms) | |||||||
| Change in mean CDAI | NS | |||||||
| Change in SF-36 scores | NS | |||||||
| Feagan, 2008 (EPIC-2) | Double-blind, placebo-controlled, Multicenter 58 weeks | 375 adults: 187 (n-3 PUFAs arm) 188 (placebo arm) CDAI < 150 after 8 weeks of steroid-induced remission | n-3 PUFAs arm: 38.5 ±13.8 # Placebo arm: 40 ± 13.6 # | Enteric-coated, time-released n-3 PUFAs capsules (2.2 g/day EPA + 0.8 g/day DHA as FFAs) * | Four 1 g of medium-chain triglyceride oil | Prednisone 20 mg/day or budesonide 6 mg/day tapered off over 8 weeks from randomisation | Relapse rate (CDAI >150 or >70 points increase from baseline) | 47.8% vs. 48.8% HR 0.90, 95% CI: 0.67–1.21 (p = 0.48) |
| Adverse events | 23.2% vs. 19.7% (diarrhoea) 5.3% vs. 1.1% (dysgeusia) 15.9% vs. 10.1% (nausea) 35.4% vs. 23.9% (all upper GI symptoms) | |||||||
| Change in mean CDAI | NS | |||||||
| Change in SF-36 scores | NS |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Scaioli, E.; Liverani, E.; Belluzzi, A. The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A Comprehensive Review and Future Therapeutic Perspectives. Int. J. Mol. Sci. 2017, 18, 2619. https://doi.org/10.3390/ijms18122619
Scaioli E, Liverani E, Belluzzi A. The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A Comprehensive Review and Future Therapeutic Perspectives. International Journal of Molecular Sciences. 2017; 18(12):2619. https://doi.org/10.3390/ijms18122619
Chicago/Turabian StyleScaioli, Eleonora, Elisa Liverani, and Andrea Belluzzi. 2017. "The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A Comprehensive Review and Future Therapeutic Perspectives" International Journal of Molecular Sciences 18, no. 12: 2619. https://doi.org/10.3390/ijms18122619
APA StyleScaioli, E., Liverani, E., & Belluzzi, A. (2017). The Imbalance between n-6/n-3 Polyunsaturated Fatty Acids and Inflammatory Bowel Disease: A Comprehensive Review and Future Therapeutic Perspectives. International Journal of Molecular Sciences, 18(12), 2619. https://doi.org/10.3390/ijms18122619




