The Inhibitory Effects of Cobalt Protoporphyrin IX and Cannabinoid 2 Receptor Agonists in Type 2 Diabetic Mice
Abstract
:1. Introduction
2. Results
2.1. Diabetic Neuropathy
2.2. Effects of CoPP on Mechanical Allodynia
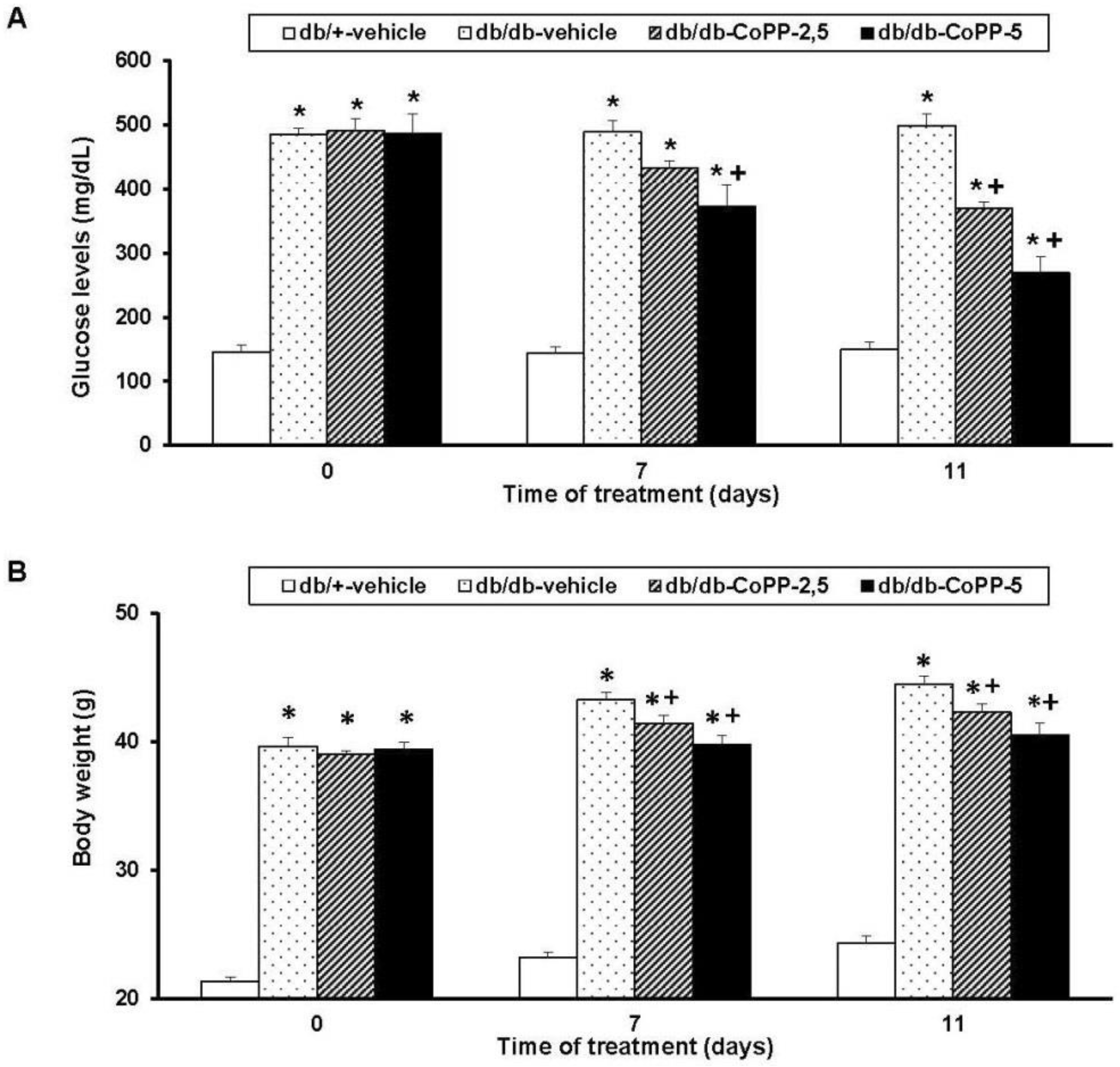
2.3. Effects of CoPP on Blood Glucose Levels and Body Weight
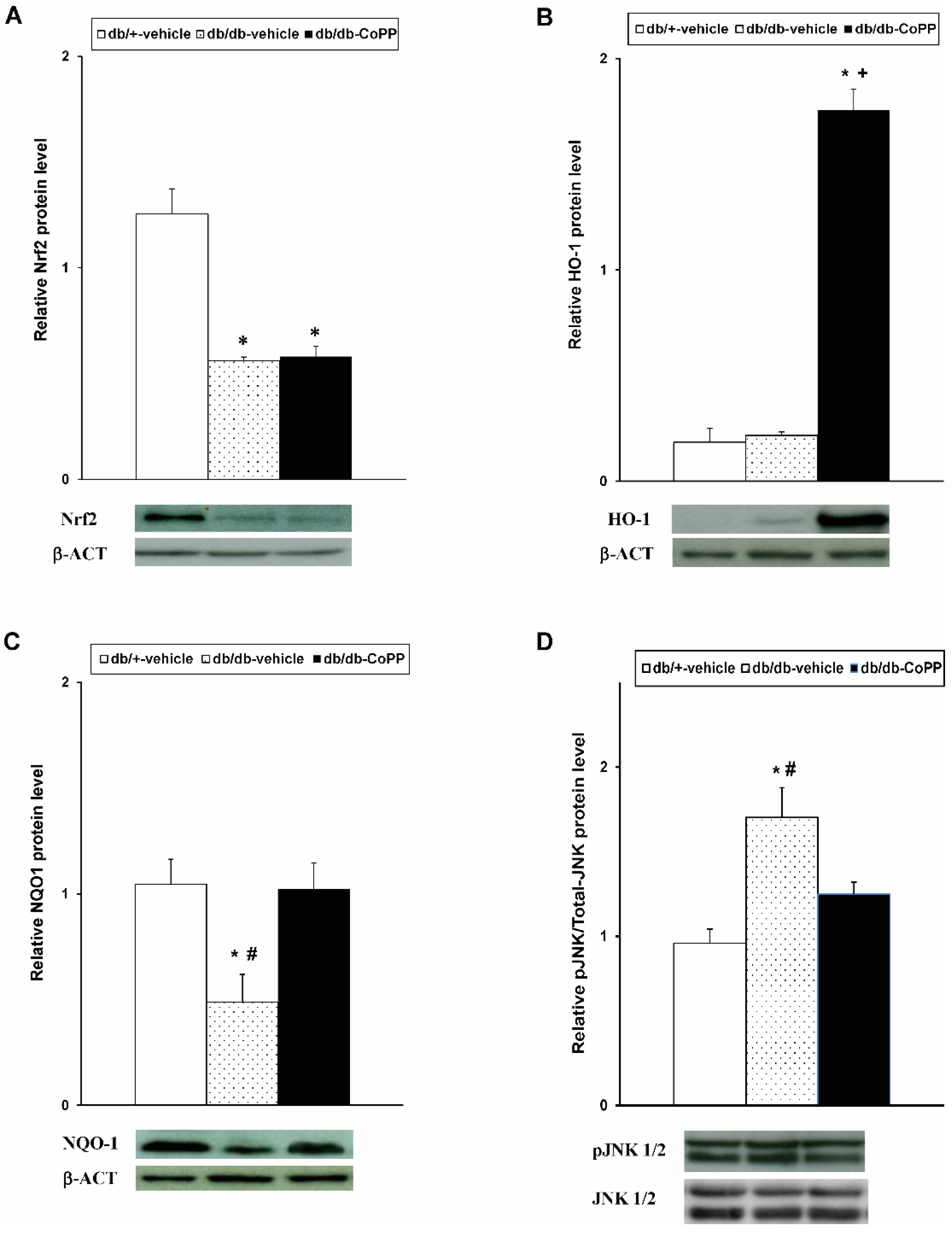
2.4. Effects of CoPP on Nrf2, HO-1, NQO1 and JNK Expression in Sciatic Nerve
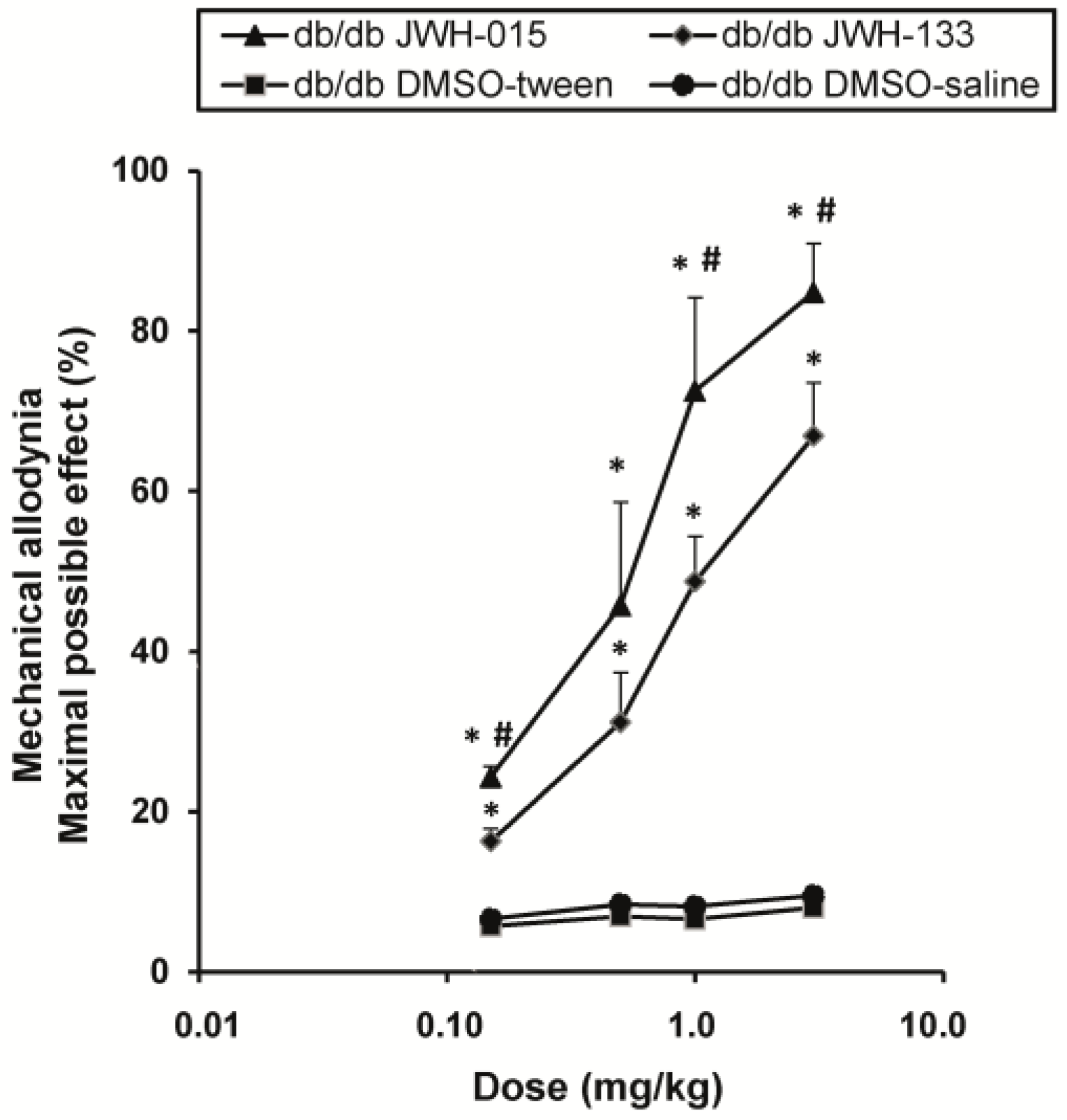
2.5. Effects of JWH-015 and JWH-133 on the Mechanical Allodynia
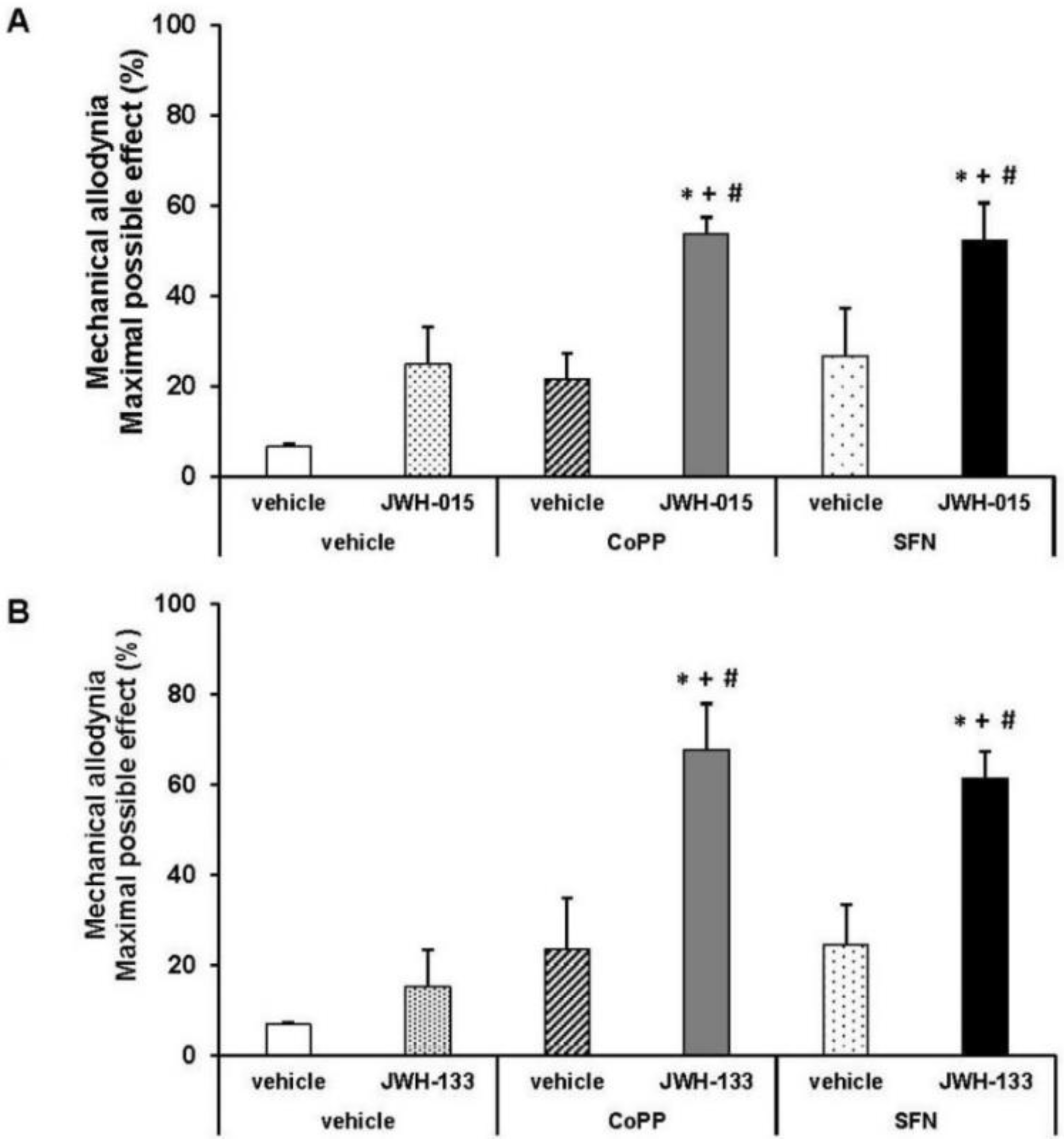
2.6. Effects of CoPP and SFN Treatments on the Antiallodynic Effects of JWH-015 and JWH-133
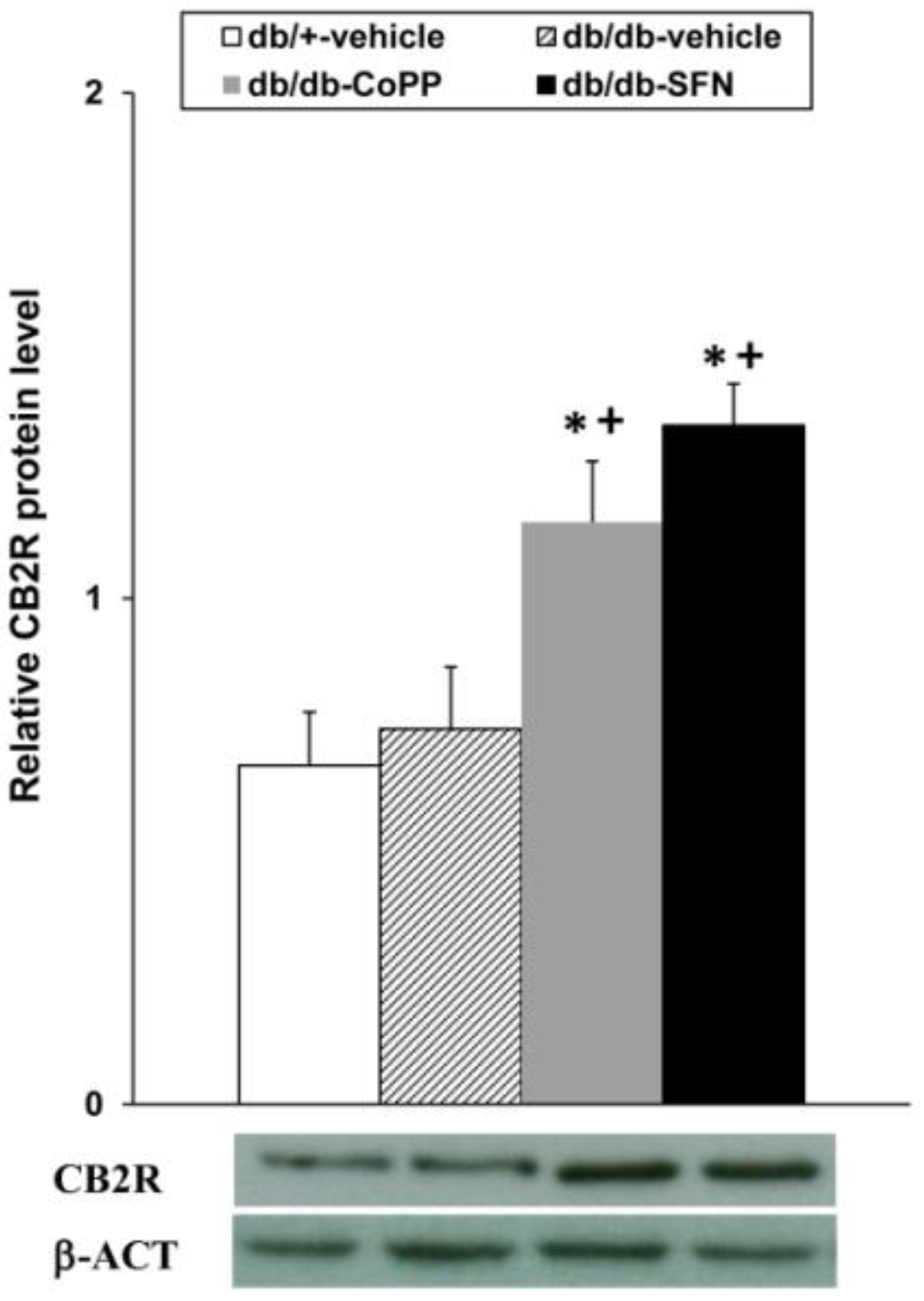
2.7. Effects of CoPP and SFN Treatments on CB2R Levels in the Dorsal Root Ganglia of db/db Mice
3. Discussion
4. Material and Methods
4.1. Animals
4.2. Nociceptive Behavioral Test
4.3. Western Blot Analysis
4.4. Experimental Procedure
4.5. Drugs
4.6. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Callaghan, B.; Cheng, H.; Stables, C.; Smith, A.; Feldman, E. Diabetic neuropathy: Clinical manifestations and current treatments. Lancet Neurol. 2012, 11, 521–534. [Google Scholar] [CrossRef]
- Schreiber, A.; Nones, C.; Reis, R.; Chichorro, J.; Cunha, J. Diabetic neuropathic pain: Physiopathology and treatment. World J. Diabetes 2015, 6, 432. [Google Scholar] [CrossRef] [PubMed]
- Peltier, A.; Goutman, S.A.; Callaghan, B.C. Painful diabetic neuropathy. BMJ 2014, 348, g1799. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Kishore, L.; Kaur, N. Diabetic peripheral neuropathy: Current perspective and future directions. Pharmacol. Res. 2014, 80, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Abraham, N.G.; Junge, J.M.; Drummond, G.S. Translational Significance of Heme Oxygenase in Obesity and Metabolic Syndrome. Trends Pharmacol. Sci. 2016, 37, 17–36. [Google Scholar] [CrossRef] [PubMed]
- Negi, G.; Nakkina, V.; Kamble, P.; Sharma, S.S. Heme oxygenase-1, a novel target for the treatment of diabetic complications: Focus on diabetic peripheral neuropathy. Pharmacol. Res. 2015, 102, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Hervera, A.; Leánez, S.; Negrete, R.; Motterlini, R.; Pol, O. Carbon monoxide reduces neuropathic pain and spinal microglial activation by inhibiting nitric oxide synthesis in mice. PLoS ONE 2012, 7, e43693. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hervera, A.; Gou, G.; Leánez, S.; Pol, O. Effects of treatment with a carbon monoxide-releasing molecule and a heme oxygenase 1 inducer in the antinociceptive effects of morphine in different models of acute and chronic pain in mice. Psychopharmacology 2013, 228, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Negrete, R.; Hervera, A.; Leánez, S.; Pol, O. Treatment with a carbon monoxide-releasing molecule inhibits chronic inflammatory pain in mice: Nitric oxide contribution. Psychopharmacology 2014, 231, 853–861. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.; Zhang, Z.J.; Zhu, M.D.; Jiang, B.C.; Yang, T.; Gao, Y.J. Exogenous induction of HO-1 alleviates vincristine-induced neuropathic pain by reducing spinal glial activation in mice. Neurobiol. Dis. 2015, 79, 100–110. [Google Scholar] [CrossRef] [PubMed]
- Castany, S.; Carcolé, M.; Leánez, S.; Pol, O. The induction of heme oxygenase 1 decreases painful diabetic neuropathy and enhances the antinociceptive effects of morphine in diabetic mice. PLoS ONE 2016, 11, e0146427. [Google Scholar] [CrossRef] [PubMed]
- Patrick, B.A.; Das, A.; Jaiswal, A.K. NAD(P)H:quinone oxidoreductase 1 protects bladder epithelium against painful bladder syndrome in mice. Free Radic. Biol. Med. 2012, 53, 1886–1893. [Google Scholar] [CrossRef] [PubMed]
- Negi, G.; Kumar, A.; Sharma, S.S. Nrf2 and NF-κB modulation by sulforaphane counteracts multiple manifestations of diabetic neuropathy in rats and high glucose-induced changes. Curr. Neurovasc. Res. 2011, 8, 294–304. [Google Scholar] [CrossRef] [PubMed]
- Purves, T.; Middlemas, A.; Agthong, S.; Jude, E.B.; Boulton, A.J.; Fernyhough, P.; Tomlinson, D.R. A role for mitogen-activated protein kinases in the etiology of diabetic neuropathy. FASEB J. 2001, 15, 2508–2514. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.T.; Dauch, J.R.; Oh, S.S.; Hayes, J.M.; Hong, Y.; Feldman, E.L. p38 mediates mechanical allodynia in a mouse model of type 2 diabetes. Mol. Pain 2010, 6, 28. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Chen, H.; Ling, B.Y.; Xu, L.; Cao, H.; Zhang, Y.Q. Extracellular signal-regulated protein kinase activation in spinal cord contributes to pain hypersensitivity in a mouse model of type 2 diabetes. Neurosci. Bull. 2014, 30, 53–66. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.J.; Ji, R.R. Activation of JNK pathway in persistent pain. Neurosci. Lett. 2008, 437, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Ji, R.R.; Gereau, R.W.; Malcangio, M.; Strichartz, G.R. MAP kinase and pain. Brain Res. Rev. 2009, 60, 135–148. [Google Scholar] [CrossRef] [PubMed]
- Grote, C.W.; Groover, A.L.; Ryals, J.M.; Geiger, P.C.; Feldman, E.L.; Wright, D.E. Peripheral nervous system insulin resistance in ob/ob mice. Acta Neuropathol. Commun. 2013, 1, 15. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, C.; Leánez, S.; Pol, O. The induction of the transcription factor Nrf2 enhances the antinociceptive effects of delta-opioid receptors in diabetic mice. PLoS ONE, 2017, 12, e0180998. [Google Scholar] [CrossRef] [PubMed]
- Castany, S.; Carcolé, M.; Leánez, S.; Pol, O. The role of carbon monoxide on the anti-nociceptive effects and expression of cannabinoid 2 receptors during painful diabetic neuropathy in mice. Psychopharmacology 2016, 233, 2209–2219. [Google Scholar] [CrossRef] [PubMed]
- Ulugol, A.; Karadag, H.C.; Ipci, Y.; Tamer, M.; Dokmeci, I. The effect of WIN 55,212–2, a cannabinoid agonist, on tactile allodynia in diabetic rats. Neurosci. Lett. 2004, 371, 167–170. [Google Scholar] [CrossRef] [PubMed]
- Vera, G.; López-Miranda, V.; Herradón, E.; Martín, M.I.; Abalo, R. Characterization of cannabinoid-induced relief of neuropathic pain in rat models of type 1 and type 2 diabetes. Pharmacol. Biochem. Behav. 2012, 102, 335–343. [Google Scholar] [CrossRef] [PubMed]
- Akamine, T.; Koyanagi, S.; Kusunose, N.; Hashimoto, H.; Taniguchi, M.; Matsunaga, N.; Ohdo, S. Dosing time-dependent changes in the analgesic effect of pregabalin on diabetic neuropathy in mice. J. Pharmacol. Exp. Ther. 2015, 354, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Saito, T.; Hasegawa-Moriyama, M.; Kurimoto, T.; Yamada, T.; Inada, E.; Kanmura, Y. Resolution of Inflammation by Resolvin D1 Is essential for peroxisome proliferator-activated receptor-γ-mediated analgesia during postincisional pain development in type 2 diabetes. Anesthesiology 2015, 123, 1420–1434. [Google Scholar] [CrossRef] [PubMed]
- Paine, A.; Eiz-Vesper, B.; Blasczyk, R.; Immenschuh, S. Signaling to heme oxygenase-1 and its anti-inflammatory therapeutic potential. Biochem. Pharmacol. 2010, 80, 1895–1903. [Google Scholar] [CrossRef] [PubMed]
- Carcolé, M.; Castany, S.; Leánez, S.; Pol, O. Treatment with a heme oxygenase 1 inducer enhances the antinociceptive effects of µ-opioid, δ-opioid, and cannabinoid 2 receptors during inflammatory pain. J. Pharmacol. Exp. Ther. 2014, 351, 224–232. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Kim, D.H.; Tsenovoy, P.L.; Peterson, S.J.; Rezzani, R.; Rodella, L.F.; Rodella, L.F.; Aronow, W.S.; Ikehara, S.; Abraham, N.G. Treatment of obese diabetic mice with a heme oxygenase inducer reduces visceral and subcutaneous adiposity, increases adiponectin levels, and improves insulin sensitivity and glucose tolerance. Diabetes 2008, 57, 1526–1535. [Google Scholar] [CrossRef] [PubMed]
- Uruno, A.; Furusawa, Y.; Yagishita, Y.; Fukutomi, T.; Muramatsu, H.; Negishi, T.; Sugawara, A.; Kensler, T.W.; Yamamoto, M. The Keap1-Nrf2 system prevents onset of diabetes mellitus. Mol. Cell. Biol. 2013, 33, 2996–3010. [Google Scholar] [CrossRef] [PubMed]
- Ndisang, J.F.; Jadhav, A.; Mishra, M. The heme oxygenase system suppresses perirenal visceral adiposity, abates renal inflammation and ameliorates diabetic nephropathy in Zucker diabetic fatty rats. PLoS ONE, 2014, 9, e87936. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.; Wakabayashi, N.; Misra, V.; Biswal, S.; Lee, G.H.; Agoston, E.S.; Yamamoto, M.; Kensler, T.W. NRF2 modulates aryl hydrocarbon receptor signaling: Influence on adipogenesis. Mol. Cell. Biol. 2007, 27, 7188–7197. [Google Scholar] [CrossRef] [PubMed]
- Nicolai, A.; Li, M.; Kim, D.H.; Peterson, S.J.; Vanella, L.; Positano, V.; Gastaldelli, A.; Rezzani, R.; Rodella, L.F.; Drummond, G.; et al. Heme oxygenase-1 induction remodels adipose tissue and improves insulin sensitivity in obesity-induced diabetic rats. Hypertension 2009, 53, 508–515. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Peterson, S.; Husney, D.; Inaba, M.; Guo, K.; Kappas, A.; Ikehara, S.; Abraham, N.G. Long-lasting expression of HO-1 delays progression of type I diabetes in NOD mice. Cell Cycle 2007, 6, 567–571. [Google Scholar] [CrossRef] [PubMed]
- Kong, Q.; Liu, K.; Wu, L.; Wang, L. Heme oxygenase-1 inhibits neuropathic pain in rats with diabetic mellitus. Neural Regen. Res. 2012, 7, 2305–2311. [Google Scholar] [PubMed]
- Yeo, S.H.; Noh, J.R.; Kim, Y.H.; Gang, G.T.; Kim, S.W.; Kim, K.S.; Hwang, J.H.; Shong, M.; Lee, C.H. Increased vulnerability to β-cell destruction and diabetes in mice lacking NAD(P)H: Quinone oxidoreductase 1. Toxicol. Lett. 2013, 219, 35–41. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Zhou, S. Inflammation: Therapeutic targets for diabetic neuropathy. Mol. Neurobiol. 2014, 49, 536–546. [Google Scholar] [CrossRef] [PubMed]
- Ndisang, JF. Role of heme oxygenase in inflammation, insulin-signalling, diabetes and obesity. Mediat. Inflamm. 2010, 2010, 359732. [Google Scholar] [CrossRef] [PubMed]
- Hervera, A.; Negrete, R.; Leánez, S.; Martín-Campos, J.; Pol, O. The role of nitric oxide in the local antiallodynic and antihyperalgesic effects and expression of delta-opioid and cannabinoid-2 receptors during neuropathic pain in mice. J. Pharmacol. Exp. Ther. 2010, 334, 887–896. [Google Scholar] [CrossRef] [PubMed]
- Vincenzi, F.; Targa, M.; Corciulo, C.; Tabrizi, M.A.; Merighi, S.; Gessi, S.; Saponaro, G.; Baraldi, P.G.; Borea, P.A.; Varani, K. Antinociceptive effects of the selective CB2 agonist MT178 in inflammatory and chronic rodent pain models. Pain, 2013, 154, 864–873. [Google Scholar] [CrossRef] [PubMed]
- Gruden, G.; Barutta, F.; Kunos, G.; Pacher, P. Role of the endocannabinoid system in diabetes and diabetic complications. Br. J. Pharmacol. 2016, 173, 1116–1127. [Google Scholar] [CrossRef] [PubMed]
- Romero-Sandoval, E.A.; Asbill, S.; Paige, C.A.; Byrd-Glover, K. Peripherally Restricted Cannabinoids for the Treatment of Pain. Pharmacotherapy 2015, 35, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Castany, S.; Carcolé, M.; Leánez, S.; Pol, O. The antinociceptive effects of a δ-opioid receptor agonist in mice with painful diabetic neuropathy: Involvement of heme oxygenase 1. Neurosci. Lett. 2016, 614, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Chaplan, S.R.; Bach, F.W.; Pogrel, J.W.; Chung, J.M.; Yaksh, T.L. Quantitative assessment of tactile allodynia in the rat paw. J. Neurosci. Methods 1998, 453, 55–63. [Google Scholar] [CrossRef]

| Treatments | von Frey Filaments Strength (g) | ||
|---|---|---|---|
| vehicle + vehicle | 1.7 | ± | 0.1 |
| JWH-015 + vehicle | 2.8 | ± | 0.1 * |
| JWH-133 + vehicle | 2.6 | ± | 0.1 * |
| vehicle + AM630 | 1.6 | ± | 0.1 |
| JWH-015 + AM630 | 1.7 | ± | 0.1 |
| JWH-133 + AM630 | 1.7 | ± | 0.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McDonnell, C.; Leánez, S.; Pol, O. The Inhibitory Effects of Cobalt Protoporphyrin IX and Cannabinoid 2 Receptor Agonists in Type 2 Diabetic Mice. Int. J. Mol. Sci. 2017, 18, 2268. https://doi.org/10.3390/ijms18112268
McDonnell C, Leánez S, Pol O. The Inhibitory Effects of Cobalt Protoporphyrin IX and Cannabinoid 2 Receptor Agonists in Type 2 Diabetic Mice. International Journal of Molecular Sciences. 2017; 18(11):2268. https://doi.org/10.3390/ijms18112268
Chicago/Turabian StyleMcDonnell, Christina, Sergi Leánez, and Olga Pol. 2017. "The Inhibitory Effects of Cobalt Protoporphyrin IX and Cannabinoid 2 Receptor Agonists in Type 2 Diabetic Mice" International Journal of Molecular Sciences 18, no. 11: 2268. https://doi.org/10.3390/ijms18112268
APA StyleMcDonnell, C., Leánez, S., & Pol, O. (2017). The Inhibitory Effects of Cobalt Protoporphyrin IX and Cannabinoid 2 Receptor Agonists in Type 2 Diabetic Mice. International Journal of Molecular Sciences, 18(11), 2268. https://doi.org/10.3390/ijms18112268






