Abstract
Recent years have seen considerable progress in explaining the mechanisms of the pathogenesis of psoriasis, with a significant role played in it by the hyper-reactivity of Th1 and Th17 cells, Treg function disorder, as well as complex relationships between immune cells, keratinocytes, and vascular endothelium. The effect of stem cells in the epidermis and stem cells on T cells has been identified and the dysfunction of various types of stem cells may be a prime cause of dysregulation of the inflammatory response in psoriasis. However, exploring these mechanisms in detail could provide a chance to develop new therapeutic strategies. In this paper, the authors reviewed data on the role played by stem cells in the pathogenesis of psoriasis and initial attempts at using them in treatment.
1. Introduction
Psoriasis is a chronic disease affecting over 125 million people all over the world and an increasing trend has been observed [1,2]. As a systemic inflammatory process, it predisposes, twice as frequently than in the general population, to the development of metabolic disorders (insulin resistance, atherogenic dyslipidemia, arterial hypertension, and cardio-vascular diseases) and, consequently, to life shortening [3]. Moreover, this disease decreases the quality of life of the affected and leads to withdrawal from social life and development of depressive disorders. Patients with psoriasis present a significantly higher likelihood of suicidal thoughts, suicide attempts, and completed suicides [4]. Psoriasis constitutes a serious socioeconomic burden not only for patients but also for health care systems [5].
Unfortunately there is no fully satisfactory therapy against psoriasis and patients frequently report dissatisfaction with the treatment [6]. Although there has been some progress due to the introduction of biological therapies, it is still necessary to seek new therapeutic methods.
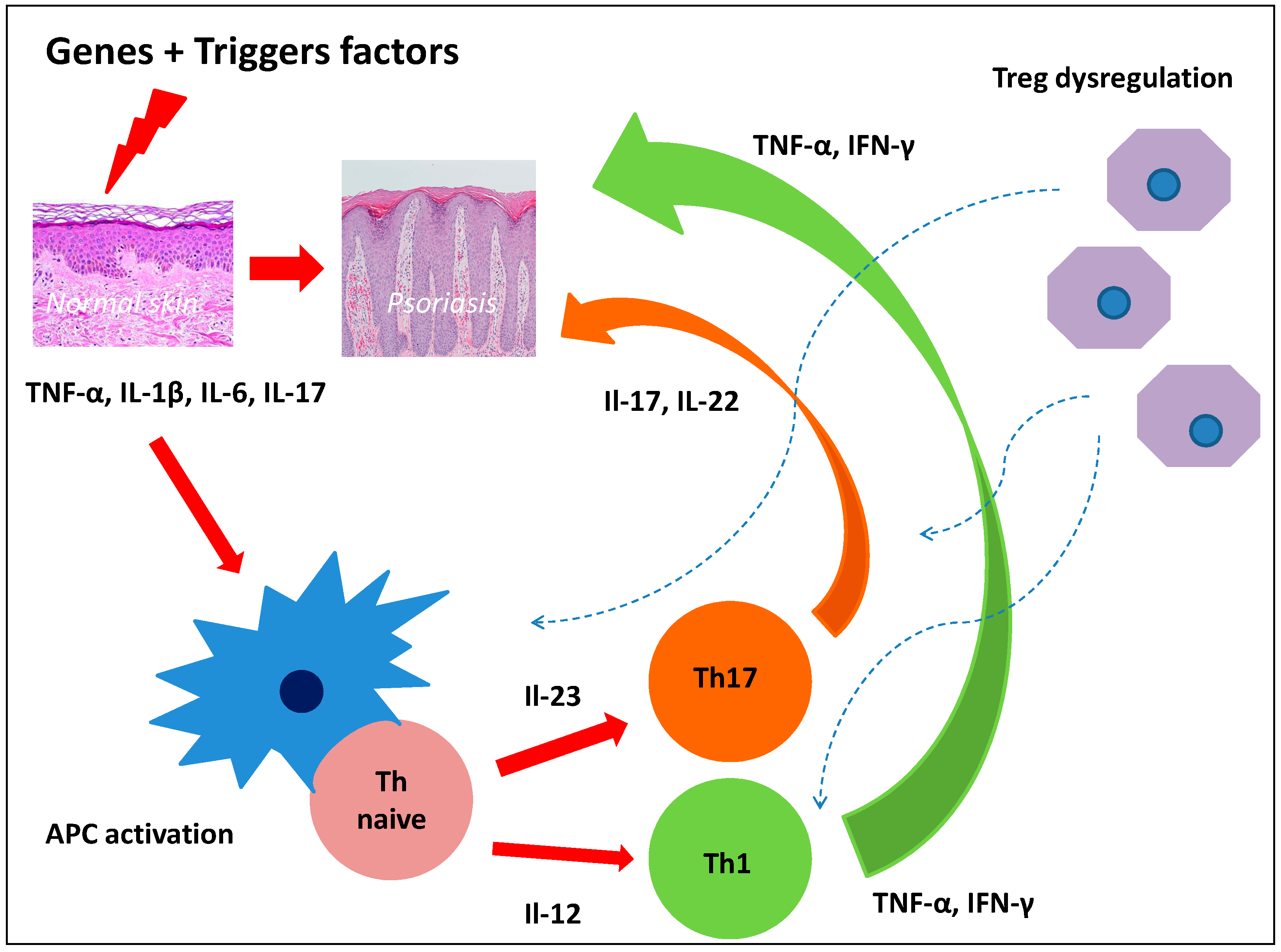
Although recent years have seen considerable progress in discovering the mechanisms of psoriasis pathogenesis, its full elucidation is still a long way away. Hyper-reactivity of Th1, Th17, dysregulation of Treg, and the complex relationships between immune system cells and keratinocytes and vascular endothelium obviously play a significant role [7]. Interleukin-23/Th17/Il-17 axis and Th1/IFN-γ axis play a key role in psoriasis inflammation [8]. Currently, attention is drawn to the effect of epidermal stem cells and stem cells on T cells. Therefore, the dysfunction of certain types of stem cells could be the root cause of dysregulation of the inflammatory response in psoriasis (Figure 1).
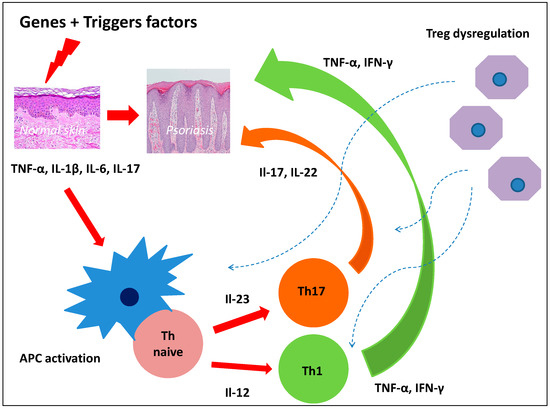
Figure 1.
Dysregulation of the inflammatory response in psoriasis.
2. Pathogenesis of Psoriasis and Dermal Stem Cells
2.1. Epidermal Stem Cells in Psoriasis
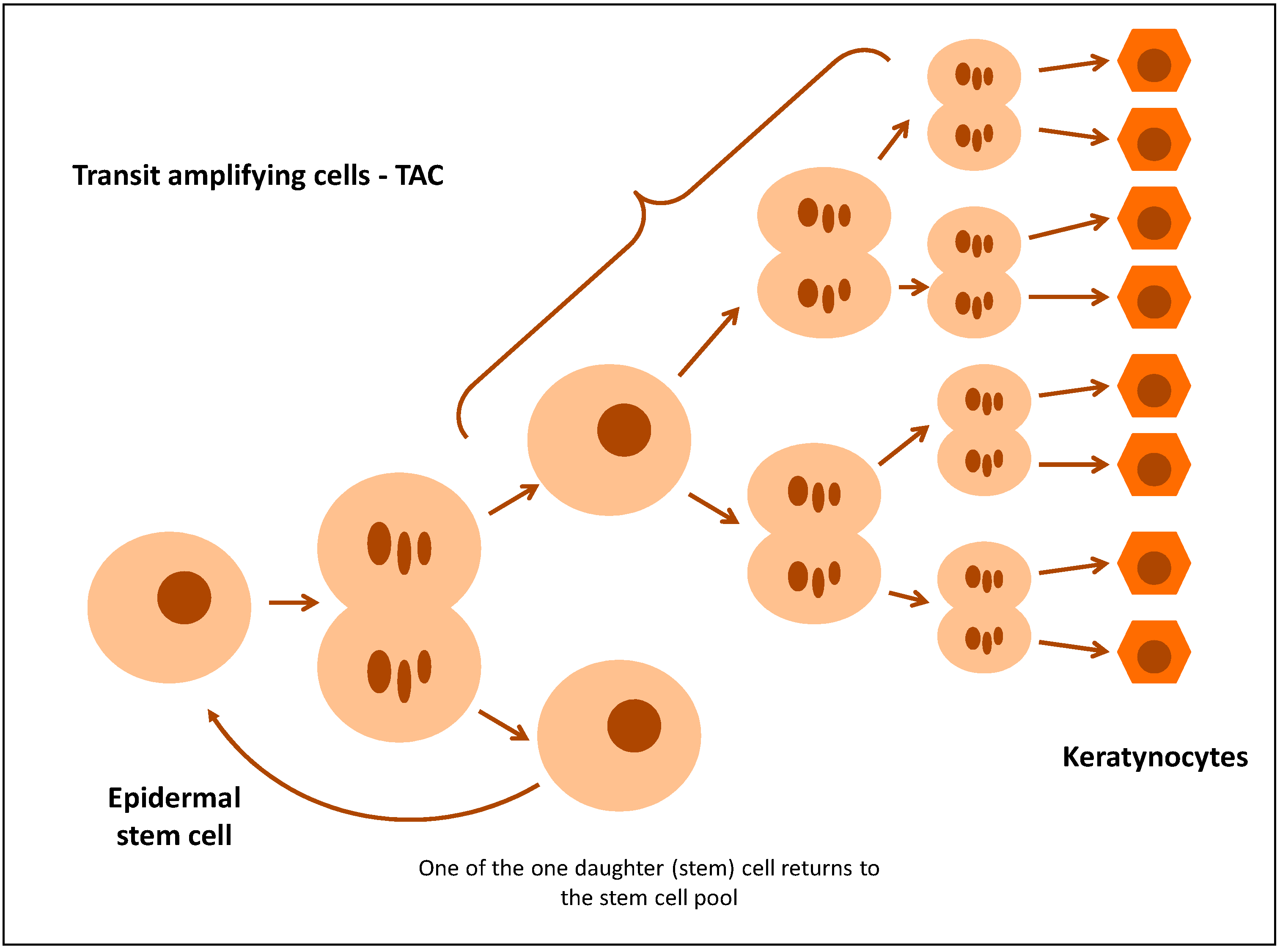
Maintaining the right homeostasis in the epidermis requires constant recreation of the pool of keratinocytes. This is possible owing to the existence of stem cells (SC) and transit-amplifying cells (TAC) formed from them [9,10]. According to the classic stem cell division theory, it divides by asymmetric mitosis, yielding one daughter (stem) cell and one TAC. A TAC has limited potential for proliferation, as it divides a specific number of times before it is ultimately differentiated (Figure 2) [9,11,12]. TAC markers include nestin and E-FABP (Epidermal fatty acid-binding protein) and, to a lesser extent, β1-integrin, whose expression in normal epidermis is detected in the basal layer. It has been proposed that the integrin β1 expression level on the cell surface of basal keratinocytes is a key determinant which controls the transition of cells from the basal layer to the suprabasal layer [10,13]. Stem cells in which the level of expression of integrin β1 is higher than in TAC cells adhere strongly to the basal membrane, whereas TAC cells decrease the expression of integrin β1, which deprives them of adhesion capability. TAC cells with an integrin β1 deficit leave the basal layer and enter the suprabasal layer, while at the same time turning K15 transcription off and K10 transcription on [9,14]. This indicates that hyperproliferation of the epidermis in psoriasis is caused by an increase in the number of TAC cells [15].
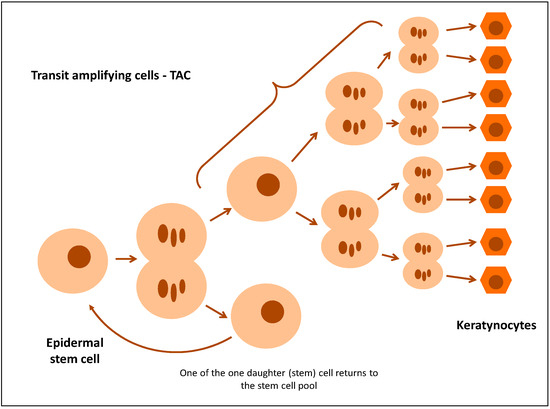
Figure 2.
Regeneration of epidermis and the role of transit-amplifying cells.
An expanded suprabasal layer appears in psoriasis, which is caused by an increased population of transgenic progenitors [16]. According to Jia et al., excessive expansion of TAC cells is observed in the psoriatic epidermis, which exhausts the stem cell pool to the extent that K15-positive cells were barely detectable in the psoriatic basal layer [9]. The balance between the symmetric and asymmetric divisions of stem cells is the key to tissue homeostasis. Jia et al. suggest that a higher percentage of both symmetric and asymmetric cell division is observed in psoriasis, which is a mechanism responsible for acanthosis [9]. However, Charruyer et al. discovered that the number of symmetrical divisions increases in planoepithelial cancer and the number of asymmetrical divisions increases in psoriasis hyperproliferation (human and murine). Moreover, researchers discovered that IL-17A increases the number of asymmetrical divisions in human and murine keratinocytes [16]. On the other hand, Jia et al. proved that IL-17A and IL-22 inhibit differentiation of keratinocytes in order to maintain the stem cell phenotype and control the time of transition of cells from the basal layer to the suprabasal layer (an increase in the expression of K15 and integrin β1, markers of stem cells) [9].
E-FABP (interchangeably referred to as FABP5) is included in the FABP family of cytoplasmic proteins that bind long-chain fatty acids and other hydrophobic ligands. Their role also includes capture, transport, and metabolism of fatty acids. They participate not only in metabolic processes, but also act as signal molecules, thereby regulating inflammatory pathways [15,17]. Disorders of the E-FABP encoding gene were first identified in psoriasis. E-FABP regulates differentiation of keratinocytes, but the mechanism is not fully clear [18,19]. Moreover, it is known that E-FABP forms a complex with psoriasin (also known as S100A7). The protein also inhibits the activity of tyrosinase in normal melanocytes and melanoma cells, which reduces their proliferation [20]. Moreover, expression of E-FABP-naive T CD4+ cells promotes maturation of the Th17 line and inhibits Treg. This results in increased production of IL-17, which directly stimulates hyperproliferation of keratinocytes [18]. Blocking E-FABP may be a new therapeutic goal in psoriasis, preventing the development of metabolic syndrome in patients.
Yamamoto et al. examined the activity of the stem cell factor (SCF), which is also a mast cell growth factor. An immunohistochemical examination demonstrated the presence of SCF in keratinocytes of psoriatic plaques, endothelium cells, and fibroblasts [21]. The serum levels of SCF were determined by ELISA. They were found to be elevated in patients with psoriasis compared to a healthy population. However, no correlation with the severity of the disease (PASI) was observed. SCF, released by keratinocytes, may cause the number of mast-cells in psoriatic eruptions to increase and induce pruritus within the eruptions [21].
2.2. Mesenchymal Stem Cells (MSC) in Psoriasis
Mesenchymal stem cells (MSC) in psoriatic plaques have an abnormal profile of cytokines that they secrete, which can influence the keratinocytes stimulating their proliferation and limiting their capability of apoptosis [22]. Liu et al. confirmed the influence of MSC from lesions on proliferation and apoptosis of HaCaT-cells (aneuploid immortal keratinocyte cell line from adult human skin). MSC exhibited similar expression of CD29, CD44, CD73, CD90, and CD105 to that in healthy skin, but with limited expression of CD34, CD45, and HLA-DR. This may explain their role in stimulation of the proliferation of psoriatic keratinocytes, which leads to acanthosis [22].
On the other hand, Orciani et al. assessed the basic functions of mesenchymal stem cells of skin, isolated from psoriatic patients: expression of inducible nitric oxide synthase (iNOS) and vascular endothelial growth factor (VEGF). They then compared them with the cells isolated from the skin of healthy people. The content of VEGF was the highest in MSC from the areas surrounding the psoriatic eruptions and that of iNOS—from lesions [23]. The secretion of these factors normalised after treatment with TNF-α inhibitors (etanercept, adalimumab) [24].
Similar conclusions were published by Hou et al. who found that the expression of genes associated with inflammation and angiogenesis, i.e., LITAF (lipopolysaccharide-induced tumor necrosis factor-alpha transcription factor), DUSP1 (dual-specificity protein phosphatase 1), VEGFα (vascular endothelial growth factor α), and IGFBP5 (insulin-like growth factor-binding protein-5) is increased in mesenchymal stem cells of the skin in psoriatic patients [25].
Recently, the authors compared the expression profile of miRNA in skin MSC in psoriatic patients and in healthy people by means of a micromatrix. Expression of pro-inflammatory miRNA miR-155 was found to be strongly increased in MCS in psoriatic patients [26]. As reported earlier by Xu et al., expression of mir-155 in psoriasis can be induced by proinflammatory cytokines, i.e., IFN-γ, TNF-α, or IL-1 [27]. Expression of mi-155 in MSC, DC cells, macrophages, and NK cells results in impaired immunosuppression, apoptosis of DC and an increase in the number of proinflammatory cytokines [28,29,30].
Additionally, miR-155 indirectly inhibits iNOS by inhibiting its target gene TAB2 [27]. This study showed that an increased level of mir-155 and decreased expression of iNOS in MSC in psoriatic patients can affect expression of cytokines and functions of immunoregulation in MSC. It was found that although expression of TAB2 and iNOS was low in MCS in psoriatic patients, expression of TGF-β and IL-10 (factors important for the function of MCS) was not significantly different in both groups, which suggests that MSC dysfunction in psoriasis is not induced by IGF-β and LI-10, but by mir-155 and iNOS [27].
MSC have immunosuppressive properties; they inhibit proliferation of activated T and B cells and cytotoxic activity of NK cells. Moreover, they change the profile of cytokines and chemokines secreted by dendritic cells and macrophages [31,32]. Psoriatic MSC have a decreased immunosuppressive capability. Liu et al. found a decreased ability to inhibit cell proliferation in psoriatic MSC co-cultured with activated T-cells in vitro, even after stimulation with IFN-γ together with TNF-α or IL-1β [32,33]. Moreover, a decreased antioxidative capability in psoriatic MSC was observed by Sah et al. They observed a decreased capability of psoriatic MSC using extracellular superoxide dismutase in eruptions induced by imiquimod [34].
MSC can differentiate into vascular endothelium cells. Excessive proliferation of capillary veins in psoriatic eruptions is observed earlier than in fully formed psoriatic plaque and it is responsible for Auspitz’s sign [35,36]. Niu et al. analyzed the expression of genes (measurement of mRNA) that induce angiogenesis in mesenchymal stem cells from the skin of psoriatic patients for PECAM 1 (platelet endothelial cell adhesion molecule-1), FGD5 (faciogenital dysplasia-5), PTGS1 (prostaglandin-endoperoxide synthase-1), MCAM (melanoma cell adhesion molecule), VASH2 (vasohibin-2), and STAB1 (stabilin-1). Considerably decreased expansion of PECAM1, PTGS1, FGD5, and MCAM in MCS was found, with no difference observed in the expression of VASH2 and STAB1 compared to healthy people. According to the authors, low expression of PECAM1 and MCAM may be interpreted as a method of inflammation modulation by dental matrix components (DMCs). However, their ability to differentiate in vitro into adipocytes and osteoblasts is preserved [37].
2.3. Role of Telocytes in Psoriasis
Human skin contains telocytes, which are parts of niches for stem cells interact with them closely, taking part in skin regeneration [38]. These are cells which have long, very thin projections called telopods, whose thickness is comparable to that of collagen fibers [39]. Manole et al. detected a reduced number of telocytes in psoriasis plaques and their degradation involves membrane disintegration, fragmentation of cytoplasm, and loss of the cell nuclei. On the other hand, topical treatment with corticosteroids increased their number [39].
3. Psoriasis and Bone Marrow Stem Cells
Although the role of T-cells in psoriasis has been very well elucidated, the cause of their hyper-reactivity remains unknown. Moreover, disorders in the function of other immune cells (monocytes, neutrophils, mast cells) are observed in psoriatic patients [40].
3.1. Associations of Psoriasis with Bone Marrow Cells
Although various factors upset the balance of the immune system in initiation of psoriatic lesions, the most important of them are associated with T cells (spontaneous activation and proliferation, production of proinflammatory cytokines). It has been suggested that hematopoietic stem cells (HSC) of bone marrow may be responsible for dysregulation of the T cells function in psoriasis. It has been shown in several recent studies that abnormal relations between T cells and HSC may be genetically conditioned. This has been proven by reported cases of remission of psoriasis, or vice-versa—the development of diseases after an allogeneic bone marrow transplant [40,41,42,43,44]. Immunological disorders typical of psoriasis can be transmitted through a bone marrow cell transplant. Leukemia in psoriatic patients can also result in the disease remission [44].
3.2. Proliferation of Bone Marrow Stem Cells (Progenitor Cells) in Psoriatic Patients Is Not Normal
In vitro studies with 99mTc bone marrow scintigraphy showed that the activity of monocytopoiesis precursors is increased in psoriasis. In consequence, the number of phagocytic cells in patients’ blood increases. Moreover, peripheral immunocytes and cytokines secreted by them (IFN-γ, IL-2, IL-8, and TNF-α) can affect the hematopoietic environment and even hematopoiesis [40,44].
Zhang et al. demonstrated that the activity of HPP-CFC (high proliferative potential colony-forming cell) and the ability to produce CFU-GM (granulocyte-macrophage colony-forming units), but not CFU-E (Erythrocyte colony-forming units), is decreased in psoriatic patients [44].
3.3. T-Cells from Haematopoietic Cells of Bone Marrow of Psoriatic Patients Are Functionally Different from T-Cells in Healthy People
Zhang et al. observed a culture of CD34+ cells of bone marrow from psoriatic patients and the process of their differentiation into T cells and regulatory CD25+ CD4+ cells. Studies have shown that CD4+ CD25+ cells inhibit effector T cells to a lesser extent both in peripheral blood and in skin lesions, which leads to their accelerated proliferation. Although the proportions of CD4+ CD25+ cells and expression of the FOXP3 gene are comparable in psoriatic patients and in healthy people, their proliferation and secretion of cytokines IL-2 and IL-10 is decreased in patients in response to the streptococcal antigen (Strep-A). Additionally, CD4+ CD25+ cells in patients cannot inhibit effector T cells [40,44].
3.4. The Role of Bone Marrow Mesenchymal Stem Cells in Psoriatic Patients
Bone marrow mesenchymal stem cells (BMSCs), also called bone marrow stromal cells, exhibit a pleiotropic immunomodulating effect, e.g., by participating in inhibition of T and NK cell proliferation and in maturation of dendritic cells [25,36]. Cytokines secreted by BMSC affect hematopoiesis. This is a group comprising over 30 hematopoietic growth factors and pro-inflammatory cytokines, including TNF-α, IL-1, IL-6, IL-7, IL-8, IL-10, IL-12, IFN-γ, and IL-18, which also affect immune response in peripheral blood. Secretion of granulocyte colony stimulation factor (G-CSF) and IL-6 increases in cultured BMSCs from psoriatic patients, whereas the levels of IL-1α, IL-1β, IL-3, IL-8, epidermal growth factor (EGF), vascular endothelial growth factor (VEGF), TNF-α, leukaemia inhibitory factor (LIF), hepatocyte growth factor (HGF), and platelet-derived growth factor (PDGF) is decreased and that of GM-CSF, IL-11, and IL-7 is unchanged. However, the concentrations of these cytokines are not correlated with PASI, which shows that their abnormal secretion results from an anomaly of BMSCs rather than from a systemic inflammatory response [25,36,44].
Mesenchymal stem cells isolated from psoriatic eruptions inhibit proliferation of T-cells less strongly than those from healthy people. Moreover, they exhibited increased secretion of IL-11, decreased secretion of IL-6 and HGF, while the production of TGF-β1 was unchanged [45].
BMSCs can differentiate into multiple mesenchymal lines, including osteocytes, chondrocytes, adipose cells, endothelial cells, and cells of skeletal muscles. Studies have shown that BMSCs taken from psoriatic patients are less able to proliferate and differentiate into these types, but they are more able to differentiate into vascular endothelial cells compared to those taken from healthy people. What is more, this ability is associated with the severity of the disease (PASI) [44].
Campanati et al. evaluated the expression of 43 genes encoding cytokines, typical of Th1, Th2, and Th17 in mesenchymal stem cells isolated from psoriatic eruptions. It was shown that expression of genes for cytokines of the Th1 and Th17 profile (INF-γ, CCR5, CXCL9, CXCL10, IL6, IL8, TNF-a, IL23A, CCL2, CCL20, CXCL2, CXCL5, IL17C, IL17F, IL17RA, IL21, TLR2) is higher in healthy people and it was similar in both groups for the Th2 profile (CCL1, CCL22, CXCL12, IL2, IL3, IL4, IL13B, IL22, IL27, TGF-β1) [24].
Furthermore, Hou et al. demonstrated disorders in the methylation of receptor genes of MSC, involved in signaling pathways [25].
3.5. Reduction of the Number of Progenitor Cells in Psoriatic Patients
Being a systemic inflammatory process, psoriasis leads to the development of metabolic disorders. By exhausting the pool of endothelial progenitor cells (EPCs) and being a source of pro-inflammatory cytokines, psoriatic eruptions that stay on the skin for years affect the cardiovascular system. It is a population of bone marrow cells with expression of CD34 and VGEFR-2 cells [45]. EPCs are responsible for integrity and regeneration of endothelium and the formation of new vessels in adults. They are recruited from bone marrow and migrate to areas of ischemia or damaged endothelium, where they repair the damage. Their number is reduced in arterial hypertension, diabetes, obesity, rheumatoid arthritis, and in tobacco smokers. Their number in blood is also reduced in patients with psoriasis and in those with psoriatic arthritis. However, the number of EPCs was found to increase following treatment with etanercept (TNF-α inhibitor), which is indicative of an improvement of the regenerative abilities of endothelium [46,47,48,49]. On the other hand, Ablin et al. did not find any significant differences between the number of EPCs in healthy people and those with psoriasis and psoriatic arthritis [45].
4. Stem Cells in Psoriasis Treatment
4.1. Autologous Haematopoietic Stem Cell Transplantation
Currently, the first clinical trials are being conducted into the use of stem cells in the treatment of psoriasis. This idea originated from observation of the remission of lesions in patients treated with mesenchymal or hematopoietic stem cells because of lymphomas and leukemias as well as other autoimmune diseases (diabetes, sclerosis multiplex, rheumatoid arthritis, systemic lupus erythematosus) [41,42,43,50] (Table 1). More than 30 patients with psoriasis who had bone marrow transplantation (BMT) which resulted in prolonged remission of psoriasis have been described in the literature in the past 25 years [42,51]. However, there have also been a few who acquired psoriasis after allogeneic BMT and blood transfusion from donors with this disease [42].

Table 1.
Analysis of the potential effect of stem cells on inhibition of the inflammatory process in psoriasis.
According to the pathogenesis of psoriasis, T-cells can provoke the development of changes after a blood transfusion, but peripheral T-cells have a relatively short life time. Psoriasis patients are known to enter remission after allogeneic transplants (but not after autologous transplants) of hematopoietic stem cells; this indicates that hematopoietic stem cells are the major factors provoking the disease [42,44].
Adkins et al. described the case of a 55-year-old female patient with chronic myeloid leukemia and concurrent refractory psoriasis, who achieved complete remission after an allogeneic bone marrow transplant. Admittedly, such quick remission may have been caused by medicines preparing the patient for the transplant (busulfan, cyclophosphamide) [41].
Likewise, Mori et al. described the case of a 54-year-old male patient with a 10-year history of psoriasis, who was treated by an allogeneic bone marrow transplant with preceding myeloablation with busulfan and cyclophosphamide because of a myelodysplastic syndrome, who achieved complete remission of psoriasis, which remained throughout the 8-month follow-up. The authors emphasize the role of the elimination of autoreactive lymphocytes during an allo-BMT, as well as immunosuppressive treatment in maintaining the state of remission [75].
Braiteh et al. presented the case of a 35-year-old patient with psoriasis (BSA 50%) and psoriatic arthritis (PsA), who had an autologous BMT (preceded by myeloablation with mephalen) in the treatment of multiple myeloma. A one-year remission of myeloma was achieved. No symptoms of psoriasis or PsA were observed during the follow-up program (more than two years). The authors emphasize that spectacular remissions were observed in patients with concurrent autoimmune diseases, who had auto hematopoietic stem cell transplantation (HSCT), but the mechanism of the effect remains unclear. Other studies have suggested that it is related to the “resetting” of the immune memory, which may result from the myeloablation preceding auto HSCT. It eliminates reactive lymphocytes and reduces reactive B cells and, in consequence, reduces auto-antibodies and developing immune tolerance. Repeated autologous infusion of stem cells may promote the regeneration of a population of lymphocytes which are able to distinguish the body’s own cells. Renewal of the population of regulatory T-cells and reactive B cells from naive progenitor cells exposed to the body’s own cells can be responsible for auto-tolerance. Moreover, auto HSCT can increase the production of cytokines and play a role in the regeneration of tissues damaged by inflammation [76].
Zurita et al. presented the results of a study of patients with severe psoriasis treated by an autologous transplant of haematopoietic cells (n = 30), compared with PUVA therapy (n = 19). Taking bone marrow from both iliac crests and isolation of the CD34+ fraction was followed by a single intravenous administration of autologous cells. The therapeutic effects were controlled for up to six months and compared with the effects of PUVA. PASI 75 reached a statistically significant level in the group treated with stem cells, but no significant difference was observed compared to the effects of PUVA [50].
4.2. Umbilical Cord-Wharton’s Jelly Stem Cells
Umbilical cord-Wharton’s Jelly stem cells (WJSCs) seem to be an ideal candidate for this therapy (Table 2). WJSCs are plastic-adherent when maintained in standard culture conditions. They express CD105, CD73, and CD90, as well as more recently recognized markers such as CD44, CD146, and CD166. However, they do not express CD3, CD45, CD34, CD14 or CD11b, CD45, CD144, CD79α or CD19, vascular endothelial growth factor (VEGF)-R1, VEGF-R2, and HLA-DR surface molecules [77,78]. Some UCB-derived cell populations show inherent ‘immunoprivileged’ properties because they exhibit class I HLA antigens, and class II HLA antigens are seen only in response to INF-γ [79]. These features fulfil the stipulated minimum criteria of ‘plastic adherence’, ‘immunological profile’, and ‘differentiation’ as stated in the position paper of the International Society for Cellular Therapy [77]. MSCs from within WJSCs are a relatively young cell type compared to most other MSCs. Among the many sources of stem cells, the human umbilical cord matrix, i.e., Wharton’s jelly (WJ), has recently become a preferential source of stem cells, because of its rapid availability with a large donor pool, non-invasive and painless collection, no risk for the donor, no ethical constraints, hypo-immunogenic and non-tumorigenic, high in vitro expandable rates and multi-potent differentiation potential, which makes them important sources for the isolation and banking of stem cells [80,81,82]. In addition, since they are rarely exposed to infectious agents, they represent a safe donor [83]. Chen et al. reported good results for psoriasis treatment using WJSCs in two cases. In the first, a patient (a 35-old-man with psoriasis and diffuse large B-cell lymphoma, stage IV) after hematopoietic stem cell transplantation failure, was successfully WJSCs-treated, with no recurrence of lymphoma or psoriasis. in the second patient, (a 26-year-old woman with psoriasis vulgaris), after three infusions, 1 × 106/kg each time over three successive weeks and two more three months later) a complete remission of the disease was observed [84]. No recurrence of the disease was observed during the 4-year follow-up [84]. Similar effects were achieved in the treatment of psoriatic arthritis [43].

Table 2.
Psoriasis remission due to autologous haematopoietic stem cell transplantation.
Human Wharton’s Jelly-derived Mesenchymal Stem Cells (hWJ-MSCs) display immunosuppressive properties and may be able to play an important role in autoimmune disorders [61]. Regulatory T-cells (Treg) are important in preventing autoimmune disease development. Interleukin-35 induces the proliferation of Treg cell populations and reduces the activity of Th17 and Th1 cells, which play a central role in the initiation of inflammation and autoimmune diseases. Recent studies identified IL-35 as a new inhibitory cytokine required for the suppressive function of Treg cells. Amari et al. revealed hWJ-MSCs as a good source of IL-35 for reduction of inflammation and autoimmune diseases [61].
Th17 cell homeostasis is the relationship with Tregs, whose imbalance may lead to the development of psoriasis. Rafei et al. confirmed that MSCs inhibit the activity of the Thl7 cell, reducing the expression of interleukin IL-17 and decreasing inflammatory cell infiltration in the central nervous system [68]. Park et al. found that TGF-β-transduced MSCs in experimental autoimmune arthritis suppressed T-cell proliferation, and down-regulated pro-inflammatory cytokine production. Moreover, these therapeutic effects were associated with an increase in CD4+FoxP3+ Treg cells, inhibition of Th17 cell formation, and inhibited osteoclast differentiation [89]. Alluno et al. revealed that only IFN-γ-pretreated microencapsulated (CpS)-hUCMS (not free) have properties to suppress T cell proliferation and restore the Treg/Th17 ratio in Sjögren syndrome [57]. They reciprocally modulate FoxP3 and RORγt expression, eventually leading to the conversion of Th17 into Treg cells, despite the presence of hUCMS-derived IL-6. Indeed, CD4+IL-17+FoxP3+RORγt− T-cells may represent an intermediate phenotype of Th17 cells turning into Treg cells [57]. Moreover, mesenchymal stem cells can up-regulate Treg transcription factor FoxP3 and Th17/Treg plasticity is well established [90]. Wang et al. assessed the effect of WJ-MSCs on Th1, Th2, Th17 cytokines production and Treg augmentation. WJ-MSCs co-cultured with peripheral blood mononuclear, showed an immunosuppressive function by inhibiting the proliferative response of Th1 and Th17 but augmenting Th2 and Treg. Moreover, co-cultures with WJ-MSCs resulted in an increase in IL-10 and IL-6 levels, especially in co-cultures with IFN-γ treated WJ-MSCs [58]. Additionally, they can affect maturation and activation of precursors of dendritic cells (DC) [56]. hWJSCs express CD14, a common marker for marcrophages. The soluble form of CD14 can down-regulate T cell activation [56,91].
WJSCs produce large amounts of tolerogenic IL-10, higher levels of TGF-β than BM-MSCs, and express HLA-G, which is not expressed in BM-MSCs [56,57]. Chen et al. detected therapeutic effects and the mechanism of WJ-MSC in a spontaneous-abortion rat model. The mechanism may be related to its up-regulation of IL-10 and down-regulation of IFN-γ and IL-17 [55].
hWJSCs had an up-regulation of the induction of apoptosis [64]. The increase in tumor cell death driven by WJ-MSCs such as Bcl-2, Bcl-xL, Survivin, Mcl-1, and cIAP-1 have been observed [74]. By cleaved caspase 3/9 up-regulation in cancer cells, WJ-MSC executed its pro-apoptotic effect [92,93].
hWJSCs express HLA-G, which is important for immune tolerance [64,65]. HLA-G is a non-classical major histo-compatibility complex (MHC) class I protein which is expressed in both membrane-bound and soluble isoforms that can display tolerogenic properties via interaction with inhibitory receptors on dendritic cells, natural killer cells, and T cells. Soluble HLA-G exerts an immunosuppressive effect by inducing apoptosis in CD8+ T cells, while also down-modulating CD4+ T cell proliferation. Unfortunately, the expressional level of HLA-G is higher in adult tissue compared to foetal tissue MSC [58,94].
5. Conclusions
The data presented here suggest that stem cells in psoriatic patients affect proliferation and differentiation of keratinocytes and immune system cells and the ability to secrete cytokines. This confirms the view that psoriasis is a multi-system disease, which affects not only the skin, but is also related to the function of the hematopoietic and neuroendocrine systems and with metabolic disorders. Although it requires a lot of new research (Table 3). The application of stem cells raises hopes for developing a new, safe, and effective therapy for psoriatic patients.

Table 3.
List of past and present clinical adipose-derived stem cell trials in psoriasis [95].
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Danielsen, K.; Olsen, A.O.; Wilsgaard, T.; Furberg, A.S. Is the prevalence of psoriasis increasing? A 30-year follow-up of a population-based cohort. Br. J. Dermatol. 2013, 168, 1303–1310. [Google Scholar] [CrossRef] [PubMed]
- Parisi, R.; Symmons, D.P.; Griffiths, C.E.; Ashcroft, D.M. Identification and Management of Psoriasis and Associated ComorbidiTy (IMPACT) project team. Global epidemiology of psoriasis: A systematic review of incidence and prevalence. J. Investig. Dermatol. 2013, 133, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Young, P.; Armstrong, A.W. An update on psoriasis and metabolic syndrome: A meta-analysis of observational studies. PLoS ONE 2017, 12, e0181039. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Taylor, C.; Kornmehl, H.; Armstrong, A.W. Psoriasis and suicidality: A systematic review and meta-analysis. J. Am. Acad. Dermatol. 2017, 77, 425–440. [Google Scholar] [CrossRef] [PubMed]
- Randa, H.; Todberg, T.; Skov, L.; Larsen, L.S.; Zachariae, R. Health-related Quality of Life in Children and Adolescents with Psoriasis: A Systematic Review and Meta-analysis. Acta Derm. Venereol. 2017, 97, 555–563. [Google Scholar] [CrossRef] [PubMed]
- Belinchón, I.; Rivera, R.; Blanch, C.; Comellas, M.; Lizán, L. Adherence, satisfaction and preferences for treatment in patients with psoriasis in the European Union: A systematic review of the literature. Patient Preference Adherence 2016, 10, 2357–2367. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Chang, C.; Lu, Q. The inflammatory response in psoriasis: A comprehensive review. Clin. Rev. Allergy Immunol. 2016, 50, 377–389. [Google Scholar] [CrossRef] [PubMed]
- Martin, D.A.; Towne, J.E.; Kricorian, G.; Klekotka, P.; Gudjonsson, J.E.; Krueger, J.E.; Russel, C.B. The emerging role of IL-17 in the pathogenesis of psoriasis: Preclinical and clinical findings. J. Investig. Dermatol. 2013, 133, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.Y.; Shi, Y.; Luo, L.F.; Jiang, G.; Zhou, Q.; Xu, S.Z.; Lei, T.C. Asymmetric stem-cell division ensures sustained keratinocyte hyperproliferation in psoriatic skin lesions. Int. J. Mol. Med. 2016, 37, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Franssen, M.E.; Zeeuwen, P.L.; Vierwinden, G.; van de Kerkhof, P.C.; Schalkwijk, J.; van Erp, P.E. Phenotypical and functional differences in germinative subpopulations derived from normal and psoriatic epidermis. J. Investig. Dermatol. 2005, 124, 373–383. [Google Scholar] [CrossRef] [PubMed]
- Joachimiak, R.; Bajek, A.; Drewa, T. Hair follicle as a novel source of stem cells. Postepy Hig. Med. Dosw. 2012, 66, 181–186. [Google Scholar] [CrossRef]
- Ma, D.R.; Yang, E.N.; Lee, S.T. A review: The location, molecular characterisation and multipotency of hair follicle epidermal stem cells. Ann. Acad. Med. Singap. 2004, 33, 784–788. [Google Scholar] [PubMed]
- O’Shaughnessy, R.F.; Seery, J.P.; Celis, J.E.; Frischauf, A.; Watt, F.M. PA-FABP, a novel marker of human epidermal transit amplifying cells revealed by 2D protein gel electrophoresis and cDNA array hybridisation. FEBS Lett. 2000, 486, 149–154. [Google Scholar] [CrossRef]
- Jones, P.; Simons, B.D. Epidermal homeostasis: Do committed progenitors work while stem cells sleep? Nat. Rev. Mol. Cell Biol. 2008, 9, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Watarai, A.; Amoh, Y.; Maejima, H.; Hamada, Y.; Katsuoka, K. Nestin expression is increased in the suprabasal epidermal layer in psoriasis vulgaris. Acta Derm. Venereol. 2013, 93, 39–43. [Google Scholar] [CrossRef] [PubMed]
- Charruyer, A.; Fong, S.; Vitcov, G.G.; Sklar, S.; Tabernik, L.; Taneja, M.; Caputo, M.; Soeung, C.; Yue, L.; Uchida, Y.; et al. Brief report: Interleukin-17A-dependent asymmetric stem cell divisions are increased in human psoriasis: A mechanism underlying benign hyperproliferation. Stem Cells 2017, 35, 2001–2007. [Google Scholar] [CrossRef] [PubMed]
- Furuhashi, T.; Saito, C.; Torii, K.; Nishida, E.; Yamazaki, S.; Morita, A. Photo(chemo)therapy reduces circulating Th17 cells and restores circulating regulatory T cells in psoriasis. PLoS ONE 2013, 8, e54895. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Reynolds, J.M.; Stout, R.D.; Bernlohr, D.A.; Suttles, J. Regulation of Th17 differentiation by epidermal fatty acid-binding protein. J. Immunol. 2009, 182, 7625–7633. [Google Scholar] [CrossRef] [PubMed]
- Reynolds, J.M.; Liu, Q.; Brittingham, K.C.; Liu, Y.; Gruenthal, M.; Gorgun, C.Z.; Liu, Y.; Gruenthal, M.; Gorgun, C.Z.; Hotamisligil, G.S.; et al. Deficiency of fatty acid-binding proteins in mice confers protection from development of experimental autoimmune encephalomyelitis. J. Immunol. 2007, 179, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Gutiérrez-González, L.H.; Ludwig, C.; Hohoff, C.; Rademacher, M.; Hanhoff, T.; Rüterjans, H.; Spener, F.; Lücke, C. Solution structure and backbone dynamics of human epidermal-type fatty acid-binding protein (E-FABP). Biochem. J. 2002, 15, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Katayama, I.; Nishioka, K. Possible contribution of stem cell factor in psoriasis vulgaris. J. Dermatol. Sci. 2000, 24, 171–176. [Google Scholar] [CrossRef]
- Liu, R.F.; Wang, F.; Wang, Q.; Zhao, X.C.; Zhang, K.M. Research note mesenchymal stem cells from skin lesions of psoriasis patients promote proliferation and inhibit apoptosis of HaCaT cells. Genet. Mol. Res. 2015, 14, 17758–17767. [Google Scholar] [CrossRef] [PubMed]
- Orciani, M.; Campanati, A.; Salvolini, E.; Lucarini, G.; Di Benedetto, G.; Offidani, A.; Di Primio, R. The mesenchymal stem cell profile in psoriasis. Br. J. Dermatol. 2011, 165, 585–592. [Google Scholar] [CrossRef] [PubMed]
- Campanati, A.; Orciani, M.; Consales, V.; Lazzarini, R.; Ganzetti, G.; Di Benedetto, G.; Di Primio, R.; Offidani, A. Characterization and profiling of immunomodulatory genes in resident mesenchymal stem cells reflect the Th1-Th17/Th2 imbalance of psoriasis. Arch. Dermatol. Res. 2014, 306, 915–920. [Google Scholar] [CrossRef] [PubMed]
- Hou, R.; Yan, H.; Niu, X.; Chang, W.; An, P.; Wang, C.; Yang, Y.; Yan, X.; Li, J.; Liu, R.; et al. Gene expression profile of dermal mesenchymal stem cells from patients with psoriasis. J. Eur. Acad. Dermatol. Venereol. 2014, 28, 1782–1791. [Google Scholar] [CrossRef] [PubMed]
- Hou, R.X.; Liu, R.F.; Zhao, X.C.; Jia, Y.R.; An, P.; Hao, Z.P.; Li, J.Q.; Li, X.H.; Yin, G.H.; Zhang, K.M. Increased miR-155-5p expression in dermal mesenchymal stem cells of psoriatic patients: Comparing the microRNA expression profile by microarray. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.; Ren, G.; Cao, G.; Chen, Q.; Zheng, C.; Du, L.; Han, X.; Jiang, M.; Yang, Q.; Lin, L.; et al. miR-155 Regulates immune modulatory properties of mesenchymal stem cells by targeting TAK1-binding protein 2. J. Biol. Chem. 2013, 288, 11074–11079. [Google Scholar] [CrossRef] [PubMed]
- Lu, C.; Huang, X.; Zhang, X.; Roensch, K.; Cao, Q.; Nakayama, K.I.; Blazar, B.R.; Zeng, Y.; Zhou, X. miR-221 and miR-155 regulate human dendritic cell development, apoptosis, and IL-12 production through targeting of p27kip1, KPC1, and SOCS-1. Blood 2011, 16, 4293–4303. [Google Scholar] [CrossRef] [PubMed]
- Tili, E.; Michaille, J.J.; Wernicke, D.; Alder, H.; Costinean, S.; Volinia, S.; Croce, C.M. Mutator activity induced by microRNA-155 (miR-155) links inflammation and cancer. Proc. Natl. Acad. Sci. USA 2011, 108, 4908–4913. [Google Scholar] [CrossRef] [PubMed]
- Trotta, R.; Chen, L.; Ciarlariello, D.; Josyula, S.; Mao, C.; Costinean, S.; Yu, L.; Butchar, J.P.; Tridandapani, S.; Croce, C.M.; et al. miR-155 regulates IFN-g production in natural killer cells. Blood 2012, 15, 3478–3485. [Google Scholar] [CrossRef] [PubMed]
- English, K. Mechanisms of mesenchymal stromal cell immunomodulation. Immunol. Cell Biol. 2013, 91, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Hou, R.; Li, J.; Niu, X.; Liu, R.; Chang, W.; Zhao, X.; Wang, Q.; Li, X.; Yin, G.; Zhang, K. Stem cells in psoriasis. J. Dermatol. Sci. 2017, 86, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Wang, Y.; Zhao, X.; Yang, Y.; Zhang, K. Lymphocyte inhibition is compromised in mesenchymal stem cells from psoriatic skin. Eur. J. Dermatol. 2014, 24, 560–567. [Google Scholar] [PubMed]
- Sah, S.K.; Park, K.H.; Yun, C.O.; Kang, K.S.; Kim, T.Y. Effects of human mesenchymal stem cells transduced with superoxide dismutase on imiquimod-induced psoriasis-like skin inflammation in mice. Antioxid. Redox Signal. 2016, 24, 233–248. [Google Scholar] [CrossRef] [PubMed]
- Henno, A.; Blacher, S.; Lambert, C.; Colige, A.; Seidel, L.; Noël, A.; Lapière, C.; de la Brassinne, M.; Nusgens, B.V. Altered expression of angiogenesis and lymphangiogenesis markers in the uninvolved skin of plaque-type psoriasis. Br. J. Dermatol. 2009, 160, 581–590. [Google Scholar] [CrossRef] [PubMed]
- Hou, R.; Liu, R.; Niu, X.; Chang, W.; Yan, X.; Wang, C.; Li, J.; An, P.; Li, X.; Yin, G.; et al. Biological characteristics and gene expression pattern of bone marrow mesenchymal stem cells in patients with psoriasis. Exp. Dermatol. 2014, 23, 521–523. [Google Scholar] [CrossRef] [PubMed]
- Niu, X.; Chang, W.; Liu, R.; Hou, R.; Li, J.; Wang, C.; Li, X.; Zhang, K. Expression of pro-angiogenic genes in mesenchymal stem cells derived from dermis of patients with psoriasis. Int. J. Dermatol. 2016, 55, e280–e288. [Google Scholar] [CrossRef] [PubMed]
- Ceafalan, L.; Gherghiceanu, M.; Popescu, L.M.; Simionescu, O. Telocytes in human skin-are they involved in skin regeneration? J. Cell. Mol. Med. 2012, 16, 1405–1420. [Google Scholar] [CrossRef] [PubMed]
- Manole, C.G.; Gherghiceanu, M.; Simionescu, O. Telocyte dynamics in psoriasis. J. Cell. Mol. Med. 2015, 19, 1504–1519. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Li, X.; Yin, G.; Liu, Y.; Niu, X.; Hou, R. Functional characterization of CD4+CD25+ regulatory T cells differentiated in vitro from bone marrow-derived haematopoietic cells of psoriasis patients with a family history of the disorder. Br. J. Dermatol. 2008, 158, 298–305. [Google Scholar] [CrossRef] [PubMed]
- Adkins, D.R.; Abidi, M.H.; Brown, R.A.; Khoury, H.; Goodnough, L.T.; Vij, R.; Westervelt, P.; DiPersio, J.F. Resolution of psoriasis after allogeneic bone marrow transplantation for chronic myelogenous leukemia: Late complications of therapy. Bone Marrow Transplant. 2000, 26, 1239–1241. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Li, J.; Wang, L.; Niu, X.; Hou, R.; Liu, R.; Hao, Z.; Wang, C.; Yin, G.; Zhang, K. Transmission of psoriasis by allogeneic bone marrow transplantation and blood transfusion. Blood Cancer J. 2015, 13, e288. [Google Scholar] [CrossRef] [PubMed]
- Woods, A.C.; Mant, M.J. Amelioration of severe psoriasis with psoriatic arthritis for 20 years after allogeneic haematopoietic stem cell transplantation. Ann. Rheum. Dis. 2006, 65, 697. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Hou, R.; Niu, X.; Zhang, J.; Yin, G.; Li, X.; Jia, Y. Decreased colony formation of high proliferative potential colony-forming cells and granulocyte-macrophage colony-forming units and increased Hes-1 expression in bone marrow mononuclear cells from patients with psoriasis. Br. J. Dermatol. 2010, 163, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Ablin, J.N.; Goldstein, Z.; Aloush, V.; Matz, H.; Elkayam, O.; Caspi, D.; Swartzenberg, S.; George, J.; Wohl, Y. Normal levels and function of endothelial progenitor cells in patients with psoriatic arthritis. Rheumatol. Int. 2009, 29, 257–262. [Google Scholar] [CrossRef] [PubMed]
- Batycka-Baran, A.; Paprocka, M.; Krawczenko, A.; Kantor, A.; Duś, D.; Szepietowski, J.C. Reduced number of circulating endothelial progenitor cells (CD133+/KDR+) in patients with plaque psoriasis. Dermatology 2012, 22, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Głowińska-Olszewska, B.; Łuczyński, W.; Bossowski, A. Endothelial progenitor cells as a new marker of endothelial function with respect to risk of cardiovascular disorders. Postepy Hig. Med. Dosw. 2011, 65, 8–15. [Google Scholar] [CrossRef]
- Hayek, S.S.; Neuman, R.; Kavtaradze, N.; Sher, S.; Jones, D.; Li, Q.; Zhao, L.; Chen, S.C.; Waller, E.; Quyyumi, A. Tumor necrosis factor-alpha antagonism with etanercept improves endothelial progenitor cell counts in patients with psoriasis: Etanercept, vascular function and endothelial progenitor cells in psoriasis. Int. J. Cardiol. 2015, 182, 387–389. [Google Scholar] [CrossRef] [PubMed]
- Hristov, M.; Erl, W.; Weber, P.C. Endothelial progenitor cells: Mobilization, differentiation, and homing. Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1185–1189. [Google Scholar] [CrossRef] [PubMed]
- Zurita, G.; Geffner, L.; Maldonado, B.; Uraga, E.; Armijos, L. Autologous Stem Cells Implants in Psoriasis. Available online: http://www.medicosecuador.com/espanol/articulos_medicos/ autologous_stem_cells_implants.htm (accessed on 20 September 2017).
- Kaffenberger, B.H.; Wong, H.K.; Jarjour, W.; Andritsos, L.A. Remission of psoriasis after allogeneic, but not autologous, hematopoietic stem-cell transplantation. J. Am. Acad. Dermatol. 2013, 68, 489–492. [Google Scholar] [CrossRef] [PubMed]
- Al-Robaee, A.A.; Al-Zolibani, A.A.; Al-Shobili, H.A.; Kazamel, A.; Settin, A. IL-10 implications in psoriasis. Int. J. Health Sci. Qassim 2008, 2, 53–58. [Google Scholar] [PubMed]
- Flisiak, I.; Zaniewski, P.; Chodynicka, B. Plasma TGF-beta1, TIMP-1, MMP-1 and IL-18 as a combined biomarker of psoriasis activity. Biomarkers 2008, 13, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Zaher, H.; Shaker, O.G.; EL-Komy, M.H.; El-Tawdi, A.; Fawzi, M.; Kadry, D. Serum and tissue expression of transforming growth factor beta 1 in psoriasis. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 406–409. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Niu, J.W.; Ning, H.M.; Li, X.B.; Li, Y.; Wang, D.H.; Hu, L.D.; Sheng, H.X.; Xu, M.; Zhang, L.; et al. Treatment of psoriasis with Mesenchymal Stem Cells. Am. J. Med. 2016, 29, 13–14. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.W.; Staples, M.; Shinozuka, K.; Pantcheva, P.; Kang, S.D.; Borlongan, C.V. Wharton’s jelly-derived mesenchymal stem cells: Phenotypic characterization and optimizing their therapeutic potential for clinical applications. Int. J. Mol. Sci. 2013, 14, 11692–11712. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, S.J.; Gopalakrishnan, D.; Shankar, S.R.; Vasandan, A.B. Pro-inflammatory cytokines, IFNgamma and TNFalpha, influence immune properties of human bone marrow and Wharton jelly mesenchymal stem cells differentially. PLoS ONE 2010, 5, e9016. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, Q.; Wang, Z.; Tong, H.; Ma, L.; Zhang, Y.; Shan, F.; Meng, Y.; Yuan, Z. Comparative analysis of human mesenchymal stem cells from fetal-bone marrow, adipose tissue, and Warton’s jelly as sources of cell immunomodulatory therapy. Hum. Vaccin. Immunother. 2016, 12, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.Y.; Chen, X.Y.; Li, J.; Zhang, H.Y.; Liu, J.; Sun, L.D. MiR-200a expression in CD4+ T cells correlates with the expression of Th17/Treg cells and relevant cytokines in psoriasis vulgaris: A case control study. Biomed. Pharmacother. 2017, 93, 1158–1164. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Yang, X.Q.; Cheng, J.; Hui, R.S.; Gao, T.W. Increased Th17 cells are accompanied by FoxP3(+) Treg cell accumulation and correlated with psoriasis disease severity. Clin. Immunol. 2010, 135, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Amari, A.; Ebtekar, M.; Moazzeni, S.M.; Soleimani, M.; Mohammadi-Amirabad, L.; Tahoori, M.T.; Massumi, M. In vitro generation of IL-35-expressing Human Wharton’s Jelly-derived Mesenchymal Stem Cells using lentiviral vector. Iran. J. Allergy Asthma Immunol. 2015, 14, 416–426. [Google Scholar] [PubMed]
- Aractingi, S.; Briand, N.; Le Danff, C.; Viguier, M.; Bachelez, H.; Michel, L.; Dubertret, R.; Carosella, E.D. HLA-G and NK receptors are expressed in psoriatic skin: A possible pathway for regulating infiltrating T cells? Am. J. Pathol. 2001, 159, 71–77. [Google Scholar] [CrossRef]
- Borghi, A.; Fogli, E.; Stignani, M.; Melchiorri, L.; Altieri, E.; Baricordi, O.; Rizzo, R.; Virgili, A. Soluble human leukocyte antigen-G and interleukin-10 levels in plasma of psoriatic patients: Preliminary study on a possible correlation between generalized immune status, treatments and disease. Arch. Dermatol. Res. 2008, 300, 551–559. [Google Scholar] [CrossRef] [PubMed]
- Fong, C.Y.; Chak, L.L.; Biswas, A.; Tan, J.H.; Gauthaman, K.; Chan, W.K.; Bongso, A. Human Wharton’s jelly stem cells have unique transcriptome profiles compared to human embryonic stem cells and other mesenchymal stem cells. Stem Cell Rev. 2011, 7, 1–16. [Google Scholar] [CrossRef] [PubMed]
- La Rocca, G.; Anzalone, R.; Corrao, S.; Magno, F.; Loria, T.; Lo Iacono, M.; Di Stefano, A.; Giannuzzi, P.; Marasà, L.; Cappello, F.; et al. Isolation and characterization of Oct-4+/HLA-G+ mesenchymal stem cells from human umbilical cord matrix: Differentiation potential and detection of new markers. Histochem. Cell Biol. 2009, 131, 267–282. [Google Scholar] [CrossRef] [PubMed]
- Keijsers, R.R.; van der Velden, H.M.; van Erp, P.E.; de Boer-van Huizen, R.T.; Joosten, I.; Koenen, H.J.; van de Kerkhof, P.C. Balance of Treg vs. T-helper cells in the transition from symptomless to lesional psoriatic skin. Br. J. Dermatol. 2013, 168, 1294–1302. [Google Scholar] [CrossRef] [PubMed]
- Owczarczyk-Saczonek, A.; Placek, W. Interleukin-17 as a factor linking the pathogenesis of psoriasis with metabolic disorders. Int. J. Dermatol. 2017, 56, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Rafei, M.; Campeau, P.M.; Aguilar-Mahecha, A.; Buchanan, M.; Williams, P.; Birman, E.; Yuan, S.; Young, Y.K.; Boivin, M.N.; Forner, K.; et al. Mesenchymal stromal cells ameliorate experimental autoimmune encephalomyelitis by inhibiting CD4 Thl7 T cells in a CC chemokine ligand 2-dependent manner. Immunology 2009, 182, 5994–6002. [Google Scholar] [CrossRef] [PubMed]
- Bovenschen, H.J.; van de Kerkhof, P.C.; van Erp, P.E.; Woestenenk, R.; Joosten, I.; Koenen, H.J. Foxp3+ regulatory T cells of psoriasis patients easily differentiate into IL-17A-producing cells and are found in lesional skin. J. Investig. Dermatol. 2011, 131, 1853–1860. [Google Scholar] [CrossRef] [PubMed]
- Alunno, A.; Montanucci, P.; Bistoni, O.; Basta, G.; Caterbi, S.; Pescara, T.; Pennoni, I.; Bini, V.; Bartoloni, E.; Gerli, R.; et al. In vitro immunomodulatory effects of microencapsulated umbilical cord Wharton jelly-derived mesenchymal stem cells in primary Sjögren’s syndrome. Rheumatol. Oxf. 2015, 54, 163–168. [Google Scholar] [CrossRef] [PubMed]
- Karamehic, J.; Zecevic, L.; Resic, H.; Jukic, M.; Jukic, T.; Ridjic, O.; Panjeta, M.; Coric, J. Immunophenotype lymphocyte of peripheral blood in patients with psoriasis. Med. Arch. 2014, 68, 236–238. [Google Scholar] [CrossRef] [PubMed]
- Khasawneh, A.; Baráth, S.; Medgyesi, B.; Béke, G.; Dajnoki, Z.; Gáspár, K.; Jenei, A.; Pogácsás, L.; Pázmándi, K.; Gaál, J.; et al. Myeloid but not plasmacytoid blood DCs possess Th1 polarizing and Th1/Th17 recruiting capacity in psoriasis. Immunol. Lett. 2017, 189, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Kastelan, M.; Prpić-Massari, L.; Brajac, I. Apoptosis in psoriasis. Acta Dermatovenerol. Croat. 2009, 17, 182–186. [Google Scholar] [PubMed]
- Han, I.; Yun, M.; Kim, E.O.; Kim, B.; Jung, M.H.; Kim, S.H. Umbilical cord tissue-derived mesenchymal stem cells induce apoptosis in PC-3 prostate cancer cells through activation of JNK and downregulation of PI3K/AKT signaling. Stem Cell Res. Ther. 2014, 5, 54. [Google Scholar] [CrossRef] [PubMed]
- Mori, J.; Kakihana, K.; Ohashi, K. Sustained remission of psoriasis vulgaris after allogeneic bone marrow transplantation. Br. J. Haematol. 2012, 159, 121. [Google Scholar] [CrossRef] [PubMed]
- Braiteh, F.; Hymes, S.R.; Giralt, S.A.; Jones, R. Complete remission of psoriasis after autologous hematopoietic stem-cell transplantation for multiple myeloma. J. Clin. Oncol. 2008, 26, 4511–4513. [Google Scholar] [CrossRef] [PubMed]
- Damien, P.; Allan, D.S. Regenerative therapy and immune modulation using Umbilical Cord Blood-Derived Cells. Biol. Blood Marrow Transplant. 2015, 21, 1545–1554. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Chamberlain, G.; Fox, J.; Ashton, B.; Middleton, J. Concise review: Mesenchymal stem cells: Their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells 2007, 25, 2739–2749. [Google Scholar] [CrossRef] [PubMed]
- Batsali, A.K.; Kastrinaki, M.C.; Papadaki, H.A.; Pontikoglou, C. Mesenchymal stem cells derived from Wharton’s Jelly of the umbilical cord: Biological properties and emerging clinical applications. Curr. Stem Cell Res. Ther. 2013, 8, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Richardson, S.M.; Kalamegam, G.; Pushparaj, P.N.; Matta, C.; Memic, A.; Khademhosseini, A.; Mobasheri, R.; Poletti, F.L.; Hoyland, J.A.; Mobasheri, A. Mesenchymal stem cells in regenerative medicine: Focus on articularcartilage and intervertebral disc regeneration. Methods 2016, 99, 69–80. [Google Scholar] [CrossRef] [PubMed]
- Sabapathy, V.; Sundaram, B.; Mankuzhy, P.; Kumar, S. Human Wharton’s Jelly Mesenchymal Stem Cells plasticity augments scar-free skin wound healing with hair growth. PLoS ONE 2014, 9, e93726. [Google Scholar] [CrossRef] [PubMed]
- Mohren, M.; Daikeler, T.; Benz, D.; Günaydin, I.; Kanz, L.; Kötter, I. Myeloablative immunosuppressive treatment with autologous haematopoietic stem cell transplantation in a patient with psoriatic arthropathy and monoclonal gammopathy of undetermined significance. Ann. Rheum. Dis. 2004, 63, 466–467. [Google Scholar] [CrossRef] [PubMed]
- Held, K.; Rahmetulla, R.; Loew, T.W.; Radhi, M.A. Complete resolution of guttate psoriasis following autologous SCT for Ewing’s sarcoma in a pediatric patient. Bone Marrow Transplant. 2012, 47, 1585–1586. [Google Scholar] [CrossRef] [PubMed]
- Kishimoto, Y.; Yamamoto, Y.; Ito, T.; Matsumoto, N.; Ichiyoshi, H.; Katsurada, T.; Date, M.; Ohga, S.; Kitajima, H.; Ikehara, S.; et al. Transfer of autoimmune thyroiditis and resolution of palmoplantar pustular psoriasis following allogeneic bone marrow transplantation. Bone Marrow Transplant. 1997, 19, 1041–1043. [Google Scholar] [CrossRef] [PubMed]
- Rossi, H.A.; Becker, P.S.; Emmons, R.V.; Westervelt, P.; Levy, W.; Liu, Q.; Clark, Y.; Ballen, K. High-dose cyclophosphamide, BCNU, and VP-16 (CBV) conditioning before allogeneic stem cell transplantation for patients with non-Hodgkin's lymphoma. Bone Marrow Transplant. 2003, 31, 441–446. [Google Scholar] [CrossRef] [PubMed]
- Kanamori, H.; Tanaka, M.; Kawaguchi, H.; Yamaji, S.; Fujimaki, K.; Tomita, N.; Fujisawa, S.; Ishigatsubo, J. Resolution of psoriasis following allogeneic bone marrow transplantation for chronic myelogenous leukemia: Case report and review of the literature. Am. J. Hematol. 2002, 71, 41–44. [Google Scholar] [CrossRef] [PubMed]
- Slavin, S.; Nagler, A.; Varadi, G.; Or, R. Graft vs autoimmunity following allogeneic non-myeloablative blood stem cell transplantation in a patient with chronic myelogenous leukemia and severe systemic psoriasis and psoriatic polyarthritis. Exp. Hematol. 2000, 28, 853–857. [Google Scholar] [CrossRef]
- Park, M.J.; Park, H.; Cho, M.L.; Oh, H.J.; Cho, Y.G.; Min, S.Y.; Chung, B.H.; Lee, J.W.; Kim, H.Y.; Cho, S.G. Transforming growth factor beta-transduced mesenchymal stem cells ameliorate experimental autoimmune arthritis through reciprocal regulation of Treg/Thl7 cells and osteoclastogenesis. Arthritis Rheum. 2011, 63, 1668–1680. [Google Scholar] [CrossRef] [PubMed]
- Sharma, M.D.; Hou, D.Y.; Liu, Y.; Koni, P.; Metz, R.; Chandler, P.; Mellor, A.L.; He, Y.; Munn, D.H. Indoleamine 2,3-dioxygenase controls conversion of Foxp3+ Tregs to TH17-like cells in tumor-draining lymph nodes. Blood 2009, 113, 6102–6111. [Google Scholar] [CrossRef] [PubMed]
- Rey Nores, J.E.; Bensussan, A.; Vita, N.; Stelter, F.; Arias, M.A.; Jones, M.; Lefort, S.; Borysiewicz, L.K.; Ferrara, P.; Labéta, M.O. Soluble CD14 acts as a negative regulator of human T cell activation and function. Eur. J. Immunol. 1999, 29, 265–276. [Google Scholar] [CrossRef]
- Kalaszczynska, I.; Ferdyn, K. Wharton’s jelly derived mesenchymal stem cells: Future of regenerative medicine? Recent findings and clinical significance. Biomed. Res. Int. 2015, 2015, 430847. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Ju, G.Q.; Du, T.; Zhu, Y.J.; Liu, G.H. Microvesicles derived from human umbilical cord Wharton’s jelly mesenchymal stem cells attenuate bladder tumor cell growth in vitro and in vivo. PLoS ONE 2013, 8, e61366. [Google Scholar] [CrossRef] [PubMed]
- Gotherstrom, C.; Ringden, O.; Tammik, C.; Zetterberg, E.; Westgren, M.; Le Blanc, K. Immunologic properties of human fetal mesenchymal stem cells. Am. J. Obstet. Gynecol. 2004, 190, 239–245. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials. Available online: https://clinicaltrials.gov/ (accessed on 20 September 2017).
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).