Using Vital Dyes to Trace Uptake of dsRNA by Green Peach Aphid Allows Effective Assessment of Target Gene Knockdown
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification of Appropriate Dyes to Trace Uptake in Aphids
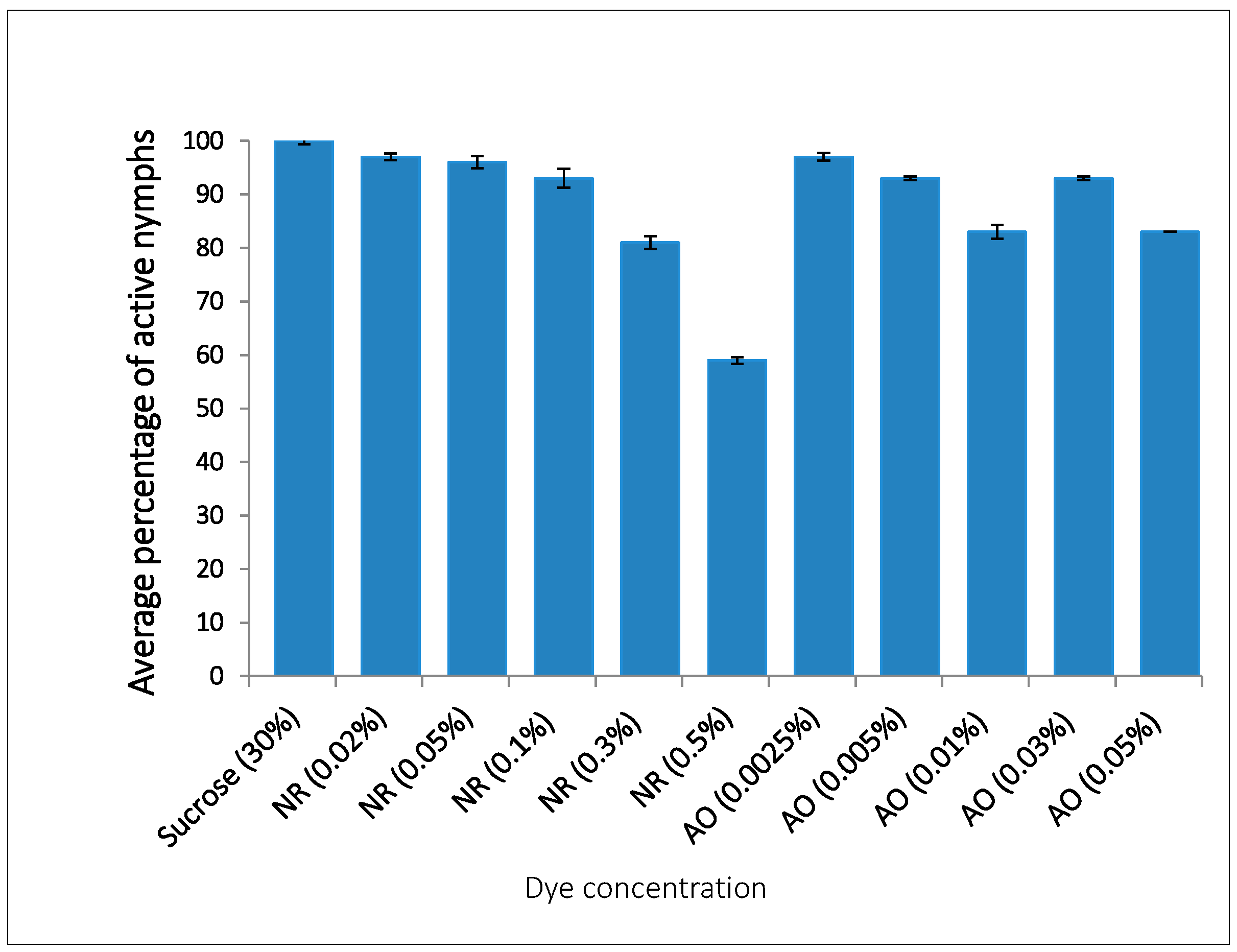
2.2. Optimisation of Non-Toxic Concentrations of AO and NR Visible in Aphid Bodies
2.3. Double-Stranded RNA Quality Is Not Affected by Neutral Red or Acridine Orange
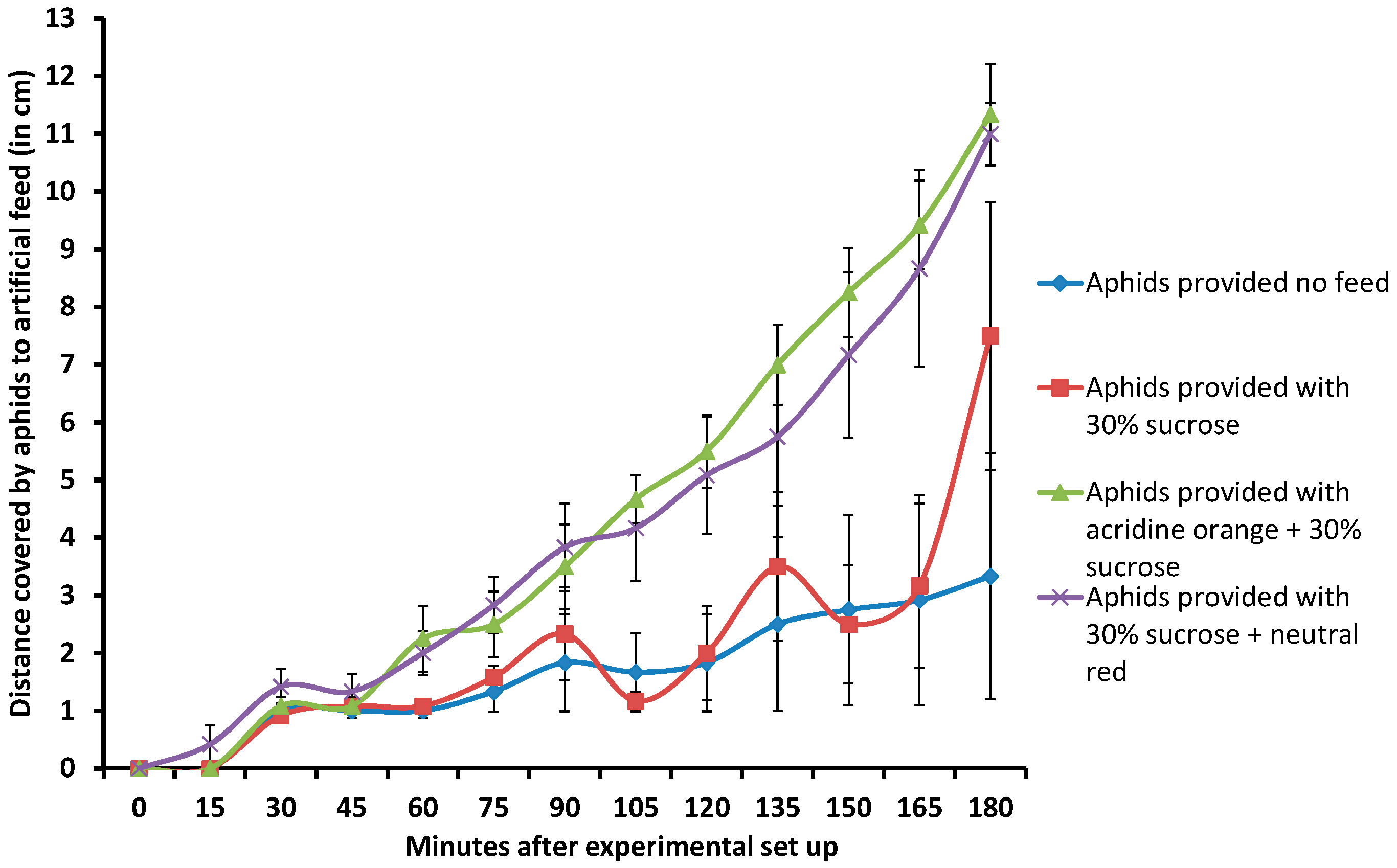
2.4. Addition of Dyes to Feed Also Attracts Aphids to Artificial Feed
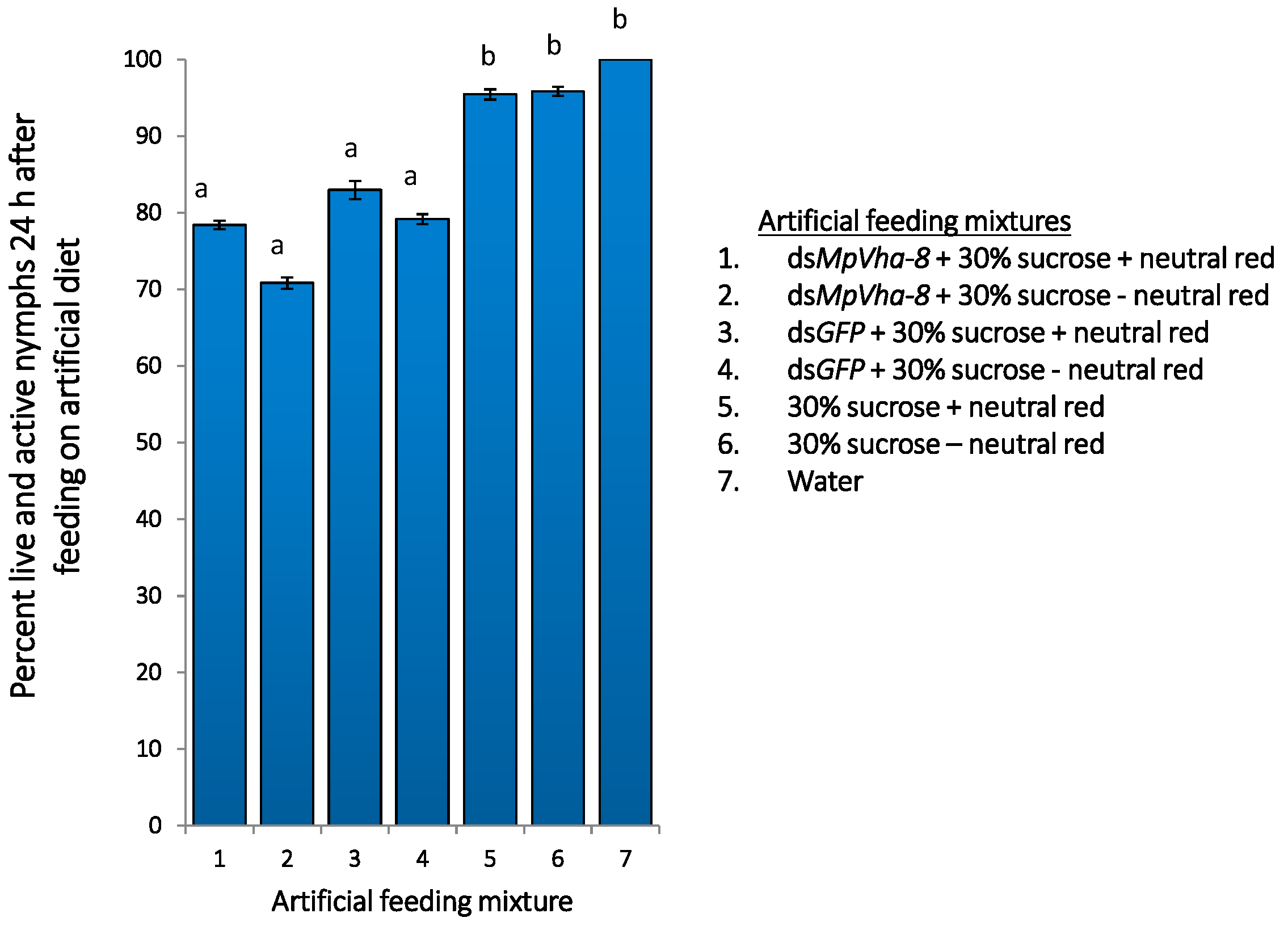
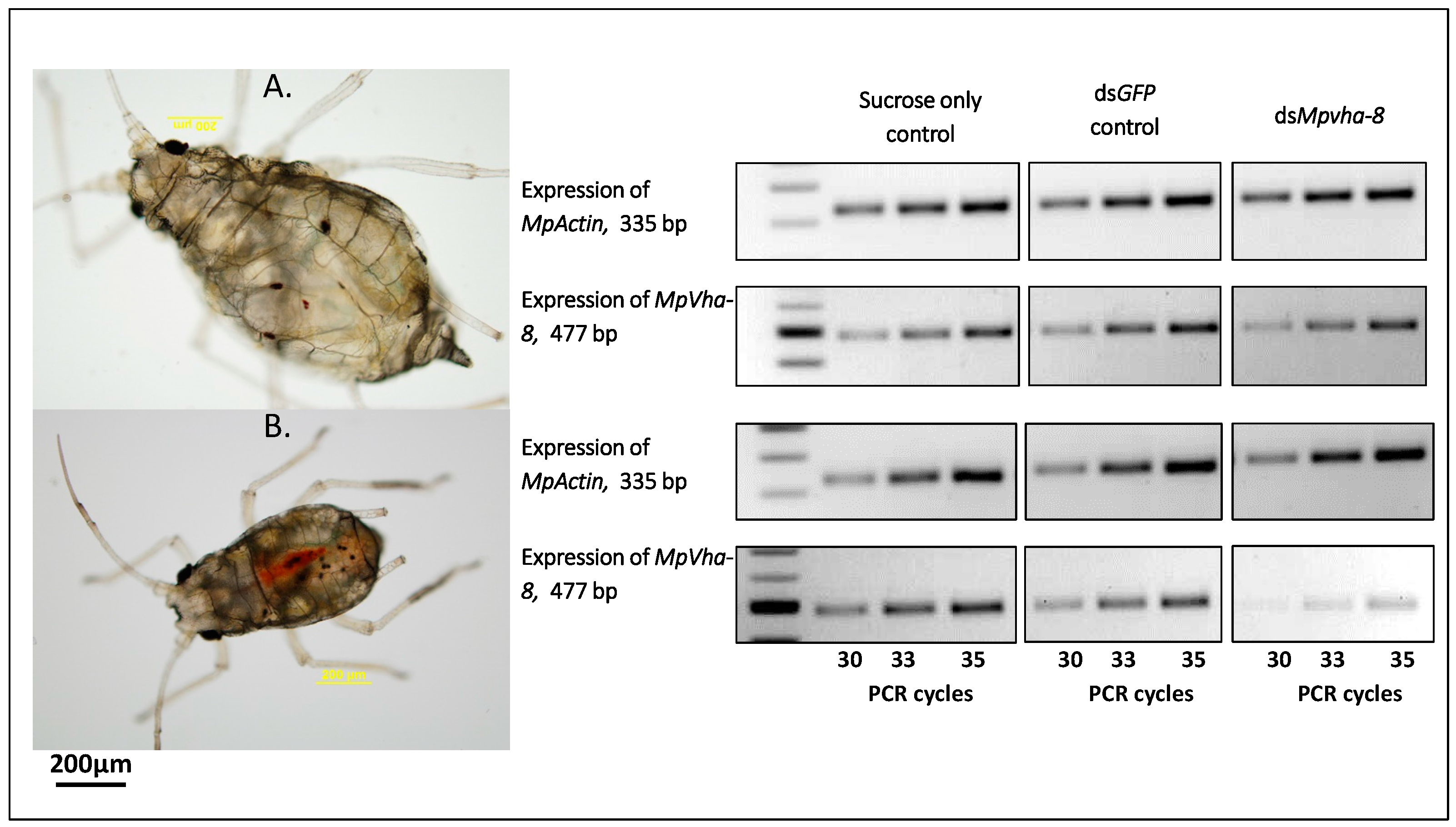
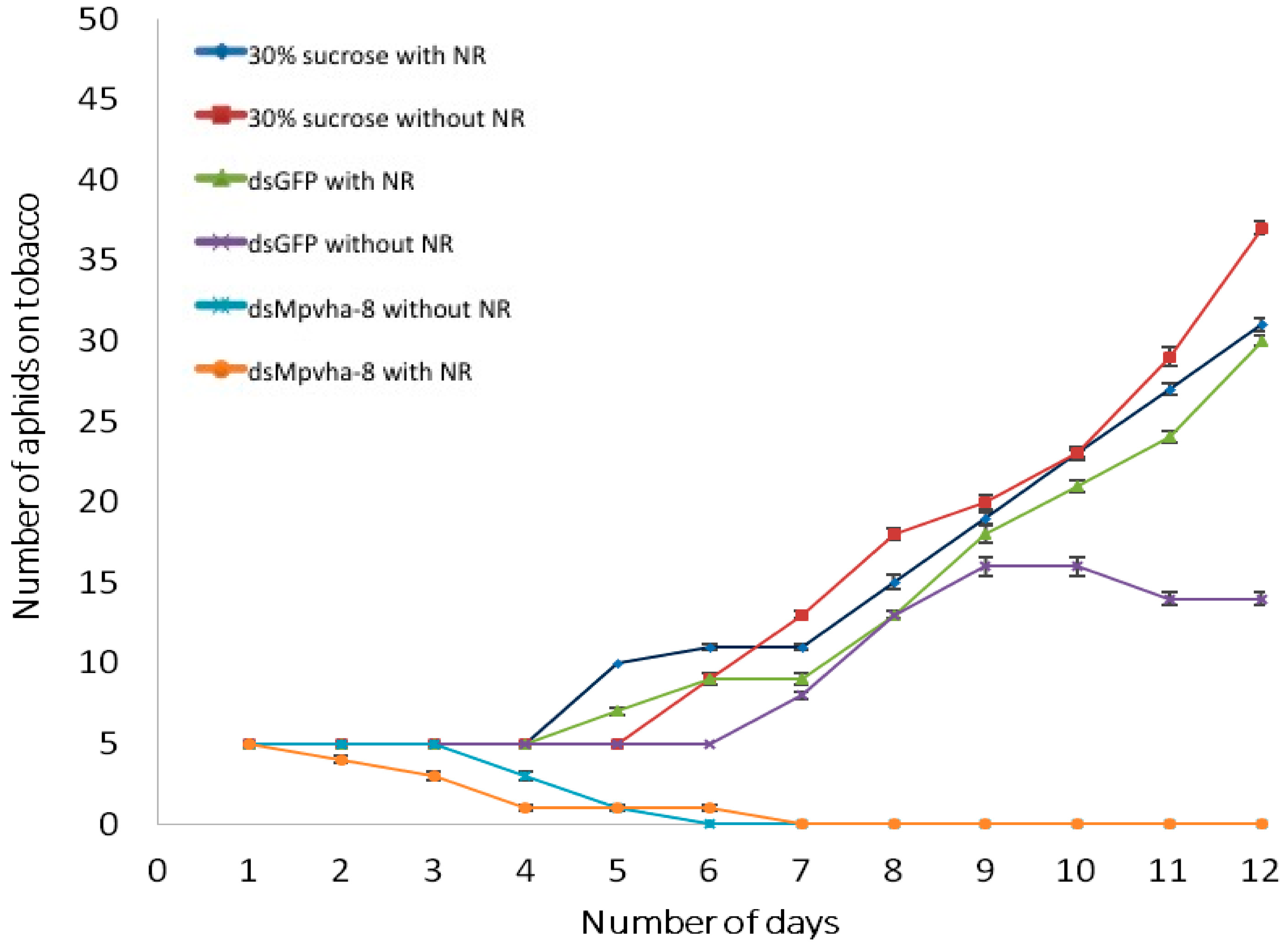
2.5. Tracing Ingestion of dsRNA of Vha-8 by GPAs Allows Efficient Assessment of RNAi Effects
3. Materials and Methods
3.1. Insect Rearing on Tobacco Plants
3.2. In Vitro Artificial Feeding Assays
3.3. Identification and Amplification of MpVha-8
3.4. Semi-Quantitative RT-PCRs
3.5. Synthesis of dsRNA Corresponding to MpVha-8 and GFP
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blackman, R.L.; Eastop, V.F. Aphids on the World’s Crops. An Identification and Information Guide; John Wiley: Chichester, UK, 1984. [Google Scholar]
- Nault, L.R. Arthropod transmission of plant viruses: A new synthesis. Ann. Entomol. Soc. Am. 1997, 90, 521–541. [Google Scholar] [CrossRef]
- Moran, P.J.; Cheng, Y.F.; Cassell, J.L.; Thompson, G.A. Gene expression profiling of Arabidopsis thaliana in compatible plant-aphid interactions. Arch. Insect Biochem. Physiol. 2002, 51, 182–203. [Google Scholar] [CrossRef] [PubMed]
- Blackman, R.L.; Eastop, V.F. Aphids on the World’s Crops: An Identification and Information Guide, 2nd ed.; John Wiley: Chichester, UK, 2000. [Google Scholar]
- Dixon, A.F.G. Aphid Ecology; Chapman & Hall: London, UK, 1998. [Google Scholar]
- Koch, C.K.; Waterhouse, D.F. Distribution and Importance of Arthropods Associated with Agriculture and Forestry in Chile; Aciar Monograph Series, No. 68; Australian Centre for International Agricultural Research: Canberra, ACT, Australia, 2000.
- Baum, J.A.; Bogaert, T.; Clinton, W.; Heck, G.R.; Feldmann, P.; Ilagan, O.; Johnson, S.; Plaetinck, G.; Munyikwa, T.; Pleau, M.; et al. Control of coleopteran insect pests through RNA interference. Nat. Biotechnol. 2007, 25, 1322–1326. [Google Scholar] [CrossRef] [PubMed]
- Fairbairn, D.J.; Cavallaro, A.S.; Bernard, M.; Mahalinga-Iyer, J.; Graham, M.W.; Botella, J.R. Host-delivered RNAi: An effective strategy to silence genes in plant parasitic nematodes. Planta 2007, 226, 1525–1533. [Google Scholar] [CrossRef] [PubMed]
- Fosu-Nyarko, J.; Jones, M.G.K. Application of biotechnology for nematode control in crop plants. Adv. Bot. Res. 2015, 73, 339–376. [Google Scholar]
- Tan, J.-A.C.H.; Jones, M.G.K.; Fosu-Nyarko, J. Gene silencing in root lesion nematodes (Pratylenchus spp.) significantly reduces reproduction in a plant host. Exp. Parasitol. 2013, 133, 166–178. [Google Scholar] [CrossRef] [PubMed]
- Baulcombe, D. RNA silencing in plants. Nature 2004, 431, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Fire, A.; Xu, S.Q.; Montgomery, M.K.; Kostas, S.A.; Driver, S.E. Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998, 391, 806–811. [Google Scholar] [CrossRef] [PubMed]
- Watson, J.M.; Fusaro, A.F.; Wang, M.; Waterhouse, P.M. RNA silencing platforms in plants. FEBS Lett. 2005, 579, 5982–5987. [Google Scholar] [CrossRef] [PubMed]
- Guo, S.; Kemphues, K.J. Par-1, a gene required for establishing polarity in C. elegans embryos, encodes a putative Ser/Thr kinase that is asymmetrically distributed. Cell 1995, 81, 611–620. [Google Scholar] [CrossRef]
- Napoli, C.; Lemieux, C.; Jorgensen, R. Introduction of a chimeric chalcone synthase gene into petunia results in reversible co-suppression of homologous genes in trans. Plant Cell 1990, 2, 279–289. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Zhang, D.; Yao, Q.; Zhang, J.; Dong, X.; Tian, H.; Chen, J.; Zhang, W. Feeding-based RNA interference of a trehalose phosphate synthase gene in the brown planthopper, Nilaparvata lugens. Insect Mol. Biol. 2010, 19, 777–786. [Google Scholar] [CrossRef] [PubMed]
- Clemens, J.C.; Worby, C.A.; Simonson-Leff, N.; Muda, M.; Maehama, T.; Hemmings, B.A.; Dixon, J.E. Use of double-stranded RNA interference in drosophila cell lines to dissect signal transduction pathways. Proc. Natl. Acad. Sci. USA 2000, 97, 6499–6503. [Google Scholar] [CrossRef] [PubMed]
- Eaton, B.A.; Fetter, R.D.; Davis, G.W. Dynactin is necessary for synapse stabilization. Neuron 2002, 34, 729–741. [Google Scholar] [CrossRef]
- Mao, J.; Zeng, F. Feeding-based RNA interference of a gap gene is lethal to the pea aphid, Acyrthosiphon pisum. PLoS ONE 2012, 7, e48718. [Google Scholar] [CrossRef] [PubMed]
- Tian, H.; Peng, H.; Yao, Q.; Chen, H.; Xie, Q.; Tang, B.; Zhang, W. Developmental control of a lepidopteran pest Spodoptera exigua by ingestion of bacteria expressing dsRNA of a non-midgut gene. PLoS ONE 2009, 4, e6225. [Google Scholar] [CrossRef] [PubMed]
- Burand, J.P.; Hunter, W.B. RNAi: Future in insect management. J. Investig. Pathol. 2013, 112 (Suppl. 1), S68–S74. [Google Scholar] [CrossRef] [PubMed]
- Gu, L.; Knipple, D.C. Recent advances in RNA interference research in insects: Implications for future insect pest management strategies. Crop Protect. 2013, 45, 36–40. [Google Scholar] [CrossRef]
- Huvenne, H.; Smagghe, G. Mechanisms of dsRNA uptake in insects and potential of RNAi for pest control: A review. J. Insect Physiol. 2010, 56, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, X.P.; Wang, M.Q.; Ma, W.H.; Hua, H.X. Advances in the use of the RNA interference technique in hemiptera. Insect Sci. 2013, 20, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Li, H.C.; Miao, X.X. Feasibility, limitation and possible solutions of RNAi-based technology for insect pest control. Insect Sci. 2013, 20, 15–30. [Google Scholar] [CrossRef] [PubMed]
- Ghanim, M.; Kontsedalov, S.; Czosnek, H. Tissue-specific gene silencing by RNA interference in the whitefly Bemisia tabaci (gennadius). Insect Biochem. Mol. Biol. 2007, 37, 732–738. [Google Scholar] [CrossRef] [PubMed]
- Jaubert-Possamai, S.; le Trionnaire, G.; Bonhomme, J.; Christophides, G.K.; Rispe, C.; Tagu, D. Gene knockdown by RNAi in the pea aphid Acyrthosiphon pisum. BMC Biotechnol. 2007, 7, 63. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Ding, Z.; Zhang, C.; Yang, B.; Liu, Z. Gene knockdown by intro-thoracic injection of double-stranded RNA in the brown planthopper, Nilaparvata lugens. Insect Biochem. Mol. Biol. 2010, 40, 666–671. [Google Scholar] [CrossRef] [PubMed]
- Mutti, N.S.; Park, Y.; Reese, J.C.; Reeck, G.R. RNAi knockdown of a salivary transcript leading to lethality in pea aphid. J. Insect Sci. 2006, 6, 38. [Google Scholar] [CrossRef] [PubMed]
- Whangbo, J.S.; Hunter, C.P. Environmental RNA interference. Trends Genet. 2008, 24, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Yu, N.; Christiaens, O.; Liu, J.; Niu, J.; Cappelle, K.; Caccia, S.; Huvenne, H.; Smagghe, G. Delivery of dsRNA for RNAi in insects: An overview and future directions. Insect Sci. 2013, 20, 4–14. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Chen, Q.; Lin, Y.; Jiang, T.; Wu, G.; Hua, H. RNA interference in Nilaparvata lugens (homoptera: Delphacidae) based on dsRNA ingestion. Pest Manag. Sci. 2011, 67, 852–859. [Google Scholar] [CrossRef] [PubMed]
- Shakesby, A.J.; Wallace, I.S.; Isaacs, H.V.; Pritchard, J.; Roberts, D.M.; Douglas, A.E. A water-specific aquaporin involved in aphid osmoregulation. Insect Biochem. Mol. Biol. 2009, 39, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Soares, C.A.G.; Lima, C.M.R.; Dolan, M.C.; Piesman, J.; Beard, C.B.; Zeidner, N.S. Capillary feeding of specific dsRNA induces silencing of the isac gene in nymphal Ixodes scapularis ticks. Insect Mol. Biol. 2005, 14, 443–452. [Google Scholar] [CrossRef] [PubMed]
- Rosa, C.; Kamita, S.G.; Falk, B.W. RNA interference is induced in the glassy winged sharpshooter Homalodisca vitripennis by actin dsRNA. Pest Manag. Sci. 2012, 68, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhou, Y.; Wang, H.; Jones, H.; Gao, Q.; Wang, D.; Ma, Y.; Xia, L. Identifying potential RNAi targets in grain aphid (Sitobion avenae F.) based on transcriptome profiling of its alimentary canal after feeding on wheat plants. BMC Genom. 2013, 14, 560. [Google Scholar] [CrossRef] [PubMed]
- Wuriyanghan, H.; Rosa, C.; Falk, B.W. Oral delivery of double-stranded RNAs and siRNAs induces RNAi effects in the potato/tomato psyllid, Bactericerca cockerelli. PLoS ONE 2011, 6, e27736. [Google Scholar] [CrossRef] [PubMed]
- Brenner, R. An in vivo fluorescent marker for spermatozoa of the screwworm (diptera: Calliphoridae): A first report. Proc. Entomol. Soc. Wash. 1984, 86, 714–719. [Google Scholar]
- Van der Goes van Naters, W.; Carlson, J.R. Insects as chemosensors of humans and crops. Nature 2006, 444, 302–307. [Google Scholar] [CrossRef] [PubMed]
- Ribble, D.; Goldstein, N.; Norris, D.; Shellman, Y. A simple technique for quantifying apoptosis in 96-well plates. BMC Biotechnol. 2005, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Strand, M.R.; Roitberg, B.D.; Papag, D.R. Acridine orange: A potentially useful internal marker of hymenoptera and diptera. J. Kans. Entomol. Soc. 1990, 63, 634–637. [Google Scholar]
- Mittler, T.E.; Dadd, R.H. Studies on the artificial feeding of the aphid Myzus persicae (Sulzer)-I. Relative uptake of water and sucrose solutions. J. Insect Physiol. 1963, 9, 623–645. [Google Scholar] [CrossRef]
- Whyard, S.; Singh, A.D.; Wong, S. Ingested double-stranded RNAs can act as species-specific insecticides. Insect Biochem. Mol. Biol. 2009, 39, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, S.K.; Chandrashekar, K.; Thakur, N.; Verma, P.C.; Borgio, J.F.; Singh, P.K.; Tuli, R. RNA interference for the control of whiteflies (Bemisia tabaci) by oral route. J. Biosci. 2011, 36, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Fosu-Nyarko, J.; Tan, J.A.C.H.; Gill, R.; Agrez, V.G.; Rao, U.; Jones, M.G.K. De novo analysis of the transcriptome of Pratylenchus zeae to identify transcripts for proteins required for structural integrity, sensation, locomotion and parasitism. Mol. Plant Pathol. 2015, 17, 532–552. [Google Scholar] [CrossRef] [PubMed]
- Bilgi, V. Effects of Silencing Green Peach Aphid (Myzus persicae) Genes via RNA Interference. Ph.D. Thesis, Murdoch University, Perth, WA, Australia, 2016. [Google Scholar]
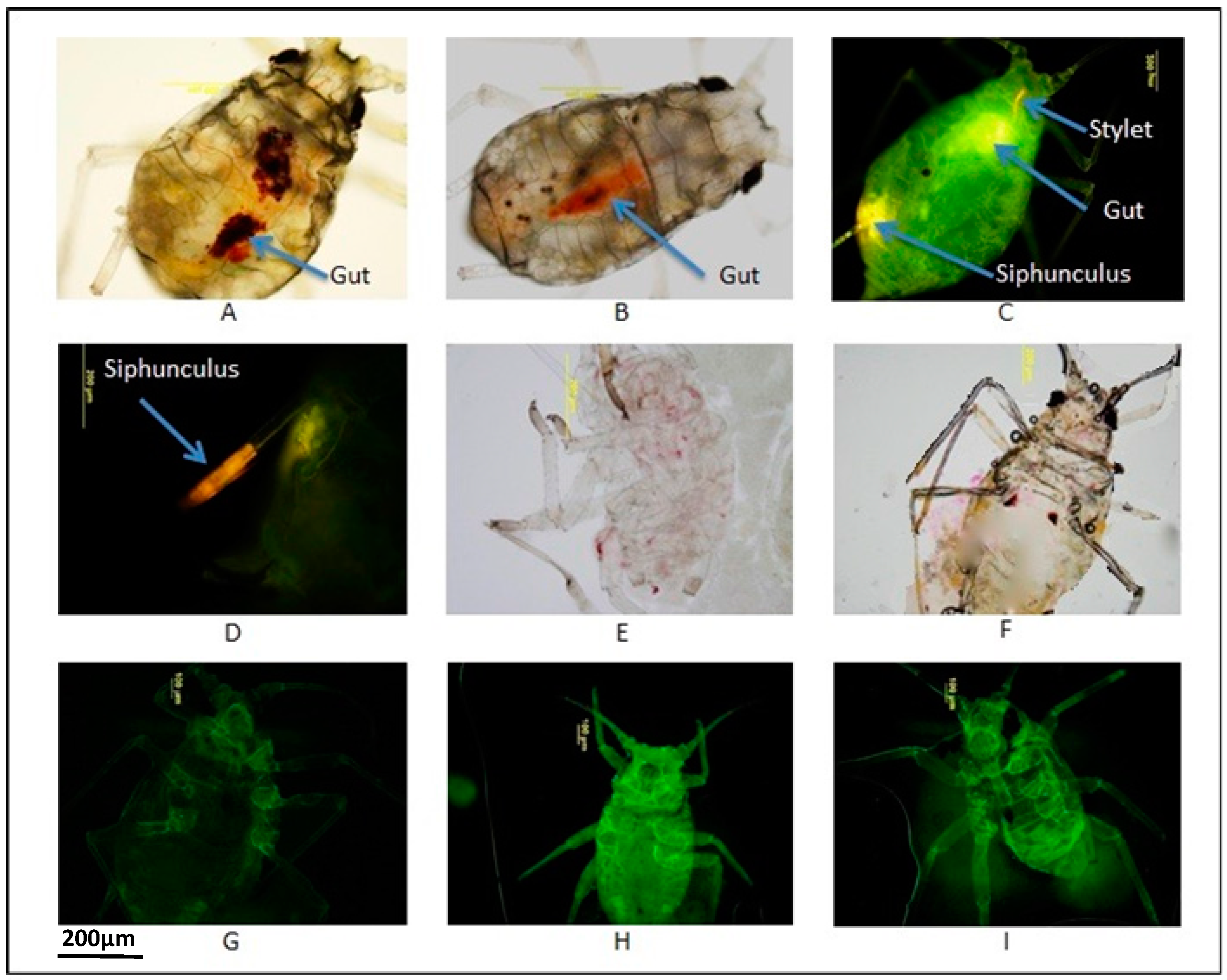


| Dye Tested with 30% Sucrose | Concentration | Number of GPA Active * | Observation of Dye |
|---|---|---|---|
| Fluorescein isothiocyanate | 0.5 mg/mL | 10 | Similar fluorescence as in untreated aphids |
| 1 mg/mL | 10 | Similar fluorescence as in untreated aphids | |
| 2 mg/mL | 12 | Similar fluorescence as in untreated aphids | |
| Fluorescein diacetate | 0.0001 w/v | 13 | Similar fluorescence as in untreated aphids |
| 0.001 w/v | 10 | Similar fluorescence as in untreated aphids | |
| 0.01 w/v | 9 | Similar fluorescence as in untreated aphids | |
| Red food color | 0.50% | 13 | Undetected |
| Yellow food color | 0.50% | 14 | Undetected |
| Aqueous acid fuchsin | 0.25% | 6 | Undetected |
| Aqueous methylene blue | 0.50% | 9 | Undetected |
| Aqueous fast green | 0.50% | 9 | Undetected |
| Phloxine B | 0.75% | 7 | Dye detected in one aphid |
| Congo red | 0.50% | 13 | Dye detected in two aphids |
| Neutral red | 0.50% | 15 | Dye detected in five aphids |
| Acridine orange | 0.05% | 16 | Dye detected in six aphids |
| Treatment | Concentration (%) | Percentage of GPA with Dye |
|---|---|---|
| Neutral red with 30% sucrose | 0.02 | 96.7 |
| 0.05 | 76.7 | |
| 0.1 | 86.7 | |
| 0.3 | 83.3 | |
| 0.5 | 86.7 | |
| Neutral red with water | 0.02 | 73.3 |
| Acridine orange with 30% sucrose | 0.0025 | 10 |
| 0.005 | 90 | |
| 0.01 | 60 | |
| 0.03 | 86.7 | |
| 0.05 | 60 | |
| Acridine orange with water | 0.0025 | 40 |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bilgi, V.; Fosu-Nyarko, J.; Jones, M.G.K. Using Vital Dyes to Trace Uptake of dsRNA by Green Peach Aphid Allows Effective Assessment of Target Gene Knockdown. Int. J. Mol. Sci. 2017, 18, 80. https://doi.org/10.3390/ijms18010080
Bilgi V, Fosu-Nyarko J, Jones MGK. Using Vital Dyes to Trace Uptake of dsRNA by Green Peach Aphid Allows Effective Assessment of Target Gene Knockdown. International Journal of Molecular Sciences. 2017; 18(1):80. https://doi.org/10.3390/ijms18010080
Chicago/Turabian StyleBilgi, Vineeta, John Fosu-Nyarko, and Michael G. K. Jones. 2017. "Using Vital Dyes to Trace Uptake of dsRNA by Green Peach Aphid Allows Effective Assessment of Target Gene Knockdown" International Journal of Molecular Sciences 18, no. 1: 80. https://doi.org/10.3390/ijms18010080
APA StyleBilgi, V., Fosu-Nyarko, J., & Jones, M. G. K. (2017). Using Vital Dyes to Trace Uptake of dsRNA by Green Peach Aphid Allows Effective Assessment of Target Gene Knockdown. International Journal of Molecular Sciences, 18(1), 80. https://doi.org/10.3390/ijms18010080







