1. Introduction
Chronic crebral hypoperfusion (CCH) is a prevalent pathophysiological state in patients with Alzheimer’s disease (AD) and vascular dementia (VaD). CCH has been identified as one of the initial conditions that are critical to the development of cognitive dysfunction [
1]. Current studies have indicated that deranged energy metabolism, glial activation, apoptosis, oxidative stress, neuronal damage, and white matter lesions caused by cerebral hypoperfusion might be the pathophysiological mechanisms that contribute to cognitive impairment [
2,
3]. Despite the detrimental effects of hypoperfusion, the decline in cellular oxygen levels and energy supplies also induces compensatory or endogenous adaptive mechanisms to defend against hypoperfusion injury and to allow recovery of brain function [
2,
4]. In agreement, gradual recovered cerebral blood flow (CBF), increased capillary diameter, neovascularization, and enhanced expression of vascular endothelial growth factor (
VEGF) were noted in the cortex and hippocampus in rodent models of CCH [
2], which indicates the compensatory or adaptive mechanisms. Because of the multifactorial pathogenesis of dementia, a large number of clinical trials which sought to identify protective strategies against this disease have failed, so the recruitment of endogenous neuroprotective pathways represents a potential strategy for developing therapeutics for the cognitive impairment. The regulation of the endogenous adaptive and neuroprotective processes involves the concerted activation of various transcription factors. In the condition of CCH, hypoxia inducible factor 1 (
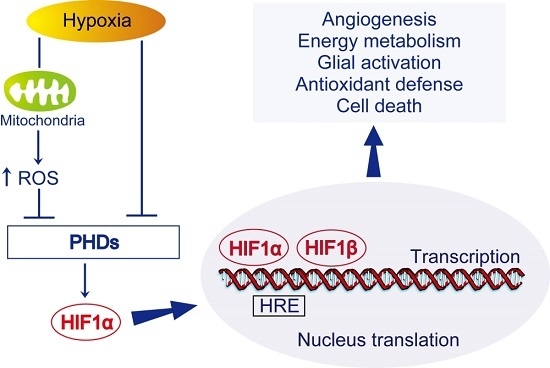
HIF-1) is one of the most important transcription factors involved in endogenous adaptive response.
HIF-1 is a pivotal regulator of gene expression in response to hypoxia or ischemia. It is composed of an oxygen-regulated subunit known as
HIF-1α and a constitutively expressed
HIF-1β subunit. In most cases,
HIF-1 activity is controlled by the availability and activity of the
HIF-1α subunit. Under normoxic conditions,
HIF-1α is hydroxylated and rapidly degraded. Under hypoxic-ischemic conditions, hydroxylases are less active, thus the
HIF-1 complex accumulates and translocates to the nucleus, which results in the transcription of
HIF-1 target genes.
HIF-1 target genes include pro-survival genes involved in angiogenesis, energy metabolism, erythropoiesis, vasomotor control, and cell proliferation, such as vascular endothelial growth factor (
VEGF), glucose transporter-1 (
GLUT-1), and erythropoietin (
EPO), which, in turn, promotes neuronal adaptation for survival under hypoxic conditions [
5,
6].
Neuron-specific knockdown of
HIF-1α aggravates brain damage after a 30 min middle cerebral artery occlusion (MCAO) and reduces the survival rate of MCAO mice [
7]. Inhibiting
HIF-1α hydroxylases to trigger the accumulation of HIF-1α protects the brain from cerebral stroke damage [
8,
9]. Thus,
HIF-1α appears to be a potential target for the development of novel therapeutic interventions in ischemia-related neurological disease.
Paradoxically,
HIF-1α is also involved in hypoxia-dependent inflammation, apoptosis, and cellular stress.
HIF-1α governs the expression of several pro-apoptotic genes including Bcl-2/adenovirus EIB 19 kDa-interacting protein 3 (
BNIP-3), NADPH oxidase activator 1 (
Noxa), NIP3-like protein X (
NIX) [
10,
11,
12]. Brain-specific knockout of
HIF-1α may be neuroprotective after acute global ischemic injury for 75 min, and genes involved in apoptotic pathways are downregulated in
HIF-1 knockout mice [
13].
Consequently, both neuroprotective and detrimental effects of
HIF-1α have been observed in animal models of acute ischemia [
14]. The role for
HIF-1α in mediating either pro-death or pro-survival responses may depend on the severity and duration of the ischemia insult [
15] and the types of pathological stimuli [
16]. Mild hypoxia induced adaptive gene expression can occur transiently or persistently depending on the duration of the stimulus, whereas with severe or sustained hypoxic conditions
HIF-1α can promote apoptotic cell death [
17].
CCH is a less severe form of ischemia that develops more slowly than acute ischemic stroke [
18,
19], therefore,
HIF-1α may play a more important role in this pathological process. Our previous study has shown that chronic global cerebral hypoperfusion triggered long-lasting accumulation of
HIF-1α protein in rat hippocampus [
20]. However, the role of the stabilized
HIF-1α during CCH is still obscure. The aim of this study was to investigate the role of endogenous
HIF-1α stabilization during CCH in rats.
3. Discussion
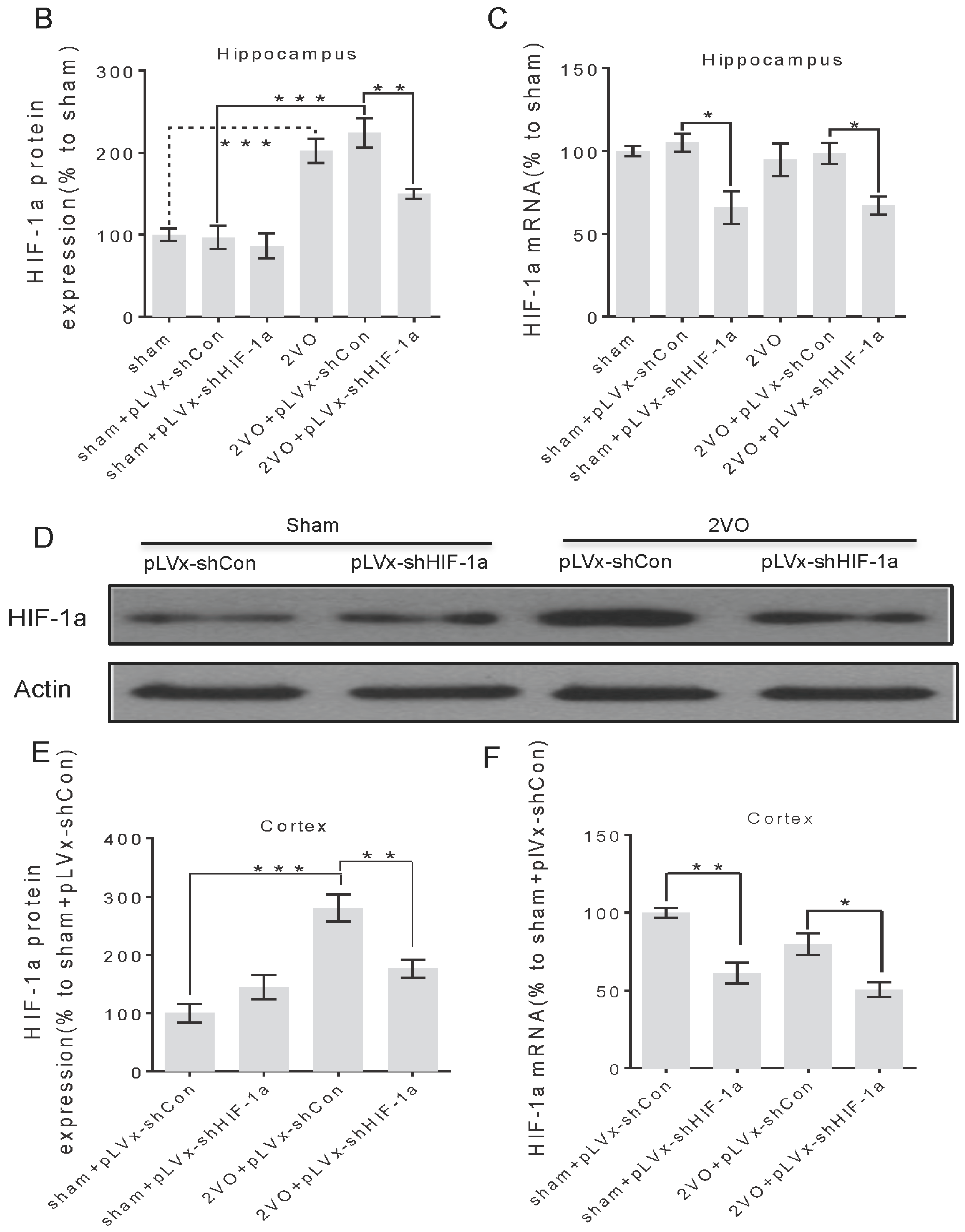
We previously reported that CCH induced by 2VO triggered a robust and long-lasting HIF-1α protein accumulation in rats: both pro-apoptotic and pro-survival downstream genes were upregulated only during the early stage after 2VO. Based on this finding, we further investigated the role of HIF-1α in the chronic stage following 2VO. To this end, we knocked down HIF-1α using lentivirus-mediated shRNAi against HIF-1α mRNA. The results demonstrated that the downregulation of HIF-1α accumulation during 2VO reduced the compensatory angiogenesis, and aggravated neuronal damage and cognitive impairment induced by 2VO.
3.1. Lentivirus-Mediated shRNAi Lowers the Expression of HIF-1α In Vivo and Reduces the Accumulation of HIF-1α during CCH
RNA interference (RNAi) has been established as a powerful tool to characterize gene function in mammals [
22]. RNAi is triggered by the presence of double-stranded RNA in the cell and results in rapid destruction of the mRNA containing the identical sequence, exerting its gene-silencing activity. Recombinant lentiviral vector is particularly useful for gene therapy in CNS injury because of its safety and stability. In this study, we knocked down
HIF-1α using shRNAi (short-hairpin RNAi) against
HIF-1α mRNA, introduced into brain cells using lentivirus. The lentivirus could efficiently transfect bilateral cortices and hippocampus one week after being injected into the bilateral hippocampus and ventricles, and lower
HIF-1α mRNA expression one week post-transfection. It ensures that the
HIF-1α mRNA expression can be decreased before treating rats with 2VO operation. Moreover, lentiviral-mediated shRNAi can reduce endogenous
HIF-1α protein accumulation under hypoperfusion.
3.2. HIF-1α Accumulation during CCH Protects against Cognitive Impairment via Its Protective Effects
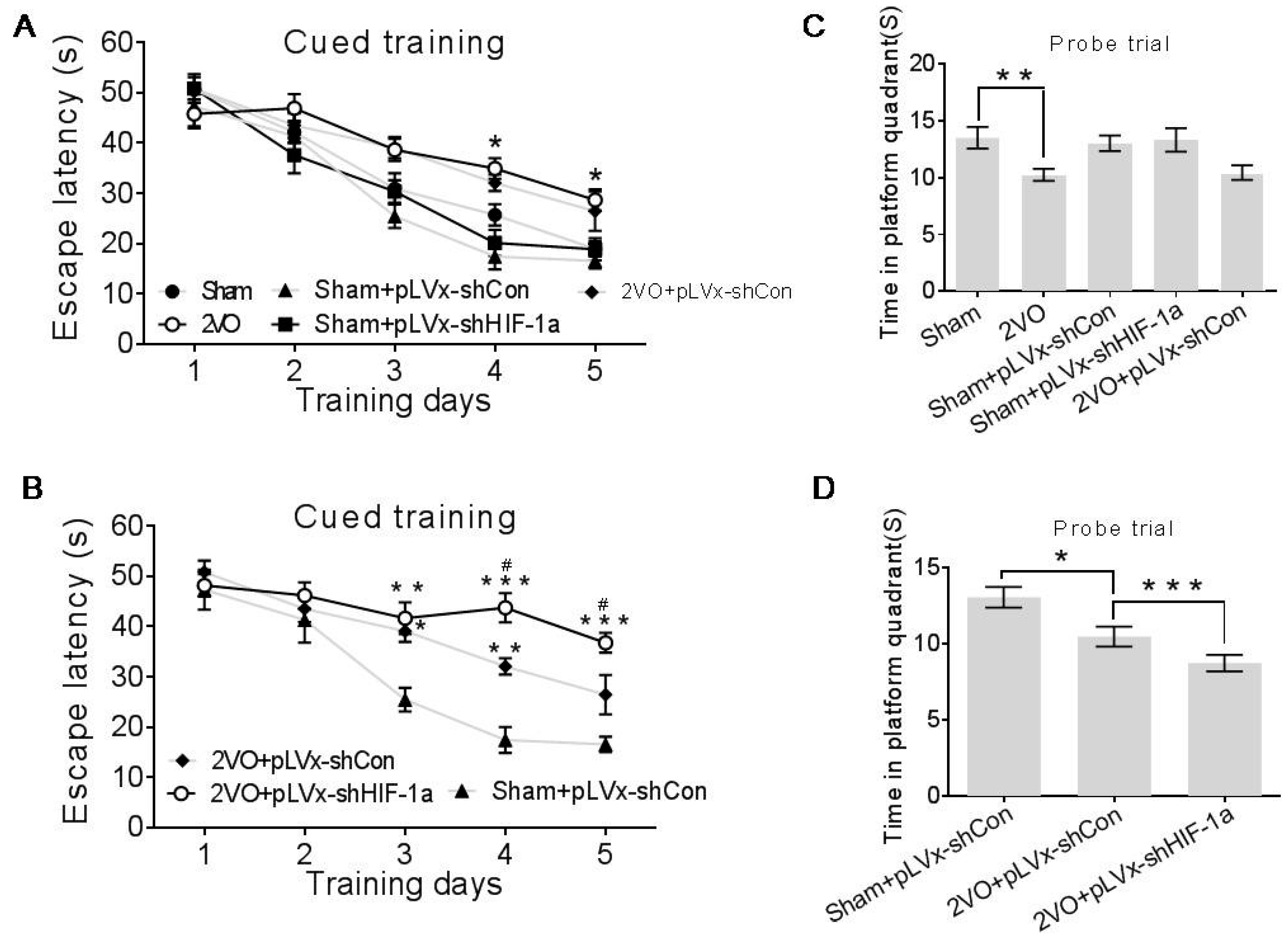
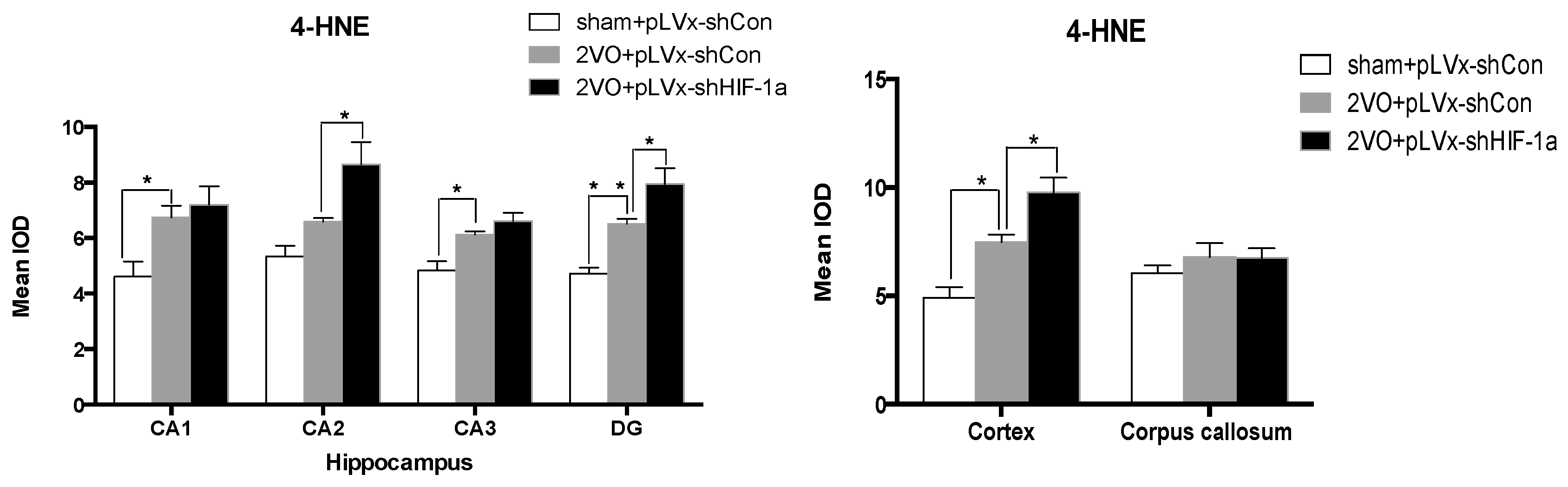
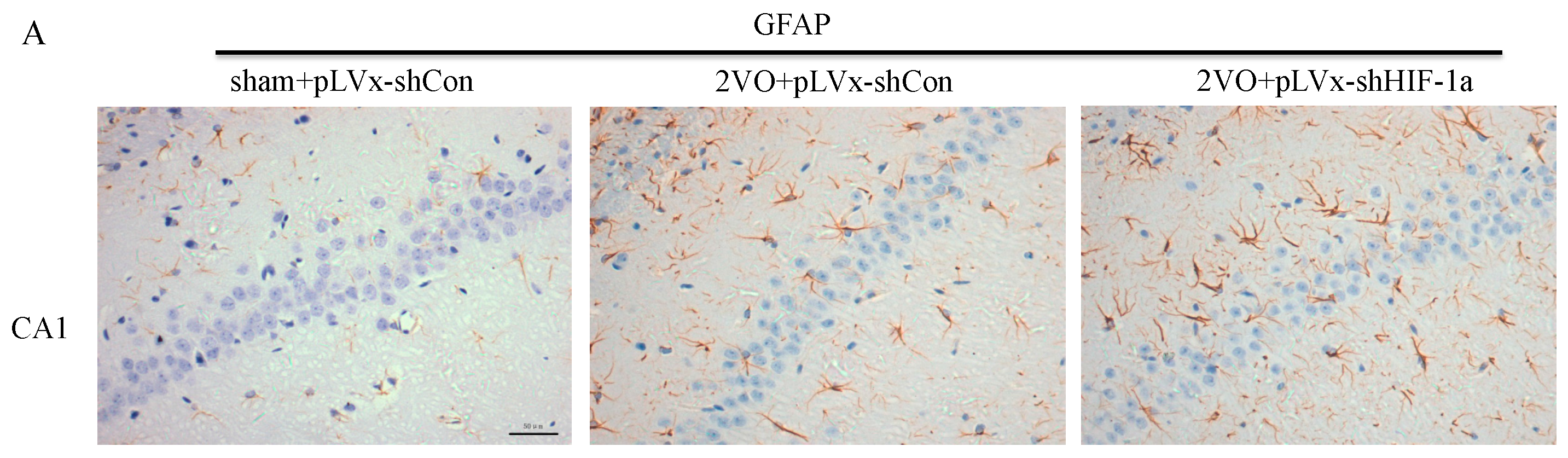
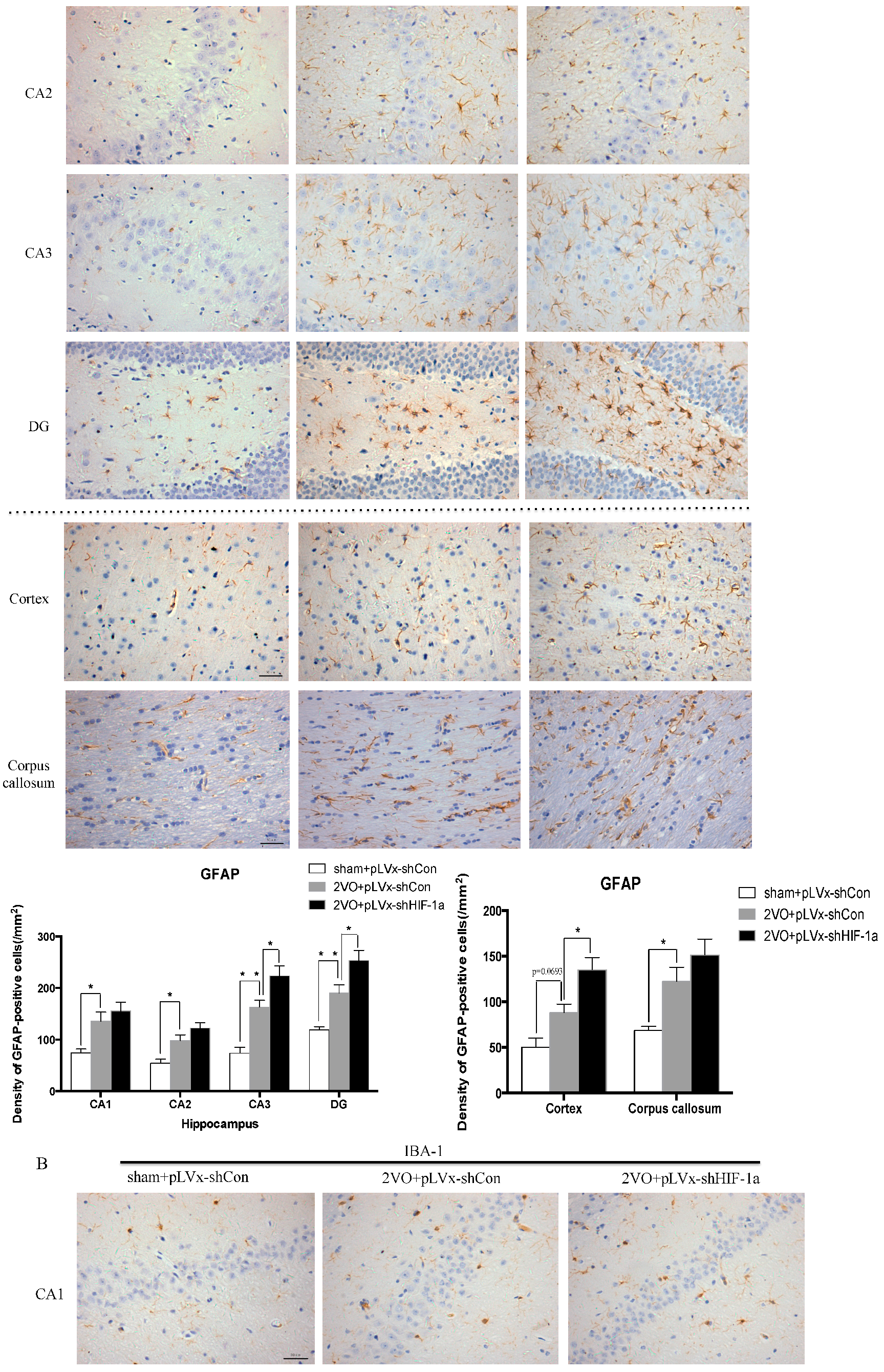
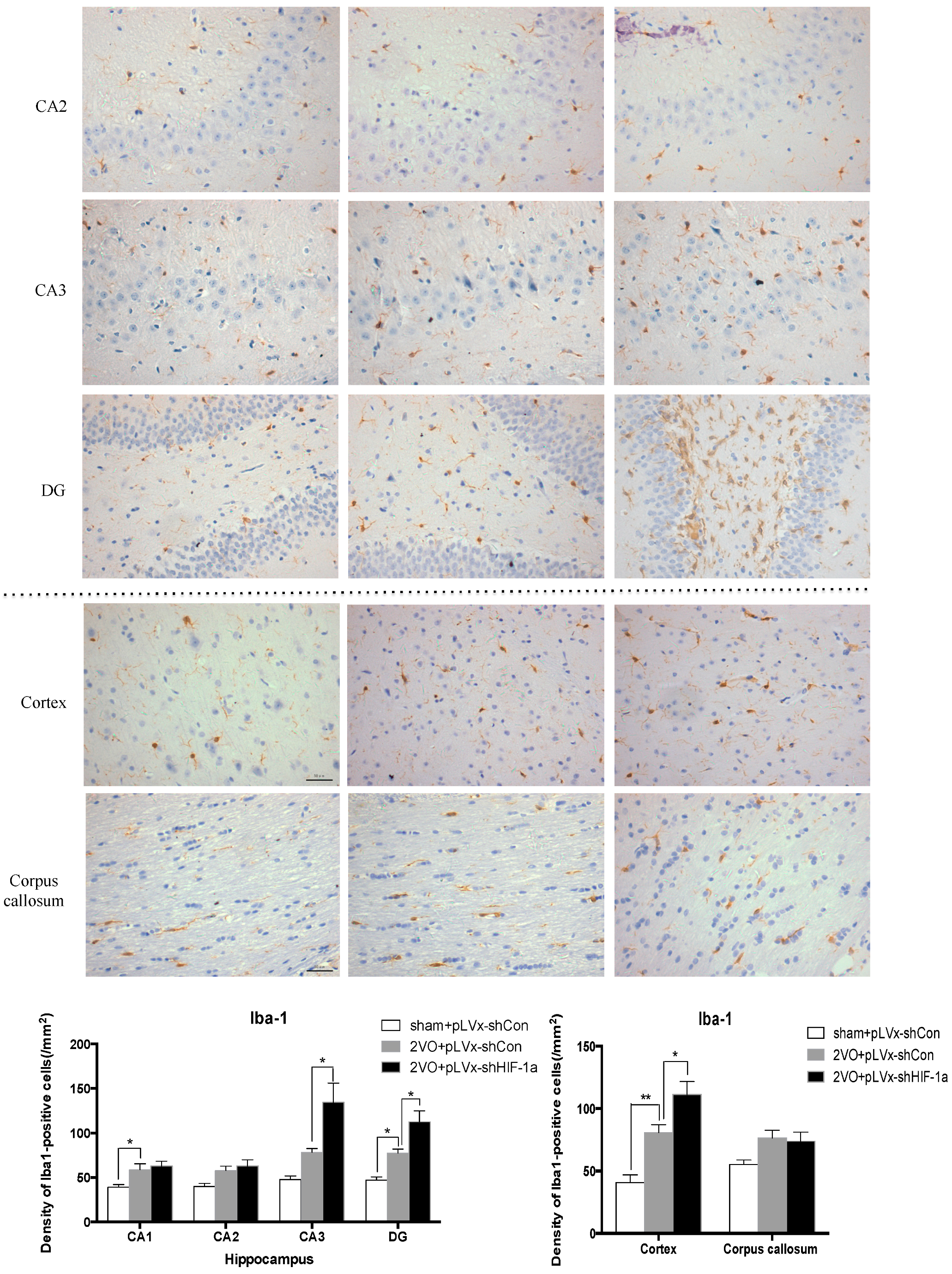
Our results suggest that reducing HIF-1α accumulation during CCH suppressed CCH-induced angiogenesis in the cortex, and enhanced oxidative damage and glial activation in the cortex and hippocampus. Moreover, it aggravated the CCH-induced spatial learning and memory deficits.
Considerable evidence indicates that CCH contributes to the development and progression of dementia through pathways of inflammation and oxidative stress and results in damage to learning and memory functions [
23]. Oxidative stress has been regarded as a common pathophysiological change and an essential factor in the development of CCH-related cognitive impairment [
3]. Studies focused on the neuroprotective effects of antioxidants indicated that antioxidants could attenuate oxidative damage in rats’ brains and reverse the cognitive deficits induced by CCH [
24,
25,
26]. Inflammatory-related microglia and astrocytes are also considered to be involved in the cognitive impairment in CCH [
27]. Previous studies have demonstrated that chronic cerebral hypoperfusion could induce the activation of inflammatory glial cells [
28]. These inflammatory glial cells are directly toxic to neurons, which causes particularly severe damage to the central nervous system [
29]. Consistent with previous reports, our study verified that CCH by 2VO triggered oxidative damage and glial activation. Moreover, when HIF-1α protein accumulation in CCH was reduced, the oxidative damage and glial activation were even more severe. HIF-1α regulated downstream genes
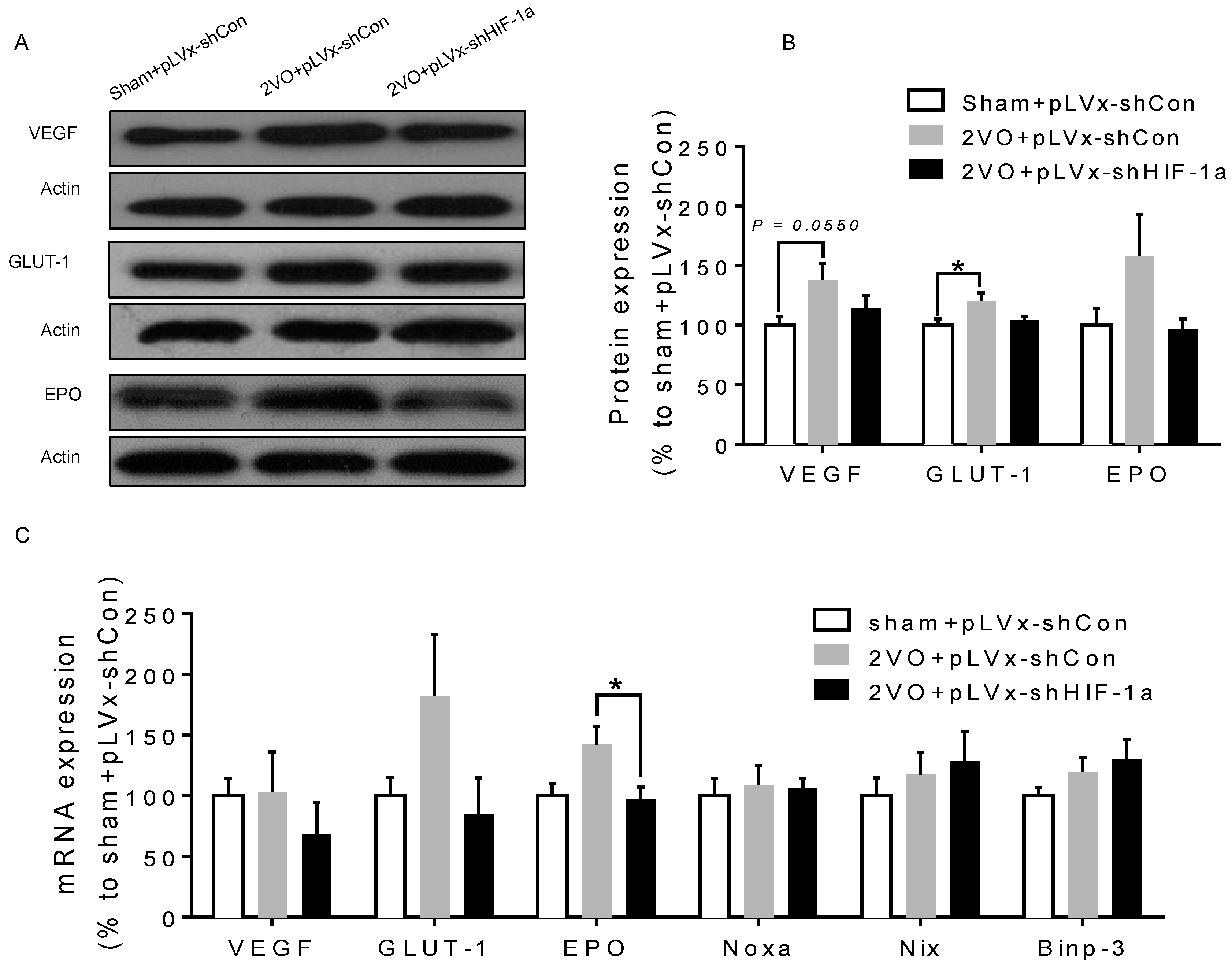
VEGF and
EPO have direct neurotrophic effects and can prevent neuronal injury due to oxidative stress evoked by hypoxia or ischemia and can enhance recovery following stroke [
16].
GLUT-1 is also a
HIF-1α target gene which could enhance the concentration of glucose and thereby ameliorate neuroprotection. Glucose transport and glycolytic flow as a result of
HIF-1α activation by hypoxia has been linked to cell survival [
30]. Thus, we concluded that
HIF-1α is required for neuroprotection from CCH.
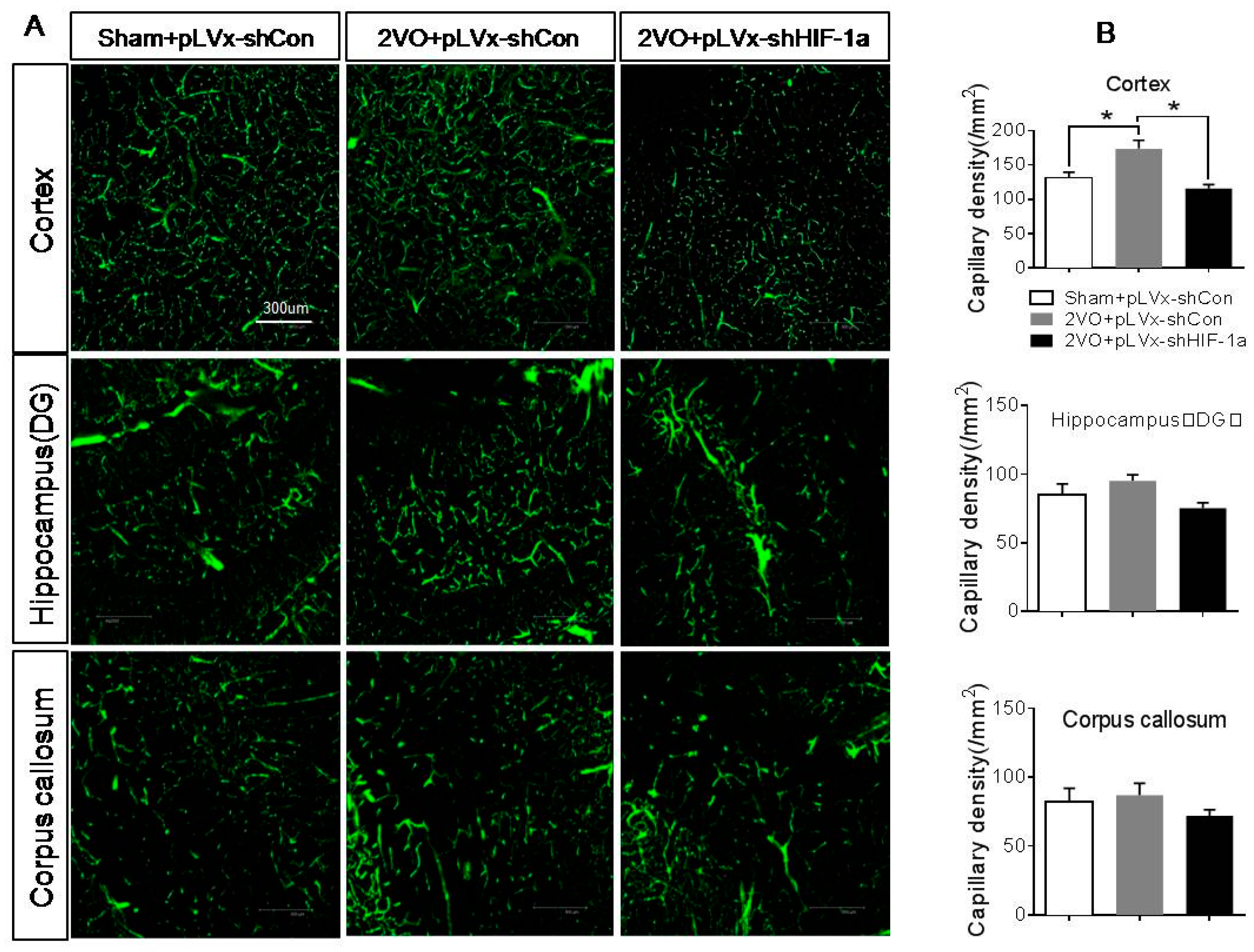
Besides the detrimental effects caused by CCH, some compensatory and endogenous responses can also be induced during CCH in order to adapt to the decreased perfusion. As our study indicated, the density of perfused cerebral capillaries was increased after 2VO, especially in the cortex. During cerebral ischemia, the damaged tissue tries to increase oxygen delivery by the induction of angiogenesis. HIF-1α-regulated cytokines (VEGF and EPO), which have been implicated in angiogenesis, were increased after CCH as well. The angiogenesis induced by CCH was decreased in HIF-1α knockdown rats which further confirmed that HIF-1α is involved in endogenous adaptive response.
Bartosz et al. showed that hypoxia inducible factor stabilization leads to increased expression of
VEGF and
EPO in the hippocampus and causes lasting improvement of hippocampal memory in healthy mice [
31]. Our research also suggests that activation of
HIF-1 appears to have a beneficial role in the hypoperfusion brain as indicated by decreased angiogenesis, and increased oxidative damage and glial activation in
HIF-1α-knockdown rats. Therefore,
HIF-1α accumulation induced by 2VO contributes to endogenous neuroprotective responses. Aggravated learning and memory disorders were associated with more severe neuronal damage caused by suppressing HIF-1α.
3.3. The Role of HIF-1α in the Regulation of Target Genes
The effect of
HIF-1 is determined by its regulation of target genes during hypoxia or ischemia. Here, we observed that 2VO triggered the increases of protein levels of
HIF-1 target genes (
VEGF,
GLUT-1) involved in angiogenesis and energy metabolism, which was in line with the increased accumulation of
HIF-1α protein. Lentiviral-mediated shRNAi could prevent the increase of protein levels of
HIF-1-target pro-survival genes by suppressing
HIF-1α accumulation. However, no significant changes were observed in the expressions of pro-apoptosis genes. Our previous study also found that the tendency of an increase in pro-survival genes was sustained for at least one week, whereas the increase in pro-apoptosis genes was relatively temporary [
20].
HIF-1 may regulate pro-survival genes more than pro-apoptosis genes, explaining why the neuroprotective effects of
HIF-1 prevailed over its pro-death effects during CCH. Thus, it is possible that the reduction in the expression of some pro-survival genes in shRNAi-treated 2VO rats may be attributed to a reduction of neuronal
HIF-1 activity, which results in a suboptimal adaptive response to hypoperfusion that exacerbates the neuronal damage and cognitive impairment.
In addition to
VEGF,
GLUT-1, and
EPO, some other
HIF-1 downstream genes involved in the regulation of energy metabolism, oxidative stress, and mitochondrial integrity may also contribute to the neuroprotective role of
HIF-1. These genes include manganese superoxide dismutase (
MnSOD), pyruvate dehydrogenase (
PKD-1), peroxisome proliferator-activated receptor-c coactivator-1a (
PGC-1), etc. [
5,
32]. Further measuring the expressions of these genes may be useful to support the beneficial role of
HIF-1.
4. Materials and Methods
4.1. Animals
Rats for this experiment were obtained from Vital River Laboratory Animal Technology Co., Ltd. (Beijing, China). Their use was approved by the Animal Ethics Committee of the Medical School of Wuhan University (WHAE0321178, 21 April 2013). The procedures performed all followed protocols approved by the National Institutes of Health Guide for the Care and Use of Laboratory Animals. 160 male Wistar rates aged 7 weeks and weighing between 220 and 250 g at the outset were obtained. Housing conditions were as follows: group-housing in polypropylene cages, ad libitum access to food and water, a temperature of 22 ± 2 °C, humidity levels of 55% ± 5%, and a twelve-hour artificial dark/light cycle (light from 7:00 a.m.–7:00 p.m.).
4.2. Experimental Design
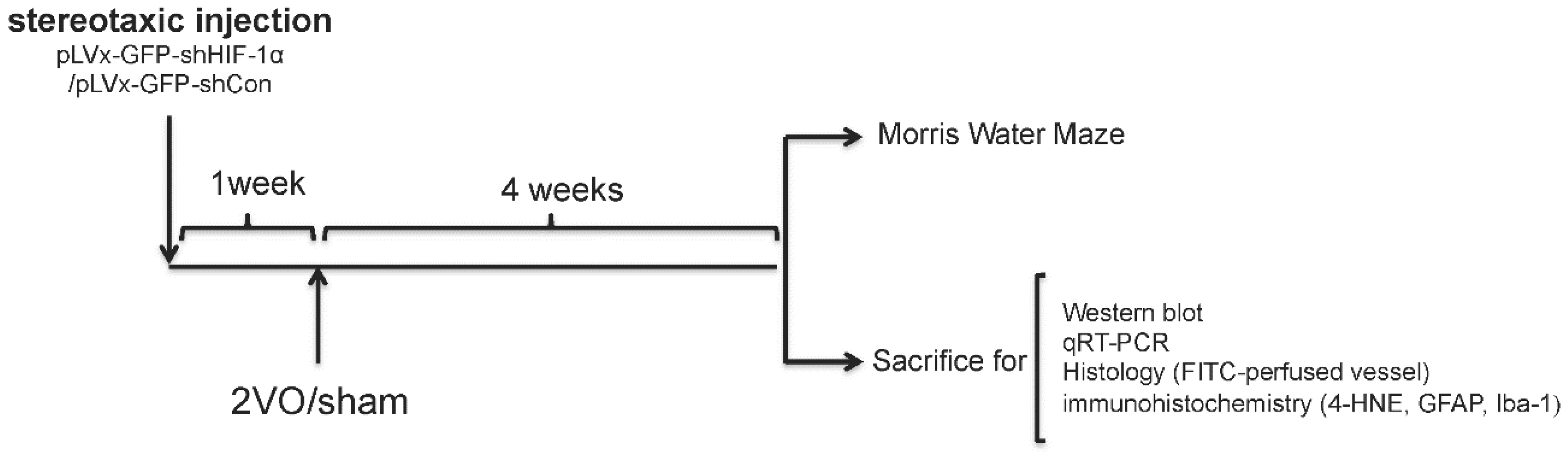
We used RNA interference (RNAi) technology to suppress HIF-1α expression in the rat brain. Lentivirus constructs that express green fluorescent protein (GFP) and short-hairpin RNA (shRNA) directed against the HIF-1α mRNA (pLVx-GFP-shHIF-1α) were stereotaxic injected into the bilateral hippocampus and bilateral ventricles to knockdown HIF-1α gene expression in the hippocampus and some other brain areas. As a negative control, lentivirus constructs that express GFP and shRNA with a scrambled sequence (pLVx-GFP-shCon) were injected in the same way. Rats without stereotaxic injection were used as blank controls.
One week after stereotaxic injection, permanent bilateral common carotid artery occlusion (2-vessel occlusions (2VOs)) was used to induce chronic global cerebral hypoperfusion in the rats. Spatial learning and memory performances were evaluated four weeks after 2VO via the Morris Water Maze (MWM) test. Samples for Western blot assays, quantitative real-time (qRT)-PCR, histopathological and immunohistochemical analyses were obtained from the brains of other rats without performing the MWM test.
The experimental design is illustrated in
Figure 8.
4.3. Construction of Lentivirus Vectors
The lentivirus vectors (pLVx-GFP-shHIF-1α and pLVx-GFP-shCon) used in our study were constructed by Land Biology Technology Co., Ltd. (Shenzhen, China).
The HIF-1α short hairpin RNA interference (shRNAi) sequence was as follows: 5′-gatccAATCAAAAGCAGTGACGAActcgagTTCGTCACTGCTTTTGATtttttg-3′ (Hif1aBamHI F) and 5′-aattcaaaaaAATCAAAAGCAGTGACGAActcgagTTCGTCACTGCTTTTGATTGg-3′ (Hif1aE croRI R). HIF-1α target sequence is 5′-AATCAAAAGCAGTGACGAA-3′, which corresponds to bases 238–256 bp of the HIF-1α mRNA (GenBank accession number NM_023459). shRNAi with a random sequence was also designed to use as a control lentivector.
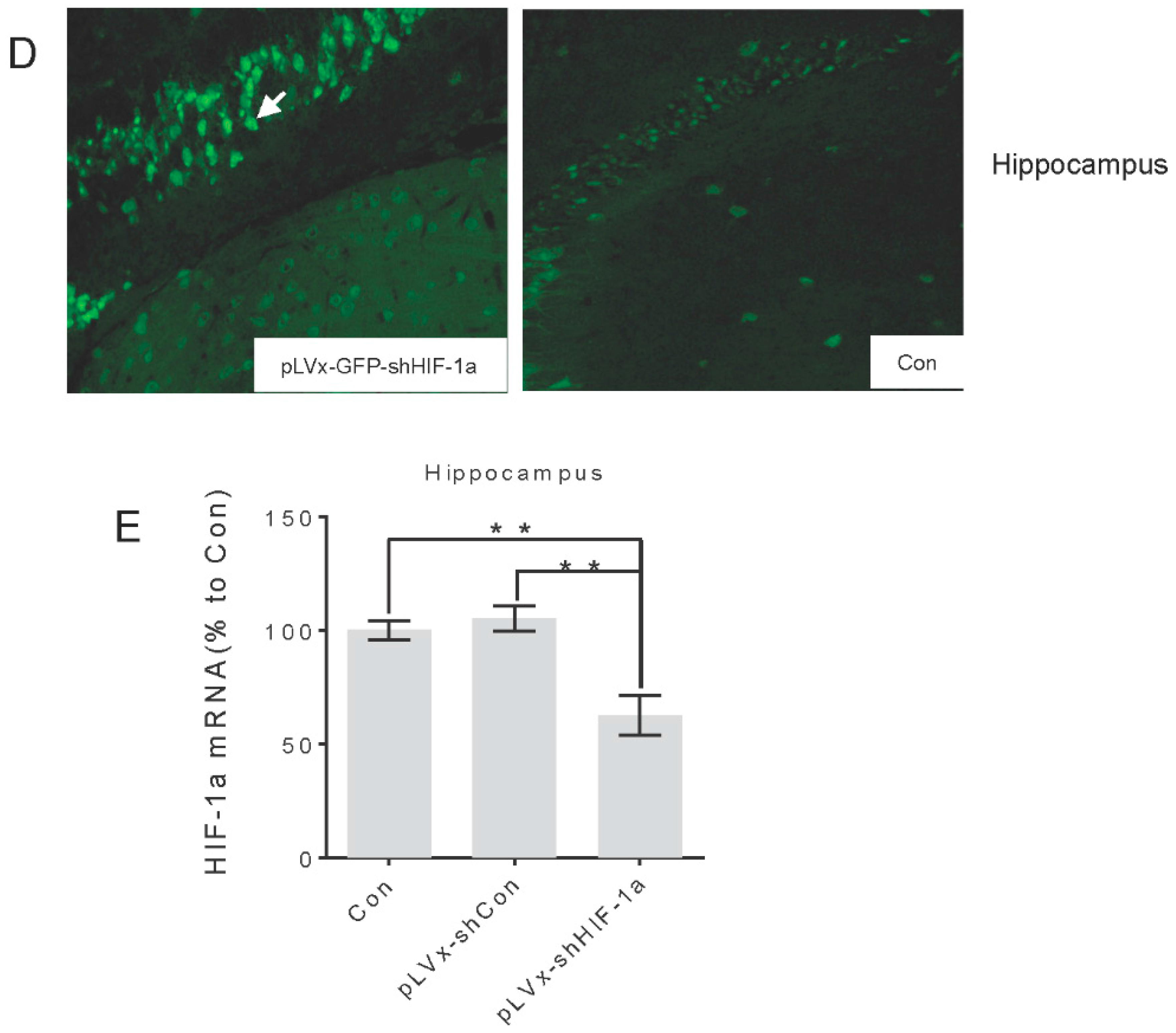
The titers of pLVx-GFP-shHIF-1α vectors is 2.5 × 108 transducing U/mL. Application of pLVx-GFP-shHIF-1α resulted in significantly lower expression of HIF-1α mRNA (>70%) in 293T cells. (These results are from Land Biology Technology Co., Ltd.).
Virus suspensions were stored at −80 °C until use and were briefly centrifuged and kept on ice immediately before injection.
4.4. Stereotaxic Injection of the Lentiviral Vectors
After anesthesia with 10% chloral hydrate (350 mg/kg, intraperitoneal injection), rats were placed into a stereotaxic frame (RWB Life Science, Shenzhen, China). In order to transfect neurons in as many brain areas as possible, pLVx-GFP-shHIF-1α or pLVx-GFP-shCon was injected into the bilateral hippocampus and bilateral ventricles. Rats without stereotaxic injection were used as blank controls.
Two microliters of pLVx-GFP-shHIF-1α or pLVx-GFP-shCon were injected into the hippocampus using a 10 µL Hamilton syringe with a 33 gauge tip needle (Hamilton). Injection coordinates relative to the bregma were as follows: anteroposterior, −4.00 mm; mediolateral, ±2.5 mm; dorsoventral −3.8 mm below the surface of skullusing coordinates derived from the atlas of Paxinos and Watson (Version 2004). Four microliters of pLVx-GFP-shHIF-1α or pLVx-GFP-shCon were injected into bilateral ventricles. Injection coordinates relative to the bregma were as follows: anteroposterior, −1.00 mm; mediolateral, ±1.5 mm; dorsoventral, −3.6 mm below the surface of dura using coordinates derived from the atlas of Paxinos and Watson (Version 2004).
The injection rate was 0.5 μL/min. At the end of the injection, the needle was maintained in the place for another 5 min and then withdrawn very slowly to prevent backflow of solution.
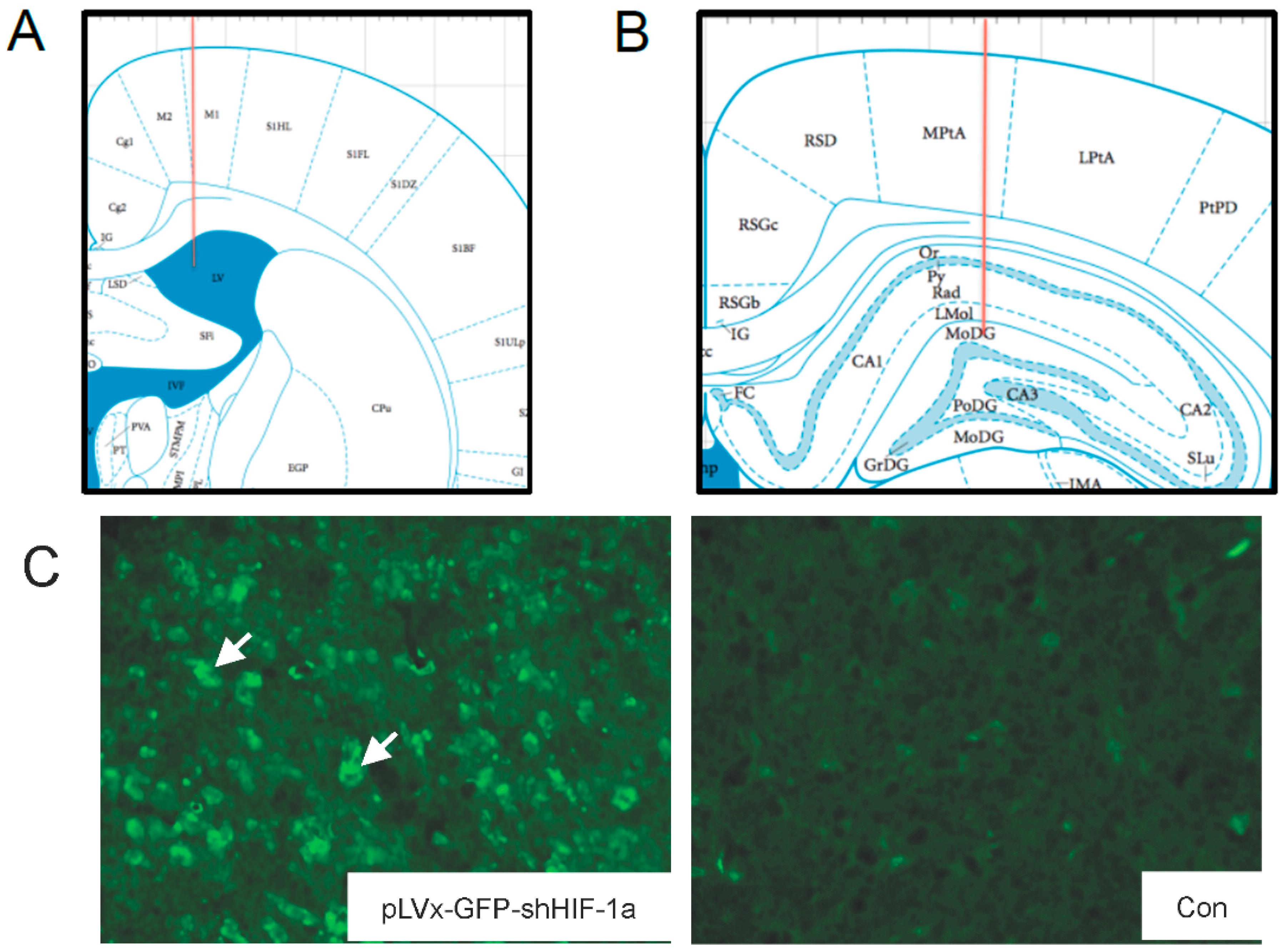
The accuracy of injection sites was identified by the Evans blue (Sigma; St. Louis, MO, USA) stereotaxic injection directly into the hippocampus and ventricle. The needle track in the ventricle and hippocampus was shown in
Figure 1A,B.
Green fluorescent protein (GFP) was observed one week after injection under fluorescence microscope for detection of virus delivery. To test the interference efficiency, the hippocampus was chosen as representative and its HIF-1α mRNA level was detected by qRT-PCR one week after injection of lentiviral vectors.
4.5. 2VO Surgery
One week after stereotaxic injection, the 2VO surgery to induce CCH was carried out as previously described [
33]. Food and water were withheld for one day prior to surgery. Under chloral hydrate (350 mg/kg, i.p.) anesthesia, a midline ventral incision was used to expose the bilateral common carotid arteries. The arteries were carefully separated from the vagal nerves and permanently ligated with silk sutures. The procedure was the same for the sham operations but for the ligation of the common carotid arteries. The surgical wounds were then sutured and the rats were returned to their cages once they had recovered from the anesthesia. Body temperature of the rats was maintained at 37.5 ± 0.5 °C throughout surgery.
4.6. Morris Water Maze Performance
Spatial learning and memory were measured using a Morris Water Maze (MWM) four weeks after the 2VO surgery.
The Maze consisted of a 2.0 m-diameter round pool filled to 32 cm deep with 20 ± 1 °C water. The surface area of the tank was divided into four equal quadrants. A platform (9 cm in diameter and 30 cm in height) was placed inside the tank and was not visible. During the entire test, the platform was kept in the same quadrant. The walls of the tank, the water and the platform were made opaque with nontoxic black paint. The surroundings were dimly lit, and the illumination was kept constant throughout the test. A number of orientation cues were placed around the tank to aid the rats in learning the location of the platform. Cued training consisted of four trials per day for the first five days. Each day the rats were released at a different location into the water facing the pool walls. Rats were given 60 s to find the platform without assistance before being guided to it and allowed to rest there for at least 20 s. The escape latencies were recorded during the 5-day cued training after which the platform was removed from the pool and each rat was given one 30-s swim probe trial (day 6); the time spent in the target quadrant in which the platform had previously been located was recorded. Data were acquired using the SMART 2.5 computerized video imaging analysis system (Panlab, Barcelona, Spain).
4.7. Western Blot
Immunoblotting analysis kits were obtained from the Keygen Institute of Biotechnology (Keygen, Nanjing, China). Protein samples were extracted from the hippocampus and cortex of the rat-nuclear protein samples for HIF-1α protein analysis, and total proteins for HIF-1 downstream protein analysis. Samples of 40 µg each underwent the following procedure: fractionation with 10% SDS-PAGE; transfer to 0.45-mm PVDF membranes (Invitrogen, Carlsbad, CA, USA) at 200 mA for one hour; staining with Ponceau red to confirm equal loading. For one hour at room temperature the blots were blocked with 5% nonfat milk in Tris-buffered saline containing 0.1% Tween 20 (TBST). Overnight incubation (at 4 °C) of the membranes with primary antibodies (anti-HIF-1α, anti-VEGF, anti-GLUT-1, and anti-EPO, 1:1000 dilution in TBST, BD Transduction Laboratories, San Diego, CA, USA; anti-β-actin, 1:2000 in TBST, Santa Cruz, Dallas, TX, USA) was performed. The membranes were then washed three times with TBST and a 30 min incubation with peroxidase-conjugated secondary antibodies (anti-mouse: 1:3000 in TBST, KPL, Gaithersburg, MD, USA) was performed. The membranes were again washed three times in TBST and chemiluminescence (SuperSignal West Pico, Pierce, Rockford, IL, USA) was used to visualize labeled proteins. Optical density analysis (HPIAS 2000, Tongji Qianping Company, Wuhan, China) was used for semi-quantification of immunoreactive bands on X-ray film with all densities normalized to β-actin.
4.8. qRT-PCR
qRT-PCR was used to observe the mRNA levels of HIF-1α and its downstream genes after 2VO and HIF-1α knockdown.
The Trizol method (Invitrogen, Carlsbad, CA, USA) was used on total RNA extracted from the hippocampus and cortex. A First Strand cDNA Synthesis kit (TOYOBO, Kita-ku, Japan) was used on 4 µg of RNA to reverse transcribe it to cDNA, of which 0.2 µg was then amplified using a qRT-PCR kit. The reaction used THUNDERBIRD SYBR qPCR Mix (TOYOBO, Kita-ku, Japan). The gene-specific primers used can be seen in
Table 1. The following PCR cycles were applied: initial denaturation at 50 °C for 2 min, then 95 °C for 2 min; 40 cycles of 15 s at 95 °C, annealing at 58 °C for 15 s, and extension at 72 °C for 45 s; and extension at 72 °C for 10 min. β-actin was used as an endogenous control and reactions were all performed in triplicate. The formula (2
-ΔΔCt) was applied to express changes in mRNAs.
4.9. Density of Capillaries
To identify the network of capillaries and evaluate angiogenesis after 2VO and HIF-1α knockdown, rats were anesthetized with 10% chloral hydrate (350 mg/kg, i.p.) and transcardially perfused at 4 weeks after 2VO with 5 mL FITC-dextran (2 × 106 molecular weight, Sigma; St. Louis, MO, USA; 50 mg/mL) at a perfusion pressure of 120–140 mmHg. The perfused vessels can be marked by FITC-dextran. The brains were removed, immersed in 4% paraformaldehyde at 4 °C for 48 h. Coronal sections (150 µm thick) were made by vibratome microtome, and fluorescence images were obtained using a laser scanning confocal microscope (ZEISS LSM 700, Carl Zeiss AG, Germany) under 488 nm wave excitation. Cortex, hippocampus and corpus callosum areas of brain were scanned under a ×40 objective lens. The full-focused images were generated from the Z-stack images using ZEISS application (Carl Zeiss AG, Germany). A FITC-perfused vessel (<12 µm) that is separated from adjacent vessels was regarded as a single capillary and counted in number. Capillary density was expressed as the number of capillaries per square millimeter.
4.10. Immunohistochemical Staining
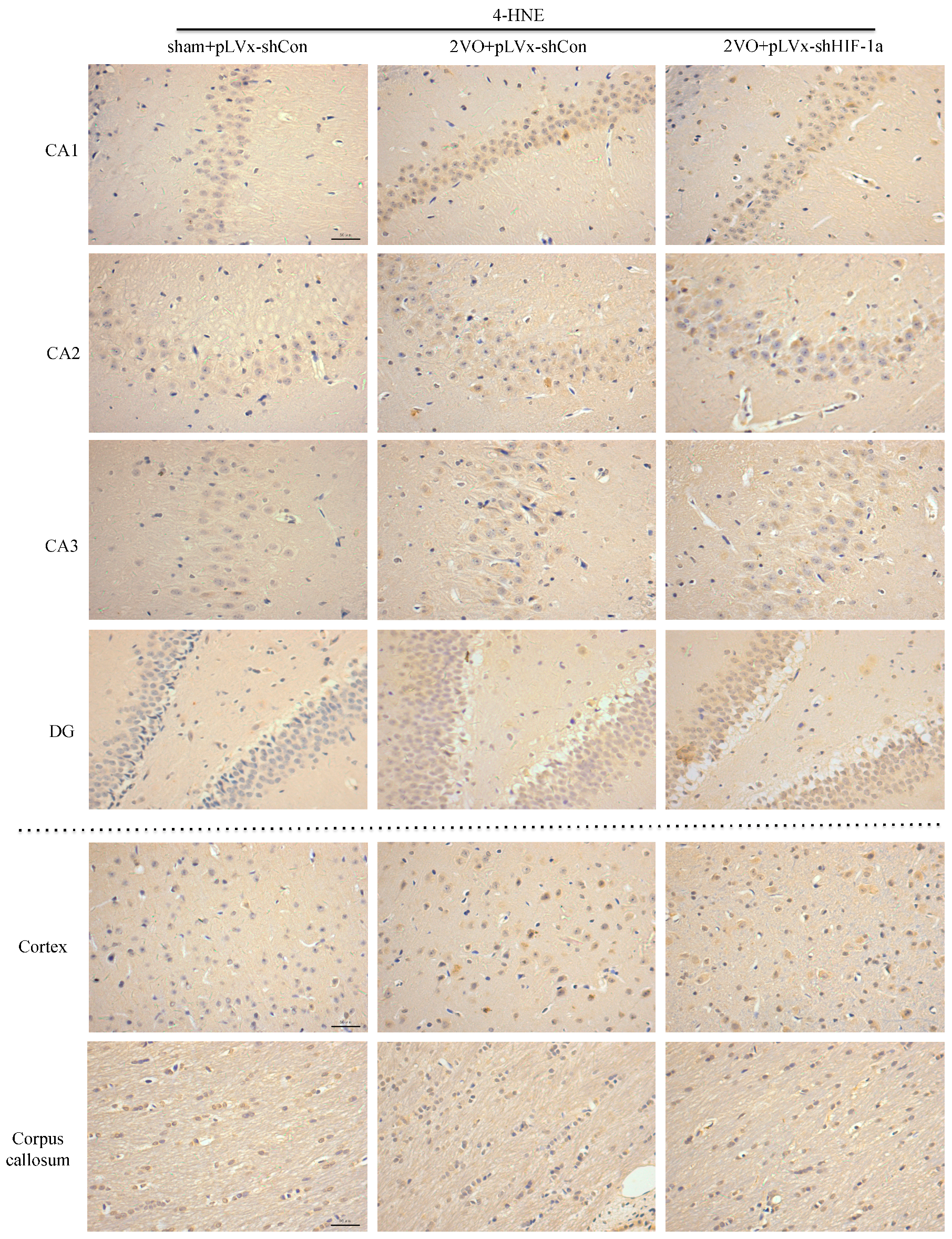
Immunohistochemical staining was used to detect oxidative damage and inflammatory-related activation of glial cells in brain.
A 10% chloral hydrate (350 mg/kg, i.p.) anaesthetic was administered to the rats. Once they were perfused transcardially using 4% buffered PFA (pH 7.4), the brain was removed. It was stored for at least 24 h in fixative then embedded in paraffin wax and sectioned coronally (4 µm thickness). Once blocked, the brain slices were incubated overnight at 4 °C with the primary antibodies used for immunohistochemistry. In this procedure, the primary antibodies used were directed against the lipid peroxidation marker 4-hydroxynonenal (
4-HNE; Millipore Chemicon International, Temecula, CA, USA, 1:500 dilution), the astrocyte marker glial fibrillary acidic protein (
GFAP; GeneTex, Irvine, CA, USA, 1:250 dilution) and the microglial marker ionized calcium-binding adapter molecule 1 (
Iba-1; Abcam, Cambridge, MA, USA, 1:100 dilution). Secondary antibodies were then used to wash and incubate the brain sections for one hour at room temperature. A solution of 50 µL diaminobenzidine solution was used to develop the reaction products and was also used to stain similar brain sections that were not treated with primary antibodies as a negative control. Image-Pro Plus 6.0 image analysis software (Media Cybernetics, Silver Spring, MD, USA) was used to magnify to 100× the sections immunostained for
GFAP or
Iba-1. The number of immunopositive cells were then counted in three visual fields (cells/mm
2) in the corpus callosum, the cerebral cortex, and certain subregions (CA1, CA2, CA3, and DG) of the hippocampus. For cells positively immunostained for
4-HNE, mean integral optical density (IOD) values were determined. To achieve this, five slices with the greatest differences in staining intensity were identified. They were assessed according to the manual to choose the most appropriate parameters to ensure the largest ratio of hippocampal pyramidal cells to other background cells. These parameters were applied to analyze all stained slices. Differences between groups were determined using GraphPad Prism 5.0 software [
1].
4.11. Statistical Analyses
All results are presented as mean ± SEM. The statistical analyses were performed via GraphPad Prism 5.0 software (GraphPad Software, Inc., La Jolla, CA, USA). Differences in escape latencies were analyzed with a two-way repeated measures ANOVA, and Bonferroni post-tests or unpaired student’s t tests were used for post hoc multiple treatment comparisons. All other measurements were analyzed using one-way ANOVAs followed by Bonferroni post hoc tests. Statistical significance was defined as p < 0.05.