Therapeutic Potential of Induced Neural Stem Cells for Parkinson’s Disease
Abstract
:1. Introduction
2. Results
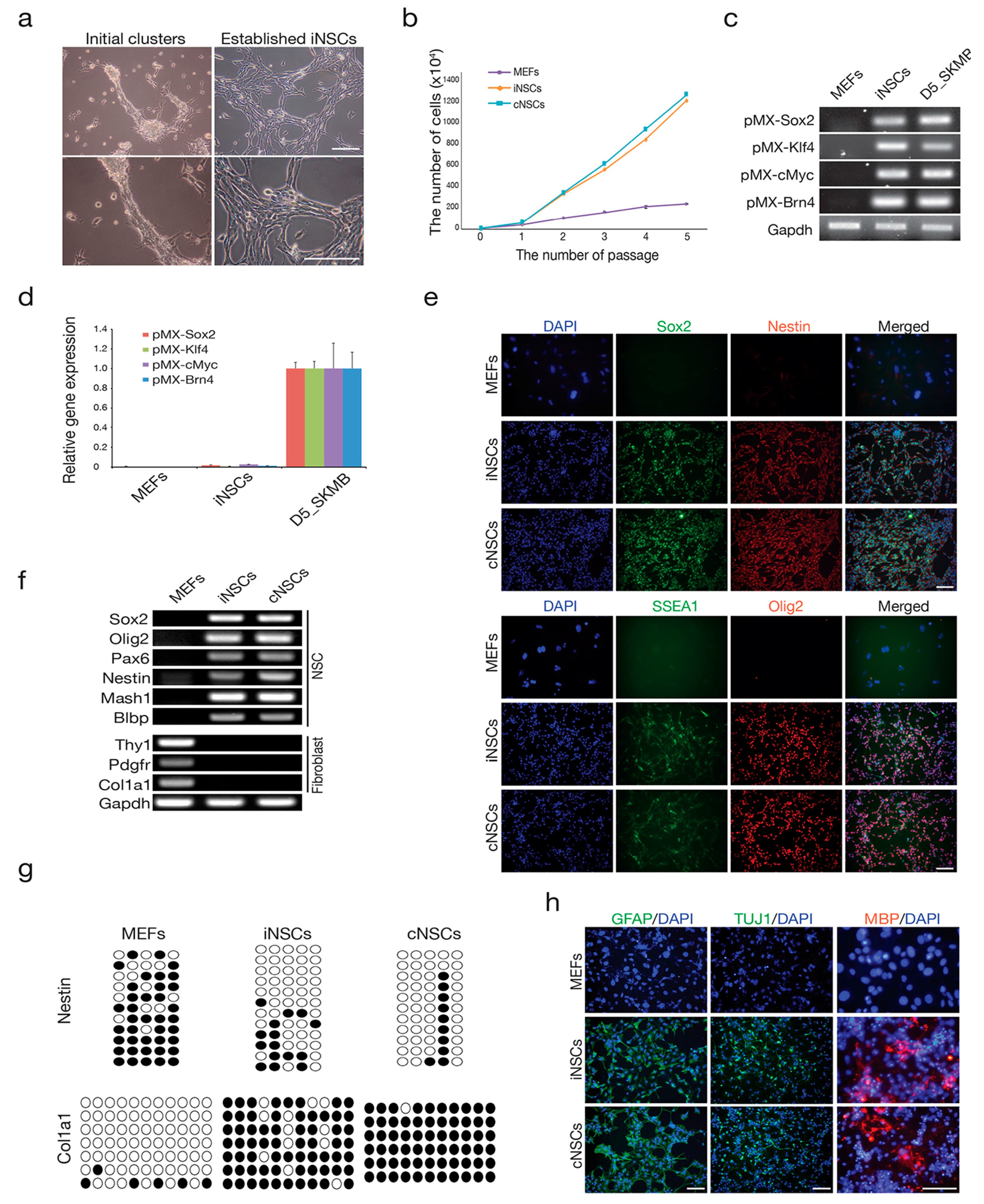
2.1. Direct Conversion of Fibroblasts into iNSCs
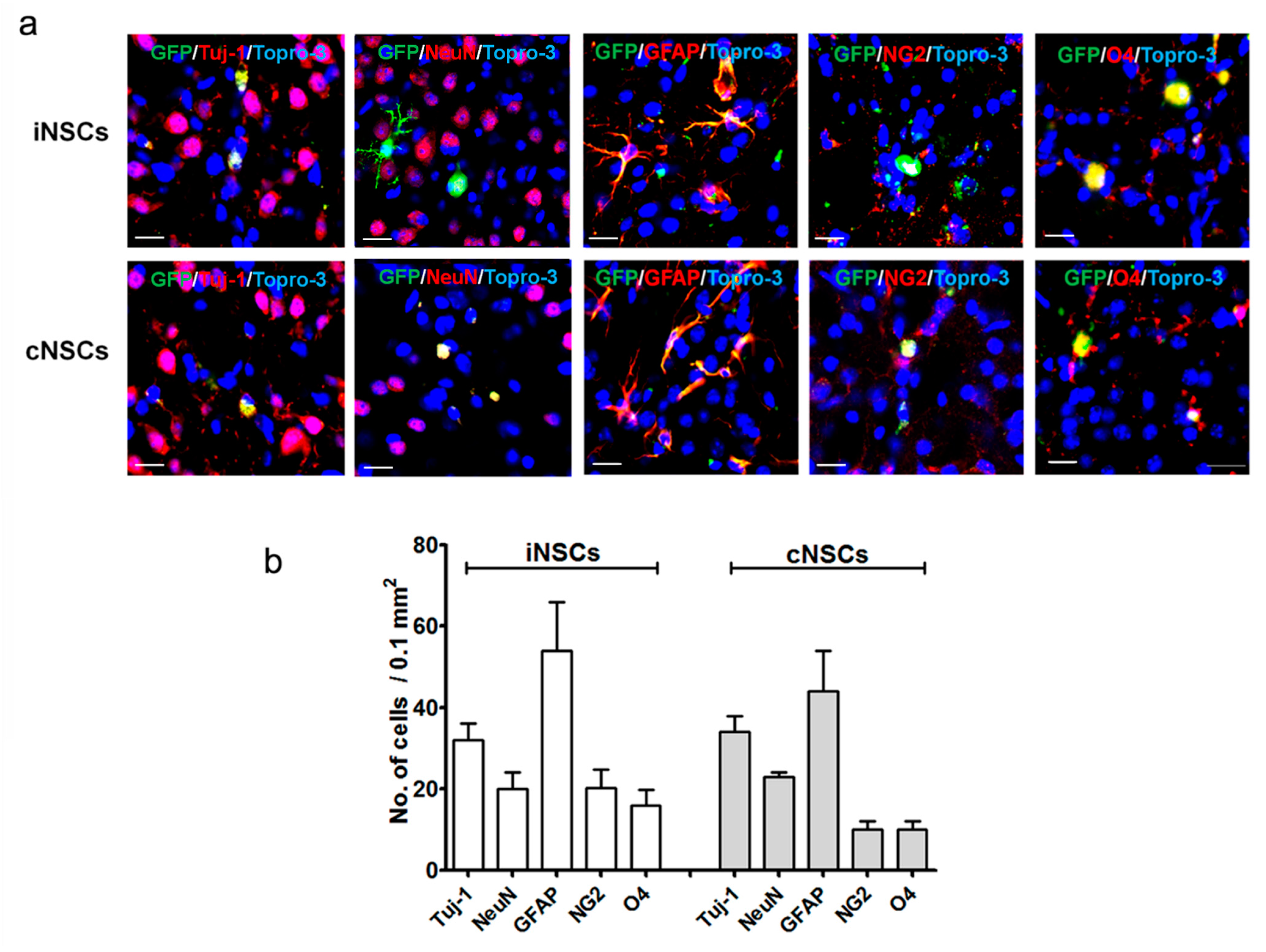
2.2. Engrafted iNSCs Could Differentiate into All Neuronal Lineages
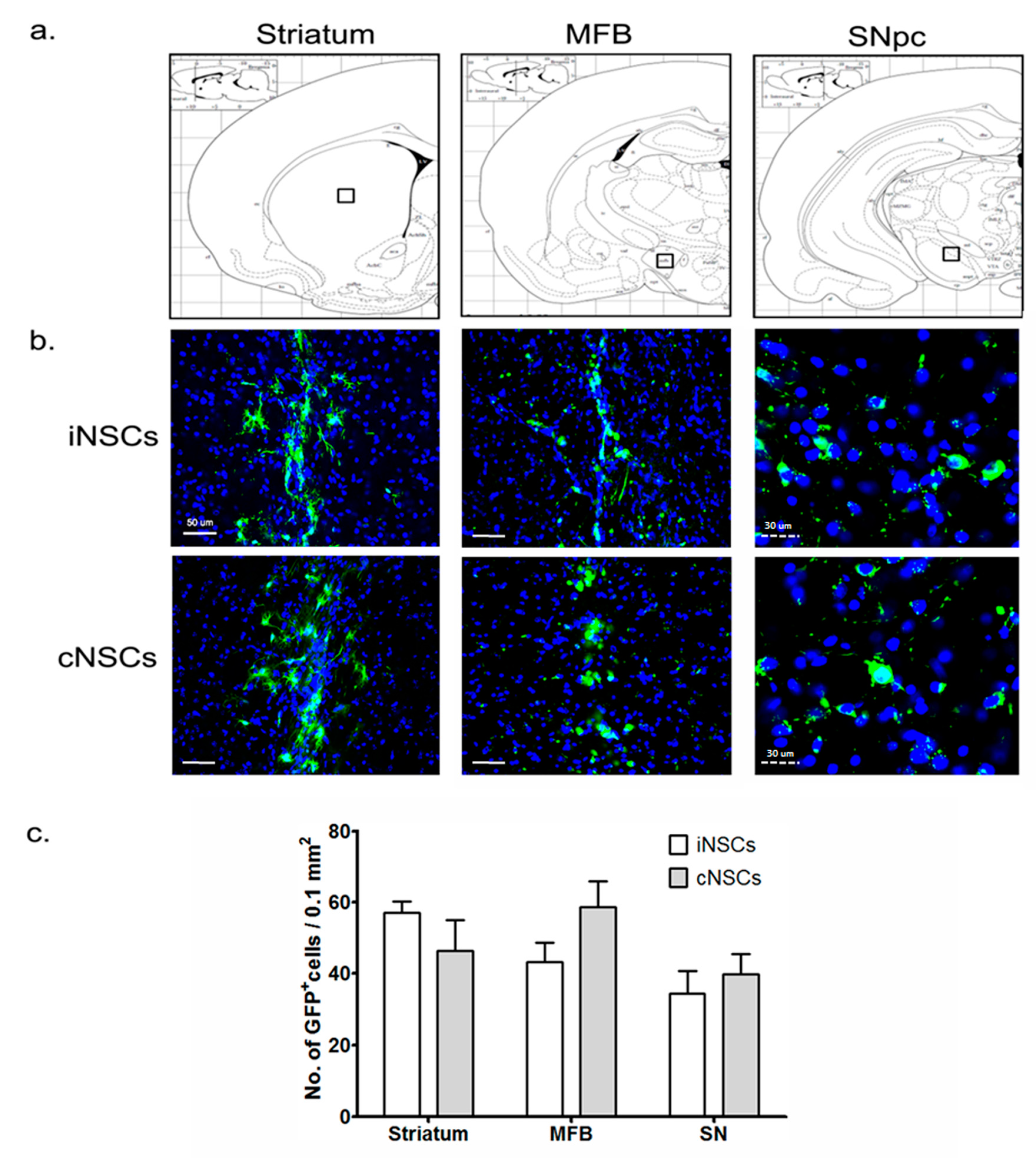
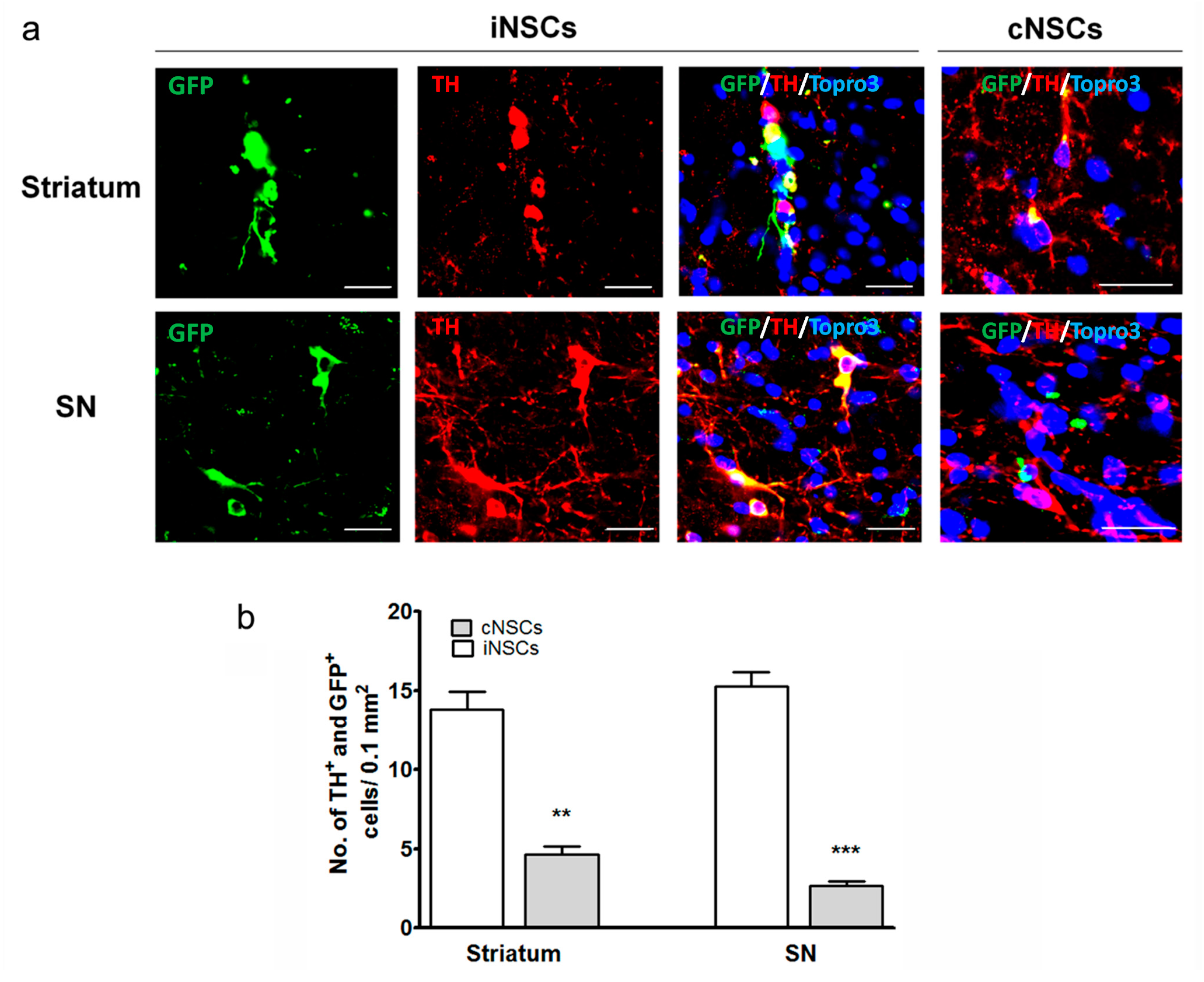
2.3. Engrafted iNSCs Differentiated to DA Neurons and Migrated into the SN
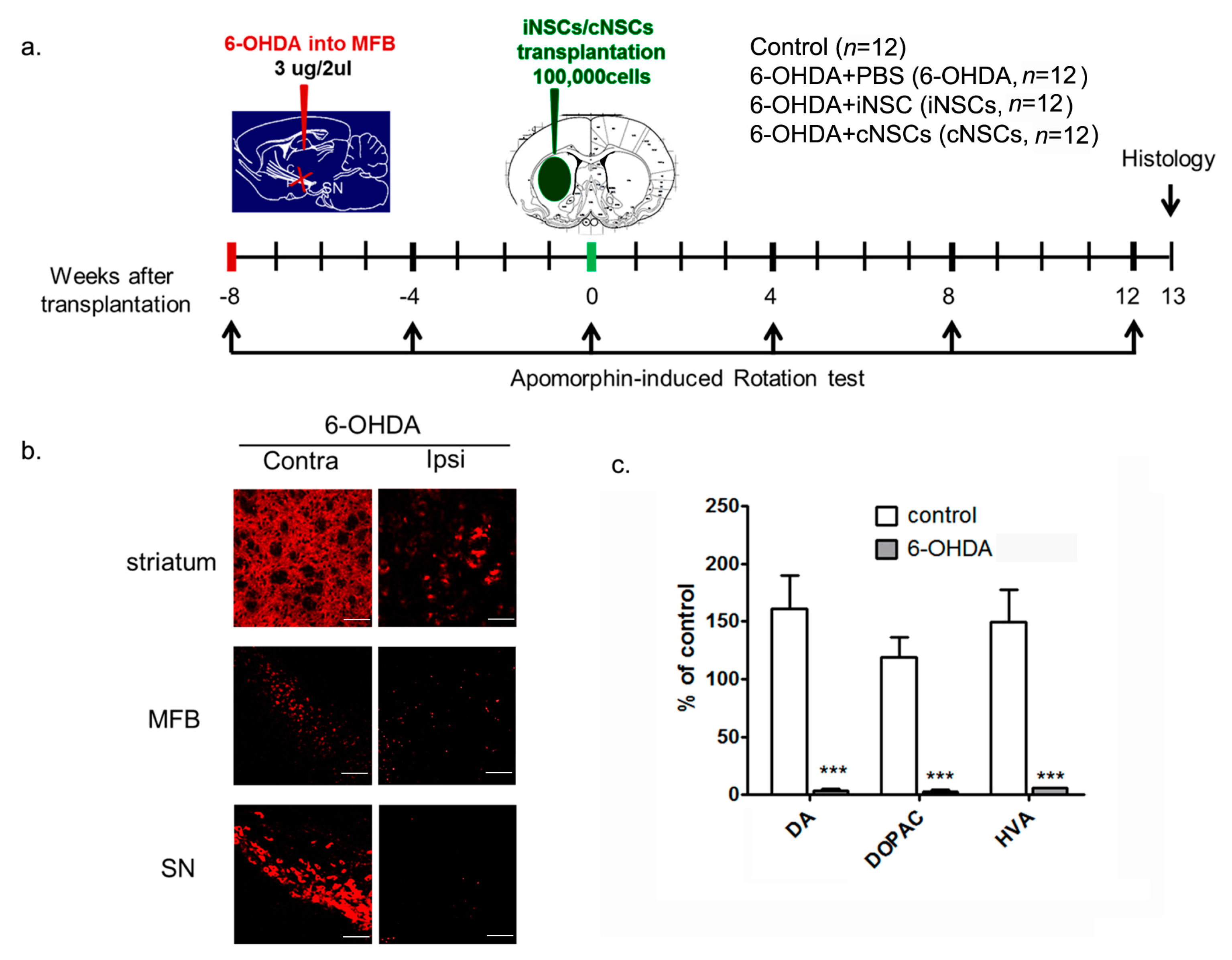
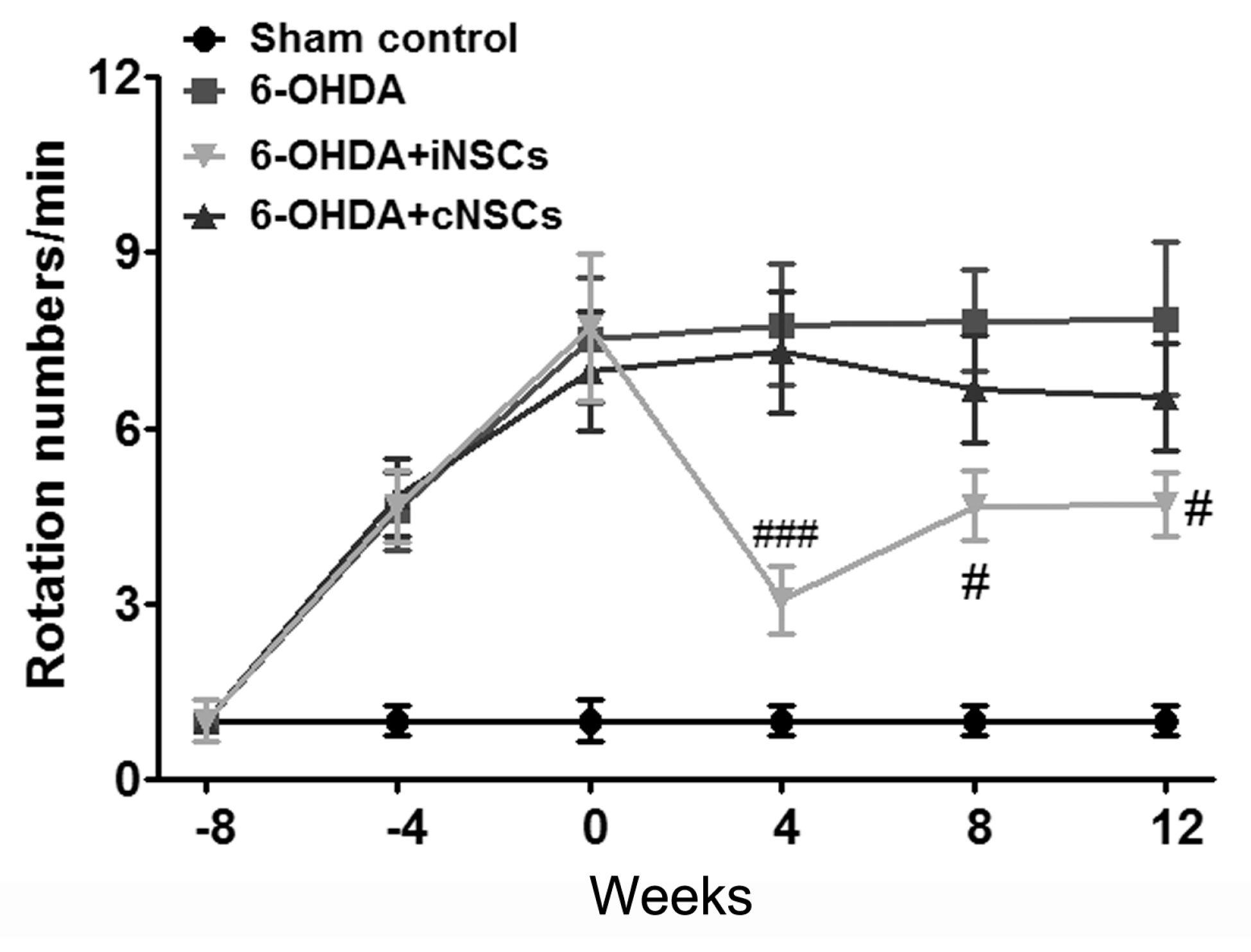
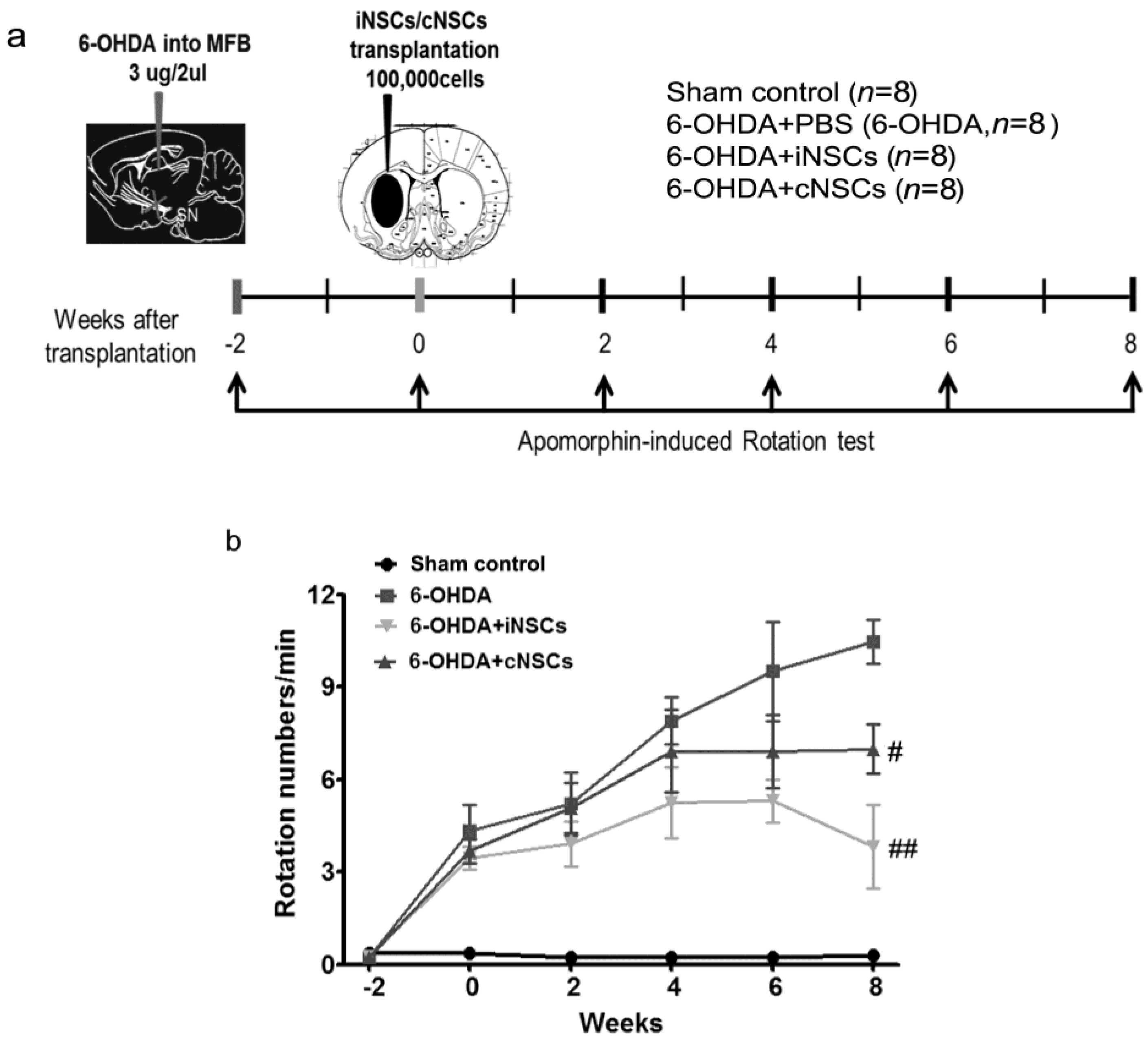
2.4. Engrafted iNSCs Improved Motor Performance in the 6-OHDA-Induced PD Mouse Model
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.2. Generation of iNSCs
4.3. Gene Expression Analysis by RT-PCR and qPCR
4.4. DNA Methylation Analysis
4.5. Immunocytochemistry
4.6. In Vitro Differentiation
4.7. Animals
4.8. Animal Treatment and Unilateral 6-OHDA Lesion in MFB
4.9. Measurement of DA and DA Metabolites (DOPAC and HVA)
4.10. Transplantation
4.11. Histology
4.12. Apomorphine-Induced Rotation
4.13. Statistics
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| PD | parkinson’s disease |
| DA | dopaminergic |
| SN | substantia nigra |
| iNSCs | induced neural stem cells |
| 6-OHDA | 6-hydroxydopamine |
| NSCs | neural stem cells |
| iPSCs | induced pluripotent stem cells |
| iDAs | induced DA neurons |
| mRNA | messenger RNA |
| MEFs | mouse embryonic fibroblasts |
| Tuj-1 | neuron-specific Class III β-tubulin |
| GFAP | glial fibrillary acidic protein |
| MBP | myelin basic protein |
| MFB | Medial forebrain buddle |
| SNpC | SN pars compacta |
| RT-PCR | reverse transcription polymerase chain reaction |
| TH | tyrosine hydroxylase |
| DOPAC | 3,4-dihydroxyphenylacetic acid |
| HVA | homovanillic acid |
| PBS | phosphate-buffered saline |
| GFP | green fluorescent protein |
| NeuN | neuronal specific nuclear protein |
| NG2 | neural/glial antigen 2 |
| O4 | oligodendrocyte marker O4 |
References
- Han, F.; Baremberg, D.; Gao, J.; Duan, J.; Lu, X.; Zhang, N.; Chen, Q. Development of stem cell-based therapy for parkinson’s disease. Trans. Neurodegener. 2015, 4, 16. [Google Scholar] [CrossRef] [PubMed]
- Fahn, S.; Oakes, D.; Shoulson, I.; Kieburtz, K.; Rudolph, A.; Lang, A.; Olanow, C.W.; Tanner, C.; Marek, K. Levodopa and the progression of parkinson’s disease. N. Engl. J. Med. 2004, 351, 2498–2508. [Google Scholar] [PubMed]
- Olanow, C.W.; Watts, R.L.; Koller, W.C. An algorithm (decision tree) for the management of parkinson’s disease: Treatment guidelines. Neurology 2001, 56, 1–88. [Google Scholar] [CrossRef]
- Limousin, P.; Pollak, P.; Benazzouz, A.; Hoffmann, D.; Le Bas, J.F.; Broussolle, E.; Perret, J.E.; Benabid, A.L. Effect of parkinsonian signs and symptoms of bilateral subthalamic nucleus stimulation. Lancet 1995, 345, 91–95. [Google Scholar] [CrossRef]
- Barker, R.A.; Barrett, J.; Mason, S.L.; Bjorklund, A. Fetal dopaminergic transplantation trials and the future of neural grafting in parkinson’s disease. Lancet Neurol. 2013, 12, 84–91. [Google Scholar] [CrossRef]
- Lindvall, O.; Brundin, P.; Widner, H.; Rehncrona, S.; Gustavii, B.; Frackowiak, R.; Leenders, K.L.; Sawle, G.; Rothwell, J.C.; Marsden, C.D.; et al. Grafts of fetal dopamine neurons survive and improve motor function in parkinson’s disease. Science 1990, 247, 574–577. [Google Scholar] [CrossRef] [PubMed]
- Piccini, P.; Brooks, D.J.; Bjorklund, A.; Gunn, R.N.; Grasby, P.M.; Rimoldi, O.; Brundin, P.; Hagell, P.; Rehncrona, S.; Widner, H.; et al. Dopamine release from nigral transplants visualized in vivo in a parkinson’s patient. Nat. Neurosci. 1999, 2, 1137–1140. [Google Scholar] [CrossRef] [PubMed]
- Freed, C.R.; Breeze, R.E.; Rosenberg, N.L.; Schneck, S.A.; Kriek, E.; Qi, J.X.; Lone, T.; Zhang, Y.B.; Snyder, J.A.; Wells, T.H.; et al. Survival of implanted fetal dopamine cells and neurologic improvement 12 to 46 months after transplantation for parkinson’s disease. N. Engl. J. Med. 1992, 327, 1549–1555. [Google Scholar] [CrossRef] [PubMed]
- Kordower, J.H.; Freeman, T.B.; Snow, B.J.; Vingerhoets, F.J.; Mufson, E.J.; Sanberg, P.R.; Hauser, R.A.; Smith, D.A.; Nauert, G.M.; Perl, D.P.; et al. Neuropathological evidence of graft survival and striatal reinnervation after the transplantation of fetal mesencephalic tissue in a patient with parkinson’s disease. N. Engl. J. Med. 1995, 332, 1118–1124. [Google Scholar] [CrossRef] [PubMed]
- Widner, H.; Tetrud, J.; Rehncrona, S.; Snow, B.; Brundin, P.; Gustavii, B.; Bjorklund, A.; Lindvall, O.; Langston, J.W. Bilateral fetal mesencephalic grafting in two patients with parkinsonism induced by 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP). N. Engl. J. Med. 1992, 327, 1556–1563. [Google Scholar] [CrossRef] [PubMed]
- Bjorklund, L.M.; Sanchez-Pernaute, R.; Chung, S.; Andersson, T.; Chen, I.Y.; McNaught, K.S.; Brownell, A.L.; Jenkins, B.G.; Wahlestedt, C.; Kim, K.S.; et al. Embryonic stem cells develop into functional dopaminergic neurons after transplantation in a parkinson rat model. Proc. Natl. Acad. Sci. USA 2002, 99, 2344–2349. [Google Scholar] [CrossRef] [PubMed]
- Hargus, G.; Cooper, O.; Deleidi, M.; Levy, A.; Lee, K.; Marlow, E.; Yow, A.; Soldner, F.; Hockemeyer, D.; Hallett, P.J.; et al. Differentiated parkinson patient-derived induced pluripotent stem cells grow in the adult rodent brain and reduce motor asymmetry in parkinsonian rats. Proc. Natl. Acad. Sci. USA 2010, 107, 15921–15926. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Su, S.C.; Wang, H.; Cheng, A.W.; Cassady, J.P.; Lodato, M.A.; Lengner, C.J.; Chung, C.Y.; Dawlaty, M.M.; Tsai, L.H.; et al. Functional integration of dopaminergic neurons directly converted from mouse fibroblasts. Cell Stem Cell 2011, 9, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Auerbach, J.M.; Rodriguez-Gomez, J.A.; Velasco, I.; Gavin, D.; Lumelsky, N.; Lee, S.H.; Nguyen, J.; Sanchez-Pernaute, R.; Bankiewicz, K.; et al. Dopamine neurons derived from embryonic stem cells function in an animal model of parkinson’s disease. Nature 2002, 418, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Kriks, S.; Shim, J.W.; Piao, J.; Ganat, Y.M.; Wakeman, D.R.; Xie, Z.; Carrillo-Reid, L.; Auyeung, G.; Antonacci, C.; Buch, A.; et al. Dopamine neurons derived from human ES cells efficiently engraft in animal models of parkinson’s disease. Nature 2011, 480, 547–551. [Google Scholar] [CrossRef] [PubMed]
- Roy, N.S.; Cleren, C.; Singh, S.K.; Yang, L.; Beal, M.F.; Goldman, S.A. Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomerase-immortalized midbrain astrocytes. Nat. Med. 2006, 12, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- Studer, L.; Tabar, V.; McKay, R.D. Transplantation of expanded mesencephalic precursors leads to recovery in parkinsonian rats. Nat. Neurosci. 1998, 1, 290–295. [Google Scholar] [PubMed]
- Yang, D.; Zhang, Z.J.; Oldenburg, M.; Ayala, M.; Zhang, S.C. Human embryonic stem cell-derived dopaminergic neurons reverse functional deficit in parkinsonian rats. Stem Cells 2008, 26, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Khoo, M.L.; Tao, H.; Meedeniya, A.C.; Mackay-Sim, A.; Ma, D.D. Transplantation of neuronal-primed human bone marrow mesenchymal stem cells in hemiparkinsonian rodents. PLoS ONE 2011, 6, e19025. [Google Scholar] [CrossRef] [PubMed]
- Blandini, F.; Cova, L.; Armentero, M.T.; Zennaro, E.; Levandis, G.; Bossolasco, P.; Calzarossa, C.; Mellone, M.; Giuseppe, B.; Deliliers, G.L.; et al. Transplantation of undifferentiated human mesenchymal stem cells protects against 6-hydroxydopamine neurotoxicity in the rat. Cell Transplant. 2010, 19, 203–217. [Google Scholar] [CrossRef] [PubMed]
- Shukla, S.; Chaturvedi, R.K.; Seth, K.; Roy, N.S.; Agrawal, A.K. Enhanced survival and function of neural stem cells-derived dopaminergic neurons under influence of olfactory ensheathing cells in parkinsonian rats. J. Neurochem. 2009, 109, 436–451. [Google Scholar] [CrossRef] [PubMed]
- Miura, K.; Okada, Y.; Aoi, T.; Okada, A.; Takahashi, K.; Okita, K.; Nakagawa, M.; Koyanagi, M.; Tanabe, K.; Ohnuki, M.; et al. Variation in the safety of induced pluripotent stem cell lines. Nat. Biotechnol. 2009, 27, 743–745. [Google Scholar] [CrossRef] [PubMed]
- Nori, S.; Okada, Y.; Nishimura, S.; Sasaki, T.; Itakura, G.; Kobayashi, Y.; Renault-Mihara, F.; Shimizu, A.; Koya, I.; Yoshida, R.; et al. Long-term safety issues of iPSC-based cell therapy in a spinal cord injury model: Oncogenic transformation with epithelial-mesenchymal transition. Stem Cell Rep. 2015, 4, 360–373. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, O.; Miura, K.; Okada, Y.; Fujiyoshi, K.; Mukaino, M.; Nagoshi, N.; Kitamura, K.; Kumagai, G.; Nishino, M.; Tomisato, S.; et al. Therapeutic potential of appropriately evaluated safe-induced pluripotent stem cells for spinal cord injury. Proc. Natl. Acad. Sci. USA 2010, 107, 12704–12709. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Flasskamp, H.; Hermann, A.; Arauzo-Bravo, M.J.; Lee, S.C.; Lee, S.H.; Seo, E.H.; Lee, S.H.; Storch, A.; Lee, H.T.; et al. Direct conversion of mouse fibroblasts into induced neural stem cells. Nat. Protoc. 2014, 9, 871–881. [Google Scholar] [CrossRef] [PubMed]
- Han, D.W.; Tapia, N.; Hermann, A.; Hemmer, K.; Hoing, S.; Arauzo-Bravo, M.J.; Zaehres, H.; Wu, G.; Frank, S.; Moritz, S.; et al. Direct reprogramming of fibroblasts into neural stem cells by defined factors. Cell Stem Cell 2012, 10, 465–472. [Google Scholar] [CrossRef] [PubMed]
- Thier, M.; Worsdorfer, P.; Lakes, Y.B.; Gorris, R.; Herms, S.; Opitz, T.; Seiferling, D.; Quandel, T.; Hoffmann, P.; Nothen, M.M.; et al. Direct conversion of fibroblasts into stably expandable neural stem cells. Cell Stem Cell 2012, 10, 473–479. [Google Scholar] [CrossRef] [PubMed]
- Hong, J.Y.; Lee, S.H.; Lee, S.C.; Kim, J.W.; Kim, K.P.; Kim, S.M.; Tapia, N.; Lim, K.T.; Kim, J.; Ahn, H.S.; et al. Therapeutic potential of induced neural stem cells for spinal cord injury. J. Biol. Chem. 2014, 289, 32512–32525. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Kim, J.W.; Kwak, T.H.; Park, S.W.; Kim, K.P.; Park, H.; Lim, K.T.; Kang, K.; Kim, J.; Yang, J.H.; et al. Generation of integration-free induced neural stem cells from mouse fibroblasts. J. Biol. Chem. 2016, 291, 14199–14212. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.M.; Lim, K.T.; Kwak, T.H.; Lee, S.C.; Im, J.H.; Hali, S.; In Hwang, S.; Kim, D.; Hwang, J.; Kim, K.P.; et al. Induced neural stem cells from distinct genetic backgrounds exhibit different reprogramming status. Stem Cell Res. 2016, 16, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.S.; Kim, H.J.; Oh, J.H.; Park, H.G.; Ra, J.C.; Chang, K.A.; Suh, Y.H. Therapeutic potentials of human adipose-derived stem cells on the mouse model of parkinson’s disease. Neurobiol. Aging 2015, 36, 2885–2892. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z. Cell therapy for parkinson’s disease: New hope from reprogramming technologies. Aging Dis. 2015, 6, 499–503. [Google Scholar] [CrossRef] [PubMed]
- Freed, C.R.; Greene, P.E.; Breeze, R.E.; Tsai, W.Y.; DuMouchel, W.; Kao, R.; Dillon, S.; Winfield, H.; Culver, S.; Trojanowski, J.Q.; et al. Transplantation of embryonic dopamine neurons for severe parkinson’s disease. N. Engl. J. Med. 2001, 344, 710–719. [Google Scholar] [CrossRef] [PubMed]
- Defer, G.L.; Geny, C.; Ricolfi, F.; Fenelon, G.; Monfort, J.C.; Remy, P.; Villafane, G.; Jeny, R.; Samson, Y.; Keravel, Y.; et al. Long-term outcome of unilaterally transplanted parkinsonian patients. I. Clinical approach. Brain 1996, 119, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Olanow, C.W.; Goetz, C.G.; Kordower, J.H.; Stoessl, A.J.; Sossi, V.; Brin, M.F.; Shannon, K.M.; Nauert, G.M.; Perl, D.P.; Godbold, J.; et al. A double-blind controlled trial of bilateral fetal nigral transplantation in parkinson’s disease. Ann. Neurol. 2003, 54, 403–414. [Google Scholar] [CrossRef] [PubMed]
- Lindvall, O.; Rehncrona, S.; Brundin, P.; Gustavii, B.; Astedt, B.; Widner, H.; Lindholm, T.; Bjorklund, A.; Leenders, K.L.; Rothwell, J.C.; et al. Human fetal dopamine neurons grafted into the striatum in two patients with severe parkinson’s disease. A detailed account of methodology and a 6-month follow-up. Arch. Neurol. 1989, 46, 615–631. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.H.; Li, C.L.; Lin, H.L.; Chen, P.C.; Calkins, M.J.; Chang, Y.F.; Cheng, P.H.; Yang, S.H. Stem cell transplantation therapy in parkinson’s disease. SpringerPlus 2015, 4, 597. [Google Scholar] [CrossRef] [PubMed]
- Hemmer, K.; Zhang, M.; van Wullen, T.; Sakalem, M.; Tapia, N.; Baumuratov, A.; Kaltschmidt, C.; Kaltschmidt, B.; Scholer, H.R.; Zhang, W.; et al. Induced neural stem cells achieve long-term survival and functional integration in the adult mouse brain. Stem Cell Rep. 2014, 3, 423–431. [Google Scholar] [CrossRef] [PubMed]
- Sheng, C.; Zheng, Q.; Wu, J.; Xu, Z.; Wang, L.; Li, W.; Zhang, H.; Zhao, X.Y.; Liu, L.; Wang, Z.; et al. Direct reprogramming of sertoli cells into multipotent neural stem cells by defined factors. Cell Res. 2012, 22, 208–218. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Sheng, C.; Liu, Z.; Jia, W.; Wang, B.; Li, M.; Fu, L.; Ren, Z.; An, J.; Sang, L.; et al. Lmx1a enhances the effect of iNSCs in a PD model. Stem Cell Res. 2015, 14, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Han, D.W.; Do, J.T.; Arauzo-Bravo, M.J.; Lee, S.H.; Meissner, A.; Lee, H.T.; Jaenisch, R.; Scholer, H.R. Epigenetic hierarchy governing Nestin expression. Stem Cells 2009, 27, 1088–1097. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, L.; Jette, N.; Frolkis, A.; Steeves, T.; Pringsheim, T. The incidence of parkinson’s disease: A systematic review and meta-analysis. Neuroepidemiology 2016, 46, 292–300. [Google Scholar] [CrossRef] [PubMed]
- Das, N.R.; Sharma, S.S. Cognitive impairment associated with parkinson’s disease: Role of mitochondria. Curr. Neuropharmacol. 2016, 14, 584–592. [Google Scholar] [CrossRef] [PubMed]
- Franklin, K.B.J.; Paxinos, G. The Mouse Brain in Stereotaxic Coordinates; Academic Press: San Diego, CA, USA, 1997. [Google Scholar]
- Boix, J.; Padel, T.; Paul, G. A partial lesion model of parkinson’s disease in mice—Characterization of a 6-OHDA-induced medial forebrain bundle lesion. Behav. Brain Res. 2015, 284, 196–206. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.T.; Son, H.J.; Choi, J.H.; Ji, I.J.; Hwang, O. Vertical grid test and modified horizontal grid test are sensitive methods for evaluating motor dysfunctions in the MPTP mouse model of parkinson’s disease. Brain Res. 2010, 1306, 176–183. [Google Scholar] [CrossRef] [PubMed]
| Gene Name | Accession Number | Sequence | Annealing Temperature |
|---|---|---|---|
| Pax6 | NM_001244198 | 5′-CAAGTTCCCGGGAGTGAACC-3′ | 60 °C |
| 5′-TCCACATAGTCATTGGCAGA-3′ | |||
| Sox2 | NM_011443 | 5′-ACGGCCATTAACGGCACACT-3′ | 60 °C |
| 5′-TTTTGCACCCCTCCCAATTC-3′ | |||
| Nes gene | NM_016701 | 5′-TCCTGGTCCTCAGGGGAAGA-3′ | 60 °C |
| 5′-TCCACGAGAGATACCACAGG-3′ | |||
| Olig2 | NM_016967 | 5′-ACCACCACGTGTCGGCTATG-3′ | 60 °C |
| 5′-TGGTCCAGCTCCCCTTCTTG-3′ | |||
| Blbp | NM_021272 | 5′-GGATGGCAAGATGGTCGTGA-3′ | 60 °C |
| 5′-TGGGACTCCAGGAAACCAAG-3′ | |||
| Mash1 | NM_008553 | 5′-CAGAGGAACAAGAGCTGCTG-3′ | 60 °C |
| 5′-GATCTGCTGCCATCCTGCTT-3′ | |||
| Col1a1 | NM_007742 | 5′-CCCTGCCTGCTTCGTGTAAA-3′ | 60 °C |
| 5′-TCGTCTGTTTCCAGGGTTGG-3′ | |||
| Thy1 | NM_009382 | 5′-TTCCCTCTCCCTCCTCCAAGC-3′ | 60 °C |
| 5′-TCGAGGGCTCCTGTTTCTCCTT-3′ | |||
| Pdgfrβ | NM_001146268 | 5′-CAGGACCTCTGGCTGAAGCA-3′ | 60 °C |
| 5′-TCTGGGAGGCAGAAGGGAGAT-3′ | |||
| Gapdh | NM_008084 | 5′-CCAATGTGTCCGTCGTGGAT-3′ | 60 °C |
| 5′-TGCCTGCTTCACCACCTTCT-3′ |
© 2017 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, D.-H.; Kim, J.-H.; Kim, S.M.; Kang, K.; Han, D.W.; Lee, J. Therapeutic Potential of Induced Neural Stem Cells for Parkinson’s Disease. Int. J. Mol. Sci. 2017, 18, 224. https://doi.org/10.3390/ijms18010224
Choi D-H, Kim J-H, Kim SM, Kang K, Han DW, Lee J. Therapeutic Potential of Induced Neural Stem Cells for Parkinson’s Disease. International Journal of Molecular Sciences. 2017; 18(1):224. https://doi.org/10.3390/ijms18010224
Chicago/Turabian StyleChoi, Dong-Hee, Ji-Hye Kim, Sung Min Kim, Kyuree Kang, Dong Wook Han, and Jongmin Lee. 2017. "Therapeutic Potential of Induced Neural Stem Cells for Parkinson’s Disease" International Journal of Molecular Sciences 18, no. 1: 224. https://doi.org/10.3390/ijms18010224
APA StyleChoi, D.-H., Kim, J.-H., Kim, S. M., Kang, K., Han, D. W., & Lee, J. (2017). Therapeutic Potential of Induced Neural Stem Cells for Parkinson’s Disease. International Journal of Molecular Sciences, 18(1), 224. https://doi.org/10.3390/ijms18010224






