1. Introduction
The use of bioelectrochemical systems (BES) for the production of organic compounds such as alcohols or short- to medium-chain fatty acids through microbial electrosynthesis (MES) holds potential [
1]. By exploiting the capabilities of microorganisms to metabolize using electrodes as electron donors—A feature that is found to be widespread amongst most diverse groups of microorganisms [
2]—A variety of organic compounds can be synthesized in mild operational conditions [
3,
4,
5]. As such, these systems provide an environmentally friendly means for producing useful chemicals using cheap and available substrates (carbon dioxide and water), while avoiding the requirement of scarce metal catalysts in the cathode, as would be the case in most conventional electrosynthetic processes.
While the products potentially formed in MES are found to be diverse, the main scientific efforts in this field have so far focused on the specific production of acetate by the reduction of carbon dioxide [
6,
7,
8,
9,
10,
11]. There are two general considerations at the basis of this product choice: (1) specificity; and (2) reaction rate. With the standard reduction potential of acetate from CO
2 being the highest amongst all carboxylates (and considerably higher than that of formate), the potential window within which the cathodic production of acetate can take place shows minimal overlap with respect to most alternative CO
2 reduction products. This provides a thermodynamic advantage regarding the specificity towards the production of acetate. In addition, with the number of electrons (eight) still being considerably low compared to higher reduction products like butyrate, reaction rates are not severely kinetically limited, and production of acetate at high specificity (up to 100%), and in some cases at high rates (up to 66 kg·m
−3·day
−1), has been achieved in biocathodes [
7].
Troubling to the specific and efficient production of acetate in BES is the production of methane by reduction of carbon dioxide, with a very comparable standard reduction potential (
Table 1).
As follows from
Table 1, given its standard potential methanogenesis may always be regarded as competitive to acetate production from a thermodynamic perspective, with methane either produced from H
2 and CO
2 instead of acetate (in case higher overpotentials are used), or produced acetate being consumed through the disproportionation reaction used in acetoclastic methanogenesis.
While the production of methane at biocathodes may form a target in itself, with applications in, for instance, biogas upgrading or the conversion of organic waste into natural gas [
13,
14], the distinct uses and higher economical value of acetate make taking control over specificity in biocathodes with respect to production of both compounds worthwhile. Substantial scientific efforts to this end have been made using diverse methodologies [
3,
8,
10,
15,
16,
17]. The findings are summarized in
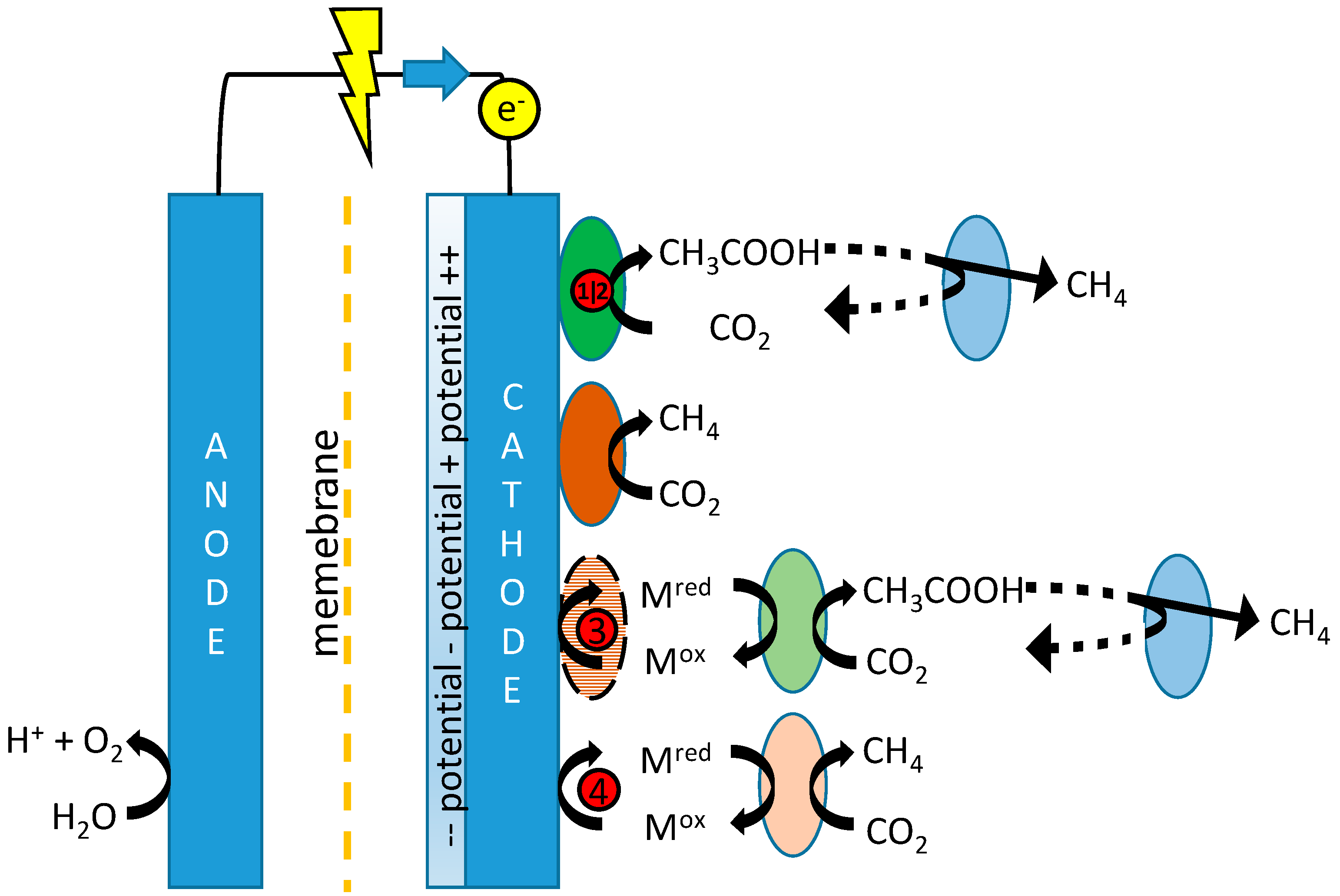
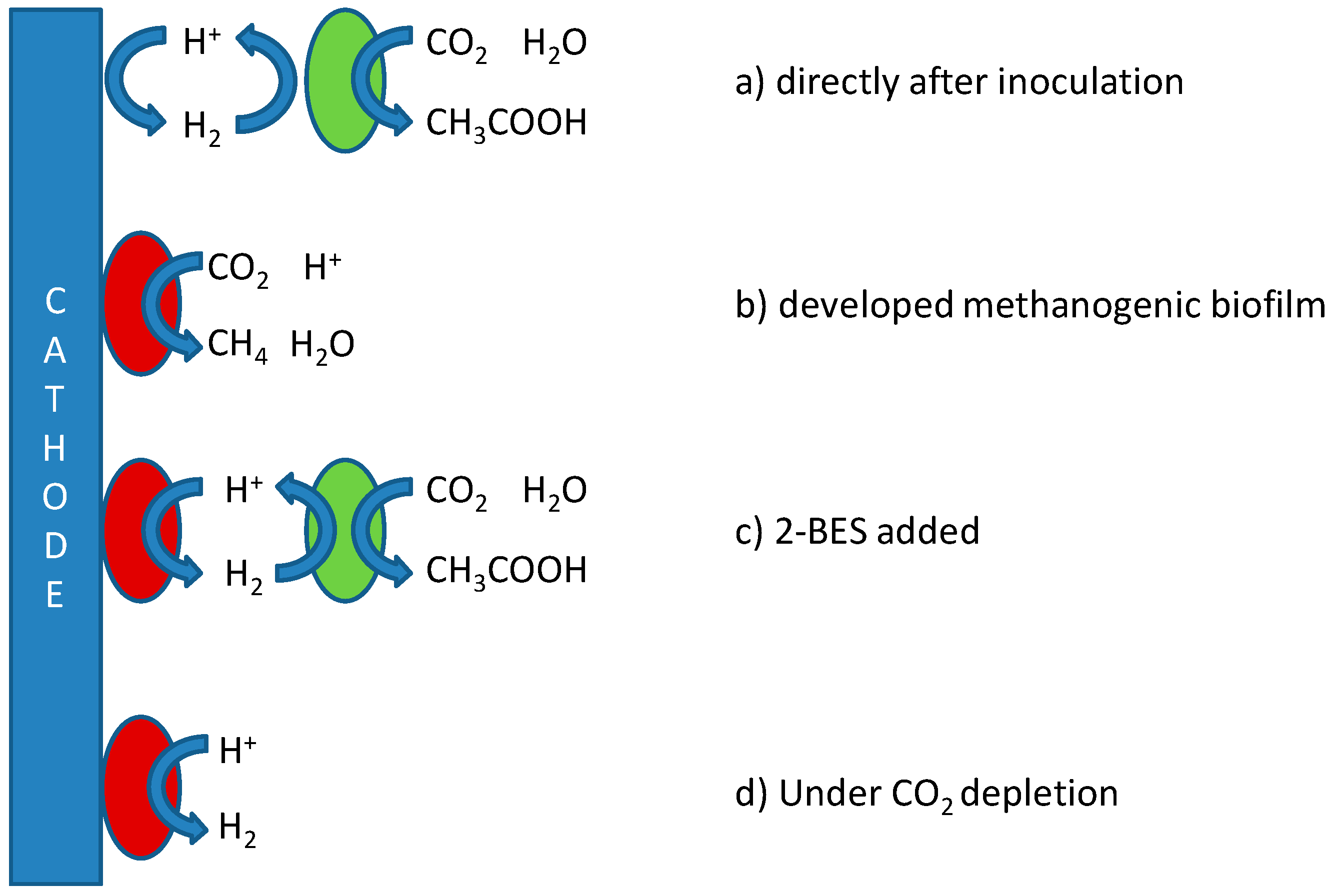
Figure 1, which depicts the possible mechanisms for production of both acetate and methane in MES, including references proposing these mechanisms.
For the production of acetate, two strategies to prevent methanogenesis may be distinguished: (1) pre-enrichment of inoculum or the use of pure cultures while working with aseptic conditions to prevent contamination of the systems with methanogens, thus effectively keeping out competitive microorganisms [
17]; and (2) chemical inhibition of methanogenesis [
7]. Although these techniques have been shown successful to some extent, none of these practices alone provide a means for continuous, larger-scale and long-term septic reactor operation. The use of pure cultures or enriched inocula is prone to contamination and use of chemical inhibitors is unattractive both from economical as well as environmental perspective. Additionally, methanogens may develop immunity towards the inhibiting compounds used, rendering them less effective over time [
19].
The need thus exists for a more integrated approach towards the selective and stable production of acetate, which takes into account (1) structural differences between microbial groups; (2) differences in thermodynamics; and (3) kinetics associated with the proposed pathways in
Figure 1 to affect product specificity. Based on these aspects, reactor operational parameters need to be found at which biomass retention time for acetogens is higher than for methanogens, and/or keeping growth rates (and thus metabolic activity) of acetogens higher than those of methanogens.
Tangent to the considerations of fast kinetics and long acetogenic biomass retention is the question as to whether biocathodes are best operated using potentiostatic or galvanostatic control. A constant potential may be set to narrow down the possibly involved electron transfer mechanisms (
Figure 1) and may lead to the formation of a more energy efficient microbial community. Constant current, on the other hand, allows for better control over production rates and possibly selectivity as it is less sensitive or dependent on (local) changes in e.g., pH, and the electron flux can be controlled to match stoichiometrically with the rate of CO
2 supply.
In this study, the performance of graphite felt electrodes was tested for the cathodic production of acetate using a mixed culture inoculum from AD sludge, using continuously operated reactors. The effect of using both potentiostatic and galvanostatic control on reactor performance was analysed and compared. By using a mixed culture inoculum, the presence of both acetogens and methanogens was assured. Different cathode potentials were tested during potentiostatic experiments, and acetate production was compared to current controlled experiments, in which reducing equivalents were provided at constant rate. Results were interpreted to provide more insight into the mechanisms at play in biocathodes, and the consequential perspectives on future reactor optimization.
3. Discussion
A well-working acetate producing MES (1) has high product selectivity; (2) has high stability/robustness against contamination and/or washout; and (3) does not require costive and/or toxic additives (e.g., 2-BES). In addition, from an application perspective, attention should be paid to: (1) obtaining high product concentrations; (2) high volume specific reaction rates, resulting in smaller reactor footprints; and (3) high energy efficiency, producing the desired compound at the lowest energy expenses.
In the presented research, the effect of different electrochemical control modes on both start-up and steady-state characteristics was investigated. Here, the obtained results will be discussed in relation to abovementioned aspects/criteria.
3.1. Current Density as Control Parameter for MES Selectivity towards Acetate
From the perspective of reaction specificity, methanogen abundance needs to be kept as low as possible. To achieve this, reactor conditions should be so that in the steady state established, achieved specific growth rates for acetogenic organisms are as high as possible when compared to those of methanogens. For the sake of brevity, in the discussion that follows no distinction is made between hydrogenotrophic methanogenesis and homoacetogenesis followed by acetoclastic methanogenesis, as the overall reaction is identical for both.
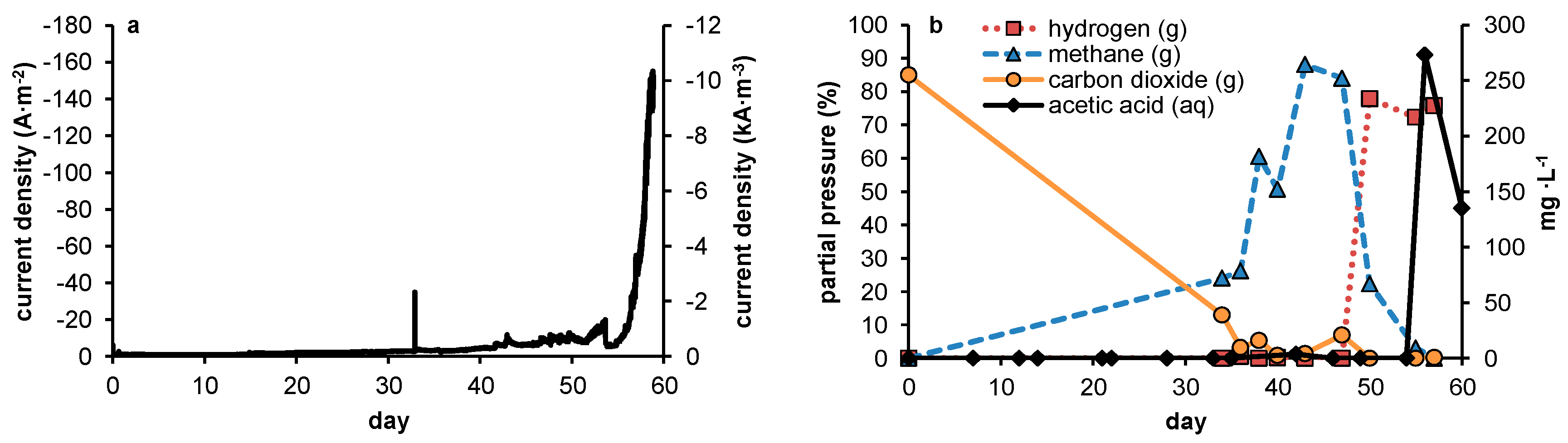
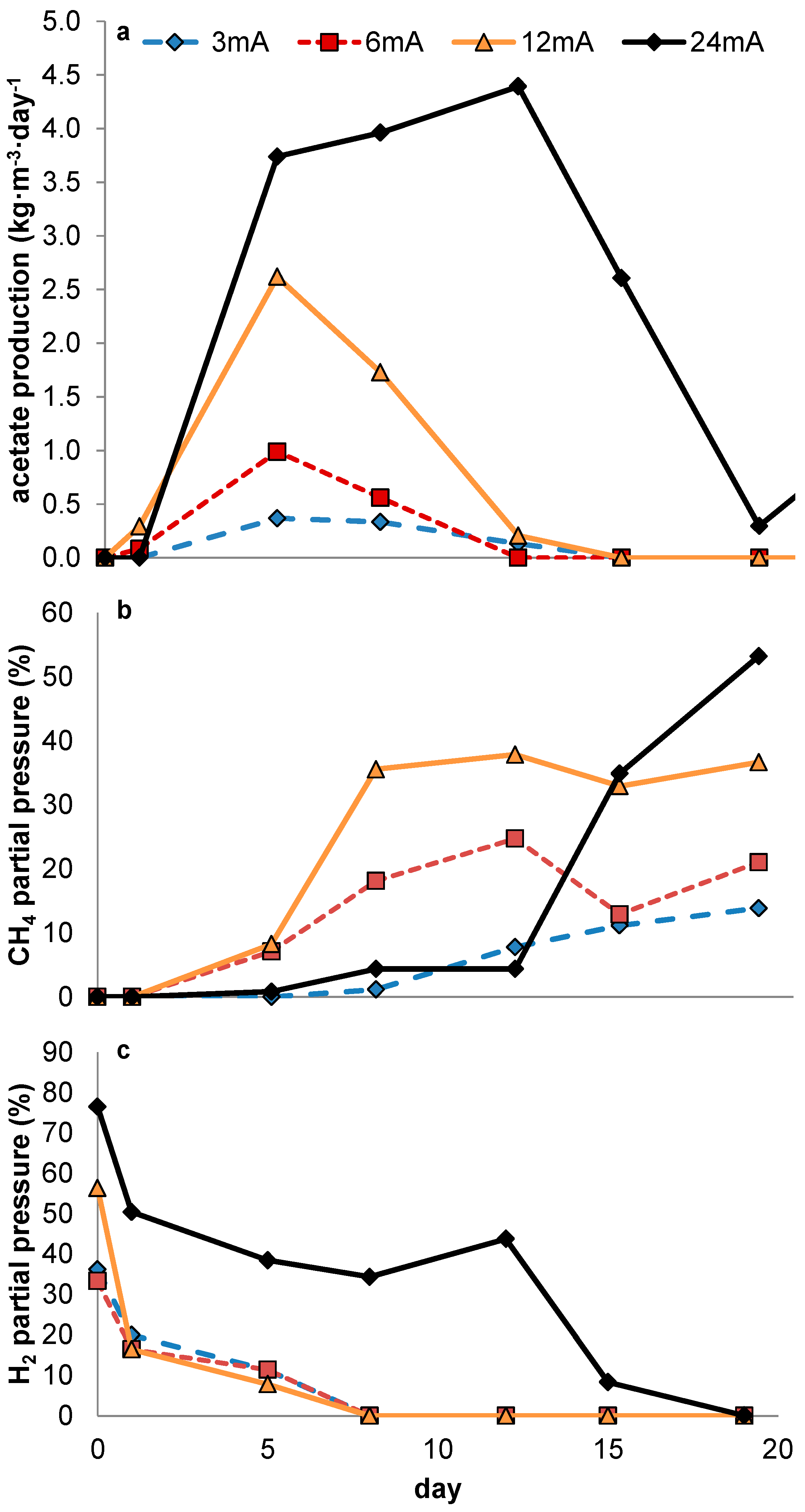
In none of the experiments presented here in which no 2-BES was added was a steady-state achieved in which measurable quantities of acetate were produced. The experimental conditions were thus rendered ineffective in outcompeting methanogens sufficiently during steady state reactor operation. However, during the current controlled experiments, acetate production was positively related to current density. In potential controlled experiments, net acetate production only occurred when current increased rapidly, thus clearly not reflecting a steady state. As was already stated in the introduction (
Table 1), from a thermodynamic perspective, methanogens can gain more energy than acetogens from the consumption of hydrogen and CO
2.
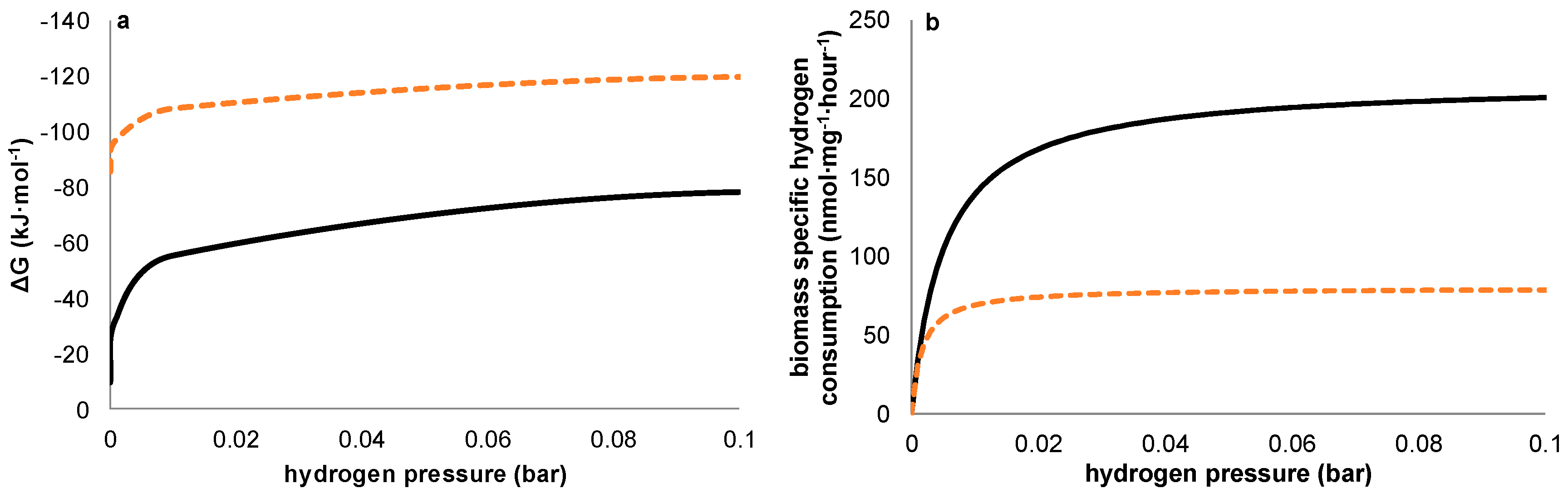
Figure 6a shows this thermodynamic advantage for methanogens is valid at any hydrogen pressure. Our observations however suggest that—under the conditions chosen—a kinetic difference exists between the acetogens and methanogens, both present in the used inoculum, resulting in a higher net growth rate for acetogens during start-up despite the higher thermodynamic available energy for methanogens. Although theoretically, an over-representation of acetogenic organisms in the used inoculum could have resulted in similar results without the requirement of higher growth rates for acetogens, the observations on the potentiostatic experiment with a prolonged period of methanogenic activity prior to acetogenesis strongly indicates this not being the case. Further supporting our hypothesis, differences in growth kinetics have been described previously for methanogens and acetogens using hydrogen as common electron donor [
20,
21]. To allow comparison between thermodynamics and kinetics,
Figure 6a,b illustrates both the thermodynamic equilibria and biomass specific substrate consumption rates which result at different hydrogen pressures. As substrate consumption is directly related to growth via yield, this shows homoacetogens thrive better than methanogens at higher hydrogen partial pressures, despite the higher potential energy gain available to methanogens.
As in our experiments the production of acetate was always accompanied by elevated hydrogen partial pressures, we presume electron transport from electrode to acetate to be primarily mediated through hydrogen. As such, the relative growth rates of both methanogens and acetogens were directly affected by the hydrogen partial pressure, in turn resulting from the fluxes obtained by, on the one hand, production of hydrogen by the set current density and, on the other hand, its consumption by microbial activity. At this point, little is known about the relationship between current density and local hydrogen partial pressures at the electrode, and this is likely to be specific for different electrode materials, depending on how accessible the material is to biofilm formation (e.g., hydrophobicity, roughness, decorated by functional groups etc.). Therefore, rather than predicting an optimal current density based on reported growth parameters, a more empirical approach towards optimization of current density with relation to biocathode selectivity will be required. This is the case both when hydrogen shows an inevitable intermediate in biocathodic production of acetate—as too high current density will result in loss of selectivity by evading hydrogen gas—and in the case alternative mechanisms turn out to play a role in electron transfer from electrode to microbe. The uptake of molecular hydrogen by both acetogens and methanogens is mediated by ferredoxin reducing hydrogenases [
22,
23], a group of enzymes known to possess typically high turnover rates [
24]. Given the mechanistic similarities and characteristics occurring at the start of both pathways [
25,
26], the kinetic advantage of acetogens compared to methanogens at higher hydrogen pressures is probably due to a conversion further down the electron transfer chain (ETC) of methanogens becoming rate limiting under these conditions. In contrast, at low hydrogen pressures, the first electron step (thus from hydrogen to the ETCs initial electron acceptor) in acetogens is likely to be thermodynamically controlled (instead of kinetically). The observation that the rate-limiting step for methane formation occurs further down the ETC is of importance, as it makes it more likely that this limitation will remain in case of an alternative mechanism of electron transfer between cathode and microbe. This would give the homoacetogens a competitive advantage over methanogens at higher current densities regardless of the exact nature of electron transfer between electrode and microbe. Therefore, we hypothesize current density to be a defining parameter for selectivity in acetate-producing MES. At the same time it should be noted that in the presented data current densities were probably not high enough to establish long term stable production of acetate.
3.2. Considerations on the Effect of Current Density on Overall Reactor Performance
As previously stated, selectivity is not the only criterion defining reactor performance in MES, and it therefore holds relevance to further delineate the effects current density presumably has on product concentration, reaction rates and energy efficiency.
In terms of volume specific reaction rates, the application of high current densities is attractive as it drives acetogens close to their maximum turnover rates. However, to sustain production at high current densities, high biomass densities will be required as well. To achieve this, assuming that a biofilm forms on the electrode surface, it becomes crucial to use an electrode material with high specific surface area (with pores accessible to microorganisms).
In case of biofilm formation, the solid/hydraulic retention time ratio (SRT/HRT) can be tuned e.g., by inducing various levels of sheer stress on the biofilm, for instance by recirculation at different flowrates, and an optimum with respect to process selectivity might be sought. To this end, one has to bear in mind that a trade-off will show between biomass retention time and product selectivity, as also methanogens will be washed out slower and may win competition if conditions are suitable (
Figure 6).
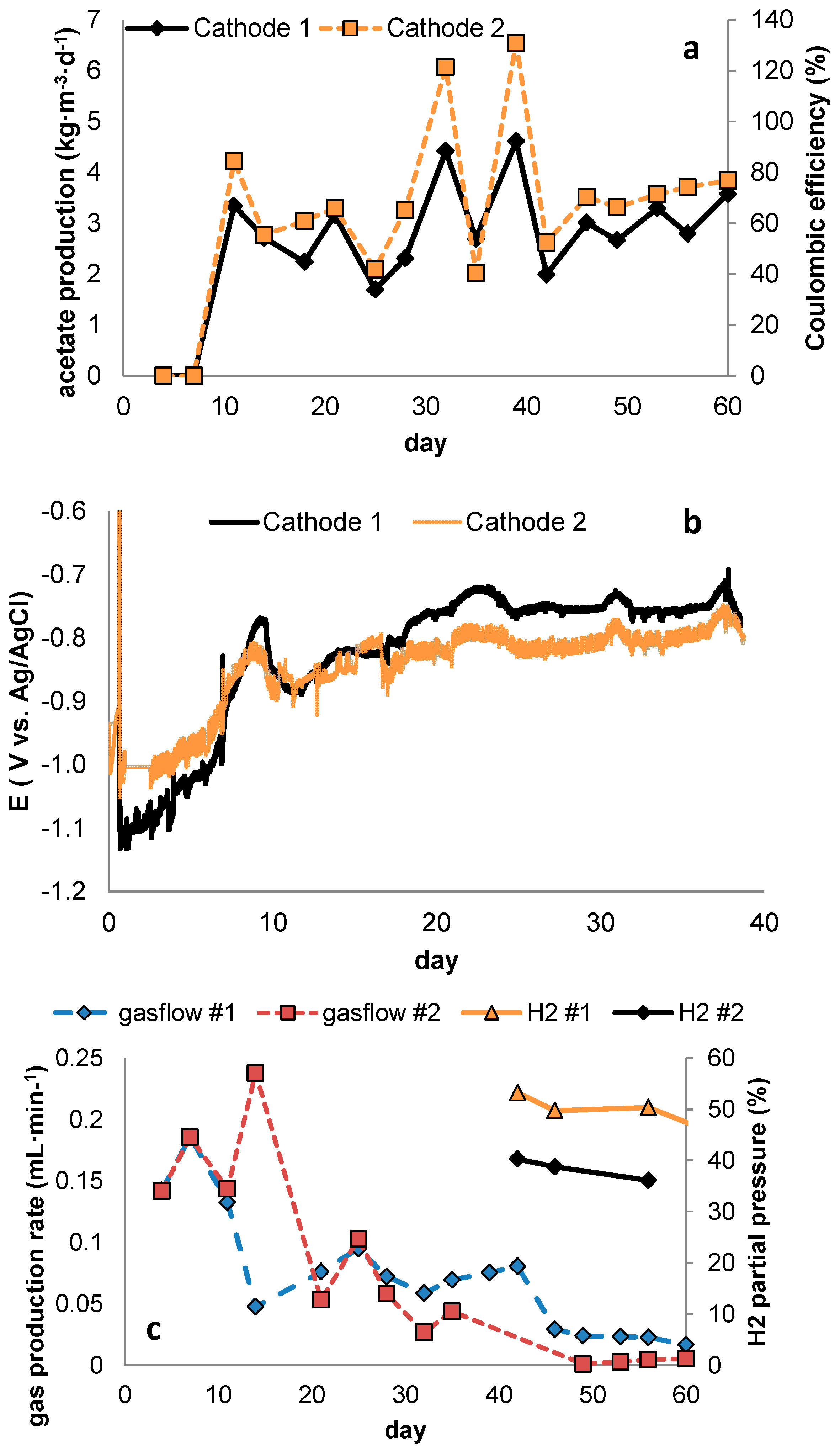
Inherent to higher current densities is also the occurrence of higher overpotential, and although the use of high specific surface area electrode materials may lower this, this will lead to lower voltage efficiency. At the same time, higher current density presumably leads to higher Coulombic efficiency towards acetate (through improved selectivity). Thus, a compromise has to be found between selectivity and voltage efficiency to obtain optimal overall energy efficiency.
To summarize, we hypothesize that, in order for an acetate-producing cathode to perform well both in terms of product selectivity, production rate and obtained product concentrations, a high current density will have to be combined with a high specific surface area electrode, allowing a (relatively) high biomass density at a (relatively) low biomass retention time.
4. Materials and Methods
4.1. System Design and Reactor Assembly
Experiments were conducted in electrochemical cells consisting of two plexiglass flow compartments, two flat plate electrodes and two plexiglass support plates as described previously [
27]. The assembly had an effective projected electrode surface area of 22 cm
2 for both the cathode and anode and an effective cathode volume of 33 cm
3. Cathodes consisted of a stainless steel (SS316) plate serving as current collector, with a plain sheet of graphite paper and five layers of graphite felt (thickness 3 mm, FMI Composites Ltd., Biddeford, Scotland, UK) firmly held between current collector and membrane, thus with the felt serving as the actual electrode material in contact with the electrolyte. Anodes consisted of flat platinum/iridium coated titanium plates (Pt/IrO
2 80:20, Magneto special anodes BV, Schiedam, The Netherlands) with the anode flow compartment filled with polypropylene spacer material sandwiched between the anode and membrane, ensuring proper flow distribution and providing support to the membrane. An Ag/AgCl reference electrode (model QM711X/Gel, Prosense, Oosterhout, The Netherlands) was connected to the cathode using a Haber-Lugging capillary filled with saturated 3 M KCl (+0.205 V vs. NHE). All potentials reported were measured relative to this reference. The anode and the cathode compartments were separated by a cation exchange membrane (Ralex™ C(M)H-PES, Vysočany, Czech Republic). Total catholyte recirculation volume was 180 mL, anolyte recirculation volume 120 mL. Electrolytes were recirculated at a pump speed of 80 mL∙min
−1. In-line pH/temperature monitoring was performed on the catholyte (Endress + Hauser, sensor model CP571D-7BV21 connected to a Liquiline datalogger). Temperature of reactors was maintained constant at 32 ± 1 °C using climate control of the research cabinet, and systems were shielded from light to avoid the possibility of photoautotrophic activities during the experiments. An Ivium N-stat multichannel DC potentiostat (Eindhoven, The Netherlands) was used in a three-electrode configuration (working, counter, and reference) to perform electrochemical measurements and experiments. Recording and analysis of potentiostat data was carried out using the Iviumstat software (Iviumsoft 2.693, ivium, Eindhoven, The Netherlands).
4.2. Electrolyte Composition
Catholyte was made using MilliQ water containing 0.4 g∙L
−1 NH
4HCO
3, 0.05 g∙L
−1 Ca(OH)
2, 0.1 g∙L
−1 MgSO
4∙7H
2O, 0.87 g∙L
−1 K
2HPO
4, 0.68 g∙L
−1 KH
2PO
4, 1 mL∙L
−1 of vitamin solution [
3] and 1 mL∙L
−1 of trace metal solution [
28]. In order to ensure anaerobic conditions and high concentrations of CO
2 in the influent, the medium was continuously purged with CO
2 prior to injection. Using this practice, a total inorganic carbon concentration (TIC) of 11 mM was dissolved in the catholyte, buffering the pH of the catholyte at 5.5.
In later current controlled experiments, the sodium salt of the methanogen inhibitor 2-bromoethanosulfonate (2-BES) was added in order to suppress methanogenic activities at a concentration of 2.1 g∙L−1. Anolyte consisted of a 10 mM phosphate buffer adjusted to pH 7 before use. Anolytes were sparged continuously with CO2 to remove any formed oxygen throughout the experiments while preventing cross-membrane CO2 stripping.
4.3. Inoculum
A mixture of activated sludge from an anaerobic digester and cow manure was used as a source of microorganisms. Homogenized activated sludge and cow manure were centrifuged separately for 5 min at 3700 rpm and supernatant from both solutions were mixed in a 2:1 ratio, resulting in the final inoculum with a COD of 12.5 g∙L−1. Upon inoculation, each cell was injected with 10 mL of inoculum leading to an initial COD of 0.7 g∙L−1 in the catholyte at the start of biotic experiments.
4.4. Hydraulic and Electrochemical Operational Conditions
Systems were filled and continuously fed with fresh electrolyte giving an HRT of 40 h (potentiostatic experiments) or 20 h (current controlled experiments). Prior to inoculation, potentials/currents were applied for at least 2 days until potentials/currents stabilized, thus serving as baseline. In the first run, four cells were poised each at a different potential. Based on the equilibrium potentials for formation of hydrogen and acetate from CO2, the following four potentials were chosen:
The first was −560 mV; the standard equilibrium potential of direct carbon dioxide reduction into acetate is −490 mV. The first setup aimed at facilitating this reaction by imposing only a small overpotential of 70 mV. Doing so, the formation of hydrogen gas is thermodynamically unfeasible and alternative electron transfer is therefore required. This may imply direct electron transfer, providing no other electron mediators are present in the system.
The second potential was −630 mV; selected based on the same rationale as for setup 1, but with a slightly increased overpotential.
The third potential was −700 mV; at this potential, hydrogen formation in theory becomes feasible but due to large overpotentials associated with hydrogen formation a catalyst would be required.
The fourth potential was −900 mV; well below the equilibrium potential for hydrogen formation in these systems, a small cathodic current of 2–4 mA was observed during the abiotic operation of the system, providing sufficient reductants at start.
During the potential controlled experiments, systems were put in batch mode by stopping influent pumps directly after inoculation for 72 h to prevent washout. After this initial batch phase, influents were resumed at the set HRT of 40 h.
During the current controlled experiments all cells were set to an HRT of 20 h, thereby allowing a fixed and equal flux of CO2 to all cathodes. HRT was maintained also directly after inoculation, as preliminary results showed good retention of biomass in the graphite felt even after prolonged periods of inactivity and continuous flushing.
4.5. Analytical Techniques
Liquid and head space samples (1 mL) were taken from catholytes, once a week or more frequently when observed changes in currents/potentials or gas fluxes justified this. Occasionally, the anolyte was sampled for its ionic composition to assess crossover rates of ions. TIC/TOC measurements were done by using a Shimadzu TIC/TOC analyser (Kyoto, Japan); model TOC-VCPH in combination with Shimadzu ASI-V Autosampler (Kyoto, Japan). Gas samples were taken from the headspace in the cathode chamber and analysed for H2, CO2, CH4 and O2 using a gas chromatograph (Varian CP-4900 microGC, TCD detector, Mol Sieve 5A PLOT and PoraPlot U columns in parallel, Santa Clara, California, USA). The catholyte COD was determined using a spectrophotometer HACH XION 500 (HACH, Loveland, CO, USA) with test kit (No. 414, Hach-Lange, Tiel, The Netherlands). For obtaining estimates on biomass concentrations, the COD of both unfiltered and filtered (0.45 µm) electrolytes were compared. HPLC analyses for detection of organic acids (formic acid, acetic acid, propionic acid and butyric acid) were performed with a Dionex UHPLC system (Waltham, MA, USA).
4.6. Performance Calculations
With acetate being the target product, Coulombic efficiency (CE) expresses the fraction of the electron flow (which is dictated by the current) which is used to form acetate:
where
F is Faraday’s constant (96,485 C∙mol
−1), Δ
cAc− is the difference in acetate concentration in the effluent (mol∙L
−1) as compared with the influent, φ is the total volumetric flow of influent (L∙s
−1),
n is the number of electrons consumed per mol acetate produced (8), and
I (A) is the steady state electrical current.