Dose and Radioadaptive Response Analysis of Micronucleus Induction in Mouse Bone Marrow
Abstract
:1. Introduction
2. Results
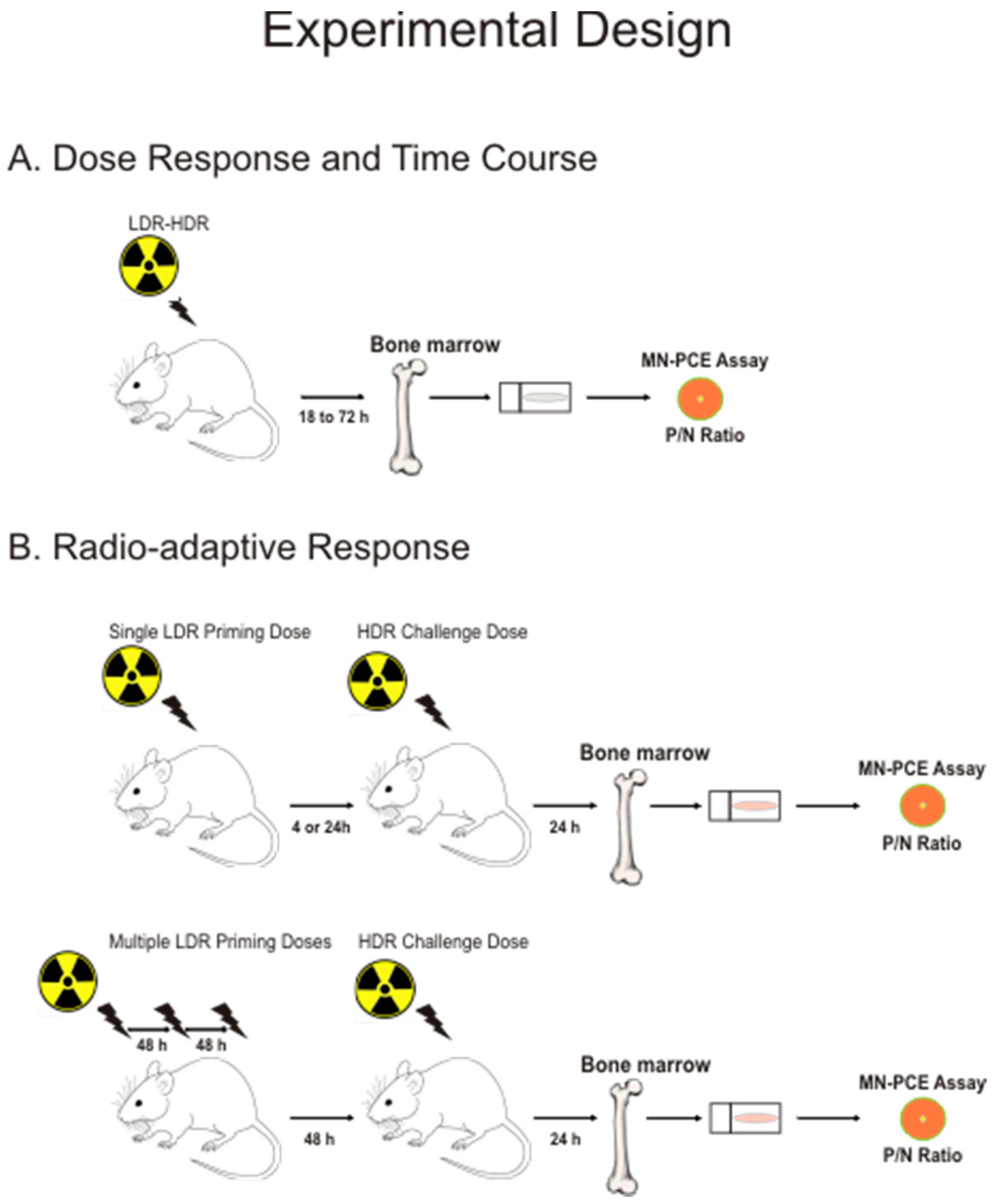
2.1. Experimental Design and Rationale
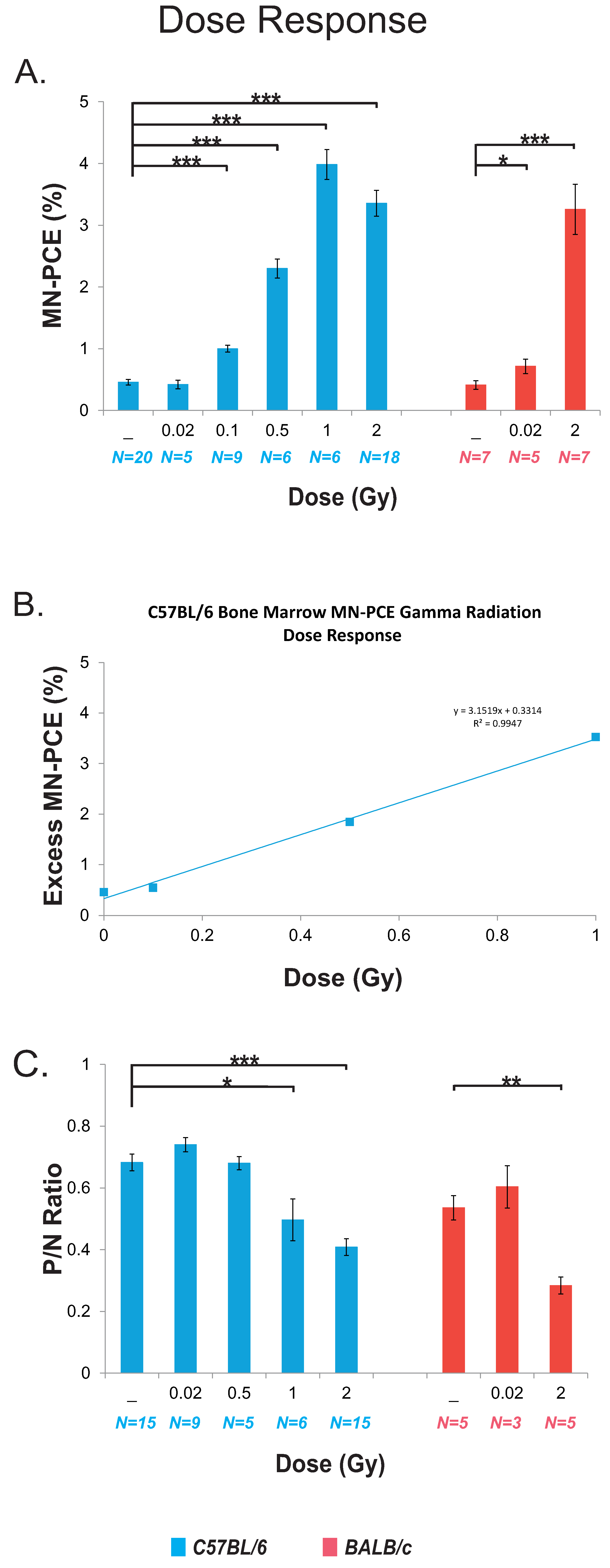
2.2. Dose Response for Bone Marrow Micronucleated Polychromatic Erythrocytes (MN-PCE) Frequency and Polychromatic/Normochromatic Erythrocyte (P/N) Ratio
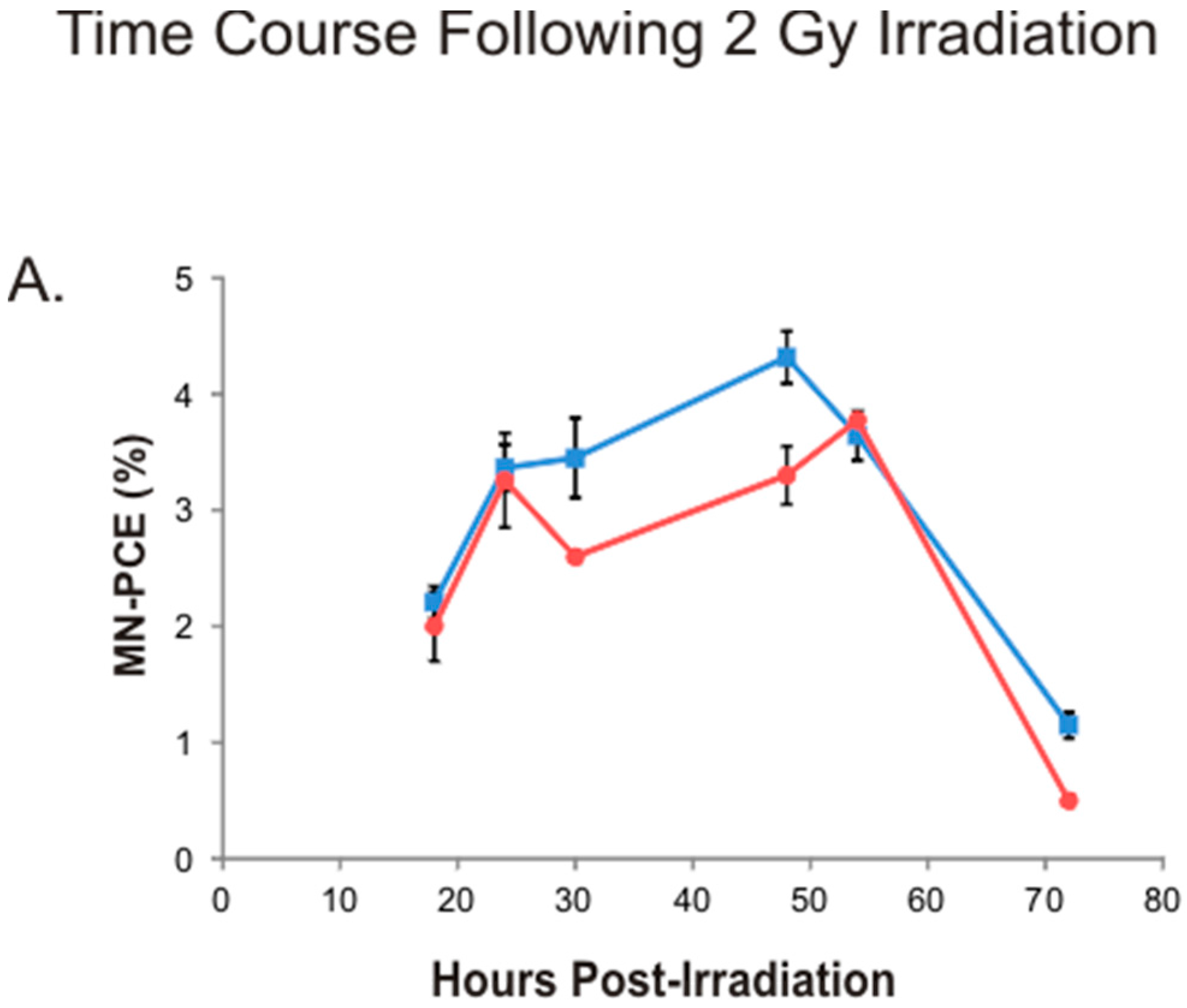
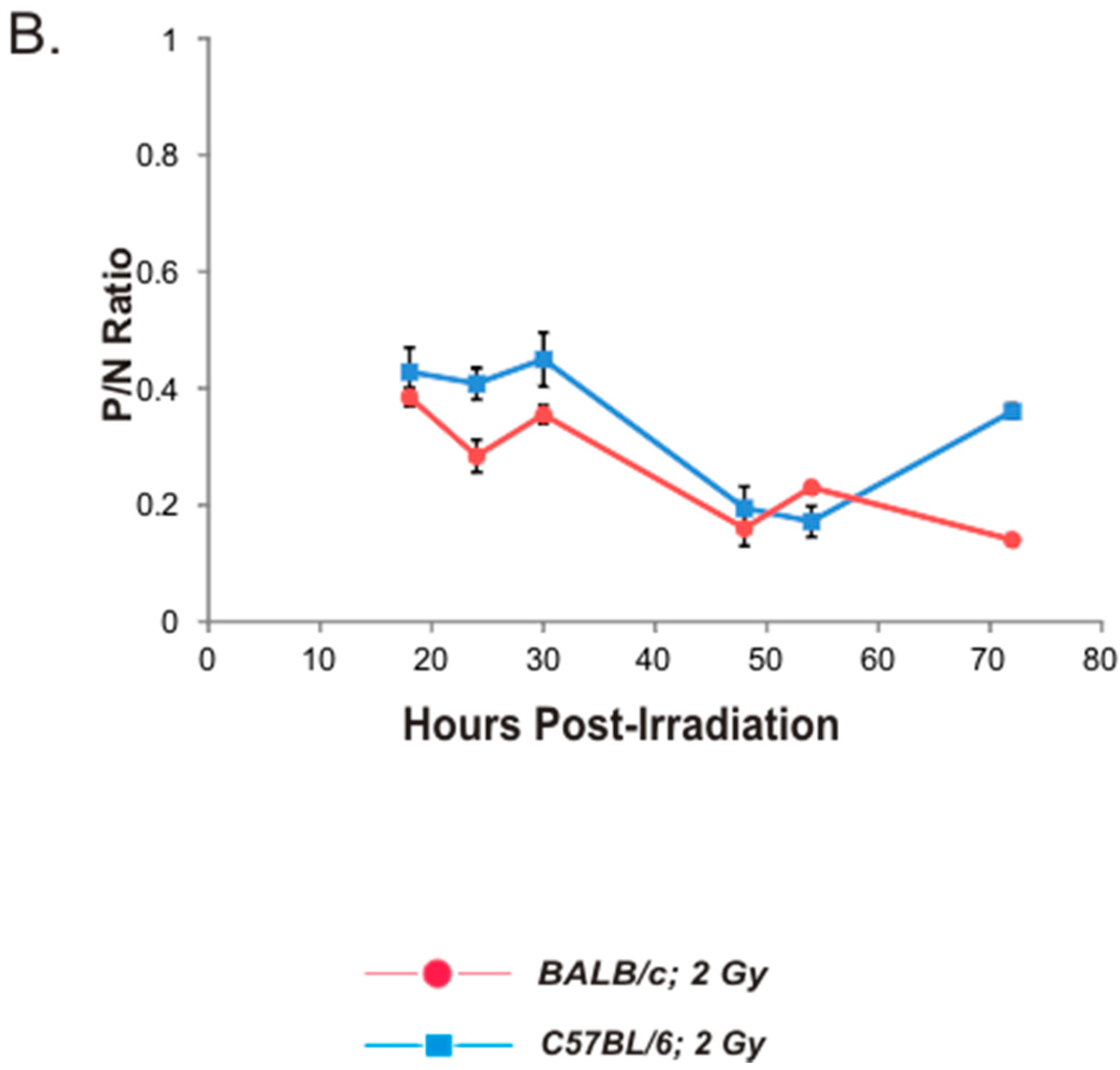
2.3. Kinetics of Bone Marrow MN-PCE Induction and P/N Ratio Suppression Following 2 Gy Irradiation
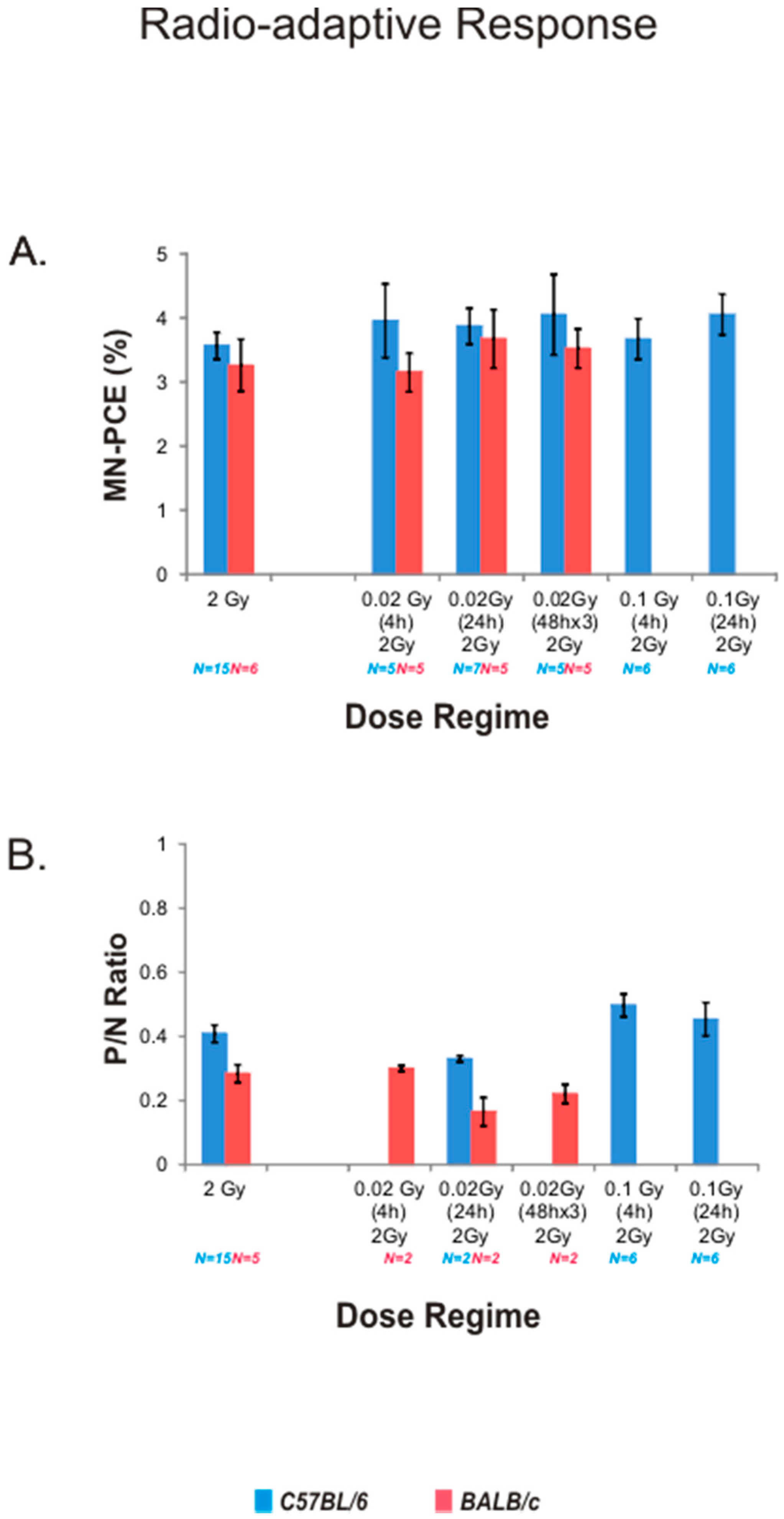
2.4. Radio-Adaptive Response for Bone Marrow MN-PCE Frequency and P/N Ratio
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Irradiations
4.3. Bone Marrow MN-PCE Frequency and P/N Ratio Determination
4.4. Statistical Analysis
Acknowledgments
Author Contributions
Conflicts of Interest
References
- 1990 Recommendations of the International Commission on Radiological Protection. Available online: http://www.ncbi.nlm.nih.gov/pubmed/2053748 (accessed on 15 May 2014).
- ICRP Statement on Tissue Reactions and Early and Late Effects of Radiation in Normal Tissues and Organs: Threshold Doses for Tissue Reactions in a Radiation Protection Context. Available online: http://www.sciencedirect.com/science/article/pii/S0146645312000024 (accessed on 15 May 2014).
- Low-Dose Extrapolation of Radiation-Related Cancer Risk. Available online: http://www.icrp.org/publication.asp?id=ICRP%20Publication%2099 (accessed on 15 May 2014).
- Mothersill, C.; Seymour, C. Implications for human and environmental health of low-doses of ionising radiation. J. Environ. Radioact. 2014, 133, 5–9. [Google Scholar] [CrossRef] [PubMed]
- Kadhim, M.; Salomaa, S.; Wright, E.; Hildebrandt, G.; Belyakov, O.V.; Prise, K.M.; Little, M.P. Non-targeted effects of ionising radiation-Implications for low-dose risk. Mutat. Res. Rev. Mutat. 2012, 752, 84–98. [Google Scholar] [CrossRef] [PubMed]
- Averbeck, D. Non-targeted effects as a paradigm breaking evidence. Mutat. Res. Fundam. Mol. Mech. Mutagenes. 2010, 687, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Tomita, M.; Otsuka, K.; Hatashita, M. A new paradigm in radioadaptive response developing from microbeam research. J. Radiat. Res. 2009, 50, A67–A79. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; O’Connor, M.K. Estimating risk of low radiation doses—A critical review of the BEIR VII report and its use of the linear no-threshold (LNT) hypothesis. Radiat. Res. 2014, 182, 463–474. [Google Scholar] [CrossRef] [PubMed]
- Seong, K.M.; Seo, S.; Lee, D.; Kim, M.J.; Lee, S.S.; Park, S.; Jin, Y.W. Is the Linear No-threshold dose-response paradigm still necessary for the assessment of health effects of low-dose radiation? J. Korean Med. Sci. 2016, 31, S10. [Google Scholar] [CrossRef] [PubMed]
- Dauer, L.T.; Brooks, A.L.; Hoel, D.G.; Morgan, W.F.; Stram, D.; Tran, P. Review and evaluation of updated research on the health effects associated with low-dose ionising radiation. Radiat. Prot. Dosim. 2010, 140, ncq141. [Google Scholar] [CrossRef] [PubMed]
- Sacks, B.; Meyerson, G.; Siegel, J.A. Epidemiology without biology: False paradigms, unfounded assumptions, and specious statistics in radiation science (with commentaries by Inge Schmitz-Feuerhake and Christopher Busby and a reply by the authors). Biol. Theory 2016, 11, 69–101. [Google Scholar] [CrossRef] [PubMed]
- Wolff, S. The adaptive response in radiobiology: Evolving insights and implications. Environ. Health Perspect. 1998, 106, 277. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, H.; Hamada, N.; Takahashi, A.; Kobayashi, Y.; Ohnishi, T. Vanguards of paradigm shift in radiation biology: Radiation-induced adaptive and bystander responses. J. Radiat. Res. 2007, 48, 97–106. [Google Scholar] [CrossRef] [PubMed]
- Tapio, S.; Jacob, V. Radioadaptive response revisited. Radiat. Environ. Biophys. 2007, 46, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Zhong, R.; Sun, L.; Jia, J.; Ma, S.; Liu, X. Ionizing radiation-induced adaptive response in fibroblasts under both monolayer and 3-dimensional conditions. PLoS ONE 2015, 10, e0121289. [Google Scholar] [CrossRef] [PubMed]
- Park, H.S.; You, G.E.; Yang, K.H.; Kim, J.Y.; An, S.; Song, J.Y.; Lee, S.J.; Lim, Y.K.; Nam, S.Y. Role of AKT and ERK pathways in controlling sensitivity to ionizing radiation and adaptive response induced by low-dose radiation in human immune cells. Eur. J. Cell Biol. 2015, 94, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Toprani, S.M.; Das, B. Radio-adaptive response of base excision repair genes and proteins in human peripheral blood mononuclear cells exposed to γ radiation. Mutagenesis 2015, 30. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Liu, S.Z. Induction of cytogenetic adaptive response of somatic and germ cells in vivo and in vitro by low-dose X-irradiation. Int. J. Radiat. Biol. 1990, 58, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Wolff, S.; Afzal, V.; Wiencke, J.K.; Olivieri, G.; Michaeli, A. Human lymphocytes exposed to low-doses of ionizing radiations become refractory to high doses of radiation as well as to chemical mutagens that induce double-strand breaks in DNA. Int. J. Radiat. Biol. 1988, 53, 39–48. [Google Scholar] [CrossRef]
- Shadley, J.D.; Afzal, V.; Wolff, S. Characterization of the adaptive response to ionizing radiation induced by low-doses of X rays to human lymphocytes. Radiat. Res. 1987, 111, 511–517. [Google Scholar] [CrossRef] [PubMed]
- Azzam, E.I.; Raaphorst, G.P.; Mitchel, R.E. Radiation-induced adaptive response for protection against micronucleus formation and neoplastic transformation in C3H 10T1/2 mouse embryo cells. Radiat. Res. 1994, 138, S28–S31. [Google Scholar] [CrossRef] [PubMed]
- Zaichkina, S.I.; Dyukina, A.R.; Rozanova, O.M.; Simonova, N.B.; Romanchenko, S.P.; Sorokina, S.S.; Zakrzhevskaya, D.T.; Yusupov, V.I.; Bagratashvili, V.N. Induction of the adaptive response in mice exposed to He-Ne laser and X-ray radiation. Bull. Exp. Biol. Med. 2016, 161, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Premkumar, K.; Shankar, B.S. Involvement of MAPK signalling in radioadaptive response in BALB/c mice exposed to low-dose ionizing radiation. Int. J. Radiat. Biol. 2016, 92, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Lacoste-Collin, L.; Jozan, S.; Pereda, V.; Courtade-Saidi, M. Influence of a continuous very low-dose of γ-rays on cell proliferation, apoptosis and oxidative stress. Dose Response 2015, 13. [Google Scholar] [CrossRef] [PubMed]
- Grdina, D.J.; Murley, J.S.; Miller, R.C.; Mauceri, H.J.; Sutton, H.G.; Thirman, M.J.; Li, J.J.; Woloschak, G.E.; Weichselbaum, R.R. A manganese superoxide dismutase (SOD2)-mediated adaptive response. Radiat. Res. 2013, 179, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Day, T.K.; Zeng, G.; Hooker, A.M.; Bhat, M.; Turner, D.R.; Sykes, P.J. Extremely low-doses of X-radiation can induce adaptive responses in mouse prostate. Dose Response 2007, 5. [Google Scholar] [CrossRef] [PubMed]
- Farooqi, Z.; Kesavan, P.C. Low-dose radiation-induced adaptive response in bone marrow cells of mice. Mutat. Res. Lett. 1993, 302, 83–89. [Google Scholar] [CrossRef]
- Phan, N.; de Lisio, M.; Parise, G.; Boreham, D.R. Biological effects and adaptive response from single and repeated computed tomography scans in reticulocytes and bone marrow of C57BL/6 mice. Radiat. Res. 2012, 177, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Mitchel, R.E.; Jackson, J.S.; McCann, R.A.; Boreham, D.R. The adaptive response modifies latency for radiation-induced myeloid leukemia in CBA/H mice. Radiat. Res. 1999, 152, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Mitchel, R.E.; Jackson, J.S.; Morrison, D.P.; Carlisle, S.M. Low-doses of radiation increase the latency of spontaneous lymphomas and spinal osteosarcomas in cancer-prone, radiation-sensitive Trp53 heterozygous mice. Radiat. Res. 2003, 159, 320–327. [Google Scholar] [CrossRef]
- Ishii, K.; Hosoi, Y.; Yamada, S.; Ono, T.; Sakamoto, K. Decreased incidence of thymic lymphoma in AKR mice as a result of chronic, fractionated low-dose total-body X irradiation. Radiat. Res. 1996, 146, 582–585. [Google Scholar] [CrossRef] [PubMed]
- Hosoi, Y.; Sakamoto, K. Suppressive effect of low-dose total body irradiation on lung metastasis: Dose dependency and effective period. Radiother. Oncol. 1993, 26, 177–179. [Google Scholar] [CrossRef]
- Ina, Y.; Tanooka, H.; Yamada, T.; Sakai, K. Suppression of thymic lymphoma induction by life-long low-dose-rate irradiation accompanied by immune activation in C57BL/6 mice. Radiat. Res. 2005, 163, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Ina, Y.; Sakai, K. Prolongation of life span associated with immunological modification by chronic low-dose-rate irradiation in MRL-lpr/lpr mice. Radiat. Res. 2004, 161, 168–173. [Google Scholar] [CrossRef] [PubMed]
- Yonezawa, M.; Takeda, A.; Misonoh, J. Acquired radioresistance after low-dose X-irradiation in mice. J. Radiat. Res. 1990, 31, 256–262. [Google Scholar] [CrossRef] [PubMed]
- Mitchel, R.E.; Burchart, P.; Wyatt, H. A lower dose threshold for the in vivo protective adaptive response to radiation. Tumorigenesis in chronically exposed normal and Trp53 heterozygous C57BL/6 mice. Radiat. Res. 2008, 170, 765–775. [Google Scholar] [CrossRef] [PubMed]
- Mitchel, R.E.; Jackson, J.S.; Carlisle, S.M. Upper dose thresholds for radiation-induced adaptive response against cancer in high-dose-exposed, cancer-prone, radiation-sensitive Trp53 heterozygous mice. Radiat. Res. 2004, 162, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Hooker, A.M.; Bhat, M.; Day, T.K.; Lane, J.M.; Swinburne, S.J.; Morley, A.A.; Sykes, P.J. The linear no-threshold model does not hold for low-dose ionizing radiation. Radiat. Res. 2004, 162, 447–452. [Google Scholar] [CrossRef] [PubMed]
- James, S.J.; Enger, S.M.; Makinodan, T. DNA strand breaks and DNA repair response in lymphocytes after chronic in vivo exposure to very low-doses of ionizing radiation in mice. Mutat. Res. Fundam. Mol. Mech. Mutagenes. 1991, 249, 255–263. [Google Scholar] [CrossRef]
- Liu, S.Z.; Cai, L.; Sun, J.B. Effect of low-dose radiation on repair of DNA and chromosome damage. Acta Biol. Hung. 1990, 41, 149–157. [Google Scholar] [PubMed]
- Schwartz, J.L. Variability: The common factor linking low-dose-induced genomic instability, adaptation and bystander effects. Mutat. Res. Fundam. Mol. Mech. Mutagenes. 2007, 616, 196–200. [Google Scholar] [CrossRef] [PubMed]
- Blimkie, M.S.; Fung, L.C.; Petoukhov, E.S.; Girard, C.; Klokov, D. Repair of DNA double-strand breaks is not modulated by low-dose γ radiation in C57BL/6J mice. Radiat. Res. 2014, 181, 548–559. [Google Scholar] [CrossRef] [PubMed]
- Bannister, L.A.; Serran, M.L.; Mantha, R.R. Low-dose γ radiation does not induce an adaptive response for micronucleus induction in mouse splenocytes. Radiat. Res. 2015, 184, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Heddle, J.A. A rapid in vivo test for chromosomal damage. Mutat. Res. Fundam. Mol. Mech. Mutagenes. 1973, 18, 187–190. [Google Scholar] [CrossRef]
- Schmid, W. The micronucleus test. Mutat. Res./Environ. Mutagenes. Relat. Subj. 1975, 31, 9–15. [Google Scholar] [CrossRef]
- Test No. 475: Mammalian Bone Marrow Chromosomal Aberration Test. Available online: http://www.oecd-ilibrary.org/environment/test-no-475-mammalian-bone-marrow-chromosome-aberration test_9789264071308-en (accessed on 30 September 2015).
- Heddle, J.A.; Hite, M.; Kirkhart, B.; Mavournin, K.; MacGregor, J.T.; Newell, G.W.; Salamone, M.F. The induction of micronuclei as a measure of genotoxicity. A report of the US environmental protection agency gene-tox program. Mutat. Res. Rev. Genet. Toxicol. 1983, 123, 61–118. [Google Scholar] [CrossRef]
- Feinendegen, L.E.; Pollycove, M.; Sondhaus, C.A. Responses to low-doses of ionizing radiation in biological systems. Nonlinearity Biol. Toxicol. Med. 2004, 2, 143–171. [Google Scholar] [CrossRef] [PubMed]
- Feinendegen, L.E.; Pollycove, M.; Neumann, R.D. Whole-body responses to low-level radiation exposure: New concepts in mammalian radiobiology. Exp. Hematol. 2007, 35, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Adler, I.D. Cytogenetic Tests in Mammals; IRL Press Oxford: Oxford, UK, 1984; Volume 212, pp. 275–306. [Google Scholar]
- Mavournin, K.H.; Blakey, D.H.; Cimino, M.C.; Salamone, M.F.; Heddle, J.A. The in vivo micronucleus assay in mammalian bone marrow and peripheral blood. A report of the US environmental protection agency gene-tox program. Mutat. Res. Rev. Genet. Toxicol. 1990, 239, 29–80. [Google Scholar] [CrossRef]
- Krishna, G.; Hayashi, M. In vivo rodent micronucleus assay: Protocol, conduct and data interpretation. Mutat. Res. Fundam. Mol. Mech. Mutagenes. 2000, 455, 155–166. [Google Scholar] [CrossRef]
- Jenssen, D.; Ramel, C. Factors affecting the induction of micronuclei at low-doses of X-rays, MMS and dimethylnitrosamine in mouse erythroblasts. Mutat. Res. Genet. Toxicol. 1978, 58, 51–65. [Google Scholar] [CrossRef]
- Cole, R.J.; Taylor, N.; Cole, J.; Arlett, C.F. Short-term tests for transplacentally active carcinogens. I. Micronucleus formation in fetal and maternal mouse erythroblasts. Mutat. Res./Fundam. Mol. Mech. Mutagenes. 1981, 80, 141–157. [Google Scholar] [CrossRef]
- Hart, J.W.; Hartley-Asp, B. Induction of micronuclei in the mouse: Revised timing of the final stage of erythropoiesis. Mutat. Res. Lett. 1983, 120, 127–132. [Google Scholar] [CrossRef]
- Devi, P.U.; Sharma, A.S. Mouse bone-marrow response to low-doses of whole-body γ irradiation: Induction of micronuclei. Int. J. Radiat. Biol. 1990, 57, 97–101. [Google Scholar] [CrossRef]
- Abramsson-Zetterberg, L.; Zetterberg, G.; Grawe, J. The time-course of micronucleated polychromatic erythrocytes in mouse bone marrow and peripheral blood. Mutat. Res./Fundam. Mol. Mech. Mutagenes. 1996, 350, 349–358. [Google Scholar] [CrossRef]
- Cornforth, M.N.; Goodwin, E.H. Transmission of radiation-induced acentric chromosomal fragments to micronuclei in normal human fibroblasts. Radiat. Res. 1991, 126, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Osipov, A.N.; Klokov, D.Y.; Elakov, A.L.; Rozanova, O.M.; Zaichkina, S.I.; Aptikaeva, G.F.; Akhmadieva, A.K. Comparison in vivo study of genotoxic action of high-versus very low dose-rate γ-irradiation. Nonlinearity Biol. Toxicol. Med. 2004, 2. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.S.; Unnikrishnan, M.K.; Devi, P.U. Effect of 5-aminosalicylic acid on radiation-induced micronuclei in mouse bone marrow. Mutat. Res. Fundam. Mol. Mech. Mutagenes. 2003, 527, 7–14. [Google Scholar] [CrossRef]
- Jagetia, G.C.; Ganapathi, N.G. Radiation-induced micronucleus formation in mouse bone marrow after low-dose exposures. Mutat. Res./Fundam. Mol. Mech. Mutagenes. 1994, 304, 235–342. [Google Scholar] [CrossRef]
- Mozdarani, H.; Gharbali, A. Radioprotective effects of cimetidine in mouse bone marrow cells exposed to γ-rays as assayed by the micronucleus test. Int. J. Radiat. Biol. 1993, 64, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Zetterberg, G.; Grawe, J. Flow cytometric analysis of micronucleus induction in mouse erythrocytes by γ-irradiation at very low-dose-rates. Int. J. Radiat. Biol. 1993, 64, 555–564. [Google Scholar] [CrossRef] [PubMed]
- Dertinger, S.D.; Tsai, Y.; Nowak, I.; Hyrien, O.; Sun, H.; Bemis, J.C.; Torous, D.K.; Keng, P.; Palis, J.; Chen, Y. Reticulocyte and micronucleated reticulocyte responses to γ irradiation: Dose-response and time-course profiles measured by flow cytometry. Mutat. Res. Genet. Toxicol. Environ. Mutagenes. 2007, 634, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Salamone, M.F.; Mavournin, K.H. Bone marrow micronucleus assay: A review of the mouse stocks used and their published mean spontaneous micronucleus frequencies. Environ. Mol. Mutagenes. 1994, 23, 239–273. [Google Scholar] [CrossRef]
- Sex difference in the micronucleus test. The collaborative study group for the micronucleus Test. Mutat. Res. 1986, 171, 151–163.
- Grahn, D.; Hamilton, K.F. Genetic variation in the acute lethal response of four inbred mouse strains to whole body X-irradiation. Genetics 1957, 42, 189–198. [Google Scholar] [PubMed]
- Yuhas, J.M.; Storer, J.B. On mouse strain differences in radiation resistance: Hematopoietic death and the endogenous colony-forming unit. Radiat. Res. 1969, 39, 608–622. [Google Scholar] [CrossRef] [PubMed]
- Okayasu, R.; Suetomi, K.; Yu, Y.; Silver, A.; Bedford, J.S.; Cox, R.; Ullrich, R.L. A deficiency in DNA repair and DNA-PKcs expression in the radiosensitive BALB/c mouse. Cancer Res. 2000, 60, 4342–4345. [Google Scholar] [PubMed]
- Yu, Y.; Okayasu, R.; Weil, M.M.; Silver, A.; McCarthy, M.; Zabriskie, R.; Long, S.; Cox, R.; Ullrich, R.L. Elevated breast cancer risk in irradiated BALB/c mice associates with unique functional polymorphism of the Prkdc (DNA-dependent protein kinase catalytic subunit) gene. Cancer Res. 2001, 61, 1820–1824. [Google Scholar] [PubMed]
- Roderick, T.H. The response of twenty-seven inbred strains of mice to daily doses of whole-body X-irradiation. Radiat. Res. 1963, 20, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Storer, J.B.; Mitchell, T.J.; Fry, R.J. Extrapolation of the relative risk of radiogenic neoplasms across mouse strains and to man. Radiat. Res. 1988, 114, 331–353. [Google Scholar] [CrossRef] [PubMed]
- Rithidech, K.N.; Udomtanakunchai, C.; Honikel, L.M.; Whorton, E.B. No evidence for the in vivo induction of genomic instability by low doses of 137Cs γ rays in bone marrow cells of BALB/CJ and C57BL/6J mice. Dose Response 2012, 10. [Google Scholar] [CrossRef] [PubMed]
- Ullrich, R.L.; Davis, C.M. Radiation-induced cytogenetic instability in vivo. Radiat. Res. 1999, 152, 170–173. [Google Scholar] [CrossRef] [PubMed]
- Ponnaiya, B.; Cornforth, M.N.; Ullrich, R.L. Radiation-induced chromosomal instability in BALB/c and C57BL/6 mice: The difference is as clear as black and white. Radiat. Res. 1997, 147, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Hamasaki, K.; Imai, K.; Hayashi, T.; Nakachi, K.; Kusunoki, Y. Radiation sensitivity and genomic instability in the hematopoietic system: Frequencies of micronucleated reticulocytes in whole-body X-irradiated BALB/c and C57BL/6 mice. Cancer Sci. 2007, 98, 1840–1844. [Google Scholar] [CrossRef] [PubMed]
- Fomenko, L.A.; Kozhanovskaia, K.; Gaziev, A.I. Micronucleus formation in the bone marrow cells of chronically irradiated mice with subsequent acute γ irradiation. Radiobiologiia 1991, 31, 709–715. [Google Scholar] [PubMed]
- Zhang, L. Cytogenetic adaptive response induced by pre-exposure in human lymphocytes and marrow cells of mice. Mutat. Res. Genet. Toxicol. Environ. Mutagenes. 1995, 334, 33–37. [Google Scholar] [CrossRef]
- Cao, Y.; Xu, Q.; Jin, Z.D.; Zhou, Z.; Nie, J.H.; Tong, J. Induction of adaptive response: Pre-exposure of mice to 900 MHz radiofrequency fields reduces hematopoietic damage caused by subsequent exposure to ionising radiation. Int. J. Radiat. Biol. 2011, 87, 720–728. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Zong, C.; Zhao, H.; Ji, Y.; Tong, J.; Cao, Y. Induction of adaptive response in mice exposed to 900MHz radiofrequency fields: Application of micronucleus assay. Mutat. Res. Genet. Toxicol. Environ. Mutagenes. 2013, 751, 127–129. [Google Scholar] [CrossRef] [PubMed]
- Mosse, I.; Kostrova, L.; Subbot, S.; Maksimenya, I.; Molophei, V. Melanin decreases clastogenic effects of ionizing radiation in human and mouse somatic cells and modifies the radioadaptive response. Radiat. Environ. Biophys. 2000, 39, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Watson, G.E.; Pocock, D.A.; Papworth, D.; Lorimore, S.A.; Wright, E.G. In vivo chromosomal instability and transmissible aberrations in the progeny of haemopoietic stem cells induced by high- and low-LET radiations. Int. J. Radiat. Biol. 2001, 77, 409–417. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, D.; Coates, P.J.; Lorimore, S.A.; Wright, E.G. The in vivo expression of radiation-induced chromosomal instability has an inflammatory mechanism. Radiat. Res. 2012, 177, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Bieging, K.T.; Mello, S.S.; Attardi, L.D. Unravelling mechanisms of p53-mediated tumour suppression. Nat. Rev. Cancer 2014, 14, 359–370. [Google Scholar] [CrossRef] [PubMed]
- Tinwell, H.; Ashby, J. Comparison of acridine orange and Giemsa stains in several mouse bone marrow micronucleus assays—Including a triple dose study. Mutagenesis 1989, 4, 476–481. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, M.; Sofuni, T.; Ishidate, M. An application of Acridine Orange fluorescent staining to the micronucleus test. Mutat. Res. Lett. 1983, 120, 241–247. [Google Scholar] [CrossRef]

© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bannister, L.A.; Mantha, R.R.; Devantier, Y.; Petoukhov, E.S.; Brideau, C.L.A.; Serran, M.L.; Klokov, D.Y. Dose and Radioadaptive Response Analysis of Micronucleus Induction in Mouse Bone Marrow. Int. J. Mol. Sci. 2016, 17, 1548. https://doi.org/10.3390/ijms17091548
Bannister LA, Mantha RR, Devantier Y, Petoukhov ES, Brideau CLA, Serran ML, Klokov DY. Dose and Radioadaptive Response Analysis of Micronucleus Induction in Mouse Bone Marrow. International Journal of Molecular Sciences. 2016; 17(9):1548. https://doi.org/10.3390/ijms17091548
Chicago/Turabian StyleBannister, Laura A., Rebecca R. Mantha, Yvonne Devantier, Eugenia S. Petoukhov, Chantal L. A. Brideau, Mandy L. Serran, and Dmitry Y. Klokov. 2016. "Dose and Radioadaptive Response Analysis of Micronucleus Induction in Mouse Bone Marrow" International Journal of Molecular Sciences 17, no. 9: 1548. https://doi.org/10.3390/ijms17091548
APA StyleBannister, L. A., Mantha, R. R., Devantier, Y., Petoukhov, E. S., Brideau, C. L. A., Serran, M. L., & Klokov, D. Y. (2016). Dose and Radioadaptive Response Analysis of Micronucleus Induction in Mouse Bone Marrow. International Journal of Molecular Sciences, 17(9), 1548. https://doi.org/10.3390/ijms17091548




