Role of Matrix Metalloproteinases in Photoaging and Photocarcinogenesis
Abstract
:1. Introduction
2. MMPs
3. Photoaging
4. Photocarcinogenesis
4.1. BCC
4.2. SCC
4.3. Malignant Melanoma
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 1O2 | Singlet oxygen |
| AP-1 | Activator protein 1 |
| BCC | Basal cell carcinoma |
| bFGF | Basic fibroblast growth factor |
| COX-2 | Cyclooxygenase 2 |
| CXCR4 | C-X-C chemokine receptor type 4 |
| ECM | Extracellular matrix |
| EGCG | Epigallocatechin gallate |
| EGFR | Epidermal growth factor receptor |
| EMT | Epithelial-mesenchymal transition |
| ERK | Extracellular signal-regulated kinase |
| GM-CSF | Granulocyte macrophage colony stimulatory factor |
| H2O2 | Hydrogen peroxide |
| HB-EGF | Heparin-binding epidermal-like growth factor |
| HIF-1α | Hypoxia-inducible factor-1α |
| IL-1α | Interleukin 1 alpha |
| IL-6 | Interleukin 6 |
| JNK | c-Jun NH2-terminal kinase |
| kDa | Kilodalton |
| LN | Lymph node |
| MAPK | Mitogen-activated protein kinase |
| MMPs | Matrix metalloproteases |
| MT-MMP | Membrane-type matrix metalloprotease |
| NEP | Neutral endopeptidase |
| NF-κB | Nuclear factor-kappa B |
| O2− | Superoxide anion |
| OH groups | Hydroxyl group |
| OH. | Hydroxyl radical |
| PAR-1 | Protease activator receptor 1 |
| PDGF | Platelet-derived growth factor |
| RGP | Radial growth phase |
| ROS | Reactive oxygen species |
| SCC | Squamous cell carcinoma |
| SDF-1 | Stromal cell-derived factor 1 |
| TAMs | Tumor-associated macrophages |
| TCF | T-cell transcription factor |
| TGF-β | Transforming growth factor-beta |
| TIMP | Tissue inhibitors of matrix metalloprotease |
| TNF-α | Tumor necrosis factor alpha |
| UV | Ultraviolet |
| VEGF | Vascular endothelial growth factor |
| VGP | Vertical growth phase |
References
- Steinbrenner, H.; Ramos, M.C.; Stuhlmann, D.; Sies, H.; Brenneisen, P. UVA-mediated downregulation of MMP-2 and MMP-9 in human epidermal keratinocytes. Biochem. Biophys. Res. Commun. 2003, 308, 486–491. [Google Scholar] [CrossRef]
- Vicentini, F.T.M.C.; He, T.; Shao, Y.; Fonseca, M.J.V.; Verri, W.A., Jr.; Fisher, G.J.; Xu, Y. Quercetin inhibits UV irradiation-induced inflammatory cytokine production in primary human keratinocytes by suppressing NF-κB pathway. J. Dermatol. Sci. 2011, 61, 162–168. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; Qin, Z.; Xia, W.; Shao, Y.; Voorhees, J.J.; Fisher, G.J. Matrix-degrading metalloproteinases in photoaging. J. Investig. Dermatol. Symp. Proc. 2009, 14, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, C.W.; Kim, E.K.; Lee, S.J.; Park, N.H.; Kim, H.S.; Kim, H.K.; Char, K.; Jang, Y.P.; Kim, J.W. Inhibition effect of Gynura procumbens extract on UVB-induced matrix-metalloproteinase expression in human dermal fibroblasts. J. Ethnopharmacol. 2011, 137, 427–433. [Google Scholar] [CrossRef] [PubMed]
- O’Grady, A.; Dunne, C.; O’Kelly, P.; Murphy, G.M.; Leader, M.; Kay, E. Differential expression of matrix metalloproteinase (MMP)-2, MMP-9 and tissue inhibitor of metalloproteinase (TIMP)-1 and TIMP-2 in non-melanoma skin cancer: Implications for tumour progression. Histopathology 2007, 51, 793–804. [Google Scholar]
- Ham, S.A.; Yoo, T.; Hwang, J.S.; Kang, E.S.; Paek, K.S.; Park, C.; Kim, J.H.; Do, J.T.; Seo, H.G. Peroxisome proliferator-activated receptor d modulates MMP-2 secretion and elastin expression in human dermal fibroblasts exposed to ultraviolet B radiation. J. Dermatol. Sci. 2014, 76, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Choi, J.H.; Kim, H.G.; Choi, J.M.; Hwang, S.K.; Chung, Y.C.; Jeong, H.G. Cultivated ginseng suppresses ultraviolet B–induced collagenase activation via mitogen-activated protein kinases and nuclear factor κB/activator protein-1–dependent signaling in human dermal fibroblasts. Nutr. Res. 2012, 32, 428–438. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.K.; Lee, K.W.; Kim, H.Y.; Oha, M.H.; Byun, S.; Lim, S.H.; Heo, Y.S.; Kang, N.J.; Bode, A.M.; Dong, Z.; et al. Myricetin suppresses UVB-induced wrinkle formation and MMP-9 expression by inhibiting Raf. Biochem. Pharmacol. 2010, 79, 1455–1461. [Google Scholar] [CrossRef] [PubMed]
- Sbardella, D.; Fasciglione, G.F.; Gioia, M.; Ciaccio, C.; Tundo, G.R.; Marini, S.; Coletta, M. Human matrix metalloproteinases: An ubiquitarian class of enzymes involved in several pathological processes. Mol. Asp. Med. 2012, 33, 119–208. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.Y.; Choi, J.S.; Choi, Y.J.; Shin, S.Y.; Kang, S.W.; Han, S.J.; Kang, Y.H. Epigallocatechin gallate hampers collagen destruction and collagenase activation in ultraviolet-B-irradiated human dermal fibroblasts: Involvement of mitogen-activated protein kinase. Food Chem. Toxicol. 2008, 46, 1298–1307. [Google Scholar] [CrossRef] [PubMed]
- Hwang, B.M.; Noh, E.M.; Kim, J.S.; Kim, J.M.; Hwang, J.K.; Kim, H.K.; Kang, J.S.; Kim, D.S.; Chae, H.J.; You, Y.O.; et al. Decursin inhibits UVB-induced MMP expression in human dermal fibroblasts via regulation of nuclear factor-κB. Int. J. Mol. Med. 2013, 31, 477–483. [Google Scholar] [PubMed]
- Wen, K.C.; Fan, P.C.; Tsai, S.Y.; Shih, I.C.; Chiang, H.M. Ixora parviflora protects against UVB-induced photoaging by inhibiting the expression of MMPs, MAP kinases, and COX-2 and by promoting type I procollagen synthesis. Evid. Based Complement. Altern. Med. 2011, 2012, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.P.; Kim, H.G.; Choi, J.H.; Han, E.H.; Kwon, K.I.; Lee, Y.C.; Choi, J.M.; Chung, Y.C.; Jeong, T.C.; Jeong, H.G. Saponins from the roots of Platycodon grandiflorum suppress ultraviolet A-induced matrix metalloproteinase-1 expression via MAPKs and NF-κB/AP-1-dependent signaling in HaCaT cells. Food Chem. Toxicol. 2011, 49, 3374–3382. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.M.; Chen, H.C.; Lin, T.J.; Shih, I.C.; Wen, K.C. Michelia alba extract attenuates UVB-induced expression of matrix metalloproteinases via MAP kinase pathway in human dermal fibroblasts. Food Chem. Toxicol. 2012, 50, 4260–4269. [Google Scholar] [CrossRef] [PubMed]
- Tewari, A.; Grys, K.; Kollet, J.; Sarkany, R.; Young, A.R. Upregulation of MMP12 and its activity by UVA1 in human skin: potential implications for photoaging. J. Investig. Dermatol. 2014, 134, 2598–2609. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, L.G.; Toro, A.; Zhao, H.; Brown, K.; Tebbutt, S.J.; Granville, D.J. Granzyme B mediates both direct and indirect cleavage of extracellular matrix in skin after chronic low-dose ultraviolet light irradiation. Aging Cell 2015, 14, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.M.; Chen, H.C.; Chiu, H.H.; Chen, C.W.; Wang, S.M.; Wen, K.C. Neonauclea reticulata (Havil.) merr stimulates skin regeneration after UVB exposure via ROS scavenging and modulation of the MAPK/MMPs/collagen pathway. Evid. Based Complement. Altern. Med. 2013, 2013, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Ham, S.A.; Kang, E.S.; Lee, H.; Hwang, J.S.; Yoo, T.; Paek, K.S.; Park, C.; Kim, J.H.; Lim, D.S.; Seo, H.G. PPAR-delta inhibits UVB-induced secretion of MMP-1 through MKP-7-mediated suppression of JNK signaling. J. Investig. Dermatol. 2013, 133, 2593–2600. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Chen, H.; Wang, W.; Wang, R.; Liu, Z.L.; Zhu, W.; Lian, S. N-terminal 5-mer peptide analog P165 of amyloid precursor protein inhibits UVA-induced MMP-1 expression by suppressing the MAPK pathway in human dermal fibroblasts. Eur. J. Pharmacol. 2014, 734, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Pyun, H.B.; Woo, S.W.; Jeong, J.H.; Hwang, J.K. The protective effect of Kaempferia parviflora extract on UVB-induced skin photoaging in hairless mice. Photodermatol. Photoimmunol. Photomed. 2014, 30, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.W.; Hwang, E.; Lee, H.J.; Lee, T.Y.; Song, H.G.; Park, S.Y.; Shin, H.S.; Lee, D.G.; Yi, T.H. Effects of Galla chinensis extracts on UVB-irradiated MMP-1 production in hairless mice. J. Nat. Med. 2015, 69, 22–34. [Google Scholar] [CrossRef] [PubMed]
- Hwang, E.; Lee, D.G.; Park, S.H.; Oh, M.S.; Kim, S.Y. Coriander leaf extract exerts antioxidant activity and protects against UVB-induced photoaging of skin by regulation of procollagen type I and MMP-1 expression. J. Med. Food 2014, 17, 985–995. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Li, R.; Yan, N.; Chen, G.; Qian, W.; Jiang, H.L.; Ji, C.; Bi, Z.G. Astragaloside IV controls collagen reduction in photoaging skin by improving transforming growth factor-β/Smad signaling suppression and inhibiting matrix metalloproteinase-1. Mol. Med. Rep. 2015, 11, 3344–3348. [Google Scholar] [PubMed]
- Lee, Y.R.; Noh, E.M.; Han, J.H.; Kim, J.M.; Hwang, J.K.; Hwang, B.M.; Chung, E.Y.; Kim, B.S.; Lee, S.H.; Lee, S.J.; et al. Brazilin inhibits UVB-induced MMP-1/3 expressions and secretions by suppressing the NF-κB pathway in human dermal fibroblasts. Eur. J. Pharmacol. 2012, 674, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Yao, C.; Lee, D.H.; Oh, J.H.; Kim, M.K.; Kim, K.H.; Park, C.H.; Chung, J.H. Poly(I:C) induces expressions of MMP-1, -2, and -3 through various signaling pathways including IRF3 in human skin fibroblasts. J. Dermatol. Sci. 2015, 80, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Son, W.C.; Yun, J.W.; Kim, B.H. Adipose-derived mesenchymal stem cells reduce MMP-1 expression in UV-irradiated human dermal fibroblasts: Therapeutic potential in skin wrinkling. Biosci. Biotechnol. Biochem. 2015, 79, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Wertz, K.; Seifert, N.; Hunziker, P.B.; Riss, G.; Wyss, A.; Lankin, C.; Goralczyk, R. Beta-carotene inhibits UVA-induced matrix metalloprotease 1 and 10 expression in keratinocytes by a singlet oxygen-dependent mechanism. Free Radic. Biol. Med. 2004, 37, 654–670. [Google Scholar] [CrossRef] [PubMed]
- Onoue, S.; Kobayashi, T.; Takemoto, Y.; Sasaki, I.; Shinkai, H. Induction of matrix metalloproteinase-9 secretion from human keratinocytes in culture by ultraviolet-B irradiation. J. Dermatol. Sci. 2003, 33, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.S.; Song, J.H.; Youn, U.J.; Hyun, J.W.; Jeong, W.S.; Lee, M.Y.; Choi, H.J.; Lee, H.K.; Chae, S. Inhibition of UVB-induced wrinkle formation and MMP-9 expression by mangiferin isolated from Anemarrhena asphodeloides. Eur. J. Pharmacol. 2012, 689, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Fortino, V.; Maioli, E.; Torricelli, C.; Davis, P.; Valacchi, G. Cutaneous MMPs are differently modulated by environmental stressors in old and young mice. Toxicol. Lett. 2007, 173, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Vayalil, P.K.; Mittal, A.; Hara, Y.; Elmets, C.A.; Katiyar, S.K. Green tea polyphenols prevent ultraviolet light-induced oxidative damage and matrix metalloproteinases expression in mouse skin. J. Investig. Dermatol. 2004, 122, 1480–1487. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.; Bhatti, H.; Nerusu, K.C.; Bhagavathula, N.; Kang, S.; Fisher, G.J.; Varani, J.; Voorhees, J.J. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem. Photobiol. 2003, 78, 43–48. [Google Scholar] [CrossRef]
- Chen, Z.; Seo, J.Y.; Kim, Y.K.; Lee, S.R.; Kim, K.H.; Cho, K.H.; Eun, H.C.; Chung, J.H. Heat modulation of tropoelastin, fibrillin-1, and matrix metalloproteinase-12 in human skin in vivo. J. Investig. Dermatol. 2005, 124, 70–78. [Google Scholar] [CrossRef] [PubMed]
- Taddese, S.; Weiss, A.S.; Neubert, R.H.; Schmelzer, C.E. Mapping of macrophage elastase cleavage sites in insoluble human skin elastin. Matrix Biol. 2008, 27, 420–428. [Google Scholar]
- Taddese, S.; Weiss, A.S.; Jahreis, G.; Neubert, R.H.; Schmelzer, C.E. In vitro degradation of human tropoelastin by MMP-12 and the generation of matrikines from domain 24. Matrix Biol. 2009, 28, 84–91. [Google Scholar] [PubMed]
- Imokawa, G.; Nakajima, H.; Ishida, K. Biological mechanisms underlying the ultraviolet light-induced formation of skin wrinkling and sagging II: over-expression of Neprilysin/Neutral endopeptidase via epithelial-mesenchymal interaction plays an essential role in wrinkling and sagging. Int. J. Mol. Sci. 2015, 16, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Morisaki, N.; Moriwaki, S.; Sugiyama-Nakagiri, Y.; Haketa, K.; Takema, Y.; Imokawa, G. Neprilysin is identical to skin fibroblast elastase. J. Biol. Chem. 2010, 285, 39819–39827. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Ezaki, Y.; Nagai, T.; Yoshioka, R.; Imokawa, G. Epithelial–mesenchymal interaction during UVB-induced up-regulation of neutral endopeptidase. Biochem. J. 2012, 443, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Chiang, H.M.; Lin, T.J.; Chiu, C.Y.; Chang, C.W.; Hsu, K.C.; Fan, P.C.; Wen, K.C. Coffea arabica extract and its constituents prevent photoaging by suppressing MMPs expression and MAP kinase pathway. Food Chem. Toxicol. 2011, 49, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Mantena, S.K.; Roy, A.M.; Katiyar, S.K. Epigallocatechin-3-Gallate inhibits photocarcinogenesis through inhibition of angiogenic factors and activation of CD8 + T cells in tumors. Photochem. Photobiol. 2005, 81, 1174–1179. [Google Scholar] [CrossRef] [PubMed]
- Poswar, F.O.; Fraga, C.A.; Farias, L.C.; Feltenberger, J.D.; Cruz, V.P.; Santos, S.H.; Silveira, C.M.; de Paula, A.M.B.; Guimarães, A.L.S. Immunohistochemical analysis of TIMP-3 and MMP-9 in actinic keratosis, squamous cell carcinoma of the skin, and basal cell carcinoma. Pathol. Res. Pract. 2013, 209, 705–709. [Google Scholar] [CrossRef] [PubMed]
- Ciurea, M.E.; Cernea, D.; Georgescu, C.C.; Cotoi, O.S.; Patrascu, V.; Parvanescu, H.; Popa, D.; Pârvănescu, V.; Ciurea, R.N.; Mercut, R. Expression of CXCR4, MMP-13 and β-catenin in different histological subtypes of facial basal cell carcinoma. Rom. J. Morphol. Embryol. 2013, 54, 949–951. [Google Scholar]
- De Oliveira Poswar, F.; de Carvalho Fraga, C.A.; Gomes, E.S.B.; Farias, L.C.; Souza, L.W.F.; Santos, S.H.S.; Gomez, R.S.; de-Paula, A.M.B.; Guimarães, A.L.S. Protein expression of MMP-2 and MT1-MMP in actinic keratosis, squamous cell carcinoma of the skin, and basal cell carcinoma. Int. J. Surg. Pathol. 2015, 23, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Tan, E.S.; Ee, M.; Shen, L.; Chua, H.; Chan, Y.H.; Tan, S.H. Basal cell carcinoma in Singapore: A prospective study on epidemiology and clinicopathological characteristics with a secondary comparative analysis between Singaporean Chinese and Caucasian patients. Australas. J. Dermatol. 2015, 56, 175–179. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.G.; Bennett, R.G. Basal cell carcinoma in Asians: A retrospective analysis of ten patients. J. Skin Cancer 2012, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Boyd, S.; Tolvanen, K.; Virolainen, S.; Kuivanen, T.; Kyllönen, L.; Saarialho-Kere, U. Differential expression of stromal MMP-1, MMP-9 and TIMP-1 in basal cell carcinomas of immunosuppressed patients and controls. Virchows. Arch. 2008, 452, 83–90. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.Y.; Cha, S.T.; Chang, C.C.; Hsiao, C.H.; Tan, C.T.; Lu, Y.C.; Jee, S.H.; Kuo, M.L. Involvement of matrix metalloproteinase-13 in stromal-cell-derived factor 1a-directed invasion of human basal cell carcinoma cells. Oncogene 2007, 26, 2491–2501. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.T.; Kim, H.S.; Yoo, N.J.; Lee, W.S.; Cho, B.K.; Reichrath, J. Increased immunoreactivity of membrane type-1 matrix metalloproteinase (MT1-MMP) and b-catenin in high-risk basal cell carcinoma. Br. Assoc. Dermatol. 2011, 165, 1197–1204. [Google Scholar] [CrossRef] [PubMed]
- Nan, H.; Niu, T.; Hunter, D.J.; Han, J. Missense polymorphisms in matrix metalloproteinase genes and skin cancer risk. Cancer Epidemiol. Biomark. Prev. 2008, 17, 3551–3557. [Google Scholar] [CrossRef] [PubMed]
- Chuang, H.C.; Su, C.Y.; Huang, H.Y.; Huang, C.C.; Chien, C.Y.; Du, Y.Y.; Chuang, J.H. Active matrix metalloproteinase-7 is associated with invasion in buccal squamous cell carcinoma. Mod. Pathol. 2008, 21, 1444–1450. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.S.; Lu, M.P.; Wu, M.T. Differential expression of matrix metalloproteinase-2 by fibroblasts in co-cultures with keratinocytes, basal cell carcinoma and melanoma. J. Dermatol. 2006, 33, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Hartmann-Petersen, S.; Tammi, R.H.; Tammi, M.I.; Kosma, V.M. Depletion of cell surface CD44 in nonmelanoma skin tumours is associated with increased expression of matrix metalloproteinase 7. Br. J. Dermatol. 2009, 160, 1251–1257. [Google Scholar] [CrossRef] [PubMed]
- Tjiu, J.W.; Chen, J.S.; Shun, C.T.; Lin, S.J.; Liao, Y.H.; Chu, C.Y.; Tsai, T.F.; Chiu, H.C.; Dai, Y.S.; Inoue, H.; et al. Tumor-associated macrophage-induced invasion and angiogenesis of human basal cell carcinoma cells by cyclooxygenase-2 induction. J. Investig. Dermatol. 2009, 129, 1016–1025. [Google Scholar] [CrossRef] [PubMed]
- Kivisaari, A.K.; Kallajoki, M.; Mirtti, T.; McGrath, J.A.; Bauer, J.W.; Weber, F.; Königová, R.; Sawamura, D.; Sato-Matsumura, K.C.; Shimizu, H.; et al. Transformation-specific matrix metalloproteinases (MMP)-7 and MMP-13 are expressed by tumour cells in epidermolysis bullosa-associated squamous cell carcinomas. Br. J. Dermatol. 2008, 158, 778–785. [Google Scholar] [CrossRef] [PubMed]
- Goodison, S.; Urquidi, V.; Tarin, D. CD44 cell adhesion molecules. J. Clin. Pathol. Mol. Pathol. 1999, 52, 189–196. [Google Scholar] [CrossRef]
- Ahokas, K.; Skoog, T.; Suomela, S.; Jeskanen, L.; Impola, U.; Isaka, K.; Saarialho-Kere, U. Matrilysin-2 (matrix metalloproteinase-26) is upregulated in keratinocytes during wound repair and early skin carcinogenesis. J. Investig. Dermatol. 2005, 124, 849–856. [Google Scholar] [CrossRef] [PubMed]
- Roh, M.R.; Zheng, Z.; Kim, H.S.; Kwon, J.E.; Jeung, H.C.; Rha, S.Y.; Chung, K.Y. Differential expression patterns of MMPs and their role in the invasion of epithelial premalignant tumors and invasive cutaneous squamous cell carcinoma. Exp. Mol. Pathol. 2012, 92, 236–242. [Google Scholar] [CrossRef] [PubMed]
- Briso, E.M.; Guinea-Viniegra, J.; Bakiri, L.; Rogon, Z.; Petzelbauer, P.; Eils, R.; Wolf, R.; Rincón, M.; Angel, P.; Wagner, E.F. Inflammation-mediated skin tumorigenesis induced by epidermal c-Fos. Genes Dev. 2013, 27, 1959–1973. [Google Scholar] [CrossRef] [PubMed]
- Prasad, N.B.; Fischer, A.C.; Chuang, A.Y.; Wright, J.M.; Yang, T.; Tsai, H.L.; Westra, W.H.; Liegeois, N.J.; Hess, A.D.; Tufaro, A.P. Differential expression of degradome components in cutaneous squamous cell carcinomas. Mod. Pathol. 2014, 27, 945–957. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.J.; Sanderson, B.J.; Zhang, W. Significant anti-invasive activities of α-mangostin from the mangosteen pericarp on two human skin cancer cell lines. Anticancer Res. 2012, 32, 3805–3816. [Google Scholar] [PubMed]
- Kessenbrock, K.; Wang, C.Y.; Werb, Z. Matrix metalloproteinases in stem cell regulation and cancer. Matrix Biol. 2015, 44, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Junttila, M.; Ala-aho, R.; Jokilehto, T.; Peltonen, J.; Kallajoki, M.; Grenman, R.; Jaakkola, P.; Westermarck, J.; Kähäri, V.M. p38a and p38d mitogen-activated protein kinase isoforms regulate invasion and growth of head and neck squamous carcinoma cells. Oncogene 2007, 26, 5267–5279. [Google Scholar] [CrossRef] [PubMed]
- Xia, Y.H.; Li, M.; Fu, D.D.; Xu, S.L.; Li, Z.G.; Liu, D.; Tian, Z.W. Effects of PTTG down-regulation on proliferation and metastasis of the SCL-1 cutaneous squamous cell carcinoma cell line. Asian Pac. J. Cancer Prev. 2013, 14, 6245–6248. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Pérez, M.; El-hajahmad, M.; Massaro, J.; Mahalingam, M. Expression of gelatinases (MMP-2, MMP-9) and gelatinase activator (MMP-14) in actinic keratosis and in in situ and invasive squamous cell carcinoma. Am. J. Dermatopathol. 2012, 7, 723–728. [Google Scholar] [CrossRef] [PubMed]
- Kuivanen, T.; Jeskanen, L.; Kyllonen, L.; Isaka, K.; Saarialho-Kere, U. Matrix metalloproteinase-26 is present more frequently in squamous cell carcinomas of immunosuppressed compared with immunocompetent patients. J. Cutan. Pathol. 2009, 36, 929–936. [Google Scholar] [CrossRef] [PubMed]
- Ahmed Haji Omar, A.; Haglund, C.; Virolainen, S.; Häyry, V.; Atula, T.; Kontio, R.; Salo, T.; Sorsa, T.; Hagström, J. MMP-7, MMP-8, and MMP-9 in oral and cutaneous squamous cell carcinomas. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2015, 119, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Lederle, W.; Hartenstein, B.; Meides, A.; Kunzelmann, H.; Werb, Z.; Angel, P.; Mueller, M.M. MMP13 as a stromal mediator in controlling persistent angiogenesis in skin carcinoma. Carcinogenesis 2010, 31, 1175–1184. [Google Scholar] [CrossRef] [PubMed]
- Shen, L.; Lio, L.; Yang, Z.; Jiang, N. Identification of genes and signaling pathways associated with squamous cell carcinoma by bioinformatics analysis. Oncol. Lett. 2016, 11, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Xu, G.; Yang, L.; Wang, Y.; Li, Y.; Mchepange, U.O.; Shen, G.; Tu, Y.; Tao, J. Expression of hypoxia-inducible factor-1alpha, vascular endothelial growth factor and prolyl hydroxylase domain protein 2 in cutaneous squamous cell carcinoma and precursor lesions and their relationship with histological stages and clinical features. J. Dermatol. 2014, 41, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Vosseler, S.; Lederle, W.; Airola, K.; Obermueller, E.; Fusenig, N.E.; Mueller, M.M. Distinct progression-associated expression of tumor and stromal MMPs in HaCaT skin SCCs correlates with onset of invasion. Int. J. Cancer 2009, 125, 2296–2306. [Google Scholar] [CrossRef] [PubMed]
- Meides, A.; Gutschalk, C.M.; Devel, L.; Beau, F.; Czarny, B.; Hensler, S.; Neugebauer, J.; Dive, V.; Angel, P.; Mueller, M.M. Effects of selective MMP-13 inhibition in squamous cell carcinoma depend on estrogen. Int. J. Cancer 2014, 135, 2749–2759. [Google Scholar] [CrossRef] [PubMed]
- Lederle, W.; Depner, S.; Schnur, S.; Obermueller, E.; Catone, N.; Just, A.; Fusenig, N.E.; Mueller, M.M. IL-6 promotes malignant growth of skin SCCs by regulating a network of autocrine and paracrine cytokines. Int. J. Cancer 2011, 128, 2803–2814. [Google Scholar] [CrossRef] [PubMed]
- Leivonen, S.K.; Ala-aho, R.; Koli, K.; Grénman, R.; Peltonen, J.; Kahari, V.M. Activation of Smad signaling enhances collagenase-3 (MMP-13) expression and invasion of head and neck squamous carcinoma cells. Oncogene 2006, 25, 2588–2600. [Google Scholar] [CrossRef] [PubMed]
- Boyd, S.; Virolainen, S.; Parssinen, J.; Skoog, T.; Van Hogerlinden, M.; Latonen, L.; Kyllönen, L.; Toftgard, R.; Saarialho-Kere, U. MMP-10 (Stromelysin-2) and MMP-21 in human and murine squamous cell cancer. Exp. Dermatol. 2009, 18, 1044–1052. [Google Scholar] [CrossRef] [PubMed]
- McCawley, L.J.; Wright, J.; LaFleur, B.J.; Crawford, H.C.; Matrisian, L.M. Keratinocyte expression of MMP3 enhances differentiation and prevents tumor establishment. Am. J. Pathol. 2008, 173, 1528–1539. [Google Scholar] [CrossRef] [PubMed]
- Meephansan, J.; Komine, M.; Tsuda, H.; Ohtsuki, M. Suppressive effect of calcipotriol on the induction of matrix metalloproteinase (MMP)-9 and MMP-13 in a human squamous cell carcinoma cell line. Clin. Exp. Dermatol. 2012, 37, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Leight, J.L.; Tokuda, E.Y.; Jones, C.E.; Lin, A.J.; Anseth, K.S. Multifunctional bioscaffolds for 3D culture of melanoma cells reveal increased MMP activity and migration with BRAF kinase inhibition. Proc. Natl. Acad. Sci. USA 2015, 112, 5366–5371. [Google Scholar] [CrossRef] [PubMed]
- Rotte, A.; Martinka, M.; Li, G. MMP2 expression is a prognostic marker for primary melanoma patients. Cell Oncol. 2012, 35, 207–216. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Chen, Y.; Huang, L.; Yu, J. Evaluation of heparanase and matrix metalloproteinase-9 in patients with cutaneous malignant melanoma. J. Dermatol. 2012, 39, 339–343. [Google Scholar] [CrossRef] [PubMed]
- Ye, S.; Dhillon, S.; Turner, S.J.; Bateman, A.C.; Theaker, J.M.; Pickering, R.M.; Day, I.; Howell, W.M. Invasiveness of cutaneous malignant melanoma is influenced by matrix metalloproteinase-1 gene polymorphism. Cancer Res. 2001, 61, 1296–1298. [Google Scholar] [PubMed]
- Blackburn, J.S.; Liu, I.; Coon, C.I.; Brinckerhoff, C.E. A matrix metalloproteinase-1/Protease activated receptor-1 signaling axis promotes melanoma invasion and metastasis. Oncogene 2009, 28, 4237–4248. [Google Scholar] [CrossRef] [PubMed]
- Kondratiev, S.; Gnepp, D.R.; Yakirevich, E.; Sabo, E.; Annino, D.J.; Rebeiz, E.; Laver, N.V. Expression and prognostic role of MMP2, MMP9, MMP13, and MMP14 matrix metalloproteinases in sinonasal and oral malignant melanomas. Hum. Pathol. 2008, 39, 337–343. [Google Scholar] [CrossRef] [PubMed]
- Väisänen, A.H.; Kallioinen, M.; Turpeenniemi-Hujanen, T. Comparison of the prognostic value of matrix metalloproteinases 2 and 9 in cutaneous melanoma. Hum. Pathol. 2008, 39, 377–385. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, U.B.; Houben, R.; Bröcker, E.B.; Becker, J.C. Role of matrix metalloproteinases in melanoma cell invasion. Biochimie 2005, 87, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Foda, H.D.; Zucker, S. Matrix metalloproteinases in cancer invasion, metastasis and angiogenesis. Drug Discov. Today 2001, 6, 478–482. [Google Scholar] [CrossRef]
- Hwang, H.W.; Baxter, L.L.; Loftus, S.K.; Cronin, J.C.; Trivedi, N.S.; Borate, B.; Pavan, W.J. Distinct microRNA expression signatures are associated with melanoma subtypes and are regulated by HIF1A. Pigment. Cell Melanoma Res. 2014, 27, 777–787. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Zhang, D.; Zhang, S.; Zhang, W.; Guo, H.; Zhao, X. Hypoxia influences vasculogenic mimicry channel formation and tumor invasion-related protein expression in melanoma. Cancer Lett. 2007, 249, 188–197. [Google Scholar] [CrossRef] [PubMed]
- Hanna, S.C.; Krishnan, B.; Bailey, S.T.; Moschos, S.J.; Kuan, P.F.; Shimamura, T.; Osborne, L.D.; Siegel, M.B.; Duncan, L.M.; O’Brien III, E.T.; et al. HIF1α and HIF2α independently activate SRC to promote melanoma metastasis. J. Clin. Investig. 2013, 123, 2078–2093. [Google Scholar] [CrossRef] [PubMed]
- Kim, A.; Im, M.; Yim, N.H.; Ma, J.Y. Reduction of metastatic and angiogenic potency of malignant cancer by Eupatorium fortunei via suppression of MMP-9 activity and VEGF production. Sci. Rep. 2014, 4, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Tas, F.; Duranyildiz, D.; Oguz, H.; Camlica, H.; Yasasever, V.; Topuz, E. Circulating levels of vascular endothelial growth factor (VEGF), matrix metalloproteinase-3 (MMP-3), and BCL-2 in malignant melanoma. Med. Oncol. 2008, 25, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Shaverdashvili, K.; Zhang, K.; Osman, I.; Honda, K.; Jobava, R.; Bedogni, B. MT1-MMP dependent repression of the tumor suppressor SPRY4 contributes to MT1-MMP driven melanoma cell motility. Oncotarget 2015, 6, 33512–33522. [Google Scholar] [PubMed]
- Moro, N.; Mauch, C.; Zigrino, P. Metalloproteinases in melanoma. Eur. J. Cell Biol. 2014, 93, 23–29. [Google Scholar] [CrossRef] [PubMed]
- Tatti, O.; Arjama, M.; Ranki, A.; Weiss, S.J.; Keski-Oja, J.; Lehti, K. Membrane-type-3 matrix metalloproteinase (MT3-MMP) functions as a matrix composition-dependent effector of melanoma cell invasion. PLoS ONE 2011, 6, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Whipple, C.A.; Brinckerhoff, C.E. BRAFV600E melanoma cells secrete factors that activate stromal fibroblasts and enhance tumourigenicity. Br. J. Cancer 2014, 111, 1625–1633. [Google Scholar] [CrossRef] [PubMed]
- Vlaykova, T.; Kurzawski, M.; Tacheva, T.; Dimov, D.; Gulubova, M.; Yovchev, Y.; Chakarov, S.; Drozdzik, M. Investigation of the role of MMP3 -1171insA polymorphism in cutaneous malignant melanoma a preliminary study. Biotechnol. Biotechnol. Equip. 2014, 28, 904–910. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, K.; Kawakami, T.; Watabe, H.; Itoh, F.; Mizoguchi, M.; Soma, Y. Expression of matrilysin (matrix metalloproteinase-7) in primary cutaneous and metastatic melanoma. Br. J. Dermatol. 2007, 156, 613–619. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhu, S.; Yang, Y.; Ma, X.; Guo, S. Matrix metalloproteinase-12 expression is increased in cutaneous melanoma and associated with tumor aggressiveness. Tumor Biol. 2015, 36, 8593–8600. [Google Scholar] [CrossRef] [PubMed]
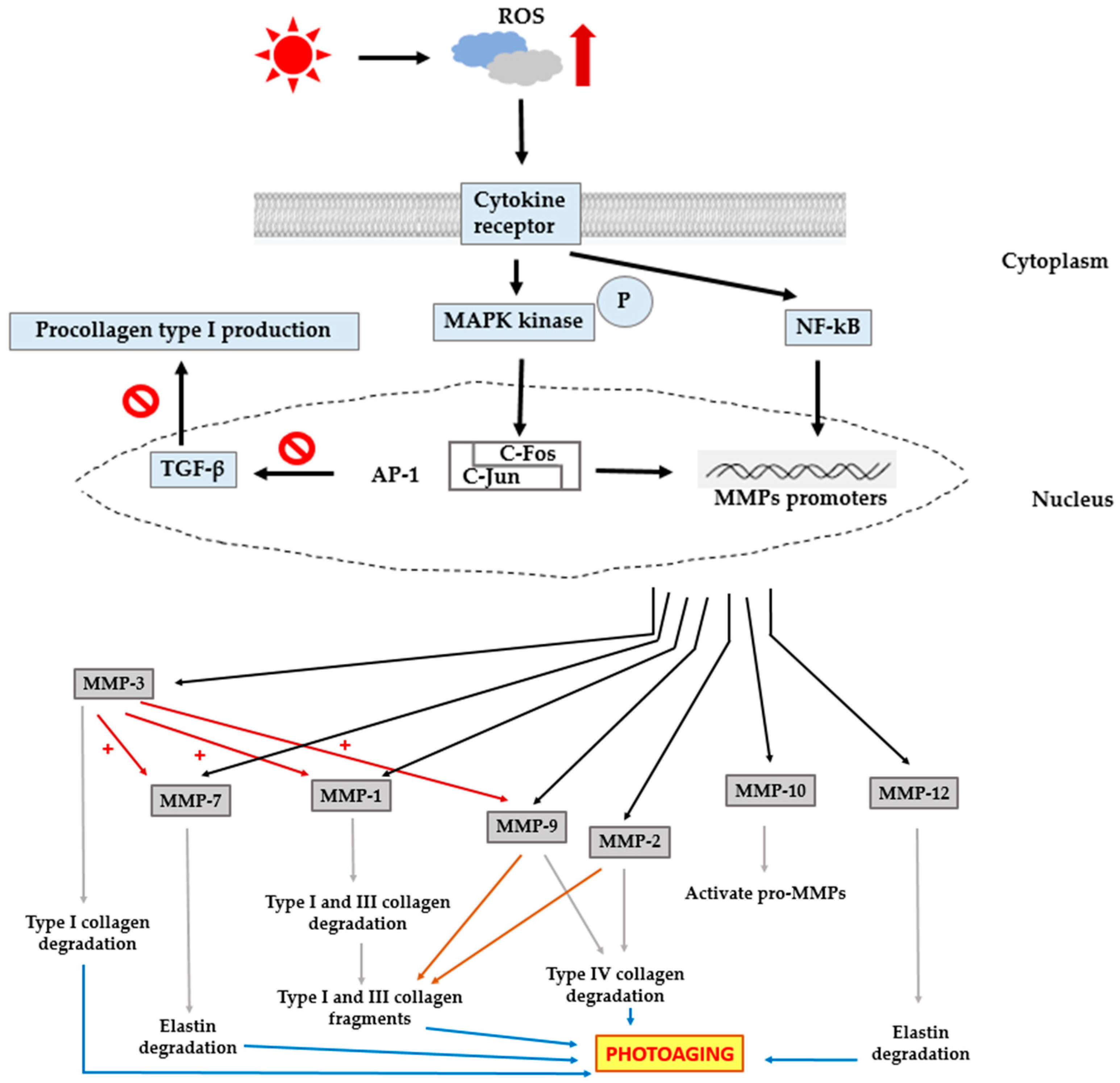
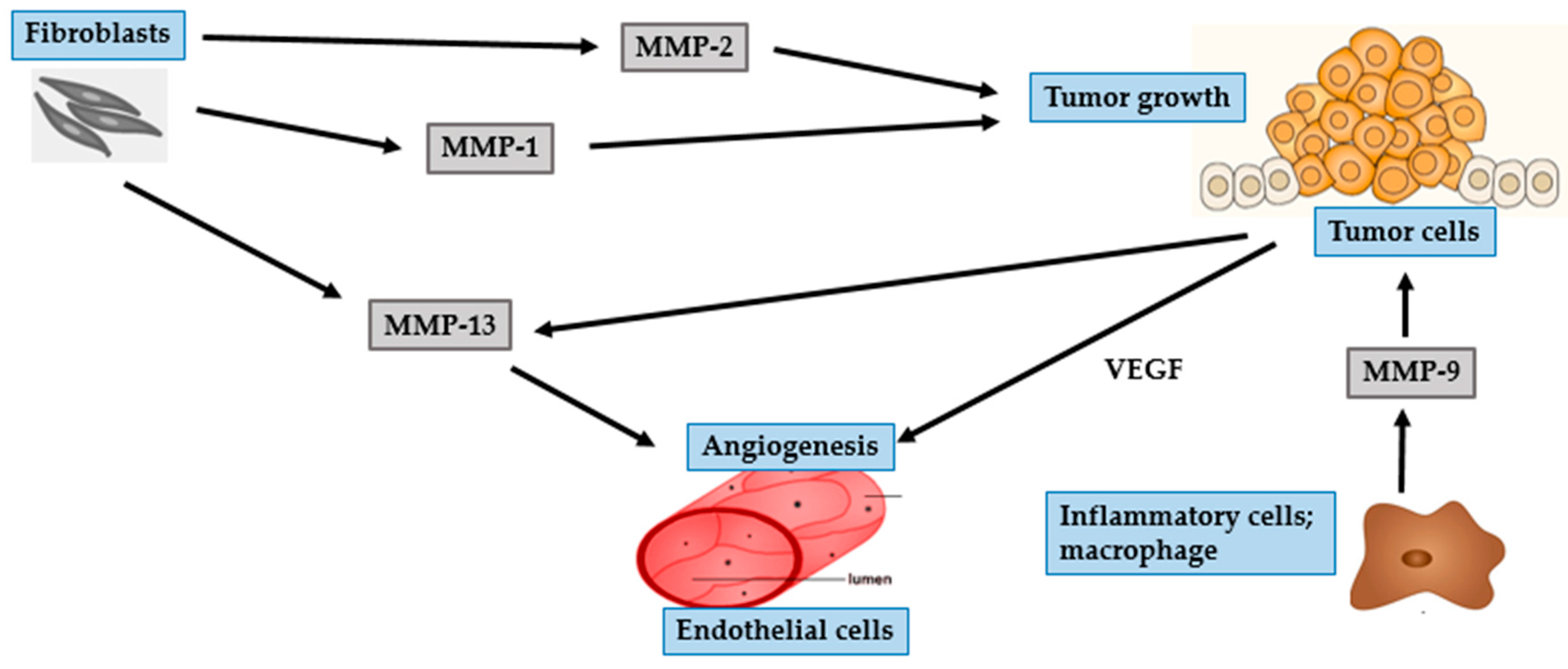
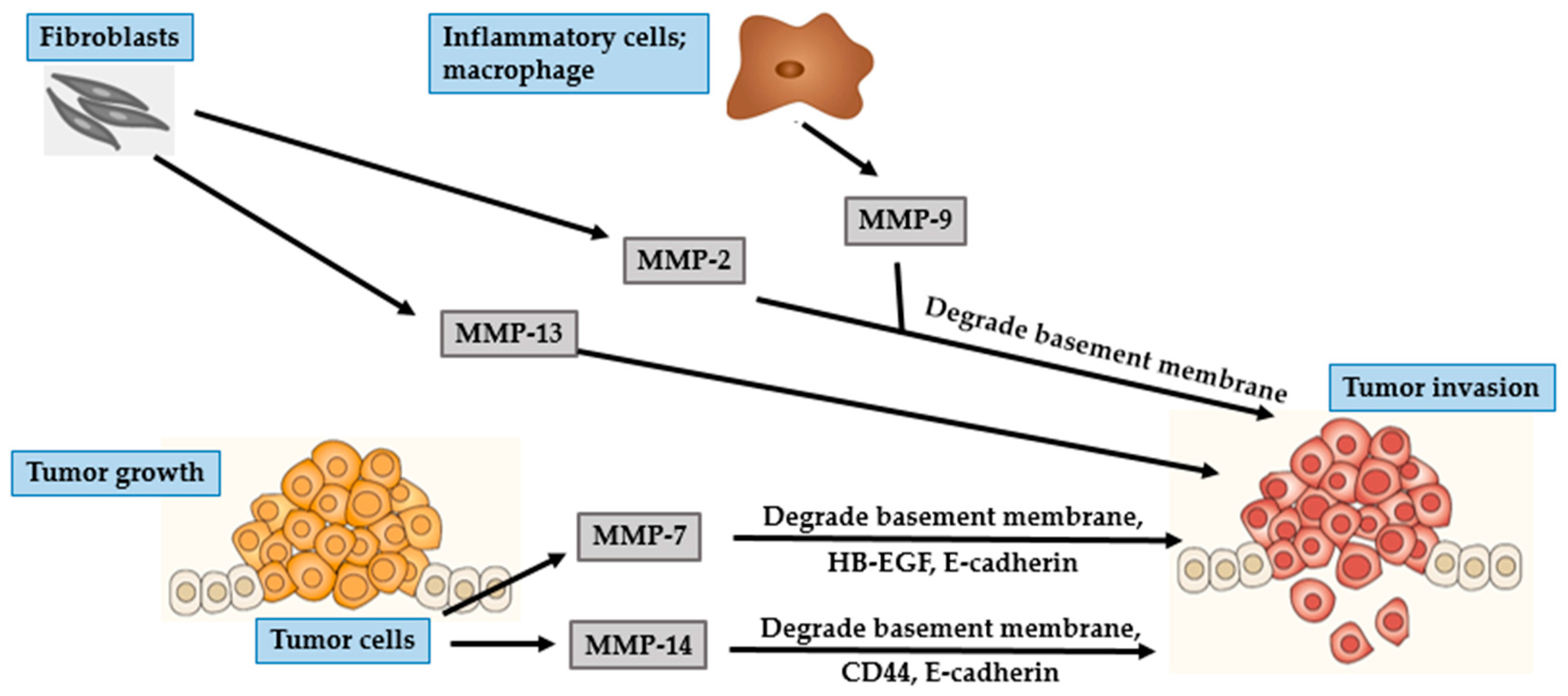
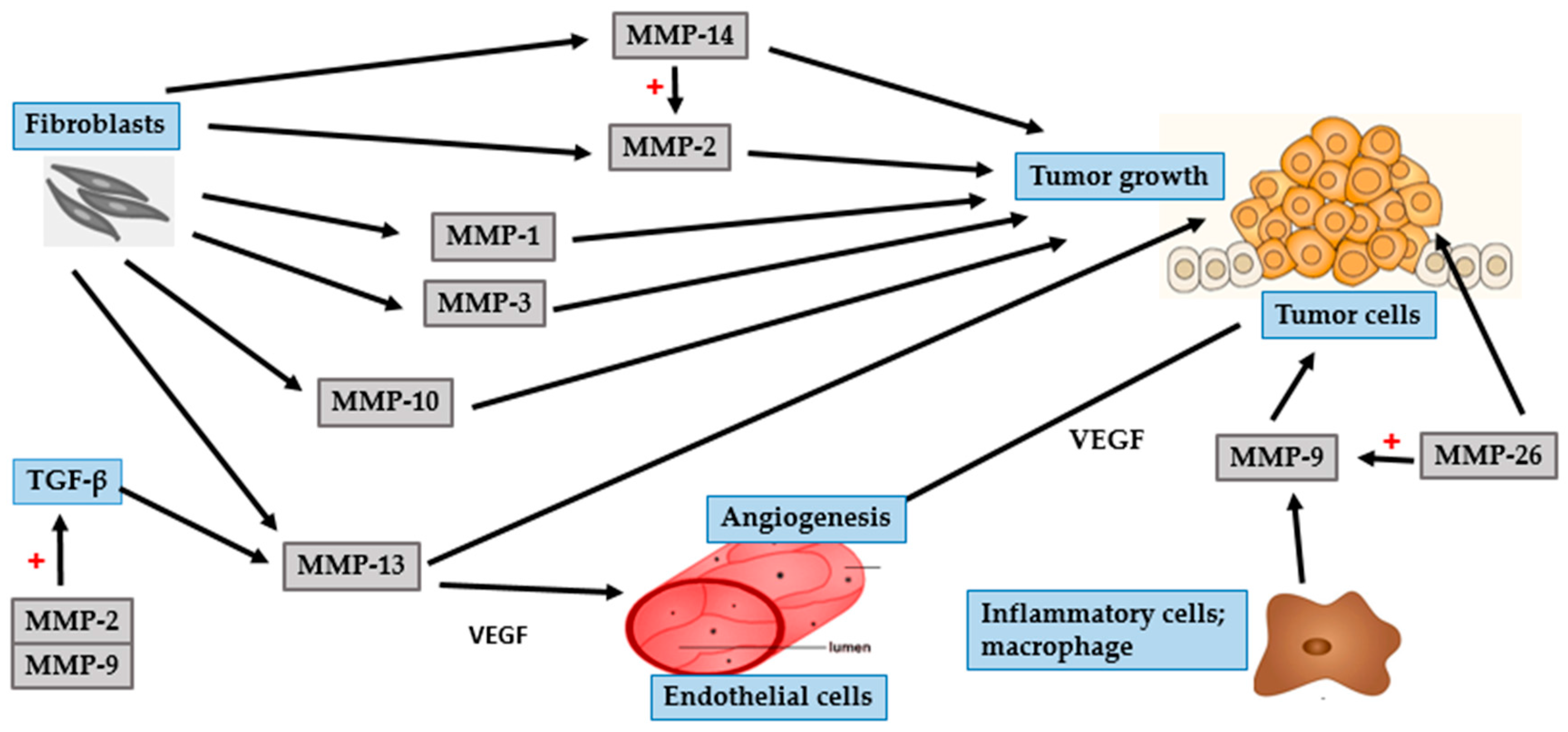
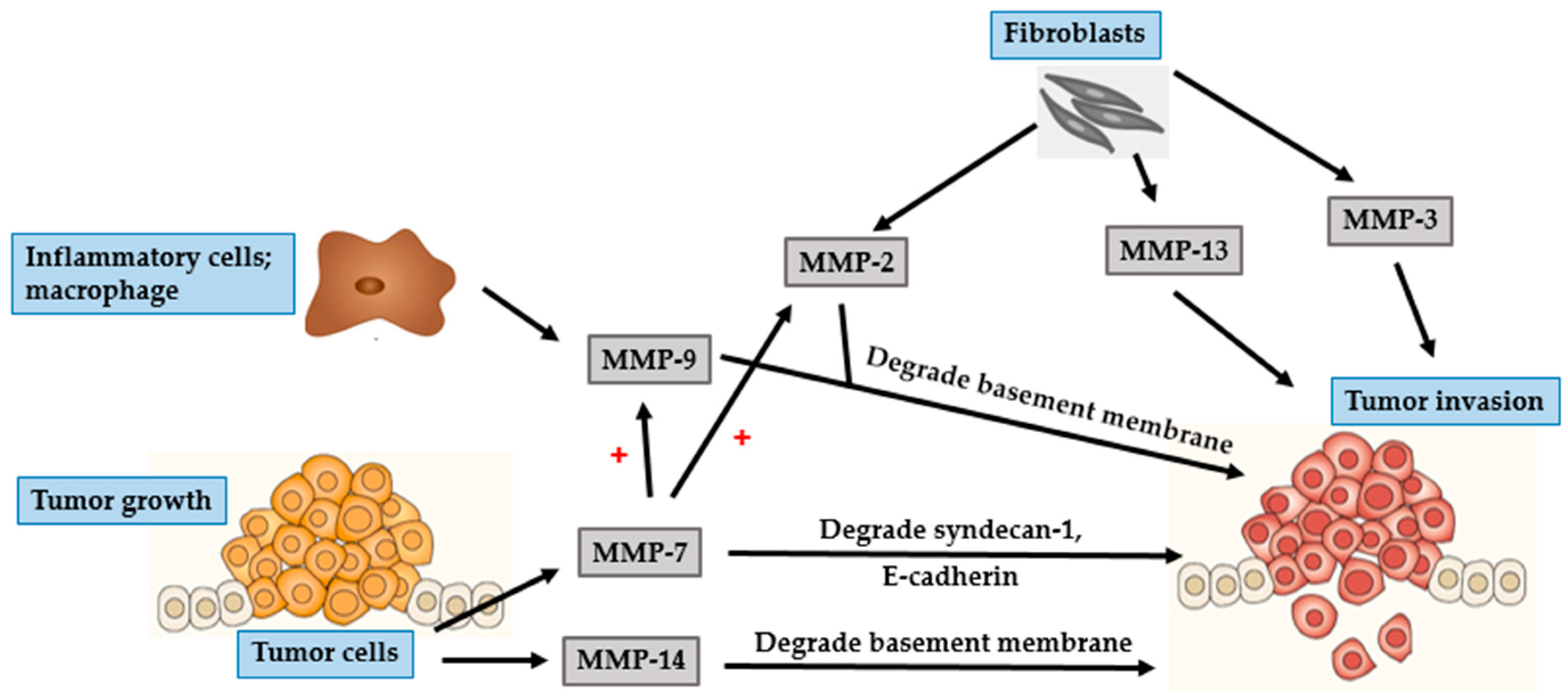
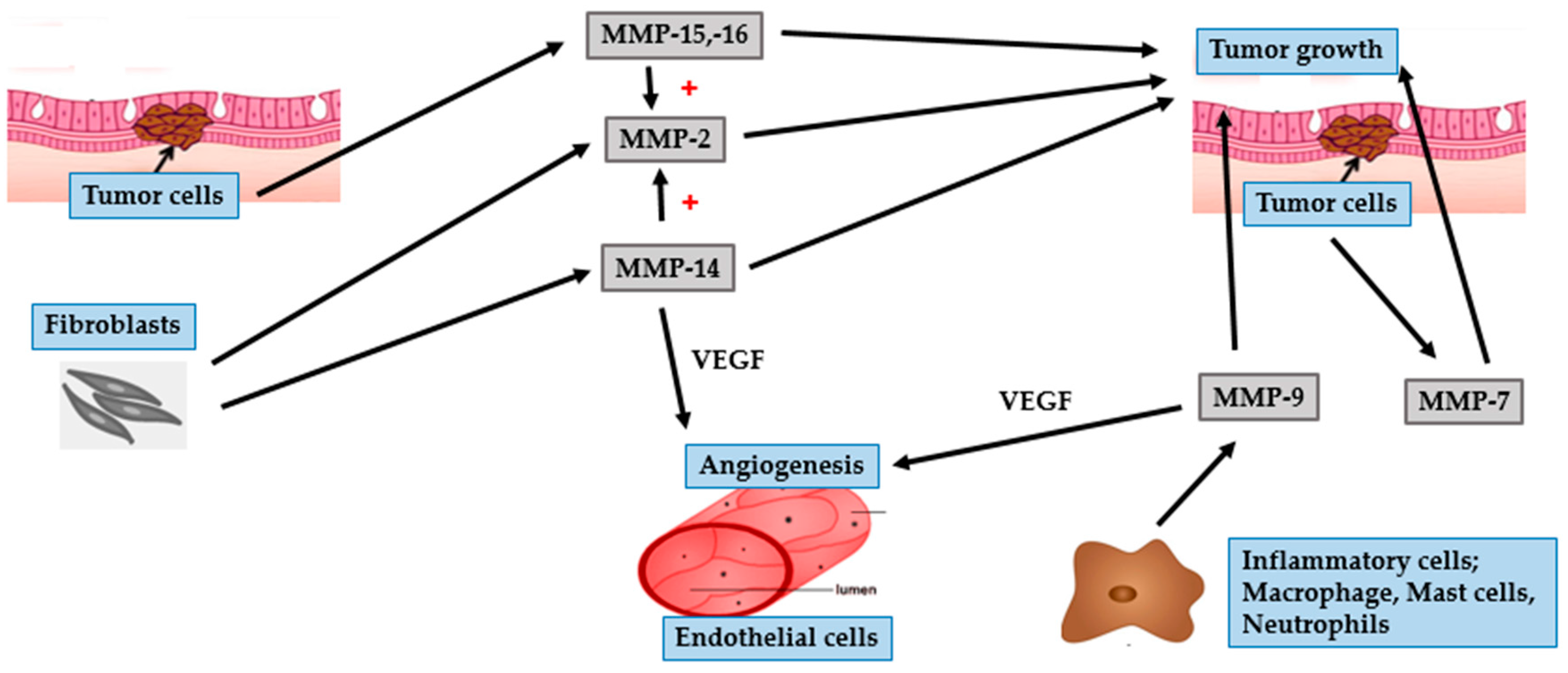
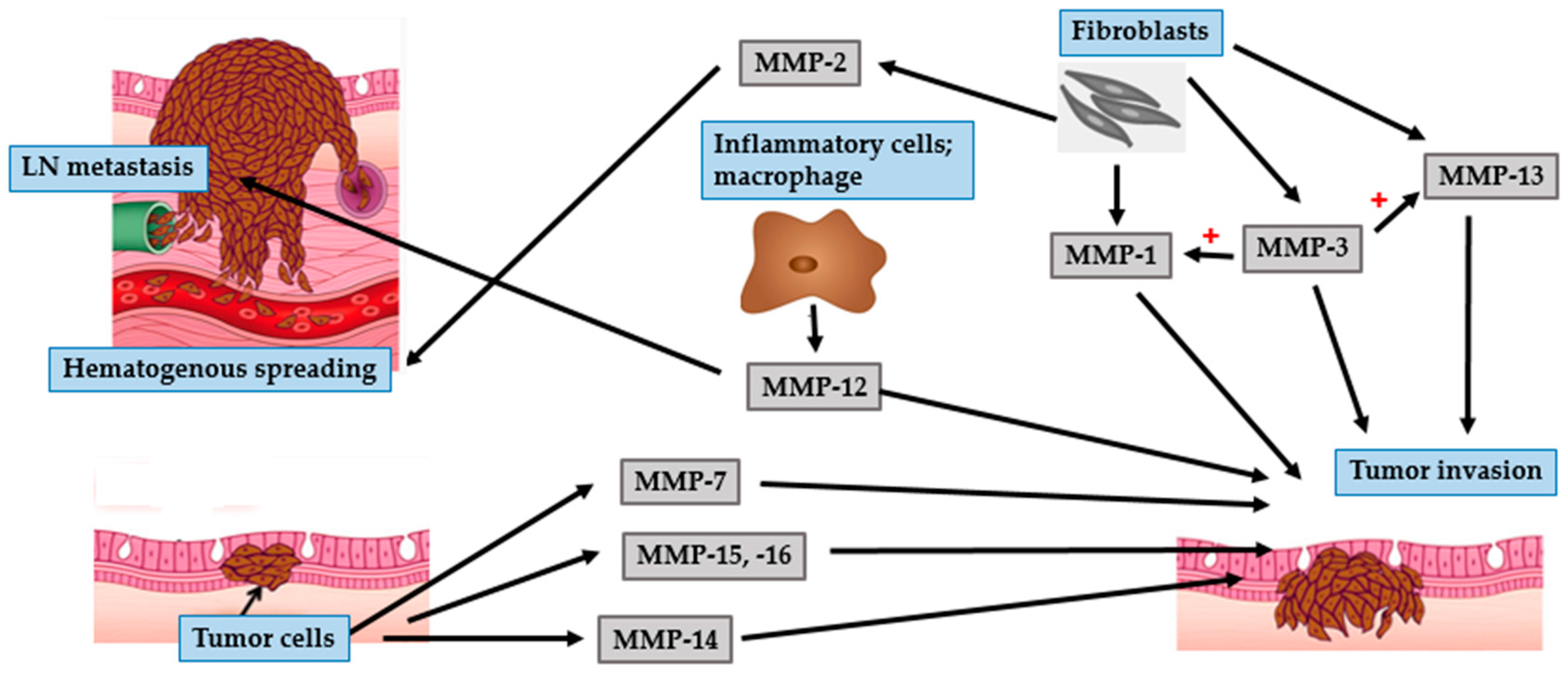
| MMP Subgroup | MMP Number | Alternate Name | Role in Photoaging | Role in Photocarcinogenesis |
|---|---|---|---|---|
| Collagenases | MMP-1 | - Interstitial collagenase
-Type I Collagenase | - Collagen type I and III degradation | - Tumor growth in BCC and SCC
- Facilitate tumor invasion in melanoma |
| MMP-8 | - Neutrophil collagenase | - Limited role | - Limited role in BCC and SCC
- Increased risk of malignant melanoma | |
| MMP-13 | - Collagenase-3 | - Limited role | - Tumor invasion and angiogenesis in BCC and SCC
- Involved in invasive VGP of melanoma | |
| Gelatinases | MMP-2 | - Gelatinase-A
- 72 kDa type IV collagenase | - Collagen type IV degradation | - Growth initiation and tumor invasion in BCC and SCC
- Hematogenous metastasis in melanoma |
| MMP-9 | - Gelatinase-B
- 92-kDa type IV collagenase | - Degrade collagen type IV | - Growth initiation and tumor invasion in BCC and SCC
- Related to the RGP of melanoma and tumor angiogenesis | |
| Stromelysins | MMP-3 | - Stromelysin-1
- Proteoglycanase - Transin-1 | - Collagen type I degradation
- Activate MMP-1, -7, and -9 | - Tumor progression and metastasis in SCC
- Activate pro-MMPs in melanoma |
| MMP-10 | - Stromelysin-2
- Transin-2 | - Activate pro-MMPs | - Tumor initiation in SCC | |
| MMP-11 | - Stromelysin-3 | - | - | |
| Matrilysins | MMP-7 | - Matrilysin-1
- Pump-1 | - Elastin degradation | - Tumor invasion |
| MMP-26 | - Matrilysin-2
- Endometase | - | -Limited role in BCC
-Activate MMP-9 and promote tumor growth in SCC | |
| Membrane-type | MMP-14 | - MT1-MMP | - | - Tumor invasion
- Activate MMP-2 - Tumor angiogenesis in melanoma |
| MMP-15 | - MT2-MMP | - | - Tumor invasion in melanoma | |
| MMP-16 | - MT3-MMP | - | - Tumor invasion in melanoma | |
| MMP-17 | - MT4-MMP | - | - | |
| MMP-24 | - MT5-MMP | - | - | |
| MMP-25 | - MT6-MMP | - | - | |
| Other types | MMP-12 | - Metalloelastase | - Elastin degradaion | - Tumor invasion, lymph node metastasis in melanoma |
| MMP-19 | - RASI-1 | - | - | |
| MMP-20 | - Enamelysin | - | - | |
| MMP-21 | - | - | - | |
| MMP-22 | - | - | - | |
| MMP-23 | - | - | - | |
| MMP-28 | - Epilysin | - | - |
| MMPs | UV-A | UV-B |
|---|---|---|
| Collagenases | ||
| MMP-1 | + | ++ |
| MMP-8 | NA | NA |
| MMP-13 | + | + |
| Gelatinases | ||
| MMP-2 | + | + |
| MMP-9 | + | + |
| Stromelysins | ||
| MMP-3 | + | ++ |
| MMP-10 | + | ++ |
| Matrilysins | ||
| MMP-7 | + | + |
| MMP-26 | NA | NA |
| MT-MMPs | ||
| MMP-14 | NA | NA |
| MMP-15,16 | NA | NA |
| Other types | ||
| MMP-12 | ++ | + |
| Mechanisms | Protective Agents |
|---|---|
| MMP inhibitors | Galla chinensis |
| Neonauclea reticulate | |
| Coffea Arabica | |
| Ixora parviflora | |
| Free radical scavengers | Coffea Arabica |
| Terminalia catappa | |
| Emblica officinalis | |
| Epigallocatechin-3-gallate (EGCG) | |
| Gynura procumbens | |
| Caesalpinia sappan L. |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pittayapruek, P.; Meephansan, J.; Prapapan, O.; Komine, M.; Ohtsuki, M. Role of Matrix Metalloproteinases in Photoaging and Photocarcinogenesis. Int. J. Mol. Sci. 2016, 17, 868. https://doi.org/10.3390/ijms17060868
Pittayapruek P, Meephansan J, Prapapan O, Komine M, Ohtsuki M. Role of Matrix Metalloproteinases in Photoaging and Photocarcinogenesis. International Journal of Molecular Sciences. 2016; 17(6):868. https://doi.org/10.3390/ijms17060868
Chicago/Turabian StylePittayapruek, Pavida, Jitlada Meephansan, Ornicha Prapapan, Mayumi Komine, and Mamitaro Ohtsuki. 2016. "Role of Matrix Metalloproteinases in Photoaging and Photocarcinogenesis" International Journal of Molecular Sciences 17, no. 6: 868. https://doi.org/10.3390/ijms17060868
APA StylePittayapruek, P., Meephansan, J., Prapapan, O., Komine, M., & Ohtsuki, M. (2016). Role of Matrix Metalloproteinases in Photoaging and Photocarcinogenesis. International Journal of Molecular Sciences, 17(6), 868. https://doi.org/10.3390/ijms17060868







