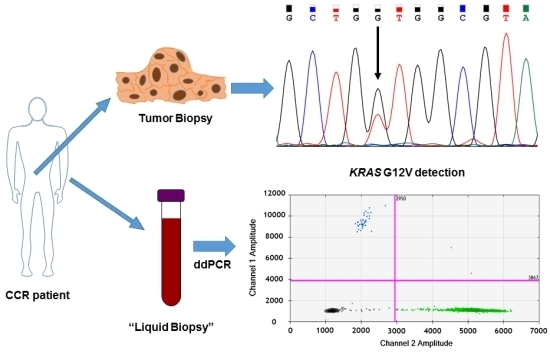
KRAS G12V Mutation Detection by Droplet Digital PCR in Circulating Cell-Free DNA of Colorectal Cancer Patients
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Cells
4.2. Patients and Healthy Subjects
4.3. Sample Collection
4.4. DNA Extraction
4.5. Mutation Detection by ddPCR
4.6. Statistical Analyses
5. Conclusion
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| KRAS | Kirsten rat sarcoma viral oncogene homolog |
| EGFR | epidermal growth factor receptor |
| BRAF | B-Raf proto-oncogene serine/threonine kinase |
| DdPCR | droplet digital PCR |
| CtDNA | circulating tumor DNA |
| COLD-PCR | coamplification at lower denaturation temperature-PCR |
| BEAMing | beads, emulsions, amplification, and magnetic |
References
- Mouliere, F.; El Messaoudi, S.; Pang, D.; Dritschilo, A.; Thierry, A.R. Multi-marker analysis of circulating cell-free DNA toward personalized medicine for colorectal cancer. Mol. Oncol. 2014, 8, 927–941. [Google Scholar] [CrossRef] [PubMed]
- Heitzer, E.; Ulz, P.; Geigl, J.B. Circulating tumor DNA as a liquid biopsy for cancer. Clin. Chem. 2015, 61, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Crowley, E.; di Nicolantonio, F.; Loupakis, F.; Bardelli, A. Liquid biopsy: Monitoring cancer-genetics in the blood. Nat. Rev. Clin. Oncol. 2013, 10, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Bettegowda, C.; Sausen, M.; Leary, R.J.; Kinde, I.; Wang, Y.; Agrawal, N.; Bartlett, B.R.; Wang, H.; Luber, B.; Alani, R.M.; et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl. Med. 2014, 6, 224ra24. [Google Scholar] [CrossRef] [PubMed]
- Herreros-Villanueva, M.; Chen, C.C.; Yuan, S.S.; Liu, T.C.; Er, T.K. KRAS mutations: Analytical considerations. Clin. Chim. Acta 2014, 431, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Huggett, J.F.; Whale, A. Digital PCR as a novel technology and its potential implications for molecular diagnostics. Clin. Chem. 2013, 59, 1691–1693. [Google Scholar] [CrossRef] [PubMed]
- Vogelstein, B.; Kinzler, K.W. Digital PCR. Proc. Natl. Acad. Sci. USA 1999, 96, 9236–9241. [Google Scholar] [CrossRef] [PubMed]
- Morley, A.A. Digital PCR: A brief history. Biomol. Detect. Quant. 2014, 1, 1–2. [Google Scholar] [CrossRef]
- Huggett, J.F.; Cowen, S.; Foy, C.A. Considerations for digital PCR as an accurate molecular diagnostic tool. Clin. Chem. 2015, 61, 79–88. [Google Scholar] [CrossRef] [PubMed]
- Bizouarn, F. Clinical applications using digital PCR. Methods Mol. Biol. 2014, 1160, 189–214. [Google Scholar] [PubMed]
- Huggett, J.F.; Foy, C.A.; Benes, V.; Emslie, K.; Garson, J.A.; Haynes, R.; Hellemans, J.; Kubista, M.; Mueller, R.D.; Nolan, T.; et al. The digital MIQE guidelines: Minimum information for publication of quantitative digital PCR experiments. Clin. Chem. 2013, 59, 892–902. [Google Scholar] [CrossRef] [PubMed]
- Azuara, D.; Ginesta, M.M.; Gausachs, M.; Rodriguez-Moranta, F.; Fabregat, J.; Busquets, J.; Pelaez, N.; Boadas, J.; Galter, S.; Moreno, V.; et al. Nanofluidic digital PCR for KRAS mutation detection and quantification in gastrointestinal cancer. Clin. Chem. 2012, 58, 1332–1341. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Irwanto, A.; Guo, Y.M.; Bei, J.X.; Wu, Q.; Chen, G.; Zhang, T.P.; Lei, J.J.; Feng, Q.S.; Chen, L.Z.; et al. Exome sequencing and digital PCR analyses reveal novel mutated genes related to the metastasis of pancreatic ductal adenocarcinoma. Cancer Biol. Ther. 2012, 13, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Janku, F.; Angenendt, P.; Tsimberidou, A.M.; Fu, S.; Naing, A.; Falchook, G.S.; Hong, D.S.; Holley, V.R.; Cabrilo, G.; Wheler, J.J.; et al. Actionable mutations in plasma cell-free DNA in patients with advanced cancers referred for experimental targeted therapies. Oncotarget 2015, 6, 12809–12821. [Google Scholar] [CrossRef] [PubMed]
- Tabernero, J.; Lenz, H.J.; Siena, S.; Sobrero, A.; Falcone, A.; Ychou, M.; Humblet, Y.; Bouche, O.; Mineur, L.; Barone, C.; et al. Analysis of circulating DNA and protein biomarkers to predict the clinical activity of regorafenib and assess prognosis in patients with metastatic colorectal cancer: A retrospective, exploratory analysis of the CORRECT trial. Lancet Oncol. 2015, 16, 937–948. [Google Scholar] [CrossRef]
- Oxnard, G.R.; Paweletz, C.P.; Kuang, Y.; Mach, S.L.; O’Connell, A.; Messineo, M.M.; Luke, J.J.; Butaney, M.; Kirschmeier, P.; Jackman, D.M.; et al. Noninvasive detection of response and resistance in EGFR-mutant lung cancer using quantitative next-generation genotyping of cell-free plasma DNA. Clin. Cancer Res. 2014, 20, 1698–1705. [Google Scholar] [CrossRef] [PubMed]
- Taly, V.; Pekin, D.; Benhaim, L.; Kotsopoulos, S.K.; Le Corre, D.; Li, X.; Atochin, I.; Link, D.R.; Griffiths, A.D.; Pallier, K.; et al. Multiplex picodroplet digital PCR to detect KRAS mutations in circulating DNA from the plasma of colorectal cancer patients. Clin. Chem. 2013, 59, 1722–1731. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.S.; Er, T.K.; Lu, H.C.; Yeh, K.T.; Chang, J.G. Detection of KRAS codon 12 and 13 mutations by mutant-enriched PCR assay. Clin. Chim. Acta 2014, 436, 169–175. [Google Scholar] [CrossRef] [PubMed]
- Tone, A.A.; McConechy, M.K.; Yang, W.; Ding, J.; Yip, S.; Kong, E.; Wong, K.K.; Gershenson, D.M.; Mackay, H.; Shah, S.; et al. Intratumoral heterogeneity in a minority of ovarian low-grade serous carcinomas. BMC Cancer 2014, 14, 982. [Google Scholar] [CrossRef] [PubMed]
- Laurent-Puig, P.; Pekin, D.; Normand, C.; Kotsopoulos, S.K.; Nizard, P.; Perez-Toralla, K.; Rowell, R.; Olson, J.; Srinivasan, P.; Le Corre, D.; et al. Clinical relevance of KRAS-mutated subclones detected with picodroplet digital PCR in advanced colorectal cancer treated with anti-EGFR therapy. Clin. Cancer Res. 2015, 21, 1087–1097. [Google Scholar] [CrossRef] [PubMed]
- Thierry, A.R.; Mouliere, F.; El Messaoudi, S.; Mollevi, C.; Lopez-Crapez, E.; Rolet, F.; Gillet, B.; Gongora, C.; Dechelotte, P.; Robert, B.; et al. Clinical validation of the detection of KRAS and BRAF mutations from circulating tumor DNA. Nat. Med. 2014, 20, 430–435. [Google Scholar] [CrossRef] [PubMed]
- Al-Mulla, F.; Milner-White, E.J.; Going, J.J.; Birnie, G.D. Structural differences between valine-12 and aspartate-12 Ras proteins may modify carcinoma aggression. J. Pathol. 1999, 187, 433–438. [Google Scholar] [CrossRef]
- Andreyev, H.J.; Norman, A.R.; Cunningham, D.; Oates, J.R.; Clarke, P.A. Kirsten ras mutations in patients with colorectal cancer: The multicenter “RASCAL” study. J. Natl. Cancer Inst. 1998, 90, 675–84. [Google Scholar] [CrossRef] [PubMed]
- Andreyev, H.J.; Norman, A.R.; Cunningham, D.; Oates, J.; Dix, B.R.; Iacopetta, B.J.; Young, J.; Walsh, T.; Ward, R.; Hawkins, N.; et al. Kirsten ras mutations in patients with colorectal cancer: The ‘RASCAL II’ study. Br. J. Cancer 2001, 85, 692–696. [Google Scholar] [CrossRef] [PubMed]
- Winder, T.; Mundlein, A.; Rhomberg, S.; Dirschmid, K.; Hartmann, B.L.; Knauer, M.; Drexel, H.; Wenzl, E.; de Vries, A.; Lang, A. Different types of K-Ras mutations are conversely associated with overall survival in patients with colorectal cancer. Oncol. Rep. 2009, 21, 1283–1287. [Google Scholar] [CrossRef] [PubMed]
- Imamura, Y.; Morikawa, T.; Liao, X.; Lochhead, P.; Kuchiba, A.; Yamauchi, M.; Qian, Z.R.; Nishihara, R.; Meyerhardt, J.A.; Haigis, K.M.; et al. Specific mutations in KRAS codons 12 and 13, and patient prognosis in 1075 BRAF wild-type colorectal cancers. Clin. Cancer Res. 2012, 18, 4753–4763. [Google Scholar] [CrossRef] [PubMed]
- Margonis, G.A.; Kim, Y.; Spolverato, G.; Ejaz, A.; Gupta, R.; Cosgrove, D.; Anders, R.; Karagkounis, G.; Choti, M.A.; Pawlik, T.M. Association between specific mutations in KRAS codon 12 and colorectal liver metastasis. JAMA Surg. 2015, 150, 722–729. [Google Scholar] [CrossRef] [PubMed]
- Sanmamed, M.F.; Fernandez-Landazuri, S.; Rodriguez, C.; Zarate, R.; Lozano, M.D.; Zubiri, L.; Perez-Gracia, J.L.; Martin-Algarra, S.; Gonzalez, A. Quantitative cell-free circulating BRAFV600E mutation analysis by use of droplet digital PCR in the follow-up of patients with melanoma being treated with BRAF inhibitors. Clin. Chem. 2015, 61, 297–304. [Google Scholar] [CrossRef] [PubMed]
- Fontanelli, G.; Barate, C.; Ciabatti, E.; Guerrini, F.; Grassi, S.; del Re, M.; Morganti, R.; Petrini, I.; Arici, R.; Barsotti, S.; et al. Real-Time PCR and Droplet Digital PCR: Two techniques for detection of the JAK2(V617F) mutation in Philadelphia-negative chronic myeloproliferative neoplasms. Int. J. Lab. Hematol. 2015, 37, 766–773. [Google Scholar] [CrossRef] [PubMed]
- Taylor, S.C.; Carbonneau, J.; Shelton, D.N.; Boivin, G. Optimization of Droplet Digital PCR from RNA and DNA extracts with direct comparison to RT-qPCR: Clinical implications for quantification of Oseltamivir-resistant subpopulations. J. Virol. Methods 2015, 224, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Drandi, D.; Kubiczkova-Besse, L.; Ferrero, S.; Dani, N.; Passera, R.; Mantoan, B.; Gambella, M.; Monitillo, L.; Saraci, E.; Ghione, P.; et al. Minimal residual disease detection by droplet digital PCR in multiple myeloma, mantle cell lymphoma, and follicular lymphoma: A comparison with real-time PCR. J. Mol. Diagn. 2015, 17, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Mukaide, M.; Sugiyama, M.; Korenaga, M.; Murata, K.; Kanto, T.; Masaki, N.; Mizokami, M. High-throughput and sensitive next-generation droplet digital PCR assay for the quantitation of the hepatitis C virus mutation at core amino acid 70. J. Virol. Methods 2014, 207, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Olsson, E.; Winter, C.; George, A.; Chen, Y.; Howlin, J.; Tang, M.H.; Dahlgren, M.; Schulz, R.; Grabau, D.; van Westen, D.; et al. Serial monitoring of circulating tumor DNA in patients with primary breast cancer for detection of occult metastatic disease. EMBO Mol. Med. 2015, 7, 1034–1047. [Google Scholar] [CrossRef] [PubMed]
- Dudley, J.; Tseng, L.H.; Rooper, L.; Harris, M.; Haley, L.; Chen, G.; Gocke, C.D.; Eshleman, J.R.; Lin, M.T. Challenges posed to pathologists in the detection of KRAS mutations in colorectal cancers. Arch. Pathol. Lab. Med. 2015, 139, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Reinert, T.; Scholer, L.V.; Thomsen, R.; Tobiasen, H.; Vang, S.; Nordentoft, I.; Lamy, P.; Kannerup, A.S.; Mortensen, F.V.; Stribolt, K.; et al. Analysis of circulating tumour DNA to monitor disease burden following colorectal cancer surgery. Gut 2015, 65, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Ogino, S.; Stampfer, M. Lifestyle factors and microsatellite instability in colorectal cancer: The evolving field of molecular pathological epidemiology. J. Natl. Cancer Inst. 2010, 102, 365–367. [Google Scholar] [CrossRef] [PubMed]
- Ogino, S.; Nishihara, R.; VanderWeele, T.J.; Wang, M.; Nishi, A.; Lochhead, P.; Qian, Z.R.; Zhang, X.; Wu, K.; Nan, H.; et al. The role of molecular pathological epidemiology in the study of neoplastic and non-neoplastic diseases in the era of precision medicine. Epidemiology 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Ogino, S.; Chan, A.T.; Fuchs, C.S.; Giovannucci, E. Molecular pathological epidemiology of colorectal neoplasia: An emerging transdisciplinary and interdisciplinary field. Gut 2011, 60, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Guttery, D.S.; Page, K.; Hills, A.; Woodley, L.; Marchese, S.D.; Rghebi, B.; Hastings, R.K.; Luo, J.; Pringle, J.H.; Stebbing, J.; et al. Noninvasive detection of activating estrogen receptor 1 (ESR1) mutations in estrogen receptor-positive metastatic breast cancer. Clin. Chem. 2015, 61, 974–982. [Google Scholar] [CrossRef] [PubMed]
- Tsao, S.C.; Weiss, J.; Hudson, C.; Christophi, C.; Cebon, J.; Behren, A.; Dobrovic, A. Monitoring response to therapy in melanoma by quantifying circulating tumour DNA with droplet digital PCR for BRAF and NRAS mutations. Sci. Rep. 2015, 5, 11198. [Google Scholar] [CrossRef] [PubMed]
| Patient ID | Age (Years) | Sex | TNM | Stage | Survival | KRAS G12V (Copies/mL) | Fractional Abundance (%) |
|---|---|---|---|---|---|---|---|
| 53 | 85 | M | T3N0M0 | IIa | 6 years | 7.50 | 62.79 |
| 113 | 80 | F | T1N0M0 | I | >2 years | 0.00 | 40.42 |
| 118 | 64 | M | T3N2M1 Liver | IV | >2 years | 25.00 | 6.16 |
| 130 | 49 | F | T3N1M1 Liver | IV | 8 months | 197.5 | 20.60 |
| 158 | 84 | M | T4N0M0 | IIb | 1 year | 11.25 | 34.87 |
| 220 | 69 | M | T4N2M1 Bone | IV | 2 months | 55.00 | 38.80 |
| 257 | 85 | M | T3N0MX | IIa | 8 days | 46.25 | 53.20 |
| 258 | 86 | M | T3N0MX | IIa | 20 days | 110.00 | 33.70 |
| 522 | 77 | M | T3N0M0 | IIa | 18 months | 13.75 | 23.76 |
| 532 | 60 | M | T4N1M2 Liver Lung | IV | 10 days | 2412.5 | 77.82 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Olmedillas López, S.; García-Olmo, D.C.; García-Arranz, M.; Guadalajara, H.; Pastor, C.; García-Olmo, D. KRAS G12V Mutation Detection by Droplet Digital PCR in Circulating Cell-Free DNA of Colorectal Cancer Patients. Int. J. Mol. Sci. 2016, 17, 484. https://doi.org/10.3390/ijms17040484
Olmedillas López S, García-Olmo DC, García-Arranz M, Guadalajara H, Pastor C, García-Olmo D. KRAS G12V Mutation Detection by Droplet Digital PCR in Circulating Cell-Free DNA of Colorectal Cancer Patients. International Journal of Molecular Sciences. 2016; 17(4):484. https://doi.org/10.3390/ijms17040484
Chicago/Turabian StyleOlmedillas López, Susana, Dolores C. García-Olmo, Mariano García-Arranz, Héctor Guadalajara, Carlos Pastor, and Damián García-Olmo. 2016. "KRAS G12V Mutation Detection by Droplet Digital PCR in Circulating Cell-Free DNA of Colorectal Cancer Patients" International Journal of Molecular Sciences 17, no. 4: 484. https://doi.org/10.3390/ijms17040484
APA StyleOlmedillas López, S., García-Olmo, D. C., García-Arranz, M., Guadalajara, H., Pastor, C., & García-Olmo, D. (2016). KRAS G12V Mutation Detection by Droplet Digital PCR in Circulating Cell-Free DNA of Colorectal Cancer Patients. International Journal of Molecular Sciences, 17(4), 484. https://doi.org/10.3390/ijms17040484