Infrared Irradiation: Toward Green Chemistry, a Review
Abstract
:1. Introduction
2. Related Aldol Condensation Reactions
3. Nucleophilic Addition to Carbonylic Substrates
4. Macrocycles.
5. Multicomponent Reactions
6. Reduction–Oxidation Reactions
7. Miscellaneous Reactions
8. Novel Results from Our Research Laboratory
9. Natural Products Extractions
10. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| aa2 | Amino acid |
| BN | Boron nitride |
| BzSH | Benzylthiol |
| CQDs | Carbon quantum dots |
| DIPP-aa1 | N-Phosphoamino acid |
| DHPs | Dihydropyridines |
| DHPDs | Dihydropyridinones |
| DHPMs | Dihydropyrimidinones |
| FIR | Far infrared irradiation |
| FRH | Far infrared–treated rice hull |
| FT-IR | Fourier transform infrared spectroscopy |
| GO | Graphene oxide |
| i-PropSH | Isopropanethiol |
| n-BuSH | n-Butanethiol |
| MCR | Multicomponent reaction |
| MH | Mantle heating |
| MIR | Middle infrared irradiation |
| MM | Mechanical milling |
| MW | Microwave irradiation |
| NIR | Near infrared irradiation |
| PhSH | Thiophenol |
| TAFF | Tonsil Actisil FF |
| US | Ultrasound |
References
- Anastas, P.T.; Warner, J.C. Green Chemistry: Theory and Practice; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Clark, J.; Macquarrie, D. Handbook of Green Chemistry and Technology; Blackwell Science: Oxford, UK, 2002. [Google Scholar]
- Victor, G.D. Strategies for cutting carbon. Nature 1998, 395, 837–838. [Google Scholar]
- Doble, M.; Kruthiventi, A.K. Green Chemistry and Engineering; Academic Press: Burlington, VT, USA, 2007. [Google Scholar]
- SShortwave Electric Infrared the Facts. Copyright© ITW BGK Finishing Systems 05/03. Available online: http://www.bgk.com/ir-the-facts (accessed on 12 January 2016).
- Pasquini, C. Near infrared spectroscopy: Fundamentals, practical aspects and analytical applications. J. Braz. Chem. Soc. 2003, 14, 198–219. [Google Scholar]
- Ortega-Jiménez, F.; Domínguez-Villa, F.X.; Rosas-Sánchez, A.; Penieres-Carrillo, G.; López-Cortés, J.G.; Ortega-Alfaro, M.C. An expedient approach to enhance Mizoroki-Heck coupling reaction by infrared irradiation using palladacycle compounds. Appl. Organometal. Chem. 2015, 29, 556–560. [Google Scholar]
- Knoevenagel, E. Method for the synthesis of glutaric acid. Ber 1894, 27, 2345–2346. [Google Scholar] [CrossRef]
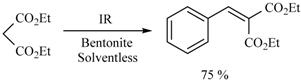
- Delgado, F.; Tamariz, J.; Zepeda, G.; Landa, M.; Miranda, R.; García, J. Knoevenagel condensation catalyzed by a Mexican bentonite using infrared irradiation. Synth. Commun. 1995, 25, 753–759. [Google Scholar] [CrossRef]
- Obrador, E.; Castro, M.; Tamaríz, J.; Zepeda, G.; Miranda, R.; Delgado, F. Knoevenagel condensation in heterogeneous phase catalyzed by IR radiation and tonsil actisil FF. Synth. Commun. 1998, 28, 4649–4663. [Google Scholar] [CrossRef]
- Alcerreca, G.; Sanabria, R.; Miranda, R.; Arroyo, G.; Tamariz, J.; Delgado, F. Preparation of benzylidene barbituric acids promoted by infrared irradiation in absence of solvent. Synth. Commun. 2000, 30, 1295–1301. [Google Scholar] [CrossRef]
- Miranda, R.; Ríos, H.; Delgado, M.; Cogordán, J.A.; Salmón, J. Characterization of TAFF, a bentonitic clay and its application in the obtention of oligotoluene. J. Appl. Catal. 2003, 224, 217–233. [Google Scholar] [CrossRef]
- Penieres, G.; Soto, V.; Alvarez, C.; García, O.; García, J.G. A new strategy for the synthesis of N-substituted 2,5-dimethylpyrroles in heterogeneous medium. Heterocycl. Commun. 1998, 4, 31–32. [Google Scholar] [CrossRef]
- Zhang, C.; Wang, J.; Li, J.H. Infrared heat aided solid state synthesis of pyrroles from 1,4-diketones and ammonium acetate. J. Heterocycl. Chem. 2012, 49, 204–207. [Google Scholar] [CrossRef]
- Penieres, C.G.; Bonifas, A.I.; López, C.J.G.; García, E.J.G.; Alvarez, T.C. Synthesis of benzimidazoles in dry medium. Synth. Commun. 2000, 30, 2191–2195. [Google Scholar] [CrossRef]
- Dandia, A.; Parewa, V.; Gupta, S.L.; Rathore, K.S. Cobalt doped ZnS nanoparticles as a recyclabe catalyst for solvent-free synthesis of heterocyclic privileged medicinal scaffolds under infrared irradiation. J. Mol. Catal. A 2013, 373, 61–71. [Google Scholar] [CrossRef]
- Vázquez, M.A.; Landa, M.; Reyes, L.; Miranda, R.; Tamariz, J.; Delgado, F. Infrared irradiation: Effective promoter in the formation of N-benzylideneanilines in the absence of solvent. Synth. Commun. 2004, 34, 2705–2718. [Google Scholar] [CrossRef]
- Tong, J.Y.; Sun, N.B.; Wu, H.K. Grinding synthesis of Schiff bases combined with infrared irradiation. Asian J. Chem. 2013, 25, 5399–5401. [Google Scholar]
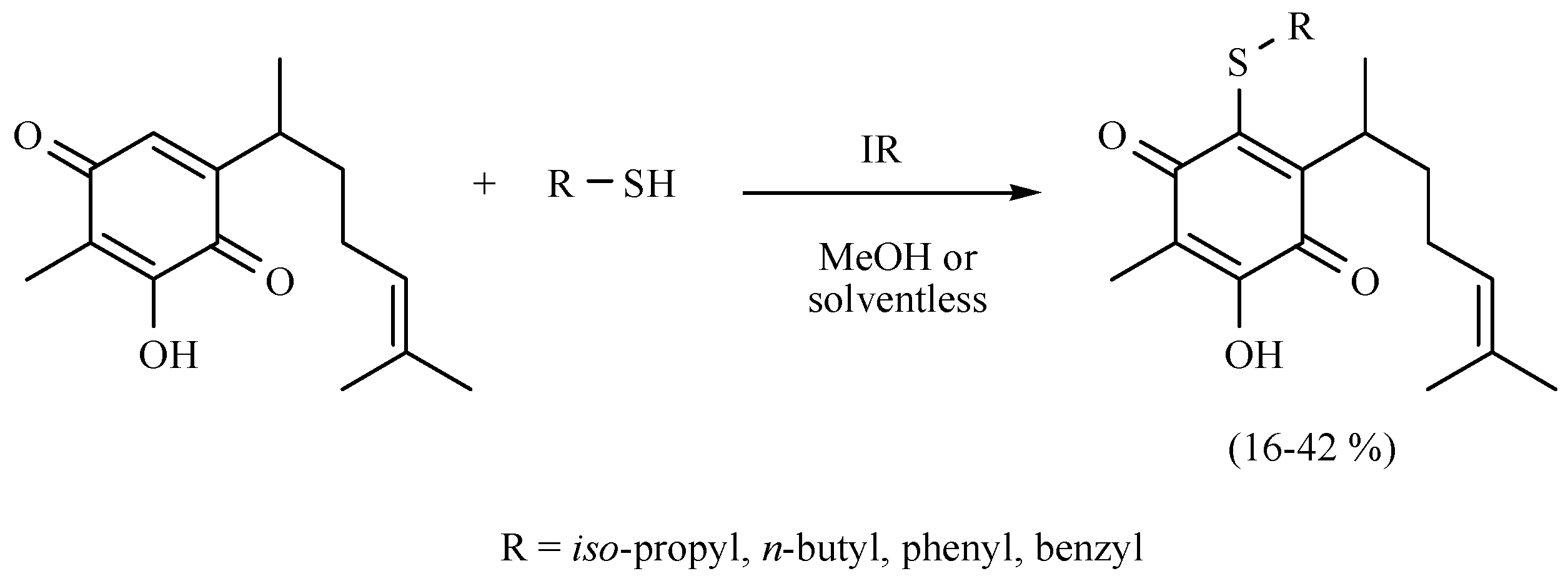
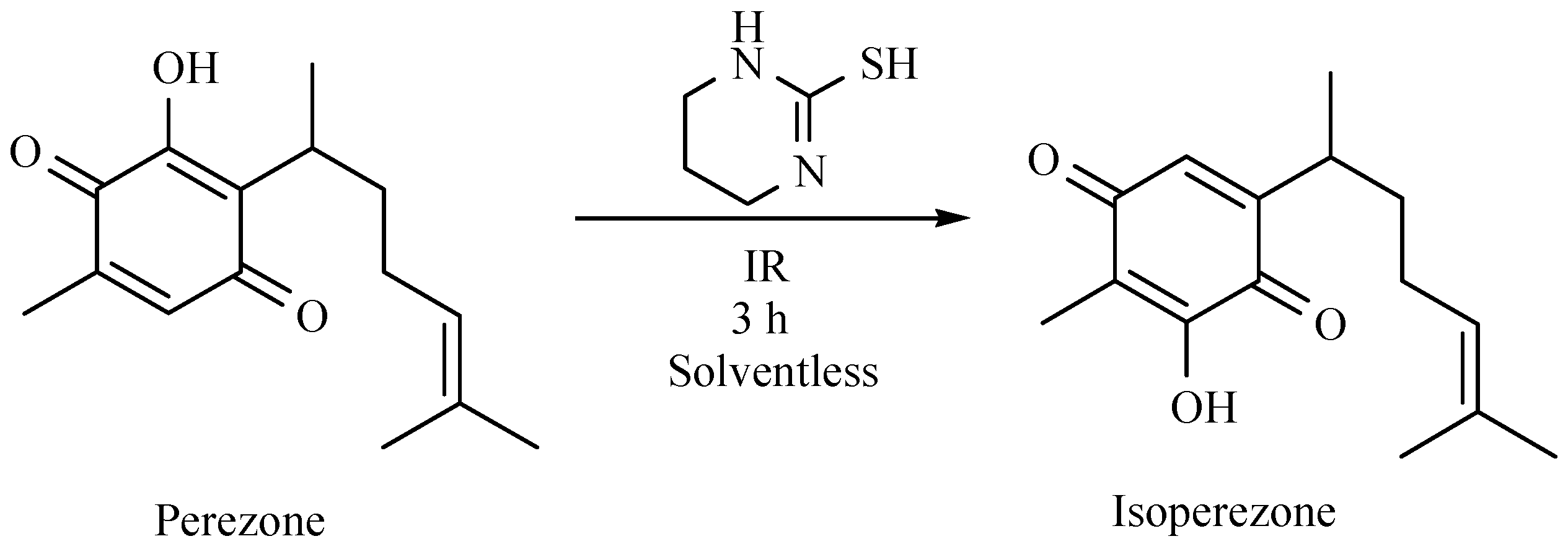
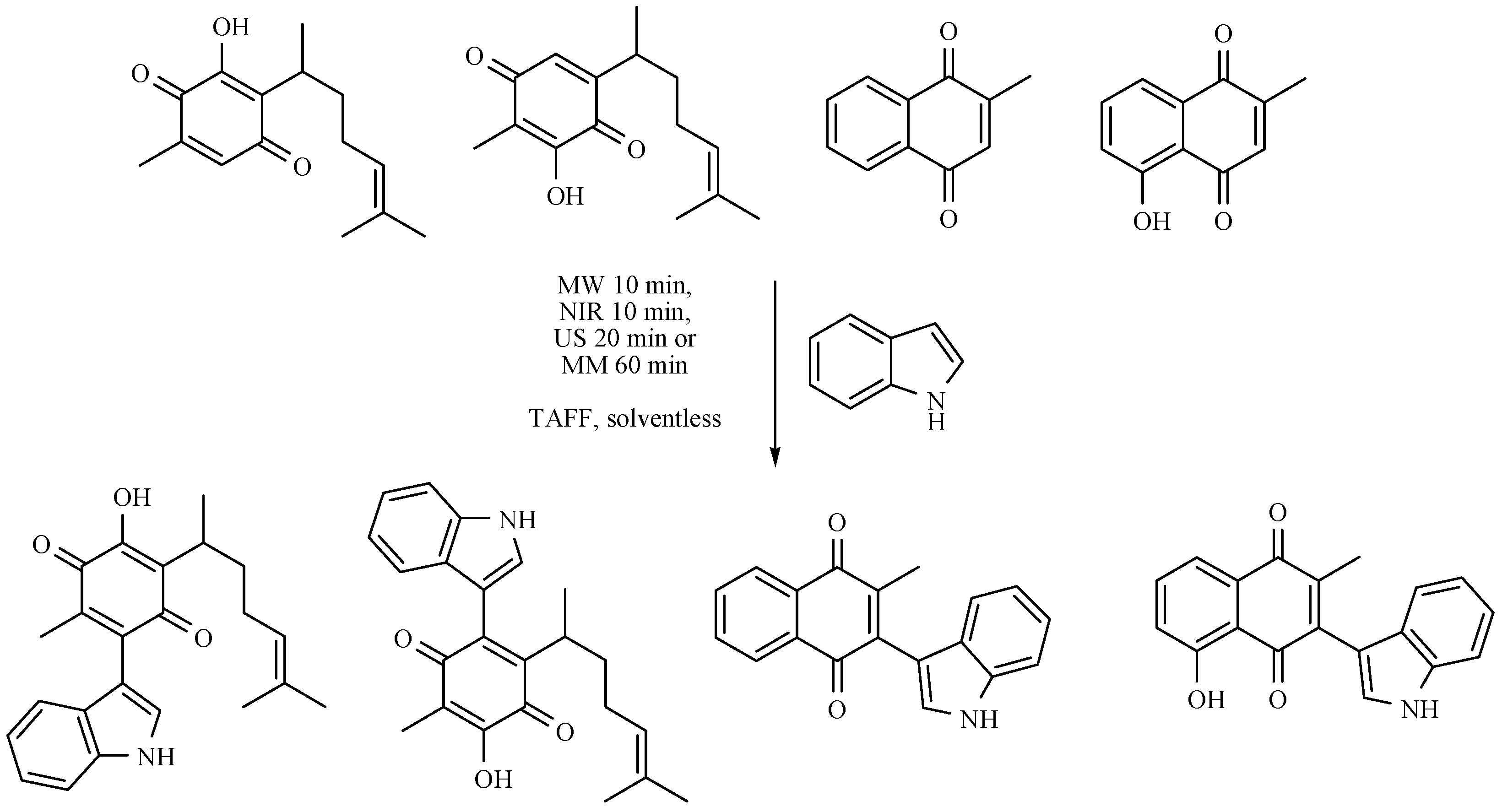
- Martínez, J.; Velasco-Bejarano, B.; Delgado, F.; Pozas, R.; Torres Domínguez, H.M.; Trujillo Ferrara, J.G.; Arroyo, G.A.; Miranda, R. Eco-contribution to the chemistry of perezone, a comparative study, using different modes of activation and solventless conditions. Nat. Prod. Commun. 2008, 3, 1465–1468. [Google Scholar]
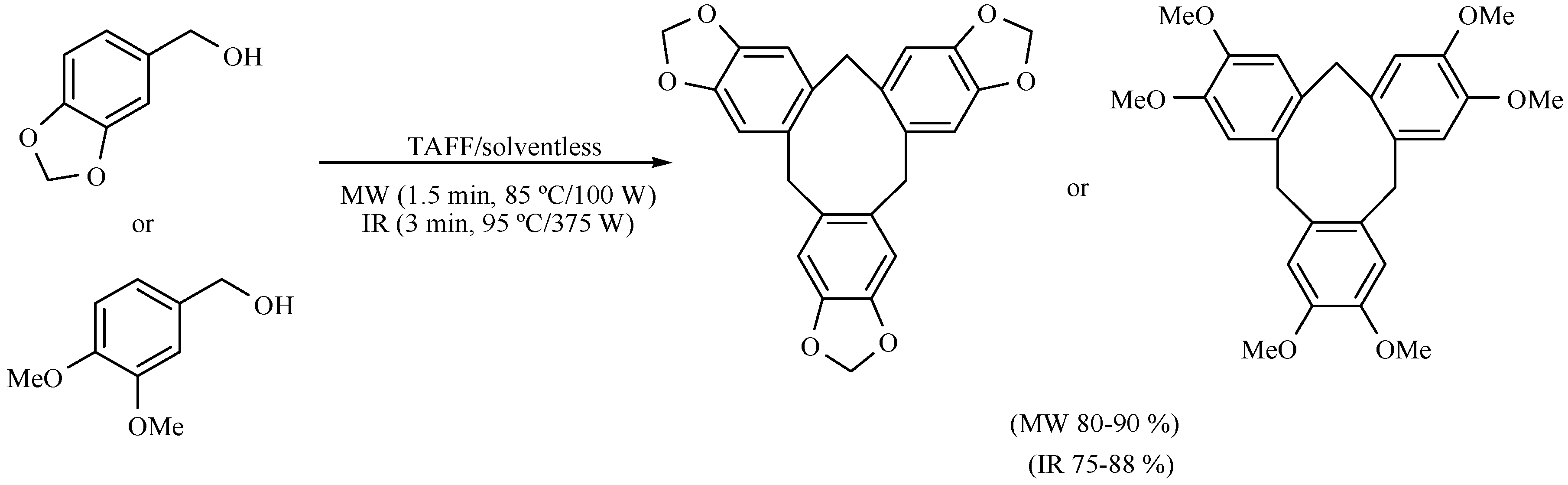
- Miranda, R.; Valencia-Vázquez, O.; Maya-Vega, C.A.; Nicolás-Vázquez, I.; Vargas-Rodriguez, Y.M.; Morales-Serna, J.A.; García-Ríos, E.; Salmón, M. Synthesis of cycloveratrylene macrocycles and benzyl oligomers catalysed by bentonitic under microwave/infrared and solvent-free conditions. Molecules 2013, 18, 12820–12844. [Google Scholar] [CrossRef] [PubMed]
- Zhu, J.; Bienaymé, H. Multicomponent Reactions; Wiley-VCH: Weinheim, Germany, 2005. [Google Scholar]
- Zarco, M.; Martínez, J.O.; Noguez, O.; Nicolás-Vazquez, M.I.; Ramírez-Apan, T.; Pérez, J.; Miranda, R.; Arroyo-Razo, G.A. A green approach to the production of hybrid diindolylmethane-phenylboronic acids via a 3MCR: Promising antineoplasic molecules. J. Chem. 2013, 2013, 1–9. [Google Scholar] [CrossRef]
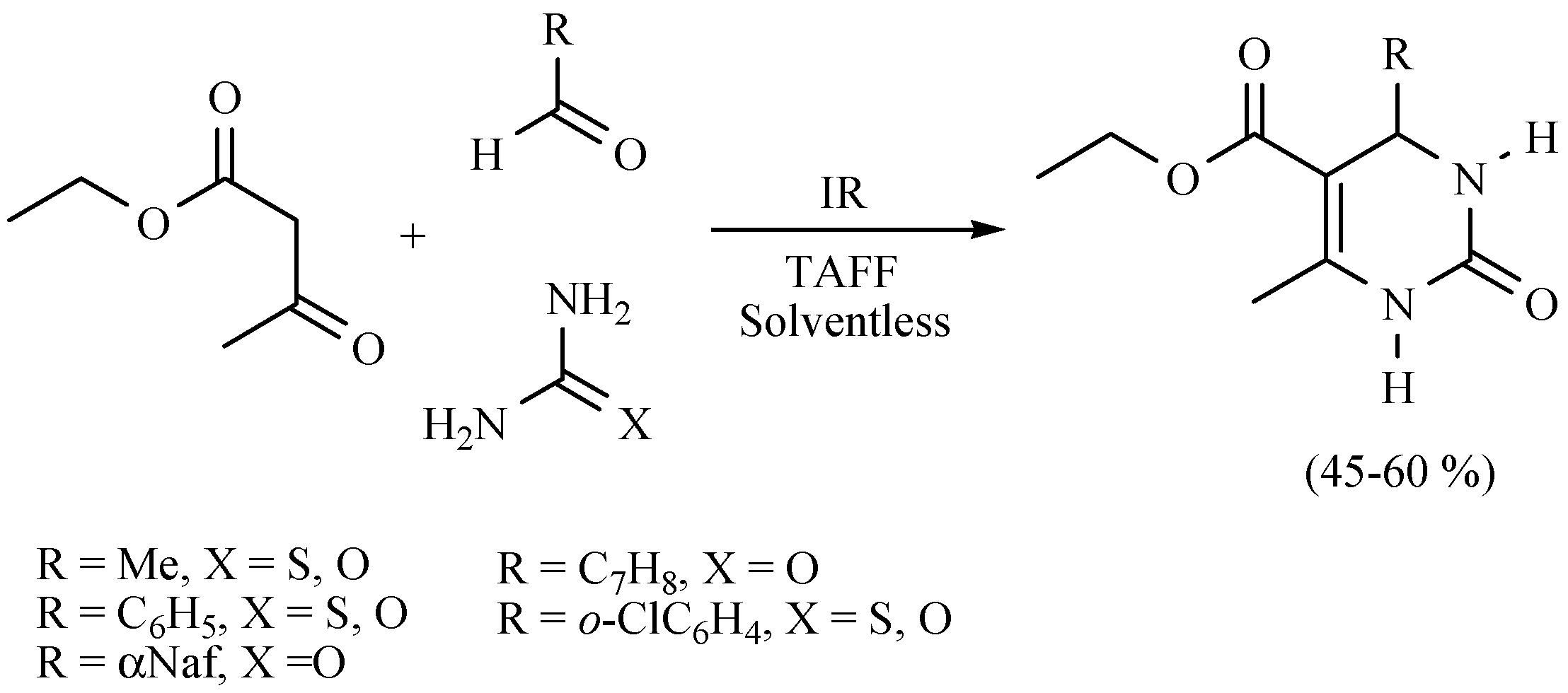
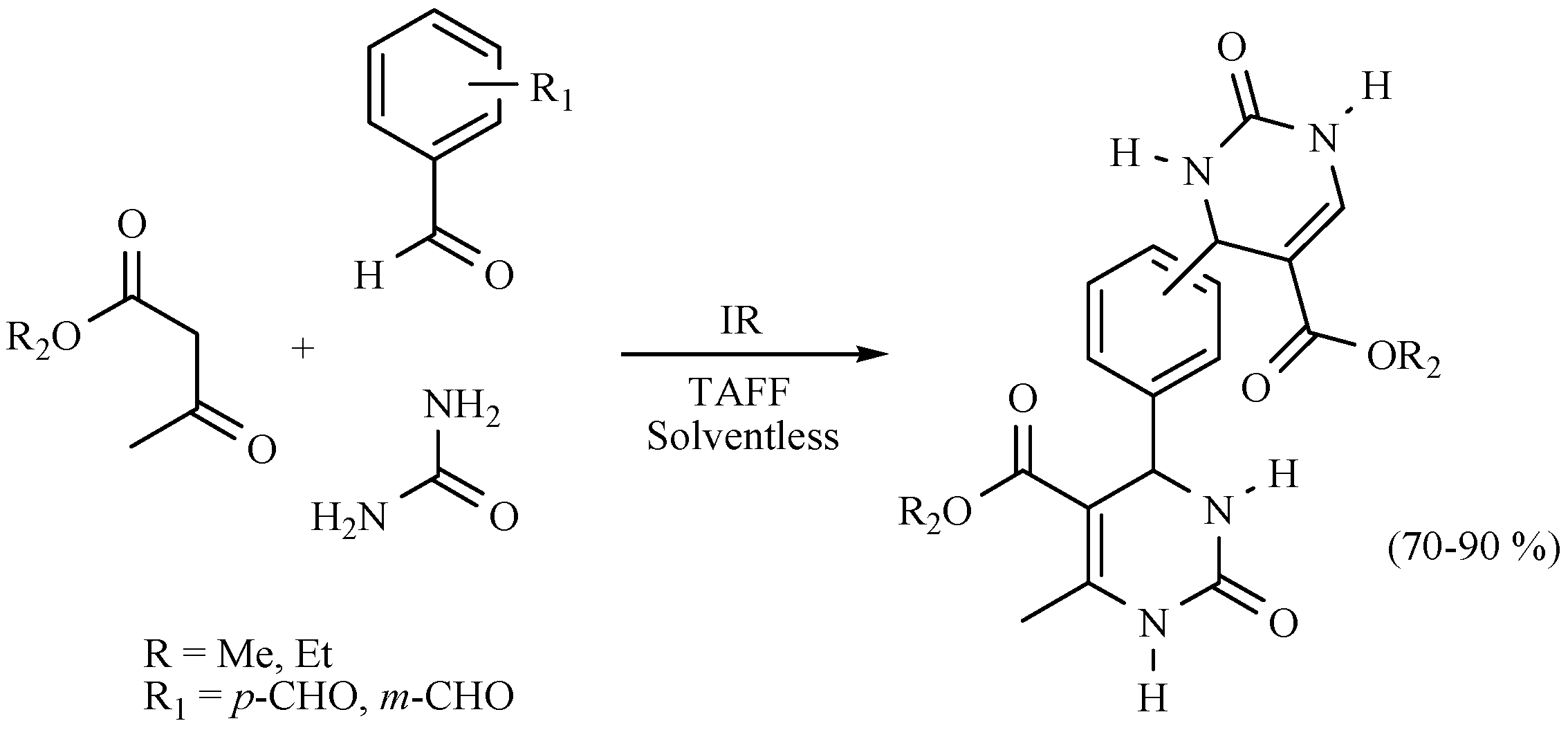
- Salmón, M.; Osnaya, R.; Gómez, L.; Arroyo, G.; Delgado, F.; Miranda, R. Contribution to the Biginelli reaction, using a bentonitic clay as catalyst and solventless procedure. J. Mex. Chem. Soc. 2001, 45, 206–207. [Google Scholar]
- Noguez, M.O.; García, A.; Ibarra, C.; Cabrera, A.; Aceves, J.M.; Nicolas, M.I.; Miranda, R. Green synthesis of bis-Biginelli esters, with vasodilatory effects, their mass spectrometric and physical studies. Trends Org. Chem. 2009, 13, 75–82. [Google Scholar]
- Osnaya, R.; Arroyo, G.A.; Parada, L.; Delgado, F.; Trujillo, J.; Salmón, M.; Miranda, R. Biginelli vs Hantzsch esters study under infrared radiation and solventless conditions. Arkivoc 2003, 2003, 112–117. [Google Scholar]
- Gómez-Pliego, R.; Osnaya, R.; Zamora, I.; Velasco-Bejarano, B.; Arroyo, G.; Ramírez-San Juan, E.; Trujillo, J.; Delgado, F.; Miranda, R. The Hantzsch ester production in a water-based biphasic medium, using infrared irradiation as the activating source. J. Mex. Chem. Soc. 2007, 51, 181–184. [Google Scholar]
- Gómez Pliego, R.; Ramírez-San Juan, E.; Miranda, R.; Villalobos-Molina, R.; Delgado, F.; Osnaya, R.; Trujillo Ferrara, J. Vasodilator effects of bis-dihydropyridines structurally related to nifedipine. Med. Chem. 2006, 2, 527–534. [Google Scholar] [CrossRef]
- Ramírez-San Juan, E.; Soriano-Ursúa, M.A.; Espinosa-Raya, J.; Correa-Basurto, J.; Trujillo-Ferrara, J.G.; Miranda Ruvalcaba, R.; Gómez-Pliego, R. Anticonvulsant effects of bis-1,4-dihydropyridines and the probable role of L-type calcium channels suggested by docking simulations. Med. Chem. Res. 2014, 23, 5149–5159. [Google Scholar] [CrossRef]
- Martínez, J.; Romero-Vega, S.; Abeja-Cruz, R.; Álvarez-Toledano, C.; Miranda, R. Green approach multicomponent production of boron containing Hantzsch and Biginelli esters. Int. J. Mol. Sci. 2013, 14, 2903–2915. [Google Scholar] [CrossRef] [PubMed]
- Chandrachood, P.; Gadkari, T.; Deshpande, N.; Kashalkar, R. Highly efficient methodology for aromatization of 1,4-dihydropyridines using silica-supported transition metal nitrates under infra-red radiations. J. Iran Chem. Soc. 2012, 9, 47–51. [Google Scholar] [CrossRef]
- Noguez, M.O.; Marcelino, V.; Rodríguez, H.; Martín, O.; Martínez, J.O.; Arroyo, G.A.; Pérez, F.J.; Suárez, M.; Miranda, R. Infrared assisted production of 3,4-dihydro-2(1H)-pyridones in solvent-free conditions. Int. J. Mol. Sci. 2011, 12, 2641–2649. [Google Scholar] [CrossRef] [PubMed]
- Penieres-Carrillo, G.; García-Estrada, J.G.; Gutiérrez-Ramírez, J.L.; Alvarez-Toledano, C. Infrared-assisted eco-friendly selective synthesis of diindolylmethanes. Green Chem. 2003, 5, 337–339. [Google Scholar] [CrossRef]
- Velasco-Bejarano, B.; Sánchez-Torres, L.E.; García-Estrada, J.G.; Miranda-Ruvalcaba, R.; Álvarez-Toledano, C.; Penieres-Carrillo, G. Diindolylmethane derivatives as apoptosis inductors in L5178Y cells. J. Mex. Chem. Soc. 2008, 52, 224–228. [Google Scholar]
- Wang, S.X.; Guo, S.B.; Gao, M.Z.; Li, J.T.; Duan, Y.F. Infrared irradiation synthesis of substituted 5-oxo-1,2,3,4,5,6,7,8-octahydroquinoline derivatives under solvent-free conditions. J. Chem. 2006, 3, 159–163. [Google Scholar]
- Sánchez, A.; Hernández, F.; Cruz, P.C.; Alcaraz, Y.; Tamariz, J.; Delgado, F.; Vázquez, M.A. Infrared irradiation-assisted multicomponent synthesis of 2-amino-3-cyano-4H-pyran derivatives. J. Mex. Chem. Soc. 2012, 56, 121–127. [Google Scholar]
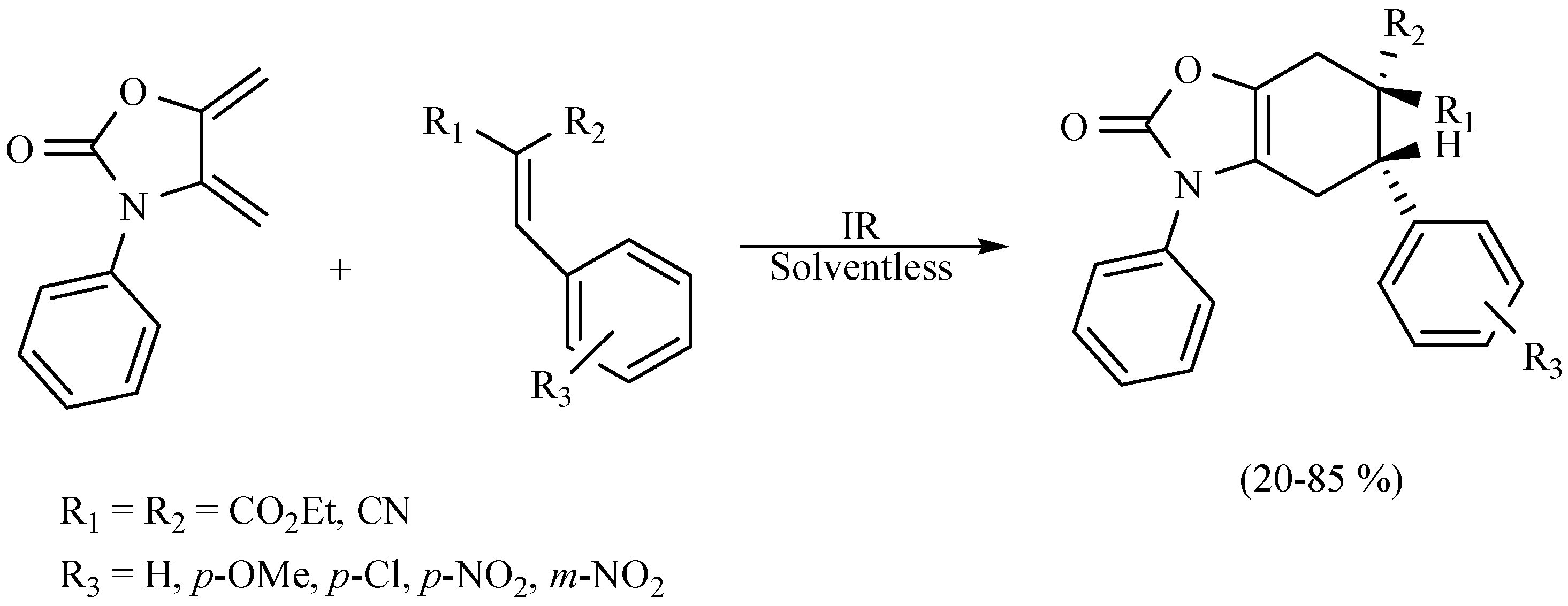
- Flores-Conde, M.I.; Reyes, L.; Herrera, R.; Rios, H.; Vazquez, M.A.; Miranda, R.; Tamariz, J.; Delgado, F. Highly regio- and stereoselective Diels–Alder cycloadditions via two-step and multicomponent reactions promoted by infrared irradiation under solvent-free conditions. Int. J. Mol. Sci. 2012, 13, 2590–2617. [Google Scholar] [CrossRef] [PubMed]
- Valdez-Rojas, J.E.; Ríos-Guerra, H.; Ramírez-Sánchez, A.L.; García-González, G.; Álvarez-Toledano, C.; López-Cortés, J.G.; Toscano, R.A.; Penieres-Carrillo, J.G. A study of the Willgerodt–Kindler reaction to obtain thioamides and α-ketothioamides under solvent-less conditions. Can. J. Chem. 2012, 90, 567–573. [Google Scholar] [CrossRef]
- Velázquez, A.M.; Torres, L.A.; Díaz, G.; Ramírez, A.; Hernández, R.; Santillán, H.; Martínez, L.; Martínez, I.; Díaz-Barriga, S.; Abrego, V.; et al. A novel one pot, solvent-free Mannich synthesis of methylpiperidinyl phenols, methylphenylmorpholinyl phenols and methylthiophenylmorpholinyl phenols using infrared light irradiation. Arkivoc 2006, 2006, 150–161. [Google Scholar]
- Velázquez, A.M.; Torres, L.A.; González, R.; Valencia, A.; Díaz-Barriga, S.; Menconi, I.; Martínez, L.; Ramírez, A.; Martínez, I.; Camacho, B.; et al. Synthesis of 4-methoxy-2-thiomorpholin-4-ylmethyl-1-phenol. Molbank 2007, 2007, M547. [Google Scholar] [CrossRef]
- Clouthier, C.M.; Pelletier, J.N. Expanding the organic toolbox: A guide to integrating biocatalysis in synthesis. Chem. Soc. Rev. 2012, 41, 1585–1605. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, F.; Zhu, J.; Yu, J.C. NaYF4:Yb,Tm/CdS composite as a novel near-infrared-driven photocatalyst. App. Catal. B 2010, 100, 433–439. [Google Scholar] [CrossRef]
- Guo, H.; Peng, M.; Zhu, Z.; Sun, L. Preparation of reduced graphene oxide by infrared irradiation induced photothermal reduction. Nanoscale 2013, 5, 9040–9048. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Lara, J.; Gutiérrez-Pérez, R.; Penieres-Carrillo, G.; López-Cortés, J.G.; Escudero-Salas, A.; Álvarez-Toledano, C. Reaction of hydroquinones with supported oxidizing reagents in solvent-free conditions. Synth. Commun. 2000, 30, 2713–2720. [Google Scholar] [CrossRef]
- Li, H.; Liu, R.; Lian, S.; Liu, Y.; Huang, H.; Kang, Z. Near-infrared light controlled photocatalytic activity of carbon quantum dots for highly selective oxidation reaction. Nanoscale 2013, 5, 3289–3297. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Huang, B.; Ma, X.; Wang, Z.; Qin, X.; Zhang, X.; Dai, Y.; Whangbo, M.H. Cu2(OH)PO4, a near-infrared-activated photocatalyst. Angew. Chem. Int. Ed. 2013, 52, 4810–4813. [Google Scholar] [CrossRef] [PubMed]
- Ikeue, T.; Sonoda, M.; Kurahashi, S.; Tachibana, H.; Teraoka, D.; Sugimori, T.; Kasuga, K.; Handa, M. Annulated dinuclear palladium(II) phthalocyanine complex as an effective photo-oxidation catalyst for near-infrared region light. Inorg. Chem. Commun. 2010, 13, 1170–1172. [Google Scholar] [CrossRef]
- Penieres, G.; Miranda, R.; García, J.; Aceves, J.; Delgado, F. Modification of the Fischer indole synthesis. Heterocycl. Commun. 1996, 2, 401–402. [Google Scholar]
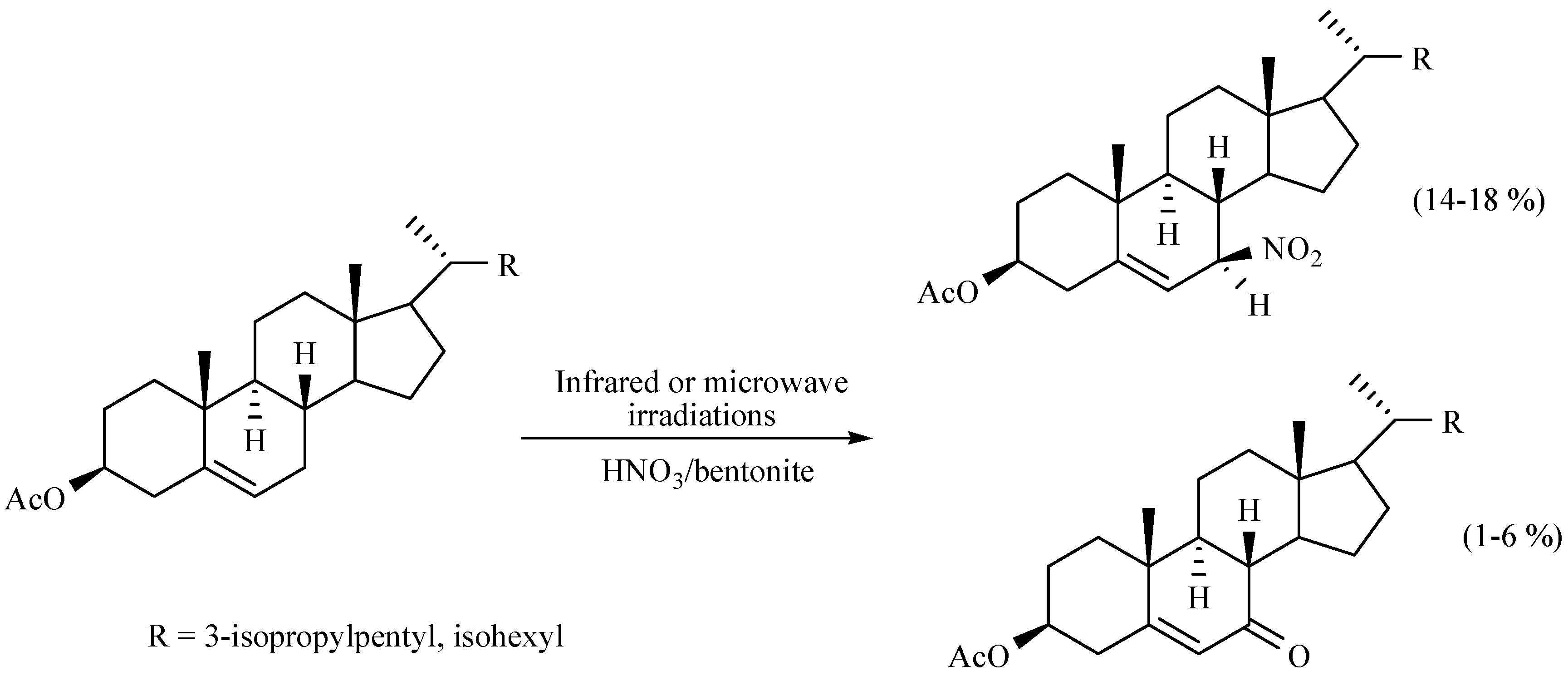
- Jiménez-Estrada, M.; García, M.O.; Navarro, A.O.; Eusebio, J.L.; Álvarez, C.T.; Penieres, G.C.; Gutiérrez, R.P. Allylic nitration of 3-sitosterol and cholesterol acetate: Preparation of 7-nitro derivatives. Steroids 1997, 62, 500–503. [Google Scholar] [CrossRef]
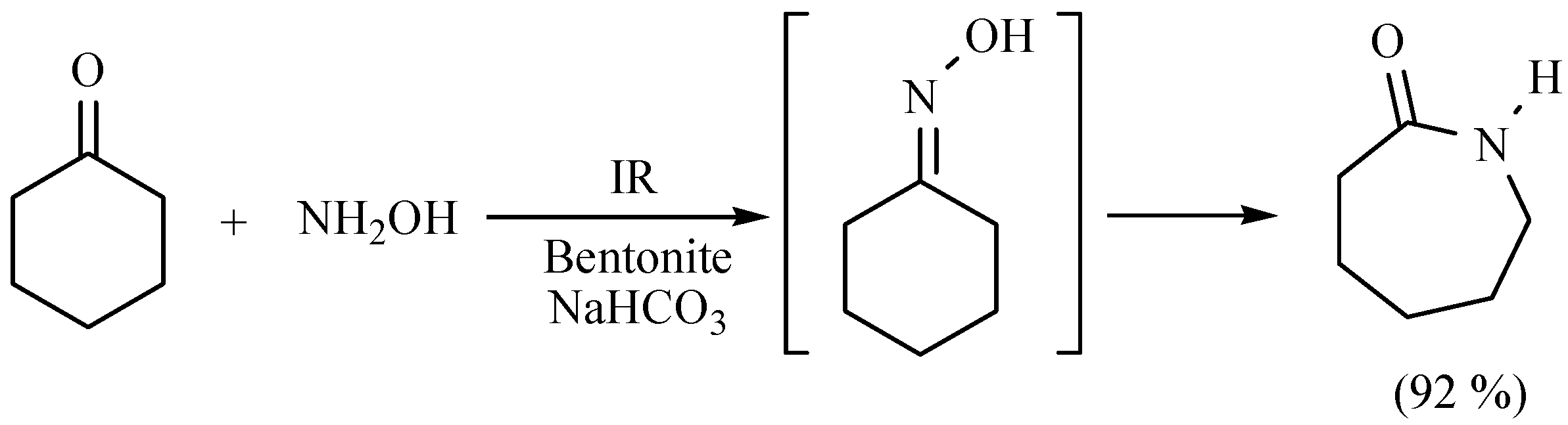
- Penieres, G.; Aceves, J.M.; Flores, A.; Mendoza, G.; García, O.; Álvarez, C. Comparative study of ε-caprolactam synthesis using different energy sources and natural clay as catalyst. Heterocycl. Commun. 1997, 3, 507–508. [Google Scholar] [CrossRef]
- Deng, C.H.; Li, Y.M.; Zhao, Y.F. Formation of oligopeptides from N-phosphoamino acid by infrared radiation. Phosphorus Sulfur Silicon 2000, 163, 203–210. [Google Scholar] [CrossRef]
- Hernández, M.T.; González, M. Synthesis of resins as α-alumina precursors by the Pechini method using microwave and infrared heating. J. Eur. Ceram. Soc. 2002, 22, 2861–2868. [Google Scholar] [CrossRef]
- Huang, L.; Wei, H.B.; Ke, F.S.; Cai, J.S.; Fan, X.Y.; Sun, S.G. Infrared irradiation-assisted one-step synthesis of nanosized tin dioxide particles and particle size effect on lithium storage performance. Colloids Surf. A 2007, 308, 87–92. [Google Scholar] [CrossRef]
- Termoss, H.; Toury, B.; Pavan, S.; Brioude, A.; Bernard, S.; Cornu, D.; Valette, S.; Benayoun, S.; Miele, P. Preparation of boron nitride-based coatings on metallic substrates via infrared irradiation of dip-coated polyborazylene. J. Mater. Chem. 2009, 19, 2671–2674. [Google Scholar] [CrossRef]
- Qin, W.; Zhang, D.; Zhao, D.; Wang, L.; Zheng, K. Near-infrared photocatalysis based on YF3:Yb3+, Tm3+/TiO2 core/shell nanoparticles. Chem. Commun. 2010, 46, 2304–2306. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Jiménez, F.; Penieres-Carrillo, J.G.; Lagunas-Rivera, S.; López-Cortés, J.G.; Álvarez-Toledano, C.; Ortega-Alfaro, M.C. Infrared irradiation assisted both the synthesis of (Z)-(aminomethyl)(aryl)phenylhydrazones via the Mannich coupling reaction and their application to the palladium-catalyzed Heck reaction. RSC Adv. 2015, 5, 80911–80918. [Google Scholar] [CrossRef]
- Noguez-Córdova, M.O.; Flores-Ramírez, C.I.; Velasco-Bejarano, B.; Arroyo-Razo, G.A.; Pérez-Flores, F.J.; Carranza-Tellez, V.; Miranda-Ruvalcaba, R. Comparative study using different infrared zones of the solventless activation of organic reaction. Int. J. Mol. Sci. 2011, 12, 8575–8580. [Google Scholar] [CrossRef] [PubMed]
- Miranda, R.; Noguez, O.; Velasco, B.; Arroyo, G.; Penieres, G.; Martínez, J.O.; Delgado, F. Irradiación infrarroja: Una alternativa para la activación de reacciones y su contribución a la química verde. Educ. Quim. 2009, 20, 421–425. [Google Scholar]
- Niu, G.G.; Xie, Y.C.; Lou, J.F.; Liu, H.Z. Isolation and purification of glycyrrhizic acid with solvent extraction. Sep. Sci. Technol. 2005, 44, 189–192. [Google Scholar] [CrossRef]
- Hartonen, K.; Parshintsev, J.; Sandberg, K.; Bergelin, E.; Nisula, L.; Riekkola, M.L. Isolation of flavonoids from aspen knotwood by pressurized hot water extraction and comparison with other extraction techniques. Talanta 2007, 74, 32–38. [Google Scholar] [CrossRef] [PubMed]
- De Rijke, E.; Zafra-Gómez, A.; Ariese, F.; Th Brinkman, U.A.; Gooijer, C. Determination of isoflavone glucoside malonates in Trifolium pretense L. (red clover) extracts: Quantification and stability studies. J. Chromatogr. A 2001, 932, 55–64. [Google Scholar] [CrossRef]
- Zhou, H.Y.; Liu, C.Z. Microwave-assisted extraction of solanesol from tobacco leaves. J. Chromatogr. A 2006, 1129, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Deng, C.; Zhang, H.; Wu, Y.; Zhang, X. Microwave-assisted steam distillation for the determination of organochlorine pesticides and pyrethroids in Chinese teas. Talanta 2007, 71, 1068–1074. [Google Scholar] [CrossRef] [PubMed]
- Hu, F.; Deng, C.; Liu, Y.; Zhang, X. Quantitative determination of chlorogenic acid in Honeysuckle using microwave-assisted extraction followed by nano-LC-ESI mass spectrometry. Talanta 2009, 77, 1299–1303. [Google Scholar] [CrossRef] [PubMed]
- Pourmortazavi, S.M.; Hajimirsadeghi, S.S. Supercritical fluid extraction in plant essential and volatile oil analysis. J. Chromatogr. A 2007, 1163, 2–24. [Google Scholar] [CrossRef] [PubMed]
- Peng, J.; Fan, G.; Wu, Y. Supercritical fluid extraction of aurentiamide acetate from Patrinia villosa Juss and subsequent isolation by silica gel and high-speed counter-current chromatography. J. Chromatogr. A 2005, 1083, 52–57. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Kim, J.H.; Jeong, S.M.; Kim, D.R.; Ha, J.U.; Nam, K.C.; Ahn, D.U. Effect of far-infrared radiation on the antioxidant activity of rice hulls. J. Agric. Food. Chem. 2003, 51, 4400–4403. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Kim, J.H.; Nam, K.C.; Ahn, D.U. Antioxidant properties of far infrared-treated rice hull extract in irradiated raw and cooked turkey breast. J. Food Sci. 2003, 68, 1904–1909. [Google Scholar] [CrossRef]
- Lee, S.C.; Jeong, S.M.; Kim, S.Y.; Nam, K.C.; Ahn, D.U. Effect of far-infrared irradiation on the antioxidant activity of defatted sesame meal extracts. J. Agric. Food Chem. 2005, 53, 1495–1498. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.C.; Jeong, S.M.; Kim, S.Y.; Park, H.R.; Nam, K.C.; Ahn, D.U. Effect of far-infrared radiation and heat treatment on the antioxidant activity of water extract from peanut hulls. Food Chem. 2006, 94, 489–493. [Google Scholar] [CrossRef]
- Lee, S.C.; Kim, S.Y.; Jeong, S.M.; Park, J.H. Effect of far-infrared irradiation on catechins and nitrite scavenging activity of green tea. J. Agric. Food Chem. 2006, 54, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Jeong, S.M.; Jo, S.C.; Lee, S.C. Application of far-infrared irradiation in the manufacturing process of green tea. J. Agric. Food Chem. 2006, 54, 9943–9947. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Lee, J.M.; Cho, Y.J.; Kim, C.T.; Kim, C.J.; Nam, K.C.; Lee, S.C. Effect of far-infrared heater on the physicochemical characteristics of green tea during processing. J. Food Biochem. 2009, 33, 149–162. [Google Scholar] [CrossRef]
- Kim, J.W.; Bae, H.C.; Kim, M.C.; Lee, S.C. Effect of far-infrared irradiation on the antioxidant activity of Puerariae radix extract. J. Food Biochem. 2008, 32, 85–95. [Google Scholar] [CrossRef]
- Wanyo, P.; Siriamornpun, S.; Meeso, N. Changes in phenolic compounds, antioxidant and physical properties of mulberry tea influenced by intensity of far-infrared radiation. Am. Eurasian J. Agric. Environ. Sci. 2009, 6, 470–479. [Google Scholar]
- Chen, Y.; Duan, G.; Xie, M.; Chen, B.; Li, Y. Infrared-assisted extraction coupled with high-performance liquid chromatography for simultaneous determination of eight active compounds in Radix Salviae miltiorrhizae. J. Sep. Sci. 2010, 33, 2888–2897. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Chen, Y.; Chen, G. Far infrared-assisted extraction followed by capillary electrophoresis for the determination of bioactive constituents in the leaves of Lycium barbarum Linn. J. Chromatogr. A 2010, 1217, 4511–4516. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.M.; Lee, S.C. The effects of far-infrared irradiation on the antioxidant activity of Licorice (Glycyrrhiza uralensis Fisch) root. J. Food Biochem. 2010, 34, 172–181. [Google Scholar] [CrossRef]
- Fu, Y.; Zhang, L.; Chen, G. Determination of carbohydrates in Folium Lysium Chinensis using capillary electrophoresis combined with far-infrared light irradiation-assisted extraction. J. Sep. Sci. 2011, 34, 3272–3278. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Yu, Y.; Duan, G.; Li, Y. Study on infrared-assisted extraction coupled with high performance liquid chromatography (HPLC) for determination of catechin, epicatechin, and procyanidin B2 in grape seeds. Food Chem. 2011, 127, 1872–1877. [Google Scholar] [CrossRef]
- Fu, Y.; Zhang, L.; Chen, G. Far infrared-assisted extraction followed by MEKC for the simultaneous determination of flavones and phenolic acids in the leaves of Rhododendron mucronulatum Turcz. J. Sep. Sci. 2012, 35, 468–475. [Google Scholar] [CrossRef] [PubMed]
- Siriamornpun, S.; Kaisoon, O.; Meeso, N. Changes in colour, antioxidant activities and carotenoids (lycopene, β-carotene, lutein) of marigold flower (Tagetes erecta L.) resulting from different drying processes. J. Funct. Food 2012, 4, 757–766. [Google Scholar] [CrossRef]
- Li, F.; Yu, Y.; Zhang, H.; Liu, T.; Li, Y.; Duan, G. Infrared-assisted non-ionic surfactant extraction as a green analytical preparatory technique for the rapid extraction and pre-concentration of picroside I and picroside II from Picrorhiza scrophulariiflora Pennell. Anal. Methods 2013, 5, 3747–3753. [Google Scholar] [CrossRef]
- Jin, C.W.; Ghimeray, A.K.; Wang, L.; Xu, M.L.; Piao, J.P.; Cho, D.H. Far infrared assisted kenaf leaf tea preparation and its effect on phenolic compounds, antioxidant and ACE inhibitory activity. J. Med. Plant Res. 2013, 7, 1121–1128. [Google Scholar]
- Ghimeray, A.K.; Sharma, P.; Hu, W.; Jin, C.W.; Park, C.H.; Rho, H.S.; Cho, D.H. Far infrared assisted conversion of isoflavones and its effect on total phenolics and antioxidant activity in black soybean seed. J. Med. Plant Res. 2013, 7, 1129–1137. [Google Scholar]
- Ghimeray, A.K.; Sharma, P.; Phoutaxay, P.; Salitxay, T.; Woo, S.H.; Park, S.U.; Park, C.H. Far infrared irradiation alters total polyphenol, total flavonoid, antioxidant property and quercetin production in tartary buckwheat sprout powder. J. Cereal Sci. 2014, 59, 167–172. [Google Scholar] [CrossRef]
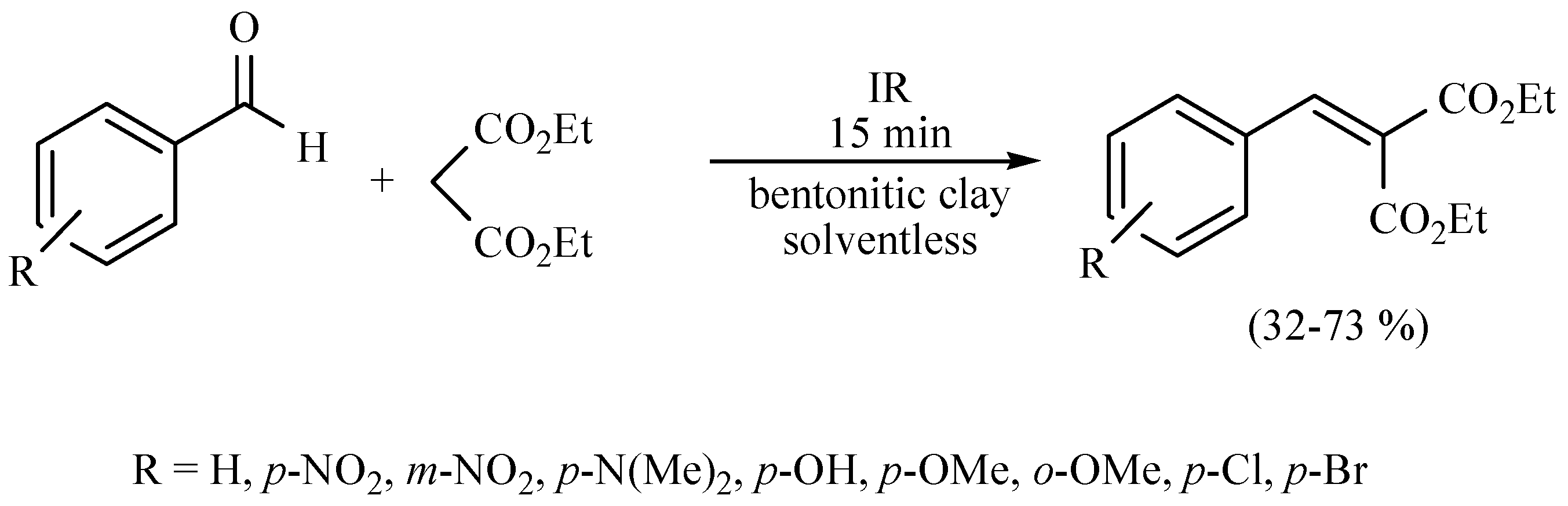


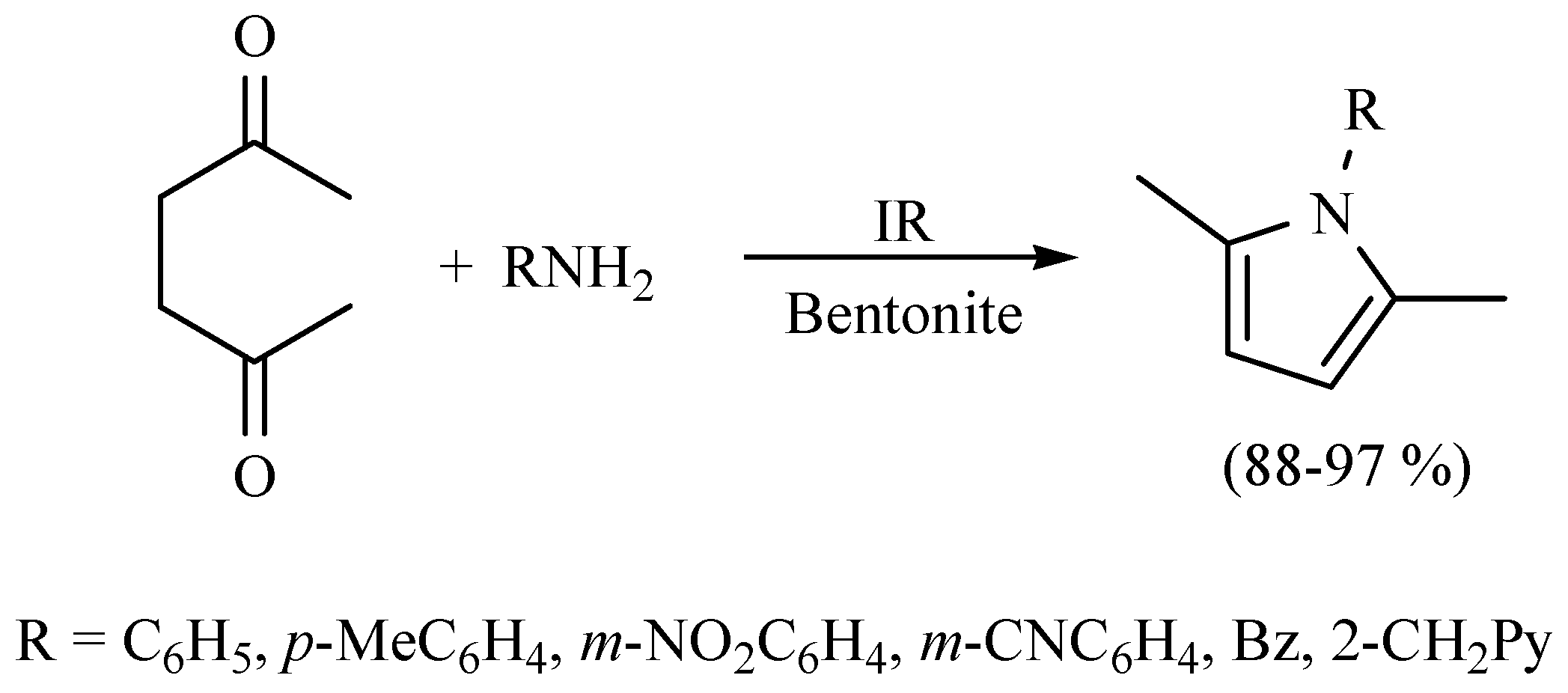
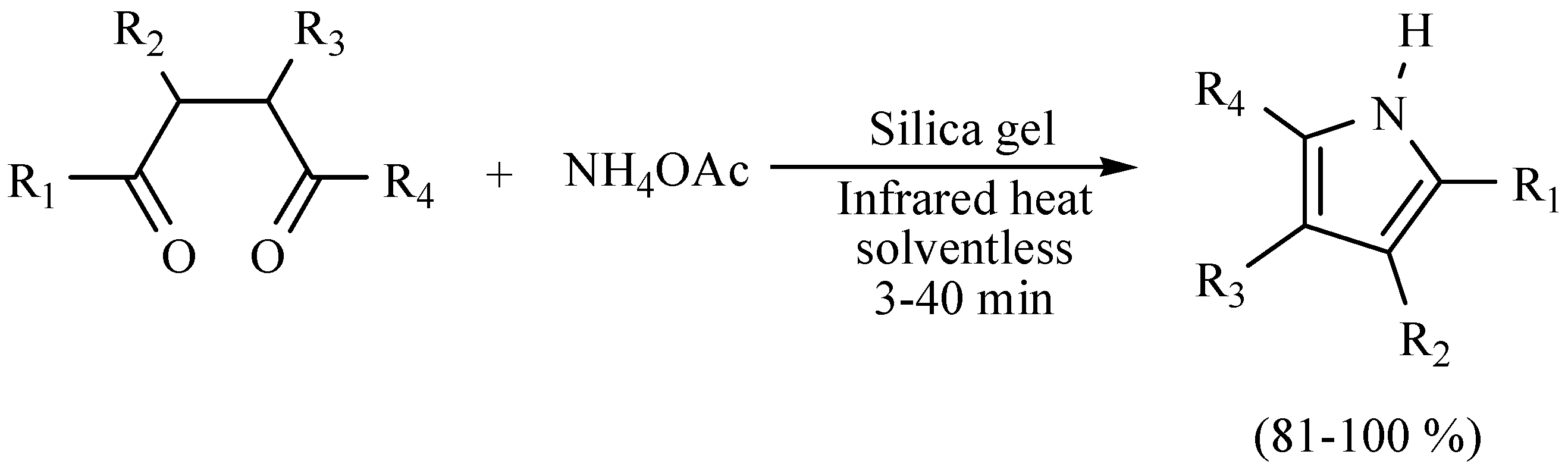
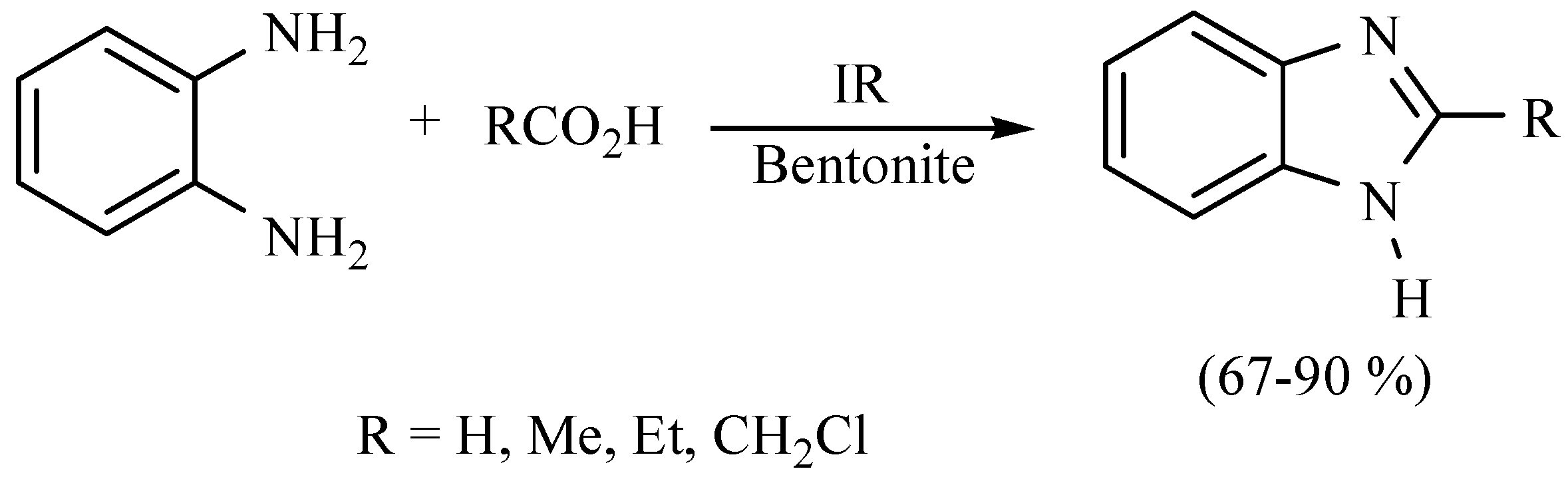
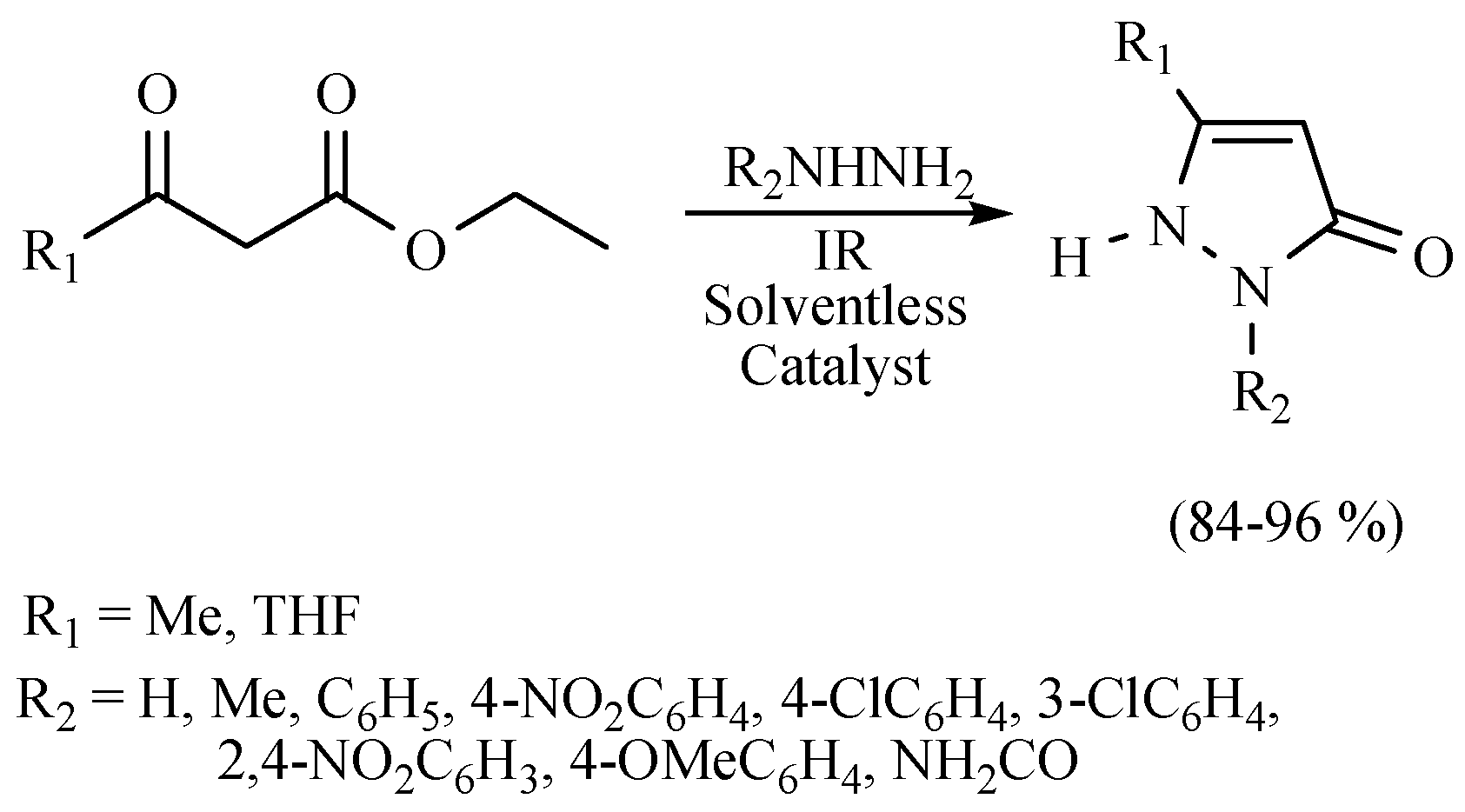
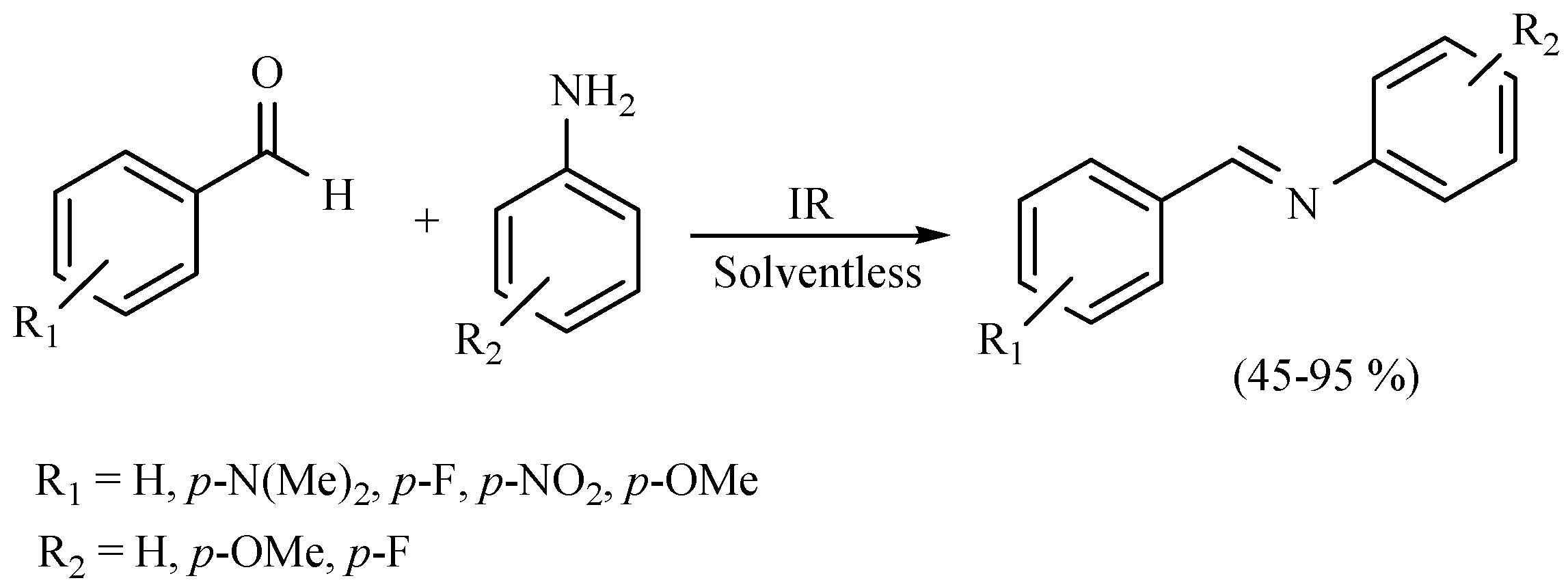
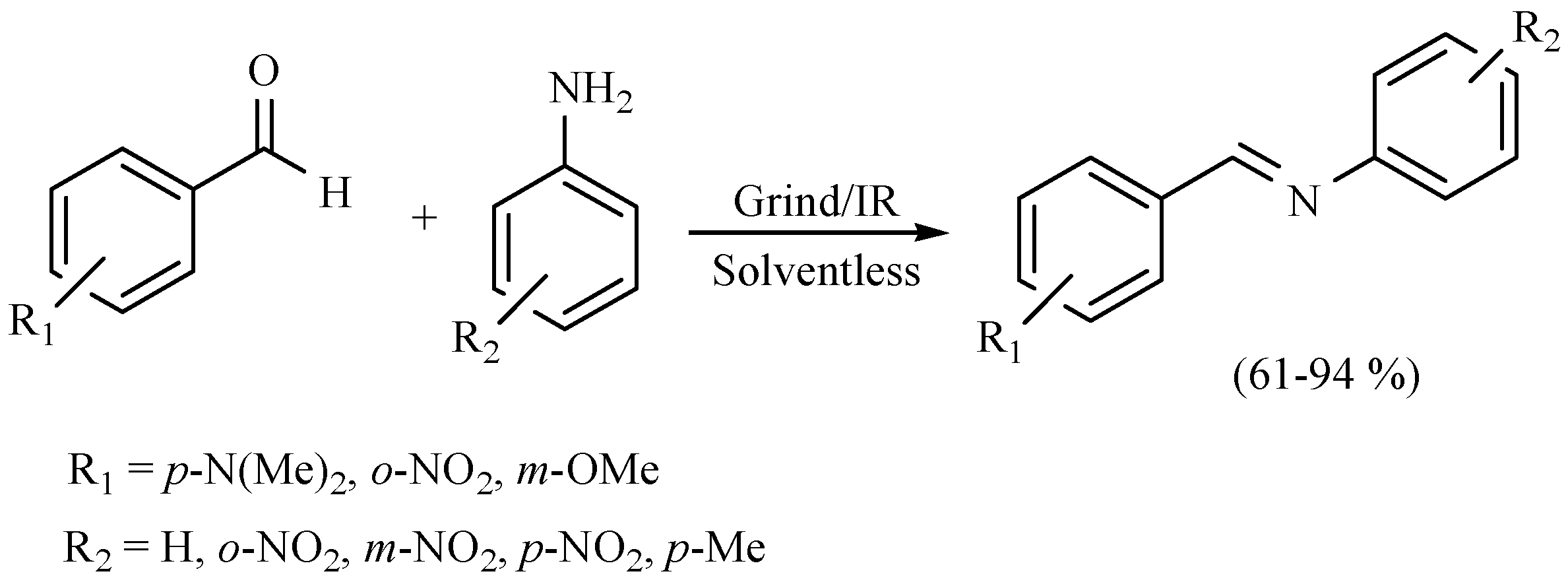
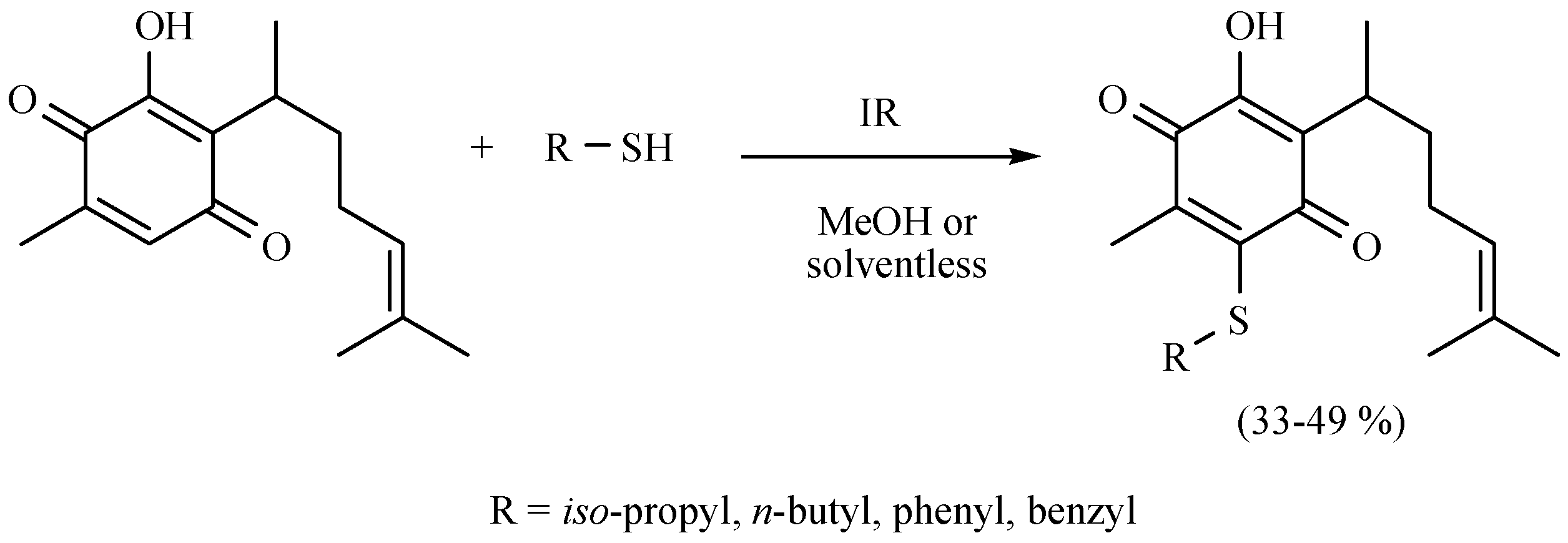

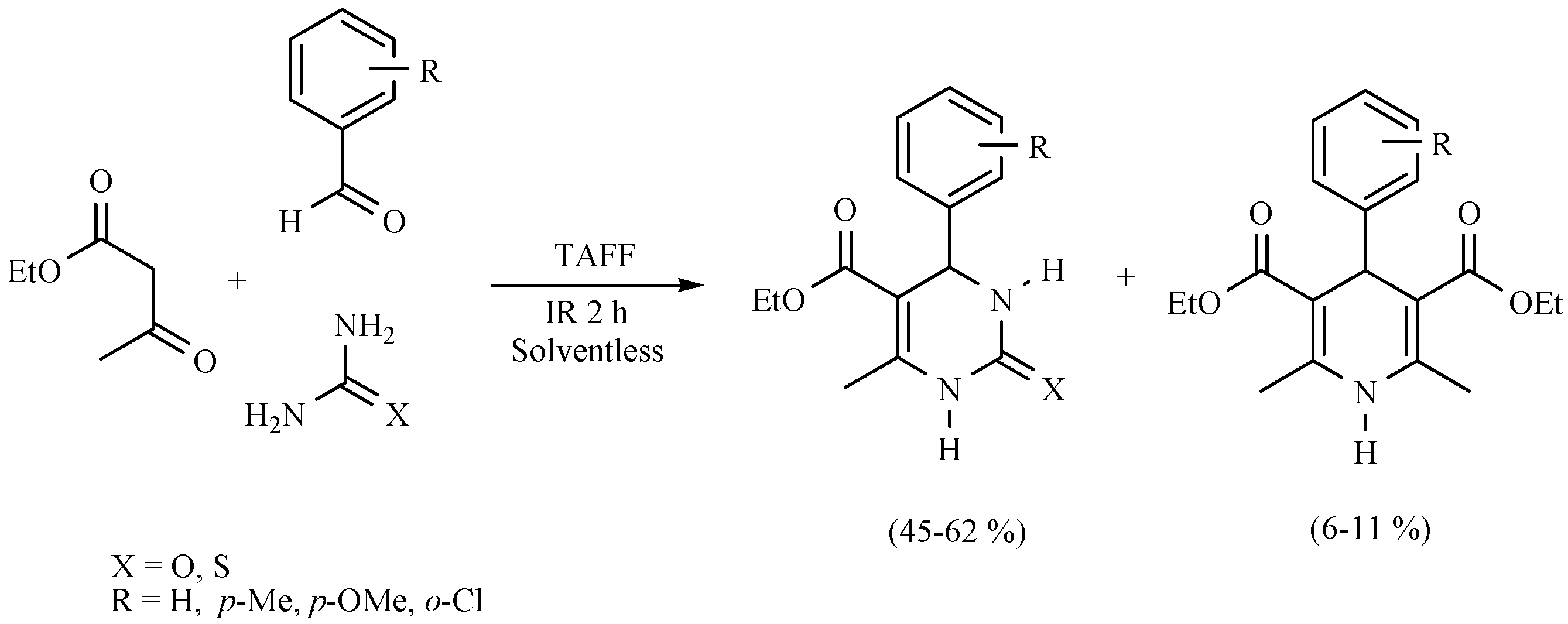
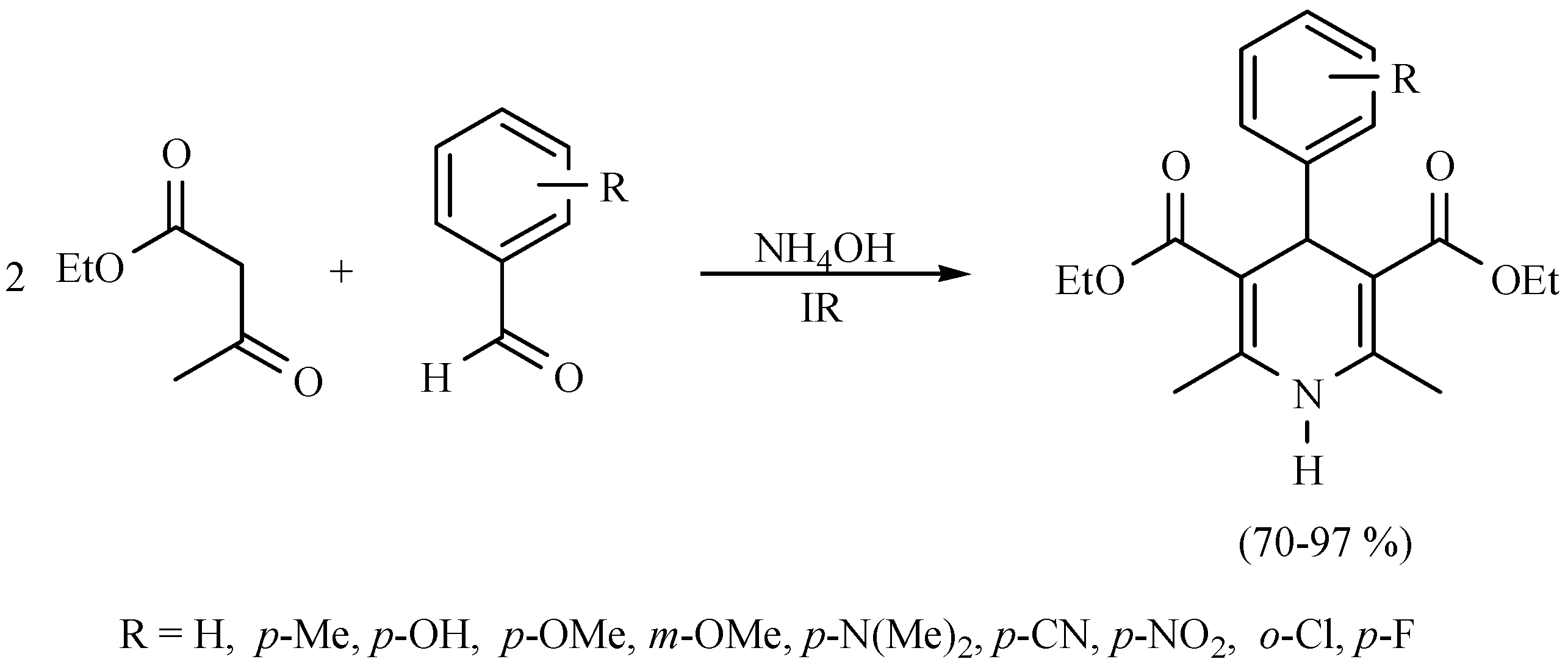
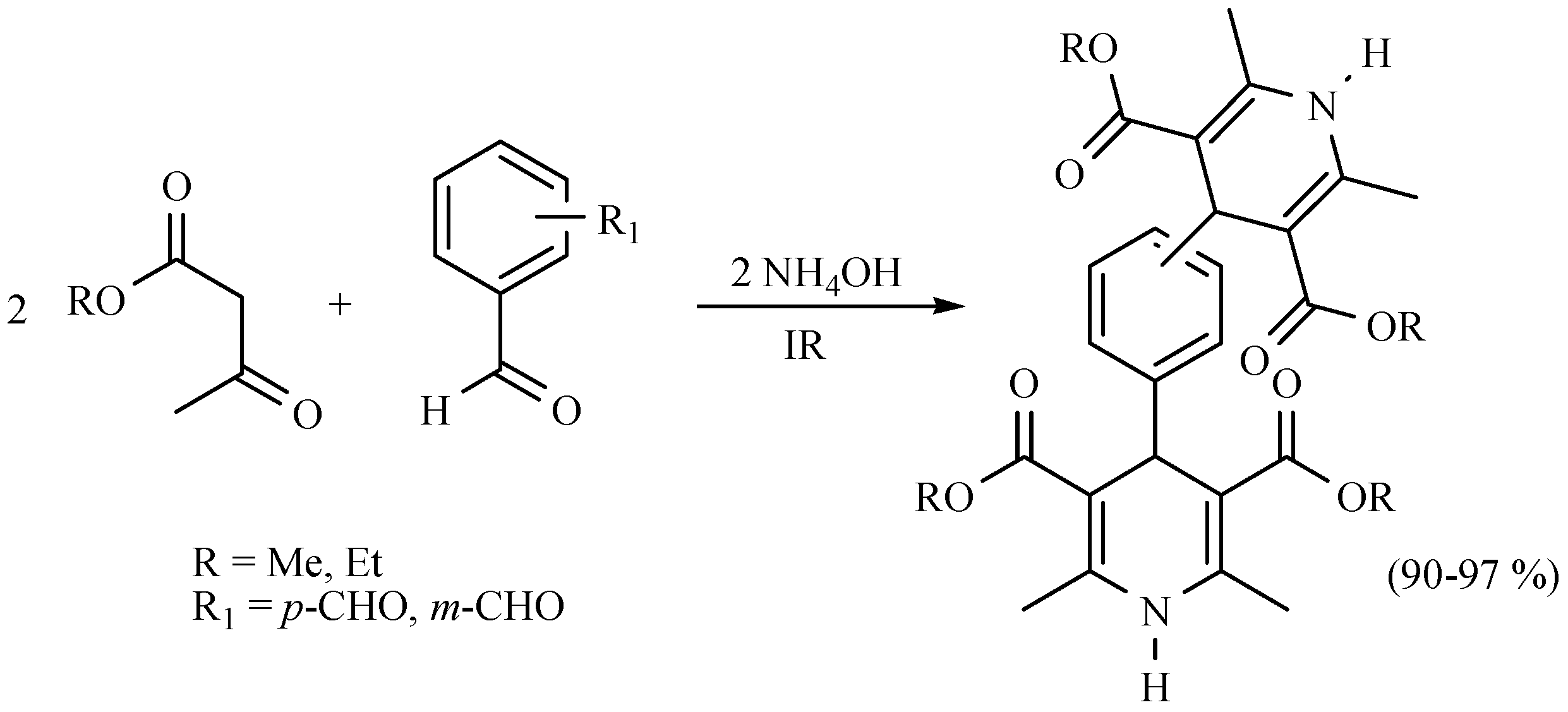
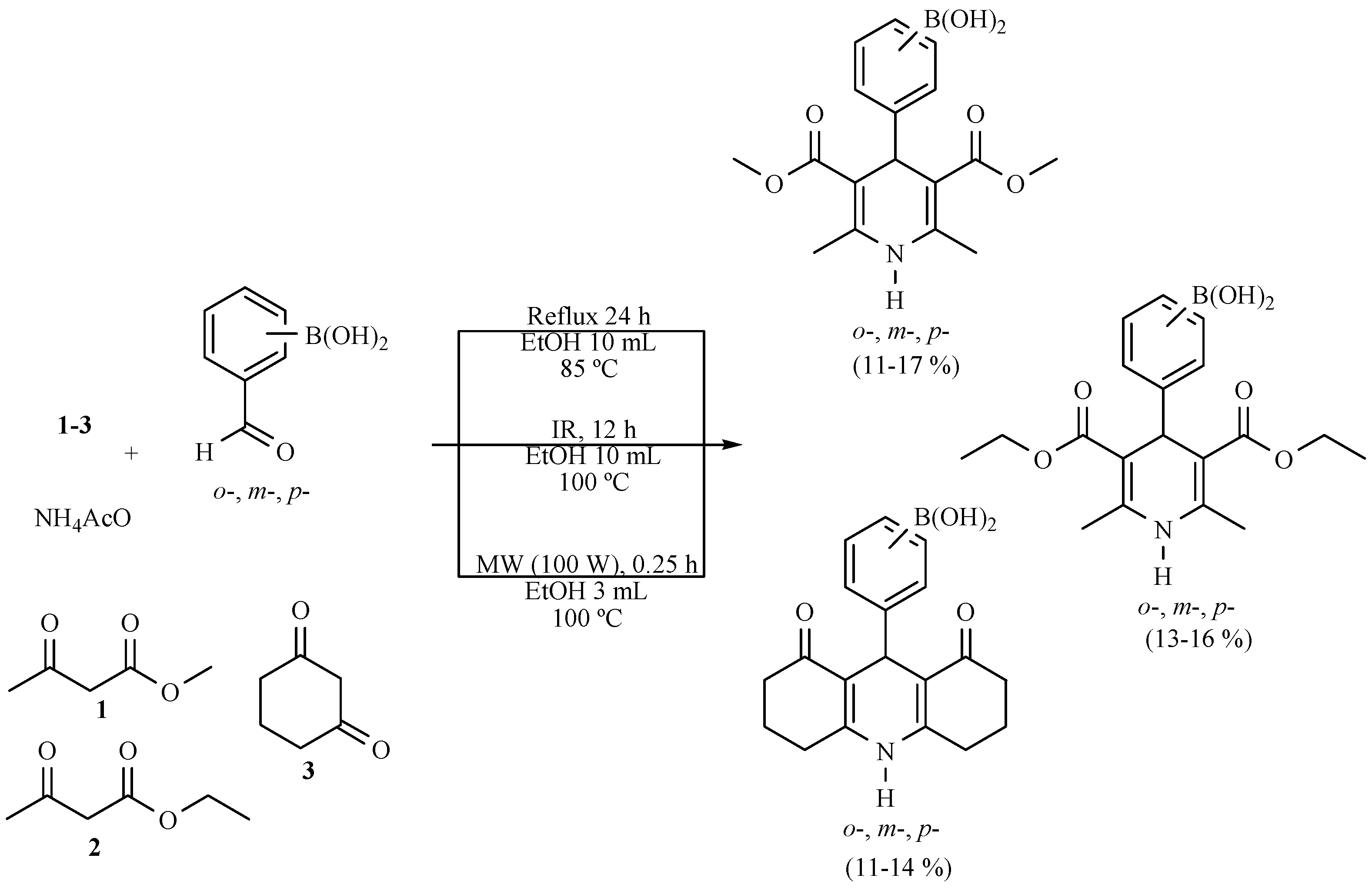
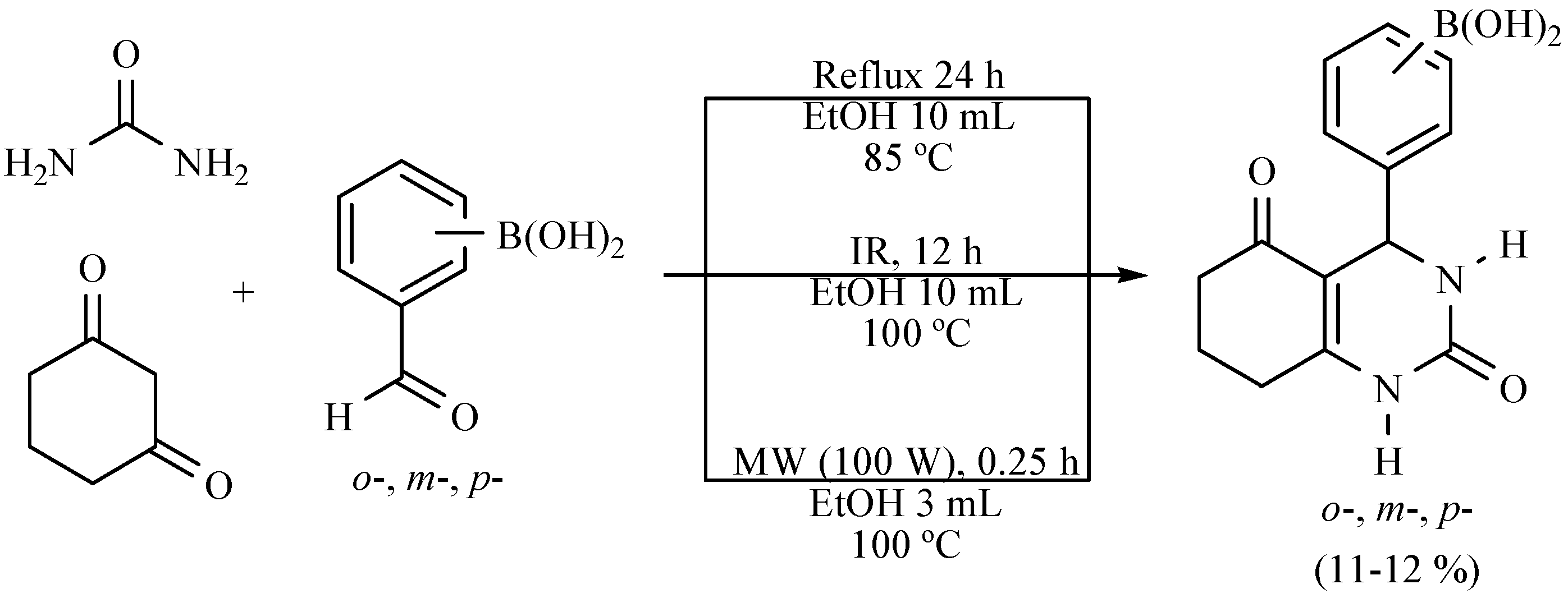
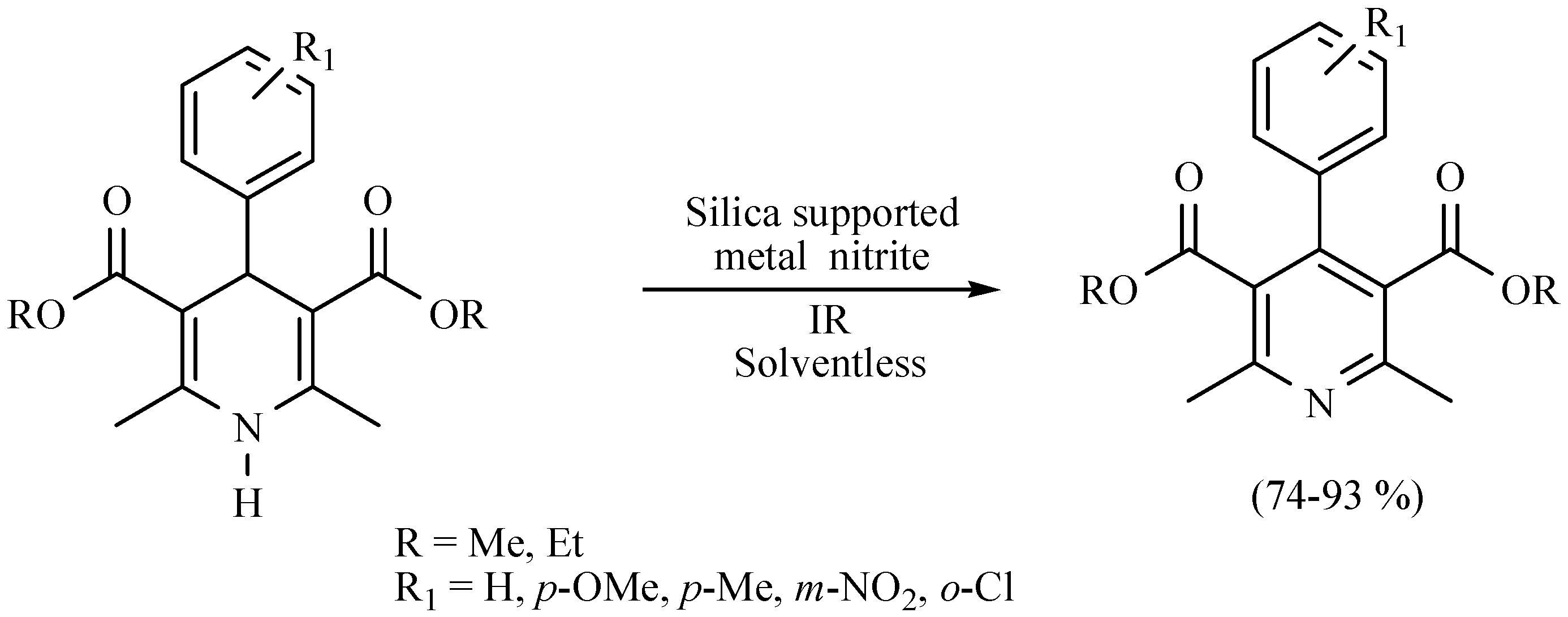
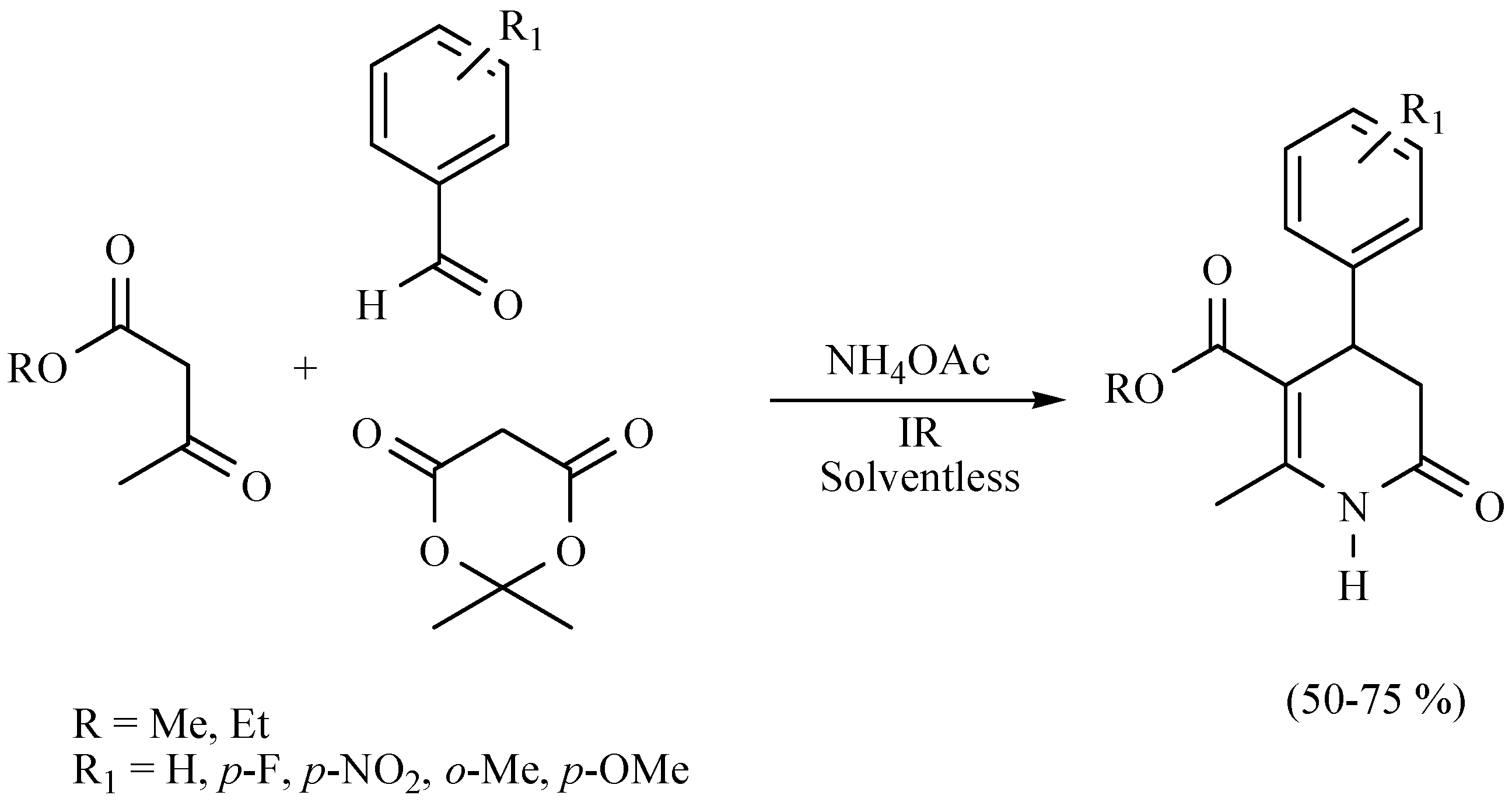
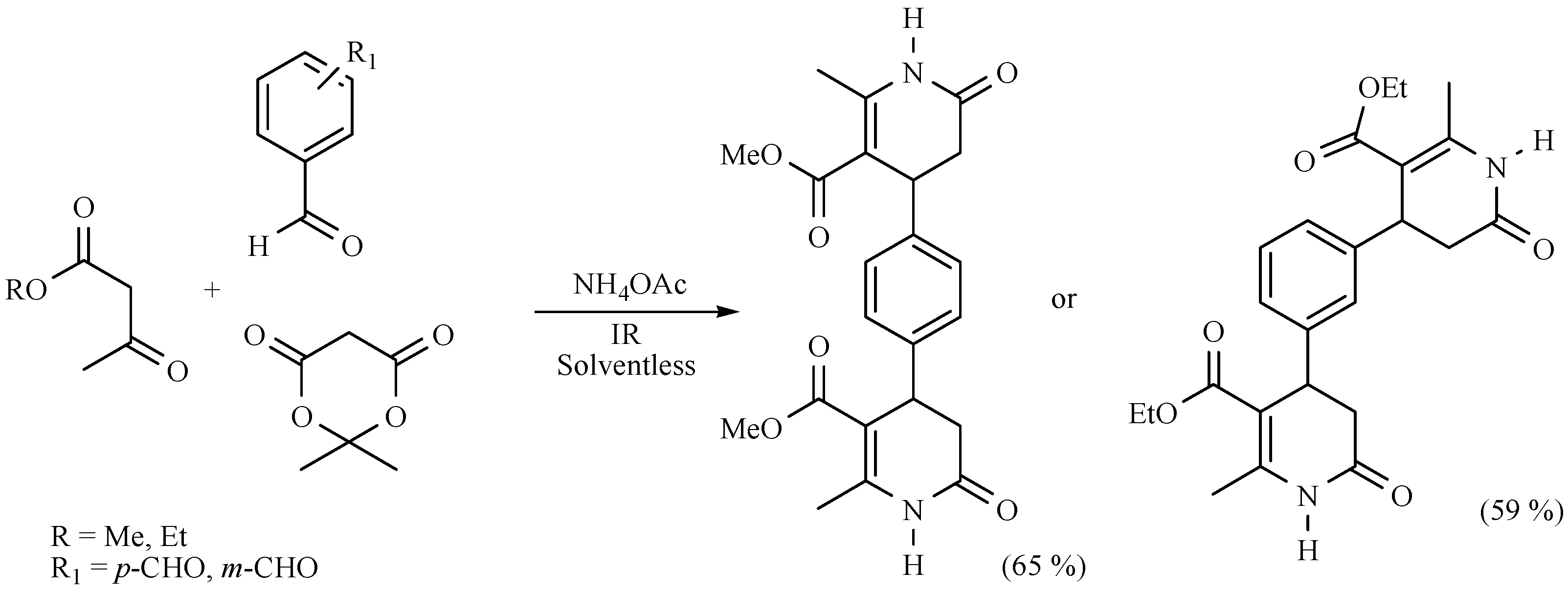

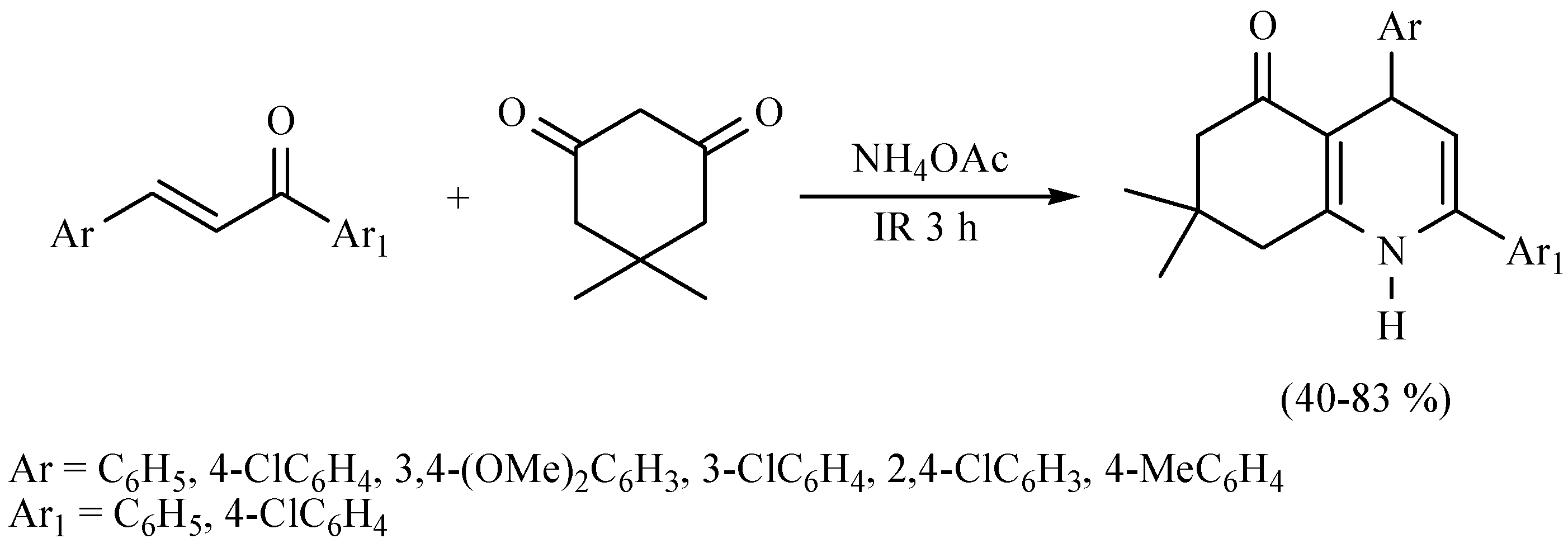
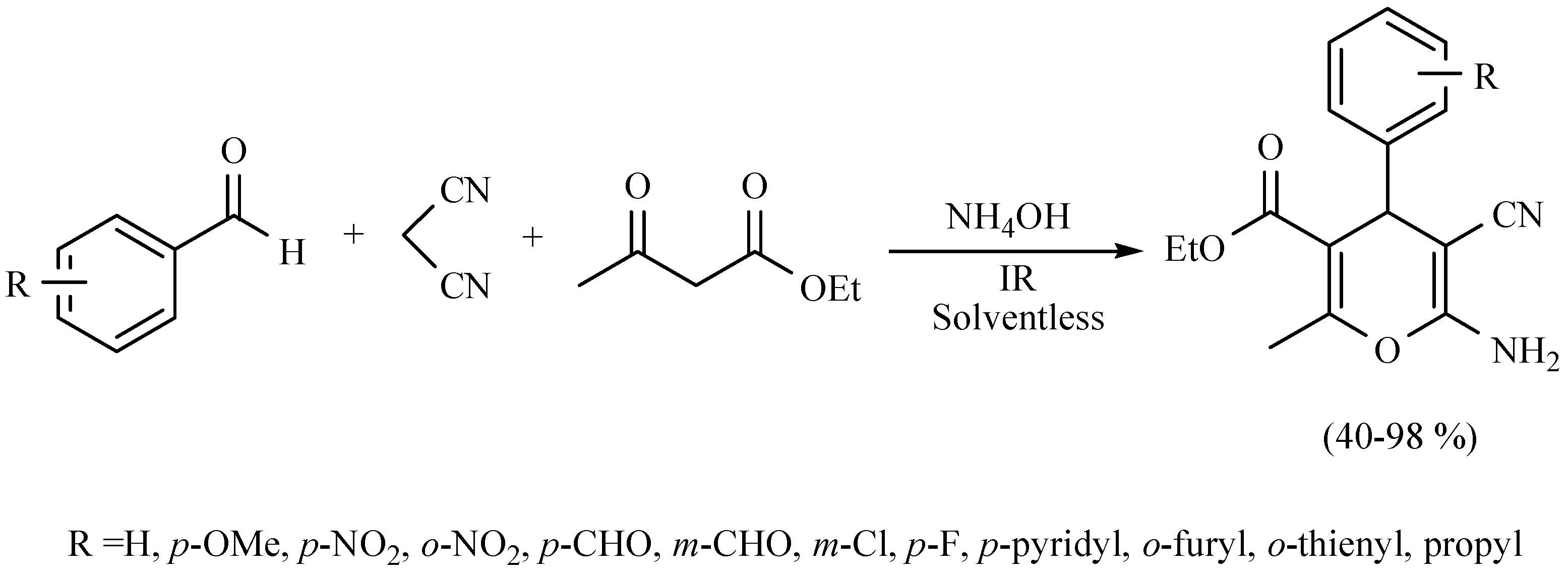
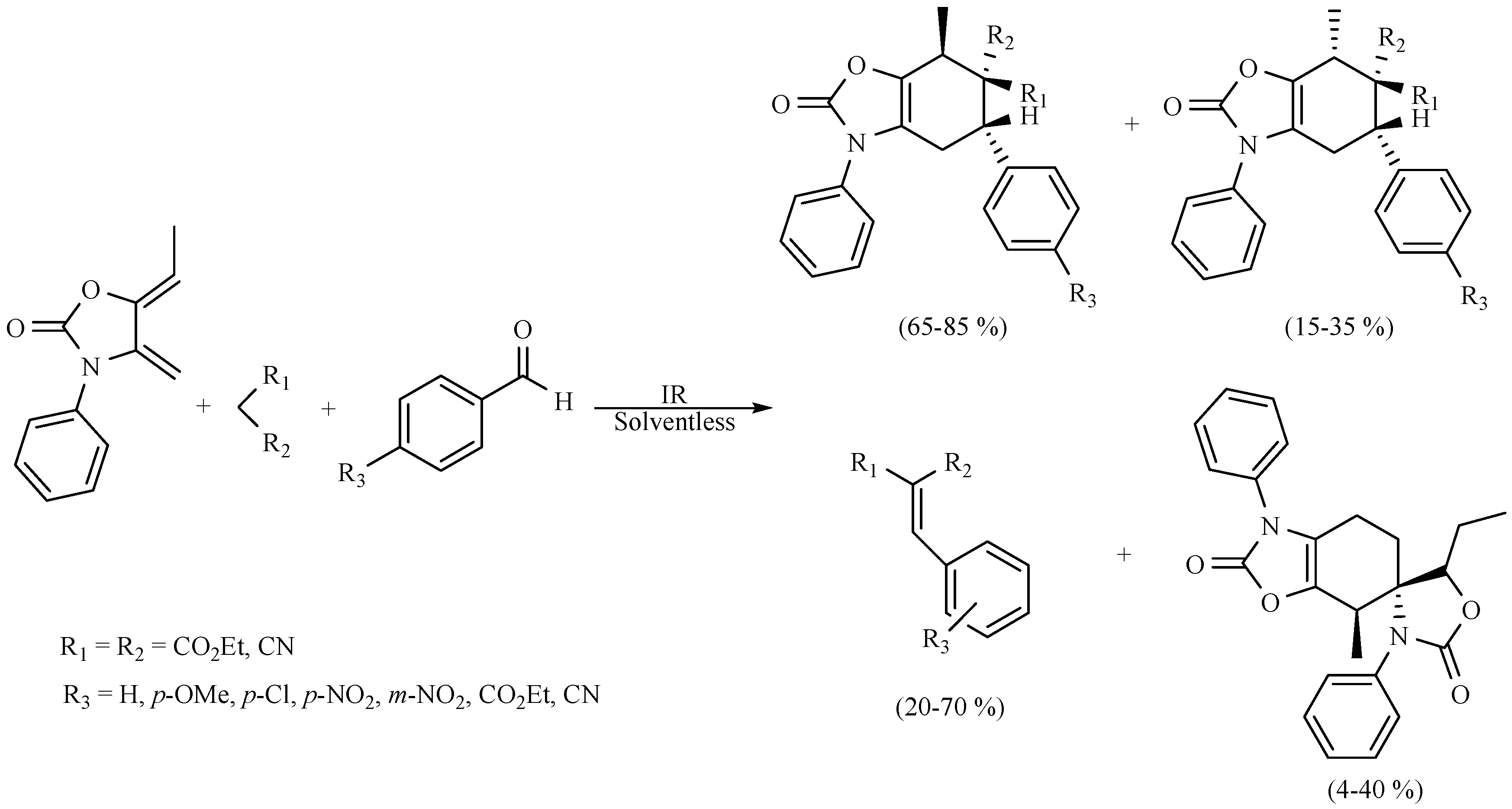
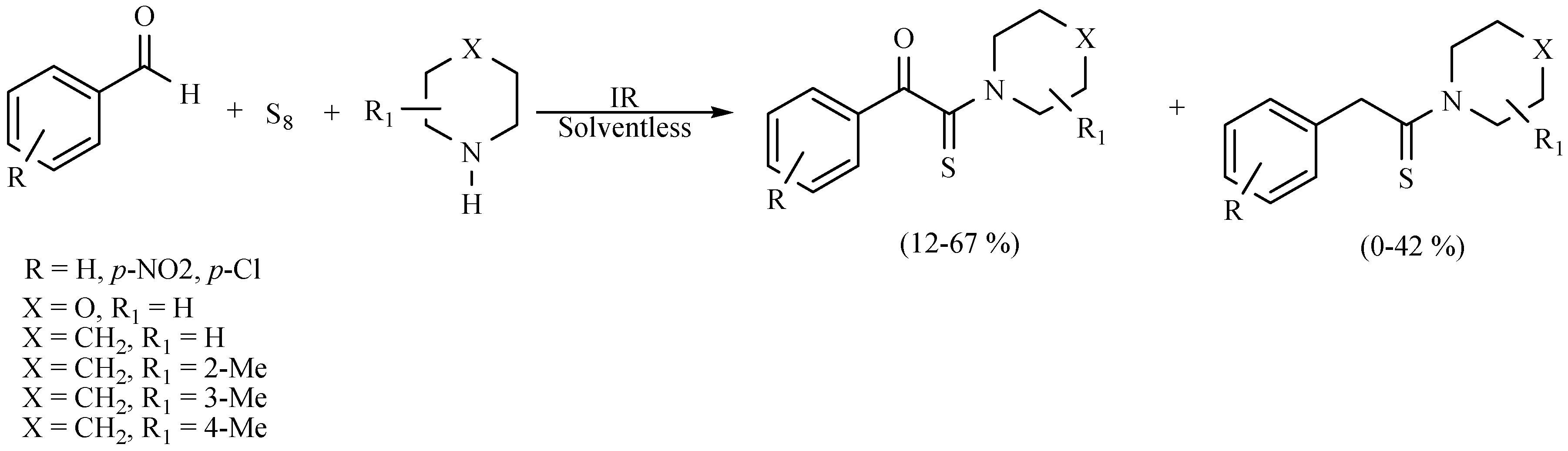
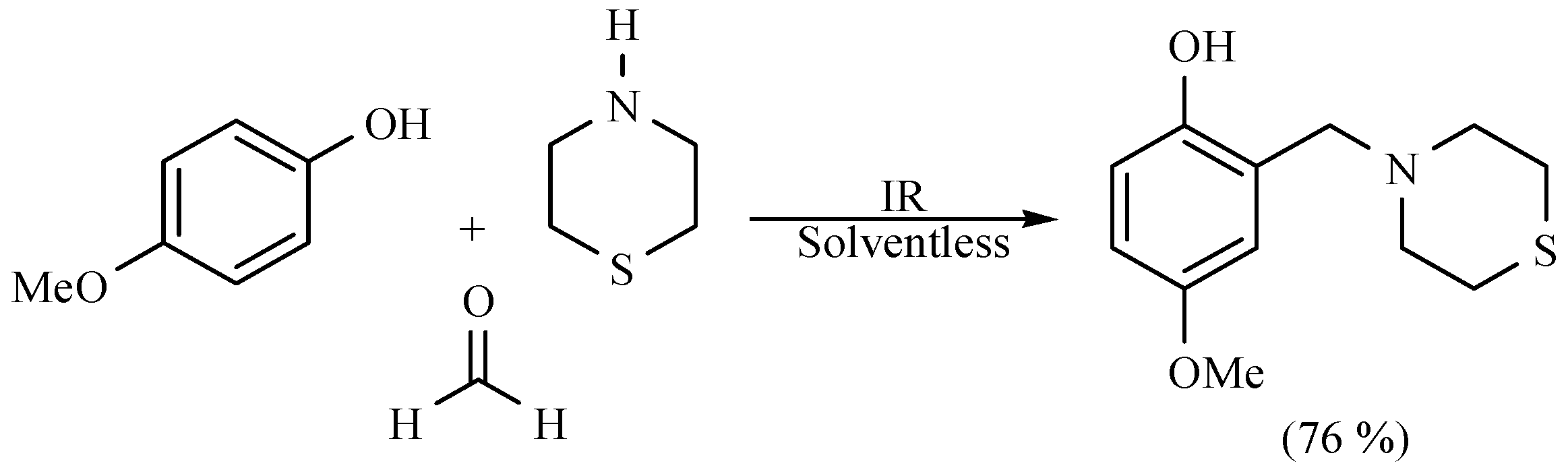
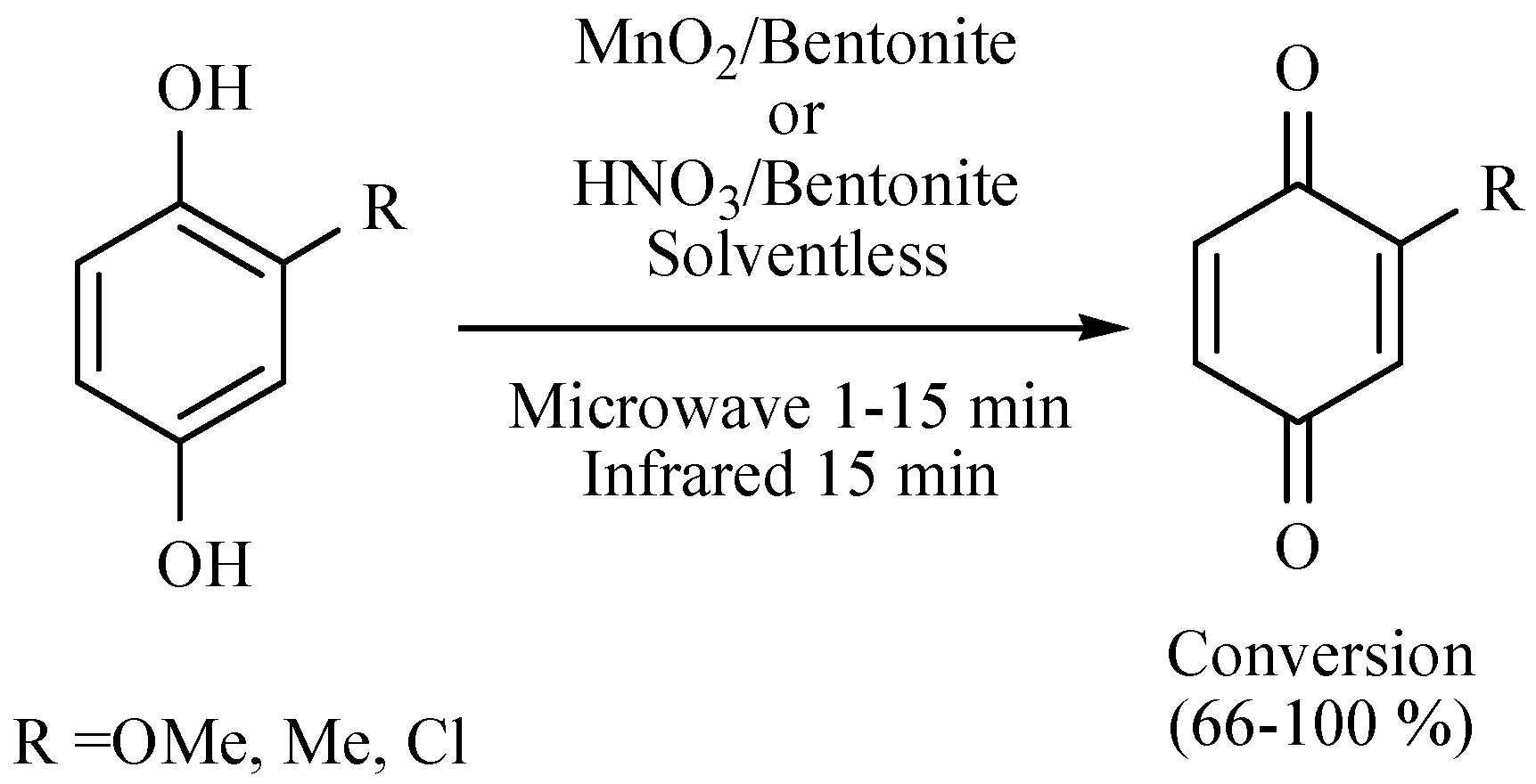
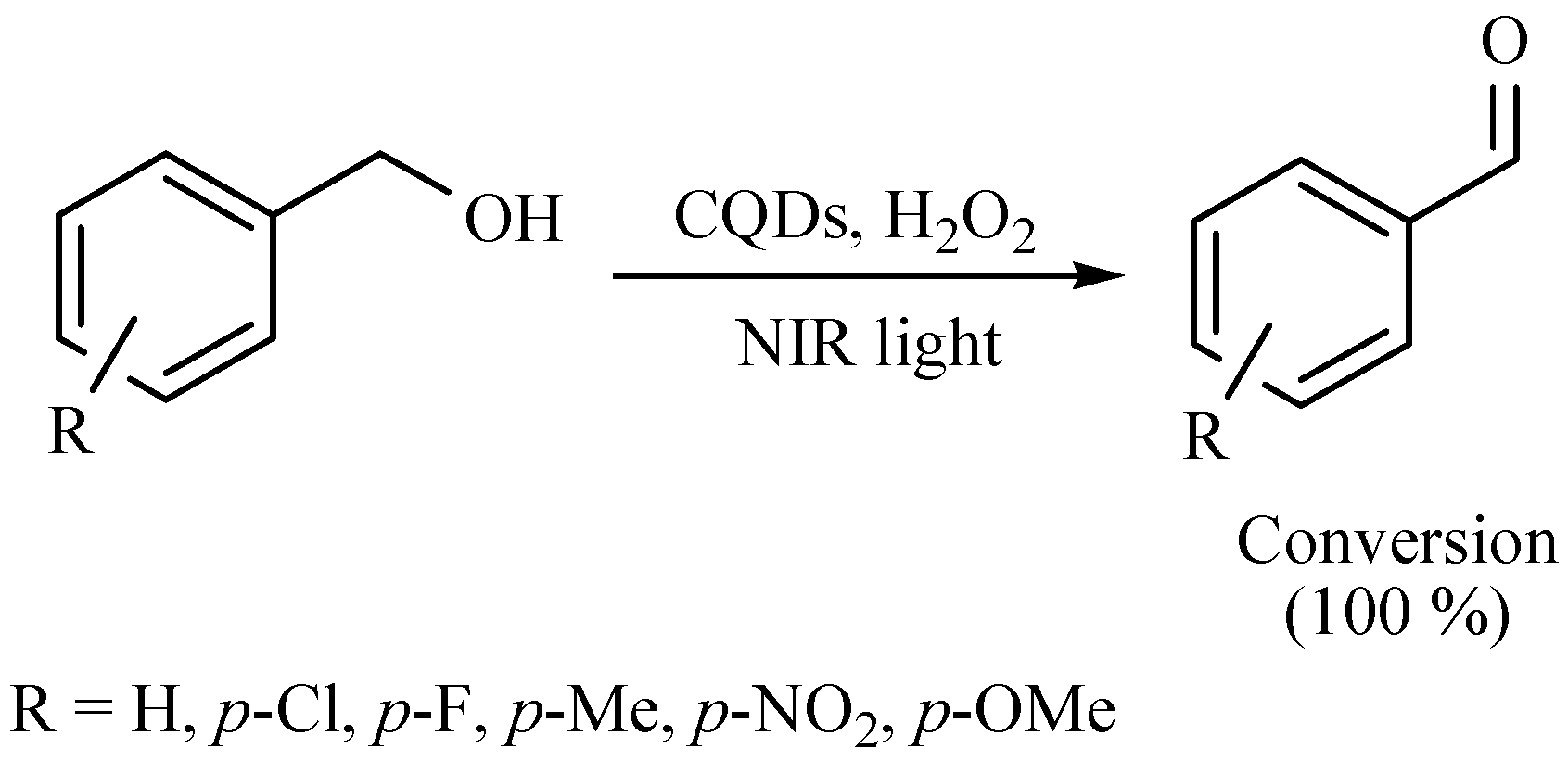
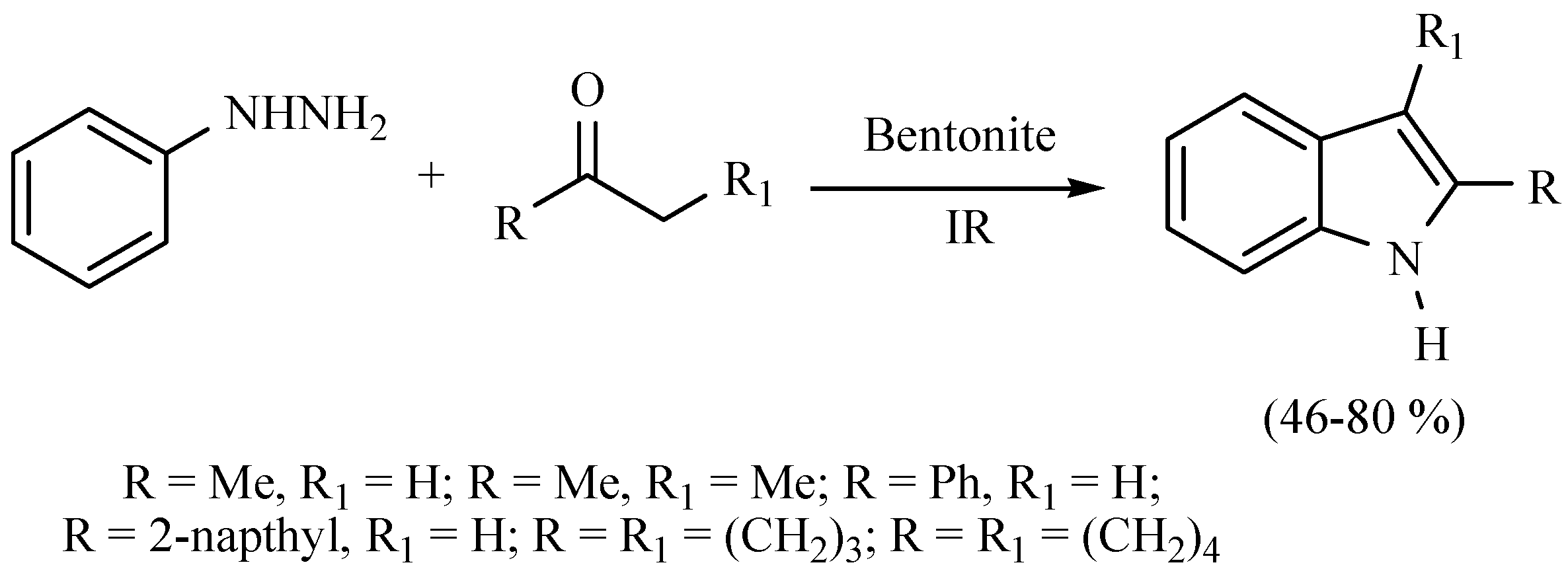
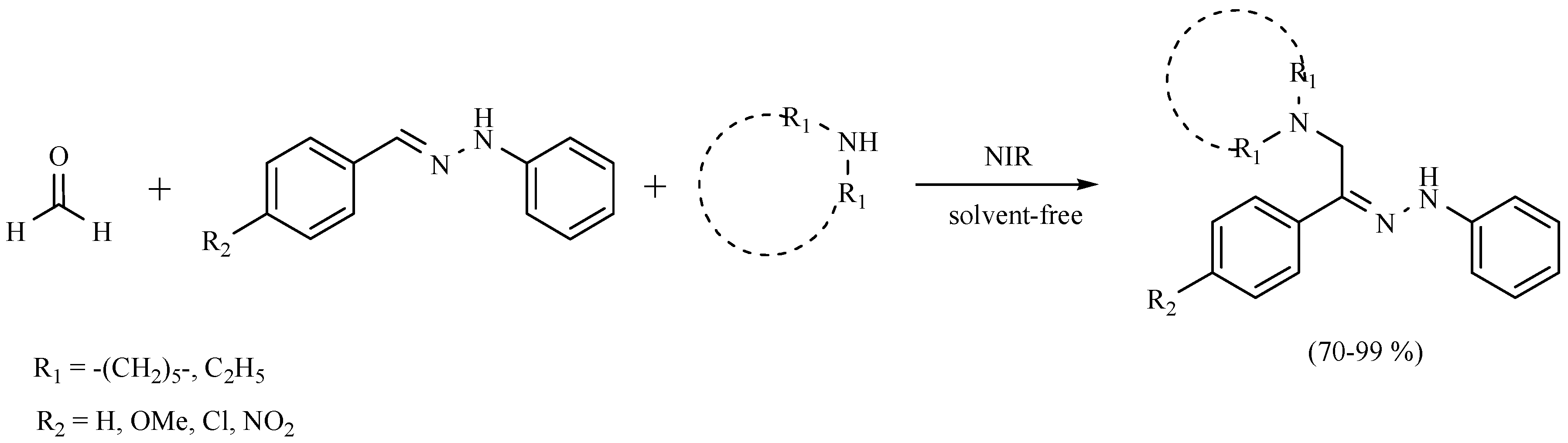
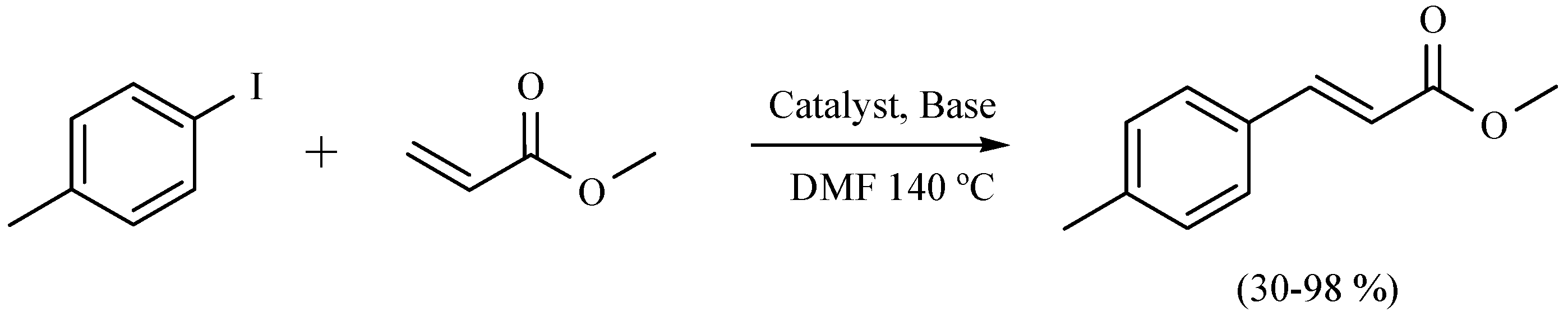
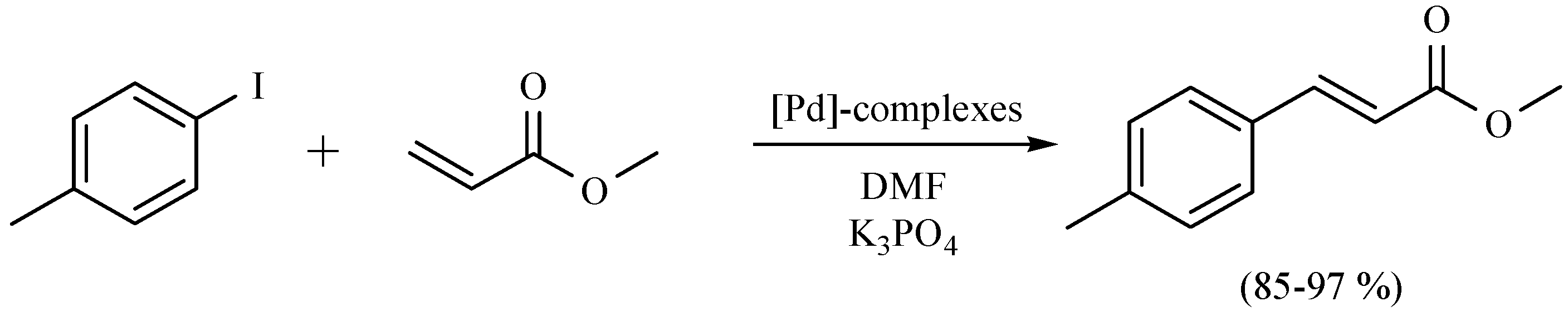
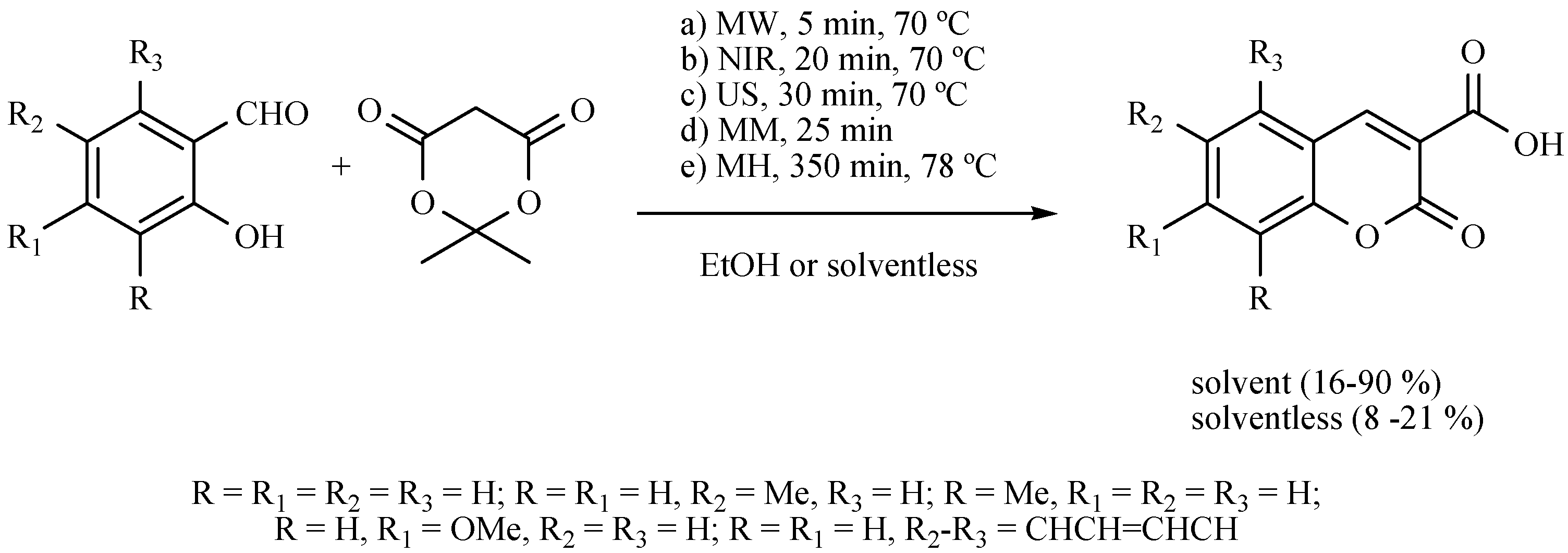

| Substrate | Reactions | MIR: Time/Temp | NIR: Time/Temp | |
|---|---|---|---|---|
| Reagent | Product | Literature [57] | This Work [56] | |
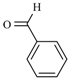 |  | 15 min/80 °C | 7 min/170 °C | |
 | 15 min/80 °C | 7 min/170 °C | ||
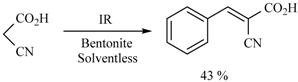 | 15 min/80 °C | 7 min/170 °C | ||
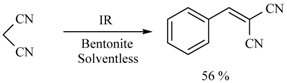 | 15 min/80 °C | 7 min/170 °C | ||
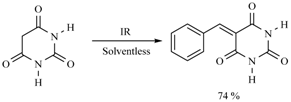 | 45 min/80 °C | 20 min/170 °C | ||
 | 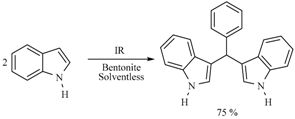 | 15 min/80 °C | 7 min/170 °C | |
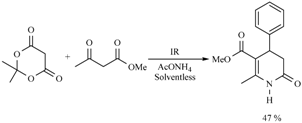 | 3 h/80 °C | 50 min/170 °C | ||
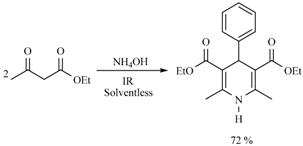 | 3 h/80 °C | 50 min/170 °C | ||
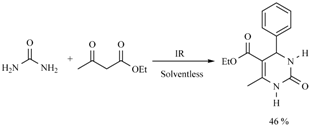 | 3 h/80 °C | 1.5 h/170 °C | ||
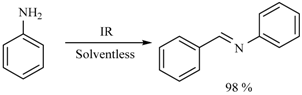 | 40 min/80 °C | 20 min/170 °C | ||
| Vegetable Material | Work Description | Compounds Analyzed | Refs. |
|---|---|---|---|
| Oriza sativa L. (Rice cultivar) | The rice hulls were treated with FIR for 30 min. After irradiation, a methanolic extract was prepared at room temperature for 1 h. The yields and antioxidant activity of the extract was better in comparison to the conventional method. In addition, The obtained results indicate that FIR could liberate and activate covalently bound phenolic compounds with antioxidant activities. | p-Coumaric acid; 3-vinyl-1-oxybenzene; 4-hydroxybenzaldehyde; vanillin; 4-hydroxybenzoic acid; and 4,7-dihydroxyvanillic acid. | [66] |
| Rice hulls pulverized and passed through a 48-mesh molecular sieve were treated with FIR for 2 h, and then a methanolic extract was prepared at room temperature overnight. The antioxidant effect of far infrared-treated rice hull (FRH) extracts in irradiated turkey breast meat was compared with that of sesamol and rosemary oleoresin. The FRH extracts significantly decreased thiobarbituric acid-reactive substances values and volatile aldehydes (hexanal, pentanal, and propanal) and was effective in reducing the production of dimethyl disulfide responsible for irradiation off-odor in irradiated raw and cooked turkey meat during aerobic storage. | [67] | ||
| Sesamum indicum L. (Sesame) | Sesame seeds were treated with FIR. The sample was defatted and the residue was used to prepare a methanolic extract by shaking overnight at room temperature. After that, the corresponding antioxidant activities were determined. | p-Hydroxybenzoic acid; o-coumaric acid, vanillic acid; p-coumaric acid, isoferulic acid; sesamol and tocopherol. | [68] |
| Arachis hypogaea L. (Peanut) | Peanut hulls dried under room temperature, were treated with FIR for 5 to 60 min at 150 °C. The irradiated material was extracted with distilled water at room temperature overnight. The total phenol contents, radical scavenging activities, and reducing power of these water extracts were determined. | 2-Methoxyphenol; 2-hydroxy-4-methoxybenzoic acid; 2-methoxy-2-vinylphenol; 4-hydroxy-3-methoxybenzaldehyde; 2,4-bis (1,1-dimethylethyl)phenol; 4-hydroxy-3-methoxybenzenacetic acid; vanillyl alcohol; methylcinnamate. | [69] |
| Camellia sinensis var. sinensis (Green tea leaves) | In a first instance, the processed green tea leaves were treated with FIR at 80–150 °C for 10 min. Then, the leaves were prepared by soaking them in boiling water and the corresponding physicochemical characteristics were determined. The applied FIR at 90 °C increased total phenol and total flavanol contents in comparison with non-irradiated control. FIR also significantly affected the amounts of epigallocatechin and epigallocatechin gallate. These results support the idea that FIR could be a useful method for increasing the health-promoting properties of green tea. | Polyphenolic compounds; caffeine; (−)-epicatechin; (−)-catechin; (−)-epicatechingallate; (−)-catechingallate; (−)-epigallocatechin; (−)-gallocatechin; (−)-epigallocatechingallate; (−)-gallocatechingallate; and ascorbic acid. | [70] |
| Green tea leaves were processed by seven modes, six of them using FIR at 90 °C for 10 min at different moments of the manufacturing process (during roasting and drying and finally on the dried product). The physicochemical characteristics of the green tea were determined. The obtained results indicate that the chemical properties of green tea are significantly affected by FIR at specific stages of the manufacturing process of green tea leaves, providing a high-quality green tea. | [71] | ||
| The effect of FIR on the physicochemical characteristic of green tea during processing was determined. Green tea leaves were irradiated by FIR during the roasting and drying step; then, the leaves were extracted by soaking them in water, and the components analyzed. The obtained results show that FIR, during the green tea process, affected its chemical properties. In addition, the authors comment the FIR can be employed to increase the physicochemical qualities of green tea. | [72] | ||
| Puerariae radix | The antioxidant activity modulated by FIR on Puerariae radix extract was evaluated. In addition, it is mentioned that this specimen has been widely used in Eastern Asia to treat the common cold and influenza and as an antidipsotropic agent. FIR at 110 °C for 40 min increased the values of total phenol contents, radical scavenging activity, reducing power and puerarin content in the corresponding methanolic extract in comparison to the non-irradiated material. The obtained results suggest that FIR could be used to increase the quality of the studied herb. | Polyphenols compounds, isoflavonoids such: puerarin, daidzein, daidzin, luteolin and biochanin A. | [73] |
| Morus alba L. (Mulberry) | The antioxidant and physical properties, and consequently the change in polyphenolic compound in mulberry tea, as influenced by FIR, were determined. This study aimed to use FIR for convection drying to improve physical quality and antioxidant properties of mulberry tea. In addition, it was demonstrated that the intensity of FIR affected the total phenolic and flavonoid contents. | p-Coumaric acid; benzoic acid; (+)-catechin; chlorogenic acid; vanillic acid; syringic acid; sinapic acid; protocatechuic acid; ferulic acid; gallic acid and caffeic acid. | [74] |
| Radix Salviae miltiorrhizae (Dashen) | In this study, FIR was employed to extract the active compounds from Radix Salviae miltiorrhizae; the optimal conditions were as follows: extraction time 15 min; extraction solvent 70% v/v methanol in water solution; and solid/liquid ratio of 0.1:15 (g/mL). Thus, four phenolic compounds and four diterpenoids were isolated. It is also worth noting that FIR was compared with conventional heat-reflux extraction, ultrasound-assisted extraction and microwave-assisted extraction. The FIR mode resulted as the best mode to extract the target molecules. | Danshensu; protocatechuic acid; protocatechuic aldehyde; salvianolic acid B; dihydrotanshinone; cryptotanshinone; tanshinone I; and tanshinone II A. | [75] |
| Lycium barbarum Linn | FIR-assisted methanolic extraction was employed to extract rutine, quercetin and gentisic acid. The effect of detection potential, irradiation time, and the voltage applied, was investigated in order to acquire the optimum analysis conditions. It is worth noting the use of capillary electrophoresis as a complementary method for the determination of bioactive constituents. The yields were similar to the conventional methods but at lower time. | Quercetin; rutin; and gentisic acid. | [76] |
| Glycyrrhiza uralensis Fisch (Licorice roots) | Licorice roots powders were treated with FIR for 30 min at 120–200 °C. After irradiation, a 50% ethanol extract was prepared at room temperature for 1 h. The total phenolic and flavanol contents in addition to the radical scavenging activities were determined. The contents of liquiritin, glycyrrhetic acid and glycyrrhzin increased. | Liquiritin; glycyrrhetic acid; and glycyrrhzin. | [77] |
| Lysium chinensis Mill | Folium Lysium Chinensis, a commonly used traditional Chinese medicine, is the dried leaf of Lysium chinensis Mill, and it has been employed to treat consumptive disease, fever, polydipsia, headache, metrorrhagia, and carbuncle. In this work, a method based on capillary electrophoresis and FIR-water assisted extraction has been developed to determine mannitol, sucrose, glucose and fructose quantities. The extraction time was significantly reduced to 7 min compared with several hours for the conventional hot solvent extraction. | Mannitol; sucrose; glucose and fructose. | [78] |
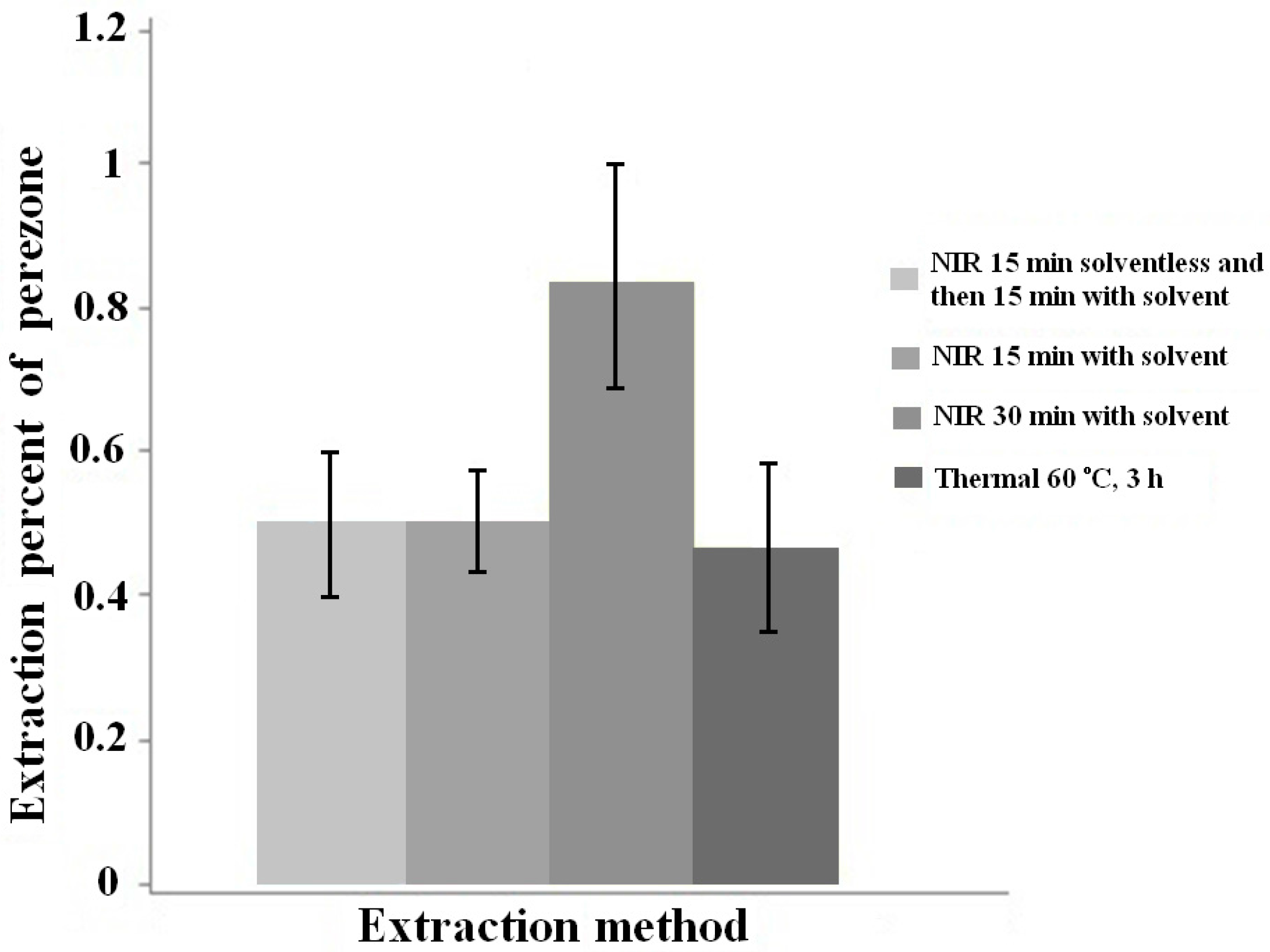
| Grape seeds | In this work, FIR-assisted extraction was combined with HPLC for the determination of catechin, epicatechin and procyanidin B2 in grape seeds. The grape seeds powder was dispersed in different mixtures of water–methanol. These mixtures were irradiated for 30 min. The extraction efficiency was compared with other methods, including microwave extraction, ultrasonic extraction and the classical mantle heating. | Catechin; epicatechin; and procyanidin B2. | [79] |
| Rhododendron mucronulatum Turcz | A combined method with FIR procedure was employed for the extraction of two flavones and three phenolic acids from the dried leaves of Rhododendron mucronulatum Turcz, commonly used in traditional Chinese medicine. The dried and pulverized leaves were dispersed in 60% aqueous methanol and treated with FIR for 6 min under reflux with methanol. The results indicate that FIR significantly enhances the extraction efficiency and reduces the time from 3 h to 6 min in comparison with the conventional heat solvent extraction. | Rutin; farrerol; syringic acid; vanillic acid and 4-hydroxybenzoic acid. | [80] |
| Tagetes erecta L. (marigold flower) | Marigold flower has long been used as a food colorant and ingredient in human food and animal feed. Drying is one the most important processes for the production of marigold powder. Therefore, in this work, the effects of different drying processes—freeze-drying, hot air drying and combined FIR-hot air convection—on the color, in addition to the carotenoids and phenolic compounds presence in the marigold flowers were evaluated. The obtained results demonstrate that FIR-hot air convection should be considered as a suitable drying method for marigold in order to preserve its color, antioxidant properties and bioactive compounds. | p-Coumaric acid; catechin; chlorogenic acid; vanillic acid; syringic acid; sinapic acid; protocatechuic; ferulic acid; gallic acid; caffeic acid and three carotenoids (lycopene, β-carotene and lutein). | [81] |
| Picrorhiza scrophulariiflora Pennell | The obtained results in this work show that FIR-assisted non-ionic surfactant extraction is good, efficient and a green analytical preparatory technique for the rapid extraction and pre-concentration of pharmacologically active ingredients from Picrorhiza scrophulariiflora Pennell. | Picroside I and picroside II. | [82] |
| Hibiscus cannabinus L. (Kenaf leaf tea) | In this work, the effect of FIR on the total polyphenol, total flavonoid, antioxidant activity and angiotensin I-converting enzyme inhibition ability were investigated in Kenaf leaf tea. The corresponding obtained data indicate that FIR at 60 °C increased the total polyphenol contents and flavonoid contents; in addition, the free radical scavenging and the lipid peroxidation inhibition activity were also increased. | Polyphenolic and flavonoid compounds; kaempferitrin, afzelin; α-rhamnoisorobin; and kaempferol. | [83] |
| Glycine max M. (black soy bean) | The goal of this work was to study the optimal temperature and time to obtain daidzein and genistein from their corresponding glycosides, employing FIR as a thermal source on black soybean. In other words, FIR is offered as a convenient green tool for the chemical conversion of isoflavone glycosides in soybean. | Daidzein and genistein. | [84] |
| Fagopyrum spp. (Buckwheat) | In this work, the FIR effect on total polyphenol, total flavonoid content, antioxidant properties and aglycone quercetin production in tartary buckwheat sprout were investigated. The powder of the herbal material was mixed with water and exposed to FIR at different temperatures (80–160 °C) for 1 h. Then, the samples were extracted with ethanol at room temperature. The corresponding results indicate that the total antioxidant capacity and metal chelating decreased, however the radical scavenging activity, the total flavonoid and polyphenol contents increased with the temperature. | Polyphenols and flavonoids (quercetin and ruthin). | [85] |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Escobedo, R.; Miranda, R.; Martínez, J. Infrared Irradiation: Toward Green Chemistry, a Review. Int. J. Mol. Sci. 2016, 17, 453. https://doi.org/10.3390/ijms17040453
Escobedo R, Miranda R, Martínez J. Infrared Irradiation: Toward Green Chemistry, a Review. International Journal of Molecular Sciences. 2016; 17(4):453. https://doi.org/10.3390/ijms17040453
Chicago/Turabian StyleEscobedo, René, René Miranda, and Joel Martínez. 2016. "Infrared Irradiation: Toward Green Chemistry, a Review" International Journal of Molecular Sciences 17, no. 4: 453. https://doi.org/10.3390/ijms17040453
APA StyleEscobedo, R., Miranda, R., & Martínez, J. (2016). Infrared Irradiation: Toward Green Chemistry, a Review. International Journal of Molecular Sciences, 17(4), 453. https://doi.org/10.3390/ijms17040453








