A Possible Role of Intestinal Microbiota in the Pathogenesis of Ankylosing Spondylitis
Abstract
:1. Introduction
2. Evidence of AS-Microbiome Interactions
3. Roles of the Intestinal Microbiome in AS
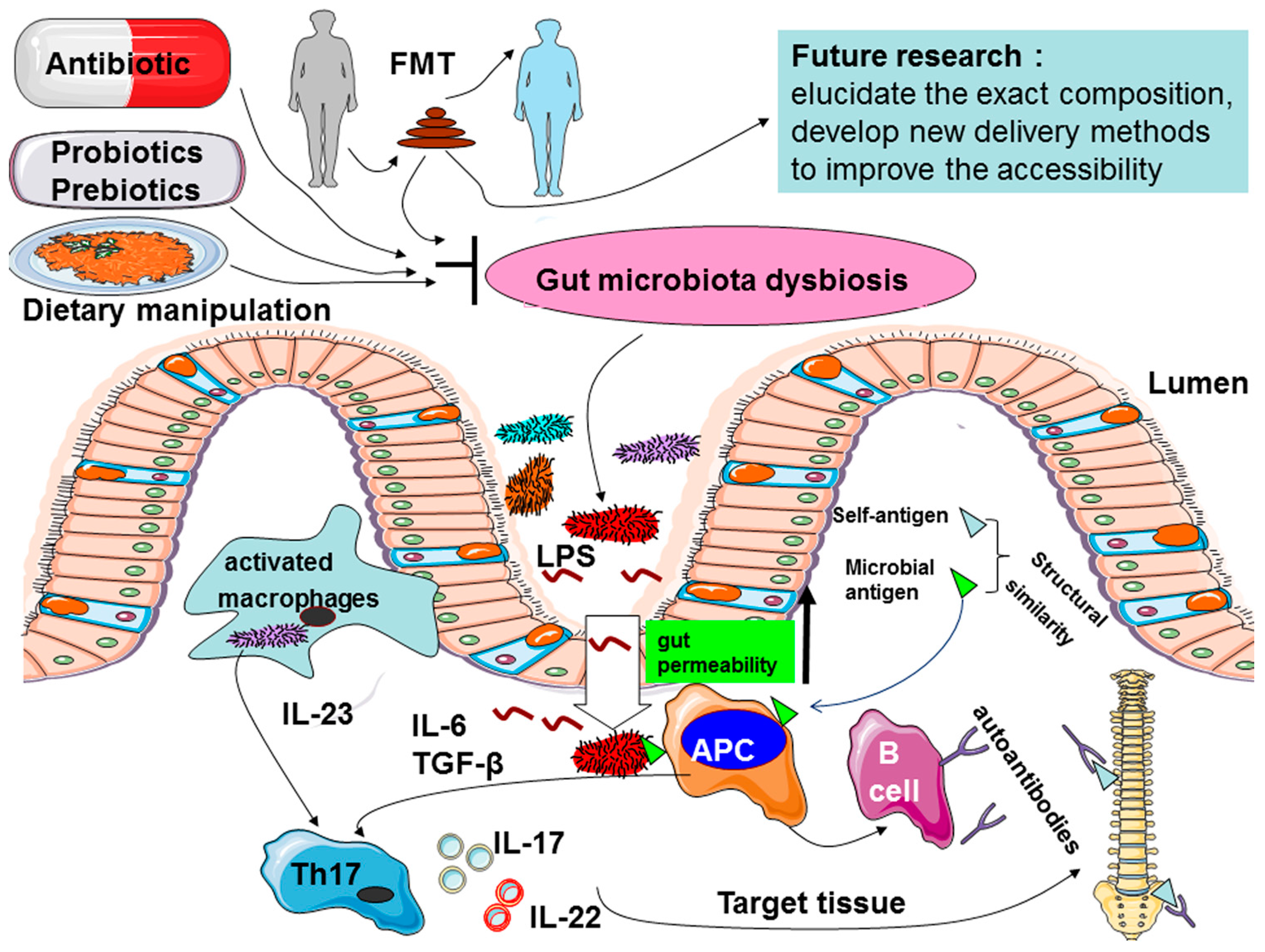
4. How the Gut Microbiome Affects AS
4.1. Increased Intestinal Permeability
4.2. Increased Gut-Joint Axis of Inflammation through Regulating Innate Immunity and IL-23/Th17 Axis
4.2.1. Innate Immunity
4.2.2. IL-23/Th17 Axis
4.3. “Molecular Mimicry” or “Cross-Reactivity”
5. Intestinal Microbiome Targeting Strategies
5.1. Antibiotics
5.2. Probiotics and Prebiotics
5.3. Dietary Manipulation
5.4. Fecal Microbial Transplantation
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Stone, M.A.; Payne, U.; Schentag, C.; Rahman, P.; Pacheco-Tena, C.; Inman, R.D. Comparative immune responses to candidate arthritogenic bacteria do not confirm a dominant role for Klebsiella pneumonia in the pathogenesis of familial ankylosing spondylitis. Rheumatology (Oxford) 2004, 43, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Coskun, B.N.; Oksuz, M.F.; Ermurat, S.; Tufan, A.N.; Orucoglu, N.; Dogan, A.; Dalkilic, E.; Pehlivan, Y. Neutrophil lymphocyte ratio can be a valuable marker in defining disease activity in patients who have started anti-tumor necrosis factor (TNF) drugs for ankylosing spondylitis. Eur. J. Rheumatol. 2014, 1, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Solmaz, D.; Kozaci, D.; Sari, I.; Taylan, A.; Onen, F.; Akkoc, N.; Akar, S. Oxidative stress and related factors in patients with ankylosing spondylitis. Eur. J. Rheumatol. 2016, 3, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Costantino, F.; Talpin, A.; Said-Nahal, R.; Goldberg, M.; Henny, J.; Chiocchia, G.; Garchon, H.J.; Zins, M.; Breban, M. Prevalence of spondyloarthritis in reference to HLA-B27 in the French population: Results of the GAZEL cohort. Ann. Rheum. Dis. 2015, 74, 689–693. [Google Scholar] [CrossRef] [PubMed]
- Pimentel-Santos, F.M.; Matos, M.; Ligeiro, D.; Mourao, A.F.; Ribeiro, C.; Costa, J.; Santos, H.; Barcelos, A.; Pinto, P.; Cruz, M.; et al. HLA alleles and HLA-B27 haplotypes associated with susceptibility and severity of ankylosing spondylitis in a Portuguese population. Tissue Antigens 2013, 82, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Malaviya, A.P.; Ostor, A.J. Early diagnosis crucial in ankylosing spondylitis. Practitioner 2011, 255, 21–24. [Google Scholar] [PubMed]
- Taurog, J.D.; Richardson, J.A.; Croft, J.T.; Simmons, W.A.; Zhou, M.; Fernandez-Sueiro, J.L.; Balish, E.; Hammer, R.E. The germfree state prevents development of gut and joint inflammatory disease in HLA-B27 transgenic rats. J. Exp. Med. 1994, 180, 2359–2364. [Google Scholar] [CrossRef] [PubMed]
- Rath, H.C.; Herfarth, H.H.; Ikeda, J.S.; Grenther, W.B.; Hamm, T.J.; Balish, E.; Taurog, J.D.; Hammer, R.E.; Wilson, K.H.; Sartor, R.B. Normal luminal bacteria, especially Bacteroides species, mediate chronic colitis, gastritis, and arthritis in HLA-B27/human β2 microglobulin transgenic rats. J. Clin. Investig. 1996, 98, 945–953. [Google Scholar] [CrossRef] [PubMed]
- Bisanz, J.E.; Suppiah, P.; Thomson, W.M.; Milne, T.; Yeoh, N.; Nolan, A.; Ettinger, G.; Reid, G.; Gloor, G.B.; Burton, J.P.; et al. The oral microbiome of patients with axial spondyloarthritis compared to healthy individuals. PeerJ 2016, 4, e2095. [Google Scholar] [CrossRef] [PubMed]
- Ogrendik, M. Periodontal pathogens are likely to be responsible for the development of ankylosing spondylitis. Curr. Rheumatol. Rev. 2015. [Google Scholar] [CrossRef]
- Rosenbaum, J.T.; Lin, P.; Asquith, M. The microbiome, HLA, and the pathogenesis of uveitis. Jpn. J. Ophthalmol. 2016, 60, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Sparks, J.A.; Costenbader, K.H. Genetics, environment, and gene-environment interactions in the development of systemic rheumatic diseases. Rheum. Dis. Clin. N. Am. 2014, 40, 637–657. [Google Scholar] [CrossRef] [PubMed]
- Scher, J.U.; Littman, D.R.; Abramson, S.B. Microbiome in inflammatory arthritis and human rheumatic diseases. Arthritis Rheumatol. 2016, 68, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Benham, H.; Robinson, P.C.; Baillet, A.C.; Rehaume, L.M.; Thomas, R. Role of genetics in infection-associated arthritis. Best Pract. Res. Clin. Rheumatol. 2015, 29, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, N.; Karadag, O.; Kimyon, G.; Yazici, A.; Yilmaz, S.; Kalyoncu, U.; Kasifoglu, T.; Temiz, H.; Baysal, B.; Tozun, N. Prevalence of hepatitis B and C infections in rheumatoid arthritis and ankylosing spondylitis: A multicenter countrywide study. Eur. J. Rheumatol. 2014, 1, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Martinez, A.; Pacheco-Tena, C.; Vazquez-Mellado, J.; Burgos-Vargas, R. Relationship between disease activity and infection in patients with spondyloarthropathies. Ann. Rheum. Dis. 2004, 63, 1338–1340. [Google Scholar] [CrossRef] [PubMed]
- Rath, H.C.; Wilson, K.H.; Sartor, R.B. Differential induction of colitis and gastritis in HLA-B27 transgenic rats selectively colonized with Bacteroides vulgatus or Escherichia coli. Infect. Immun. 1999, 67, 2969–2974. [Google Scholar] [PubMed]
- Ebringer, R.W.; Cawdell, D.R.; Cowling, P.; Ebringer, A. Sequential studies in ankylosing spondylitis. Association of Klebsiella pneumoniae with active disease. Ann. Rheum. Dis. 1978, 37, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Eastmond, C.J.; Willshaw, H.E.; Burgess, S.E.; Shinebaum, R.; Cooke, E.M.; Wright, V. Frequency of fecal Klebsiella aerogenes in patients with ankylosing spondylitis and controls with respect to individual features of the disease. Ann. Rheum. Dis. 1980, 39, 118–123. [Google Scholar] [CrossRef] [PubMed]
- Maki-Ikola, O.; Leirisalo-Repo, M.; Turunen, U.; Granfors, K. Association of gut inflammation with increased serum IgA class Klebsiella antibody concentrations in patients with axial ankylosing spondylitis (AS): Implication for different aetiopathogenetic mechanisms for axial and peripheral AS? Ann. Rheum. Dis. 1997, 56, 180–183. [Google Scholar] [CrossRef] [PubMed]
- Tani, Y.; Tiwana, H.; Hukuda, S.; Nishioka, J.; Fielder, M.; Wilson, C.; Bansal, S.; Ebringer, A. Antibodies to Klebsiella, Proteus, and HLA-B27 peptides in Japanese patients with ankylosing spondylitis and rheumatoid arthritis. J. Rheumatol. 1997, 24, 109–114. [Google Scholar] [PubMed]
- Van Kregten, E.; Huber-Bruning, O.; Vandenbroucke, J.P.; Willers, J.M. No conclusive evidence of an epidemiological relation between Klebsiella and ankylosing spondylitis. J. Rheumatol. 1991, 18, 384–388. [Google Scholar] [PubMed]
- Rashid, T.; Ebringer, A. Autoimmunity in rheumatic diseases is induced by microbial infections via crossreactivity or molecular mimicry. Autoimmune Dis. 2012, 2012, 539282. [Google Scholar] [CrossRef] [PubMed]
- Dulal, S.; Keku, T.O. Gut microbiome and colorectal adenomas. Cancer J. 2014, 20, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Burcelin, R. Gut microbiota and immune crosstalk in metabolic disease. Mol. Metab. 2016, 5, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Paun, A.; Danska, J.S. Modulation of type 1 and type 2 diabetes risk by the intestinal microbiome. Pediatr. Diabetes 2016, 17, 469–477. [Google Scholar] [CrossRef] [PubMed]
- Sampaio-Maia, B.; Simoes-Silva, L.; Pestana, M.; Araujo, R.; Soares-Silva, I.J. The Role of the gut microbiome on chronic kidney disease. Adv. Appl. Microbiol. 2016, 96, 65–94. [Google Scholar] [PubMed]
- Jostins, L.; Ripke, S.; Weersma, R.K.; Duerr, R.H.; McGovern, D.P.; Hui, K.Y.; Lee, J.C.; Schumm, L.P.; Sharma, Y.; Anderson, C.A.; et al. Host-microbe interactions have shaped the genetic architecture of inflammatory bowel disease. Nature 2012, 491, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Costello, M.E.; Ciccia, F.; Willner, D.; Warrington, N.; Robinson, P.C.; Gardiner, B.; Marshall, M.; Kenna, T.J.; Triolo, G.; Brown, M.A. Intestinal dysbiosis in ankylosing spondylitis. Arthritis Rheumatol. 2014. [Google Scholar] [CrossRef]
- Rodrigues, I.K.; Andrigueti, M.; de Oliveira, G.I.; de Lucca, S.L.; de Andrade, K.R.; Pereira, I.A.; de Castro, G.R.; Zimmermann, A.F.; Saporiti, L.N.; Bazzo, M.L.; et al. An investigation into the relationship between anti-Helicobacter pylori and anti-Saccharomyces cerevisiae antibodies in patients with axial spondyloarthritis and Crohn disease. Rheumatol. Int. 2015, 35, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Ciccia, F.; Rizzo, A.; Triolo, G. Subclinical gut inflammation in ankylosing spondylitis. Curr. Opin. Rheumatol. 2016, 28, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Stoll, M.L. Gut microbes, immunity, and spondyloarthritis. Clin. Immunol. 2015, 159, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Tito, R.Y.; Cypers, H.; Joossens, M.; Varkas, G.; van Praet, L.; Glorieus, E.; van den Bosch, F.; de Vos, M.; Raes, J.; Elewaut, D. Dialister as microbial marker of disease activity in spondyloarthritis. Arthritis Rheumatol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Asquith, M.; Rosenbaum, J.T. The interaction between host genetics and the microbiome in the pathogenesis of spondyloarthropathies. Curr. Opin. Rheumatol. 2016, 28, 405–412. [Google Scholar] [CrossRef] [PubMed]
- Schaeverbeke, T.; Truchetet, M.E.; Richez, C. Gut metagenome and spondyloarthritis. Jt. Bone Spine 2013, 80, 349–352. [Google Scholar] [CrossRef] [PubMed]
- Costello, M.E.; Elewaut, D.; Kenna, T.J.; Brown, M.A. Microbes, the gut and ankylosing spondylitis. Arthritis Res. Ther. 2013, 15, 214. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, J.T.; Davey, M.P. Time for a gut check: Evidence for the hypothesis that HLA-B27 predisposes to ankylosing spondylitis by altering the microbiome. Arthritis Rheum. 2011, 63, 3195–3198. [Google Scholar] [CrossRef] [PubMed]
- Mitulescu, T.C.; Stavaru, C.; Voinea, L.M.; Banica, L.M.; Matache, C.; Predeteanu, D. The role of Vitamin D in immuno-inflammatory responses in ankylosing spondylitis patients with and without acute anterior uveitis. J. Med. Life 2016, 9, 26–33. [Google Scholar] [PubMed]
- Ebringer, A.; Rashid, T.; Tiwana, H.; Wilson, C. A possible link between Crohn’s disease and ankylosing spondylitis via Klebsiella infections. Clin. Rheumatol. 2007, 26, 289–297. [Google Scholar] [CrossRef] [PubMed]
- Ebringer, A.; Wilson, C. The use of a low starch diet in the treatment of patients suffering from ankylosing spondylitis. Clin. Rheumatol. 1996, 15, 62–66. [Google Scholar] [CrossRef] [PubMed]
- Stebbings, S.; Munro, K.; Simon, M.A.; Tannock, G.; Highton, J. Comparison of the fecal microflora of patients with ankylosing spondylitis and controls using molecular methods of analysis. Rheumatology 2002, 41, 1395–1401. [Google Scholar] [CrossRef] [PubMed]
- Montoya, J.; Matta, N.B.; Suchon, P.; Guzian, M.C.; Lambert, N.C.; Mattei, J.P.; Guis, S.; Breban, M.; Roudier, J.; Balandraud, N. Patients with ankylosing spondylitis have been breast fed less often than healthy controls: A case-control retrospective study. Ann. Rheum. Dis. 2016, 75, 879–882. [Google Scholar] [CrossRef] [PubMed]
- Inkorová, Z.; Apková, J.; Niederlová, J.; Těpánková, R.; Inkora, J. Commensal intestinal bacterial strains trigger ankylosing enthesopathy of the ankle in inbred B10.BR (H-2k) male mice. Hum. Immunol. 2008, 69, 845–850. [Google Scholar] [CrossRef] [PubMed]
- Lin, P.; Bach, M.; Asquith, M.; Lee, A.Y.; Akileswaran, L.; Stauffer, P.; Davin, S.; Pan, Y.; Cambronne, E.D.; Dorris, M.; et al. HLA-B27 and human β2-microglobulin affect the gut microbiota of transgenic rats. PLoS ONE 2014, 9, e105684. [Google Scholar] [CrossRef] [PubMed]
- Hoentjen, F.; Welling, G.W.; Harmsen, H.J.; Zhang, X.; Snart, J.; Tannock, G.W.; Lien, K.; Churchill, T.A.; Lupicki, M.; Dieleman, L.A. Reduction of colitis by prebiotics in HLA-B27 transgenic rats is associated with microflora changes and immunomodulation. Inflamm. Bowel Dis. 2005, 11, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Ciccia, F.; Ferrante, A.; Triolo, G. Intestinal dysbiosis and innate immune responses in axial spondyloarthritis. Curr. Opin. Rheumatol. 2016, 28, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Tlaskalová-Hogenová, H.; Těpánková, R.; Hudcovic, T.; Tu Ková, L.; Cukrowska, B.E.; Lodinová-Ádn Ková, R.; Kozáková, H.; Rossmann, P.; Bártová, J.I.; Sokol, D.; et al. Commensal bacteria (normal microflora), mucosal immunity and chronic inflammatory and autoimmune diseases. Immunol. Lett. 2004, 93, 97–108. [Google Scholar] [CrossRef] [PubMed]
- Tian, P.; Li, B.; He, C.; Song, W.; Hou, A.; Tian, S.; Meng, X.; Li, K.; Shan, Y. Antidiabetic (type 2) effects of Lactobacillus G15 and Q14 in rats through regulation of intestinal permeability and microbiota. Food Funct. 2016, 7, 3789–3797. [Google Scholar] [CrossRef] [PubMed]
- Morris, G.; Berk, M.; Carvalho, A.F.; Caso, J.R.; Sanz, Y.; Maes, M. The role of microbiota and intestinal permeability in the pathophysiology of autoimmune and neuroimmune processes with an emphasis on inflammatory bowel disease type 1 diabetes and chronic fatigue syndrome. Curr. Pharm. Des. 2016. [Google Scholar] [CrossRef]
- Martinez-Gonzalez, O.; Cantero-Hinojosa, J.; Paule-Sastre, P.; Gomez-Magan, J.C.; Salvatierra-Rios, D. Intestinal permeability in patients with ankylosing spondylitis and their healthy relatives. Br. J. Rheumatol. 1994, 33, 644–647. [Google Scholar] [CrossRef] [PubMed]
- Kerr, S.W.; Wolyniec, W.W.; Filipovic, Z.; Nodop, S.G.; Braza, F.; Winquist, R.J.; Noonan, T.C. Repeated measurement of intestinal permeability as an assessment of colitis severity in HLA-B27 transgenic rats. J. Pharmacol. Exp. Ther. 1999, 291, 903–910. [Google Scholar] [PubMed]
- Hollander, D.; Vadheim, C.M.; Brettholz, E.; Petersen, G.M.; Delahunty, T.; Rotter, J.I. Increased intestinal permeability in patients with Crohn’s disease and their relatives. A possible etiologic factor. Ann. Intern. Med. 1986, 105, 883–885. [Google Scholar] [CrossRef] [PubMed]
- Toyonaga, T.; Matsuura, M.; Mori, K.; Honzawa, Y.; Minami, N.; Yamada, S.; Kobayashi, T.; Hibi, T.; Nakase, H. Lipocalin 2 prevents intestinal inflammation by enhancing phagocytic bacterial clearance in macrophages. Sci. Rep. 2016, 6, 35014. [Google Scholar] [CrossRef] [PubMed]
- Ochi, T.; Feng, Y.; Kitamoto, S.; Nagao-Kitamoto, H.; Kuffa, P.; Atarashi, K.; Honda, K.; Teitelbaum, D.H.; Kamada, N. Diet-dependent, microbiota-independent regulation of IL-10-producing lamina propria macrophages in the small intestine. Sci. Rep. 2016, 6, 27634. [Google Scholar] [CrossRef] [PubMed]
- Cipriani, G.; Gibbons, S.J.; Kashyap, P.C.; Farrugia, G. Intrinsic gastrointestinal macrophages: Their phenotype and role in gastrointestinal motility. Cell. Mol. Gastroenterol. Hepatol. 2016, 2, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Steinert, A.; Radulovic, K.; Niess, J. Gastro-intestinal tract: The leading role of mucosal immunity. Swiss Med. Wkly. 2016, 146, w14293. [Google Scholar] [CrossRef] [PubMed]
- Bain, C.C.; Mowat, A.M. Macrophages in intestinal homeostasis and inflammation. Immunol. Rev. 2014, 260, 102–117. [Google Scholar] [CrossRef] [PubMed]
- Kamada, N.; Hisamatsu, T.; Okamoto, S.; Chinen, H.; Kobayashi, T.; Sato, T.; Sakuraba, A.; Kitazume, M.T.; Sugita, A.; Koganei, K.; et al. Unique CD14 intestinal macrophages contribute to the pathogenesis of Crohn disease via IL-23/IFN-γ axis. J. Clin. Investig. 2008, 118, 2269–2280. [Google Scholar] [CrossRef] [PubMed]
- Sartor, R.B. Mechanisms of disease: Pathogenesis of Crohn’s disease and ulcerative colitis. Nat. Clin. Pract. Gastroenterol. Hepatol. 2006, 3, 390–407. [Google Scholar] [CrossRef] [PubMed]
- Redhu, N.; Shouval, D.; Bakthavatchalu, V.; Wang, C.; Conaway, E.; Goettel, J.; Mack, M.; Bleich, A.; Bry, L.; Fox, J.; et al. O-005 YI microbiota drives inflammation by altering intestinal lamina propria macrophage phenotype in a novel IL10R-deficient model of very early onset IBD. Inflamm. Bowel Dis. 2016, 22, S2–S3. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; He, C.; Cong, Y.; Liu, Z. Regulatory immune cells in regulation of intestinal inflammatory response to microbiota. Mucosal. Immunol. 2015, 8, 969–978. [Google Scholar] [CrossRef] [PubMed]
- Bystrom, J.; Evans, I.; Newson, J.; Stables, M.; Toor, I.; van Rooijen, N.; Crawford, M.; Colville-Nash, P.; Farrow, S.; Gilroy, D.W. Resolution-phase macrophages possess a unique inflammatory phenotype that is controlled by cAMP. Blood 2008, 112, 4117–4127. [Google Scholar] [CrossRef] [PubMed]
- Vandooren, B.; Noordenbos, T.; Ambarus, C.; Krausz, S.; Cantaert, T.; Yeremenko, N.; Boumans, M.; Lutter, R.; Tak, P.P.; Baeten, D. Absence of a classically activated macrophage cytokine signature in peripheral spondylarthritis, including psoriatic arthritis. Arthritis Rheum. 2009, 60, 966–975. [Google Scholar] [CrossRef] [PubMed]
- Ciccia, F.; Alessandro, R.; Rizzo, A.; Accardo-Palumbo, A.; Raimondo, S.; Raiata, F.; Guggino, G.; Giardina, A.; de Leo, G.; Sireci, G.; et al. Macrophage phenotype in the subclinical gut inflammation of patients with ankylosing spondylitis. Rheumatology (Oxford) 2014, 53, 104–113. [Google Scholar] [CrossRef] [PubMed]
- Appel, H.; Maier, R.; Bleil, J.; Hempfing, A.; Loddenkemper, C.; Schlichting, U.; Syrbe, U.; Sieper, J. In situ analysis of interleukin-23- and interleukin-12-positive cells in the spine of patients with ankylosing spondylitis. Arthritis Rheum. 2013, 65, 1522–1529. [Google Scholar] [CrossRef] [PubMed]
- Ciccia, F.; Bombardieri, M.; Principato, A.; Giardina, A.; Tripodo, C.; Porcasi, R.; Peralta, S.; Franco, V.; Giardina, E.; Craxi, A.; et al. Overexpression of interleukin-23, but not interleukin-17, as an immunologic signature of subclinical intestinal inflammation in ankylosing spondylitis. Arthritis Rheum. 2009, 60, 955–965. [Google Scholar] [CrossRef] [PubMed]
- Gupta, L.; Bhattacharya, S.; Agarwal, V.; Aggarwal, A. Elevated levels of serum MRP8/14 in ankylosing spondylitis: Associated with peripheral arthritis and active disease. Clin. Rheumatol. 2016. [Google Scholar] [CrossRef] [PubMed]
- Welsby, I.; Goriely, S. Regulation of interleukin-23 expression in health and disease. Adv. Exp. Med. Biol. 2016, 941, 167–189. [Google Scholar] [PubMed]
- Meng, P.; Zhao, S.; Niu, X.; Fu, N.; Su, S.; Wang, R.; Zhang, Y.; Qiao, L.; Nan, Y. Involvement of the interleukin-23/interleukin-17 axis in chronic hepatitis C virus infection and its treatment responses. Int. J. Mol. Sci. 2016, 17, 1070. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zou, Y.; Peng, J.; Lu, F.; Yin, Y.; Li, F.; Yang, J. Lactobacillus acidophilus suppresses colitis-associated activation of the IL-23/Th17 axis. J. Immunol. Res. 2015. [Google Scholar] [CrossRef] [PubMed]
- Sakkas, L.I.; Bogdanos, D.P. Are psoriasis and psoriatic arthritis the same disease? The IL-23/IL-17 axis data. Autoimmun. Rev. 2016. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.C.; Lu, M.C.; Huang, K.Y.; Huang, H.L.; Liu, S.Q.; Huang, H.B.; Lai, N.S. Sulfasalazine treatment suppresses the formation of HLA-B27 heavy chain homodimer in patients with ankylosing spondylitis. Int. J. Mol. Sci. 2015, 17, 46. [Google Scholar] [CrossRef] [PubMed]
- Wendling, D. An overview of investigational new drugs for treating ankylosing spondylitis. Expert. Opin. Investig. Drugs 2016, 25, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Hreggvidsdottir, H.S.; Noordenbos, T.; Baeten, D.L. Inflammatory pathways in spondyloarthritis. Mol. Immunol. 2014, 57, 28–37. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.; Colbert, R.A. Review: The interleukin-23/interleukin-17 axis in spondyloarthritis pathogenesis: Th17 and beyond. Arthritis Rheumatol. 2014, 66, 231–241. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, J.T.; Asquith, M.J. The microbiome: A revolution in treatment for rheumatic diseases? Curr. Rheumatol. Rep. 2016, 18, 62. [Google Scholar] [CrossRef] [PubMed]
- Goodall, J.C.; Wu, C.; Zhang, Y.; McNeill, L.; Ellis, L.; Saudek, V.; Gaston, J.S. Endoplasmic reticulum stress-induced transcription factor, CHOP, is crucial for dendritic cell IL-23 expression. Proc. Natl. Acad. Sci. USA 2010, 107, 17698–17703. [Google Scholar] [CrossRef] [PubMed]
- Noto, L.M.; Sarnacki, S.H.; Aya, C.M.R.; Bernal, M.I.; Giacomodonato, M.N.; Cerquetti, M.C. Consumption of Lactobacillus casei fermented milk prevents Salmonella reactive arthritis by modulating IL-23/IL-17 expression. PLoS ONE 2013, 8, e82588. [Google Scholar] [CrossRef]
- Ciccia, F.; Guggino, G.; Rizzo, A.; Saieva, L.; Peralta, S.; Giardina, A.; Cannizzaro, A.; Sireci, G.; de Leo, G.; Alessandro, R.; et al. Type 3 innate lymphoid cells producing IL-17 and IL-22 are expanded in the gut, in the peripheral blood, synovial fluid and bone marrow of patients with ankylosing spondylitis. Ann. Rheum. Dis. 2015, 74, 1739–1747. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Q.; Zhou, K.; Liang, Q.L.; Lin, S.; Wang, Y.C.; Xiong, X.Y.; Meng, Z.Y.; Zhao, T.; Zhu, W.Y.; Yang, Y.R.; et al. Interleukin-23 secreted by activated macrophages drives γΔT cell production of interleukin-17 to aggravate secondary injury after intracerebral hemorrhage. J. Am. Heart. Assoc. 2016, 5, e004340. [Google Scholar] [CrossRef] [PubMed]
- Sherlock, J.P.; Taylor, P.C.; Buckley, C.D.; Cua, D.J. Spondyloarthropathy: Interleukin 23 and disease modification. Lancet 2015, 385, 2017–2018. [Google Scholar] [CrossRef]
- Su, S.S.; Wang, S.L.; Lu, L.J.; Lin, L. Association of interleukin-23 receptor gene single nucleotide polymorphisms with ankylosing spondylitis. Ann. Clin. Lab. Sci. 2016, 46, 470–473. [Google Scholar] [PubMed]
- Ugur, M.; Baygutalp, N.K.; Melikoglu, M.A.; Baygutalp, F.; Altas, E.U.; Seferoglu, B. Elevated serum interleukin-23 levels in ankylosing spondylitis patients and the relationship with disease activity. Nagoya J. Med. Sci. 2015, 77, 621–627. [Google Scholar] [PubMed]
- Przepiera-Bedzak, H.; Fischer, K.; Brzosko, M. Serum IL-6 and IL-23 levels and their correlation with angiogenic cytokines and disease activity in ankylosing spondylitis, psoriatic arthritis, and SAPHO syndrome. Mediators Inflamm. 2015. [Google Scholar] [CrossRef] [PubMed]
- Wright, P.B.; McEntegart, A.; McCarey, D.; McInnes, I.B.; Siebert, S.; Milling, S.W. Ankylosing spondylitis patients display altered dendritic cell and T cell populations that implicate pathogenic roles for the IL-23 cytokine axis and intestinal inflammation. Rheumatology (Oxford) 2016, 55, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Asquith, M.; Elewaut, D.; Lin, P.; Rosenbaum, J.T. The role of the gut and microbes in the pathogenesis of spondyloarthritis. Best Pract. Res. Clin. Rheumatol. 2014, 28, 687–702. [Google Scholar] [CrossRef] [PubMed]
- Mease, P.J. Inhibition of interleukin-17, interleukin-23 and the Th17 cell pathway in the treatment of psoriatic arthritis and psoriasis. Curr. Opin. Rheumatol. 2015, 27, 127–133. [Google Scholar] [PubMed]
- Benham, H.; Rehaume, L.M.; Hasnain, S.Z.; Velasco, J.; Baillet, A.C.; Ruutu, M.; Kikly, K.; Wang, R.; Tseng, H.W.; Thomas, G.P.; et al. Interleukin-23 mediates the intestinal response to microbial β-1, 3-glucan and the development of spondyloarthritis pathology in SKG mice. Arthritis Rheumatol. 2014, 66, 1755–1767. [Google Scholar] [CrossRef] [PubMed]
- Hayashi, E.; Chiba, A.; Tada, K.; Haga, K.; Kitagaichi, M.; Nakajima, S.; Kusaoi, M.; Sekiya, F.; Ogasawara, M.; Yamaji, K.; et al. Involvement of mucosal-associated invariant T cells in ankylosing spondylitis. J. Rheumatol. 2016, 43, 1695–1703. [Google Scholar] [CrossRef] [PubMed]
- Gracey, E.; Qaiyum, Z.; Almaghlouth, I.; Lawson, D.; Karki, S.; Avvaru, N.; Zhang, Z.; Yao, Y.; Ranganathan, V.; Baglaenko, Y.; et al. IL-7 primes IL-17 in mucosal-associated invariant T (MAIT) cells, which contribute to the Th17-axis in ankylosing spondylitis. Ann. Rheum. Dis. 2016. [Google Scholar] [CrossRef] [PubMed]
- Serriari, N.E.; Eoche, M.; Lamotte, L.; Lion, J.; Fumery, M.; Marcelo, P.; Chatelain, D.; Barre, A.; Nguyen-Khac, E.; Lantz, O.; et al. Innate mucosal-associated invariant T (MAIT) cells are activated in inflammatory bowel diseases. Clin. Exp. Immunol. 2014, 176, 266–274. [Google Scholar] [PubMed]
- Chiba, A.; Tajima, R.; Tomi, C.; Miyazaki, Y.; Yamamura, T.; Miyake, S. Mucosal-associated invariant T cells promote inflammation and exacerbate disease in murine models of arthritis. Arthritis Rheum. 2012, 64, 153–161. [Google Scholar] [PubMed]
- Kenna, T.J.; Hanson, A.; Costello, M.E.; Brown, M.A. Functional genomics and its bench-to-bedside translation pertaining to the identified susceptibility alleles and loci in ankylosing spondylitis. Curr. Rheumatol. Rep. 2016, 18, 63. [Google Scholar] [CrossRef] [PubMed]
- Fragoulis, G.E.; Siebert, S.; McInnes, I.B. Therapeutic targeting of IL-17 and IL-23 cytokines in immune-mediated diseases. Annu. Rev. Med. 2016, 67, 337–353. [Google Scholar] [CrossRef] [PubMed]
- Cusick, M.F.; Libbey, J.E.; Fujinami, R.S. Molecular mimicry as a mechanism of autoimmune disease. Clin. Rev. Allergy Immunol. 2012, 42, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, J.T.; Lin, P.; Asquith, M. Does the microbiome cause B27-related acute anterior uveitis? Ocul. Immunol. Inflamm. 2016, 24, 440–444. [Google Scholar] [CrossRef] [PubMed]
- Burke, R.J.; Chang, C. Diagnostic criteria of acute rheumatic fever. Autoimmun. Rev. 2014, 13, 503–507. [Google Scholar] [CrossRef] [PubMed]
- De Vries, D.D.; Dekker-Saeys, A.J.; Gyodi, E.; Bohm, U.; Ivanyi, P. Absence of autoantibodies to peptides shared by HLA-B27.5 and Klebsiella pneumoniae nitrogenase in serum samples from HLA-B27 positive patients with ankylosing spondylitis and Reiter’s syndrome. Ann. Rheum. Dis. 1992, 51, 783–789. [Google Scholar] [CrossRef] [PubMed]
- Fielder, M.; Pirt, S.J.; Tarpey, I.; Wilson, C.; Cunningham, P.; Ettelaie, C.; Binder, A.; Bansal, S.; Ebringer, A. Molecular mimicry and ankylosing spondylitis: Possible role of a novel sequence in pullulanase of Klebsiella pneumoniae. FEBS Lett. 1995, 369, 243–248. [Google Scholar] [CrossRef]
- Welsh, J.; Avakian, H.; Cowling, P.; Ebringer, A.; Wooley, P.; Panayi, G.; Ebringer, R. Ankylosing spondylitis, HLA-B27 and Klebsiella. I. Cross-reactivity studies with rabbit antisera. Br. J. Exp. Pathol. 1980, 61, 85–91. [Google Scholar] [PubMed]
- Rashid, T.; Wilson, C.; Ebringer, A. The link between ankylosing spondylitis, Crohn’s disease, Klebsiella, and starch consumption. Clin. Dev. Immunol. 2013, 2013, 872632. [Google Scholar] [CrossRef] [PubMed]
- Cauli, A.; Dessole, G.; Vacca, A.; Porru, G.; Cappai, L.; Piga, M.; Bitti, P.P.; Fiorillo, M.T.; Sorrentino, R.; Carcassi, C.; et al. Susceptibility to ankylosing spondylitis but not disease outcome is influenced by the level of HLA-B27 expression, which shows moderate variability over time. Scand J. Rheumatol. 2012, 41, 214–218. [Google Scholar] [CrossRef] [PubMed]
- Rashid, T.; Ebringer, A. Gut-mediated and HLA-B27-associated arthritis: An emphasis on ankylosing spondylitis and Crohn’s disease with a proposal for the use of new treatment. Discov. Med. 2011, 12, 187–194. [Google Scholar] [PubMed]
- Moghimi, J.; Rezaei, A.A.; Ghorbani, R.; Razavi, M.R.; Pahlevan, D. Efficacy of an acquainted drug in the treatment of inflammatory low back pain: Sulfasalazine under investigation. Drug Des. Dev. Ther. 2016, 10, 3065–3069. [Google Scholar]
- Xiao, P.; Pang, C.; Zhu, X.; Wu, X. Clinical research for curing ankylosing spondylitis through combining etanercept, thalidomide and sulfasalazine. Pak. J. Pharm. Sci. 2015, 28, 359–362. [Google Scholar] [PubMed]
- Hendrix, S.; Koenig, A.; Li, W.; Singh, A. Disease-modifying effect of etanercept versus sulphasalazine on spinal mobility in patients with ankylosing spondylitis. J. Int. Med. Res. 2013, 41, 1606–1611. [Google Scholar] [CrossRef] [PubMed]
- Clegg, D.O.; Reda, D.J.; Abdellatif, M. Comparison of sulfasalazine and placebo for the treatment of axial and peripheral articular manifestations of the seronegative spondylarthropathies: A department of veterans affairs cooperative study. Arthritis Rheum. 1999, 42, 2325–2329. [Google Scholar] [CrossRef]
- Ogrendik, M. Treatment of ankylosing spondylitis with moxifloxacin. South Med. J. 2007, 100, 366–370. [Google Scholar] [CrossRef] [PubMed]
- Cock, I.E.; van Vuuren, S.F. The potential of selected South African plants with anti-Klebsiella activity for the treatment and prevention of ankylosing spondylitis. Inflammopharmacology 2015, 23, 21–35. [Google Scholar] [CrossRef] [PubMed]
- Alexandre, Y.; Le Blay, G.; Boisrame-Gastrin, S.; Le Gall, F.; Hery-Arnaud, G.; Gouriou, S.; Vallet, S.; Le Berre, R. Probiotics: A new way to fight bacterial pulmonary infections? Med. Mal. Infect. 2014, 44, 9–17. [Google Scholar] [CrossRef] [PubMed]
- Zorriehzahra, M.J.; Delshad, S.T.; Adel, M.; Tiwari, R.; Karthik, K.; Dhama, K.; Lazado, C.C. Probiotics as beneficial microbes in aquaculture: An update on their multiple modes of action: A review. Vet. Q. 2016, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Bertazzoni, E.; Donelli, G.; Midtvedt, T.; Nicoli, J.; Sanz, Y. Probiotics and clinical effects: Is the number what counts? J. Chemother. 2013, 25, 193–212. [Google Scholar] [CrossRef] [PubMed]
- Andrews, J.M.; Tan, M. Probiotics in luminal gastroenterology: The current state of play. Intern. Med. J. 2012, 42, 1287–1291. [Google Scholar] [CrossRef] [PubMed]
- Anilkumar, K.; Monisha, A.L. Role of friendly bacteria in oral health—A short review. Oral Health Prev. Dent. 2012, 10, 3–8. [Google Scholar] [PubMed]
- NeSmith, M.; Ahn, J.; Flamm, S.L. Contemporary understanding and management of overt and covert hepatic encephalopathy. Gastroenterol. Hepatol. 2016, 12, 91–100. [Google Scholar]
- Sanchez, B.; Delgado, S.; Blanco-Miguez, A.; Lourenco, A.; Gueimonde, M.; Margolles, A. Probiotics, gut microbiota, and their influence on host health and disease. Mol. Nutr. Food Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Koletzko, S. Probiotics and prebiotics for prevention of food allergy: Indications and recommendations by societies and institutions. J. Pediatr. Gastroenterol. Nutr. 2016, 63, S9–S10. [Google Scholar] [PubMed]
- Louis, P.; Flint, H.J.; Michel, C. How to manipulate the microbiota: Prebiotics. Adv. Exp. Med. Biol. 2016, 902, 119–142. [Google Scholar] [PubMed]
- Lichtenstein, L.; Avni-Biron, I.; Ben-Bassat, O. Probiotics and prebiotics in Crohn’s disease therapies. Best Pract. Res. Clin. Gastroenterol. 2016, 30, 81–88. [Google Scholar] [CrossRef] [PubMed]
- Dylag, K.; Hubalewska-Mazgaj, M.; Surmiak, M.; Szmyd, J.; Brzozowski, T. Probiotics in the mechanism of protection against gut inflammation and therapy of gastrointestinal disorders. Curr. Pharm. Des. 2014, 20, 1149–1155. [Google Scholar] [CrossRef] [PubMed]
- Tanriover, M.D.; Aksoy, D.Y.; Unal, S. Use of probiotics in various diseases: Evidence and promises. Pol. Arch. Med. Wewn. 2012, 122, 72–77. [Google Scholar] [PubMed]
- Amdekar, S.; Singh, V.; Singh, R.; Sharma, P.; Keshav, P.; Kumar, A. Lactobacillus casei reduces the inflammatory joint damage associated with collagen-induced arthritis (CIA) by reducing the pro-inflammatory cytokines: Lactobacillus casei: COX-2 inhibitor. J. Clin. Immunol. 2011, 31, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Dieleman, L.A.; Goerres, M.S.; Arends, A.; Sprengers, D.; Torrice, C.; Hoentjen, F.; Grenther, W.B.; Sartor, R.B. Lactobacillus GG prevents recurrence of colitis in HLA-B27 transgenic rats after antibiotic treatment. GUT 2003, 52, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Jenks, K.; Stebbings, S.; Burton, J.; Schultz, M.; Herbison, P.; Highton, J. Probiotic therapy for the treatment of spondyloarthritis: A randomized controlled trial. J. Rheumatol. 2010, 37, 2118–2125. [Google Scholar] [CrossRef] [PubMed]
- Brophy, S.; Burrows, C.L.; Brooks, C.; Gravenor, M.B.; Siebert, S.; Allen, S.J. Internet-based randomised controlled trials for the evaluation of complementary and alternative medicines: Probiotics in spondyloarthropathy. BMC Musculoskelet. Disord. 2008, 9, 4. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Scott, K.P.; Louis, P.; Duncan, S.H. The role of the gut microbiota in nutrition and health. Nat. Rev. Gastroenterol. Hepatol. 2012, 9, 577–589. [Google Scholar] [CrossRef] [PubMed]
- Brown, K.; DeCoffe, D.; Molcan, E.; Gibson, D.L. Diet-induced dysbiosis of the intestinal microbiota and the effects on immunity and disease. Nutrients 2012, 4, 1095–1119. [Google Scholar] [CrossRef] [PubMed]
- Van Laere, K.M.; Hartemink, R.; Bosveld, M.; Schols, H.A.; Voragen, A.G. Fermentation of plant cell wall derived polysaccharides and their corresponding oligosaccharides by intestinal bacteria. J. Agric. Food Chem. 2000, 48, 1644–1652. [Google Scholar] [CrossRef] [PubMed]
- Djuric, Z. Obesity-associated cancer risk: The role of intestinal microbiota in the etiology of the host proinflammatory state. Transl. Res. 2016. [Google Scholar] [CrossRef] [PubMed]
- Oh, B.R.; Seo, J.W.; Heo, S.Y.; Hong, W.K.; Luo, L.H.; Son, J.H.; Park, D.H.; Kim, C.H. Fermentation strategies for 1,3-propanediol production from glycerol using a genetically engineered Klebsiella pneumoniae strain to eliminate by-product formation. Bioprocess Biosyst. Eng. 2012, 35, 159–165. [Google Scholar] [CrossRef] [PubMed]
- Ebringer, A.; Baines, M.; Childerstone, M. Etiopathogenesis of ankylosing spondylitis and the cross-tolerance hypothesis. In Advances in Inflammation Research—The Spondyloarthropathies; Ziff, M., Cohen, S.B., Eds.; Raven Press: New York, NY, USA, 1985; pp. 101–128. [Google Scholar]
- Rashid, T.; Wilson, C.; Ebringer, A. Raised incidence of ankylosing spondylitis among Inuit populations could be due to high HLA-B27 association and starch consumption. Rheumatol. Int. 2015, 35, 945–951. [Google Scholar] [CrossRef] [PubMed]
- Fuentes, S.; de Vos, W.M. How to manipulate the microbiota: Fecal microbiota transplantation. Adv. Exp. Med. Biol. 2016, 902, 143–153. [Google Scholar] [PubMed]
- Sturdik, I.; Hlavaty, T.; Payer, J. Fecal microbiota transplantation. Vnitr. Lek. 2016, 62, 147–151. [Google Scholar] [PubMed]
- Pamer, E.G. Fecal microbiota transplantation: Effectiveness, complexities, and lingering concerns. Mucosal. Immunol. 2014, 7, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Zhu, A.; Benes, V.; Costea, P.I.; Hercog, R.; Hildebrand, F.; Huerta-Cepas, J.; Nieuwdorp, M.; Salojarvi, J.; Voigt, A.Y.; et al. Durable coexistence of donor and recipient strains after fecal microbiota transplantation. Science 2016, 352, 586–589. [Google Scholar] [CrossRef] [PubMed]
- Xu, M.Q.; Cao, H.L.; Wang, W.Q.; Wang, S.; Cao, X.C.; Yan, F.; Wang, B.M. Fecal microbiota transplantation broadening its application beyond intestinal disorders. World J. Gastroenterol. 2015, 21, 102–111. [Google Scholar] [CrossRef] [PubMed]
- Millan, B.; Park, H.; Hotte, N.; Mathieu, O.; Burguiere, P.; Tompkins, T.A.; Kao, D.; Madsen, K.L. Fecal microbial transplants reduce antibiotic-resistant genes in patients with recurrent clostridium difficile infection. Clin. Infect. Dis. 2016, 62, 1479–1486. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Garg, S.; Girotra, M.; Maddox, C.; von Rosenvinge, E.C.; Dutta, A.; Dutta, S.; Fricke, W.F. Microbiota dynamics in patients treated with fecal microbiota transplantation for recurrent Clostridium difficile infection. PLoS ONE 2013, 8, e81330. [Google Scholar] [CrossRef] [PubMed]
- Di Luccia, B.; Crescenzo, R.; Mazzoli, A.; Cigliano, L.; Venditti, P.; Walser, J.C.; Widmer, A.; Baccigalupi, L.; Ricca, E.; Iossa, S. Rescue of fructose-induced metabolic syndrome by antibiotics or fecal transplantation in a rat model of obesity. PLoS ONE 2015, 10, e134893. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, C.S. Microbiome disturbances and autism spectrum disorders. Drug Metab. Dispos. 2015, 43, 1557–1571. [Google Scholar] [CrossRef] [PubMed]
- Cui, B.; Li, P.; Xu, L.; Peng, Z.; Xiang, J.; He, Z.; Zhang, T.; Ji, G.; Nie, Y.; Wu, K.; et al. Step-up fecal microbiota transplantation (FMT) strategy. Gut Microbes 2016, 7, 323–328. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Allen-Vercoe, E.; Petrof, E.O. Fecal microbiota transplantation: In perspective. Ther. Adv. Gastroenterol. 2016, 9, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Vaughn, B.P.; Vatanen, T.; Allegretti, J.R.; Bai, A.; Xavier, R.J.; Korzenik, J.; Gevers, D.; Ting, A.; Robson, S.C.; Moss, A.C. Increased intestinal microbial diversity following fecal microbiota transplant for active Crohn’s disease. Inflamm. Bowel Dis. 2016, 22, 2182–2190. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.L.; Edney, R.J.; Whelan, K. Systematic review: Fecal microbiota transplantation in the management of inflammatory bowel disease. Aliment. Pharmacol. Ther. 2012, 36, 503–516. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Zhu, W.; Gong, J.; Guo, D.; Gu, L.; Li, N.; Li, J. Fecal microbiota transplantation improves the quality of life in patients with inflammatory bowel disease. Gastroenterol. Res. Pract. 2015, 2015, 517597. [Google Scholar] [CrossRef] [PubMed]
- Rossen, N.G.; Fuentes, S.; van der Spek, M.J.; Tijssen, J.G.; Hartman, J.H.; Duflou, A.; Lowenberg, M.; van den Brink, G.R.; Mathus-Vliegen, E.M.; de Vos, W.M.; et al. Findings from a randomized controlled trial of fecal transplantation for patients with ulcerative colitis. Gastroenterology 2015, 149, 110–118. [Google Scholar] [CrossRef] [PubMed]
- Petrof, E.O.; Claud, E.C.; Gloor, G.B.; Allen-Vercoe, E. Microbial ecosystems therapeutics: A new paradigm in medicine? Benef. Microbes 2013, 4, 53–65. [Google Scholar] [CrossRef] [PubMed]
- Dave, M.; Higgins, P.D.; Middha, S.; Rioux, K.P. The human gut microbioma current knowledge, challenges, and future directions. Transl. Res. 2012, 160, 246–257. [Google Scholar] [CrossRef] [PubMed]
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, L.; Wang, L.; Wang, X.; Xian, C.J.; Lu, H. A Possible Role of Intestinal Microbiota in the Pathogenesis of Ankylosing Spondylitis. Int. J. Mol. Sci. 2016, 17, 2126. https://doi.org/10.3390/ijms17122126
Yang L, Wang L, Wang X, Xian CJ, Lu H. A Possible Role of Intestinal Microbiota in the Pathogenesis of Ankylosing Spondylitis. International Journal of Molecular Sciences. 2016; 17(12):2126. https://doi.org/10.3390/ijms17122126
Chicago/Turabian StyleYang, Lianjun, Liping Wang, Xin Wang, Cory J. Xian, and Hai Lu. 2016. "A Possible Role of Intestinal Microbiota in the Pathogenesis of Ankylosing Spondylitis" International Journal of Molecular Sciences 17, no. 12: 2126. https://doi.org/10.3390/ijms17122126
APA StyleYang, L., Wang, L., Wang, X., Xian, C. J., & Lu, H. (2016). A Possible Role of Intestinal Microbiota in the Pathogenesis of Ankylosing Spondylitis. International Journal of Molecular Sciences, 17(12), 2126. https://doi.org/10.3390/ijms17122126






