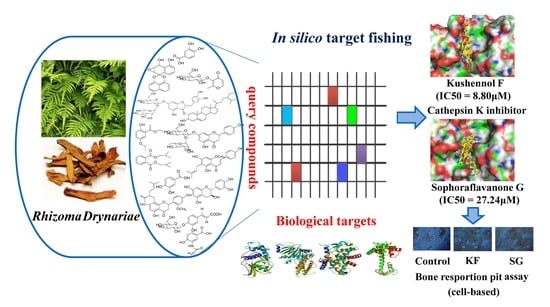
Discovery of a New Class of Cathepsin K Inhibitors in Rhizoma Drynariae as Potential Candidates for the Treatment of Osteoporosis
Abstract
:1. Introduction
2. Results
2.1. In Silico Target Fishing
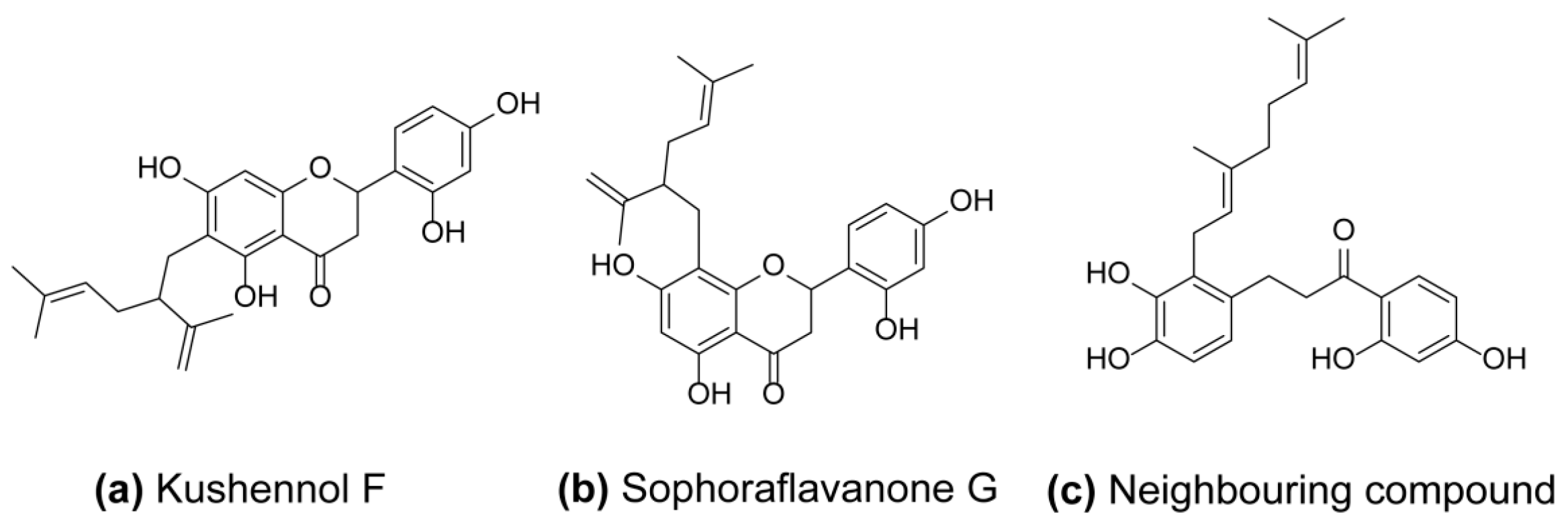
2.2. Kushennol F (KF) and Sophoraflavone G (SG) Directly Inhibited the Proteolytic Activity of Cathepsin K
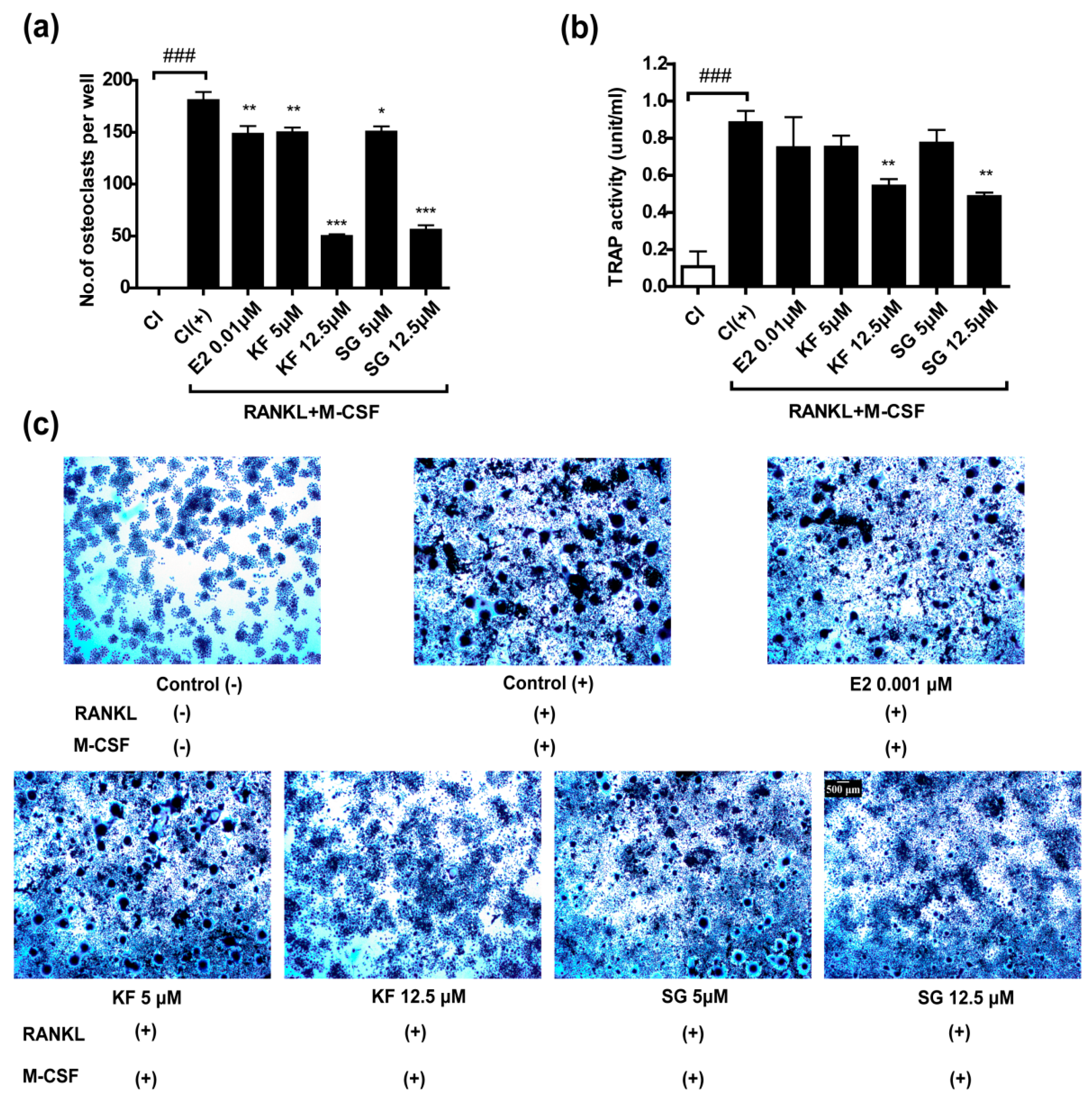
2.3. Kushennol F and Sophoraflavone G Suppressed RANKL-induced Osteoclastogenesis of RAW264.7
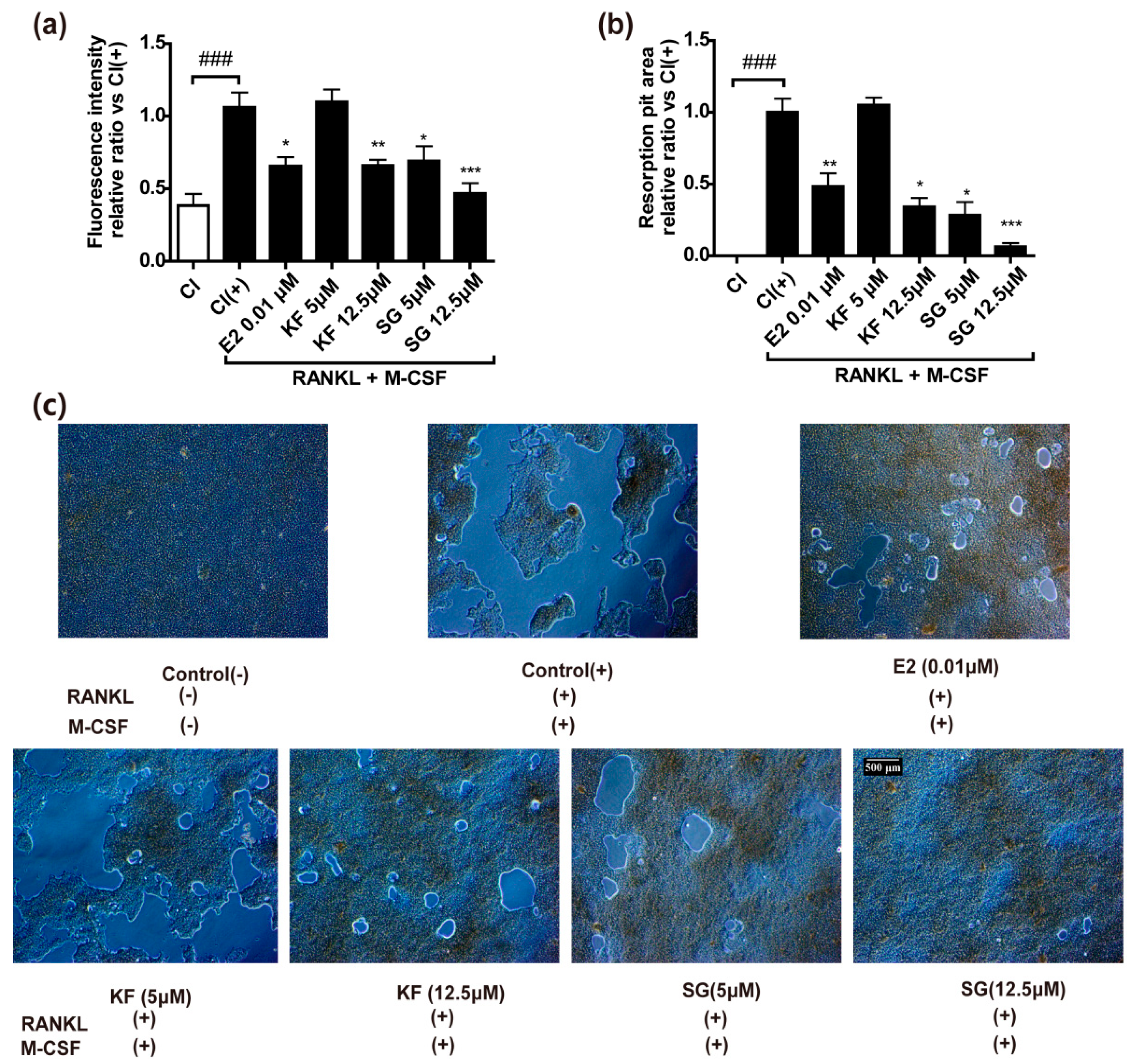
2.4. Kushennol F and Sophoraflavone G Significantly Inhibited Bone Resorption Pit Formation
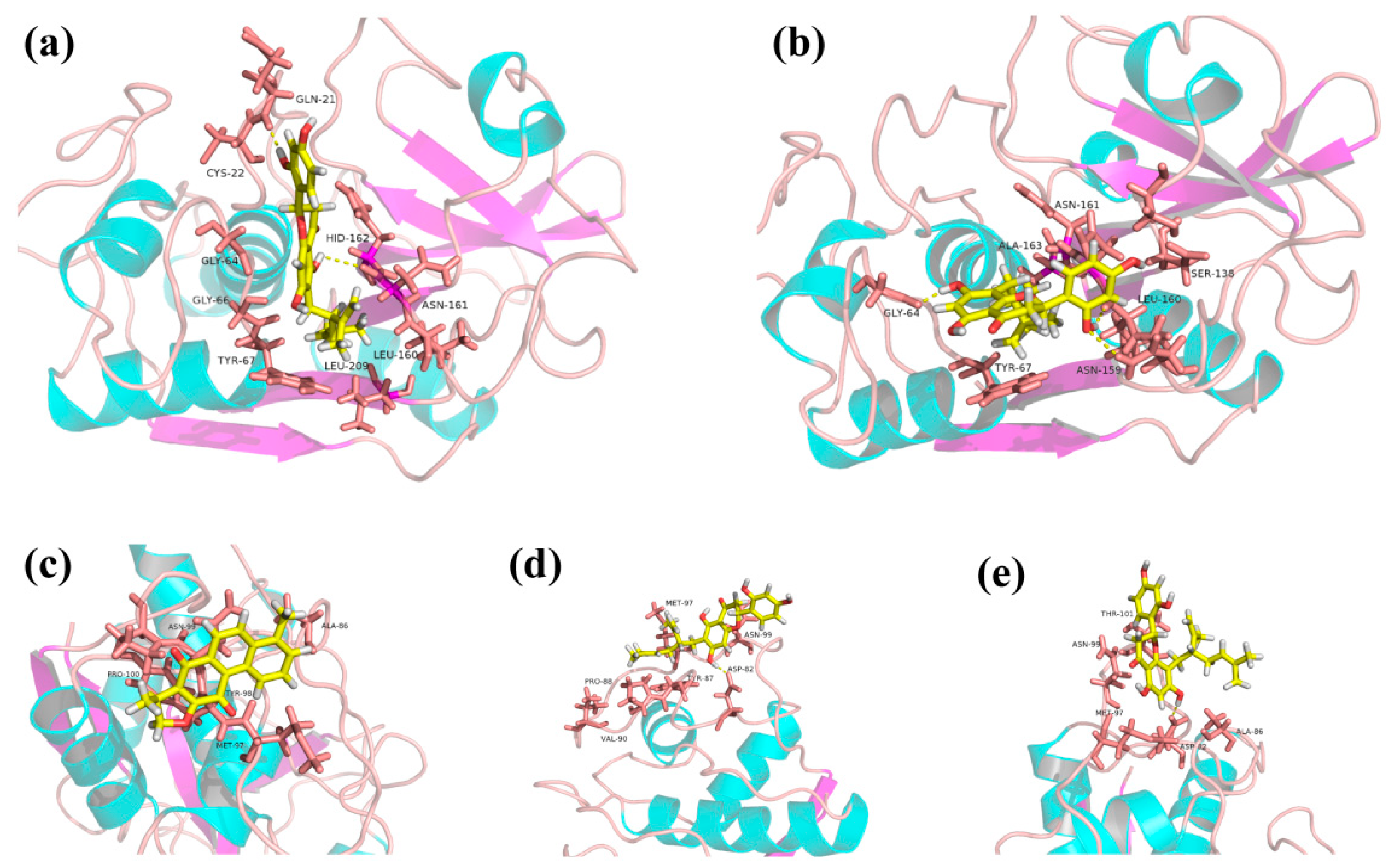
2.5. Molecular Docking Analysis
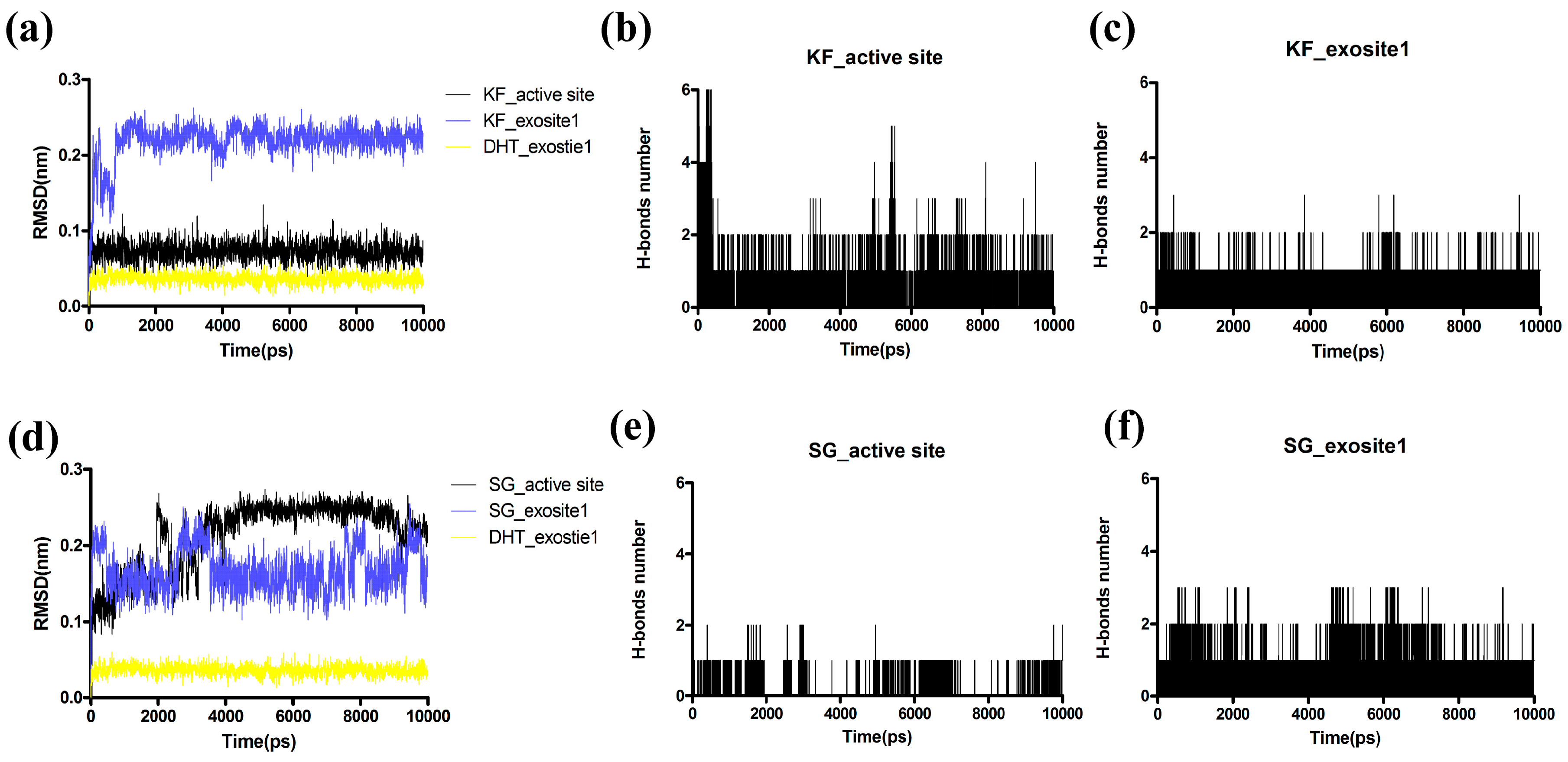
2.6. Molecular Dynamic(MD) Simulations
3. Discussion
4. Materials and Methods
4.1. The Establishment of RD Ingredients Database
4.2. In Silico Target Fishing
4.3. Experimental Validation
4.3.1. Chemicals and Reagents
4.3.2. Cathepsin K Inhibition Activity Assay
4.3.3. RAW264.7 Cell Culture
4.3.4. Cell Cytotoxicity Assay
4.3.5. TRAP Positive Multinucleated Cells Staining
4.3.6. TRAP Activity Measurement
4.3.7. Pit Formation Assay
4.4. Docking Analysis
4.5. Molecular Dynamics
4.6. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| Ctsk | Cathepsin K |
| RD | Rhizoma Drynariae |
| KF | Kushennol F |
| SG | Sophoraflavanone G |
| TCM | Traditional Chinese Medicine |
| MNCs | Multi-nucleated cells |
| TRAP | Tartrate-resistant acid phosphatase |
| MD | Molecular Dynamics |
References
- Rachner, T.D.; Khosla, S.; Hofbauer, L.C. Osteoporosis: Now and the future. Lancet 2011, 377, 1276–1287. [Google Scholar] [CrossRef]
- Makras, P.; Delaroudis, S.; Anastasilakis, A.D. Novel therapies for osteoporosis. Metab.: Clin. Exp. 2015, 64, 1199–1214. [Google Scholar] [CrossRef] [PubMed]
- Appelman-Dijkstra, N.M.; Papapoulos, S.E. Modulating bone resorption and bone formation in opposite directions in the treatment of postmenopausal osteoporosis. Drugs 2015, 75, 1049–1058. [Google Scholar] [CrossRef] [PubMed]
- Komm, B.S.; Chines, A.A. An update on selective estrogen receptor modulators for the prevention and treatment of osteoporosis. Maturitas 2012, 71, 221–226. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.Q.; Li, J.L.; Sun, Y.L.; Yao, M.; Gao, J.; Yang, Z.; Shi, Q.; Cui, X.J.; Wang, Y.J. Chinese herbal medicine for osteoporosis: A systematic review of randomized controlled trails. Evid.-Based Complement. Altern. Med. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Mukwaya, E.; Xu, F.; Wong, M.S.; Zhang, Y. Chinese herbal medicine for bone health. Pharm. Biol. 2014, 52, 1223–1228. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.C.; Lee, B.T.; Yoon, C.H.; Kim, H.M.; Kim, C.H. Effects of drynariae rhizoma on the proliferation of human bone cells and the immunomodulatory activity. Pharmacol. Res. 2005, 51, 125–136. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Lei, L.H.; Ding, P.H.; Tang, Q.; Wu, Y.M. Osteogenic effect of drynariae rhizoma extracts and naringin on mc3t3-e1 cells and an induced rat alveolar bone resorption model. Arch. Oral Biol. 2011, 56, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.-S.; Lin, C.-Y.; Dong, G.-C.; Sheu, S.-Y.; Lin, F.-H.; Chen, L.-T.; Wang, Y.-J. The effect of Gu-Sui-Bu (Drynaria fortunei J. Sm) on bone cell activities. Biomaterials 2002, 23, 3377–3385. [Google Scholar] [CrossRef]
- Jeong, J.C.; Lee, J.W.; Yoon, C.H.; Kim, H.M.; Kim, C.H. Drynariae Rhizoma promotes osteoblast differentiation and mineralization in MC3T3-E1 cells through regulation of bone morphogenetic protein-2, alkaline phosphatase, type I collagen and collagenase-1. Toxicol. In Vitro 2004, 18, 829–834. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-L.; Wang, N.-L.; Zhang, Y.; Gao, H.; Pang, W.-Y.; Wong, M.-S.; Zhang, G.; Qin, L.; Yao, X.-S. Effects of eleven flavonoids from the osteoprotective fraction of Drynaria fortunei (KUNZE) J. Sm. On osteoblastic proliferation using an osteoblast-like cell line. Chem. Pharm. Bull. 2008, 56, 46–51. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.L.; Wang, N.L.; Gao, H.; Zhang, G.; Qin, L.; Wong, M.S.; Yao, X.S. Phenylpropanoid and flavonoids from osteoprotective fraction of Drynaria fortunei. Nat. Prod. Res. 2010, 24, 1206–1213. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Zhen, L.; Zhang, G.; Wong, M.S.; Qin, L.; Yao, X. Osteogenic effects of flavonoid aglycones from an osteoprotective fraction of Drynaria fortunei—An in vitro efficacy study. Phytomedicine 2011, 18, 868–872. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Wang, X.-L.; Wang, N.L.; Yao, X.-S. Chemical constituents from Drynaria fortunei. Chin. J. Med. Chem. 2008, 18, 284–287. [Google Scholar]
- Wu, X.; Zhao, Y.M. Isolation and identification of chemical compounds from Drynaria fortunei. China J. Chin. Mater. Med. 2005, 30, 443–444. [Google Scholar]
- Jeong, J.C.; Kang, S.K.; Youn, C.H.; Jeong, C.W.; Kim, H.M.; Lee, Y.C.; Chang, Y.C.; Kim, C.H. Inhibition of drynariae rhizoma extracts on bone resorption mediated by processing of cathepsin K in cultured mouse osteoclasts. Int. Immunopharmacol. 2003, 3, 1685–1697. [Google Scholar] [CrossRef] [PubMed]
- Guo, D.; Wang, J.; Wang, X.; Luo, H.; Zhang, H.; Cao, D.; Chen, L.; Huang, N. Double directional adjusting estrogenic effect of naringin from Rhizoma Drynariae (Gusuibu). J. Ethnopharmacol. 2011, 138, 451–457. [Google Scholar] [CrossRef] [PubMed]
- Engin, H.B.; Gursoy, A.; Nussinov, R.; Keskin, O. Network-based strategies can help mono- and poly-pharmacology drug discovery: A systems biology view. Curr. Pharm. Des. 2014, 20, 1–7. [Google Scholar] [CrossRef]
- Hopkins, A.L. Network pharmacology: The next paradigm in drug discovery. Nat. Chem. Biol. 2008, 4, 682–690. [Google Scholar] [CrossRef] [PubMed]
- Iyengar, A.D.; Iyengar, R. Systems approaches to polypharmacology and drug discovery. Curr. Opin. Drug Discov. Dev. 2011, 13, 297–309. [Google Scholar]
- Koutsoukas, A.; Simms, B.; Kirchmair, J.; Bond, P.J.; Whitmore, A.V.; Zimmer, S.; Young, M.P.; Jenkins, J.L.; Glick, M.; Glen, R.C.; et al. From in silico target prediction to multi-target drug design: Current databases, methods and applications. J. Proteom. 2011, 74, 2554–2574. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, J.L.; Bender, A.; Davies, J.W. In silico target fishing: Predicting biological targets from chemical structure. Drug Discov. Today: Technol. 2006, 3, 413–421. [Google Scholar] [CrossRef]
- Cereto-Massague, A.; Ojeda, M.J.; Valls, C.; Mulero, M.; Pujadas, G.; Garcia-Vallve, S. Tools for in silico target fishing. Methods 2015, 71, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Lounkine, E.; Keiser, M.J.; Whitebread, S.; Mikhailov, D.; Hamon, J.; Jenkins, J.L.; Lavan, P.; Weber, E.; Doak, A.K.; Cote, S.; et al. Large-scale prediction and testing of drug activity on side-effect targets. Nature 2012, 486, 361–367. [Google Scholar] [CrossRef] [PubMed]
- Muller, P.; Lena, G.; Boilard, E.; Bezzine, S.; Lambeau, G.; Guichard, G.; Rognan, D. In silico-guided target identification of a scaffold-focused library: 1,3,5-triazepan-2,6-diones as novel phospholipase A2 inhibitors. J. Med. Chem. 2006, 49, 6768–6778. [Google Scholar] [CrossRef] [PubMed]
- Vidal, D.; Garcia-Serna, R.; Mestres, J. Ligand-based approaches to in silico pharmacology. Methods Mol. Biol. 2011, 672, 489–502. [Google Scholar] [PubMed]
- Sharma, V.; Panwar, P.; O’Donoghue, A.J.; Cui, H.; Guido, R.V.; Craik, C.S.; Bromme, D. Structural requirements for the collagenase and elastase activity of cathepsin K and its selective inhibition by an exosite inhibitor. Biochem. J. 2015, 465, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Gelb, B.D.; Shi, G.-P.; Chapman, H.A.; Desnick, R.J. Pycnodysostosis, a lysosomal disease caused by cathepsin K deficiency. Science 1996, 273, 1236–1238. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, Y.; Kaleta, J.; Bromme, D. The role of cathepsins in osteoporosis and arthritis: Rationale for the design of new therapeutics. Adv. Drug Deliv. Rev. 2005, 57, 973–993. [Google Scholar] [CrossRef] [PubMed]
- Drake, F.H.; Dodds, R.A.; James, I.E.; Connor, J.R.; Debouck, C.; Richardson, S.; Lee-Rykaczewski, E.; Coleman, L.; Rieman, D.; Barthlow, R.; et al. Cathepsin K, but not Cathepsins B, L, or S, is abundantly expressed in human osteoclasts. J. Biol. Chem. 1996, 271, 12511–12516. [Google Scholar] [CrossRef] [PubMed]
- Boonen, S.; Rosenberg, E.; Claessens, F.; Vanderschueren, D.; Papapoulos, S. Inhibition of cathepsin K for treatment of osteoporosis. Curr. Osteoporos. Rep. 2012, 10, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Stoch, S.A.; Zajic, S.; Stone, J.A.; Miller, D.L.; van Bortel, L.; Lasseter, K.C.; Pramanik, B.; Cilissen, C.; Liu, Q.; Liu, L.; et al. Odanacatib, a selective cathepsin K inhibitor to treat osteoporosis: Safety, tolerability, pharmacokinetics and pharmacodynamics—Results from single oral dose studies in healthy volunteers. Br. J. Clin. Pharmacol. 2013, 75, 1240–1254. [Google Scholar] [CrossRef] [PubMed]
- Eastell, R.; Nagase, S.; Ohyama, M.; Small, M.; Sawyer, J.; Boonen, S.; Spector, T.; Kuwayama, T.; Deacon, S. Safety and efficacy of the cathepsin k inhibitor ono-5334 in postmenopausal osteoporosis: The ocean study. J. Bone Miner. Res. 2011, 26, 1303–1312. [Google Scholar] [CrossRef] [PubMed]
- Jerome, C.; Missbach, M.; Gamse, R. Balicatib, a cathepsin K inhibitor, stimulates periosteal bone formation in monkeys. Osteoporos. Int. 2011, 22, 3001–3011. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.; Dare, L.; Vasko-Moser, J.A.; James, I.E.; Blake, S.M.; Rickard, D.J.; Hwang, S.M.; Tomaszek, T.; Yamashita, D.S.; Marquis, R.W.; et al. A highly potent inhibitor of cathepsin K (relacatib) reduces biomarkers of bone resorption both in vitro and in an acute model of elevated bone turnover in vivo in monkeys. Bone 2007, 40, 122–131. [Google Scholar] [CrossRef] [PubMed]
- Duong le, T.; Leung, A.T.; Langdahl, B. Cathepsin K inhibition: A new mechanism for the treatment of osteoporosis. Calcif. Tissue Int. 2016, 98, 381–397. [Google Scholar] [CrossRef] [PubMed]
- Mullard, A. Merck & Co. Drops osteoporosis drug odanacatib. Nat. Rev. Drug Discov. 2016, 15, 669. [Google Scholar] [PubMed]
- Zhao, P.; Zhang, Y.-J.; Hirobumi, Y.; Yang, C.-R. Recebt advance on the chemistry, bioactivity and biosynthesis of prenylated flavonoids from sophora flavecens. Nat. Prod. Res. Dev. 2004, 16, 172–177. [Google Scholar]
- Patil, A.D.; Freyer, A.J.; Killmer, L.; Offen, P.; Taylor, P.B.; Votta, B.J.; Johnson, R.K. A new dimeric dihydrochalcone and a new prenylated flavone from the bud covers of artocarpus altilis: Potent inhibitors of cathepsin K. J. Nat. Prod. 2002, 65, 624–627. [Google Scholar] [CrossRef] [PubMed]
- Bromme, D.; Lecaille, F. Cathepsin k inhibitors for osteoporosis and potential off-target effects. Expert Opin. Investig. Drugs 2009, 18, 585–600. [Google Scholar] [CrossRef] [PubMed]
- Li, C.S.; Deschenes, D.; Desmarais, S.; Falgueyret, J.P.; Gauthier, J.Y.; Kimmel, D.B.; Leger, S.; Masse, F.; McGrath, M.E.; McKay, D.J.; et al. Identification of a potent and selective non-basic cathepsin k inhibitor. Bioorg. Med. Chem. Lett. 2006, 16, 1985–1989. [Google Scholar] [CrossRef] [PubMed]
- Fonovic, M.; Turk, B. Cysteine cathepsins and their potential in clinical therapy and biomarker discovery. Proteom. Clin. Appl. 2014, 8, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Shevde, N.K.; Bendixen, A.C.; Dienger, K.M.; Pike, J.W. Estrogens suppress rank ligand-induced osteoclast differentiation via a stromal cell independent mechanism involving C-Jun repression. Proc. Natl. Acad. Sci. USA 2000, 97, 7829–7834. [Google Scholar] [CrossRef] [PubMed]
- Lindberg, M.K.; Erlandsson, M.; Alatalo, S.L.; Windahl, S.; Andersson, G.; Halleen, J.M.; Carlsten, H.; Gustafsson, J.A.; Ohlsson, C. Estrogen receptor α, but not estrogen receptor β, is involved in the regulation of the OPG/RANKL (osteoprotegerin/receptor activator of NF-κB ligand) ratio and serum interleukin-6 in male mice. J. Endocrinol. 2001, 171, 425–433. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Imai, Y.; Matsumoto, T.; Sato, S.; Takeuchi, K.; Igarashi, K.; Harada, Y.; Azuma, Y.; Krust, A.; Yamamoto, Y.; et al. Estrogen prevents bone loss via estrogen receptor α and induction of fas ligand in osteoclasts. Cell 2007, 130, 811–823. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kim, D.H.; Ahn, H.N.; Song, Y.S.; Lee, Y.J.; Ryu, J.H. Activation of estrogen receptor by bavachin from psoralea corylifolia. Biomol. Ther. 2012, 20, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Nomura, T.; Fukai, T.; Akiyama, T. Chemistry of phenolic compounds of licorice (Glycyrrhiza species) and their estrogenic and cytotoxic activities. Pure Appl. Chem. 2002, 74, 1199–1206. [Google Scholar] [CrossRef]
- Lim, S.H.; Ha, T.Y.; Ahn, J.; Kim, S. Estrogenic activities of psoralea corylifolia l. Seed extracts and main constituents. Phytomedicine 2011, 18, 425–430. [Google Scholar] [CrossRef] [PubMed]
- Mestres, J.; Martín-Couce, L.; Gregori-Puigjané, E.; Cases, M.; Boyer, S. Ligand-based approach to in silico pharmacology: Nuclear receptor profiling. J. Chem. Inf. Model. 2006, 46, 2725–2736. [Google Scholar] [CrossRef] [PubMed]
- Vidal, D.; Mestres, J. In silico receptorome screening of antipsychotic drugs. Mol. Inform. 2010, 29, 543–551. [Google Scholar] [CrossRef] [PubMed]
- Antolin, A.A.; Jalencas, X.; Yelamos, J.; Mestres, J. Identification of pim kinases as novel targets for pj34 with confounding effects in parp biology. ACS Chem. Biol. 2012, 7, 1962–1967. [Google Scholar] [CrossRef] [PubMed]
- Spitzmuller, A.; Mestres, J. Prediction of the p. Falciparum target space relevant to malaria drug discovery. PLoS Comput. Biol. 2013, 9, e1003257. [Google Scholar]
- Montolio, M.; Gregori-Puigjane, E.; Pineda, D.; Mestres, J.; Navarro, P. Identification of small molecule inhibitors of amyloid beta-induced neuronal apoptosis acting through the imidazoline i(2) receptor. J. Med. Chem. 2012, 55, 9838–9846. [Google Scholar] [CrossRef] [PubMed]
- Weninger, D. Smiles, a chemical language and information system. 1. Introduction to methodology and encoding rules. J. Chem. Inj. Comput. Sci. 1988, 28, 31–36. [Google Scholar] [CrossRef]
- Pronk, S.; Pall, S.; Schulz, R.; Larsson, P.; Bjelkmar, P.; Apostolov, R.; Shirts, M.R.; Smith, J.C.; Kasson, P.M.; van der Spoel, D.; et al. Gromacs 4.5: A high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 2013, 29, 845–854. [Google Scholar] [CrossRef] [PubMed]



| Parameters | Compound Identifier | |
|---|---|---|
| Kushennol F | Sophoraflavanone G | |
| ANN | PRD | -- |
| pIC50 | 6.8 | -- |
| REF_NN | FVNFXIPJDHVJGE-REZTVBANSA-N | -- |
| SIM | 0.841 | 0.838 |
| REF_pACT | 6.77 | -- |
| SOURCE_DB | ChemblDB, PubChem, Binding DB | -- |
| UNIPROT | P43235 | -- |
| TARGET NAME | Cathepsin K | -- |
| FUNCTIONAL | EC, FD, PS | -- |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qiu, Z.-C.; Dong, X.-L.; Dai, Y.; Xiao, G.-K.; Wang, X.-L.; Wong, K.-C.; Wong, M.-S.; Yao, X.-S. Discovery of a New Class of Cathepsin K Inhibitors in Rhizoma Drynariae as Potential Candidates for the Treatment of Osteoporosis. Int. J. Mol. Sci. 2016, 17, 2116. https://doi.org/10.3390/ijms17122116
Qiu Z-C, Dong X-L, Dai Y, Xiao G-K, Wang X-L, Wong K-C, Wong M-S, Yao X-S. Discovery of a New Class of Cathepsin K Inhibitors in Rhizoma Drynariae as Potential Candidates for the Treatment of Osteoporosis. International Journal of Molecular Sciences. 2016; 17(12):2116. https://doi.org/10.3390/ijms17122116
Chicago/Turabian StyleQiu, Zuo-Cheng, Xiao-Li Dong, Yi Dai, Gao-Keng Xiao, Xin-Luan Wang, Ka-Chun Wong, Man-Sau Wong, and Xin-Sheng Yao. 2016. "Discovery of a New Class of Cathepsin K Inhibitors in Rhizoma Drynariae as Potential Candidates for the Treatment of Osteoporosis" International Journal of Molecular Sciences 17, no. 12: 2116. https://doi.org/10.3390/ijms17122116
APA StyleQiu, Z.-C., Dong, X.-L., Dai, Y., Xiao, G.-K., Wang, X.-L., Wong, K.-C., Wong, M.-S., & Yao, X.-S. (2016). Discovery of a New Class of Cathepsin K Inhibitors in Rhizoma Drynariae as Potential Candidates for the Treatment of Osteoporosis. International Journal of Molecular Sciences, 17(12), 2116. https://doi.org/10.3390/ijms17122116