Autoimmune Regulator Expression in DC2.4 Cells Regulates the NF-κB Signaling and Cytokine Expression of the Toll-Like Receptor 3 Pathway
Abstract
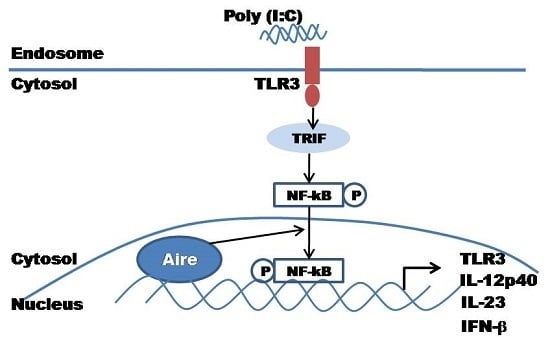
:1. Introduction
2. Results
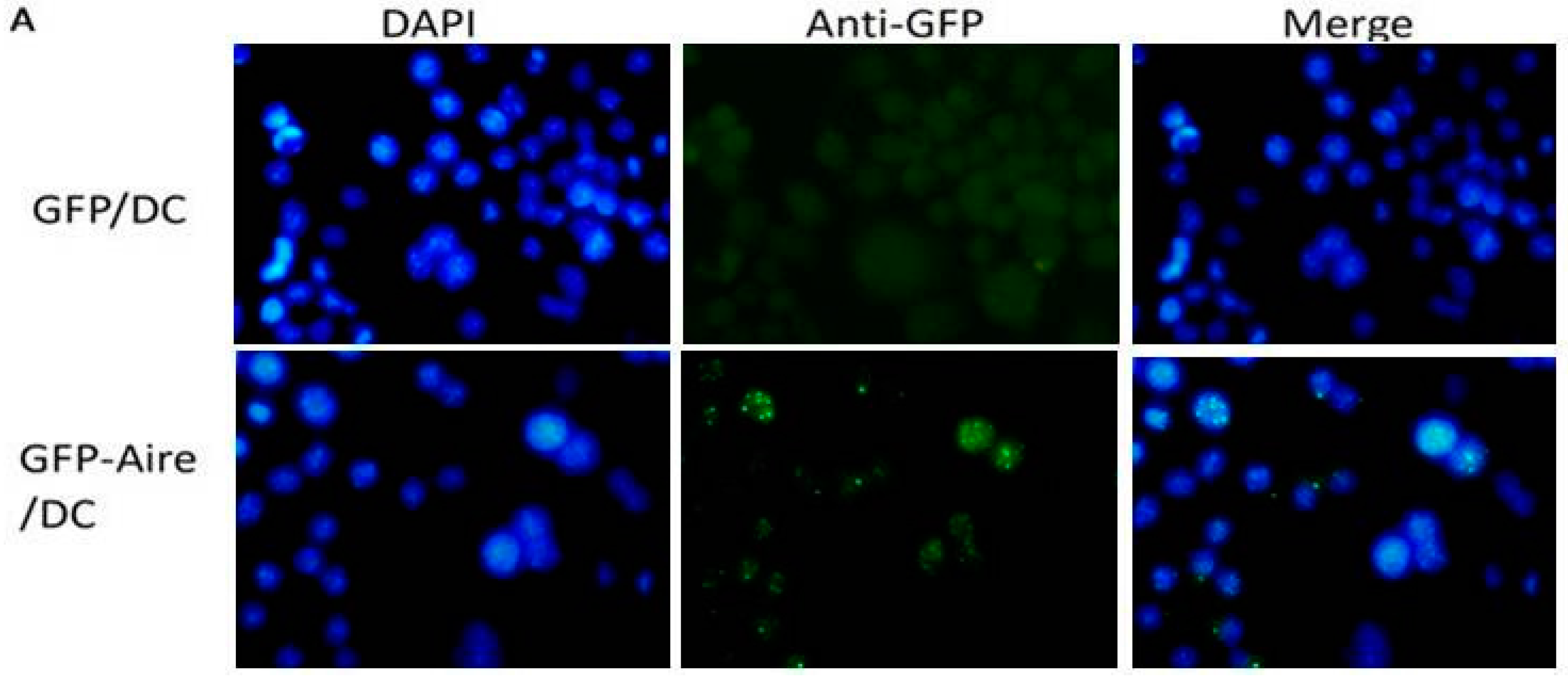
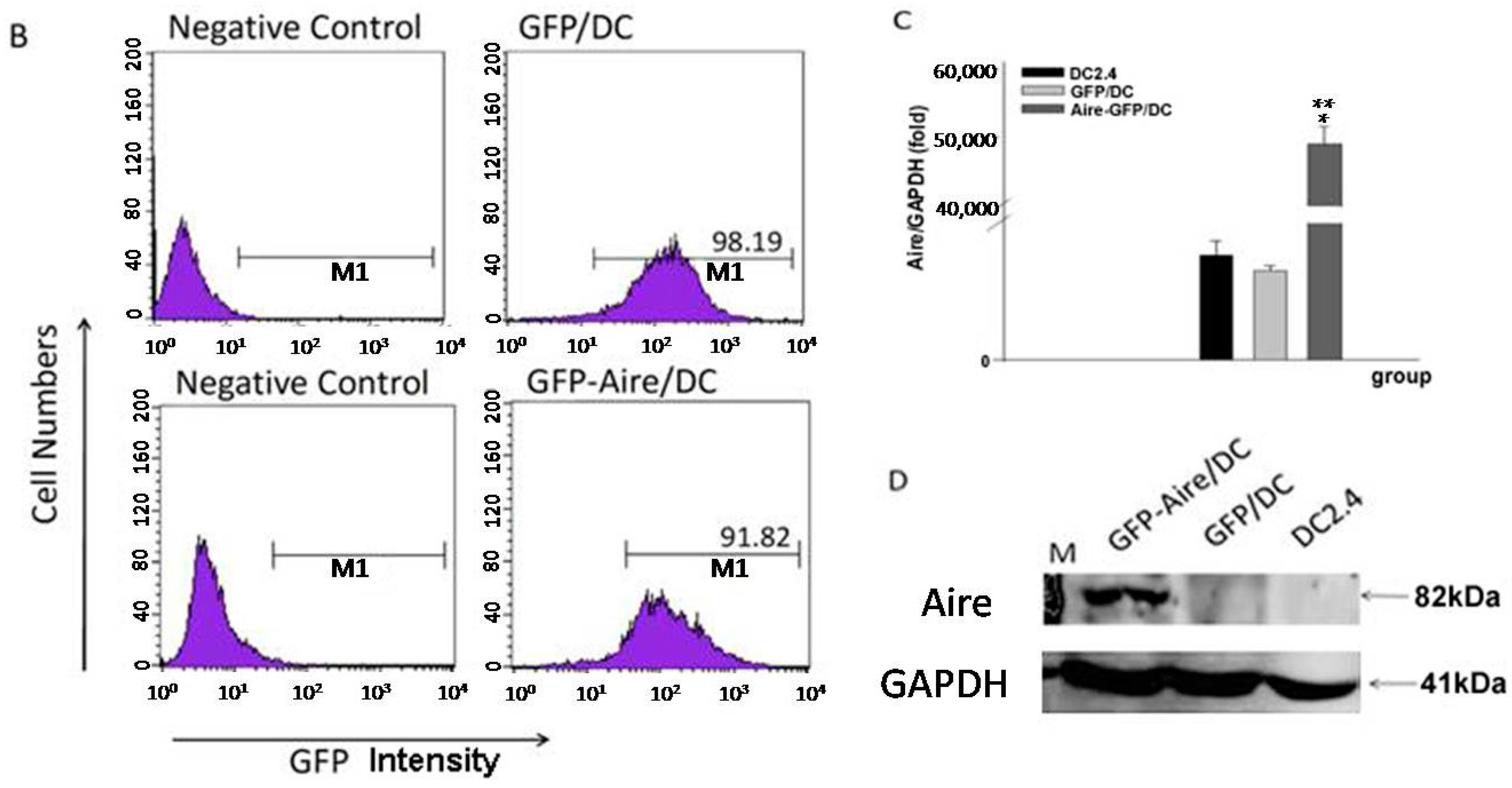
2.1. Establishment of Stable Aire-Expressing Transfectants (Stable Aire Expression in DC2.4 Cells)
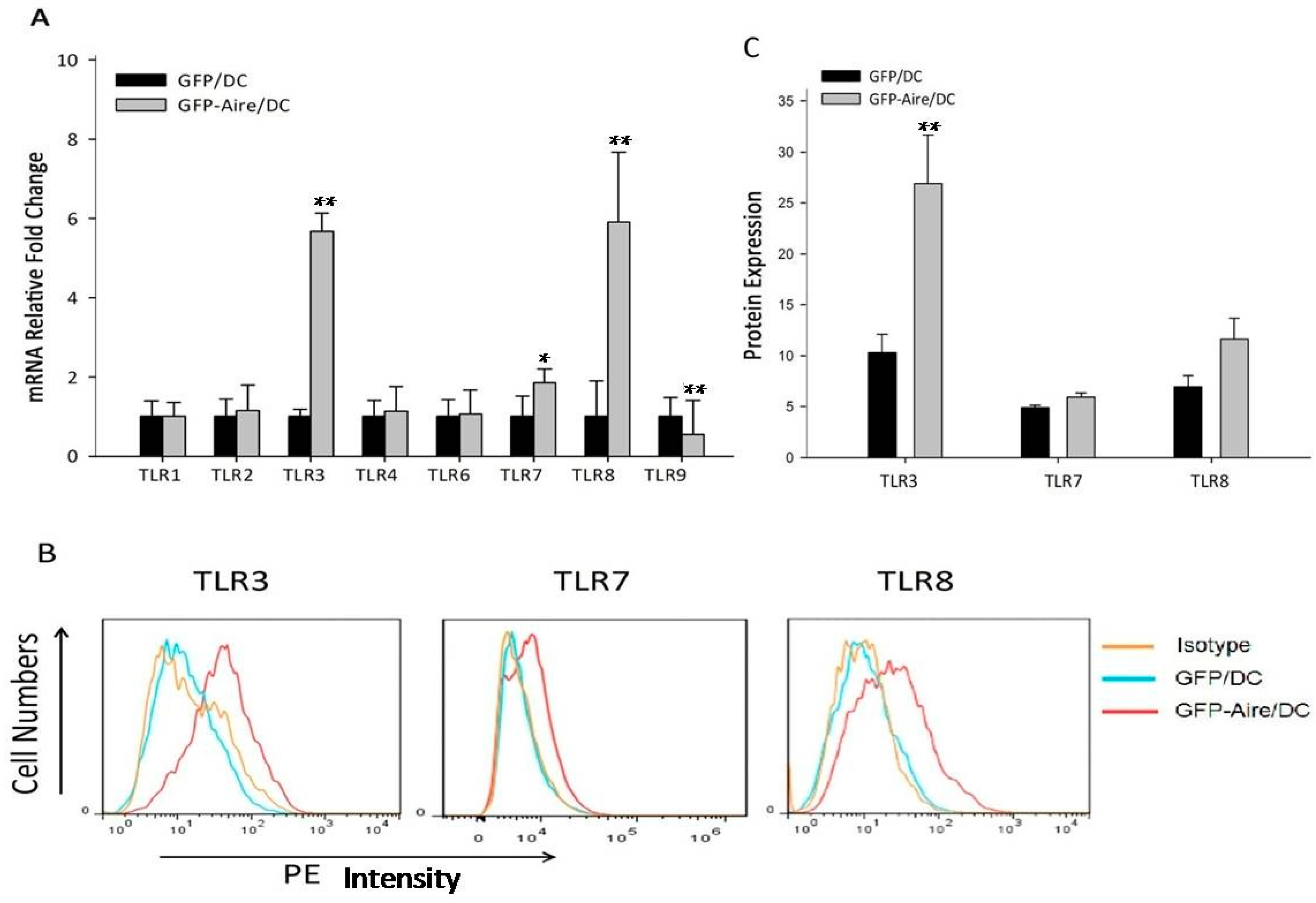
2.2. Aire Regulates TLR Expression in Stable Aire-Expressing DC2.4 Cells
2.3. Aire Influences the Phenotype of DC2.4 Cells
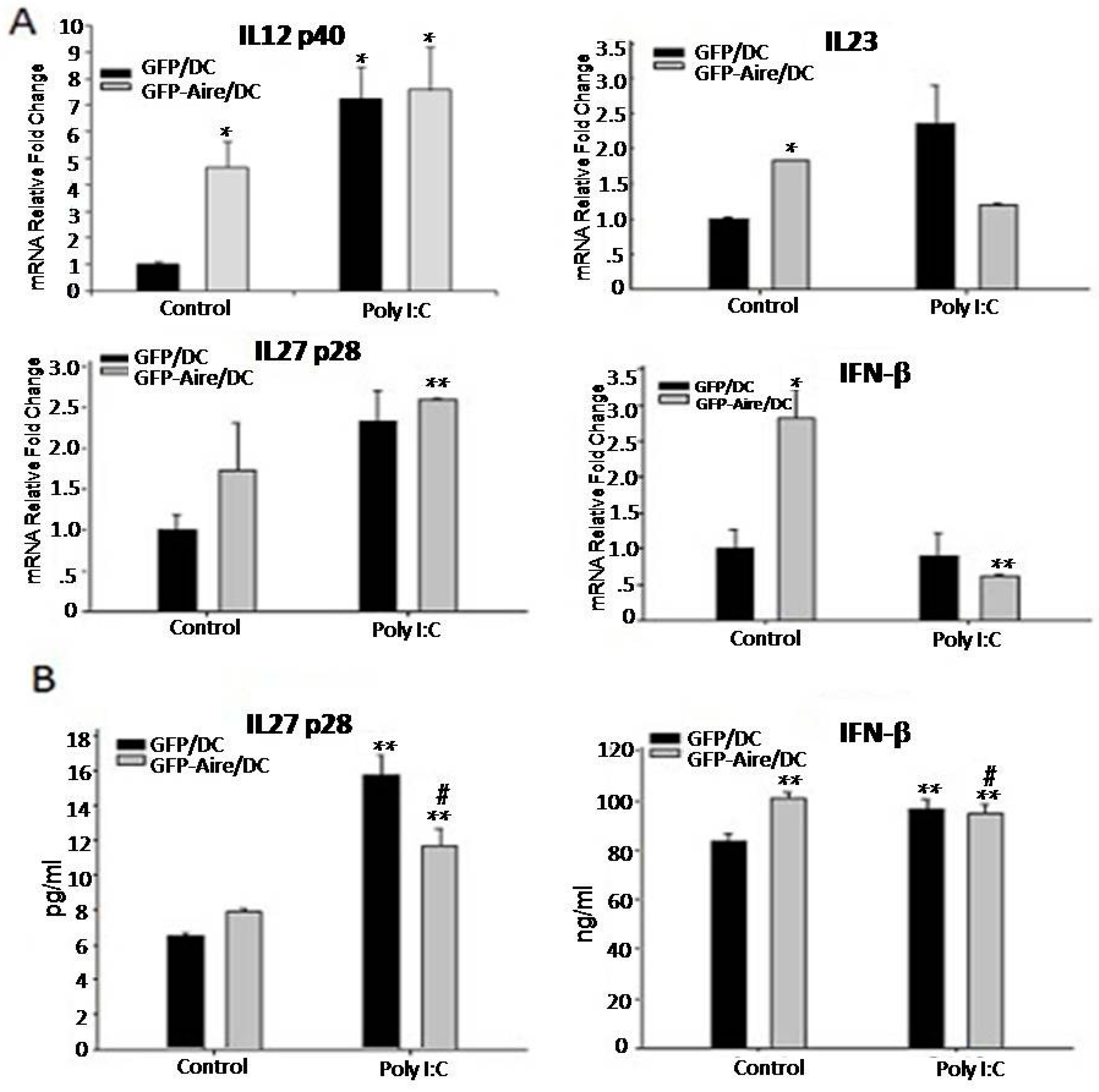
2.4. Aire Regulates Cytokine Expression Downstream of the TLR3 Pathway
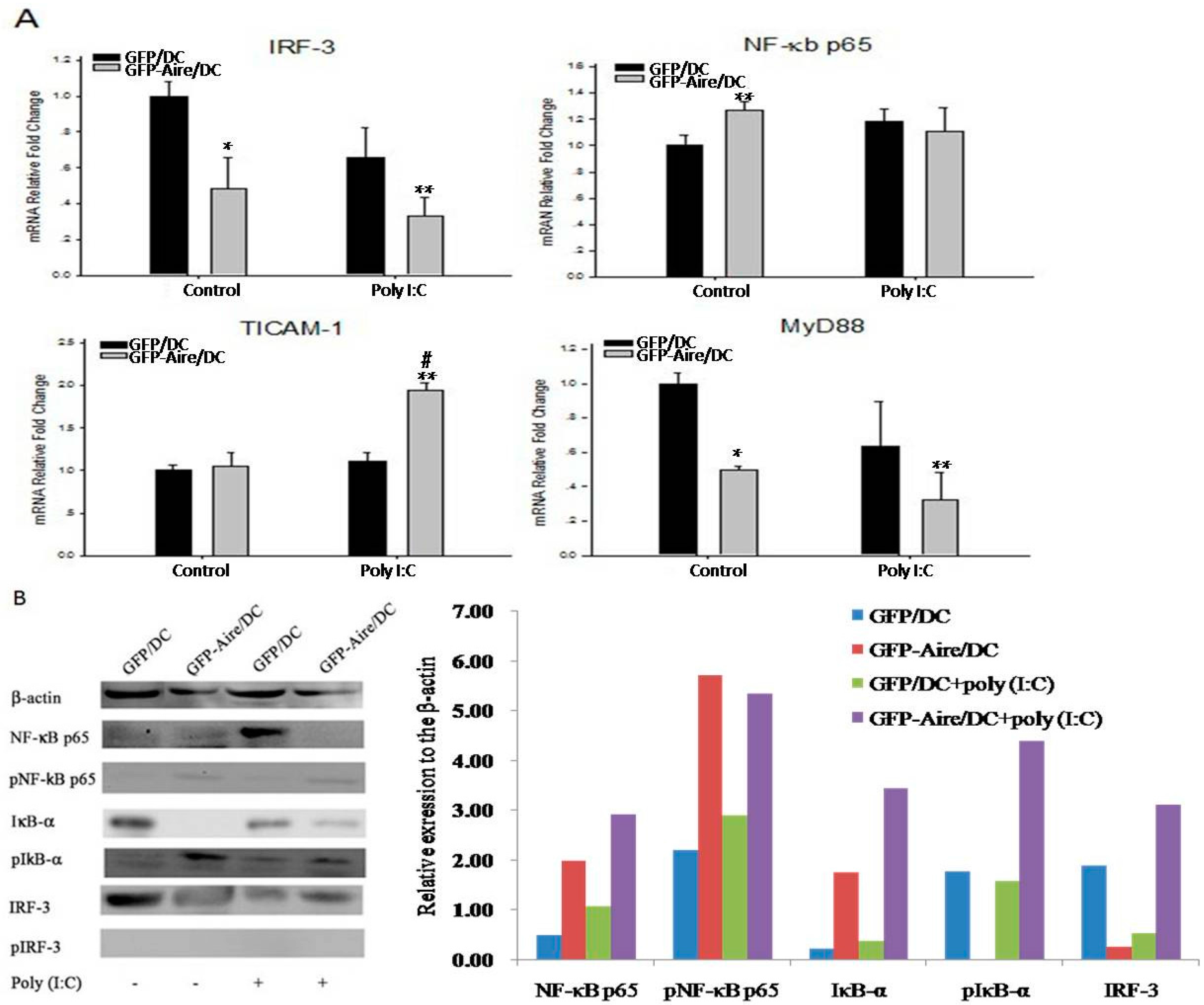
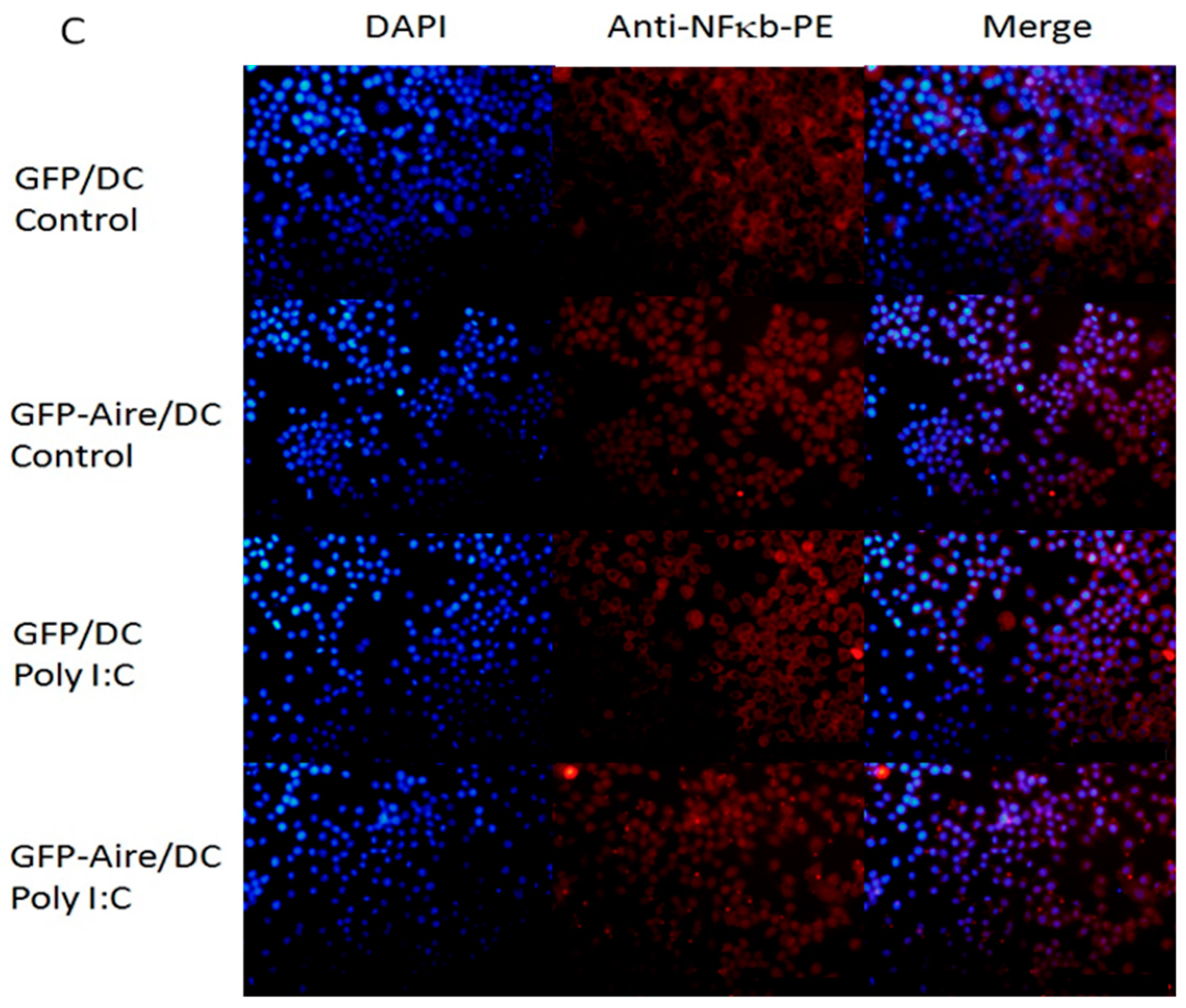
2.5. Aire Modifies Key TLR3 Pathway Molecules in DC2.4
3. Discussion
4. Experimental Section
4.1. Cells and Animals
4.2. Cell Culture and Ligand Stimulation
4.3. Transfection
4.4. Quantitative Real-Time PCR (RT-qPCR)
4.5. Flow Cytometry (FCM)
4.6. Western Blotting
4.7. Immunofluorescence Staining
4.8. ELISA
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Takeda, K.; Akira, S. Toll-like receptors. Curr. Protoc. Immunol. 2015, 109, 1–10. [Google Scholar]
- De Nardo, D. Toll-like receptors: Activation, signalling and transcriptional modulation. Cytokine 2015, 74, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Gauzzi, M.C.; del Corno, M.; Gessani, S. Dissecting TLR3 signalling in dendritic cells. Immunobiology 2010, 215, 713–723. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Ostroff, G.R.; Lee, C.K.; Wang, J.P.; Specht, C.A.; Levitz, S.M. Distinct patterns of dendritic cell cytokine release stimulated by fungal β-glucans and toll-like receptor agonists. Infect. Immun. 2009, 77, 1774–1781. [Google Scholar] [CrossRef] [PubMed]
- Rizzetto, L.; Cavalieri, D. A systems biology approach to the mutual interaction between yeast and the immune system. Immunobiology 2010, 215, 762–769. [Google Scholar] [CrossRef] [PubMed]
- Romani, L. Immunity to fungal infections. Nat. Rev. Immunol. 2011, 11, 275–288. [Google Scholar] [CrossRef] [PubMed]
- Swiecki, M.; McCartney, S.A.; Wang, Y.; Colonna, M. TLR7/9 versus TLR3/MDA5 signaling during virus infections and diabetes. J. Leukoc. Biol. 2011, 90, 691–701. [Google Scholar] [CrossRef] [PubMed]
- Leonard, M.F. Chronic idiopathic hypoparathyroidism with superimposed Addison’s disease in a child. J. Clin. Endocrinol. Metab. 1946, 6, 493–506. [Google Scholar] [CrossRef] [PubMed]
- Gardner, J.M.; Devoss, J.J.; Friedman, R.S.; Wong, D.J.; Tan, Y.X.; Zhou, X.; Johannes, K.P.; Su, M.A.; Chang, H.Y.; Krummel, M.F.; et al. Deletional tolerance mediated by extrathymicAire-expressing cells. Science 2008, 321, 843–847. [Google Scholar] [CrossRef] [PubMed]
- Akirav, E.M.; Ruddle, N.H.; Herold, K.C. The role of Aire in human autoimmune disease. Nat. Rev. Endocrinol. 2011, 7, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Malchow, S.; Leventhal, D.S.; Nishi, S.; Fischer, B.I.; Shen, L.; Paner, G.P.; Amit, A.S.; Kang, C.; Geddes, J.E.; Allison, J.P.; et al. Aire-dependent thymic development of tumor-associated regulatory T cells. Science 2013, 339, 1219–1224. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Fu, H.; Wu, J.; Zhu, W.; Li, Y.; Yang, W. Macrophages overexpressing Aire induce CD4+Foxp3+ T cells. Mol. Med. Rep. 2013, 7, 159–165. [Google Scholar] [PubMed]
- Wu, J.; Zhu, W.; Fu, H.; Zhang, Y.; Sun, J.; Yang, W.; Li, Y. DNA-PKcs interacts with Aire and regulates the expression of toll-like receptors in RAW264.7 cells. Scand. J. Immunol. 2012, 75, 479–488. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Yang, W.; He, Z.; Liao, X.; Wu, J.; Sun, J.T.; Yang, Y.; Li, Y. Overexpressing autoimmune regulator regulates the expression of toll-like receptors by interacting with their promoters in RAW264.7 cells. Cell. Immunol. 2011, 270, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Banchereau, J.; Pascual, V.; O’Garra, A. From IL-2 to IL-37: The expanding spectrum of anti-inflammatory cytokines. Nat. Immunol. 2012, 13, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Do, J.S.; Visperas, A.; Sanogo, Y.O.; Bechtel, J.J.; Dvorina, N.; Kim, S.; Jang, E.; Stohlman, S.A.; Shen, B.; Fairchild, R.L.; et al. An IL-27/Lag3 axis enhances Foxp3 regulatory T cell-suppressive function and therapeutic efficacy. Mucosal Immunol. 2016, 9, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, Y.; Fujio, K.; Okamura, T.; Yamamoto, K. Interleukin-27 in T cell immunity. Int. J. Mol. Sci. 2015, 16, 2851–2863. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.Y.; Roberts, L.L.; Robinson, C.M. The presence of interleukin-27 during monocyte-derived dendritic cell differentiation promotes improved antigen processing and stimulation of T cells. Immunology 2015, 144, 649–660. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Seya, T. TLR3: Interferon induction by double-stranded RNA including poly (I:C). Adv. Drug Deliv. Rev. 2008, 60, 805–812. [Google Scholar] [CrossRef] [PubMed]
- Honda, K.; Taniguchi, T. IRFs: Master regulators of signalling by Toll-like receptors and cytosolic pattern-recognition receptors. Nat. Rev. Immunol. 2006, 6, 644–658. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Mamane, Y.; Hiscott, J. Structural and functional analysis of interferon regulatory factor 3: Localization of the transactivation and autoinhibitory domains. Mol. Cell. Biol. 1999, 19, 2465–2474. [Google Scholar] [CrossRef] [PubMed]
- Pitkanen, J.; Doucas, V.; Sternsdorf, T.; Nakajima, T.; Aratani, S.; Jensen, K.; Will, H.; Vähämurto, P.; Ollila, J.; Vihinen, M.; et al. The autoimmune regulator protein has transcriptional transactivating properties and interacts with the common coactivator CREB-binding protein. J. Biol. Chem. 2000, 275, 16802–16809. [Google Scholar] [CrossRef] [PubMed]
- Kaisho, T.; Tanaka, T. Turning NF-κB and IRFs on and off in DC. Trends Immunol. 2008, 29, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Oeckinghaus, A.; Ghosh, S. The NF-κB family of transcription factors and its regulation. Cold Spring Harb. Perspect. Biol. 2009, 1, a000034. [Google Scholar] [CrossRef] [PubMed]
- Bonifazi, P.; Zelante, T.; D’Angelo, C.; de Luca, A.; Moretti, S.; Bozza, S.; Perruccio, K.; Iannitti, R.G.; Giovannini, G.; Volpi, C.; et al. Balancing inflammation and tolerance in vivo through dendritic cells by the commensal Candida albicans. Mucosal. Immunol. 2009, 4, 362–374. [Google Scholar] [CrossRef] [PubMed]
| MFI % | GFP/DC | GFP-Aire/DC | ||
|---|---|---|---|---|
| Control | Poly (I:C) | Control | Poly (I:C) | |
| CD40 | 0.50 ± 0.25 | 2.60 ± 0.18 1 | 0.64 ± 0.22 | 0.98 ± 0.27 2,3 |
| 1.67 ± 0.64 | 8.35 ± 0.35 2 | 6.61 ± 0.13 | 7.34 ± 1.02 | |
| CD80 | 51.60 ± 12.16 | 69.60 ± 0.64 | 26.20 ± 8.76 2 | 14.51 ± 10.57 3 |
| 30.43 ± 7.36 | 43.67 ± 1.37 | 25.99 ± 0.02 | 8.08 ± 4.57 4 | |
| CD83 | 17.07 ± 0.49 | 8.46 ± 0.85 1 | 13.60 ± 1.70 | 4.08 ± 3.29 3 |
| 13.75 ± 3.17 | 13.18 ± 9.03 | 11.75 ± 1.27 | 9.02 ± 3.89 | |
| CD11C | 7.93 ± 0.49 | 11.70 ± 0.28 2 | 6.69 ± 0.22 | 5.03 ± 0.18 4 |
| 15.59 ± 5.51 | 19.60 ± 0.87 | 17.41 ± 2.75 | 16.14 ± 0.20 | |
| CD86 | 1.82 ± 0.39 | 2.70 ± 1.00 2 | 2.89 ± 0.25 2 | 2.34 ± 0.19 2 |
| 4.03 ± 0.38 | 5.88 ± 1.75 2 | 3.59 ± 0.05 2 | 6.60 ± 0.68 | |
| MHC II | 0.29 ± 0.14 | 1.09 ± 0.11 2 | 0.42 ± 0.29 | 0.42 ± 0.16 3 |
| 0.62 ± 0.41 | 1.37 ± 0.43 | 2.42 ± 0.01 1 | 2.22 ± 1.93 | |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Niu, K.; Fu, H.; Li, H.; Li, Y.; Yang, W. Autoimmune Regulator Expression in DC2.4 Cells Regulates the NF-κB Signaling and Cytokine Expression of the Toll-Like Receptor 3 Pathway. Int. J. Mol. Sci. 2016, 17, 2002. https://doi.org/10.3390/ijms17122002
Sun J, Niu K, Fu H, Li H, Li Y, Yang W. Autoimmune Regulator Expression in DC2.4 Cells Regulates the NF-κB Signaling and Cytokine Expression of the Toll-Like Receptor 3 Pathway. International Journal of Molecular Sciences. 2016; 17(12):2002. https://doi.org/10.3390/ijms17122002
Chicago/Turabian StyleSun, Jitong, Kunwei Niu, Haiying Fu, Haijun Li, Yi Li, and Wei Yang. 2016. "Autoimmune Regulator Expression in DC2.4 Cells Regulates the NF-κB Signaling and Cytokine Expression of the Toll-Like Receptor 3 Pathway" International Journal of Molecular Sciences 17, no. 12: 2002. https://doi.org/10.3390/ijms17122002