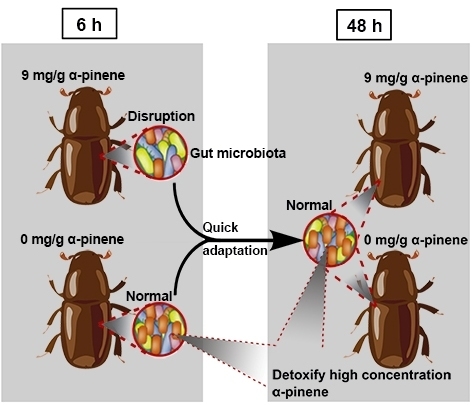
Pine Defensive Monoterpene α-Pinene Influences the Feeding Behavior of Dendroctonus valens and Its Gut Bacterial Community Structure
Abstract
:1. Introduction
2. Results
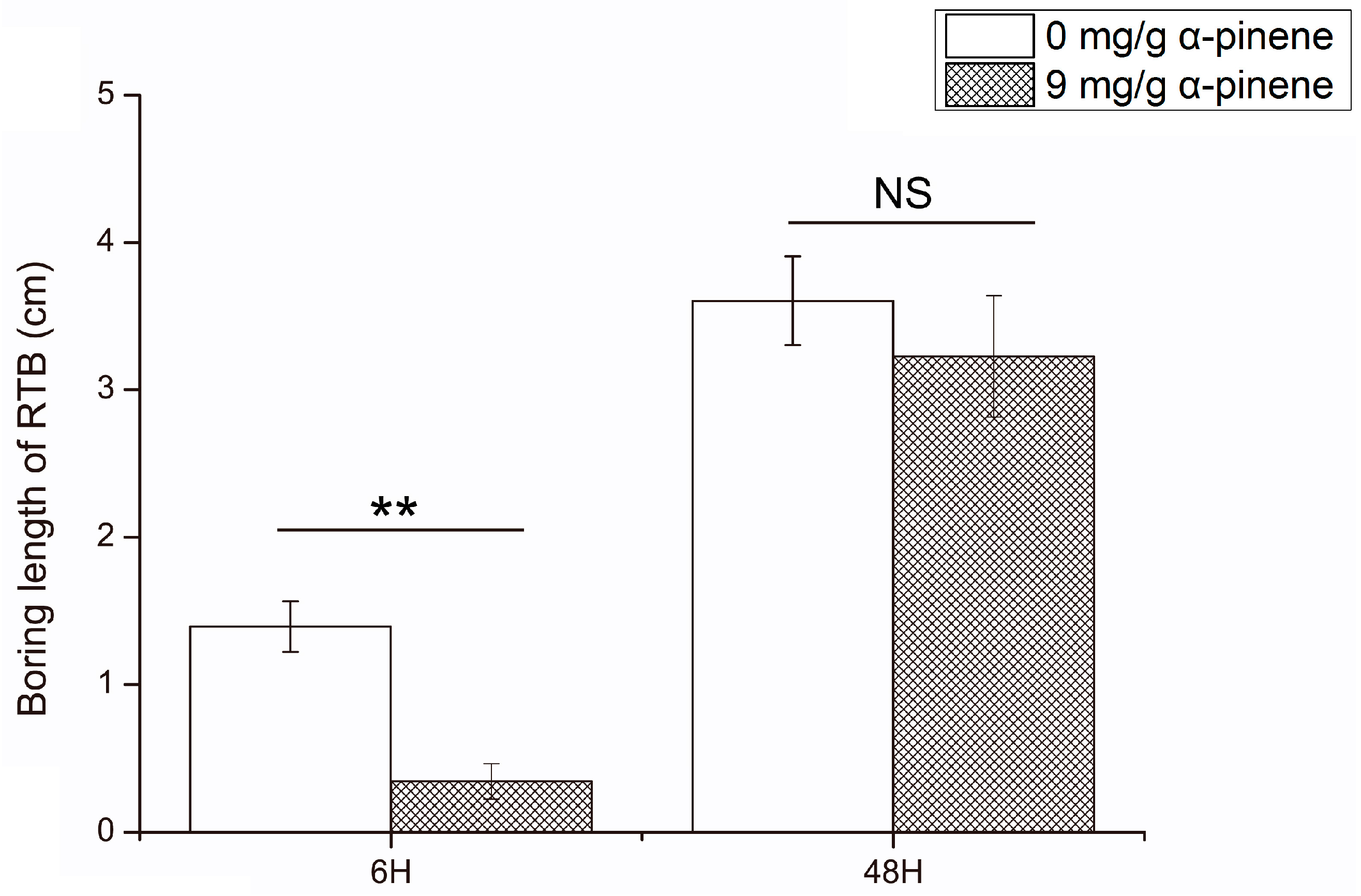
2.1. The Influence of α-Pinene on the Boring Length of Dendroctonus valens
2.2. Alpha Diversity
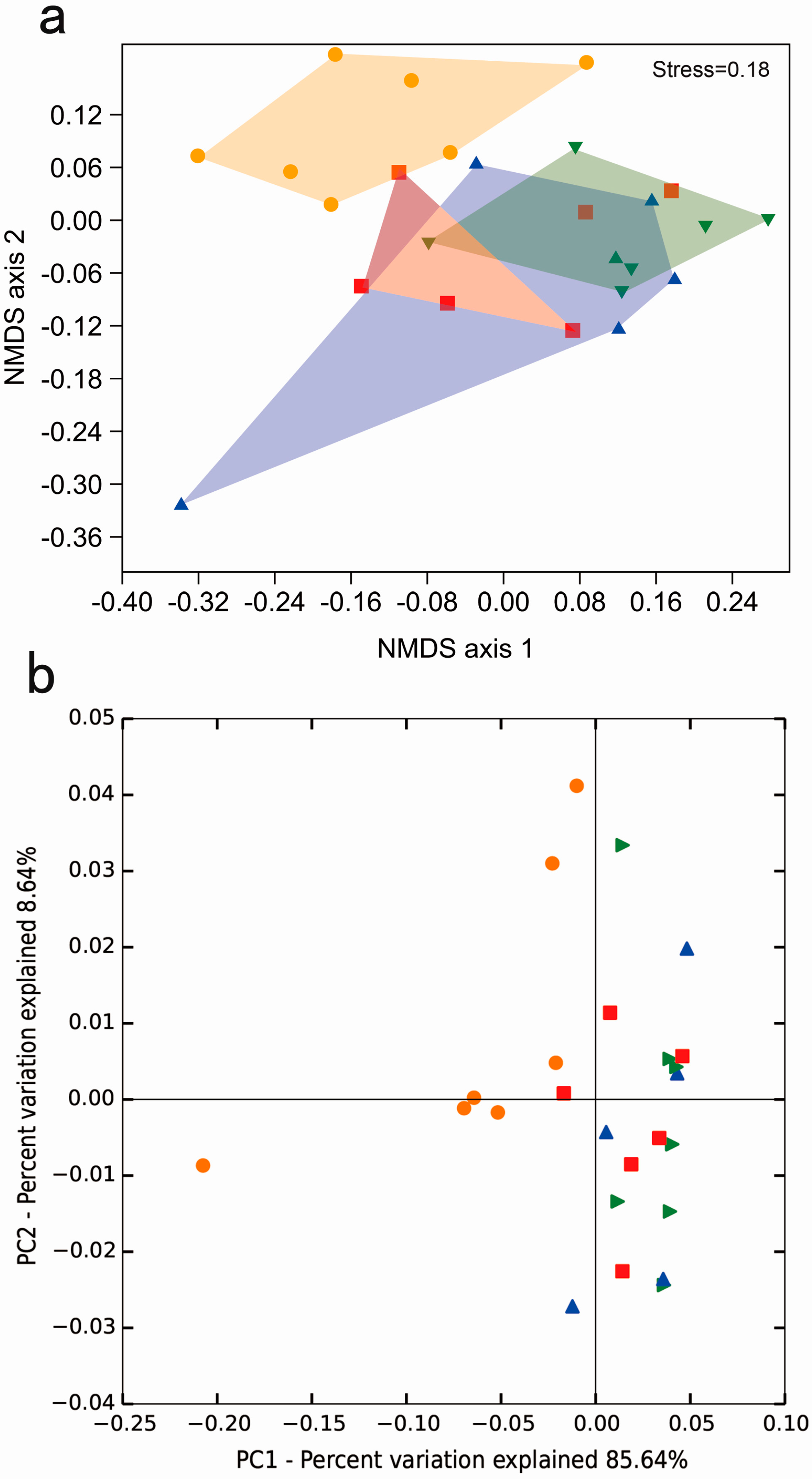
2.3. Principal Component Analysis of the Gut Microbiota of D. valens
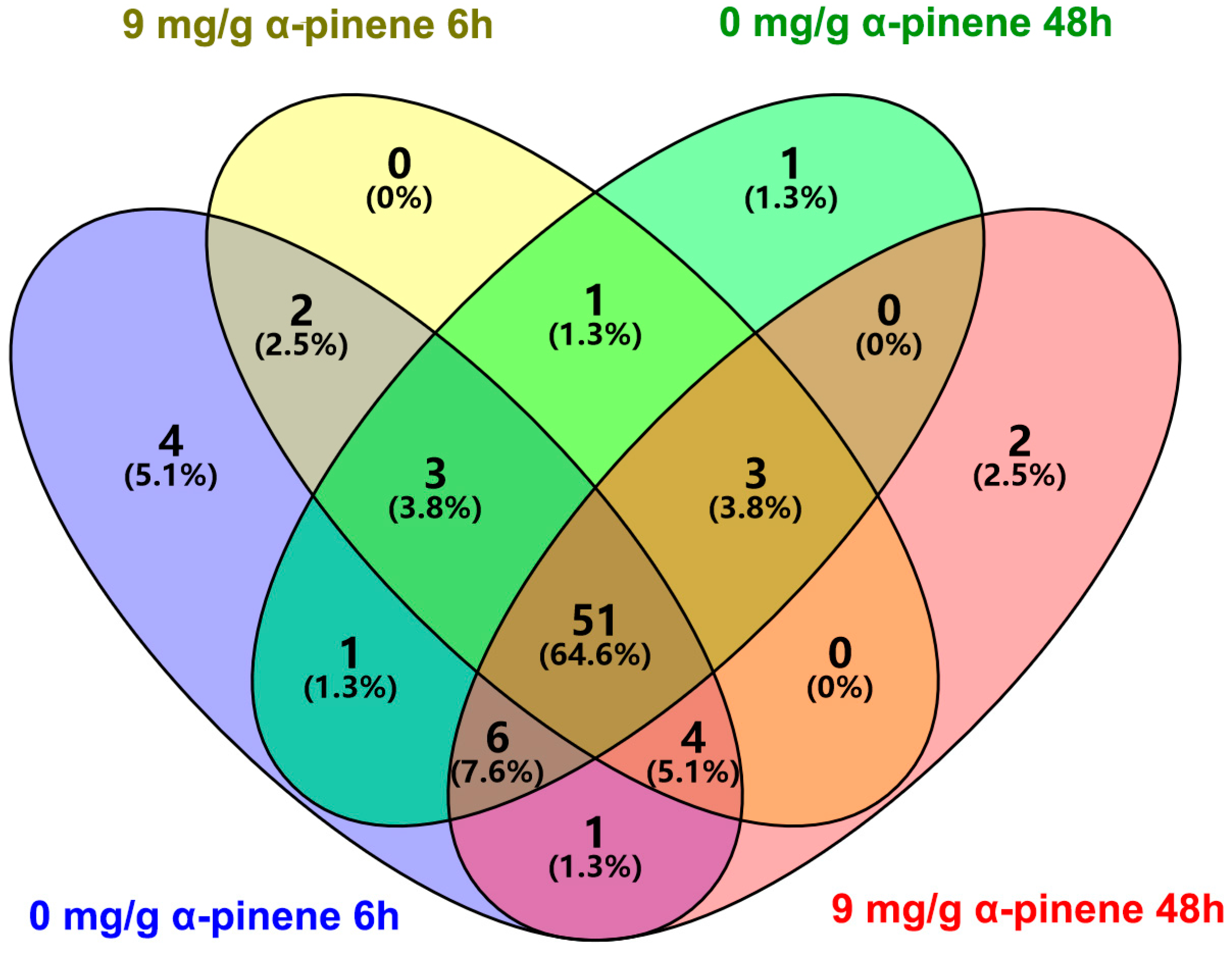
2.4. The Analysis of Community Composition at Genus Levels
3. Discussion
4. Materials and Methods
4.1. Insects
4.2. The Boring Lengths of D. valens at 0 and 9 mg/g Concentration of Host Defensive Compound α-Pinene
4.3. DNA Extraction, PCR, Pyrosequencing, and Sequence Processing
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Geib, S.M.; Filley, T.R.; Hatcher, P.G.; Hoover, K.; Carlson, J.E.; del Mar Jimenez-Gasco, M.; Nakagawa-Izumi, A.; Sleighter, R.L.; Tien, M. Lignin degradation in wood-feeding insects. Proc. Natl. Acad. Sci. USA 2008, 105, 12932–12937. [Google Scholar] [CrossRef] [PubMed]
- Morales-Jiménez, J.; de León, A.V.-P.; García-Domínguez, A.; Martínez-Romero, E.; Zúñiga, G.; Hernández-Rodríguez, C. Nitrogen-fixing and uricolytic bacteria associated with the gut of Dendroctonus rhizophagus and Dendroctonus valens (Curculionidae: Scolytinae). Microb. Ecol. 2013, 66, 200–210. [Google Scholar] [CrossRef] [PubMed]
- Ayayee, P.; Rosa, C.; Ferry, J.G.; Felton, G.; Saunders, M.; Hoover, K. Gut microbes contribute to nitrogen provisioning in a wood-feeding cerambycid. Environ. Entomol. 2014, 43, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Dillon, R.; Vennard, C.; Buckling, A.; Charnley, A. Diversity of locust gut bacteria protects against pathogen invasion. Ecol. Lett. 2005, 8, 1291–1298. [Google Scholar] [CrossRef]
- Koch, H.; Schmid-Hempel, P. Socially transmitted gut microbiota protect bumble bees against an intestinal parasite. Proc. Natl. Acad. Sci. USA 2011, 108, 19288–19292. [Google Scholar] [CrossRef] [PubMed]
- Dillon, R.; Vennard, C.; Charnley, A. A note: Gut bacteria produce components of a locust cohesion pheromone. J. Appl. Microbiol. 2002, 92, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Lou, Q.; Cheng, C.; Lu, M.; Sun, J. Gut-associated bacteria of Dendroctonus valens and their involvement in verbenone production. Microb. Ecol. 2015, 70, 1012–1023. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, Y.; Hayatsu, M.; Hosokawa, T.; Nagayama, A.; Tago, K.; Fukatsu, T. Symbiont-mediated insecticide resistance. Proc. Natl. Acad. Sci. USA 2012, 109, 8618–8622. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Lu, M.; Sun, J. Invasive bark beetle-associated microbes degrade a host defensive monoterpene. Insect Sci. 2016, 23, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Feldhaar, H. Bacterial symbionts as mediators of ecologically important traits of insect hosts. Ecol. Entomol. 2011, 36, 533–543. [Google Scholar] [CrossRef]
- Douglas, A.E. Microbial brokers of insect-plant interactions revisited. J. Chem. Ecol. 2013, 39, 952–961. [Google Scholar] [CrossRef] [PubMed]
- Hammer, T.J.; Bowers, M.D. Gut microbes may facilitate insect herbivory of chemically defended plants. Oecologia 2015, 179, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Douglas, A. The microbial dimension in insect nutritional ecology. Funct. Ecol. 2009, 23, 38–47. [Google Scholar] [CrossRef]
- Dowd, P. Symbiont-mediated detoxification in insect herbivores. In Microbial Mediation of Plant-Herbivore Interactions; Barbosa, P., Krischik, V.A., Jones, C.G., Eds.; John Wiley & Sons: New York, NY, USA, 1991; pp. 411–440. [Google Scholar]
- Boone, C.K.; Keefover-Ring, K.; Mapes, A.C.; Adams, A.S.; Bohlmann, J.; Raffa, K.F. Bacteria associated with a tree-killing insect reduce concentrations of plant defense compounds. J. Chem. Ecol. 2013, 39, 1003–1006. [Google Scholar] [CrossRef] [PubMed]
- Levin, D.A. The chemical defenses of plants to pathogens and herbivores. Annu. Rev. Ecol. Syst. 1976, 7, 121–159. [Google Scholar] [CrossRef]
- Colman, D.; Toolson, E.; Takacs-Vesbach, C. Do diet and taxonomy influence insect gut bacterial communities? Mol. Ecol. 2012, 21, 5124–5137. [Google Scholar] [CrossRef] [PubMed]
- Gayatri Priya, N.; Ojha, A.; Kajla, M.K.; Raj, A.; Rajagopal, R. Host plant induced variation in gut bacteria of Helicoverpa armigera. PLoS ONE 2012, 7, e30768. [Google Scholar] [CrossRef] [PubMed]
- Mason, C.; Rubert-Nason, K.; Lindroth, R.; Raffa, K. Aspen defense chemicals influence midgut bacterial community composition of gypsy moth. J. Chem. Ecol. 2015, 41, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Raffa, K.F.; Berryman, A.A. Interacting selective pressures in conifer-bark beetle systems: A basis for reciprocal adaptations? Am. Nat. 1987, 129, 234–262. [Google Scholar] [CrossRef]
- Paine, T.D.; Raffa, K.F.; Harrington, T.C. Interactions among scolytid bark beetles, their associated fungi, and live host conifers. Annu. Rev. Entomol. 1997, 42, 179–206. [Google Scholar] [CrossRef] [PubMed]
- Gitau, C.W.; Bashford, R.; Carnegie, A.J.; Gurr, G.M. A review of semiochemicals associated with bark beetle (Coleoptera: Curculionidae: Scolytinae) pests of coniferous trees: A focus on beetle interactions with other pests and their associates. For. Ecol. Manag. 2013, 297, 1–14. [Google Scholar] [CrossRef]
- Sun, J.; Lu, M.; Gillette, N.E.; Wingfield, M.J. Red turpentine beetle: Innocuous native becomes invasive tree killer in China. Annu. Rev. Entomol. 2013, 58, 293–311. [Google Scholar] [CrossRef] [PubMed]
- Lieutier, F. Mechanisms of resistance in conifers and bark beetle attack strategies. In Mechanisms and Deployment of Resistance in Trees to Insects; Springer: Berlin/Heidelberg, Germany, 2002; pp. 31–77. [Google Scholar]
- Smith, R.H. Toxicity of pine resin vapors to three species of Dendroctonus bark beetles. J. Econ. Entomol. 1963, 56, 827–831. [Google Scholar] [CrossRef]
- Byers, J.A. Pheromone biosynthesis in the bark beetle, Ips paraconfusus, during feeding or exposure to vapours of host plant precursors. Insect Biochem. 1981, 11, 563–569. [Google Scholar] [CrossRef]
- Phillips, M.A.; Croteau, R.B. Resin-based defenses in conifers. Trends Plant Sci. 1999, 4, 184–190. [Google Scholar] [CrossRef]
- Seybold, S.J.; Huber, D.P.W.; Lee, J.C.; Graves, A.D.; Bohlmann, J. Pine monoterpenes and pine bark beetles: A marriage of convenience for defense and chemical communication. Phytochem. Rev. 2006, 5, 143–178. [Google Scholar] [CrossRef]
- Klepzig, K.; Smalley, E.; Raffa, K. Combined chemical defenses against an insect-fungal complex. J. Chem. Ecol. 1996, 22, 1367–1388. [Google Scholar] [CrossRef] [PubMed]
- Hofstetter, R.W.; Mahfouz, J.B.; Klepzig, K.D.; Ayres, M.P. Effects of tree phytochemistry on the interactions among endophloedic fungi associated with the southern pine beetle. J. Chem. Ecol. 2005, 31, 539–560. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.S.; Boone, C.K.; Bohlmann, J.; Raffa, K.F. Responses of bark beetle-associated bacteria to host monoterpenes and their relationship to insect life histories. J. Chem. Ecol. 2011, 37, 808–817. [Google Scholar] [CrossRef] [PubMed]
- Raffa, K.F. Terpenes tell different tales at different scales: Glimpses into the chemical ecology of conifer-bark beetle-microbial interactions. J. Chem. Ecol. 2014, 40, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.S.; Aylward, F.O.; Adams, S.M.; Erbilgin, N.; Aukema, B.H.; Currie, C.R.; Suen, G.; Raffa, K.F. Mountain pine beetles colonizing historical and naïve host trees are associated with a bacterial community highly enriched in genes contributing to terpene metabolism. Appl. Environ. Microbiol. 2013, 79, 3468–3475. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.; Zhou, F.; Lu, M.; Sun, J. Inducible pine rosin defense mediates interactions between an invasive insect–fungal complex and newly acquired sympatric fungal associates. Integr. Zool. 2015, 10, 453–464. [Google Scholar] [CrossRef] [PubMed]
- López, M.F.; Cano-Ramírez, C.; Shibayama, M.; Zúñiga, G. α-Pinene and myrcene induce ultrastructural changes in the midgut of Dendroctonus valens (Coleoptera: Curculionidae: Scolytinae). Ann. Entomol. Soc. Am. 2011, 104, 553–561. [Google Scholar] [CrossRef]
- Xu, B.; Liu, Z.; Sun, J.H. The effects of α-pinene on the feeding performance and pheromone production of Dendroctonus valens. Entomol. Exp. Appl. 2014, 150, 269–278. [Google Scholar] [CrossRef]
- Adams, A.S.; Currie, C.R.; Cardoza, Y.; Klepzig, K.D.; Raffa, K.F. Effects of symbiotic bacteria and tree chemistry on the growth and reproduction of bark beetle fungal symbionts. Can. J. For. Res. 2009, 39, 1133–1147. [Google Scholar] [CrossRef]
- Hobson, K.R.; Wood, D.L.; Cool, L.G.; White, P.R.; Ohtsuka, T.; Kubo, I.; Zavarin, E. Chiral specificity in responses by the bark beetle Dendroctonus valens to host kairomones. J. Chem. Ecol. 1993, 19, 1837–1846. [Google Scholar] [CrossRef] [PubMed]
- Brand, J.M.; Bracke, J.W.; Markovetz, A.J.; Wood, D.L.; Browne, L.E. Production of verbenol pheromone by a bacterium isolated from bark beetles. Nature 1975, 254, 136–137. [Google Scholar] [CrossRef] [PubMed]
- Mason, C.J.; Couture, J.J.; Raffa, K.F. Plant-associated bacteria degrade defense chemicals and reduce their adverse effects on an insect defoliator. Oecologia 2014, 175, 901–910. [Google Scholar] [CrossRef] [PubMed]
- Kohl, K.D.; Dearing, M.D. Experience matters: Prior exposure to plant toxins enhances diversity of gut microbes in herbivores. Ecol. Lett. 2012, 15, 1008–1015. [Google Scholar] [CrossRef] [PubMed]
- Birgersson, G.; Schlyter, F.; Lofqvist, J.; Bergstrom, G. Quantitative variation of pheromone components in the spruce bark beetle Ips typographus from different attack phases. J. Chem. Ecol. 1984, 10, 1029–1055. [Google Scholar] [CrossRef] [PubMed]
- Wallin, K.F.; Raffa, K.F. Influences of host chemicals and internal physiology on the multiple steps of postlanding host acceptance behavior of Ips pini (Coleoptera: Scolytidae). Environ. Entomol. 2000, 29, 442–453. [Google Scholar] [CrossRef]
- Reid, M.; Purcell, J.R.C. Condition-dependent tolerance of monoterpenes in an insect herbivore. Arthropod Plant Interact. 2011, 5, 331–337. [Google Scholar] [CrossRef]
- Santo Domingo, J.; Kaufman, M.; Klug, M.; Holben, W.; Harris, D.; Tiedje, J. Influence of diet on the structure and function of the bacterial hindgut community of crickets. Mol. Ecol. 1998, 7, 761–767. [Google Scholar] [CrossRef]
- Sharon, G.; Segal, D.; Zilber-Rosenberg, I.; Rosenberg, E. Symbiotic bacteria are responsible for diet-induced mating preference in Drosophila melanogaster, providing support for the hologenome concept of evolution. Gut Microbes 2011, 2, 190–192. [Google Scholar] [CrossRef] [PubMed]
- Lozupone, C.A.; Stombaugh, J.I.; Gordon, J.I.; Jansson, J.K.; Knight, R. Diversity, stability and resilience of the human gut microbiota. Nature 2012, 489, 220–230. [Google Scholar] [CrossRef] [PubMed]
- Sudakaran, S.; Salem, H.; Kost, C.; Kaltenpoth, M. Geographical and ecological stability of the symbiotic mid-gut microbiota in European firebugs, Pyrrhocoris apterus (Hemiptera, Pyrrhocoridae). Mol. Ecol. 2012, 21, 6134–6151. [Google Scholar] [CrossRef] [PubMed]
- Salem, H.; Kreutzer, E.; Sudakaran, S.; Kaltenpoth, M. Actinobacteria as essential symbionts in firebugs and cotton stainers (Hemiptera, Pyrrhocoridae). Environ. Microbiol. 2013, 15, 1956–1968. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, S.; Kobayashi, T.; Songjinda, P.; Tateyama, A.; Tsubouchi, M.; Kiyohara, C.; Shirakawa, T.; Sonomoto, K.; Nakayama, J. Influence of antibiotic exposure in the early postnatal period on the development of intestinal microbiota. FEMS Immunol. Med. Microbiol. 2009, 56, 80–87. [Google Scholar] [CrossRef] [PubMed]
- White, D.C.; Sutton, S.D.; Ringelberg, D.B. The genus Sphingomonas: Physiology and ecology. Curr. Opin. Biotechnol. 1996, 7, 301–306. [Google Scholar] [CrossRef]
- Thomassin-Lacroix, E.J.; Yu, Z.; Eriksson, M.; Reimer, K.J.; Mohn, W.W. DNA-based and culture-based characterization of a hydrocarbon-degrading consortium enriched from Arctic soil. Can. J. Microbiol. 2001, 47, 1107–1115. [Google Scholar] [CrossRef] [PubMed]
- Singer, A.C. The chemical ecology of pollutant biodegradation: Bioremediation and phytoremediation from mechanistic and ecological perspectives. In Phytoremediation Rhizoremediation; Springer: Berlin/Heidelberg, Germany, 2006; pp. 5–21. [Google Scholar]
- Van der Werf, M.J.; Swarts, H.J.; de Bont, J.A. Rhodococcus erythropolis DCL14 contains a novel degradation pathway for limonene. Appl. Environ. Microbiol. 1999, 65, 2092–2102. [Google Scholar] [PubMed]
- Langolf, B.M.; Kleinheinz, G.T. A lava rock-based biofilter for the treatment of α-pinene. Bioresour. Technol. 2006, 97, 1951–1958. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.-J. Characterization of aromatic hydrocarbon degrading bacteria isolated from pine litter. Korean J. Microbiol. Biotechnol. 2009, 37, 333–339. [Google Scholar]
- Breznak, J.A. Intestinal microbiota of termites and other xylophagous insects. Ann. Rev. Microbiol. 1982, 36, 323–343. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.M.; Teakle, D.S.; MacRae, I.C. Kinetics of colonization of adult Queensland fruit flies (Bactrocera tryoni) by dinitrogen-fixing alimentary tract bacteria. Appl. Environ. Microbiol. 1994, 60, 2508–2517. [Google Scholar] [PubMed]
- Leufvén, A.; Birgersson, G. Quantitative variation of different monoterpenes around galleries of Ips typographus (Colleoptera: Scolytidae) attacking Norway spruce. Can. J. Bot. 1987, 65, 1038–1044. [Google Scholar] [CrossRef]
- Zhang, Q.H.; Birgersson, G.; Schlyter, F.; Chen, G.F. Pheromone components in the larch bark beetle, Ips cembrae, from China: Quantitative variation among attack phases and individuals. J. Chem. Ecol. 2000, 26, 841–858. [Google Scholar] [CrossRef]
- Yu, Y.; Lee, C.; Kim, J.; Hwang, S. Group-specific primer and probe sets to detect methanogenic communities using quantitative real-time polymerase chain reaction. Biotechnol. Bioeng. 2005, 89, 670–679. [Google Scholar] [CrossRef] [PubMed]
- Caporaso, J.G.; Kuczynski, J.; Stombaugh, J.; Bittinger, K.; Bushman, F.D.; Costello, E.K.; Fierer, N.; Pena, A.G.; Goodrich, J.K.; Gordon, J.I. QIIME allows analysis of high-throughput community sequencing data. Nat. Methods 2010, 7, 335–336. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C.; Haas, B.J.; Clemente, J.C.; Quince, C.; Knight, R. UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 2011, 27, 2194–2200. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Garrity, G.M.; Tiedje, J.M.; Cole, J.R. Naive Bayesian classifier for rapid assignment of rRNA sequences into the new bacterial taxonomy. Appl. Environ. Microbiol. 2007, 73, 5261–5267. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.L.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, a chimera-checked 16S rRNA gene database and workbench compatible with ARB. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef] [PubMed]
- Clarke, K.R. Non-parametric multivariate analyses of changes in community structure. Aust. J. Ecol. 1993, 18, 117–143. [Google Scholar] [CrossRef]
- Bohlmann, J. Insect-induced terpenoid defenses in spruce. In Induced Plant Resistance to Herbivory; Springer: Berlin/Heidelberg, Germany, 2008; pp. 173–187. [Google Scholar]
- Six, D.; Paine, T. Effects of mycangial fungi and host tree species on progeny survival and emergence of Dendroctonus ponderosae (Coleoptera: Scolytidae). Environ. Entomol. 1998, 27, 1393–1401. [Google Scholar] [CrossRef]
- Shrimpton, D.M. Resistance of lodgepole pine to mountain pine beetle infestation. In Theroy and Practice of Mountain Pine Beetle Management in Lodgepole Pine Forests; Berryman, A.A., Amman, G.D., Stark, R.W., Kibbee, D.L., Eds.; University of Idaho Press: Moscow, ID, USA, 1978; pp. 64–75. [Google Scholar]

| Index | 0 mg/g α-Pinene 6 h | 9 mg/g α-Pinene 6 h | 0 mg/g α-Pinene 48 h | 9 mg/g α-Pinene 48 h |
|---|---|---|---|---|
| Number of OTUs | 273.8 ± 28.62 | 306.6 ± 30.14 | 277.0 ± 26.36 | 268.1 ± 17.76 |
| ACE diversity | 455.4 ± 57.56 | 527.5 ± 54.89 | 441.2 ± 62.43 | 402.4 ± 33.63 |
| Chao diversity | 443.0 ± 49.51 | 532.7 ± 52.08 | 449.8 ± 64.44 | 403.1 ± 35.85 |
| Shannon diversity (H) | 1.62 ± 0.09 b | 1.86 ± 0.02 b | 1.61 ± 0.14 b | 1.52 ± 0.05 a |
| Simpson diversity | 0.49 ± 0.02 a,b | 0.60 ± 0.00 b | 0.48 ± 0.02 a,b | 0.46 ± 0.02 a |
| Phylum | Phylogenetic Group (Genus) | 0 mg/g α-Pinene 6 h % | 9 mg/g α-Pinene 6 h % | 0 mg/g α-Pinene 48 h % | 9 mg/g α-Pinene 48 h % |
|---|---|---|---|---|---|
| Actinobacteria | Rhodococcus | 20.42 ± 0.86 | 17.15 ± 1.99 | 20.48 ± 2.40 | 21.58 ± 0.99 |
| Salinibacterium | 0.93 ± 0.20 | 1.00 ± 0.17 | 1.17 ± 0.29 | 1.19 ± 0.20 | |
| Proteobacteria | Burkholderia | 1.05 ± 0.16 a,b | 0.69 ± 0.12 a | 1.22 ± 0.09 a,b | 1.33 ± 0.19 b |
| Erwinia | 5.80 ± 1.96 b | 24.41 ± 5.74 a | 4.05 ± 2.30 b | 2.37 ± 1.17 b | |
| Pseudomonas | 0.26 ± 0.06 | 0.10 ± 0.03 | 0.29 ± 0.09 | 0.36 ± 0.12 | |
| Sphingomonas | 70.35 ± 1.62 b | 55.85 ± 4.26 a | 71.69 ± 1.30 b | 72.24 ± 1.56 b | |
| Sum | 98.81 ± 0.00 | 99.20 ± 0.00 | 98.91 ± 0.00 | 99.06 ± 0.00 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, L.; Shi, Z.; Wang, B.; Lu, M.; Sun, J. Pine Defensive Monoterpene α-Pinene Influences the Feeding Behavior of Dendroctonus valens and Its Gut Bacterial Community Structure. Int. J. Mol. Sci. 2016, 17, 1734. https://doi.org/10.3390/ijms17111734
Xu L, Shi Z, Wang B, Lu M, Sun J. Pine Defensive Monoterpene α-Pinene Influences the Feeding Behavior of Dendroctonus valens and Its Gut Bacterial Community Structure. International Journal of Molecular Sciences. 2016; 17(11):1734. https://doi.org/10.3390/ijms17111734
Chicago/Turabian StyleXu, Letian, Zhanghong Shi, Bo Wang, Min Lu, and Jianghua Sun. 2016. "Pine Defensive Monoterpene α-Pinene Influences the Feeding Behavior of Dendroctonus valens and Its Gut Bacterial Community Structure" International Journal of Molecular Sciences 17, no. 11: 1734. https://doi.org/10.3390/ijms17111734
APA StyleXu, L., Shi, Z., Wang, B., Lu, M., & Sun, J. (2016). Pine Defensive Monoterpene α-Pinene Influences the Feeding Behavior of Dendroctonus valens and Its Gut Bacterial Community Structure. International Journal of Molecular Sciences, 17(11), 1734. https://doi.org/10.3390/ijms17111734