Prokaryotic Expression, Purification and Immunogenicity in Rabbits of the Small Antigen of Hepatitis Delta Virus
Abstract
:1. Introduction
2. Results
2.1. Expression and Purification of Small HDV Antigen
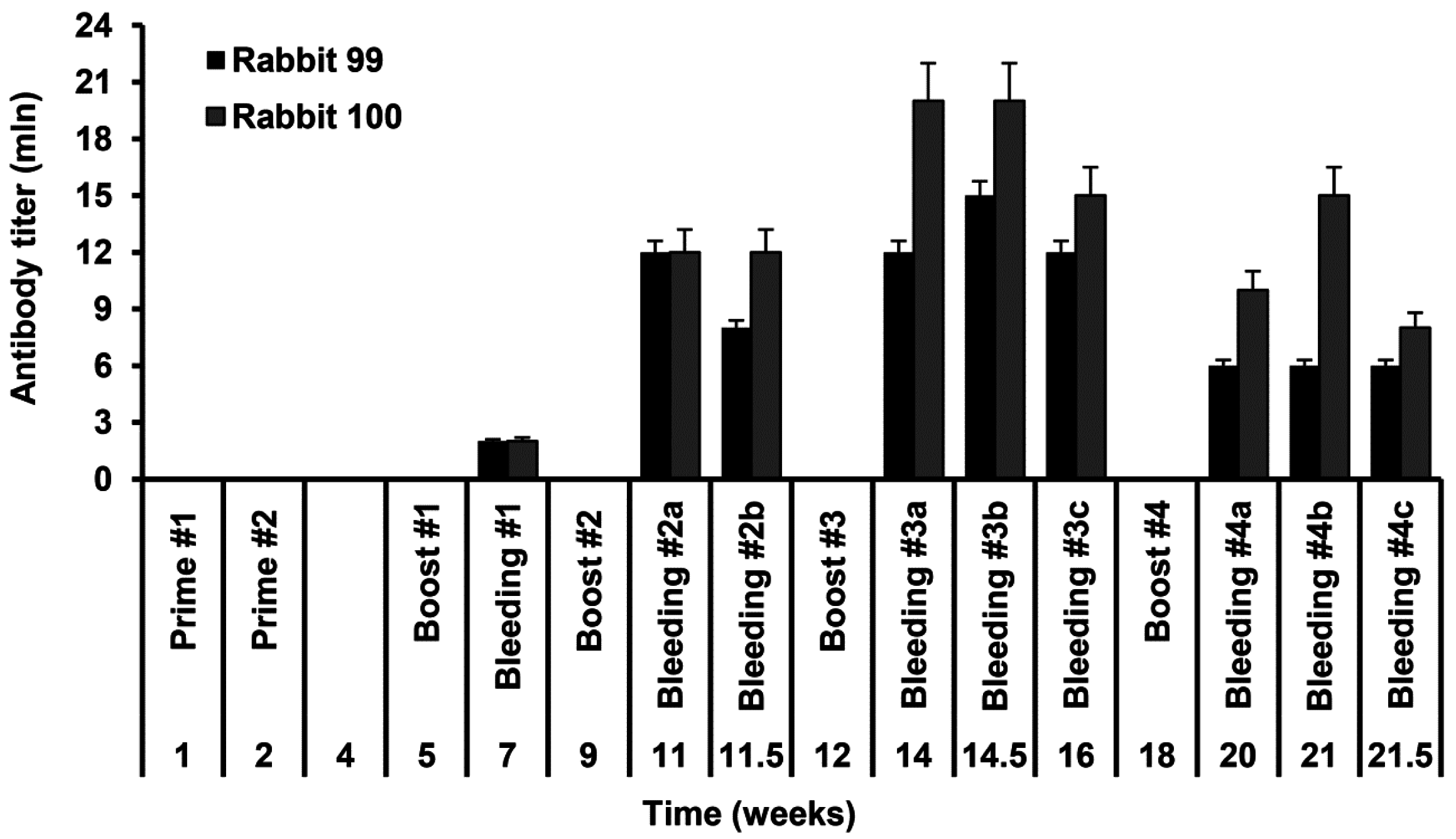
2.2. Rabbit Immunization and Evaluation of Rabbit Anti-S-HDAg
2.3. Application of Anti-S-HDAg Antibodies for Western Blotting
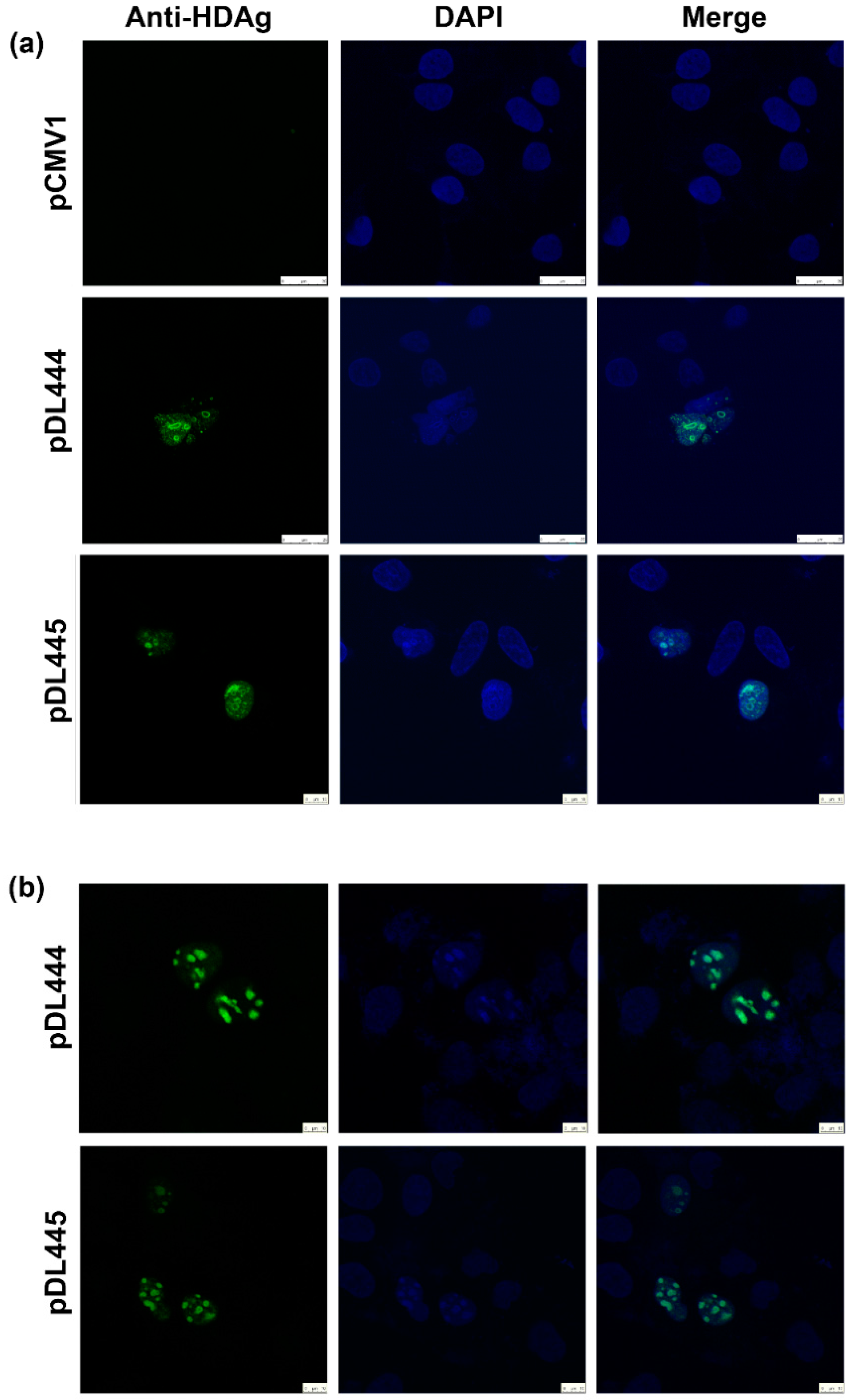
2.4. Application of Anti-S-HDAg Antibodies for Immunofluorescence
3. Materials and Methods
3.1. Reagents
3.2. Plasmid Construction
3.3. Protein Expression and Purification
3.4. Rabbit Immunization
3.5. ELISA
3.6. Cell Culture and Transfection
3.7. Western Blot Analysis
3.8. Immunofluorescence
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Alvarado-Mora, M.V.; Locarnini, S.; Rizzetto, M.; Pinho, J.R. An update on HDV: Virology, pathogenesis and treatment. Antivir. Ther. 2013, 18, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Gunsar, F. Treatment of delta hepatitis. Expert Rev. Anti Infect. Ther. 2013, 11, 489–498. [Google Scholar] [CrossRef] [PubMed]
- Bahcecioglu, I.H.; Ispiroglu, M.; Demirel, U.; Yalniz, M. Pegylated interferon α therapy in chronic delta hepatitis: A one-center experience. Hepat. Mon. 2015, 15. [Google Scholar] [CrossRef] [PubMed]
- Rizzetto, M. Hepatitis d: Clinical features and therapy. Dig. Dis. 2010, 28, 139–143. [Google Scholar] [CrossRef] [PubMed]
- Sagnelli, E.; Sagnelli, C.; Pisaturo, M.; Macera, M.; Coppola, N. Epidemiology of acute and chronic hepatitis B and delta over the last 5 decades in italy. World J. Gastroenterol. 2014, 20, 7635–7643. [Google Scholar] [CrossRef] [PubMed]
- Hughes, S.A.; Wedemeyer, H.; Harrison, P.M. Hepatitis delta virus. Lancet 2011, 378, 73–85. [Google Scholar] [CrossRef]
- Kozhanova, T.V.; Ilchenko, L.Y.; Lopatuchina, M.A.; Saryglar, A.A.; Saryg-Chaa, O.N.; Sonam-Baiyr, Y.D.; Mongusch, M.K.; Kyuregyan, K.K.; Mikhailov, M.I. Familial Clusters of Hepatitis Delta in Endemic Region (Republic Tyva). Eksp. Klin. Gastroenterol. 2015, 11, 15–22. [Google Scholar] [PubMed]
- Polson, A.G.; Bass, B.L.; Casey, J.L. RNA editing of hepatitis delta virus antigenome by dsRNA-adenosine deaminase. Nature 1996, 380, 454–456. [Google Scholar] [PubMed]
- Chen, P.J.; Chang, F.L.; Wang, C.J.; Lin, C.J.; Sung, S.Y.; Chen, D.S. Functional study of hepatitis delta virus large antigen in packaging and replication inhibition: Role of the amino-terminal leucine zipper. J. Virol. 1992, 66, 2853–2859. [Google Scholar] [PubMed]
- Giersch, K.; Dandri, M. Hepatitis B and delta virus: Advances on studies about interactions between the two viruses and the infected hepatocyte. J. Clin. Transl. Hepatol. 2015, 3, 220–229. [Google Scholar] [PubMed]
- Rizzetto, M.; Canese, M.G.; Gerin, J.L.; London, W.T.; Sly, D.L.; Purcell, R.H. Transmission of the hepatitis B virus-associated delta antigen to chimpanzees. J. Infect. Dis. 1980, 141, 590–602. [Google Scholar] [CrossRef] [PubMed]
- Turgeon, M.L. Immunology & Serology in Laboratory Medicine, 5th ed.; Elsevier/Mosby: St. Louis, MO, USA, 2014; pp. 15–584. [Google Scholar]
- Chow, S.K.; Atienza, E.E.; Cook, L.; Prince, H.; Slev, P.; Lape-Nixon, M.; Jerome, K.R. Comparison of enzyme immunoassays for detection of antibodies to hepatitis D virus in serum. Clin. Vaccine Immunol. 2016, 23, 732–734. [Google Scholar] [CrossRef] [PubMed]
- Brichler, S.; le Gal, F.; Butt, A.; Chevret, S.; Gordien, E. Commercial real-time reverse transcriptase PCR assays can underestimate or fail to quantify hepatitis delta virus viremia. Clin. Gastroenterol. Hepatol. 2013, 11, 734–740. [Google Scholar] [CrossRef] [PubMed]
- Le Gal, F.; Brichler, S.; Sahli, R.; Chevret, S.; Gordien, E. First international external quality assessment for hepatitis delta virus RNA quantification in plasma. Hepatology 2016. [Google Scholar] [CrossRef] [PubMed]
- Olivero, A.; Smedile, A. Hepatitis delta virus diagnosis. Semin. Liver Dis. 2012, 32, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Shattock, A.G.; Morris, M.C. Evaluation of commercial enzyme immunoassays for detection of hepatitis delta antigen and anti-hepatitis delta virus (HDV) and immunoglobulin M anti-HDV antibodies. J. Clin. Microbiol. 1991, 29, 1873–1876. [Google Scholar] [PubMed]
- Rosina, F.; Fabiano, A.; Garripoli, A.; Smedile, A.; Mattalia, A.; Eckart, M.R.; Houghton, M.; Bonino, F. Rabbit-derived anti-HD antibodies for HDag immunoblotting. J. Hepatol. 1991, 13, S130–S133. [Google Scholar] [CrossRef]
- Wang, J.G.; Cullen, J.; Lemon, S.M. Immunoblot analysis demonstrates that the large and small forms of hepatitis delta virus antigen have different C-terminal amino acid sequences. J. Gen. Virol. 1992, 73, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Yi, Y.; Su, Q.; Qiu, F.; Jia, Z.; Bi, S. High expression of small hepatitis D antigen in Escherichia coli and ELISA for diagnosis of hepatitis D virus. J. Virol. Methods 2014, 197, 34–38. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.; Alves, C.; Gudima, S.; Taylor, J. Intracellular localization of hepatitis delta virus proteins in the presence and absence of viral RNA accumulation. J. Virol. 2009, 83, 6457–6463. [Google Scholar] [CrossRef] [PubMed]
- Lazinski, D.W.; Taylor, J.M. Relating structure to function in the hepatitis delta virus antigen. J. Virol. 1993, 67, 2672–2680. [Google Scholar] [PubMed]
- National Research Council (U.S.); Committee for the Update of the Guide for the Care and Use of Laboratory Animals; Institute for Laboratory Animal Research (U.S.); National Academies Press (U.S.). Guide for the Care and Use of Laboratory Animals, 8th ed.; National Academies Press: Washington, WA, USA, 2011; pp. 25–220. [Google Scholar]
- Ivanov, A.V.; Smirnova, O.A.; Petrushanko, I.Y.; Ivanova, O.N.; Karpenko, I.L.; Alekseeva, E.; Sominskaya, I.; Makarov, A.A.; Bartosch, B.; Kochetkov, S.N.; et al. HCV core protein uses multiple mechanisms to induce oxidative stress in human hepatoma Huh7 cells. Viruses 2015, 7, 2745–2770. [Google Scholar] [CrossRef] [PubMed]
- Smirnova, O.A.; Ivanova, O.N.; Bartosch, B.; Valuev-Elliston, V.T.; Mukhtarov, F.; Kochetkov, S.N.; Ivanov, A.V. Hepatitis C virus NS5A protein triggers oxidative stress by inducing NADPH oxidases 1 and 4 and cytochrome P450 2E1. Oxid. Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]


© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tunitskaya, V.L.; Eliseeva, O.V.; Valuev-Elliston, V.T.; Tyurina, D.A.; Zakirova, N.F.; Khomich, O.A.; Kalis, M.; Latyshev, O.E.; Starodubova, E.S.; Ivanova, O.N.; et al. Prokaryotic Expression, Purification and Immunogenicity in Rabbits of the Small Antigen of Hepatitis Delta Virus. Int. J. Mol. Sci. 2016, 17, 1721. https://doi.org/10.3390/ijms17101721
Tunitskaya VL, Eliseeva OV, Valuev-Elliston VT, Tyurina DA, Zakirova NF, Khomich OA, Kalis M, Latyshev OE, Starodubova ES, Ivanova ON, et al. Prokaryotic Expression, Purification and Immunogenicity in Rabbits of the Small Antigen of Hepatitis Delta Virus. International Journal of Molecular Sciences. 2016; 17(10):1721. https://doi.org/10.3390/ijms17101721
Chicago/Turabian StyleTunitskaya, Vera L., Olesja V. Eliseeva, Vladimir T. Valuev-Elliston, Daria A. Tyurina, Natalia F. Zakirova, Olga A. Khomich, Martins Kalis, Oleg E. Latyshev, Elizaveta S. Starodubova, Olga N. Ivanova, and et al. 2016. "Prokaryotic Expression, Purification and Immunogenicity in Rabbits of the Small Antigen of Hepatitis Delta Virus" International Journal of Molecular Sciences 17, no. 10: 1721. https://doi.org/10.3390/ijms17101721
APA StyleTunitskaya, V. L., Eliseeva, O. V., Valuev-Elliston, V. T., Tyurina, D. A., Zakirova, N. F., Khomich, O. A., Kalis, M., Latyshev, O. E., Starodubova, E. S., Ivanova, O. N., Kochetkov, S. N., Isaguliants, M. G., & Ivanov, A. V. (2016). Prokaryotic Expression, Purification and Immunogenicity in Rabbits of the Small Antigen of Hepatitis Delta Virus. International Journal of Molecular Sciences, 17(10), 1721. https://doi.org/10.3390/ijms17101721







