Microalgae Cultivation on Anaerobic Digestate of Municipal Wastewater, Sewage Sludge and Agro-Waste
Abstract
:1. Introduction
2. Results
2.1. Production of Anaerobic Digestates
Nutrient Composition of Digestates
2.2. Growth of Micoralgae in Solid Medium
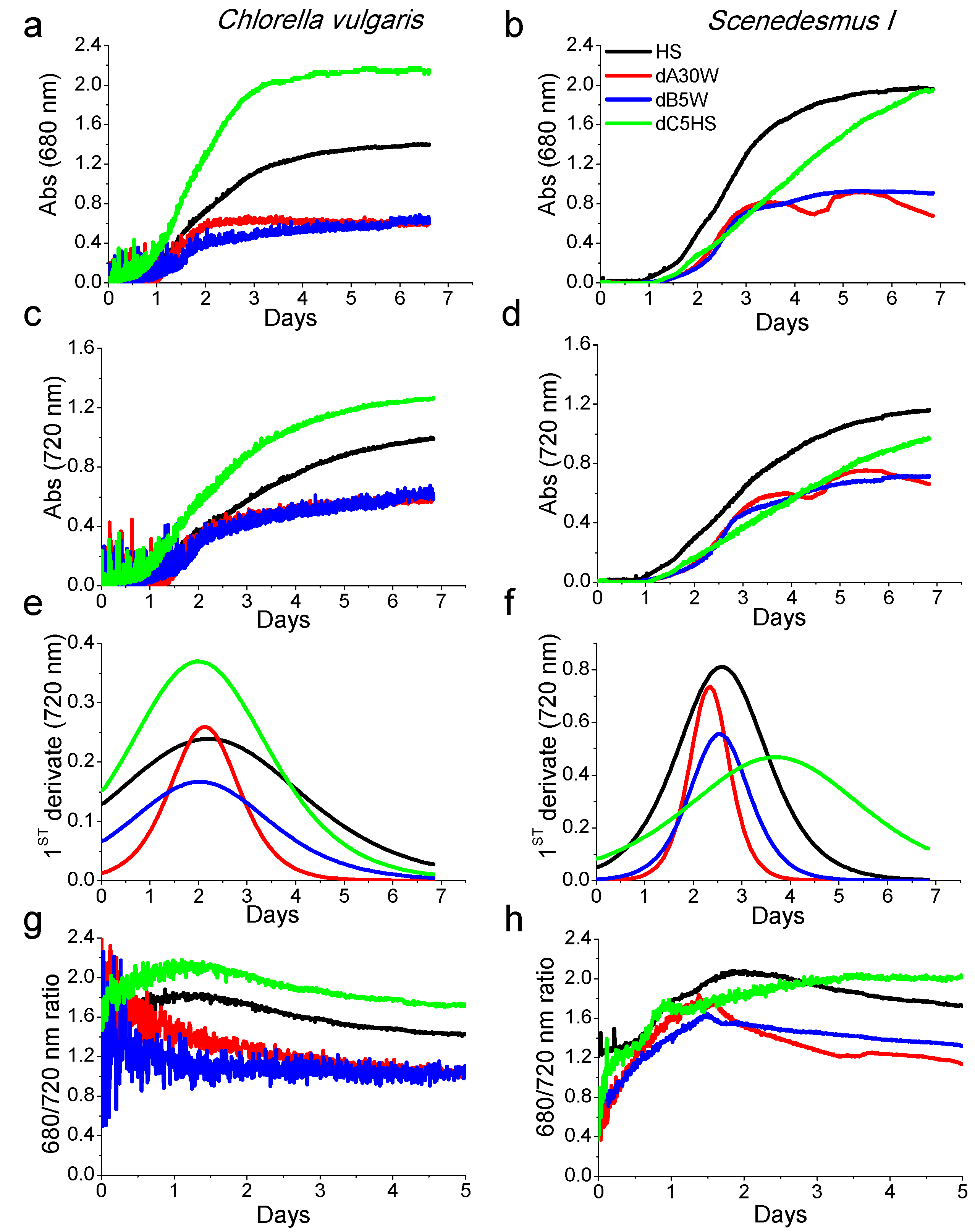
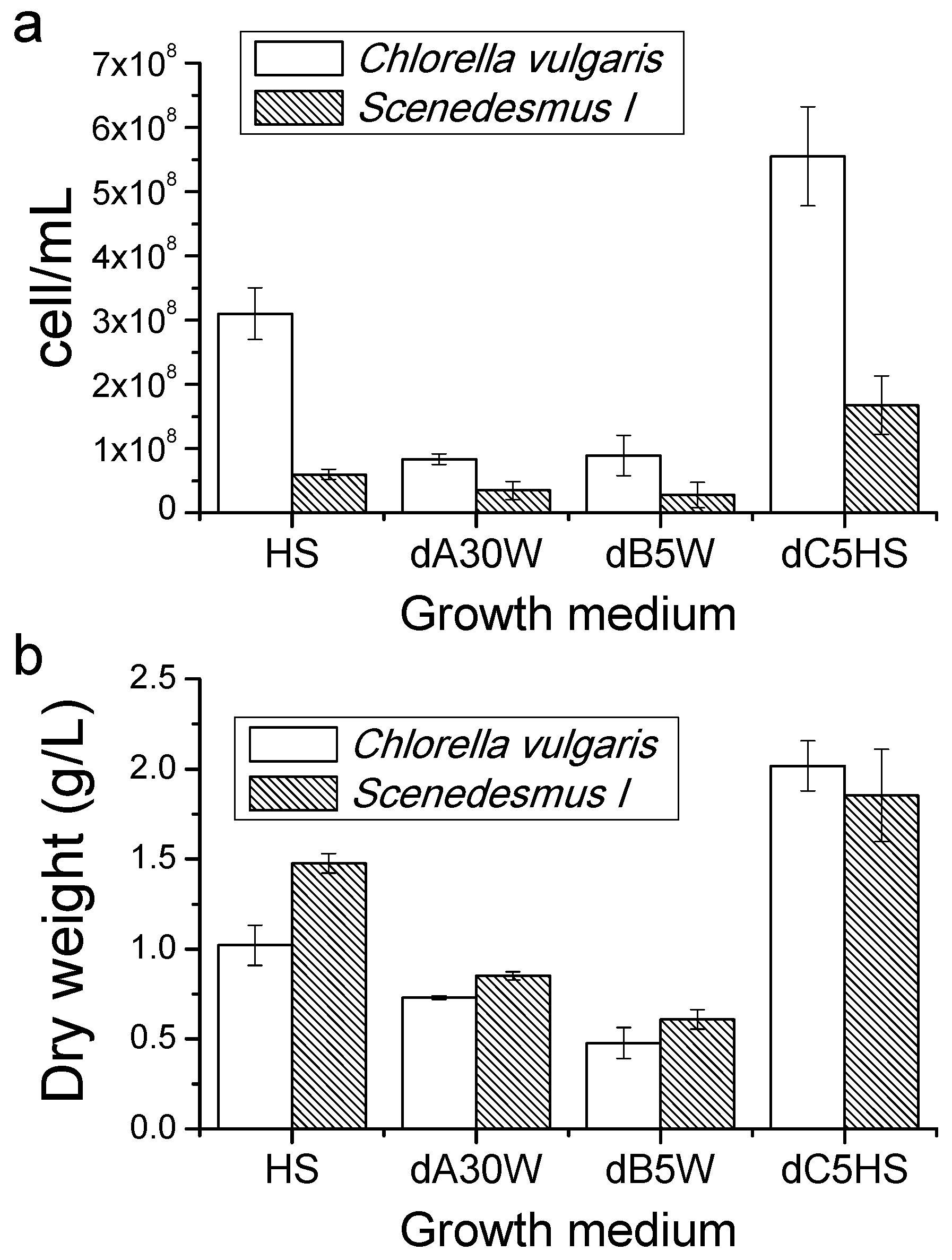
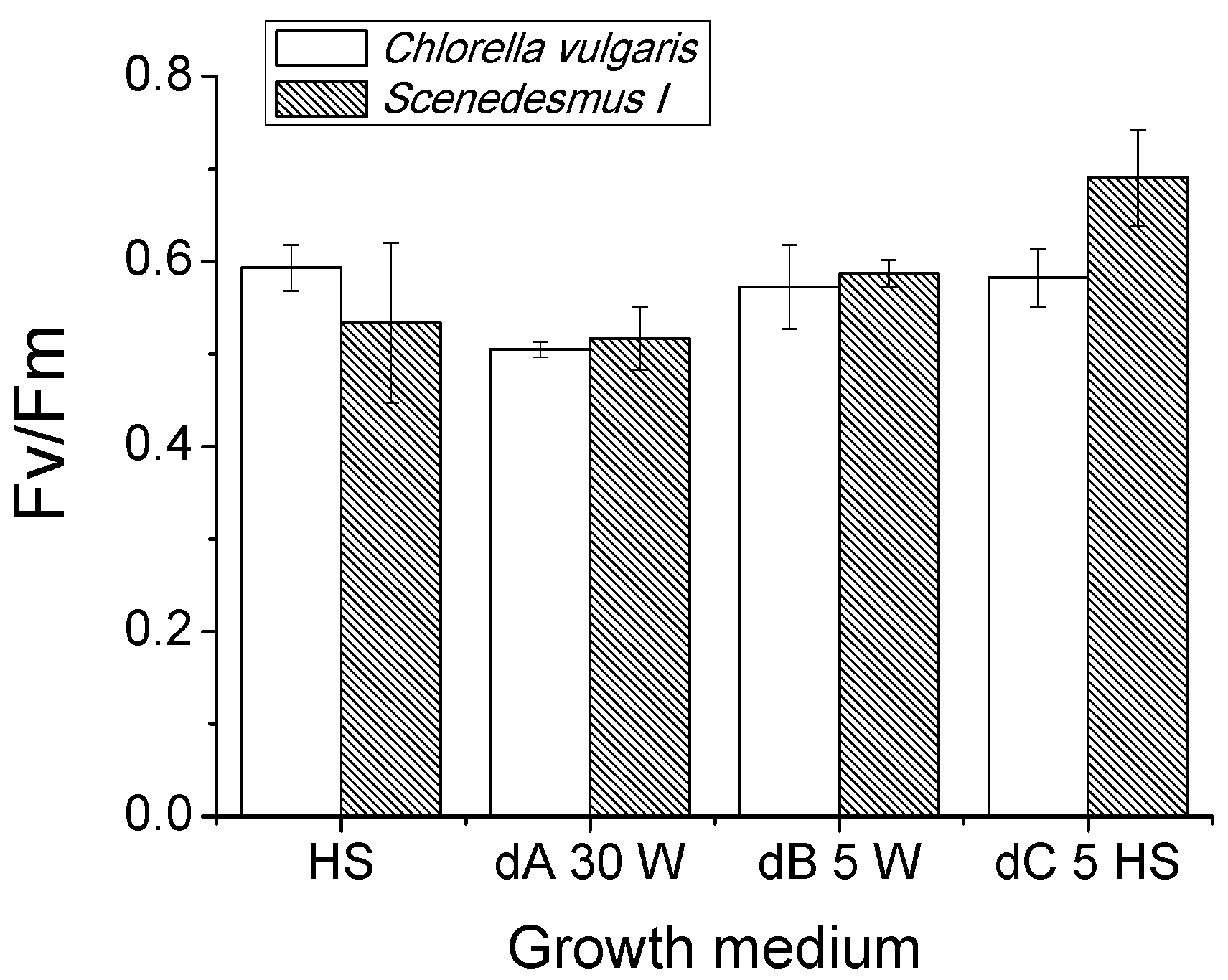
2.3. Growth Tests in Closed Photobioreactors
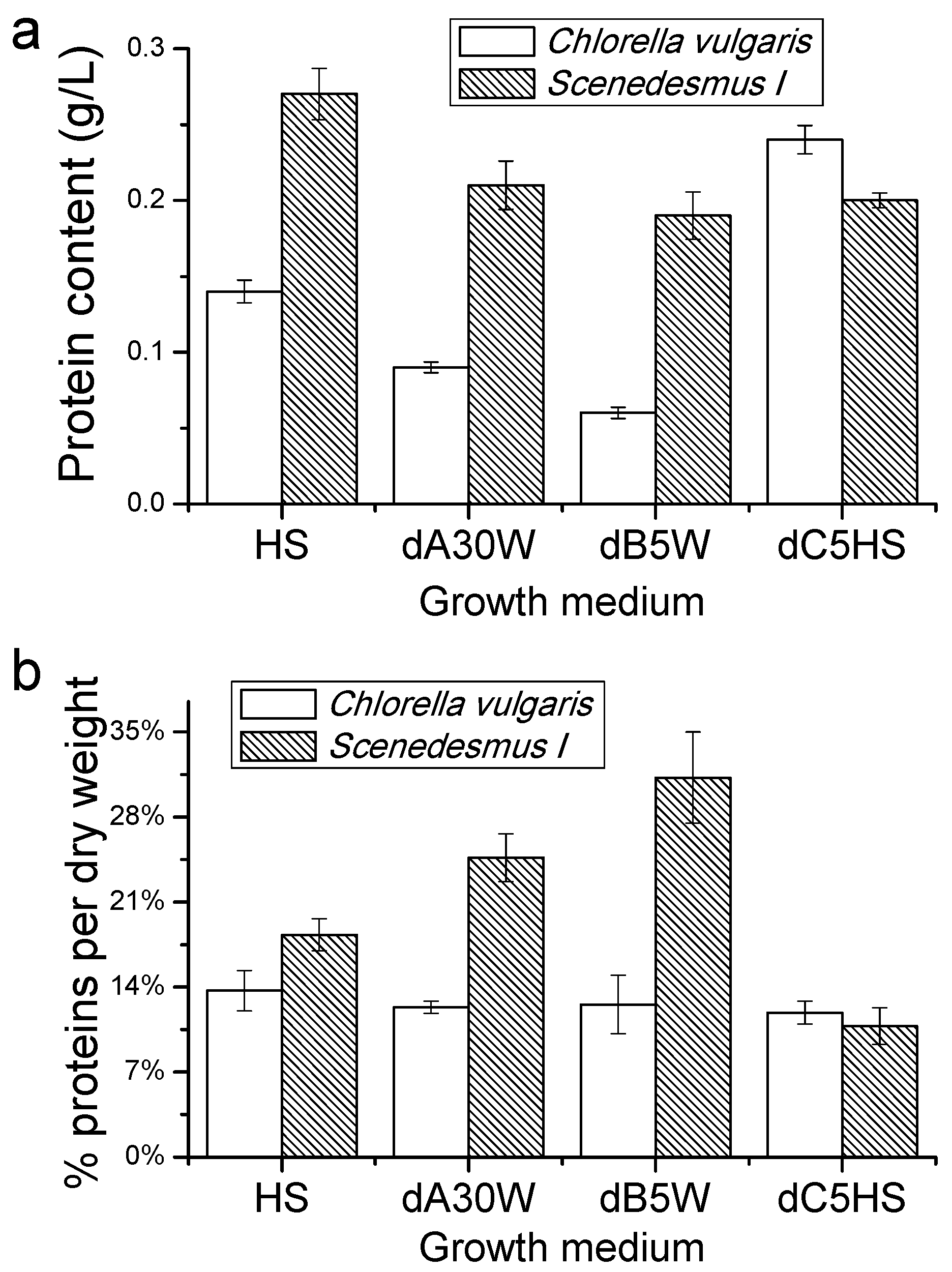
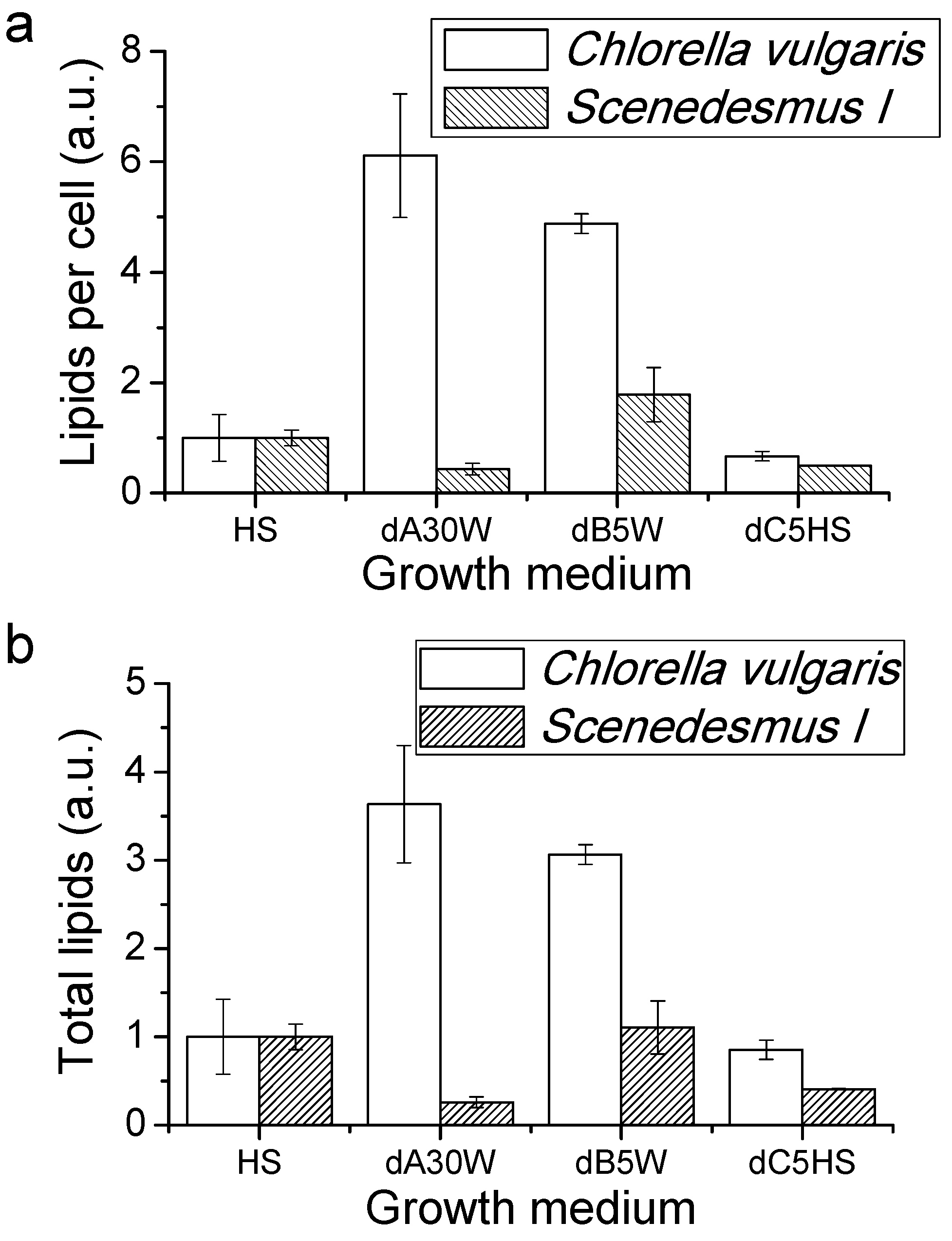
Proteins, Lipids and Pigment Accumulation in Photobioreactor Cultivation
3. Discussion
4. Materials and Methods
4.1. Digestate Production
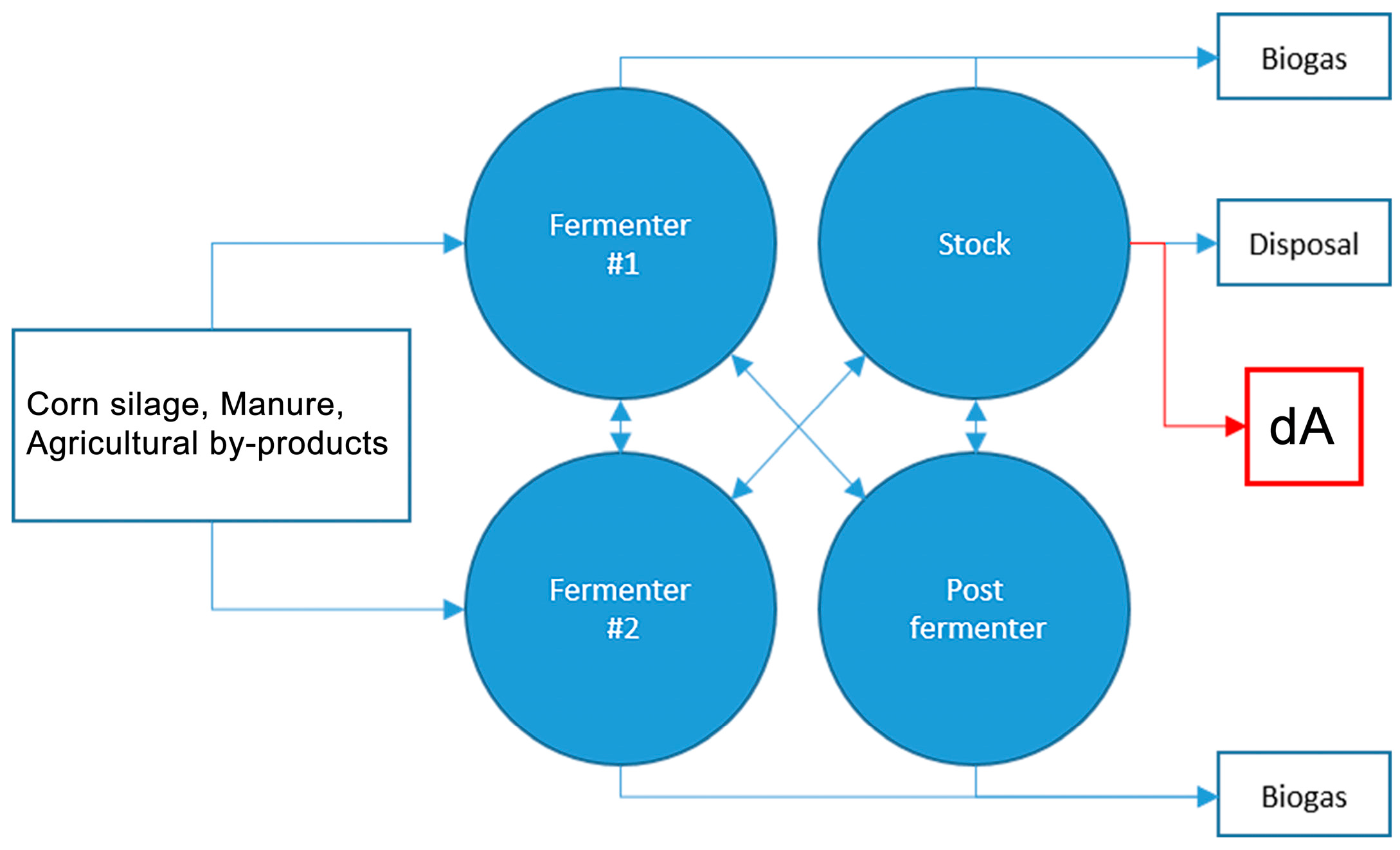
4.1.1. Substrate dA: Anaerobic Digestate from Agro-Waste
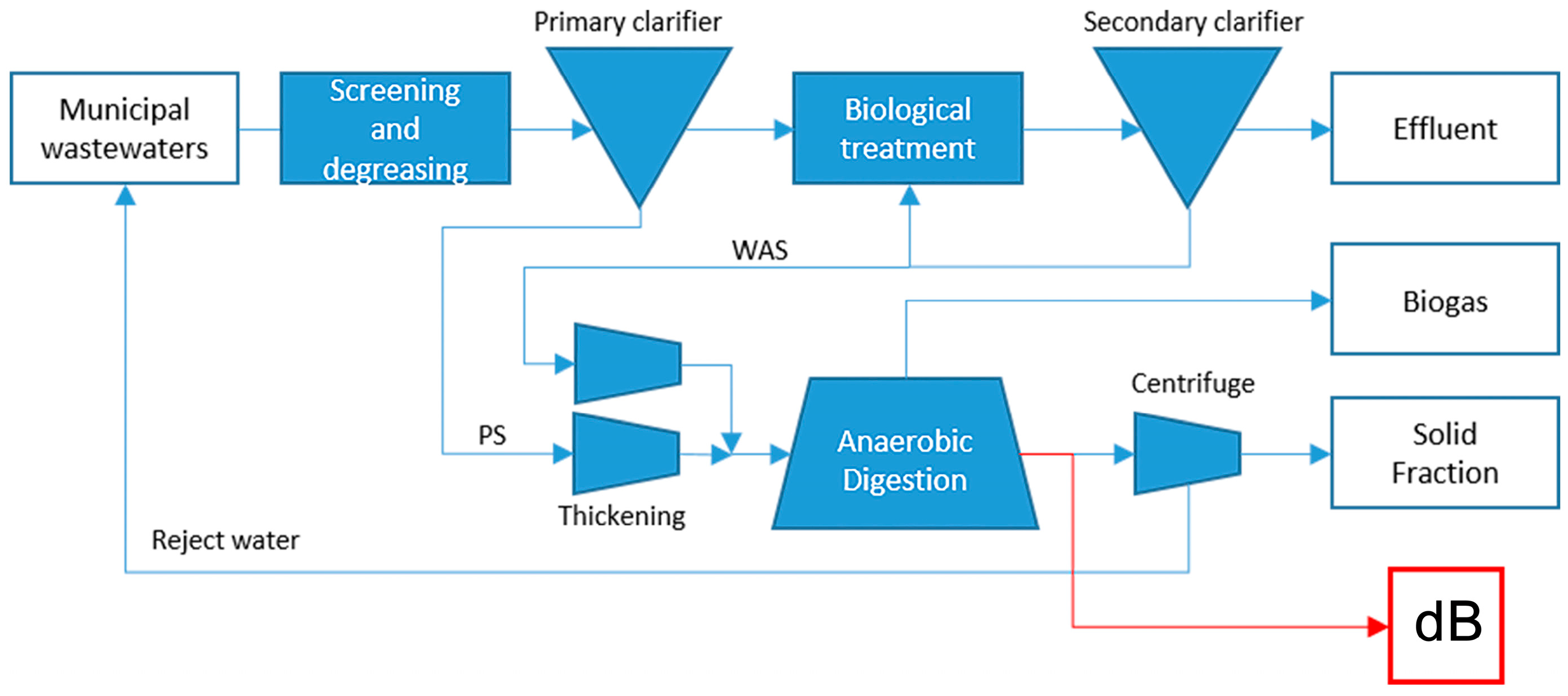
4.1.2. Substrate dB: Anaerobic Digestate from Sewage Sludge
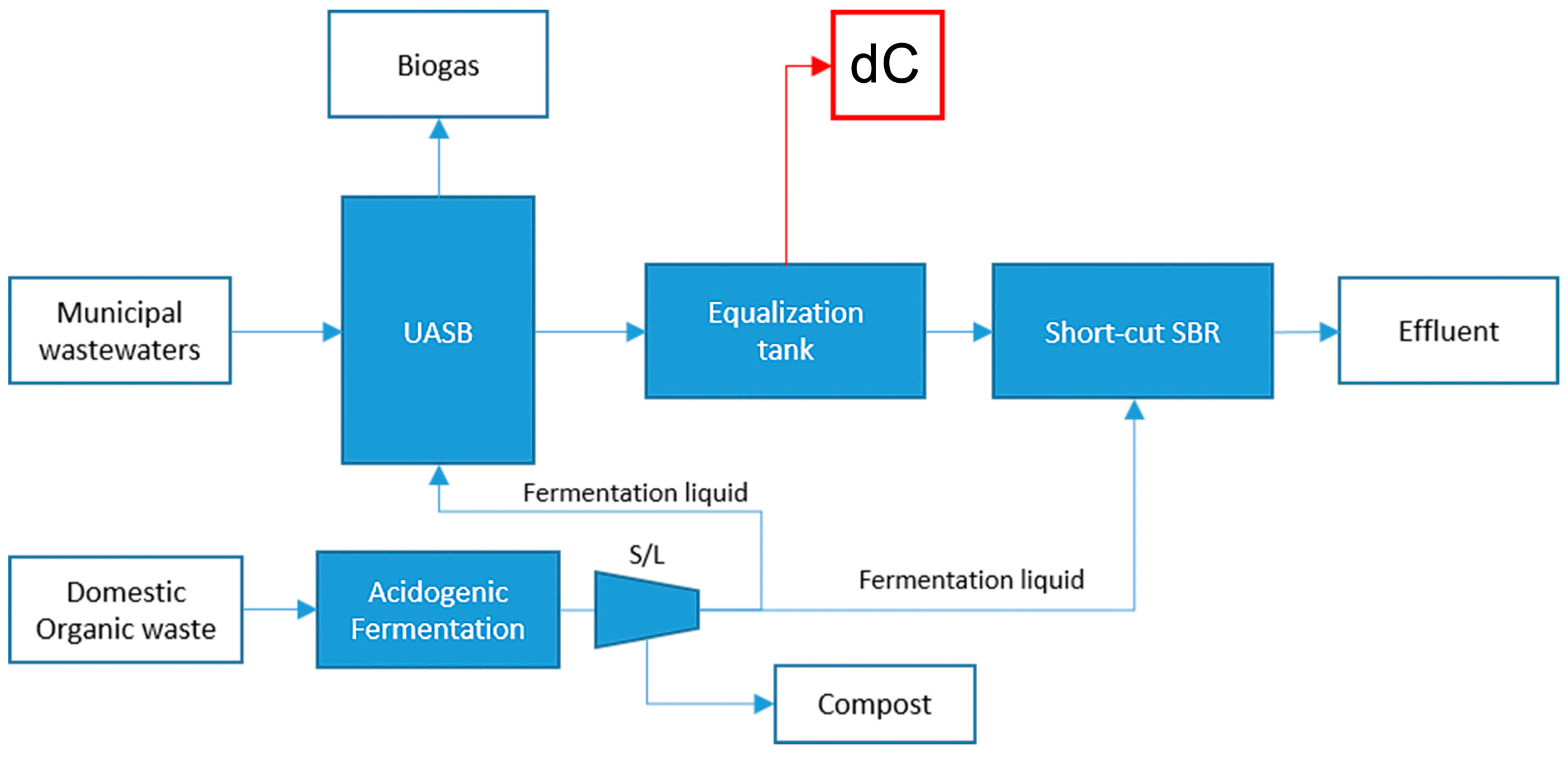
4.1.3. Substrate dC: Anaerobic Effluent from Municipal Wastewater and Organic Waste
4.2. Microalgal Cultivation
4.3. Fluorescence Measurement
4.4. Protein Quantification
4.5. Nile Red Assay
4.6. Pigment Extraction
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gangl, D.; Zedler, J.A.Z.; Rajakumar, P.D.; Martinez, E.M.R.; Riseley, A.; Włodarczyk, A.; Purton, S.; Sakuragi, Y.; Howe, C.J.; Jensen, P.E.; et al. Biotechnological exploitation of microalgae. J. Exp. Bot. 2015, 66, 6975–6990. [Google Scholar] [CrossRef] [PubMed]
- Koller, M.; Salerno, A.; Tuffner, P.; Koinigg, M.; Böchzelt, H.; Schober, S.; Pieber, S.; Schnitzer, H.; Mittelbach, M.; Braunegg, G. Characteristics and potential of micro algal cultivation strategies: A review. J. Clean. Prod. 2012, 37, 377–388. [Google Scholar] [CrossRef]
- Benemann, J.R. Feasibility analysis of photobiological hydrogen production. Int. J. Hydrogen Energy 1997, 22, 979–987. [Google Scholar] [CrossRef]
- Oswald, W.J.; Golueke, C.G. Biological transformation of solar energy. In Advances in Applied Microbiology; Wayne, W.U., Ed.; Academic Press: Waltham, MA, USA, 1960; Volume 2, pp. 223–262. [Google Scholar]
- Fernandes, B.D.; Mota, A.; Teixeira, J.A.; Vicente, A.A. Continuous cultivation of photosynthetic microorganisms: Approaches, applications and future trends. Biotechnol. Adv. 2015, 33, 1228–1245. [Google Scholar] [CrossRef] [PubMed]
- Anemaet, I.G.; Bekker, M.; Hellingwerf, K.J. Algal photosynthesis as the primary driver for a sustainable development in energy, feed, and food production. Mar. Biotechnol. 2010, 12, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Chisti, Y. Biodiesel from microalgae. Biotechnol. Adv. 2007, 25, 294–306. [Google Scholar] [CrossRef] [PubMed]
- Ghirardi, M. Microalgae: A green source of renewable H2. Trends Biotechnol. 2000, 18, 506–511. [Google Scholar] [CrossRef]
- Borowitzka, M.A. High-value products from microalgae—Their development and commercialisation. J. Appl. Phycol. 2013, 25, 743–756. [Google Scholar] [CrossRef]
- Koller, M.; Muhr, A.; Braunegg, G. Microalgae as versatile cellular factories for valued products. Algal Res. 2014, 6, 52–63. [Google Scholar] [CrossRef]
- Kazamia, E.; Smith, A.G. Assessing the environmental sustainability of biofuels. Trends Plant Sci. 2014, 19, 615–618. [Google Scholar] [CrossRef] [PubMed]
- Rodolfi, L.; Chini Zittelli, G.; Bassi, N.; Padovani, G.; Biondi, N.; Bonini, G.; Tredici, M.R. Microalgae for oil: Strain selection, induction of lipid synthesis and outdoor mass cultivation in a low-cost photobioreactor. Biotechnol. Bioeng. 2009, 102, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Taparia, T.; Mvss, M.; Mehrotra, R.; Shukla, P.; Mehrotra, S. Developments and challenges in biodiesel production from microalgae: A review. Biotechnol. Appl. Biochem. 2015. [Google Scholar] [CrossRef] [PubMed]
- Medipally, S.R.; Yusoff, F.M.; Banerjee, S.; Shariff, M. Microalgae as sustainable renewable energy feedstock for biofuel production. BioMed Res. Int. 2015, 2015, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Hannon, M.; Gimpel, J.; Tran, M.; Rasala, B.; Mayfield, S. Biofuels from algae: Challenges and potential. Biofuels 2010, 1, 763–784. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Mallick, N. Microalga scenedesmus obliquus as a potential source for biodiesel production. Appl. Microbial. Biotechnol. 2009, 84, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Arbib, Z.; Ruiz, J.; Álvarez-Díaz, P.; Garrido-Pérez, C.; Barragan, J.; Perales, J.A. Photobiotreatment: Influence of nitrogen and phosphorus ratio in wastewater on growth kinetics of scenedesmus obliquus. Int. J. Phytoremediat. 2013, 15, 774–788. [Google Scholar] [CrossRef] [PubMed]
- Ruiz, J.; Arbib, Z.; Alvarez-Díaz, P.D.; Garrido-Pérez, C.; Barragán, J.; Perales, J.A. Influence of light presence and biomass concentration on nutrient kinetic removal from urban wastewater by scenedesmus obliquus. J. Biotechnol. 2014, 178, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Gouveia, L.; Oliveira, A.C. Microalgae as a raw material for biofuels production. J. Ind. Microbial. Biotechnol. 2009, 36, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Zheng, Y.; Yu, L.; Chen, S. High productivity cultivation of a heat-resistant microalga chlorella sorokiniana for biofuel production. Bioresour. Technol. 2013, 131, 60–67. [Google Scholar] [CrossRef] [PubMed]
- Nirupama-Mallick, S.M.; Amit, K.S.; Moumita, B.; Archana, D. Green microalga chlorella vulgaris as a potential feedstock for biodiesel. J. Chem. Technol. Biotechnol. 2012, 87, 137–145. [Google Scholar] [CrossRef]
- Liang, Y.; Sarkany, N.; Cui, Y. Biomass and lipid productivities of chlorella vulgaris under autotrophic, heterotrophic and mixotrophic growth conditions. Biotechnol. Lett. 2009, 31, 1043–1049. [Google Scholar] [CrossRef] [PubMed]
- Biondi, N.; Bassi, N.; Chini Zittelli, G.; de Faveri, D.; Giovannini, A.; Rodolfi, L.; Allevi, C.; Macrì, C.; Tredici, M.R. Nannochloropsis sp. F&m-m24: Oil production, effect of mixing on productivity and growth in an industrial wastewater. Environ. Prog. Sustain. Energy 2013, 32, 846–853. [Google Scholar]
- Simionato, D.; Block, M.A.; La Rocca, N.; Jouhet, J.; Maréchal, E.; Finazzi, G.; Morosinotto, T. The response of nannochloropsis gaditana to nitrogen starvation includes de novo biosynthesis of triacylglycerols, a decrease of chloroplast galactolipids, and reorganization of the photosynthetic apparatus. Eukaryot. Cell 2013, 12, 665–676. [Google Scholar] [CrossRef] [PubMed]
- Markou, G.; Vandamme, D.; Muylaert, K. Microalgal and cyanobacterial cultivation: The supply of nutrients. Water Res. 2014, 65, 186–202. [Google Scholar] [CrossRef] [PubMed]
- Peccia, J.; Haznedaroglu, B.; Gutierrez, J.; Zimmerman, J.B. Nitrogen supply is an important driver of sustainable microalgae biofuel production. Trends Biotechnol. 2013, 31, 134–138. [Google Scholar] [CrossRef] [PubMed]
- Oswald, W.J.; Gotaas, H.B.; Golueke, C.G.; Kellen, W.R.; Gloyna, E.F.; Hermann, E.R. Algae in waste treatment [with discussion]. Sew. Ind. Wastes 1957, 29, 437–457. [Google Scholar]
- Pittman, J.K.; Dean, A.P.; Osundeko, O. The potential of sustainable algal biofuel production using wastewater resources. Bioresour. Technol. 2011, 102, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Martínez, M. Nitrogen and phosphorus removal from urban wastewater by the microalga Scenedesmus obliquus. Bioresour. Technol. 2000, 73, 263–272. [Google Scholar] [CrossRef]
- Mata-Alvarez, J.; Macé, S.; Llabrés, P. Anaerobic digestion of organic solid wastes. An overview of research achievements and perspectives. Bioresour. Technol. 2000, 74, 3–16. [Google Scholar] [CrossRef]
- Appels, L.; Baeyens, J.; Degrève, J.; Dewil, R. Principles and potential of the anaerobic digestion of waste-activated sludge. Prog. Energy Combust. Sci. 2008, 34, 755–781. [Google Scholar] [CrossRef]
- Weiland, P. Biogas production: Current state and perspectives. Appl. Microbiol. Biotechnol. 2010, 85, 849–860. [Google Scholar] [CrossRef] [PubMed]
- Katsou, E.; Malamis, S.; Frison, N.; Fatone, F. Coupling the treatment of low strength anaerobic effluent with fermented biowaste for nutrient removal via nitrite. J. Environ. Manag. 2015, 149, 108–117. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, K.; Johnson, G.N. Chlorophyll fluorescence—A practical guide. J. Exp. Bot. 2000, 51, 659–668. [Google Scholar] [CrossRef] [PubMed]
- Bonente, G.; Pippa, S.; Castellano, S.; Bassi, R.; Ballottari, M. Acclimation of chlamydomonas reinhardtii to different growth irradiances. J. Biol. Chem. 2012, 287, 5833–5847. [Google Scholar] [CrossRef] [PubMed]
- Rumin, J.; Bonnefond, H.; Saint-Jean, B.; Rouxel, C.; Sciandra, A.; Bernard, O.; Cadoret, J.-P.; Bougaran, G. The use of fluorescent nile red and bodipy for lipid measurement in microalgae. Biotechnol. Biofuels 2015, 8, 42–42. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.K.; Schuhmann, H.; Schenk, P.M. High lipid induction in microalgae for biodiesel production. Energies 2012, 5, 1532–1553. [Google Scholar] [CrossRef]
- Gilmore, A.M.; Yamamoto, H.Y. Resolution of lutein and zeaxanthin using a non-endcapped, lightly carbon-loaded C18 high-performance liquid chromatographic column. J. Chromatogr. A 1991, 543, 137–145. [Google Scholar] [CrossRef]
- Bohutskyi, P.; Liu, K.; Nasr, L.K.; Byers, N.; Rosenberg, J.N.; Oyler, G.A.; Betenbaugh, M.J.; Bouwer, E.J. Bioprospecting of microalgae for integrated biomass production and phytoremediation of unsterilized wastewater and anaerobic digestion centrate. Appl. Microbiol. Biotechnol. 2015, 99, 6139–6154. [Google Scholar] [CrossRef] [PubMed]
- Ledda, C.; Romero Villegas, G.I.; Adani, F.; Acién Fernández, F.G.; Molina Grima, E. Utilization of centrate from wastewater treatment for the outdoor production of nannochloropsis gaditana biomass at pilot-scale. Algal Res. 2015, 12, 17–25. [Google Scholar] [CrossRef]
- Cai, T.; Park, S.Y.; Racharaks, R.; Li, Y. Cultivation of nannochloropsis salina using anaerobic digestion effluent as a nutrient source for biofuel production. Appl. Energy 2013, 108, 486–492. [Google Scholar] [CrossRef]
- Sheets, J.P.; Ge, X.; Park, S.Y.; Li, Y. Effect of outdoor conditions on nannochloropsis salina cultivation in artificial seawater using nutrients from anaerobic digestion effluent. Bioresource Technol. 2014, 152, 154–161. [Google Scholar] [CrossRef] [PubMed]
- Sueoka, N. Mitotic replication of deoxyribonucleic acid in chlamydomonas reinhardi. Proc. Nat. Acad. Sci. USA 1960, 46, 83–91. [Google Scholar] [CrossRef]
- Ferrante, P.; Ballottari, M.; Bonente, G.; Giuliano, G.; Bassi, R. Lhcbm1 and lhcbm2/7 polypeptides, components of major LHCII complex, have distinct functional roles in photosynthetic antenna system of Chlamydomonas reinhardtii. J. Biol. Chem. 2012, 287, 16276–16288. [Google Scholar] [CrossRef] [PubMed]
- Kropat, J.; Hong-Hermesdorf, A.; Casero, D.; Ent, P.; Castruita, M.; Pellegrini, M.; Merchant, S.S.; Malasarn, D. A revised mineral nutrient supplement increases biomass and growth rate in chlamydomonas reinhardtii. Plant J. 2011, 66, 770–780. [Google Scholar] [CrossRef] [PubMed]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; Cohen-Bazire, G. Purification and properties of unicellular blue-green algae (order chroococcales). Bacteriol. Rev. 1971, 35, 171–205. [Google Scholar] [PubMed]
- Lowrey, J.; Brooks, M.; McGinn, P. Heterotrophic and mixotrophic cultivation of microalgae for biodiesel production in agricultural wastewaters and associated challenges—A critical review. J. Appl. Phycol. 2015, 27, 1485–1498. [Google Scholar] [CrossRef]
- Hu, B.; Min, M.; Zhou, W.; Du, Z.; Mohr, M.; Chen, P.; Zhu, J.; Cheng, Y.; Liu, Y.; Ruan, R. Enhanced mixotrophic growth of Microalga chlorella sp. On pretreated swine manure for simultaneous biofuel feedstock production and nutrient removal. Bioresour. Technol. 2012, 126C, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Laliberte, G.; Delanoue, J. Autotrophic, heterotrophic, and mixotrophic growth of chlamydomonas-humicola (chlorophyceae) on acetate. J. Phycol. 1993, 29, 612–620. [Google Scholar] [CrossRef]
- Combres, C.; Laliberte, G.; Reyssac, J.; Delanoue, J. Effect of acetate on growth and ammonium uptake in the microalga Scenedesmus-obliquus. Physiol. Plant. 1994, 91, 729–734. [Google Scholar] [CrossRef]
- Chen, F.; Johns, M. Heterotrophic growth of chlamydomonas reinhardtii on acetate in chemostat culture. Process Biochem. 1996, 31, 601–604. [Google Scholar] [CrossRef]
- Heredia-Arroyo, T.; Wei, W.; Hu, B. Oil accumulation via heterotrophic/mixotrophic chlorella protothecoides. Appl. Biochem. Biotechnol. 2010, 162, 1978–1995. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Xiong, H.; Hui, Z.; Zeng, X. Mixotrophic cultivation of chlorella pyrenoidosa with diluted primary piggery wastewater to produce lipids. Bioresour. Technol. 2012, 104, 215–220. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, N.; Noel, E.A.; Barnes, A.; Watson, A.; Rosenberg, J.N.; Erickson, G.; Oyler, G.A. Characterization of three chlorella sorokiniana strains in anaerobic digested effluent from cattle manure. Bioresour. Technol. 2013, 150, 377–386. [Google Scholar] [CrossRef] [PubMed]
- Åkerström, A.M.; Mortensen, L.M.; Rusten, B.; Gislerød, H.R. Biomass production and nutrient removal by chlorella sp. As affected by sludge liquor concentration. J. Environ. Manag. 2014, 144, 118–124. [Google Scholar] [CrossRef] [PubMed]
- Scibilia, L.; Girolomoni, L.; Berteotti, S.; Alboresi, A.; Ballottari, M. Photosynthetic response to nitrogen starvation and high light in haematococcus pluvialis. Algal Res. 2015, 12, 170–181. [Google Scholar] [CrossRef]
- Qu, C.-B.; Wu, Z.-Y.; Shi, X.-M. Phosphate assimilation by chlorella and adjustment of phosphate concentration in basal medium for its cultivation. Biotechnol. Lett. 2008, 30, 1735–1740. [Google Scholar] [CrossRef] [PubMed]
- Mulders, K.J.M.; Lamers, P.P.; Martens, D.E.; Wijffels, R.H. Phototrophic pigment production with microalgae: Biological constraints and opportunities. J. Phycol. 2014, 50, 229–242. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Matsumura, R.; Hoshino, N.; Tsuzuki, M.; Sato, N. Responsibility of regulatory gene expression and repressed protein synthesis for triacylglycerol accumulation on sulfur-starvation in Chlamydomonas reinhardtii. Front. Plant Sci. 2014, 5, 444. [Google Scholar] [CrossRef] [PubMed]
- Guckert, J.B; Cooksey, K.E. Triglyceride accumulation and fatty acid profile changes in chlorella (chlorophyta) during high pH-induced cell cycle inhibition1. J. Phycol. 1990, 26, 72–79. [Google Scholar] [CrossRef]
- Gardner, R.; Peters, P.; Peyton, B.; Cooksey, K.E. Medium pH and nitrate concentration effects on accumulation of triacylglycerol in two members of the chlorophyta. J. Appl. Phycol. 2010, 23, 1005–1016. [Google Scholar] [CrossRef]
- Park, J.; Jin, H.-F.; Lim, B.-R.; Park, K.-Y.; Lee, K. Ammonia removal from anaerobic digestion effluent of livestock waste using green alga scenedesmus sp. Bioresour. Technol. 2010, 101, 8649–8657. [Google Scholar] [CrossRef] [PubMed]
- Zhao, B.; Ma, J.; Zhao, Q.; Laurens, L.; Jarvis, E.; Chen, S.; Frear, C. Efficient anaerobic digestion of whole microalgae and lipid-extracted microalgae residues for methane energy production. Bioresour. Technol. 2014, 161, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Bohutskyi, P.; Chow, S.; Ketter, B.; Betenbaugh, M.J.; Bouwer, E.J. Prospects for methane production and nutrient recycling from lipid extracted residues and whole nannochloropsis salina using anaerobic digestion. Appl. Energy 2015, 154, 718–731. [Google Scholar] [CrossRef]
- Bohutskyi, P.; Ketter, B.; Chow, S.; Adams, K.J.; Betenbaugh, M.J.; Allnutt, F.C.T.; Bouwer, E.J. Anaerobic digestion of lipid-extracted auxenochlorella protothecoides biomass for methane generation and nutrient recovery. Bioresour. Technol. 2015, 183, 229–239. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Gao, M.; Hua, D.; Zhang, J.; Zhao, Y.; Mu, H.; Xu, H.; Liang, X.; Jin, F.; Zhang, X. One-stage and two-stage anaerobic digestion of lipid-extracted algae. Ann. Microbiol. 2015, 65, 1465–1471. [Google Scholar] [CrossRef]
- Ward, A.J.; Lewis, D.M.; Green, F.B. Anaerobic digestion of algae biomass: A review. Algal Res. 2014, 5, 204–214. [Google Scholar] [CrossRef]
- Sialve, B.; Bernet, N.; Bernard, O. Anaerobic digestion of microalgae as a necessary step to make microalgal biodiesel sustainable. Biotechnol. Adv. 2009, 27, 409–416. [Google Scholar] [CrossRef] [PubMed]
- Golueke, C.G.; Oswald, W.J.; Gotaas, H.B. Anaerobic digestion of algae. Appl. Microbiol. 1957, 5, 47–55. [Google Scholar] [PubMed]
- Mussgnug, J.H.; Klassen, V.; Schlüter, A.; Kruse, O. Microalgae as substrates for fermentative biogas production in a combined biorefinery concept. J. Biotechnol. 2010, 150, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.; Bolzonella, D.; Pavan, P.; Cavinato, C.; Cecchi, F. Co-digestion of livestock effluents, energy crops and agro-waste: Feeding and process optimization in mesophilic and thermophilic conditions. Bioresour. Technol. 2013, 128, 612–618. [Google Scholar] [CrossRef] [PubMed]
- Cavinato, C.; Fatone, F.; Bolzonella, D.; Pavan, P. Thermophilic anaerobic co-digestion of cattle manure with agro-wastes and energy crops: Comparison of pilot and full scale experiences. Bioresour. Technol. 2010, 101, 545–550. [Google Scholar] [CrossRef] [PubMed]
- Simionato, D.; Sforza, E.; Corteggiani Carpinelli, E.; Bertucco, A.; Giacometti, G.M.; Morosinotto, T. Acclimation of nannochloropsis gaditana to different illumination regimes: Effects on lipids accumulation. Bioresour. Technol. 2011, 102, 6026–6032. [Google Scholar] [CrossRef] [PubMed]
- Guillard, R.R.L. Culture of phytoplankton for feeding marine invertebrates. In Culture of Marine Invertebrate Animals: Proceedings—1st Conference on Culture of Marine Invertebrate Animals Greenport; Smith, W.L., Chanley, M.H., Eds.; Springer: Boston, MA, USA, 1975; pp. 29–60. [Google Scholar]
- Berteotti, S.; Ballottari, M.; Bassi, R. Increased biomass productivity in green algae by tuning non-photochemical quenching. Sci. Rep. 2016, 6. [Google Scholar] [CrossRef] [PubMed]
- Kaňa, R.; Špundová, M.; Ilı́k, P.; Lazár, D.; Klem, K.; Tomek, P.; Nauš, J.; Prášil, O. Effect of herbicide clomazone on photosynthetic processes in primary barley (Hordeum vulgare l.) leaves. Pestic. Biochem. Physiol. 2004, 78, 161–170. [Google Scholar] [CrossRef]
- Malasarn, D.; Kropat, J.; Hsieh, S.I.; Finazzi, G.; Casero, D.; Loo, J.A.; Pellegrini, M.; Wollman, F.-A.; Merchant, S.S. Zinc deficiency impacts CO2 assimilation and disrupts copper homeostasis in Chlamydomonas reinhardtii. J. Biol. Chem. 2013, 288, 10672–10683. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Devanadera, M.C.E.; Roddick, F.A.; Fan, L.; Dalida, M.L.P. Impact of algal organic matter released from microcystis Aeruginosa and chlorella sp. On the fouling of a ceramic microfiltration membrane. Water Res. 2016, 103, 391–400. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhang, C.; Song, L.; Sommerfeld, M.; Hu, Q. A high throughput nile red method for quantitative measurement of neutral lipids in microalgae. J. Microbiol. Meth. 2009, 77, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Cazzaniga, S.; Dall'Osto, L.; Szaub, J.; Scibilia, L.; Ballottari, M.; Purton, S.; Bassi, R. Domestication of the green alga chlorella sorokiniana: Reduction of antenna size improves light-use efficiency in a photobioreactor. Biotechnol. Biofuels 2014, 7, 1–13. [Google Scholar] [CrossRef] [PubMed]
| Parameter | dA | dB | dC | HS |
|---|---|---|---|---|
| TS (g/L) | 94 ± 5 | 28 ± 3 | <0.1 | 2.1 ± 0.1 |
| TVS (g/L) | 65 ± 2 | 15 ± 3 | <0.1 | <LOD |
| COD (gCOD/L) | 74 ± 8 | 18 ± 4 | <0.1 | <LOD |
| Total Nitrogen (mgN/L) | 5218 ± 314 | 2750 ± 112 | 37 ± 6 | 113 ± 5 |
| VFA (acetic acid, mg/L) | <100 | <100 | <20 | <LOD |
| Ammonia (mgN/L) | 2949 ± 197 | 1450 ± 65 | 35 ± 5 | 113 ± 5 |
| Total Phosphorus (mgP/L) | 4870 ± 390 | 480 ± 44 | 6.5 ± 0.9 | 436 ± 15 |
| Orthophosphate (mgP/L) | 85 ± 25 | 30.5 ± 5.2 | 6.2 ± 0.8 | 436 ± 15 |
| CaCO3 (mg/L) | 13,020 ± 750 | 3780 ± 227 | 406 ± 54 | 360 ± 18 |
| pH | 8.2 ± 0.1 | 7.9 ± 0.2 | 7.6 ± 0.5 | 6.9 ± 0.1 |
| Conductivity (mS/cm) | 26 ± 3 | 5.4 ±0.9 | 0.4 ± 0.1 | 3.2 ± 0.1 |
| OD 440 nm (liquid phase) | 13,673 ± 0.05 | 0.459 ± 0.02 | 0.0137 ± 0.01 | 0.002 ± 0.01 |
| OD 680 nm (liquid phase) | 2340 ± 0.02 | 0.221 ± 0.01 | 0.007 ± 0.01 | 0.004 ± 0.01 |
| Strain | Growth Medium | Chl a/Chl b | Car/Chl | Car Per 100 Chl |
|---|---|---|---|---|
| Sce I | HS | 1.74 | 1.25 | 79.86 |
| dA30W | 1.67 | 0.81 | 123.28 | |
| dB5W | 0.73 | 0.68 | 147.63 | |
| dC5HS | 2.84 | 2.62 | 38.14 | |
| Cv | HS | 4.02 | 1.58 | 63.43 |
| dA30W | 2.22 | 0.56 | 177.96 | |
| dB5W | 1.47 | 0.62 | 160.56 | |
| dC5HS | 3.80 | 1.87 | 53.60 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zuliani, L.; Frison, N.; Jelic, A.; Fatone, F.; Bolzonella, D.; Ballottari, M. Microalgae Cultivation on Anaerobic Digestate of Municipal Wastewater, Sewage Sludge and Agro-Waste. Int. J. Mol. Sci. 2016, 17, 1692. https://doi.org/10.3390/ijms17101692
Zuliani L, Frison N, Jelic A, Fatone F, Bolzonella D, Ballottari M. Microalgae Cultivation on Anaerobic Digestate of Municipal Wastewater, Sewage Sludge and Agro-Waste. International Journal of Molecular Sciences. 2016; 17(10):1692. https://doi.org/10.3390/ijms17101692
Chicago/Turabian StyleZuliani, Luca, Nicola Frison, Aleksandra Jelic, Francesco Fatone, David Bolzonella, and Matteo Ballottari. 2016. "Microalgae Cultivation on Anaerobic Digestate of Municipal Wastewater, Sewage Sludge and Agro-Waste" International Journal of Molecular Sciences 17, no. 10: 1692. https://doi.org/10.3390/ijms17101692
APA StyleZuliani, L., Frison, N., Jelic, A., Fatone, F., Bolzonella, D., & Ballottari, M. (2016). Microalgae Cultivation on Anaerobic Digestate of Municipal Wastewater, Sewage Sludge and Agro-Waste. International Journal of Molecular Sciences, 17(10), 1692. https://doi.org/10.3390/ijms17101692









