α6β4 Integrin Genetic Variations (A380T and R1281W) and Breast Cancer Risk in an Argentinian Population
Abstract
:1. Introduction
2. Results
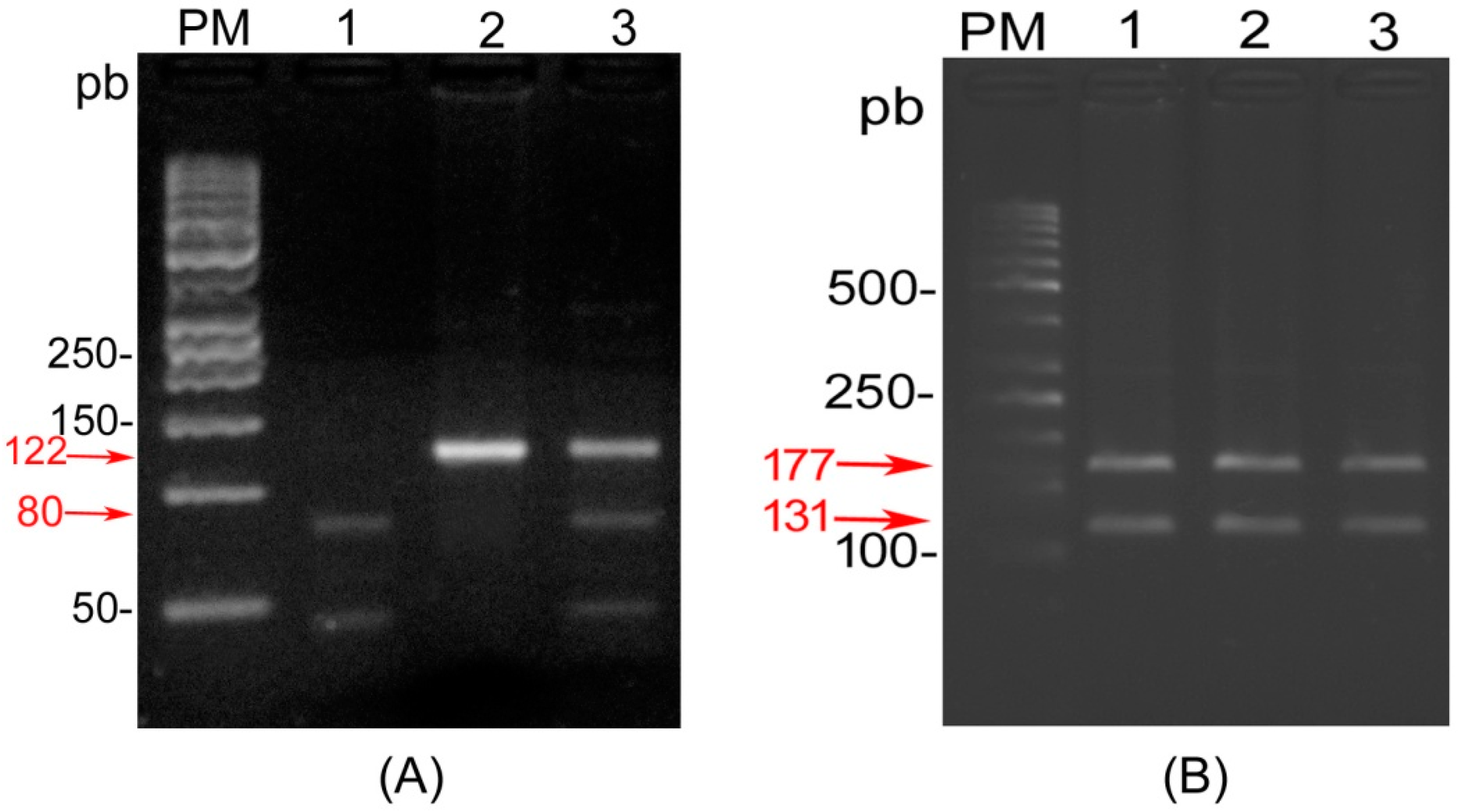
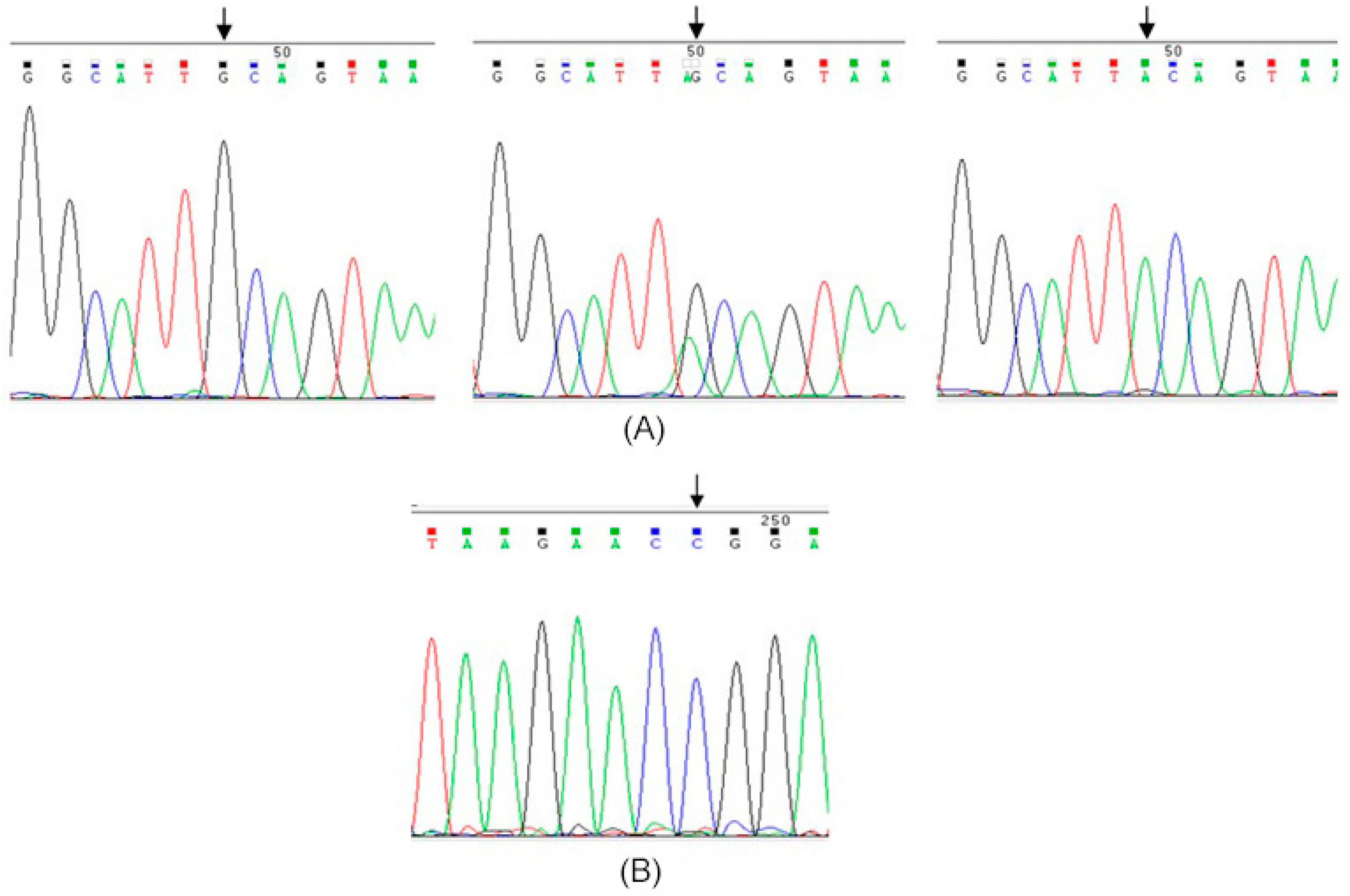
2.1. Genotyping
2.2. Genotypic/Allelic Frequency and Association Analysis
Stratification Analysis
3. Discussion
4. Materials and Methods
4.1. Study Subjects
4.2. PCR-RFLP Analysis
4.3. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Dirección de Estadística del Ministerio de Salud e Información en Salud. Estadísticas vitales, 2013, Serie 5, N-57. Available online: http://www.deis.gov.ar (accessed on 16 September 2015).
- Hynes, R.O. Integrins: Bidirectional, allosteric signaling machines. Cell 2002, 110, 673–687. [Google Scholar] [CrossRef]
- Barczyk, M.; Carracedo, S.; Gullberg, D. Integrins. Cell Tissue Res. 2010, 339, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Sinha, M.; Luxon, B.; Bresnick, A.; O´Connor, K. Integrin α6β4 controls the expression of genes associated with cell motility, invasion and metastasis, including S100A4/Mestastasin. J. Biol. Chem. 2009, 284, 1484–1494. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Giancotti, F.G. Integrin signalling during tumour progression. Nat. Rev. Mol. Cell Biol. 2004, 5, 816–826. [Google Scholar] [CrossRef] [PubMed]
- Lipscomb, E.A.; Mercurio, A.M. Mobilization and activation of a signaling competent α6β4integrin underlies its contribution to carcinoma progression. Cancer Metastasis Rev. 2005, 24, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Nikolopoulos, S.N.; Blaikie, P.; Yoshioka, T.; Guo, W.; Giancotti, F.G. Integrin β4 signaling promotes tumor angiogenesis. Cancer Cell 2004, 6, 471–483. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.M.; Rabinovitz, I.; Wang, H.H.; Toker, A.; Mercurio, A.M. Activation of phosphoinositide 3-OH kinase by the α6β4 integrin promotes carcinoma invasion. Cell 1997, 91, 949–960. [Google Scholar] [CrossRef]
- Rabinovitz, I.; Gipson, I.K.; Mercurio, A.M. Traction forces mediated by α6β4 integrin: Implications for basement membrane organization and tumor invasion. Mol. Biol. Cell 2001, 12, 4030–4043. [Google Scholar] [CrossRef] [PubMed]
- Mainiero, F.; Pepe, A.; Yeon, M.; Ren, Y.; Giancotti, F.G. The intracellular functions of α6β4 integrin are regulated by EGF. J. Cell Biol. 1996, 134, 241–253. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, K.L.; Shaw, L.M.; Mercurio, A.M. Release of cAMP gating by the α6β4 integrin stimulates lamellae formation and the chemotactic migration of invasive carcinoma cells. J. Cell Biol. 1998, 143, 1749–1760. [Google Scholar] [CrossRef] [PubMed]
- Guo, W.; Pylayeva, Y.; Pepe, A.; Yoshioka, T.; Muller, W.J.; Inghirami, G.; Giancotti, F.G. β4 integrin amplifies ErbB2 signaling to promote mammary tumorigenesis. Cell 2006, 126, 489–502. [Google Scholar] [CrossRef] [PubMed]
- Bon, G.; Folgiero, V.; di Carlo, S.; Sacchi, A.; Falcione, R. Involvement of α6β4 integrin in the mechanisms that regulate breast cancer progression. Breast Cancer Res. 2007, 9. [Google Scholar] [CrossRef] [PubMed]
- Diaz, L.; Cristofanilli, M.; Zhou, X.; Welch, K.; Smith, T.; Yang, Y.; Sneige, N.; Sahin, A.; Gilcrease, M. β4 integrin subunit gene expression correlates with tumor size and nuclear grade in early breast cancer. Mod. Pathol. 2005, 18, 1165–1175. [Google Scholar] [CrossRef] [PubMed]
- Pereda, J.; Ortega, E.; Alonso-García, N.; Gómez-Hernández, M.; Sonnenberg, A. Advances and perspectives of the architecture of hemisdesmosomes. Cell Adhes. Migr. 2009, 3, 361–364. [Google Scholar] [CrossRef] [PubMed]
- Gabarra, V.; Cho, S.; Ramirez, M.; Ren, Y.; Chen, L.L.; Cheung, A.; Cao, X.; Rennard, R.; Unruh, K.R.; Graff, C.P.; et al. Antibodies directed to α6β4 highlight the adhesive and signaling functions of the integrin in breast cancer cell lines. Cancer Biol. Ther. 2010, 9, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Stewart, R.L.; O’Connor, K.L. Clinical significance of the integrin α6β4 in human malignancies. Lab. Investig. 2015, 95, 976–986. [Google Scholar] [CrossRef] [PubMed]
- Jorde, L.; Bamshad, M.; Carey, J.; White, R. Medical Genetics, 2nd ed.; Elsevier Science Health Science Division: Philadelphia, PA, USA, 2000. [Google Scholar]
- Pulkkinen, L.M.; Rouan, F.; Bruckener-Tuderman, L.; Wallerstein, R.; Garzón, M.; Brown, T.; Smith, L.; Carter, W.; Uitto, J. Novel ITGβ4 mutations in lethal and nonlethal variants of epidermolysis bullosa with pyloric atresia: Missense versus nonsense. Am. J. Hum. Genet. 1998, 63, 1376–1387. [Google Scholar] [CrossRef] [PubMed]
- Mercurio, A.; Rabinovitz, I.; Shaw, L. The α6β4 integrin and epithelial cell migration. Cell Biol. 2001, 13, 541–545. [Google Scholar] [CrossRef]
- Koster, J.; Kuikman, I.; Kreft, M.; Sonnenberg, A. Two different mutations in the cytoplasmatic domain of the integrin β4 subunit in nonlethal forms of epidermolysis bullosa prevent interaction of β4 with plectin. J. Investig. Dermatol. 2001, 117, 1405–2001. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, D.; Oh, I.; Song, J.; Kwon, K.; Choe, B.; Kim, Y. A missense polymorphism (rs11895564, Ala380Thr) of integrin α6 is associated with the development and progression of papillary thyroid carcinoma in Korean population. J. Korean Surg. Soc. 2011, 81, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Jo, D.J. Polymorphisms of integrin, α6 contribute to the development and neurologic symptoms of intracerebral hemorrhage in Korean population. J. Korean Neurosurg. Soc. 2011, 50, 293–298. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.A.; Dykes, D.D.; Polesky, H.F. A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res. 1988, 16, 1215. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Coulouris, G.; Zaretskaya, I.; Cutcutache, I.; Rozen, S.; Madden, T.L. Primer-BLAST: A tool to design target-specific primers for polymerase chain reaction. BMC Bioinform. 2012, 13. [Google Scholar] [CrossRef] [PubMed]
| Models 1 | Genotype 2 | Cases | Controls | Odds Ratio (OR) | Confidence Interval (CI) 95% | p 4 | ||
|---|---|---|---|---|---|---|---|---|
| n = 85 | (%) 3 | n = 113 | (%) 3 | |||||
| Co | GG | 37 | 44 | 47 | 42 | 1 | - | - |
| GA | 41 | 48 | 55 | 49 | 0.94 | 0.52–1.71 | 0.881 | |
| GG | 7 | 8 | 11 | 9 | 0.81 | 0.28–2.29 | 0.795 | |
| Do | GG | 37 | 44 | 47 | 42 | 1 | - | - |
| GA + AA | 48 | 56 | 66 | 58 | 0.92 | 0.52–1.63 | 0.884 | |
| Re | GG + GA | 78 | 92 | 102 | 91 | 1 | - | - |
| AA | 7 | 8 | 11 | 9 | 0.83 | 0.31–2.24 | 0.806 | |
| Ad | - | - | - | - | - | 0.92 | 0.61–1.41 | 0.747 |
| ITGα6 Codon 380 | Cases n = 85 (%) | Controls n = 113 (%) | OR | CI 95% | p 1 |
|---|---|---|---|---|---|
| Allelic Frequencies | |||||
| Ala | 115 (68) | 149 (66) | 0.92 2 | 0.61–1.41 2 | 0.747 2 |
| Thr | 55 (32) | 77 (34) |
| Parameters | GA + AA Cases/Controls 1 | GG Cases/Controls 2 | OR Adjusted (95% CI) | p 3 |
|---|---|---|---|---|
| Age | ||||
| <55 (mean) | 21 | 17 | 1.13 (0.55–2.31) | 0.857 |
| ≥55 (mean) | 23 | 18 | 0.91 (0.44–1.87) | 0.854 |
| Tumor site | ||||
| Ductal | 21 | 13 | 1.15 (0.52–2.52) | 0.843 |
| Lobular | 1 | - | - | 1.000 |
| Lymphovascular invasion | ||||
| Positive | 24 | 16 | 1.07 (0.51–2.22) | 1.000 |
| Negative | 13 | 12 | 0.77 (0.32–1.84) | 0.656 |
| RP | ||||
| Positive | 21 | 13 | 1.15 (0.52–2.52) | 0.843 |
| Negative | 6 | 5 | 0.85 (0.25–2.96) | 1.000 |
| RE | ||||
| Positive | 22 | 13 | 1.21 (0.55–2.63) | 0.697 |
| Negative | 9 | 3 | 2.13 (0.55–8.32) | 0.359 |
| HER2 | ||||
| Positive | 10 | 9 | 0.79 (0.29–2.09) | 0.803 |
| Negative | 20 | 8 | 1.78 (0.72–4.38) | 0.280 |
| Histological grade | ||||
| Grade I | 4 | 4 | 0.71 (0.17–2.99) | 0.720 |
| Grade II | 26 | 18 | 1.02 (0.51–2.08) | 1.000 |
| Grade III | 13 | 10 | 0.92 (0.37–2.28) | 1.000 |
| Menopausal period | ||||
| Premenopausal | 12 | 14 | 0.61 (0.26–1.44) | 0.280 |
| Postmenopausal | 24 | 17 | 1.01 (0.48–2.07) | 1.000 |
| Parameters | Total Number of Cases (n = 85) |
|---|---|
| Pathological diagnosis | |
| Ductal carcinoma | 36 |
| Lobular carcinoma | 1 |
| N/D 1 | 48 |
| Lymphovascular invasion | |
| Positive | 43 |
| Negative | 30 |
| N/D | 14 |
| Histological grade | |
| Grade I | 8 |
| Grade II | 45 |
| Grade III | 26 |
| N/D | 8 |
| Menopausal period | |
| Premenopausal | 29 |
| Postmenopausal | 45 |
| N/D | 13 |
| Estrogen receptor (ER) | |
| Positive | 32 |
| Negative | 17 |
| N/D | 38 |
| Progesterone Receptor (PR) | |
| Positive | 37 |
| Negative | 12 |
| N/D | 38 |
| HER2/neu | |
| Positive | 21 |
| Negative | 28 |
| N/D | 38 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Acosta, K.B.; Lorenzini Campos, M.N.; Etcheverry, S.B.; Zapata, P.D. α6β4 Integrin Genetic Variations (A380T and R1281W) and Breast Cancer Risk in an Argentinian Population. Int. J. Mol. Sci. 2016, 17, 1540. https://doi.org/10.3390/ijms17101540
Acosta KB, Lorenzini Campos MN, Etcheverry SB, Zapata PD. α6β4 Integrin Genetic Variations (A380T and R1281W) and Breast Cancer Risk in an Argentinian Population. International Journal of Molecular Sciences. 2016; 17(10):1540. https://doi.org/10.3390/ijms17101540
Chicago/Turabian StyleAcosta, Karina Beatriz, Melina Noelia Lorenzini Campos, Susana Beatriz Etcheverry, and Pedro Dario Zapata. 2016. "α6β4 Integrin Genetic Variations (A380T and R1281W) and Breast Cancer Risk in an Argentinian Population" International Journal of Molecular Sciences 17, no. 10: 1540. https://doi.org/10.3390/ijms17101540
APA StyleAcosta, K. B., Lorenzini Campos, M. N., Etcheverry, S. B., & Zapata, P. D. (2016). α6β4 Integrin Genetic Variations (A380T and R1281W) and Breast Cancer Risk in an Argentinian Population. International Journal of Molecular Sciences, 17(10), 1540. https://doi.org/10.3390/ijms17101540





