Poly-ε-caprolactone Coated and Functionalized Porous Titanium and Magnesium Implants for Enhancing Angiogenesis in Critically Sized Bone Defects
Abstract
:1. Introduction
2. Results and Discussion
2.1. Vitality and Proliferation of Murine Osteoblasts on Polymers
2.1.1. Vitality of Murine Osteoblasts on Polymers


2.1.2. Proliferation of Murine Osteoblasts on Polymers
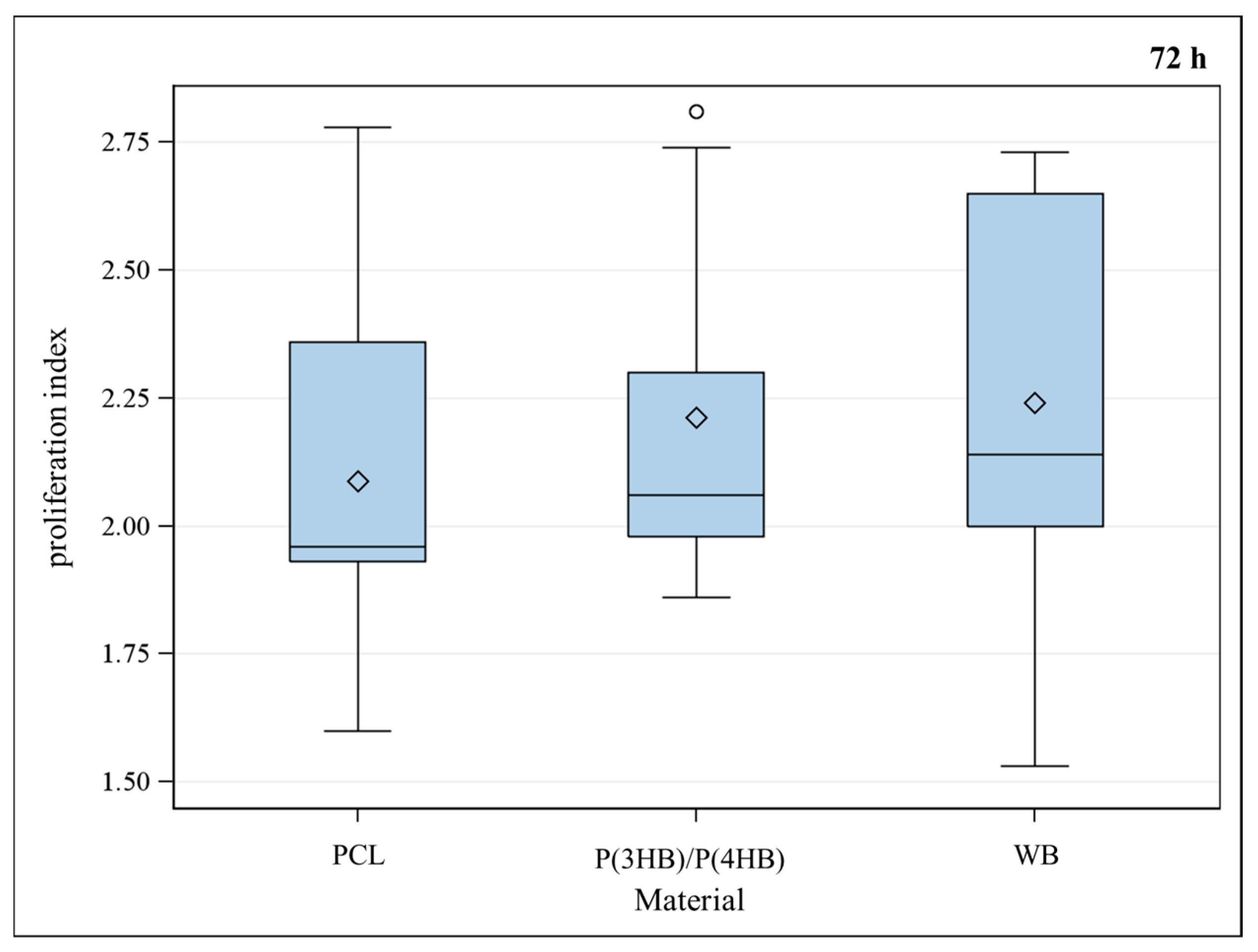
2.2. Vitality and Proliferation of Murine Osteoblasts on Titanium
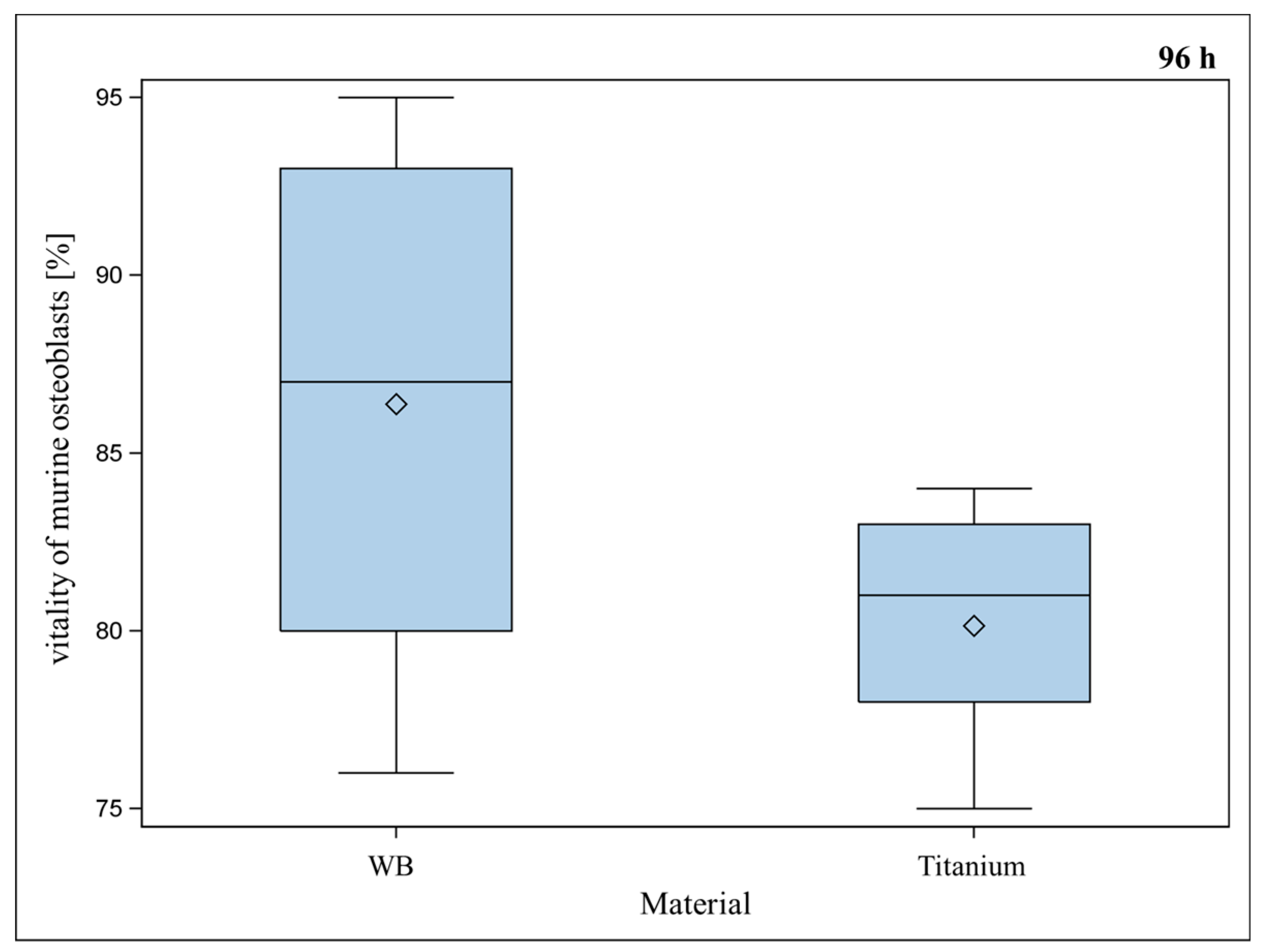
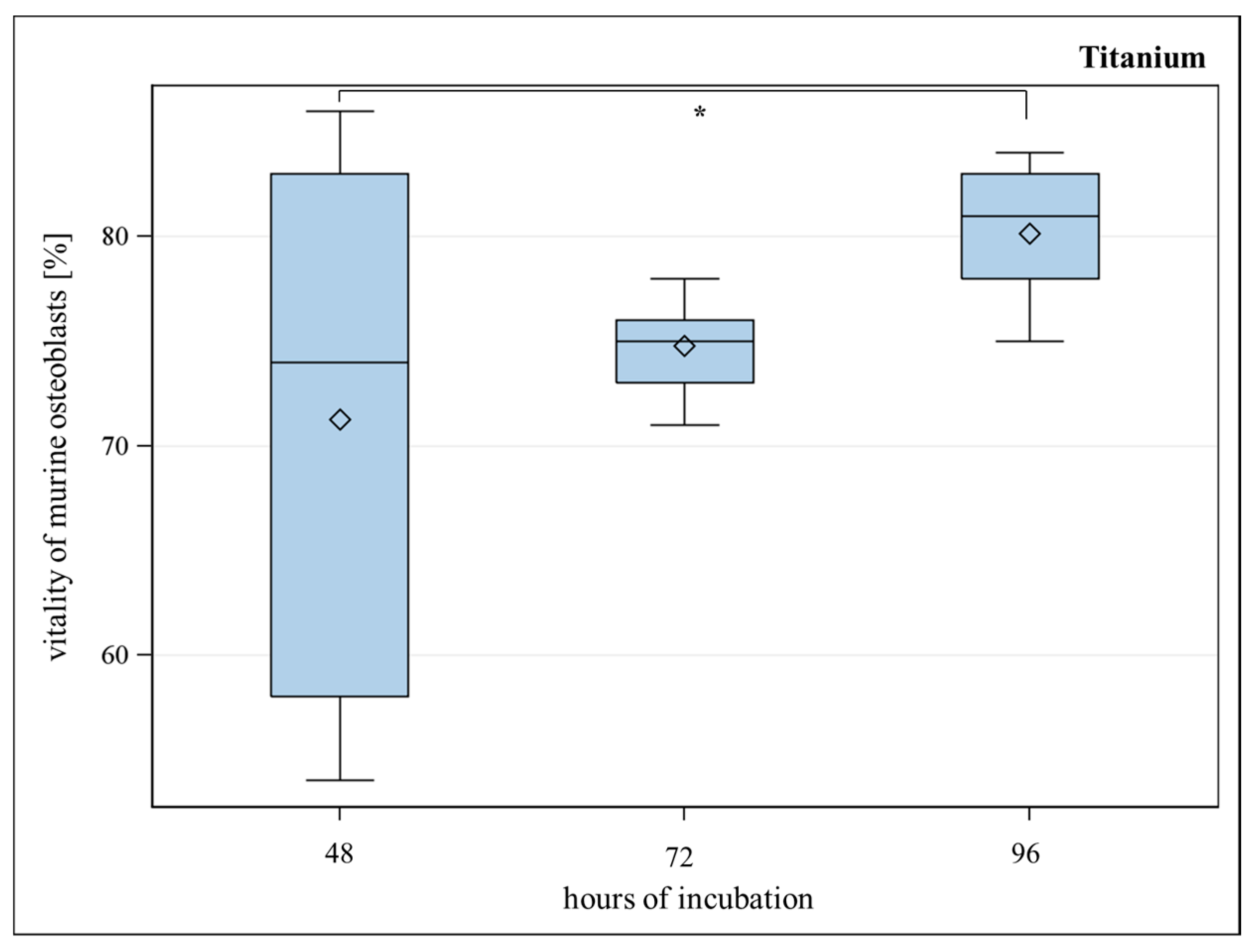
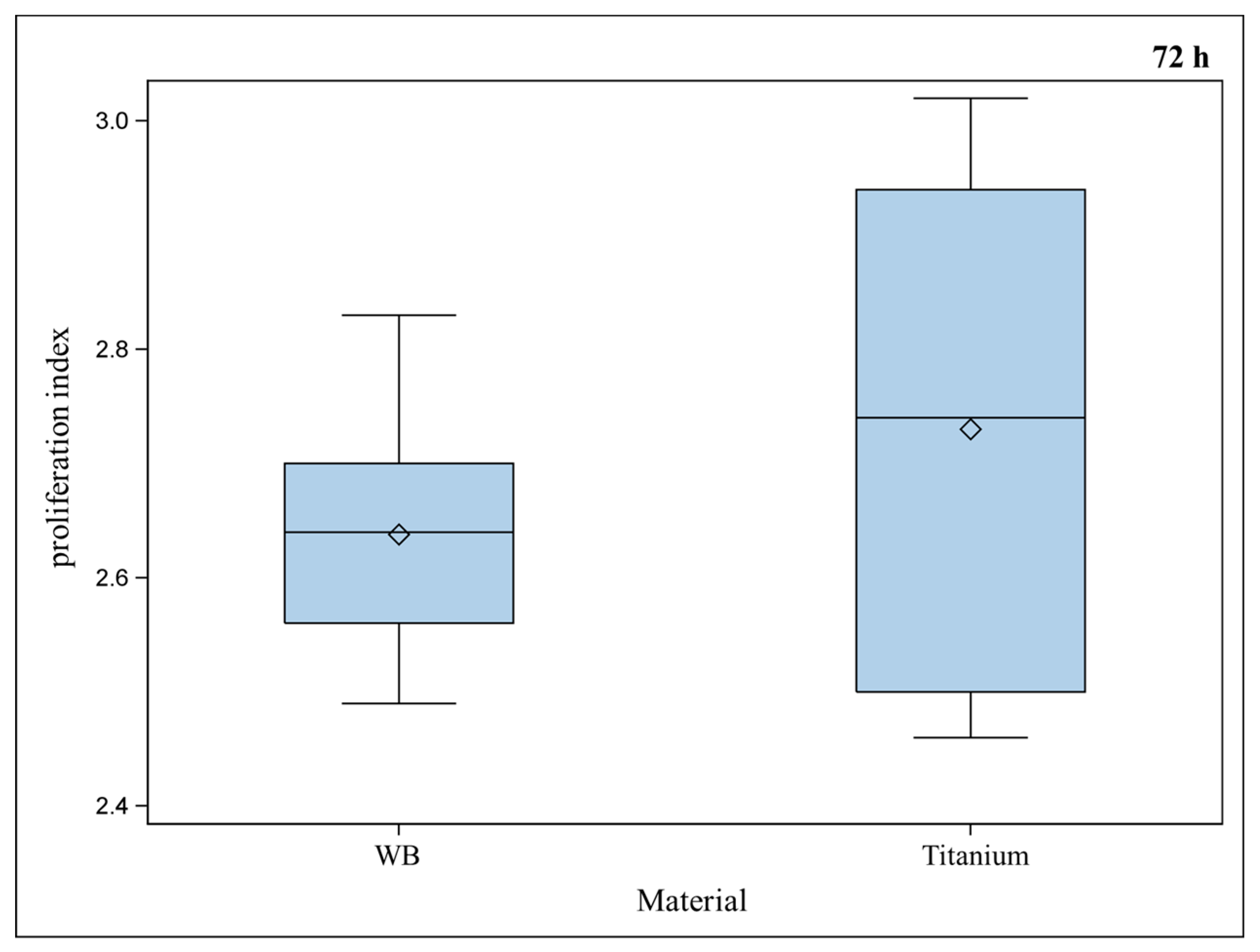
2.3. Live Cell Imaging (LCI) of Green Fluorescent Protein (GFP)-Osteoblasts on Titanium- and Titanium-poly-ε-caprolactone- Implants
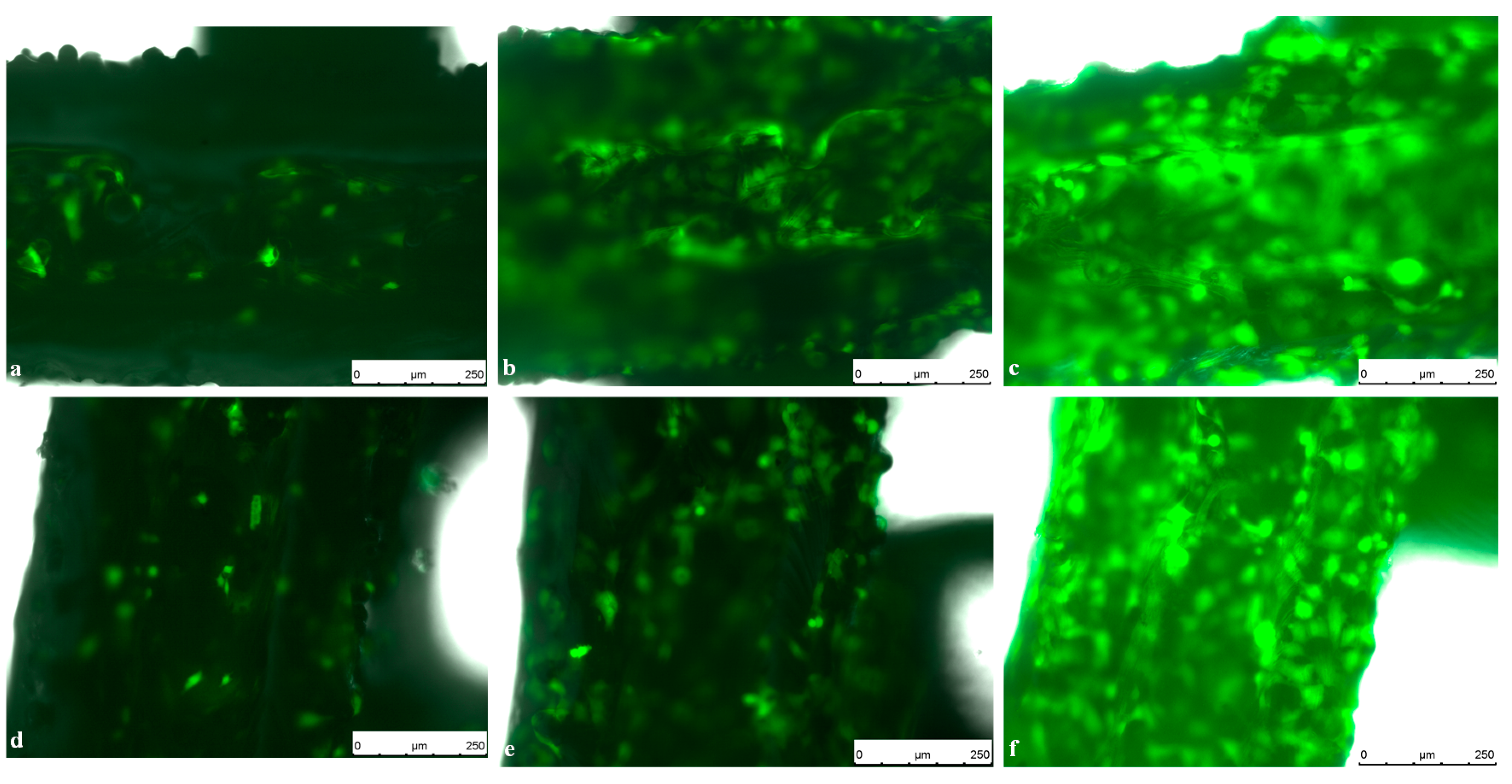
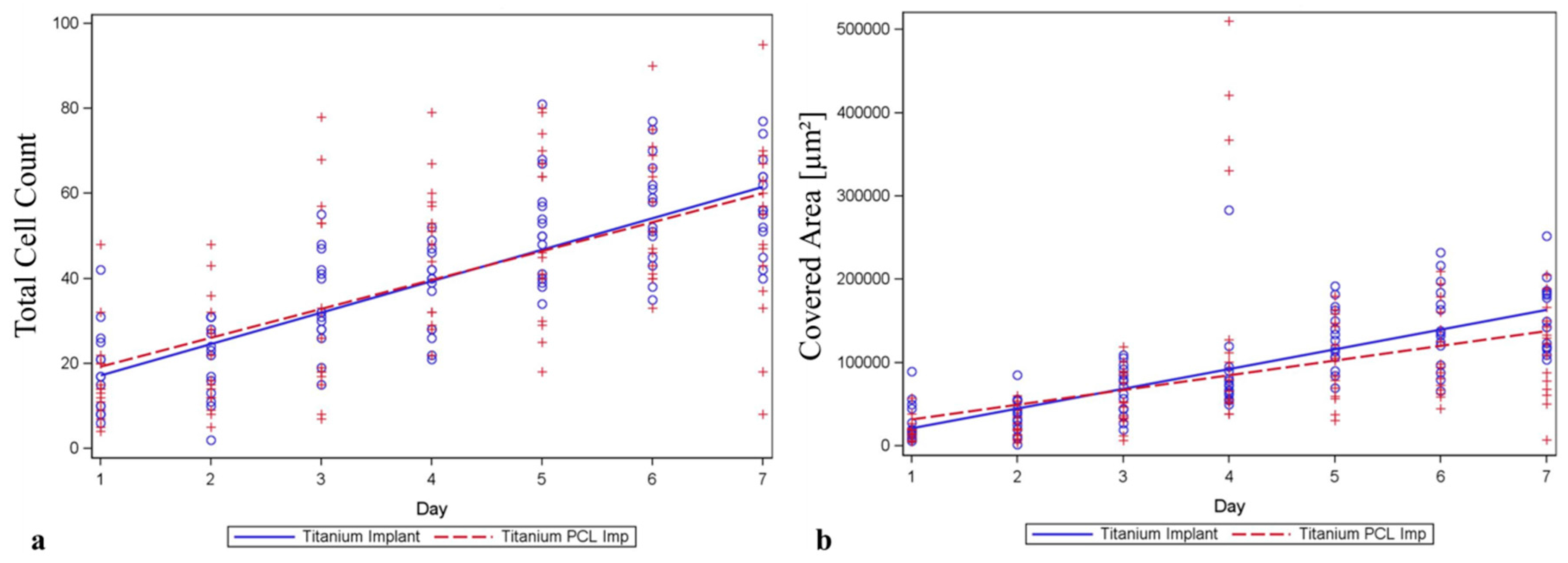
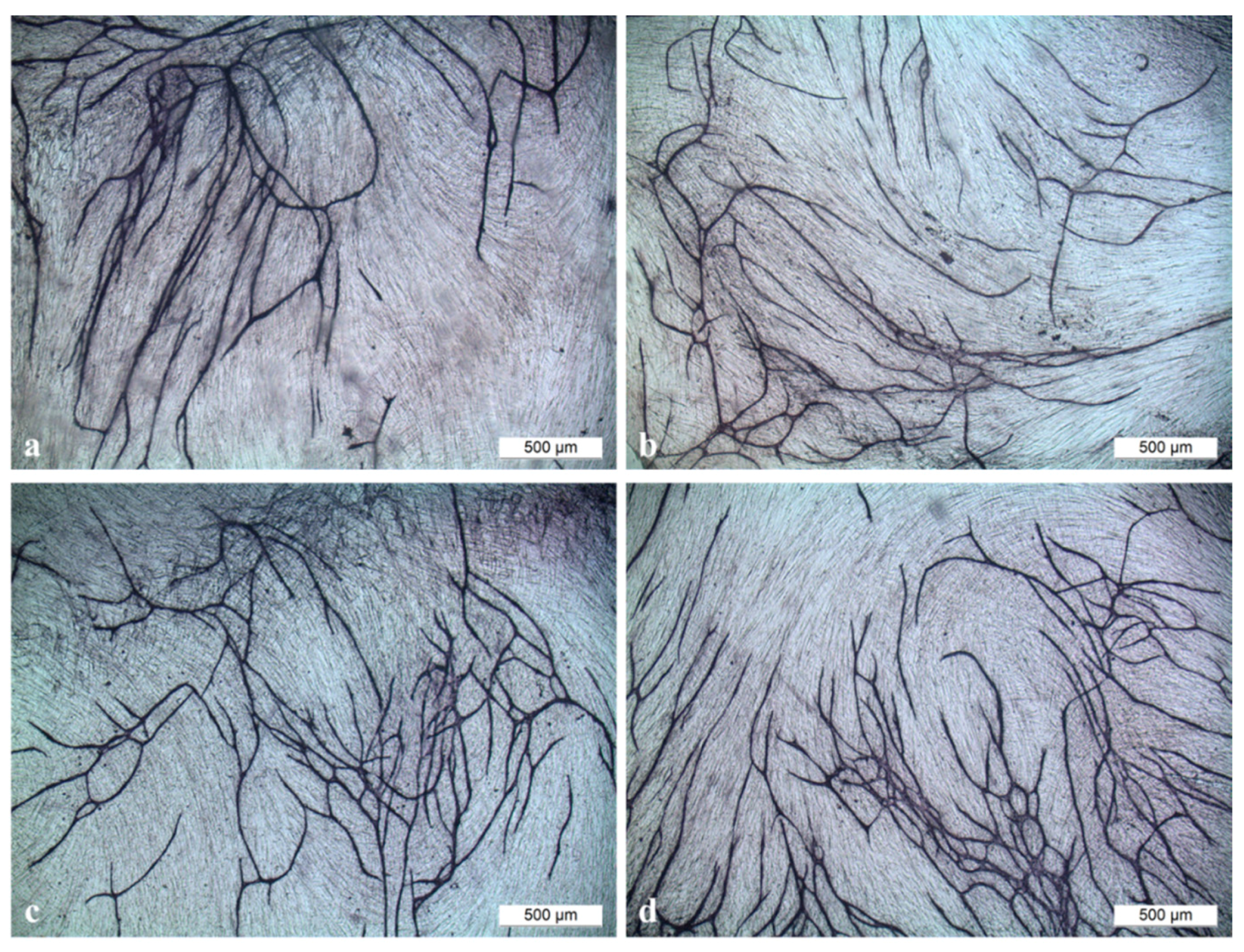
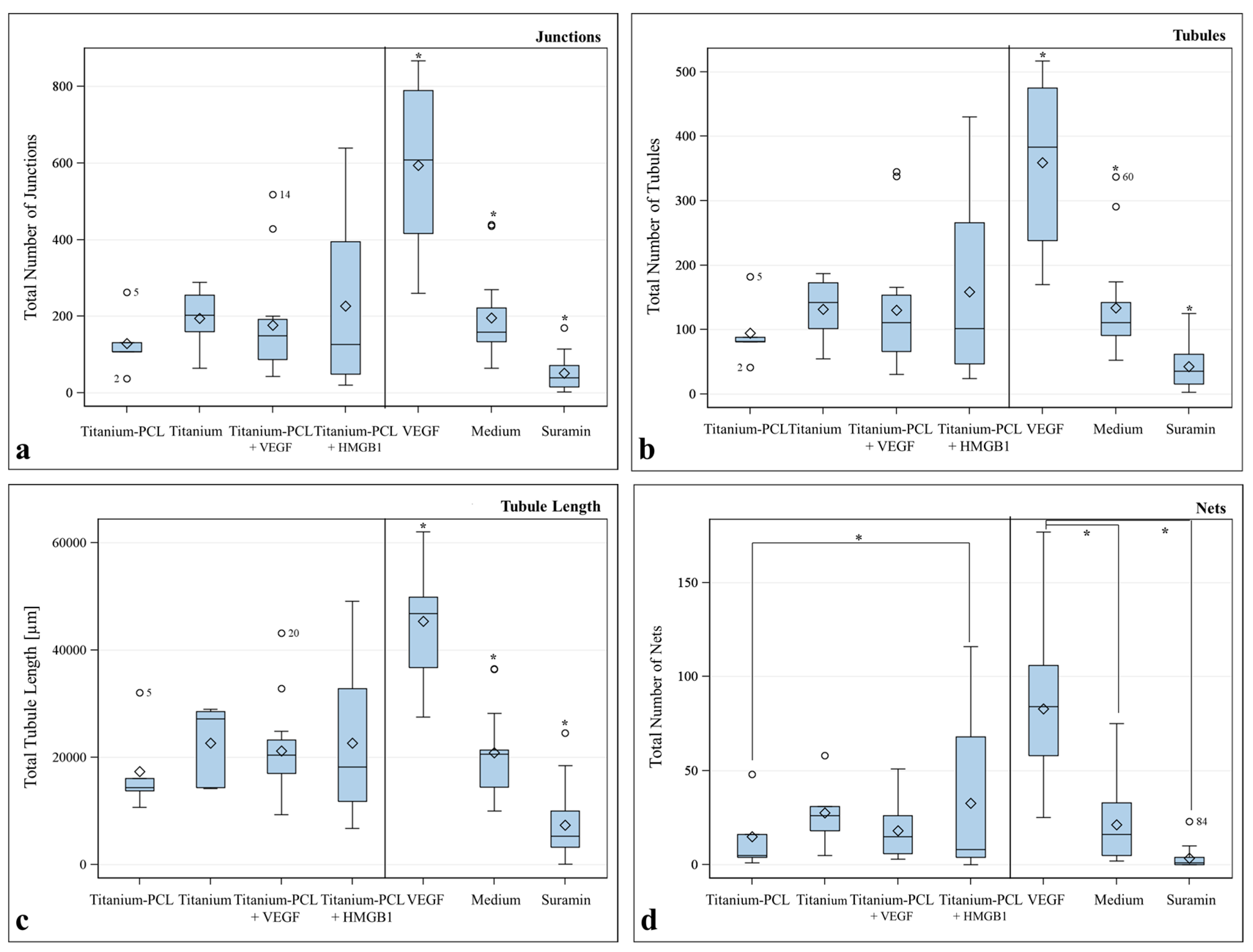
2.4. Angiogenesis Assay with Functionalized Titanium PCL and Magnesium PCL Implants
3. Experimental Section
3.1. Preparation of the PCL and Poly(3-hydroxy-butyrate)/Poly(4-hydroxy-butyrate (P(3HB)/P(4HB))
3.2. Selective Laser Melting of Titanium and Magnesium Implants
3.3. PCL Coating of Titanium and Magnesium Implants

3.4. Incorporation of Vascular Endothelial Growth Factor (VEGF) and High Mobility Group Box 1 (HMGB1) into Titanium- and Magnesium-Scaffolds
3.5. Murine Osteoblast and Murine GFP-Osteoblast Isolation [20]
3.6. Staining Of Murine Osteoblasts for Cell Division Tracking
3.7. Proliferation and Vitality of Murine Osteoblasts on Polymers (PCL and P(3HB)/P(4HB))
3.8. Proliferation and Vitality of Murine Osteoblasts on Titanium Scaffolds
3.9. Live Cell Imaging (LCI) of GFP-Osteoblasts On Titanium- and Titanium PCL Implants [28]
3.10. Angiogenesis Assay with Functionalized Titanium PCL and Magnesium PCL Implants
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Gellrich, N.C.; Held, U.; Schoen, R.; Pailing, T.; Schramm, A.; Bormann, K.H. Alveolar zygomatic buttress: A new donor site for limited preimplant augmentation procedures. J. Oral Maxillofac. Surg. 2007, 65, 275–280. [Google Scholar] [CrossRef] [PubMed]
- Carano, R.A.; Filvaroff, E.H. Angiogenesis and bone repair. Drug Discov. Today 2003, 8, 980–989. [Google Scholar] [CrossRef]
- Geiger, M.; Li, R.H.; Friess, W. Collagen sponges for bone regeneration with rhBMP-2. Adv. Drug Deliv. Rev. 2003, 55, 1613–1629. [Google Scholar] [CrossRef] [PubMed]
- Goulet, J.A.; Senunas, L.E.; DeSilva, G.L.; Greenfield, M.L. Autogenous iliac crest bone graft. Complications and functional assessment. Clin. Orthop. Relat. Res. 1997, 339, 76–81. [Google Scholar] [CrossRef] [PubMed]
- Silber, J.S.; Anderson, D.G.; Daffner, S.D.; Brislin, B.T.; Leland, J.M.; Hilibrand, A.S.; Vaccaro, A.R.; Albert, T.J. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine 2003, 28, 134–139. [Google Scholar] [CrossRef] [PubMed]
- Li, J.P.; Li, S.H.; van Blitterswijk, C.A.; de Groot, K. Cancellous bone from porous Ti6Al4V by multiple coating technique. J. Mater. Sci. Mater. Med. 2006, 17, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Li, C.; Zhu, Z.; Liu, C.S. Measurement of the dynamic young’s modulus of porous titanium and ti6al4v. J. Mater. Sci. 2007, 42, 7348–7353. [Google Scholar] [CrossRef]
- Rubshtein, A.P.; Makarova, E.B.; Rinkevich, A.B.; Medvedeva, D.S.; Yakovenkova, L.I.; Vladimirov, A.B. Elastic properties of a porous titanium-bone tissue composite. Mater. Sci. Eng. C Mater. Biol. Appl. 2015, 52, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Niinomi, M. Mechanical biocompatibilities of titanium alloys for biomedical applications. J. Mech. Behav. Biomed. Mater. 2008, 1, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Van der Stok, J.; van der Jagt, O.P.; Amin Yavari, S.; de Haas, M.F.; Waarsing, J.H.; Jahr, H.; van Lieshout, E.M.; Patka, P.; Verhaar, J.A.; Zadpoor, A.A.; et al. Selective laser melting-produced porous titanium scaffolds regenerate bone in critical size cortical bone defects. J. Orthop. Res. 2013, 31, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Loh, Q.L.; Choong, C. Three-dimensional scaffolds for tissue engineering applications: Role of porosity and pore size. Tissue Eng. B Rev. 2013, 19, 485–502. [Google Scholar] [CrossRef] [PubMed]
- Thijs, L.; Verhaeghe, F.; Craeghs, T.; Humbeeck, J.V.; Kruth, J.-P. A study of the microstructural evolution during selective laser melting of Ti–6Al–4V. Acta Mater. 2010, 58, 3303–3312. [Google Scholar] [CrossRef]
- Rotaru, H.; Schumacher, R.; Kim, S.G.; Dinu, C. Selective laser melted titanium implants: A new technique for the reconstruction of extensive zygomatic complex defects. Maxillofac. Plast. Reconstr. Surg. 2015, 37. [Google Scholar] [CrossRef]
- Gieseke, M.; Noelke, C.; Kaierle, S.; Maier, H.J.; Haferkamp, H. Selective laser melting of magnesium alloys for manufacturing individual implants. In Proceedings of the Fraunhofer Direct Digital Manufacturing Conference 2014, Berlin, Germany, 12–13 March 2014.
- Gieseke, M.; Noelke, C.; Kaierle, S.; Wesling, V.; Haferkamp, H. Selective laser melting of magnesium and magnesium alloys. In Magnesium Technology 2013; Hort, N., Alderman, M., Mathaudhu, S., Neale, N., Eds.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; pp. 65–68. [Google Scholar]
- Charyeva, O.; Thormann, U.; Lips, K.S.; Heimann, L.; Sommer, U.; Szalay, G.; Alt, V.; Hort, N.; Schnettler, R.; Rauschmann, M.; et al. Histological comparison of new biodegradable magnesium-based implants for maxillofacial applications. J. Maxillofac. Oral Surg. 2015, 14, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Janning, C.; Willbold, E.; Vogt, C.; Nellesen, J.; Meyer-Lindenberg, A.; Windhagen, H.; Thorey, F.; Witte, F. Magnesium hydroxide temporarily enhancing osteoblast activity and decreasing the osteoclast number in peri-implant bone remodelling. Acta Biomater. 2010, 6, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Waizy, H.; Seitz, J.-M.; Reifenrath, J.; Weizbauer, A.; Bach, F.-W.; Meyer-Lindenberg, A.; Denkena, B.; Windhagen, H. Biodegradable magnesium implants for orthopedic applications. J. Mater. Sci. 2013, 48, 39–50. [Google Scholar] [CrossRef]
- Shadanbaz, S.; Walker, J.; Woodfield, T.B.; Staiger, M.P.; Dias, G.J. Monetite and brushite coated magnesium: In vivo and in vitro models for degradation analysis. J. Mater. Sci. Mater. Med. 2014, 25, 173–183. [Google Scholar] [CrossRef] [PubMed]
- Matena, J.; Petersen, S.; Gieseke, M.; Kampmann, A.; Teske, M.; Beyerbach, M.; Murua Escobar, H.; Haferkamp, H.; Gellrich, N.C.; Nolte, I. SLM produced porous titanium implant improvements for enhanced vascularization and osteoblast seeding. Int. J. Mol. Sci. 2015, 16, 7478–7492. [Google Scholar] [CrossRef] [PubMed]
- Ferrara, N. Vascular endothelial growth factor: Basic science and clinical progress. Endocr. Rev. 2004, 25, 581–611. [Google Scholar] [CrossRef] [PubMed]
- Wake, H.; Mori, S.; Liu, K.; Takahashi, H.K.; Nishibori, M. High mobility group box 1 complexed with heparin induced angiogenesis in a matrigel plug assay. Acta Med. Okayama 2009, 63, 249–262. [Google Scholar] [PubMed]
- Murphy, W.L.; Simmons, C.A.; Kaigler, D.; Mooney, D.J. Bone regeneration via a mineral substrate and induced angiogenesis. J. Dent. Res. 2004, 83, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Wu, Q.; Wang, Y.; Chen, G.Q. Medical application of microbial biopolyesters polyhydroxyalkanoates. Artif. Cells Blood Substit. Immobil. Biotechnol. 2009, 37, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Woodruff, M.A.; Hutmacher, D.W. The return of a forgotten polymer—polycaprolactone in the 21st century. Prog. Polym. Sci. 2010, 35, 1217–1256. [Google Scholar] [CrossRef] [Green Version]
- Hazer, B. Amphiphilic poly(3-hydroxy alkanoate)s: Potential candidates for medical applications. Int. J. Polym. Sci. 2010, 2010. [Google Scholar] [CrossRef]
- Williams Simon, F.; Rizk, S.; Martin David, P. Poly-4-hydroxybutyrate (P4HB): A new generation of resorbable medical devices for tissue repair and regeneration. Biomed. Tech. 2013, 58, 439–452. [Google Scholar] [CrossRef] [PubMed]
- Matena, J.; Petersen, S.; Gieseke, M.; Teske, M.; Beyerbach, M.; Kampmann, A.; Murua Escobar, H.; Gellrich, N.C.; Haferkamp, H.; Nolte, I. Comparison of selective laser melted titanium and magnesium implants coated with PCL. Int. J. Mol. Sci. 2015, 16, 13287–13301. [Google Scholar] [CrossRef] [PubMed]
- Bennett, J.H.; Moffatt, S.; Horton, M. Cell adhesion molecules in human osteoblasts: Structure and function. Histol. Histopathol. 2001, 16, 603–611. [Google Scholar] [PubMed]
- Williams, J.M.; Adewunmi, A.; Schek, R.M.; Flanagan, C.L.; Krebsbach, P.H.; Feinberg, S.E.; Hollister, S.J.; Das, S. Bone tissue engineering using polycaprolactone scaffolds fabricated via selective laser sintering. Biomaterials 2005, 26, 4817–4827. [Google Scholar] [CrossRef] [PubMed]
- Kanczler, J.M.; Oreffo, R.O. Osteogenesis and angiogenesis: The potential for engineering bone. Eur. Cells Mater. 2008, 15, 100–114. [Google Scholar]
- Sharmin, F.; Adams, D.; Pensak, M.; Dukas, A.; Lieberman, J.; Khan, Y. Biofunctionalizing devitalized bone allografts through polymer-mediated short and long term growth factor delivery. J. Biomed. Mater. Res. A 2015, 103, 2847–2854. [Google Scholar] [CrossRef] [PubMed]
- Hou, J.; Wang, J.; Cao, L.; Qian, X.; Xing, W.; Lu, J.; Liu, C. Segmental bone regeneration using rhBMP-2-loaded collagen/chitosan microspheres composite scaffold in a rabbit model. Biomed. Mater. 2012, 7. [Google Scholar] [CrossRef] [PubMed]
- Wulf, K.; Teske, M.; Lobler, M.; Luderer, F.; Schmitz, K.P.; Sternberg, K. Surface functionalization of poly(ε-caprolactone) improves its biocompatibility as scaffold material for bioartificial vessel prostheses. J. Biomed. Mater. Res. B Appl. Biomater. 2011, 98, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Laschke, M.W.; Vollmar, B.; Menger, M.D. The dorsal skinfold chamber: Window into the dynamic interaction of biomaterials with their surrounding host tissue. Eur. Cells Mater. 2011, 22, 147–164. [Google Scholar]
- Gebhardt, A. Understanding additive manufacturing. In Understanding Additive Manufacturing; Carl Hanser Verlag GmbH & Co. KG: Munich, Germany, 2011; pp. 40–44. [Google Scholar]
- Chen, X.D.; Qian, H.Y.; Neff, L.; Satomura, K.; Horowitz, M.C. Thy-1 antigen expression by cells in the osteoblast lineage. J. Bone Miner. Res. 1999, 14, 362–375. [Google Scholar] [CrossRef] [PubMed]
- Matena, J.; Gieseke, M.; Kampmann, A.; Petersen, S.; Murua Escobar, H.; Sternberg, K.; Haferkamp, H.; Gellrich, N.C.; Nolte, I. Characterisation of cell growth on titanium scaffolds made by selective laser melting for tissue engineering. Biomed. Tech. 2013. [Google Scholar] [CrossRef] [PubMed]
- V2a Kit Protocol. Available online: http://www.cellworks.co.uk/v2akit.php (accessed on 1 August 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roland, L.; Grau, M.; Matena, J.; Teske, M.; Gieseke, M.; Kampmann, A.; Beyerbach, M.; Murua Escobar, H.; Haferkamp, H.; Gellrich, N.-C.; et al. Poly-ε-caprolactone Coated and Functionalized Porous Titanium and Magnesium Implants for Enhancing Angiogenesis in Critically Sized Bone Defects. Int. J. Mol. Sci. 2016, 17, 1. https://doi.org/10.3390/ijms17010001
Roland L, Grau M, Matena J, Teske M, Gieseke M, Kampmann A, Beyerbach M, Murua Escobar H, Haferkamp H, Gellrich N-C, et al. Poly-ε-caprolactone Coated and Functionalized Porous Titanium and Magnesium Implants for Enhancing Angiogenesis in Critically Sized Bone Defects. International Journal of Molecular Sciences. 2016; 17(1):1. https://doi.org/10.3390/ijms17010001
Chicago/Turabian StyleRoland, Laura, Michael Grau, Julia Matena, Michael Teske, Matthias Gieseke, Andreas Kampmann, Martin Beyerbach, Hugo Murua Escobar, Heinz Haferkamp, Nils-Claudius Gellrich, and et al. 2016. "Poly-ε-caprolactone Coated and Functionalized Porous Titanium and Magnesium Implants for Enhancing Angiogenesis in Critically Sized Bone Defects" International Journal of Molecular Sciences 17, no. 1: 1. https://doi.org/10.3390/ijms17010001
APA StyleRoland, L., Grau, M., Matena, J., Teske, M., Gieseke, M., Kampmann, A., Beyerbach, M., Murua Escobar, H., Haferkamp, H., Gellrich, N.-C., & Nolte, I. (2016). Poly-ε-caprolactone Coated and Functionalized Porous Titanium and Magnesium Implants for Enhancing Angiogenesis in Critically Sized Bone Defects. International Journal of Molecular Sciences, 17(1), 1. https://doi.org/10.3390/ijms17010001




