Effect of Dose and Administration Period of Seed Cake of Genetically Modified and Non-Modified Flax on Selected Antioxidative Activities in Rats
Abstract
:1. Introduction
2. Results and Discussion
2.1. Digestibility of Nutrients and Rat Growth Parameters
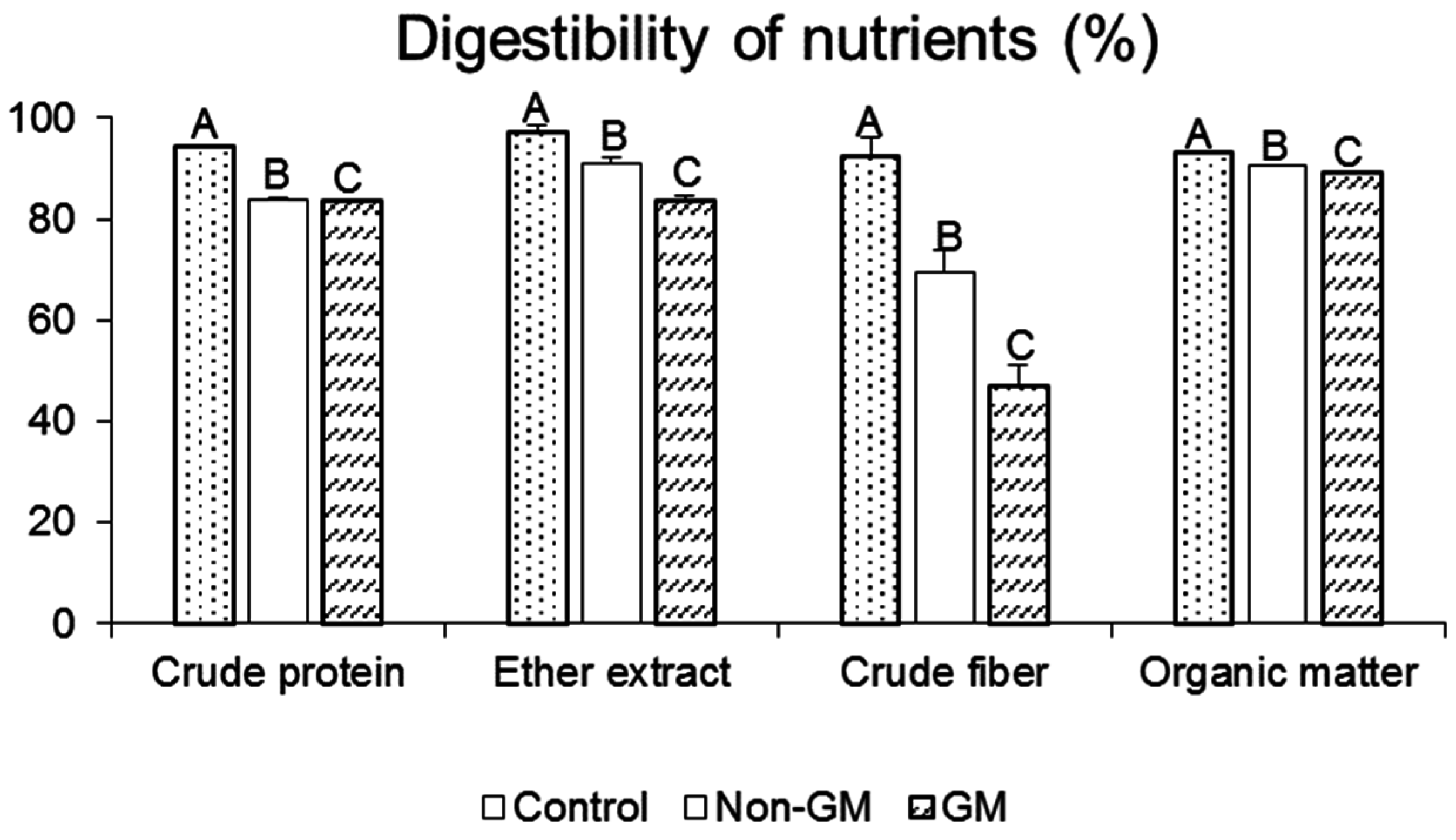
| Parameter | Diets in Experiment I | Diets in Experiment II | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | Non-GM | GM | SEM | p | Control | Non-GM | GM | SEM | p | |
| Diet intake (g/day) | 18.57 | 18.70 | 18.79 | 0.5459 | 0.9591 | 20.79 | 20.27 | 19.34 | 0.5213 | 0.1732 |
| Final body weight (g) | 260.5 | 285.23 | 273.25 | 6.7066 | 0.0605 | 401.33 a | 397.33 a,b | 391.0 b | 2.2277 | 0.0164 |
| Body weight gain (g) | 175.15 | 190.28 | 177.25 | 5.9202 | 0.1813 | 348.75 a | 344.67 a,b | 338.41 b | 2.2544 | 0.0178 |
| Relative Weight of Organs (g/100 g body weight) | ||||||||||
| Liver | 3.80 | 3.44 | 3.64 | 0.1411 | 0.2311 | 5.19 | 5.16 | 5.07 | 0.1044 | 0.6964 |
| Kidneys | 0.78 | 0.76 | 0.77 | 0.0233 | 0.7829 | 0.70 | 0.73 | 0.71 | 0.0187 | 0.6623 |
| Spleen | 0.21 | 0.23 | 0.24 | 0.0152 | 0.4410 | 0.19 | 0.20 | 0.18 | 0.0060 | 0.4050 |
2.2. Hematological and Biochemical Blood Parameters
| Parameter | Diets in Experiment I | Diets in Experiment II | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Control | Non-GM | GM | SEM | p | Control | Non-GM | GM | SEM | p | |
| Hematological Parameters | ||||||||||
| Erythrocytes (1012/L) | 7.90 | 7.60 | 7.55 | 0.1789 | 0.3408 | 6.12 | 5.93 | 6.09 | 0.1814 | 0.7296 |
| Hemoglobin (g/L) | 159.67 | 158.33 | 153.17 | 5.1462 | 0.6490 | 116.17 | 120.67 | 123.5 | 2.1850 | 0.0884 |
| Thrombocytes (G/L) | 646.50 | 508.67 | 571.67 | 63.6548 | 0.3357 | 738.83 | 740.50 | 728.00 | 47.7231 | 0.9800 |
| Leukocytes (G/L) | 5.29 | 7.42 | 9.94 | 1.2288 | 0.0617 | 3.66 | 3.18 | 3.65 | 0.2953 | 0.4381 |
| Monocytes (%) | 1.33 | 1.33 | 1.00 | 0.4303 | 0.8209 | 0.83 | 1.00 | 1.50 | 0.4513 | 0.5662 |
| Lymphocytes (%) | 92.17 | 94.33 | 93.67 | 1.0514 | 0.3540 | 92.66 | 93.00 | 91.00 | 1.4882 | 0.6058 |
| Neutrophils (%) | 6.33 | 4.33 | 5.50 | 0.8829 | 0.3029 | 6.50 | 6.00 | 7.50 | 1.35401 | 0.7323 |
| Biochemical Parameters | ||||||||||
| Alanine aminotransferase (U/L) | 29.50 | 30.33 | 30.33 | 1.1840 | 0.8493 | 30.33 | 36.33 | 34.34 | 1.6667 | 0.0623 |
| Aspartate aminotransferase (U/L) | 119.00 | 123.00 | 121.67 | 19.3710 | 0.9890 | 126.83 | 121.83 | 124.17 | 17.5152 | 0.9798 |
| Total protein (g/L) | 60.50 | 58.33 | 58.83 | 1.1910 | 0.4247 | 58.00 | 59.00 | 59.33 | 1.1418 | 0.6973 |
| Albumins (g/L) | 35.50 a | 32.83 b | 31.67 b | 0.8142 | 0.0134 | 37.17 A | 31.50 B | 31.00 B | 0.6176 | 0.0000 |
| Globulins (g/L) | 25.00 | 25.67 | 27.17 | 1.1164 | 0.3953 | 20.83 A | 27.50 B | 28.33 B | 1.2722 | 0.0014 |
2.3. Blood and Liver Redox Status
| Parameter | Diets in Experiment II | SEM | P | ||
|---|---|---|---|---|---|
| Control | Non-GM | GM | |||
| TAS (mmol/L) | 0.907 a | 0.963 b | 0.978 b | 0.0183 | 0.0342 |
| SOD (U/mL) | 162.33 | 162.83 | 172.84 | 9.8914 | 0.7278 |
| GPx (U/mL) | 137.00 | 136.17 | 140.00 | 3.5945 | 0.7348 |
| TBARS (mmol/L) | 0.198 a | 0.197 a | 0.173 b | 0.0064 | 0.0229 |
| 8-oxo-2′-dG (1/1062′-dG) | 6.8027 | 6.7665 | 6.7487 | 0.4206 | 0.9957 |
2.4. Small Intestine—Relative Weight and Histomorphometric Measurements
| Parameter | Diets in Experiment II | SEM | p | ||
|---|---|---|---|---|---|
| Control | Non-GM | GM | |||
| Relative weight of small intestine (g/100 g BW) | 2.99 | 2.80 | 2.83 | 0.0913 | 0.3224 |
| Histomorphometric Measurements of Jejunum | |||||
| Length of microvilli (nm) | 1933.17 a | 1843.17 b | 1852.83 b | 25.2961 | 0.0458 |
| Length of villi (μm) | 401.66 | 369.00 | 377.50 | 8.9099 | 0.0522 |
| Width of villi (μm) | 69.67 | 68.33 | 67.00 | 1.9512 | 0.6357 |
| Depth of crypts (μm) | 134.83 | 126.67 | 128.83 | 2.2730 | 0.0580 |
| Thickness of muscle layer (μm) | 72.00 | 69.33 | 70.00 | 1.8053 | 0.5662 |
| Thickness of mucus layer (μm) | 143.83 | 134.50 | 140.00 | 7.2620 | 0.6662 |
3. Experimental Section
3.1. Plant Material and Transformation
3.2. Feeding Experiments and Preparation of Animal Material
| Component (%) | Diets in Experiment I | Diets in Experiments II and III | ||||
|---|---|---|---|---|---|---|
| Control | Non-GM | GM | Control | Non-GM | GM | |
| Flaxseed cake | – | 15.00 | 15.00 | – | 30.00 | 30.00 |
| Maize starch | 70.52 | 66.01 | 67.00 | 70.52 | 58.5 | 56.53 |
| Casein | 15.70 | 10.70 | 10.40 | 15.70 | 5.10 | 5.70 |
| Cellulose | 4.00 | 3.00 | 2.80 | 4.00 | 1.60 | 2.00 |
| Flaxseed oil | 4.98 | 0.49 | – | 4.98 | – | 0.97 |
| Mineral mix * | 3.50 | 3.50 | 3.50 | 3.50 | 3.50 | 3.50 |
| Vitamin mix ** | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| Choline chloride | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 | 0.20 |
| dl-Methionine | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 | 0.10 |
| Nutritional Value (% Dry Matter) | ||||||
| Crude protein | 12.03 | 12.01 | 12.02 | 12.03 | 12.03 | 12.02 |
| Ether extract | 4.97 | 4.98 | 4.98 | 4.97 | 4.97 | 4.96 |
| Crude fiber | 4.00 | 3.99 | 4.01 | 4.00 | 4.01 | 4.02 |
| Metabolizable energy (kcal/kg) | 377.14 | 372.62 | 372.43 | 413.88 | 408.02 | 407.47 |
3.3. Nutrient Digestibility
3.4. Hematological Blood Parameters
3.5. Biochemical Blood Parameters
3.6. Total Antioxidant Status of Blood Plasma
3.7. Activity of Superoxide Dismutase in Erythrocytes
3.8. Activity of Glutathione Peroxidase in Blood Plasma
3.9. Peroxidation of Liver Lipids
3.10. Oxidative Damage of Liver DNA
3.11. Histological Evaluation of the Small Intestine (Jejunum)
3.12. Analysis of the Ultrastructure of the Small Intestine (Jejunum)
3.13. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Lorenc-Kukuła, K.; Amarowicz, R.; Oszmiański, J.; Doermann, P.; Starzycki, M.; Skała, J.; Żuk, M.; Kulma, A.; Szopa, J. Pleiotropic effects of phenolic compounds content increases in transgenic flax plant. J. Agric. Food Chem. 2005, 53, 3685–3692. [Google Scholar] [CrossRef] [PubMed]
- Żuk, M.; Kulma, A.; Dymińska, L.; Szołtysek, K.; Prescha, A.; Hanuza, J.; Szopa, J. Flavonoid engineering of flax potentiate its biotechnological application. BMC Biotechnol. 2011, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.J.; Papa, S.; Bolaños, J.; Bruckdorfer, R.; Carlsen, H.; Elliott, R.M.; Flier, J.; Griffiths, H.R.; Heales, S.; Holst, B.; et al. Antioxidants, reactive oxygen and nitrogen species, gene induction and mitochondrial function. Mol. Asp. Med. 2002, 23, 209–285. [Google Scholar] [CrossRef]
- Moon, H.K.; Yang, E.S.; Park, J.W. Protection of peroxynitrite-induced DNA damage by dietary antioxidants. Arch. Pharm. Res. 2006, 29, 213–217. [Google Scholar] [CrossRef] [PubMed]
- González, R.; Ballester, I.; López-Posadas, R.; Suárez, M.D.; Zarzuelo, A.; Martinez-Augustin, O.; de Medina, F.S. Effects of flavonoids and other polyphenols on inflammation. Crit. Rev. Food Sci. Nutr. 2011, 51, 331–362. [Google Scholar] [CrossRef] [PubMed]
- Soobrattee, M.A.; Neergheen, V.S.; Luximon-Ramma, A.; Aruoma, O.I.; Bahorun, T. Phenolics as potential antioxidant therapeutic agents: Mechanism and actions. Mutat. Res. 2005, 579, 200–213. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.S.; Leu, K.L.; Huang, G.J.; Yeh, C.F.; Tai, H.M.; Ho, W.Y.; Huang, M.H. Protective effects of an aqueous Pericarpium granati extract against inflammatory damage in mice. J. Funct. Foods 2014, 9, 183–191. [Google Scholar] [CrossRef]
- Mitchell, J.H.; Gardner, P.T.; McPhail, D.B.; Morrice, P.C.; Collins, A.R.; Duthie, G.G. Antioxidant efficacy of phytoestrogens in chemical and biological model systems. Arch. Biochem. Biophys. 1998, 360, 142–148. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K. Oxidative stress as a mechanism of diabetes in diabetic BB prone rats: Effect of secoisolariciresinol diglucoside (SDG). Mol. Cell. Biochem. 2000, 209, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K.; Mantha, S.V; Muir, A.D.; Westcott, N.D. Protective effect of secoisolariciresinol diglucoside against streptozotocin-induced diabetes and its mechanism. Mol. Cell. Biochem. 2000, 206, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Varga, T.K.; Diosady, L.L. Simultaneous extraction of oil and antinutritional compounds from flaxseed. J. Am. Oil Chem. Soc. 1994, 71, 603–607. [Google Scholar] [CrossRef]
- Eun-Ryong, P.; Jin-Hwan, H.; Dong-Ha, L.; Sang-Bae, H.; Kang-Bong, L.; Jae-Seok, P.; Hyung-Wook, C.; Kyung-Hyun, H.; Myung-Chul, K. Analysis and decrease of cyanogenic glucosides in flaxseed. J. Korean Soc. Food Sci. Nutr. 2005, 34, 875–879. [Google Scholar]
- Oomah, B.D.; Mazza, G. Effect of dehulling on chemical composition and physical properties of flaxseed. LWT Food Sci. Technol. 1997, 30, 135–140. [Google Scholar] [CrossRef]
- Siegień, I. Cyjanogeneza u roślin i jej efektywność w ochronie roślin przed atakiem roślinożerców i patogenów. Kosmos 2007, 56, 155–166. [Google Scholar]
- Shen, Y.; Feng, D.; Oresanya, T.F.; Chavez, E.R. Fatty acid and nitrogen utilization of processed flaxseed by adult chickens. J. Sci. Food Agric. 2005, 85, 1137–1142. [Google Scholar] [CrossRef]
- Barrett, M.D.; Hill, D.C.; Alexander, J.C.; Zitnak, A. Fate of orally dosed linamarin in the rat. Can. J. Physiol. Pharmacol. 1977, 55, 134–136. [Google Scholar] [CrossRef] [PubMed]
- Oke, O.L. Some aspects of the role of cyanogenic glycosides in nutrition. World Rev. Nutr. Diet. 1979, 33, 70–103. [Google Scholar] [PubMed]
- Skibola, C.F.; Smith, M.T. Potential health impacts of excessive flavonoid intake. Free Radic. Biol. Med. 2000, 29, 375–383. [Google Scholar] [CrossRef]
- Azevedo, L.; Gomes, J.; Stringheta, P.; Gontijo, Á.M.M.C.; Padovani, C.R.; Ribeiro, LR.; Salvadori, D.M.F. Black bean (Phaseolus vulgaris L.) as a protective agent against DNA damage in mice. Food Chem. Toxicol. 2003, 41, 1671–1676. [Google Scholar] [CrossRef]
- Orzechowski, A.; Ostaszewski, P.; Jank, M.; Berwid, S.J. Bioactive substances of plant origin in food—Impact on genomics. Reprod. Nutr. Dev. 2002, 42, 461–477. [Google Scholar] [CrossRef] [PubMed]
- Sorrenti, V.; Mazza, F.; Campisi, A.; di Giacomo, C.; Acquaviva, R.; Vanella, L.; Galvano, F. Heme oxygenase induction by cyanidin-3-O-β-glucoside in cultured human endothelial cells. Mol. Nutr. Food Res. 2007, 51, 580–586. [Google Scholar] [CrossRef] [PubMed]
- Fofana, B.; Cloutier, S.; Kirby, C.W.; McCallum, J.; Duguid, S. A well balanced ω-6/ω-3 ratio in developing flax bolls after heating and its implications for use as a fresh vegetable by humans. Food Res. Int. 2011, 44, 2459–2464. [Google Scholar] [CrossRef]
- Tominaga, S.; Nishi, K.; Nishimoto, S.; Akiyama, K.; Yamauchi, S.; Sugahara, T. (−)-Secoisolariciresinol attenuates high-fat diet-induced obesity in C57BL/6 mice. Food Funct. 2012, 3, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Oomah, B.D.; Mazza, G.; Kenaschuk, E.O. Cyanogenic compounds in flaxseed. J. Agric. Food Chem. 1992, 40, 1346–1348. [Google Scholar] [CrossRef]
- Cardoso Carraro, J.C.; Dantas, M.I.D.S.; Espeschit, A.C.R.; Martino, H.S.D.; Ribeiro, S.M.R. Flaxseed and human health: Reviewing benefits and adverse effects. Food Rev. Int. 2012, 28, 203–230. [Google Scholar] [CrossRef]
- Ortiz, L.T.; Rebole, A.; Alzueta, C.; Rodriguez, M.L.; Treviño, J. Metabolisable energy value and digestibility of fat and fatty acids in linseed determined with growing broiler chickens. Br. Poult. Sci. 2001, 42, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, M.L.; Alzueta, C.; Rebolé, A.; Ortiz, LT.; Centeno, C.; Treviño, J. Effect of inclusion level of linseed on the nutrient utilisation of diets for growing broiler chickens. Br. Poult. Sci. 2001, 42, 368–375. [Google Scholar] [CrossRef] [PubMed]
- Roth-Maier, D.A.; Kirchgessner, M. Feeding full-fat flax seed and sunflower seed to fattening chickens. Arch. Gefluegelkunde 1995, 59, 319–322. [Google Scholar]
- Treviño, J.; Rodriguez, M.L.; Ortiz, L.T.; Rebolé, A.; Alzueta, C. Protein quality of linseed for growing broiler chicks. Anim. Feed Sci. Technol. 2000, 84, 155–166. [Google Scholar] [CrossRef]
- Alzueta, C.; Rodriguez, M.L.; Cutuli, M.T.; Rebolé, A.; Ortiz, L.T.; Centeno, C.; Treviño, J. Effect of whole and demucilaged linseed in broiler chicken diets on digesta viscosity, nutrient utilization and intestinal microflora. Br. Poult. Sci. 2003, 44, 67–74. [Google Scholar] [CrossRef] [PubMed]
- Hemmings, S.J.; Barker, L. The effects of dietary flaxseed on the Fischer 344 rat: I. Development, behaviour, toxicity and the activity of liver γ-glutamyltranspeptidase. Cell Biochem. Funct. 2004, 22, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Matthews, K.R.; Homer, D.B.; Thies, F.; Calder, P.C. Effect of whole linseed (Linum usitatissiumum) in the diet of finishing pigs on growth performance and on the quality and fatty acid composition of various tissues. Br. J. Nutr. 2000, 83, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Batterham, E.S.; Andersen, L.M.; Baigent, D.R.; Green, A.G. Evaluation of meals from Linola™ low-linolenic acid linseed and conventional linseed as protein sources for growing pigs. Anim. Feed Sci. Technol. 1991, 35, 181–190. [Google Scholar] [CrossRef]
- Askar, A.; Morad, M.M. Lebensmittelvergiftigung 1. Toxine in natürlichen Lebensmittel. Alimentia 1983, 19, 59–66. [Google Scholar]
- Frankenberg, L.; Sörbo, B. Effect of cyanide antidotes on the metabolic conversion of cyanide to thiocyanate. Arch. Toxicol. 1975, 33, 81–89. [Google Scholar] [CrossRef] [PubMed]
- Shoji, T.; Akazome, Y.; Kanda, T.; Ikeda, M. The toxicology and safety of apple polyphenol extract. Food Chem. Toxicol. 2004, 42, 959–967. [Google Scholar] [CrossRef] [PubMed]
- Iyayi, E.A. Supplement effects of low- and high-cyanide cassava peels on the performance, nutrient digestibility, and serum metabolites of growing pigs. J. Agric. Trop. Subtrop. 1994, 95, 197–205. [Google Scholar]
- Ibebunjo, C.; Kamalu, B.P.; Ihemelandu, E.C. Comparison of the effects of cassava (Manihot esculenta Crantz) organic cyanide and inorganic cyanide on muscle and bone development in a Nigerian breed of dog. Br. J. Nutr. 1992, 68, 483–491. [Google Scholar] [CrossRef] [PubMed]
- Kamalu, B.P. Pathological changes in growing dogs fed on a balanced cassava (Manihot esculenta Crantz) diet. Br. J. Nutr. 1993, 69, 921–934. [Google Scholar] [CrossRef] [PubMed]
- Kamalu, B.P. Cassava (Manihot esculenta Crantz) in the aetiology of kwashiorkor. Nutr. Res. Rev. 1993, 6, 121–135. [Google Scholar] [CrossRef] [PubMed]
- Okafor, P.N.; Anoruo, K.; Bonire, A.O.; Maduagwu, E.N. The role of low-protein and cassava-cyanide intake in the aetiology of tropical pancreatitis. Glob. J. Pharmacol. 2008, 2, 6–10. [Google Scholar]
- Lim, B.O.; Yamada, K.; Nonaka, M.; Kuramoto, Y.; Hung, P.; Sugano, M. Dietary fibers modulate indices of intestinal immune function in rats. J. Nutr. 1997, 127, 663–667. [Google Scholar] [PubMed]
- Yamada, K.; Hung, P.; Yoshimura, K.; Taniguchi, S.; Lim, B.O.; Sugano, M. Effect of unsaturated fatty acids and antioxidants on immunoglobulin production by mesenteric lymph node lymphocytes of Sprague-Dawley rats. J. Biochem. 1996, 120, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Middleton, E., Jr.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar] [PubMed]
- Tudisco, R.; Lombardi, P.; Bovera, F.; DAngelo, D.; Cutrignelli, M.I.; Mastellone, V.; Terzi, V.; Avallone, L.; Infascelli, F. Genetically modified soya bean in rabbit feeding: Detection of DNA fragments and evaluation of metabolic effects by enzymatic analysis. Anim. Sci. 2006, 82, 193–199. [Google Scholar] [CrossRef]
- Tudisco, R.; Mastellone, V.; Cutrignelli, M.I.; Lombardi, P.; Bovera, F.; Mirabella, N.; Picollo, G.; Calabrò, S.; Avallone, L.; Infascelli, F. Fate of transgenic DNA and evaluation of metabolic effects in goats fed genetically modified soybean and in their offsprings. Animal 2010, 4, 1662–1671. [Google Scholar] [CrossRef] [PubMed]
- Tudisco, R.; Calabro, S.; Cutrignelli, M.I.; Moniello, G.; Grossi, M.; Mastellone, V.; Terzi, V.; Avallone, L.; Infascelli, F. Genetically modified soybean in a goat diet: Influence on kid performance. Small Rumin. Res. 2015, 126, 67–74. [Google Scholar] [CrossRef]
- Huang, C.Y.; Chen, L.H.; Osio, Y.; Cohen, D.A. Effects of diet composition on liver antioxidant defense and detoxification enzymes in mice with murine AIDS. Nutr. Res. 1994, 14, 1841–1851. [Google Scholar] [CrossRef]
- Makni, M.; Fetoui, H.; Gargouri, N.K.; Garoui, E.M.; Jaber, H.; Makni, J.; Boudawara, T.; Zeghal, N. Hypolipidemic and hepatoprotective effects of flax and pumpkin seed mixture rich in ω-3 and ω-6 fatty acids in hypercholesterolemic rats. Food Chem. Toxicol. 2008, 46, 3714–3720. [Google Scholar] [CrossRef] [PubMed]
- Makni, M.; Fetoui, H.; Garoui, E.M.; Gargouri, N.K.; Jaber, H.; Makni, J.; Boudawara, T.; Zeghal, N. Hypolipidemic and hepatoprotective seeds mixture diet rich in ω-3 and ω-6 fatty acids. Food Chem. Toxicol. 2010, 48, 2239–2246. [Google Scholar] [CrossRef] [PubMed]
- Idibie, C.A.; Davids, H.; Iyuke, S.E. Cytotoxicity of purified cassava linamarin to a selected cancer cell lines. Bioprocess Biosyst. Eng. 2007, 30, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Prasad, K. Hypocholesterolemic and antiatherosclerotic effect of flax lignan complex isolated from flaxseed. Atherosclerosis 2005, 179, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Moneim, A.E.; Dkhil, M.A.; Al-Quraishy, S. The redox status in rats treated with flaxseed oil and lead-induced hepatotoxicity. Biol. Trace Elem. Res. 2011, 143, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Kim, M. The water soluble extract of chicory affects rat intestinal morphology similarly to other non-starch polysaccharides. Nutr. Res. 2002, 22, 1299–1307. [Google Scholar] [CrossRef]
- Domenghini, C.; di Giancamillo, A.; Boshi, G.; Arrighi, S. Can nutraceuticals affect the structure of intestinal mucosa? Qualitative and quantitative microanatomy in l-glutamine diet-supplemented weaning piglets. Vet. Res. Commun. 2006, 30, 331–342. [Google Scholar] [CrossRef] [PubMed]
- Ryan, C.K.; Miller, J.H.; Seydel, A.S.; de Mesy Jensen, K.; Sax, H.C. Epidermal growth factor and neurotensin induce microvillus hypertrophy following massive enterectomy. J. Gastrointest. Surg. 1997, 1, 467–473. [Google Scholar] [CrossRef]
- Kawaguchi, A.L.; Dunn, J.C.Y.; Lam, M.; O’Connor, T.P.; Diamond, J.; Fonkalsrud, E.W. Glucose uptake in dilated small intestine. J. Ped. Surg. 1998, 33, 1670–1673. [Google Scholar] [CrossRef]
- NRC. Nutrient Requirements of Laboratory Animals. National Research Council, 4th ed.; National Academy of Sciences: Washington, DC, USA, 1996. [Google Scholar]
- AOAC. Official Methods of Analysis of the Association of Official Analytical Chemists, 15th ed.; AOAC: Arlington, TX, USA, 1996. [Google Scholar]
- Uchiyama, M.; Mihara, M. Determination of malonaldehyde precursor in tissues by thiobarbituric acid test. Anal. Biochem. 1978, 86, 271–278. [Google Scholar] [CrossRef]
- Foksinski, M.; Kotzbach, R.; Szymanski, W.; Olinski, R. The level of typical biomarker of oxidative stress 8-hydroxy-2′-deoxyguanosine is higher in uterine myomas than in control tissues and correlates with the size of the tumor. Free Radic. Biol. Med. 2000, 29, 597–601. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matusiewicz, M.; Kosieradzka, I.; Zuk, M.; Szopa, J. Effect of Dose and Administration Period of Seed Cake of Genetically Modified and Non-Modified Flax on Selected Antioxidative Activities in Rats. Int. J. Mol. Sci. 2015, 16, 14259-14275. https://doi.org/10.3390/ijms160614259
Matusiewicz M, Kosieradzka I, Zuk M, Szopa J. Effect of Dose and Administration Period of Seed Cake of Genetically Modified and Non-Modified Flax on Selected Antioxidative Activities in Rats. International Journal of Molecular Sciences. 2015; 16(6):14259-14275. https://doi.org/10.3390/ijms160614259
Chicago/Turabian StyleMatusiewicz, Magdalena, Iwona Kosieradzka, Magdalena Zuk, and Jan Szopa. 2015. "Effect of Dose and Administration Period of Seed Cake of Genetically Modified and Non-Modified Flax on Selected Antioxidative Activities in Rats" International Journal of Molecular Sciences 16, no. 6: 14259-14275. https://doi.org/10.3390/ijms160614259
APA StyleMatusiewicz, M., Kosieradzka, I., Zuk, M., & Szopa, J. (2015). Effect of Dose and Administration Period of Seed Cake of Genetically Modified and Non-Modified Flax on Selected Antioxidative Activities in Rats. International Journal of Molecular Sciences, 16(6), 14259-14275. https://doi.org/10.3390/ijms160614259







