Ultrasound Assessment of Carotid Plaque Echogenicity Response to Statin Therapy: A Systematic Review and Meta-Analysis
Abstract
:1. Introduction
2. Results
2.1. Study Selection
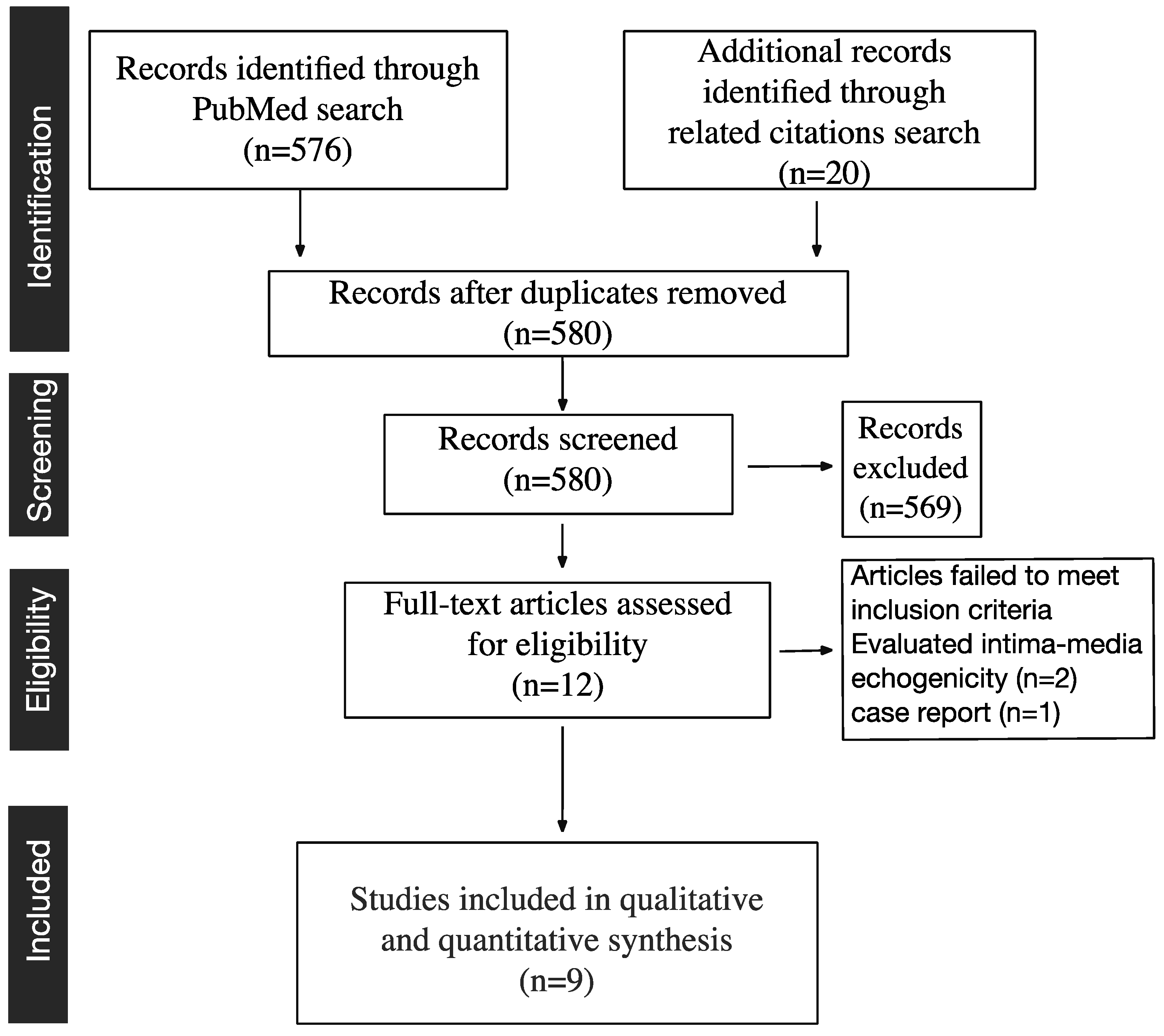
2.2. Qualitative Assessment and Study Characteristics
2.2.1. Effect of Statins on Plaque Features
2.2.2. Effect of Statins on Blood Lipids and Inflammatory Markers
| Author/Year | Population (n) | Mean Age ± SD | Gender (Male) | Hypercholesterolemic | Carotid Stenosis | Echogenicity Measured | Minor Score |
|---|---|---|---|---|---|---|---|
| 1. Watanabe et al., 2005 [12] | 30 | 69.9 ± 8.8 | 63% | No | Moderate | IBS | RT |
| 2. Yamagami et al., 2008 [13] | 41 | 63.4 ± 8.3 | 24% | Yes | Moderate | IBS | RT |
| 3. Nakamura et al., 2008 [14] | 33 | 60 ± 9 | 25% | Yes | Moderate | IBS | RT |
| 4. Kadoglou et al., 2008 [15] | 113 | 63.6 ± 9.9 | 67% | Yes | Moderate symptomatic | GSM | 20 |
| 5. Yamada et al., 2009 [16] | 40 | 71 ± 8 | 90% | No | 30%–60% | IBS | RT |
| 6. Kadoglou et al., 2009 [17] | 67 + 46 | 66.7 ± 7.3 | 40% | No | >40% | GSM | 20 |
| 7. Kadoglou et al., 2010 [18] | 66 + 65 | 64.9 ± 10 | 46% | Yes | 30%–60% | GSM | 24 |
| 8. Della-Morte et al., 2011 [19] | 40 | >45 | NA | Yes | NA | GSM | 15 |
| 9. Nohara et al., 2013 [20] | 25 | 63.9 ± 8.1 | 50% | Yes | NA | GSM | 20 |
| Author/Year | Study Design | Statin/Dose | Follow-up (Months) | % Change Echogenicity | % Change LDL | % Change HDL | % Change hsCRP |
|---|---|---|---|---|---|---|---|
| 1. Watanabe et al., 2005 [12] | Randomized case-control trial | Pravastatin | 6 | 14.1 ± 3.3 | 24.5 ± 6.4 | 10.2 ± 6.0 | 45.0 ± 58.3 |
| 3. Yamagami et al., 2008 [13] | Randomized case-control trial | Simvastatin 10 mg | 1 | 10.6 ± 4.3 | 34.2 ± 18.4 | 0 | 43.0 ± 119.8 |
| 2. Nakamura et al., 2008 [14] | Randomized case-control trial | Pitavastatin 4 mg | 12 | 32.1 ± 5.9 | 37.8 ± 12.4 | 9.3 ± 2.0 | 43.7 ± 51.5 |
| 4. Kadoglou et al., 2008 [15] | Open-label prospective trial | Atorvastatin | 6 | 36.0 ± 15.2 | 41.7 ± 19.9 | 4.5 ± 2.4 | 58.9 ± 34.0 |
| 5. Yamada et al., 2009 [16] | Randomized case-control trial | Simvastatin | 6 | 17.0 ± 5.9 | 44.0 ± 23.9 | 0 | 42.1 ± 94.6 |
| 6a. Kadoglou et al., 2009 [17] | Open-label prospective trial | Atorvastatin | 6 | 36.8 ± 9.8 | 38.6 ± 20.0 | 13.4 ± 6.6 | 78.3 ± 74.9 |
| 6b. Kadoglou et al., 2009 [17] | Open-label prospective trial | Atorvastatin + CAS | 6 | 48.4 ± 18.6 | 33.3 ± 15.0 | 4.4 ± 2.2 | 52.1 ± 39.5 |
| 7a. Kadoglou et al., 2010 [18] | Randomized case-control trial | Atorvastatin 10–20 mg | 12 | 32.6 ± 11.7 | 64.5 ± 23.6 | 5.5 ± 2.6 | 52.9 ± 55.2 |
| 7b. Kadoglou et al., 2010 [18] | Randomized case-control trial | Atorvastatin 80 mg | 12 | 51.4 ± 18.4 | 54.2 ± 37.2 | 10.3 ± 6.0 | 65.0 ± 80.0 |
| 8. Della-Morte et al., 2011 [19] | Prospective pilot study | NA | 1 | 21.9 ± 4.8 | 51.4 ± 31.0 | 2.0 ± 1.1 | NA |
| 9. Nohara et al., 2013 [20] | Prospective open label, blinded-endpoint | Rosuvastatin | 12 | 16.9 ± 33.1 | 50.1 ± 22.9 | 8.1 ± 3.6 | NA |
2.3. Meta-Analysis Results
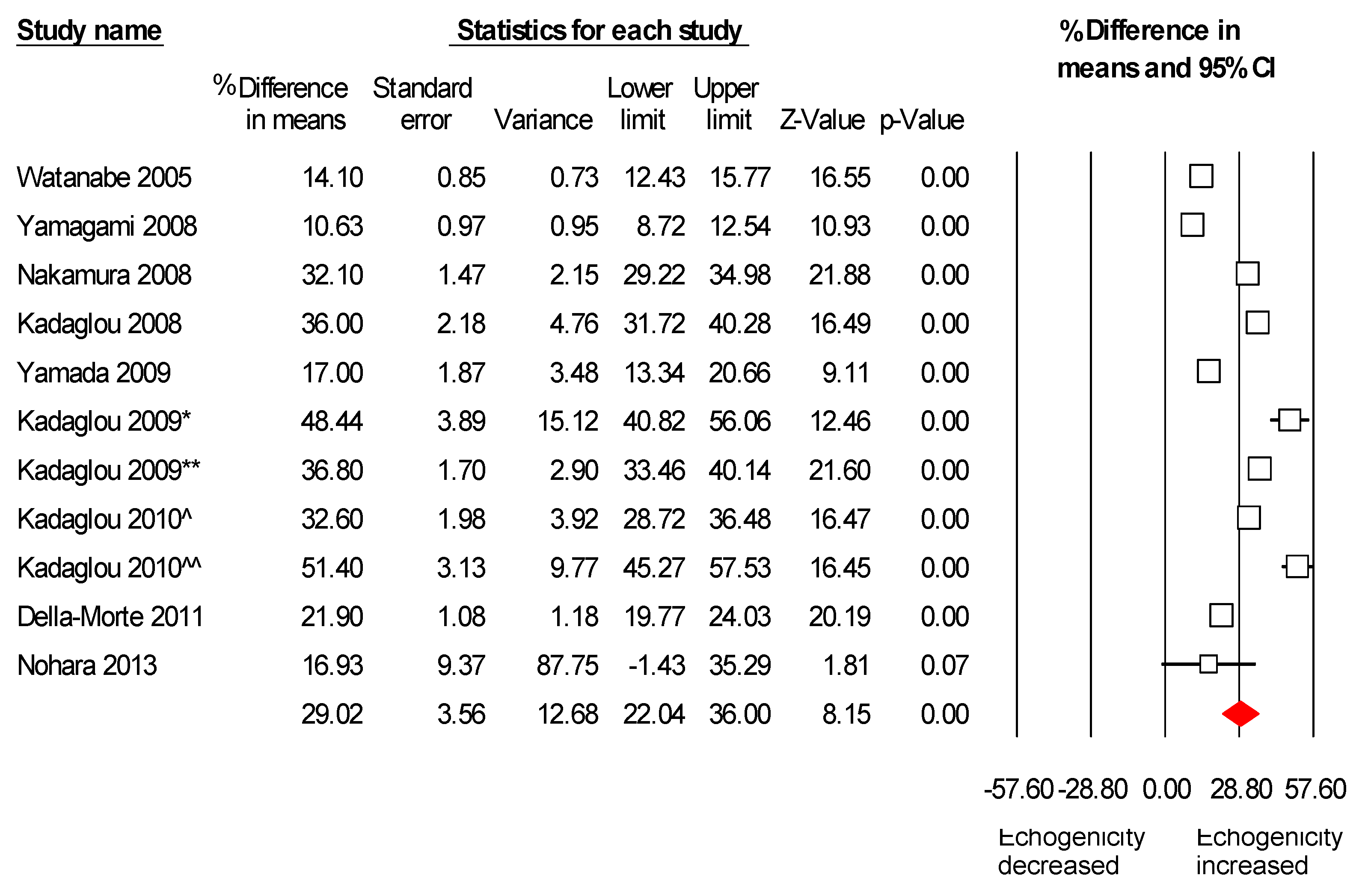
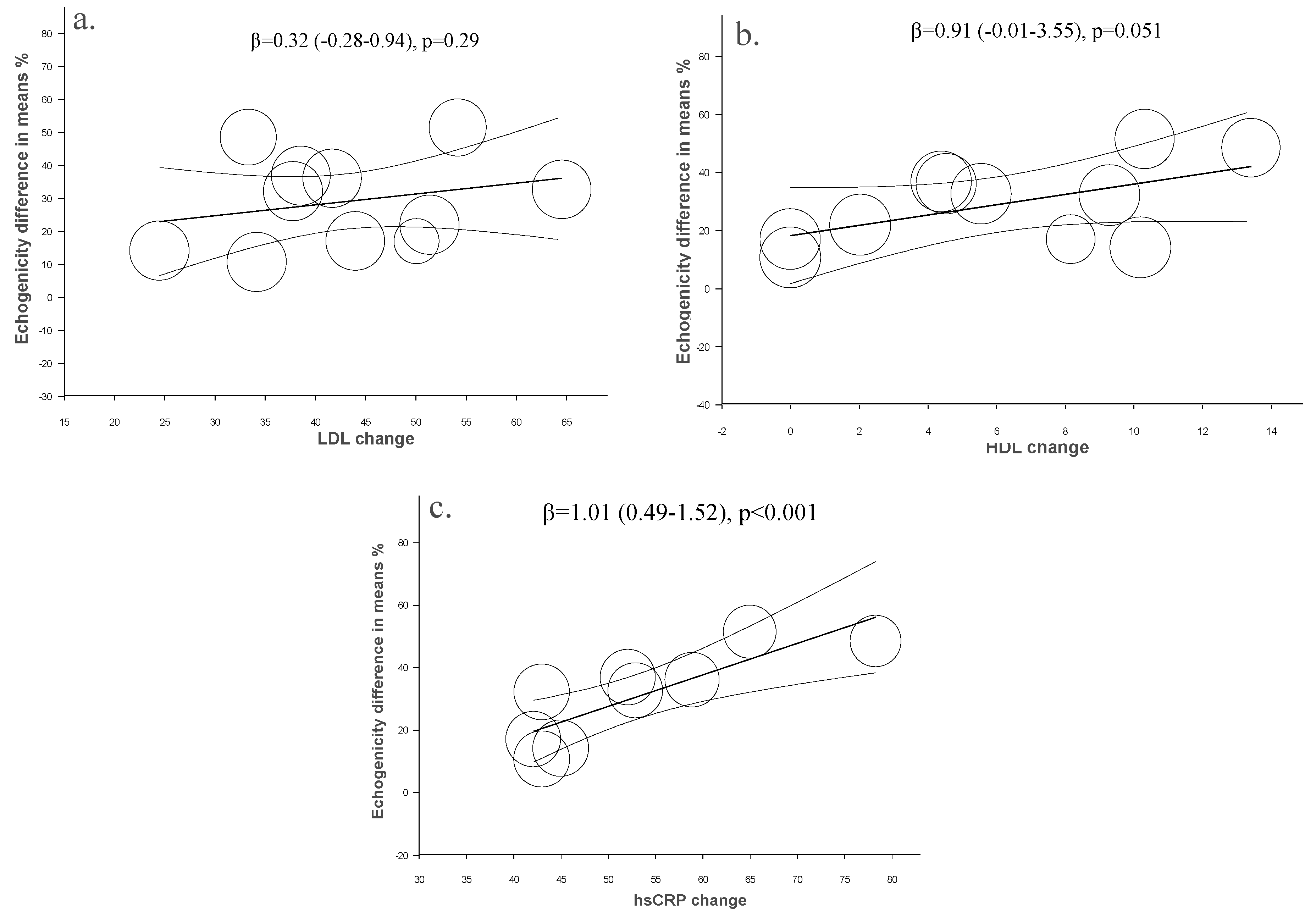
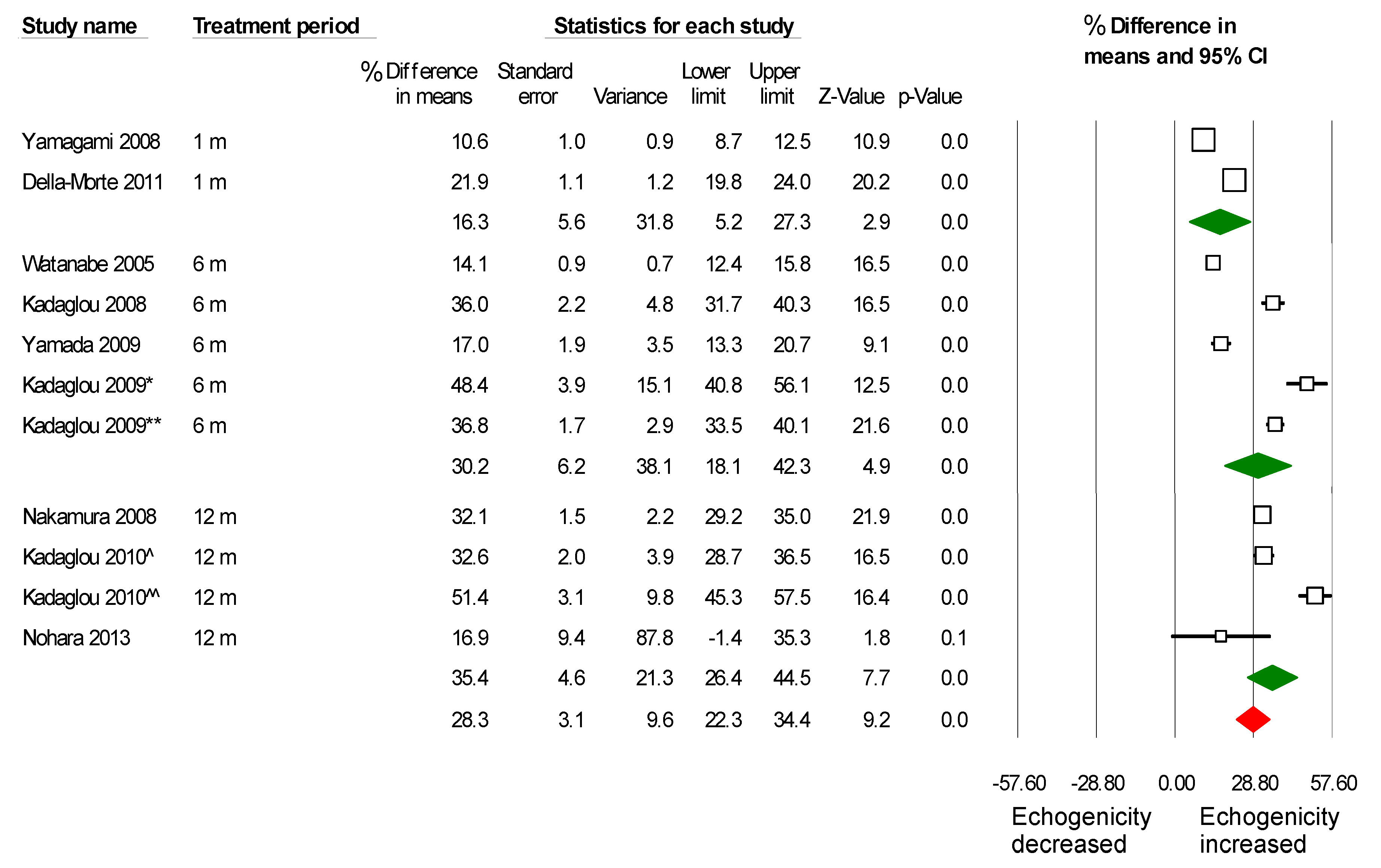
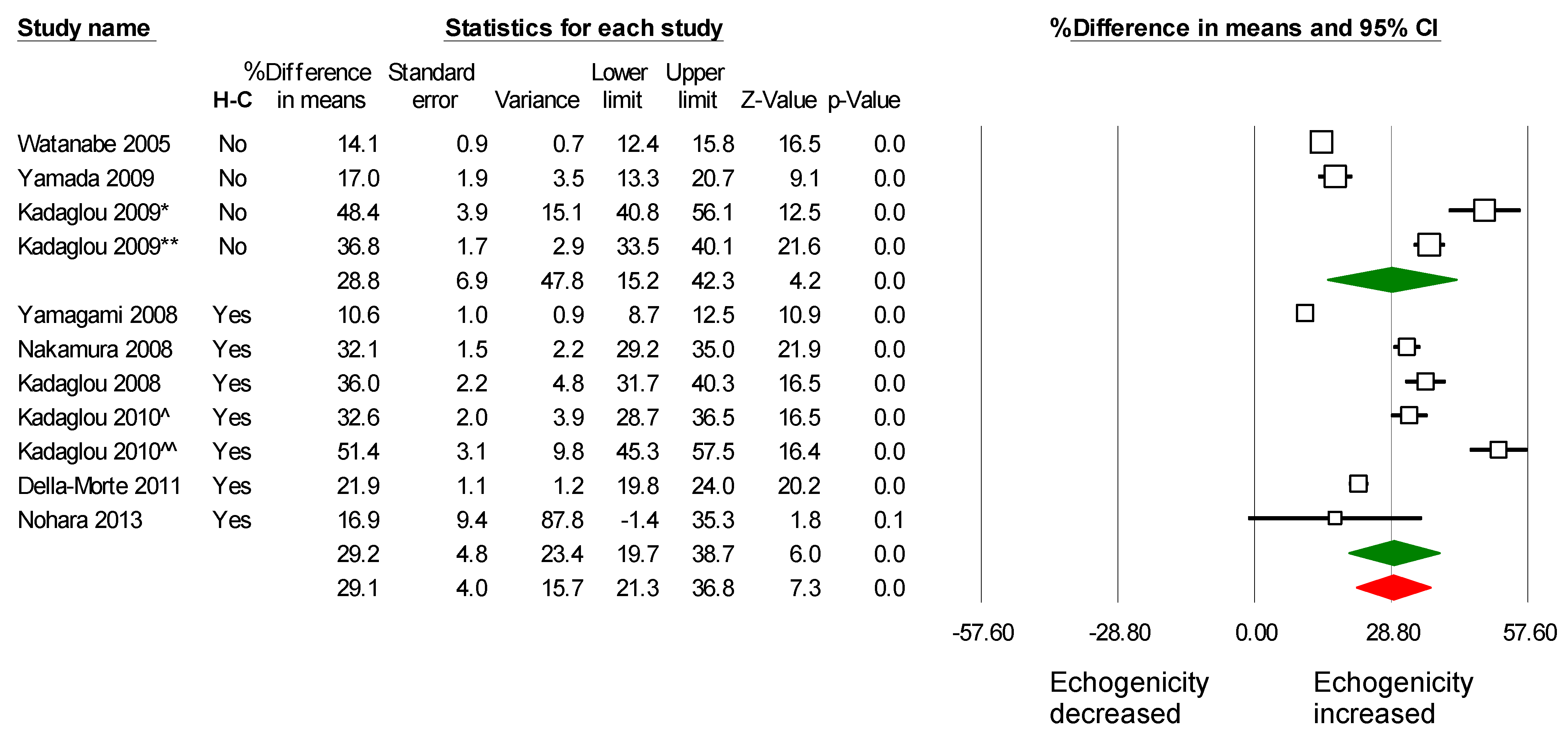
2.4. Assessment of Potential Publication Bias
| Test of Heterogeneity | Publication Bias (Begg and Mazumdar Rank Correlation) | |||
|---|---|---|---|---|
| I2 | Kendell Tau | Test Statistic Z | p-Value | |
| Echogenicity | 92.1 | 0.34 | 1.41 | 0.16 |
| LDL | 81.2 | −0.33 | 1.25 | 0.12 |
| HDL | 98.1 | 0.05 | 0.20 | 0.41 |
| hsCRP | 0 | 0 | 0 | 1.0 |
3. Discussion
4. Experimental Section
4.1. Methods
4.2. Information Search and Data Collection
4.3. Study Eligibility Criteria
4.4. Statistical Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Conflicts of Interest
References
- Ibrahimi, P.; Jashari, F.; Nicoll, R.; Bajraktari, G.; Wester, P.; Henein, M.Y. Coronary and carotid atherosclerosis: How useful is the imaging? Atherosclerosis 2013, 231, 323–333. [Google Scholar] [CrossRef] [PubMed]
- Amarenco, P.; Labreuche, J.; Lavallee, P.; Touboul, P.J. Statins in stroke prevention and carotid atherosclerosis: Systematic review and up-to-date meta-analysis. Stroke J. Cereb. Circ. 2004, 35, 2902–2909. [Google Scholar] [CrossRef]
- Martinez-Sanchez, P.; Serena, J.; Alexandrov, A.V.; Fuentes, B.; Fernandez-Dominguez, J.; Diez-Tejedor, E. Update on ultrasound techniques for the diagnosis of cerebral ischemia. Cerebrovasc. Dis. 2009, 27, 9–18. [Google Scholar] [PubMed]
- Seeger, J.M.; Barratt, E.; Lawson, G.A.; Klingman, N. The relationship between carotid plaque composition and neurologic symptoms. J. Surg. Res. 1987, 43, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Gronholdt, M.L.; Nordestgaard, B.G.; Bentzon, J.; Wiebe, B.M.; Zhou, J.; Falk, E.; Sillesen, H. Macrophages are associated with lipid-rich carotid artery plaques, echolucency on B-mode imaging, and elevated plasma lipid levels. J. Vasc. Surg. 2002, 35, 137–145. [Google Scholar] [PubMed]
- El-Barghouty, N.M.; Levine, T.; Ladva, S.; Flanagan, A.; Nicolaides, A. Histological verification of computerised carotid plaque characterisation. Eur. J. Vasc. Endovasc. Surg. 1996, 11, 414–416. [Google Scholar] [CrossRef] [PubMed]
- Makris, G.C.; Lavida, A.; Nicolaides, A.N.; Geroulakos, G. The effect of statins on carotid plaque morphology: A LDL-associated action or one more pleiotropic effect of statins? Atherosclerosis 2010, 213, 8–20. [Google Scholar] [CrossRef] [PubMed]
- Noyes, A.M.; Thompson, P.D. A systematic review of the time course of atherosclerotic plaque regression. Atherosclerosis 2014, 234, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Lind, L.P.S.; den Ruijter, H.M.; Palmer, M.K.; Grobbee, D.E.; Crouse, J.R., 3rd; O’Leary, D.H.; Evans, G.W.; Raichlen, J.S.; Bots, M.L. Effect of rosuvastatin on the echolucency of the common carotid intima-media in low-risk individuals: The METEOR trial. J. Am. Soc. Echocardiogr. 2012, 25, 1120–1127. [Google Scholar] [CrossRef] [PubMed]
- Yamagishi, T.; Kato, M.; Koiwa, Y.; Omata, K.; Hasegawa, H.; Kanai, H. Evaluation of plaque stabilization by fluvastatin with carotid intima-medial elasticity measured by a transcutaneous ultrasonic-based tissue characterization system. J. Atheroscler. Thromb. 2009, 16, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Stivali, G.; Cerroni, F.; Bianco, P.; Fiaschetti, P.; Cianci, R. Images in cardiovascular medicine. Carotid plaque reduction after medical treatment. Circulation 2005, 112, e276–e277. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Sugiyama, S.; Kugiyama, K.; Honda, O.; Fukushima, H.; Koga, H.; Horibata, Y.; Hirai, T.; Sakamoto, T.; Yoshimura, M.; et al. Stabilization of carotid atheroma assessed by quantitative ultrasound analysis in nonhypercholesterolemic patients with coronary artery disease. J. Am. Coll. Cardiol. 2005, 46, 2022–2030. [Google Scholar] [CrossRef] [PubMed]
- Yamagami, H.; Sakaguchi, M.; Furukado, S.; Hoshi, T.; Abe, Y.; Hougaku, H.; Hori, M.; Kitagawa, K. Statin therapy increases carotid plaque echogenicity in hypercholesterolemic patients. Ultrasound Med. Biol. 2008, 34, 1353–1359. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, T.; Obata, J.E.; Kitta, Y.; Takano, H.; Kobayashi, T.; Fujioka, D.; Saito, Y.; Kodama, Y.; Kawabata, K.; Mende, A.; et al. Rapid stabilization of vulnerable carotid plaque within 1 month of pitavastatin treatment in patients with acute coronary syndrome. J. Cardiovasc. Pharmacol. 2008, 51, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Kadoglou, N.P.; Gerasimidis, T.; Moumtzouoglou, A.; Kapelouzou, A.; Sailer, N.; Fotiadis, G.; Vitta, I.; Katinios, A.; Kougias, P.; Bandios, S.; et al. Intensive lipid-lowering therapy ameliorates novel calcification markers and GSM score in patients with carotid stenosis. Eur. J. Vasc. Endovasc. Surg. 2008, 35, 661–668. [Google Scholar] [CrossRef] [PubMed]
- Yamada, K.; Yoshimura, S.; Kawasaki, M.; Enomoto, Y.; Asano, T.; Minatoguchi, S.; Iwama, T. Effects of atorvastatin on carotid atherosclerotic plaques: A randomized trial for quantitative tissue characterization of carotid atherosclerotic plaques with integrated backscatter ultrasound. Cerebrovasc. Dis. 2009, 28, 417–424. [Google Scholar] [CrossRef] [PubMed]
- Kadoglou, N.P.; Gerasimidis, T.; Kapelouzou, A.; Moumtzouoglou, A.; Avgerinos, E.D.; Kakisis, J.D.; Karayannacos, P.E.; Liapis, C.D. Beneficial changes of serum calcification markers and contralateral carotid plaques echogenicity after combined carotid artery stenting plus intensive lipid-lowering therapy in patients with bilateral carotid stenosis. Eur. J. Vasc. Endovasc. Surg. 2010, 39, 258–265. [Google Scholar] [CrossRef] [PubMed]
- Kadoglou, N.P.; Moumtzouoglou, A.; Kapelouzou, A.; Gerasimidis, T.; Liapis, C.D. Aggressive lipid-lowering is more effective than moderate lipid-lowering treatment in carotid plaque stabilization. J. Vasc. Surg. 2010, 55, 114–121. [Google Scholar] [CrossRef]
- Della-Morte, D.; Moussa, I.; Elkind, M.S.; Sacco, R.L.; Rundek, T. The short-term effect of atorvastatin on carotid plaque morphology assessed by computer-assisted gray-scale densitometry: A pilot study. Neurol. Res. 2011, 33, 991–994. [Google Scholar] [CrossRef] [PubMed]
- Nohara, R.; Daida, H.; Hata, M.; Kaku, K.; Kawamori, R.; Kishimoto, J.; Kurabayashi, M.; Masuda, I.; Sakuma, I.; Yamazaki, T.; et al. Effect of long-term intensive lipid-lowering therapy with rosuvastatin on progression of carotid intima-media thickness—Justification for atherosclerosis regression treatment (JART) extension study. Circ. J. 2013, 77, 1526–1533. [Google Scholar] [CrossRef] [PubMed]
- Jashari, F.; Ibrahimi, P.; Nicoll, R.; Bajraktari, G.; Wester, P.; Henein, M.Y. Coronary and carotid atherosclerosis: Similarities and differences. Atherosclerosis 2013, 227, 193–200. [Google Scholar] [CrossRef] [PubMed]
- Nicolaides, A.N.; Kyriacou, E.; Griffin, M.; Sabetai, M.; Thomas, D.J.; Tegos, T.; Geroulakos, G.; Labropoulos, N.; Doré, C.J.; Morris, T.P.; et al. Asymptomatic internal carotid artery stenosis and cerebrovascular risk stratification. J. Vasc. Surg. 2010, 52, 1486–1496. [Google Scholar] [CrossRef] [PubMed]
- Ibrahimi, P.; Jashari, F.; Johansson, E.; Gronlund, C.; Bajraktari, G.; Wester, P.; Henein, M.Y. Vulnerable plaques in the contralateral carotid arteries in symptomatic patients: A detailed ultrasound analysis. Atherosclerosis 2014, 235, 526–531. [Google Scholar] [CrossRef] [PubMed]
- Sztajzel, R.; Momjian-Mayor, I.; Comelli, M.; Momjian, S. Correlation of cerebrovascular symptoms and microembolic signals with the stratified gray-scale median analysis and color mapping of the carotid plaque. Stroke J. Cereb. Circ. 2006, 37, 824–829. [Google Scholar] [CrossRef]
- Aburahma, A.F.; Thiele, S.P.; Wulu, J.T., Jr. Prospective controlled study of the natural history of asymptomatic 60% to 69% carotid stenosis according to ultrasonic plaque morphology. J. Vasc. Surg. 2002, 36, 437–442. [Google Scholar] [CrossRef] [PubMed]
- Takemoto, M.; Liao, J.K. Pleiotropic effects of 3-hydroxy-3-methylglutaryl coenzyme a reductase inhibitors. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 1712–1719. [Google Scholar] [CrossRef] [PubMed]
- Schmermund, A.A.S.; Budde, T.; Buziashvili, Y.; Förster, A.; Friedrich, G.; Henein, M.; Kerkhoff, G.; Knollmann, F.; Kukharchuk, V.; Lahiri, A.; et al. Effect of intensive versus standard lipid-lowering treatment with atorvastatin on the progression of calcified coronary atherosclerosis over 12 months: A multicenter, randomized, double-blind trial. Circulation 2006, 113, 427–437. [Google Scholar] [CrossRef] [PubMed]
- Henein, M.; Granåsen, G.; Wiklund, U.; Schmermund, A.; Guerci, A.; Erbel, R.; Raggi, P. High dose and long-term statin therapy accelerate coronary artery calcification. Int. J. Cardiol. 2015, 184, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Coll, B.; Feinstein, S.B. Carotid intima-media thickness measurements: Techniques and clinical relevance. Curr. Atheroscler. Rep. 2008, 10, 444–450. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.A.; Swanson, A.C.; Gandhi, P.J.; Tataronis, G.R. Statin-related adverse events: A meta-analysis. Clin. Ther. 2006, 28, 26–35. [Google Scholar] [CrossRef] [PubMed]
- Jukema, J.W.; Cannon, C.P.; de Craen, A.J.; Westendorp, R.G.; Trompet, S. The controversies of statin therapy: Weighing the evidence. J. Am. Coll. Cardiol. 2012, 60, 875–881. [Google Scholar] [CrossRef] [PubMed]
- Sattar, N.; Preiss, D.; Murray, H.M.; Welsh, P.; Buckley, B.M.; de Craen, A.J.; Seshasai, S.R.; McMurray, J.J.; Freeman, D.J.; Jukema, J.W.; et al. Statins and risk of incident diabetes: A collaborative meta-analysis of randomised statin trials. Lancet 2010, 375, 735–742. [Google Scholar] [CrossRef] [PubMed]
- Preiss, D.; Seshasai, S.R.; Welsh, P.; Murphy, S.A.; Ho, J.E.; Waters, D.D.; DeMicco, D.A.; Barter, P.; Cannon, C.P.; Sabatine, M.S.; et al. Risk of incident diabetes with intensive-dose compared with moderate-dose statin therapy: A meta-analysis. JAMA 2011, 305, 2556–2564. [Google Scholar] [CrossRef] [PubMed]
- Liberati, A.A.D.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. PLoS Med. 2009, 6, 1–28. [Google Scholar] [CrossRef]
- Slim, K.; Nini, E.; Forestier, D.; Kwiatkowski, F.; Panis, Y.; Chipponi, J. Methodological index for non-randomized studies (MINORS): Development and validation of a new instrument. ANZ J. Surg. 2003, 73, 712–716. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ibrahimi, P.; Jashari, F.; Bajraktari, G.; Wester, P.; Henein, M.Y. Ultrasound Assessment of Carotid Plaque Echogenicity Response to Statin Therapy: A Systematic Review and Meta-Analysis. Int. J. Mol. Sci. 2015, 16, 10734-10747. https://doi.org/10.3390/ijms160510734
Ibrahimi P, Jashari F, Bajraktari G, Wester P, Henein MY. Ultrasound Assessment of Carotid Plaque Echogenicity Response to Statin Therapy: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences. 2015; 16(5):10734-10747. https://doi.org/10.3390/ijms160510734
Chicago/Turabian StyleIbrahimi, Pranvera, Fisnik Jashari, Gani Bajraktari, Per Wester, and Michael Y. Henein. 2015. "Ultrasound Assessment of Carotid Plaque Echogenicity Response to Statin Therapy: A Systematic Review and Meta-Analysis" International Journal of Molecular Sciences 16, no. 5: 10734-10747. https://doi.org/10.3390/ijms160510734
APA StyleIbrahimi, P., Jashari, F., Bajraktari, G., Wester, P., & Henein, M. Y. (2015). Ultrasound Assessment of Carotid Plaque Echogenicity Response to Statin Therapy: A Systematic Review and Meta-Analysis. International Journal of Molecular Sciences, 16(5), 10734-10747. https://doi.org/10.3390/ijms160510734







