Biological Mechanisms Underlying the Ultraviolet Radiation-Induced Formation of Skin Wrinkling and Sagging II: Over-Expression of Neprilysin Plays an Essential Role
Abstract
:1. Introduction
2. Characterization and Identification of Skin Fibroblast Elastase

3. Biological Mechanism Involved in the UVB-Induced Up-Regulation of Neprilysin Expression in Human Skin Fibroblasts
4. Effect of Direct Exposure of Human Dermal Fibroblasts to UVB on Neprilysin Protein and Activity
5. Biological Mechanisms Underlying the Up-Regulation of Neprilysin in Dermal Fibroblasts by UVB-Exposed Epidermal Keratinocytes
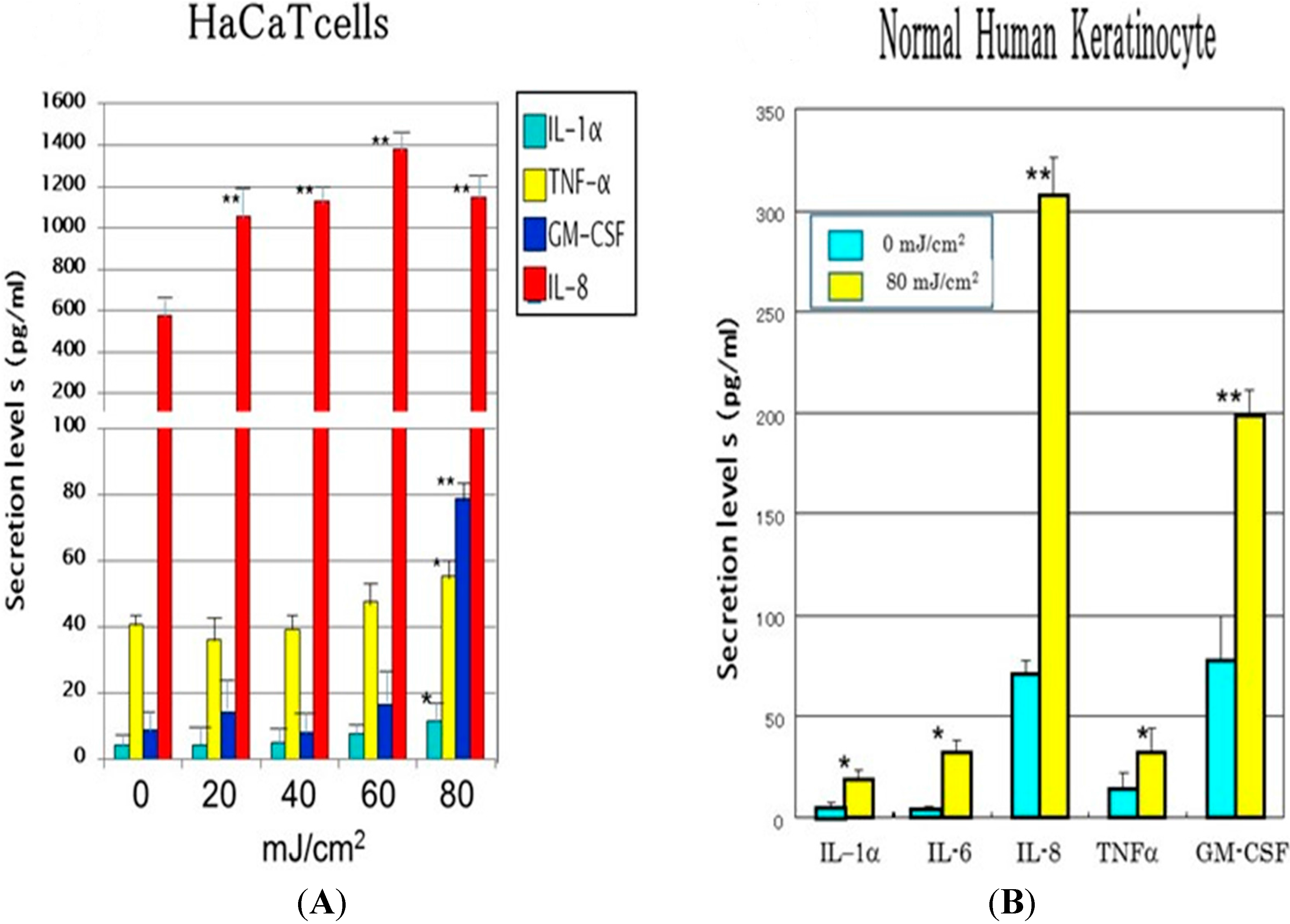


6. Identification of Paracrine Cytokines Responsible for UVB-Induced Up-Regulation of Neprilysin Expression in Human Fibroblasts
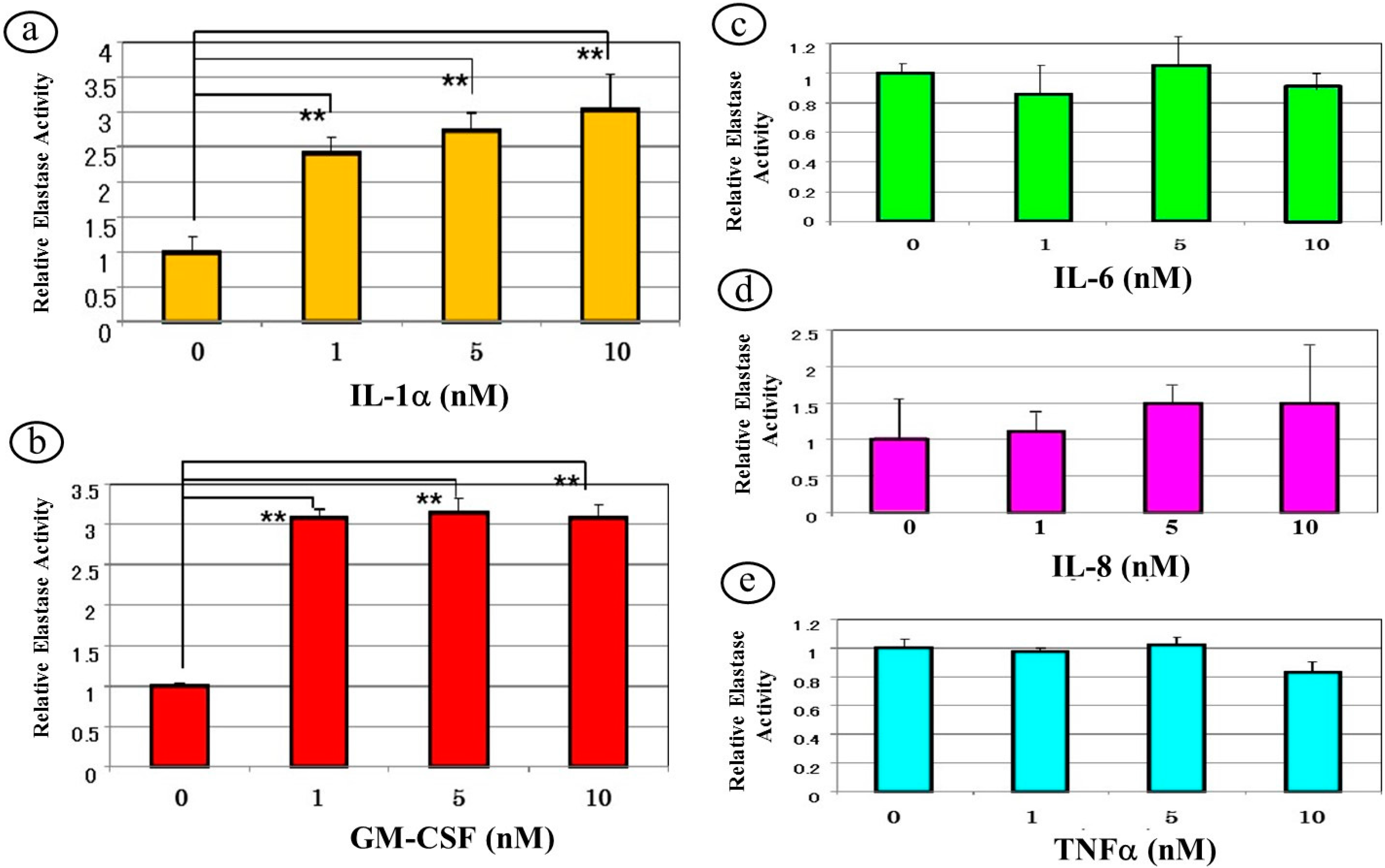
7. A Neutralizing Antibody Abrogates the Stimulated Neprilysin Activity
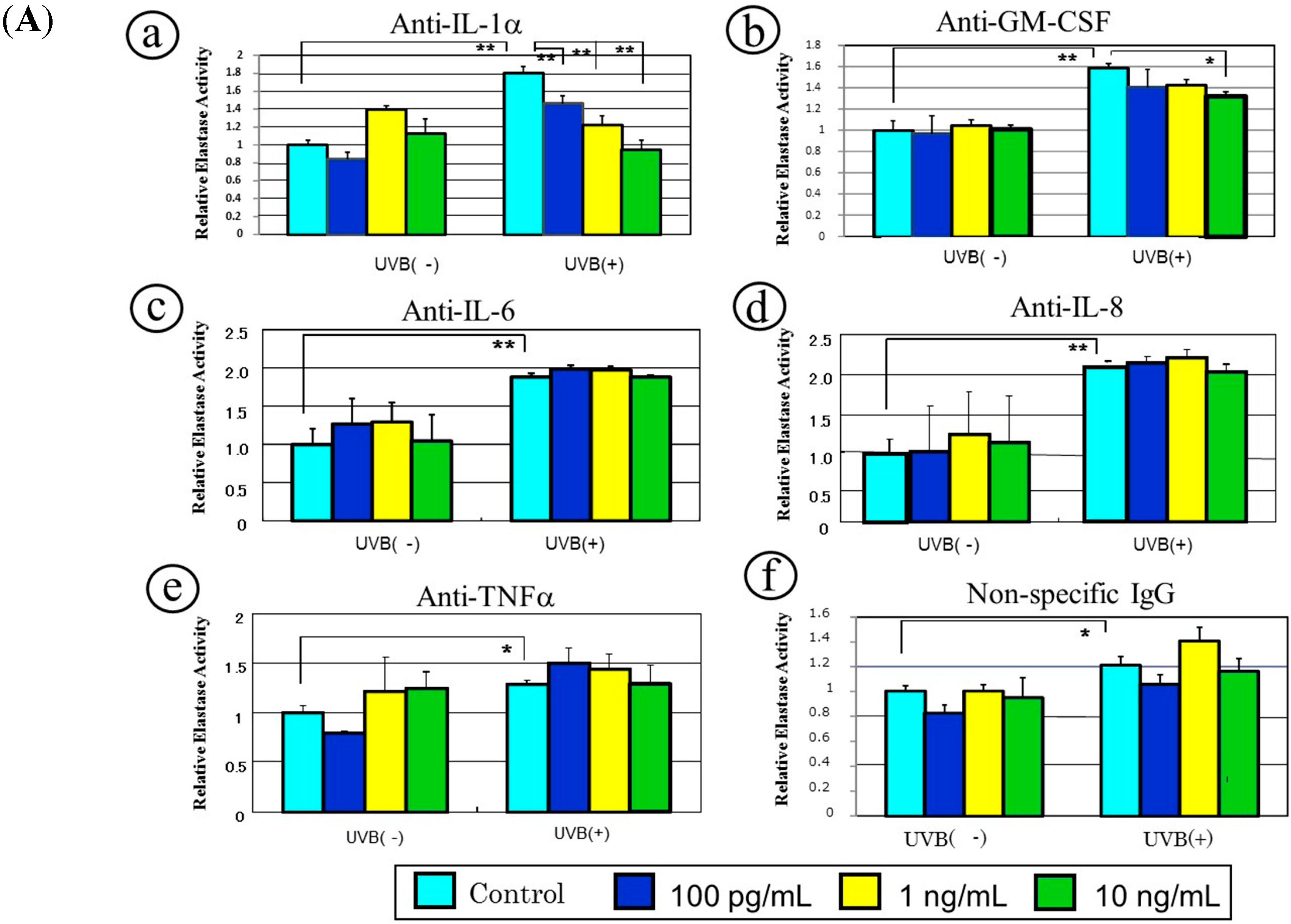
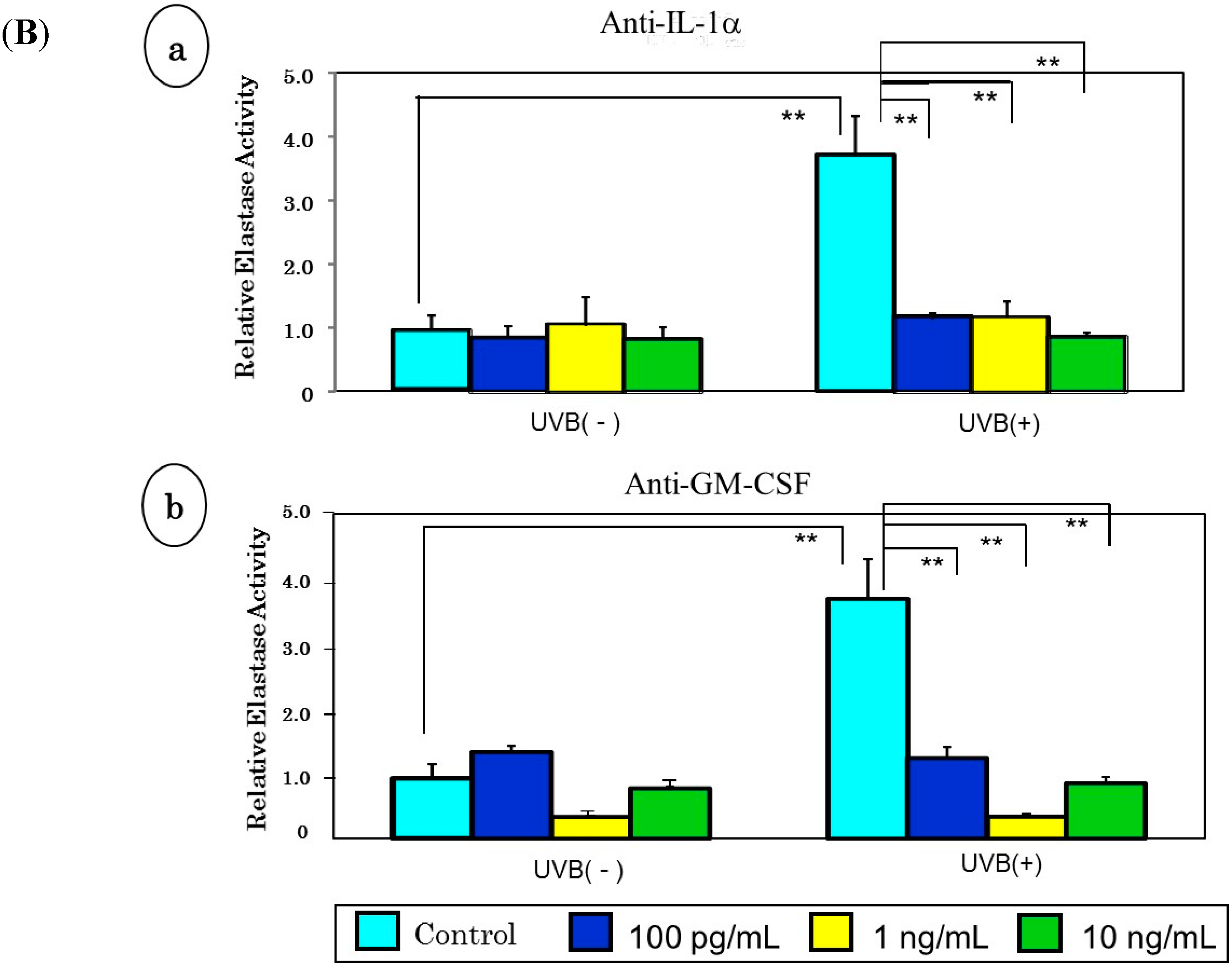
8. A Neutralizing Antibody Abrogates the Stimulated Neprilysin Protein
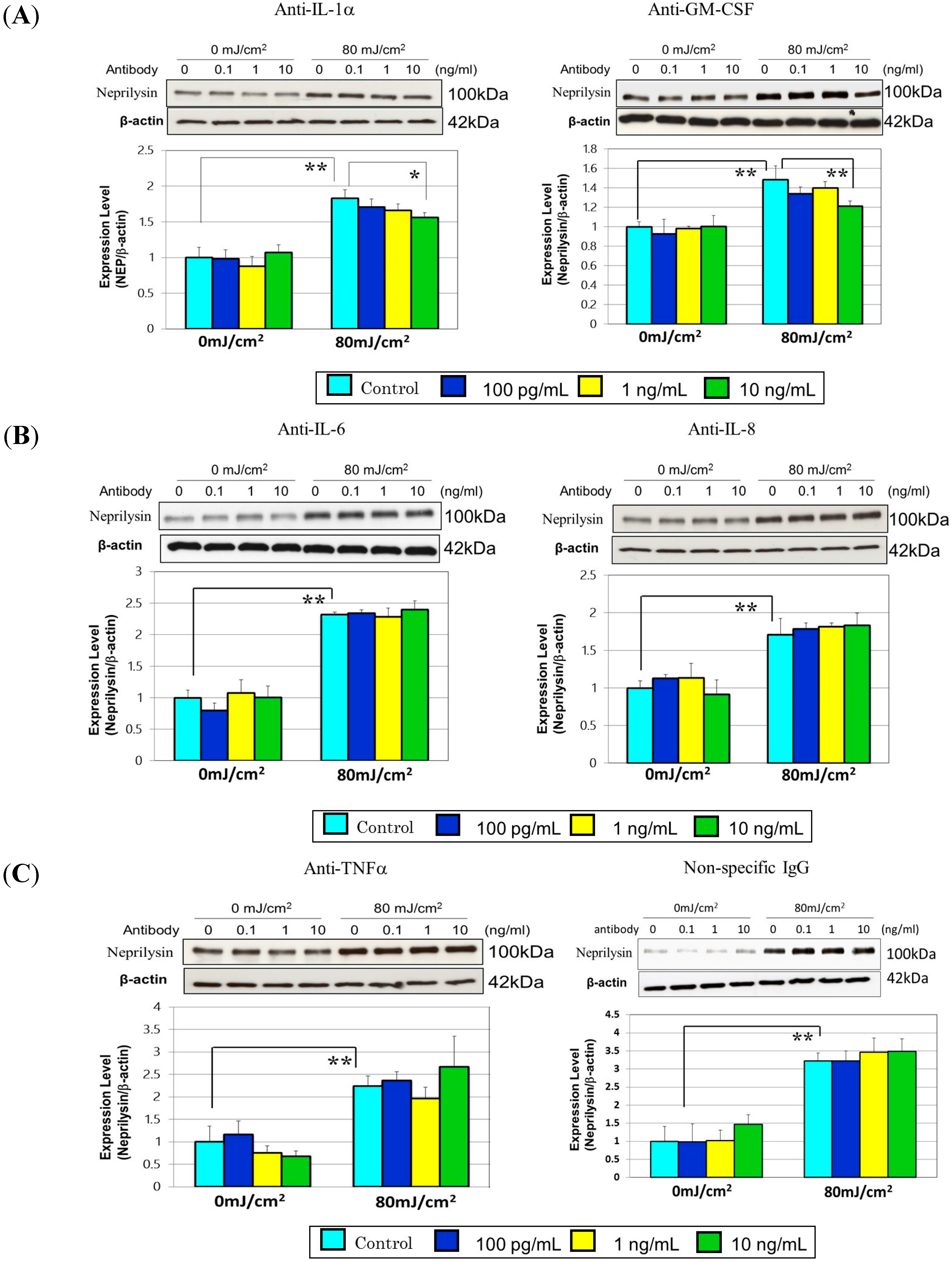
9. Effects of Cytokines on the Expression of Neprilysin in Human Fibroblasts at the Gene, Protein and Enzymatic Levels
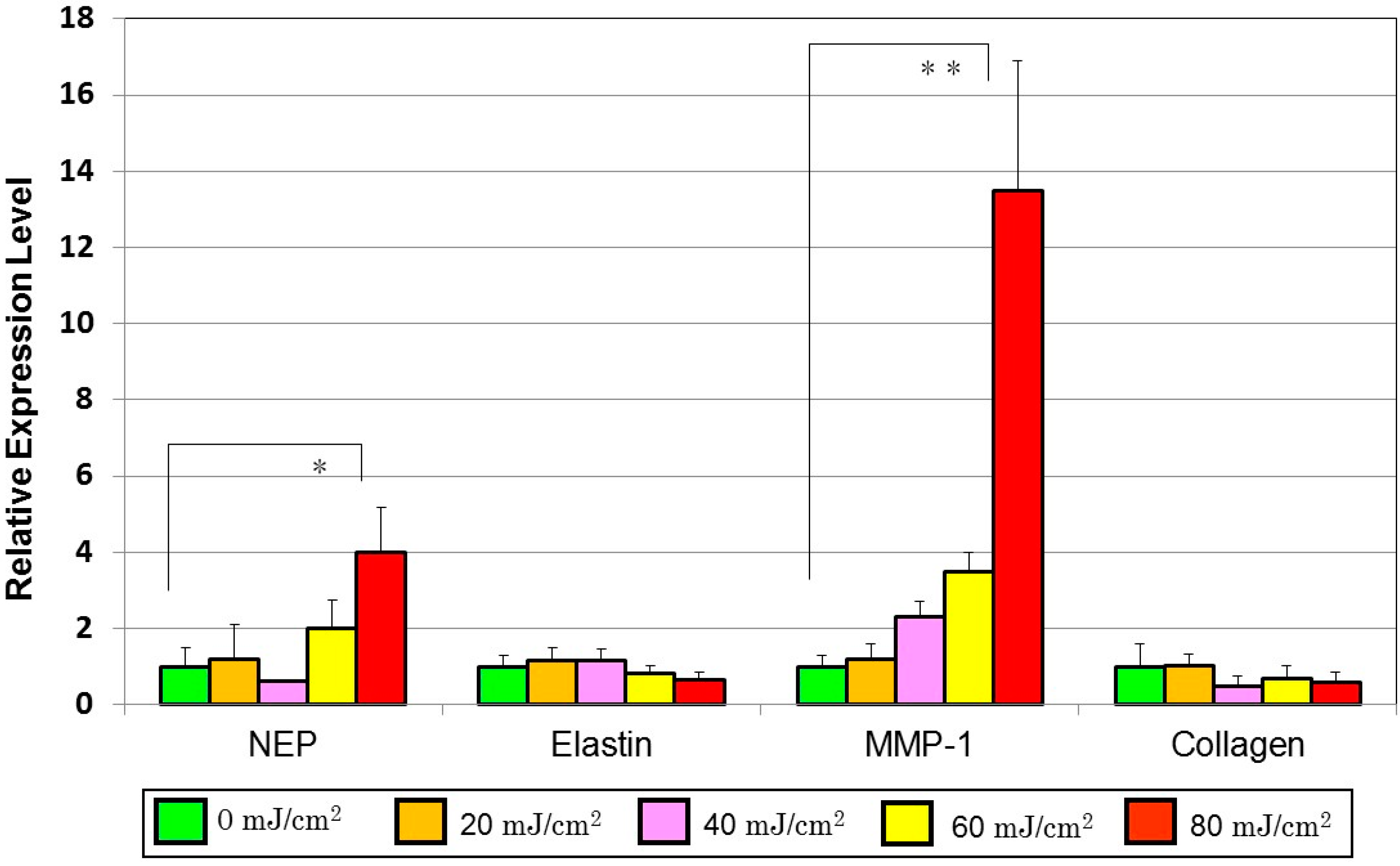
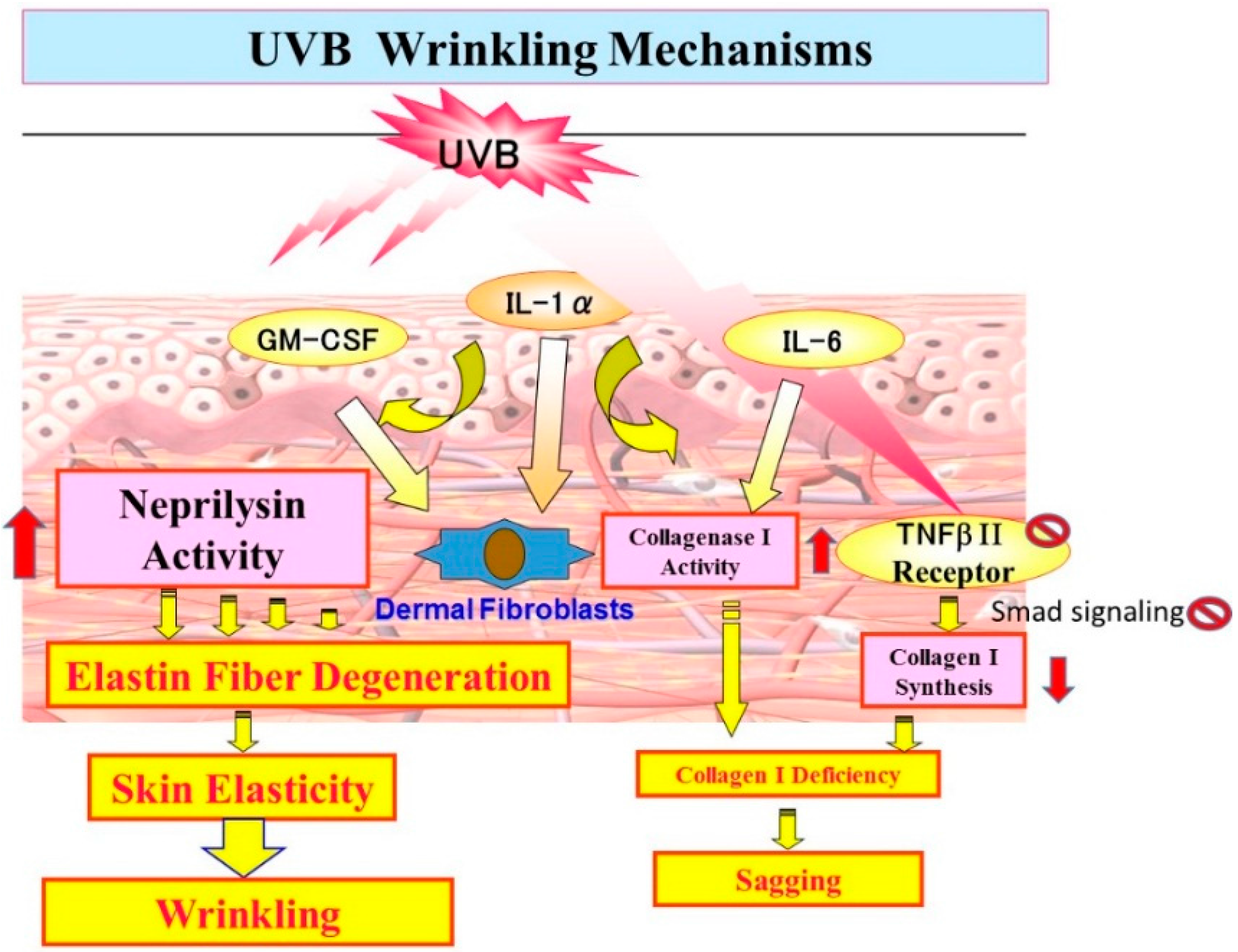
10. Mechanistic Comparison of Neprilysin/Elastin Linkage with MMP-1/TbetaRII/Collagen I Linkage Leading to UVB Radiation-Induced Wrinkling and Sagging
11. Mechanistic Study of UVA-Induced Formation of Wrinkling and Sagging
Effects of UVA Irradiation on Cytokine Secretion by Keratinocytes or Fibroblasts and on the Expression of Metalloproteinases by Fibroblasts
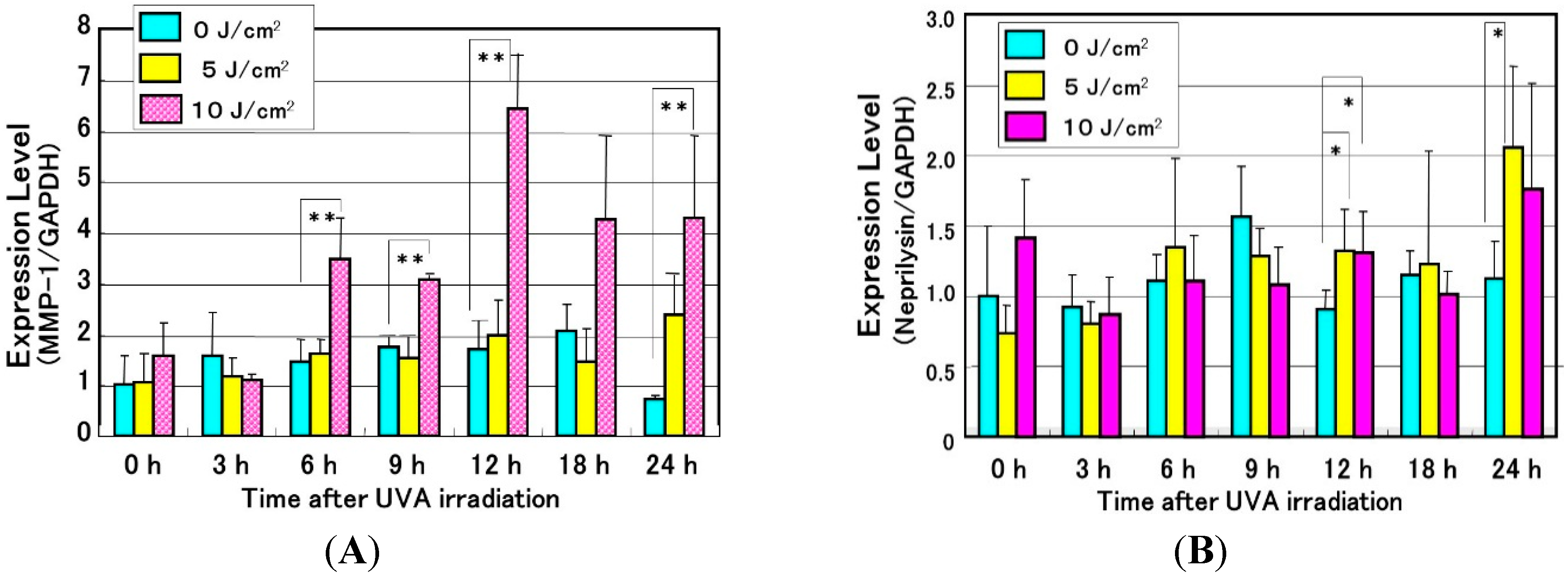
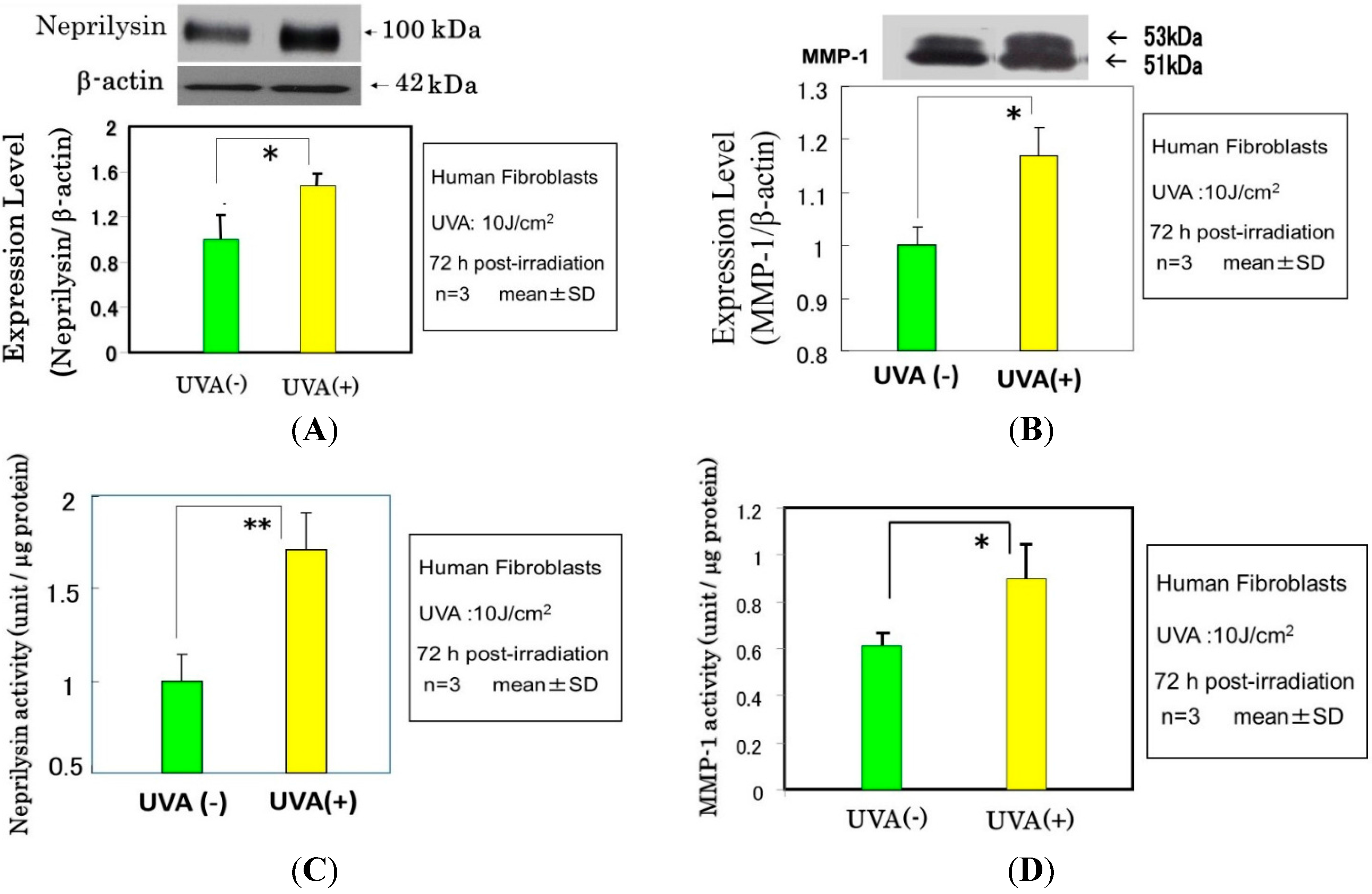
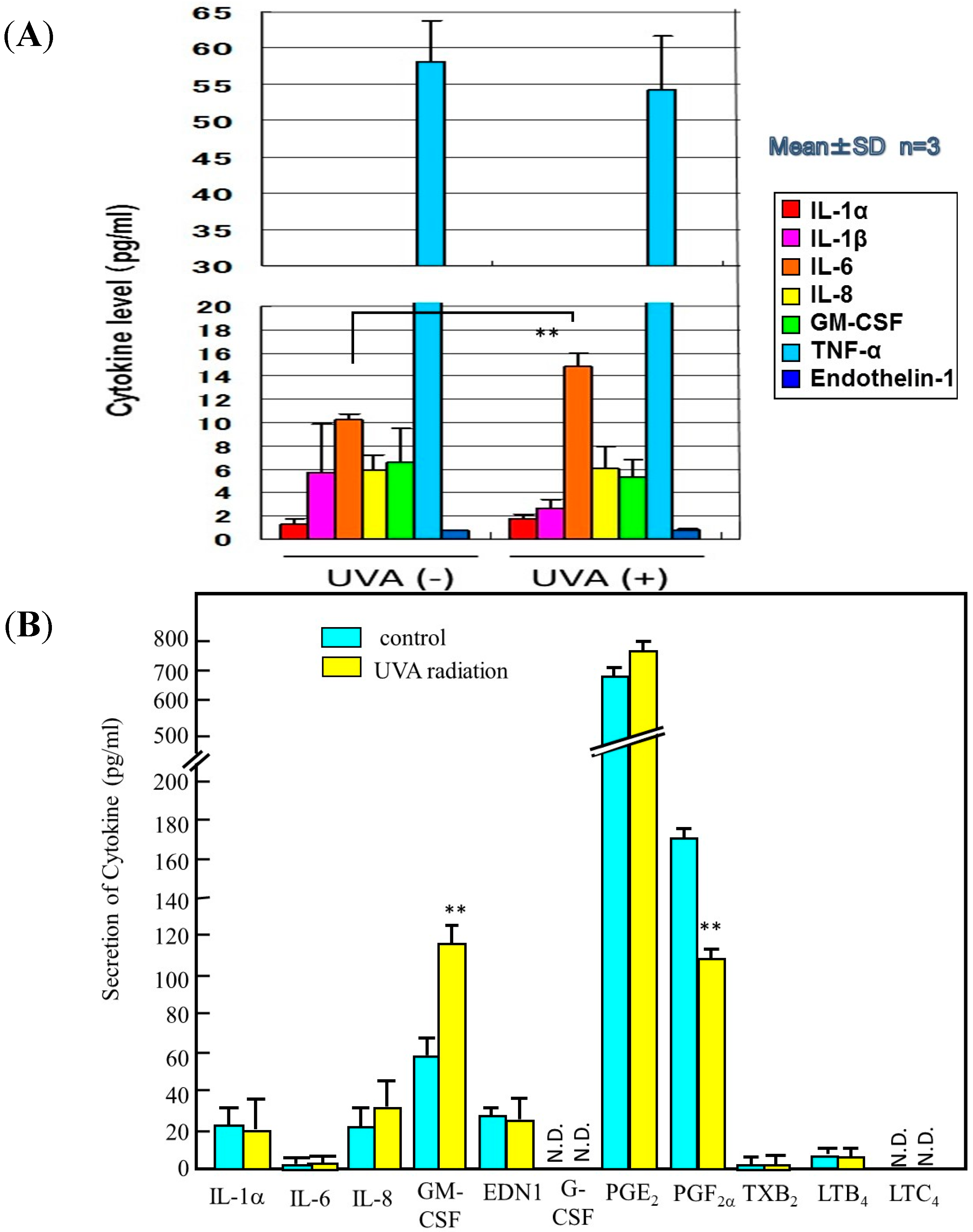
12. Conclusions
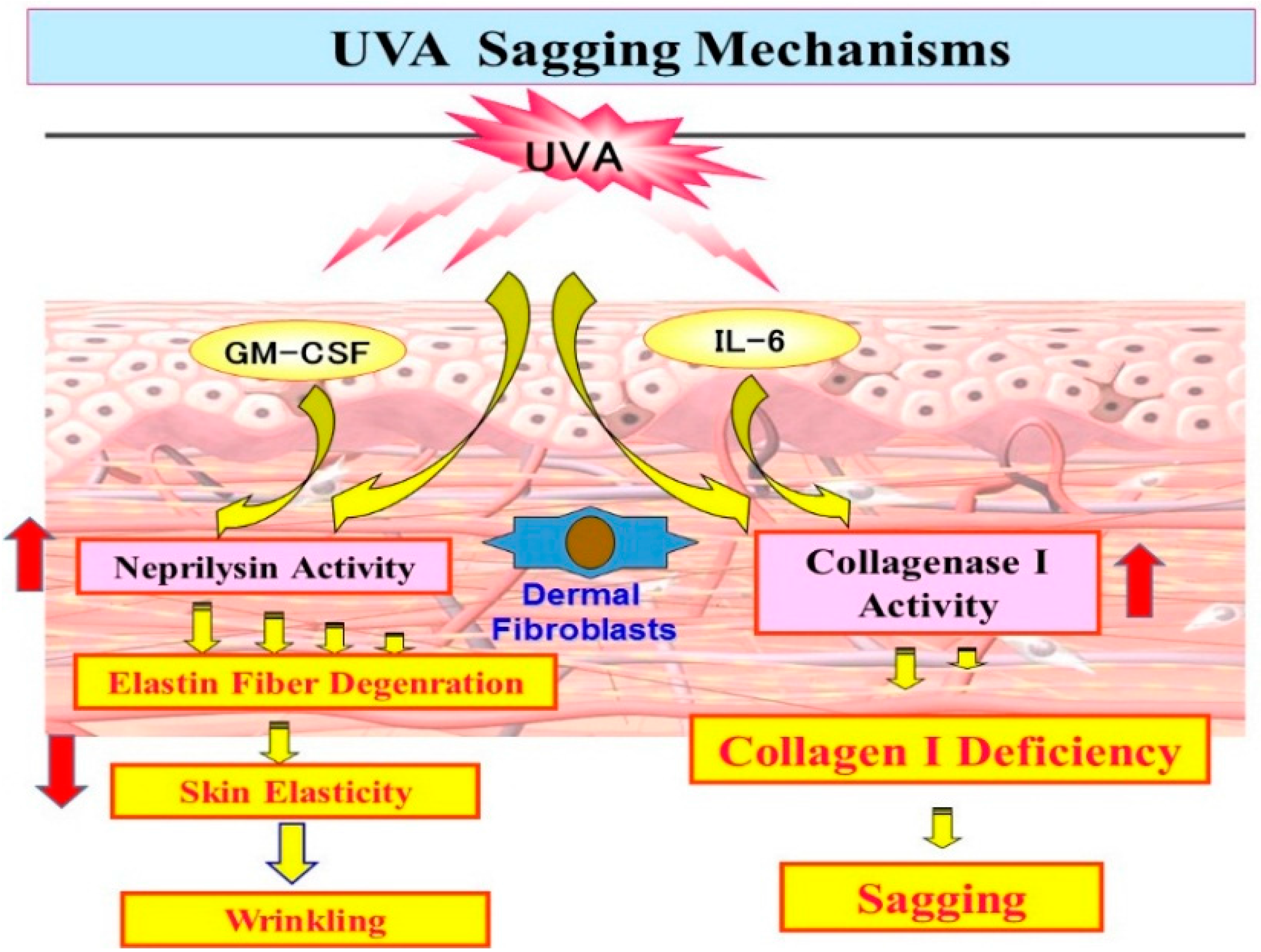
13. Perspectives
- A system to determine the inhibitory effects of several candidates on IL-1α or GM-CSF-stimulated elastase activity on human fibroblasts.
- A system to determine the inhibitory effects of several candidates on enhanced elastase activity by the conditioned medium from UVB-exposed human keratinocytes on human fibroblasts.
- A system to determine the inhibitory effects of several candidates on enhanced elastase activity in human fibroblasts co-cultured with UVB-exposed human keratinocytes.
Acknowledgments
Author Contributions
Abbreviations
| UVB | ultraviolet B |
| UVA | ultraviolet A |
| NEP | neutral endopeptidase |
| IL | interleukin |
| GM-CSF | granulocyte macrophage colony stimulatory factor |
| TNF | tumor necrosis factor |
| MMP-1 | matrix metallo-protease-1 |
Conflicts of Interest
References
- Szendri, M.; Meimon, G.; Bakala, H.; Frances, C.; Robert, L.; Godeau, G.; Hornebeck, W. On the presence of a metalloproteinase in human skin fibroblasts that degrades the human skin elastic fiber system. J. Investig. Dermatol. 1984, 83, 224–229. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, D.E.; Paller, A.S.; Lizak, P.P.; Person, R.W. Elastase and neutral cathepsin production by human fibroblasts: Effect of culture conditions on synthesis and secretion. J. Investig. Dermatol. 1986, 86, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, E.; Cruickshank, F.A.; Lebwohl, M.G. Elastase-like protease and elastolytic activities expressed in cultured dermal fibroblasts derived from lesional skin of patients with pseudoxanthoma elasticum, actinic elastosis, and cutis laxa. Clin. Chim. Acta 1988, 176, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, K.; Tsuji, T.; Kadoya, A.; Hamada, T. Elastase-like enzyme activity in cultured human fibroblast. Skin Res. 1987, 29, 793–797. [Google Scholar]
- Godeau, G.; Hornebeck, W. Morphometric analysis of the degradation of human skin elastic fibers by human leukocyte elastase (EC 3-4-21-37) and human skin fibroblast elastase (EC 3-4-24). Pathol. Biol. 1988, 36, 1133–1138. [Google Scholar] [PubMed]
- Homsy, R.; Pelletier-Lebon, P.; Tixier, J.M.; Godeau, G.; Robert, L.; Hornebeck, W. Characterization of human skin fibroblast elastase activity. J. Investig. Dermatol. 1988, 91, 472–477. [Google Scholar] [CrossRef] [PubMed]
- Croute, F.; Delaporte, E.; Bonnefoy, J.Y.; Fertin, C.; Thivolet, J.; Nicolas, J.F. Interleukin-1β stimulates fibroblast elastase activity. Br. J. Dermatol. 1991, 124, 538–541. [Google Scholar] [CrossRef] [PubMed]
- Mecham, R.P.; Broekelmann, T.J.; Fliszar, C.J.; Shapiro, S.D.; Welgus, H.G.; Senior, R.M. Elastin degradation by matrix metalloproteinases. J. Biol. Chem. 1997, 272, 18071–18076. [Google Scholar] [CrossRef] [PubMed]
- Senior, R.M.; Griffin, G.L.; Fliszar, C.J.; Shapiro, S.D.; Goldberg, G.I.; Wegus, H.G. Human 92- and 72-kilodalton type IV collagenase are elastase. J. Biol. Chem. 1991, 266, 7870–7875. [Google Scholar] [PubMed]
- Shipley, J.M.; Doyle, G.A.R.; Fliszar, C.J.; Ye, Q.-.Z.; Johnson, L.L.; Shapiro, S.D.; Welgus, H.G.; Senior, R.M. The structural basis for the elastolytic activity of the 92-kDa and 72-kDa gelatinases: Role of the fibronectin type II-like repeats. J. Biol. Chem. 1996, 271, 4335–4341. [Google Scholar] [CrossRef] [PubMed]
- Cambell, E.J.; Cambell, M.A. Pericellular proteolysis by neutrophils in the presence of proteinase inhibitors: Effects of substrate opsonization. J. Cell Biol. 1988, 106, 667–676. [Google Scholar] [CrossRef] [PubMed]
- Boudier, C.; Godeau, G.; Hornebeck, W.; Robert, L.; Bieth, J.G. The elastolytic activity of cathepsin G: An ex vivo study with dermal elastin. Am. J. Respir. Cell Mol. Biol. 1991, 4, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Kao, R.C.; Wehner, N.G.; Skubitz, K.M.; Gray, B.H.; Hoidal, J.R. Proteinase 3. A distinct human polymorphnuclear leukocyte proteinase that produces emphysema in hamsters. J. Clin. Investig. 1988, 82, 1963–1973. [Google Scholar] [CrossRef] [PubMed]
- Fulcher, I.S.; Matsas, R.; Turner, A.J.; Kenny, A.J. Kidney neutral endopeptidase and the hydrolysis of enkephalin by synaptic membranes show similar sensitivity to inhibitors. Biochem. J. 1982, 203, 519–522. [Google Scholar] [PubMed]
- Kerr, M.A.; Kenny, A.J. The purification and specificity of a neutral endopeptidase from rabbit kidney brush border. Biochem. J. 1974, 137, 477–488. [Google Scholar] [PubMed]
- Lorkowski, G.; Zijderhand-Bleekemolen, J.E.; Erdös, E.G.; von Figura, K.; Hasilik, A. Neutral endopeptidase-24.11 (enkephalinase). Biosynthesis and localization in human fibroblasts. Biochem. J. 1987, 248, 345–350. [Google Scholar] [PubMed]
- Oka, T.; Hiranuma, T.; Liu, X.F.; Ohgiya, N.; Iwao, K.; Matsumiya, T. Enkephalin-inactivating enzyme. Nippon Yakurigaku Zasshi 1993, 101, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Saria, A.; Hauser, K.F.; Traurig, H.H.; Turbek, C.S.; Hersh, L.; Gerard, C. Opioid-related changes in nociceptive threshold and in tissue levels of enkephalins after target disruption of the gene for neutral endopeptidase (EC 3.4.24.11) in mice. Neurosci. Lett. 1997, 234, 27–30. [Google Scholar] [CrossRef]
- Solan, N.J.; Ward, P.E.; Sanders, S.P.; Towns, M.C.; Bathon, J.M. Soluble recombinant neutral endopeptidase (CD10) as a potential antiinflammatory agent. Inflammation 1998, 22, 107–121. [Google Scholar] [CrossRef] [PubMed]
- Cohen, A.J.; Bunn, P.A.; Franklin, W.; Magill-Solc, C.; Hartmann, C.; Helfrich, B.; Gilman, L.; Folkvord, J.; Helm, K.; Miller, Y.E. Neutral endopeptidase: Variable expression in human lung, inactivation in lung cancer, and modulation of peptide- induced calcium flux. Cancer Res. 1996, 56, 831–839. [Google Scholar] [PubMed]
- Aguayo, S.M.; Schuyler, W.E.; Murtagh, J.J., Jr.; Roman, J. Regulation of lung branching morphogenesis by bombesin-like peptides and neutral endopeptidase. Am. J. Respir. Cell Mol. Biol. 1994, 10, 635–642. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, J.; Scicli, A.G.; Carretero, O.A.; Slaughter, C.; Moomaw, C.; Hersh, L.B. The hydrolysis of endothelins by neutral endopeptidase 24.11 (enkephalinase). J. Biol. Chem. 1990, 265, 14150–14155. [Google Scholar] [PubMed]
- Graf, K.; Schӓper, C.; Grӓfe, M.; Fleck, E.; Kunkel, G. Glucocorticoids and protein kinase C regulate neutral endopeptidase 24.11 in human vascular smooth muscle cells. Basic Res. Cardiol. 1998, 93, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Roques, B.P.; Noble, F.; Daugé, V.; Fournié-Zaluski, M.C.; Beaumont, A. Neutral endopeptidase 24.11: Structure, inhibition, and experimental and clinical pharmacology. Pharmacol. Rev. 1993, 45, 87–146. [Google Scholar] [PubMed]
- Krӓmer, H.H.; Schmidt, K.; Leis, S.; Schmelz, M.; Sommer, C.; Birklein, F. Inhibition of neutral endopeptidase (NEP) facilitates neurogenic inflammation. Exp. Neurol. 2005, 195, 179–184. [Google Scholar] [CrossRef] [PubMed]
- Roques, B.P.; Fournié-Zaluski, M.C.; Soroca, E.; Lecomte, J.M.; Malfroy, B.; Llorens, C.; Schwartz, J.C. The enkephalinase inhibitor thiorphan shows antinociceptive activity in mice. Nature 1980, 288, 286–288. [Google Scholar] [CrossRef] [PubMed]
- Mari, B.; Guerin, S.; Maulon, L.; Belhacene, N.; Farahi Far, D.; Imbert, V.; Rossi, B.; Peyron, J.F.; Auberger, P. Endopeptidase 24.11 (CD10/NEP) is required for phorbol ester-induced growth arrest in Jurkat T cells. FASEB J. 1997, 11, 869–879. [Google Scholar] [PubMed]
- Stefano, G.B.; Paemen, L.R.; Hughes, T.K., Jr. Autoimmunoregulation: Differential modulation of CD10/neutral endopeptidase 24.11 by tumor necrosis factor and neuropeptides. J. Neuroimmunol. 1992, 41, 9–14. [Google Scholar] [CrossRef] [PubMed]
- Letarte, M.; Vera, S.; Tran, R.; Addis, J.B.; Onizuka, R.J.; Quackenbush, E.J.; Jongeneel, C.V.; McInnes, R.R. Common acute lymphocytic leukemia antigen is identical to neutral endopeptidase. J. Exp. Med. 1988, 168, 1247–1253. [Google Scholar] [CrossRef] [PubMed]
- Miners, J.S.; Van Helmond, Z.; Chalmers, K.; Wilcock, G.; Love, S.; Kehoe, P.G. Decreased expression and activity of neprilysin in Alzheimer disease are associated with cerebral amyloid angiopathy. J. Neuropathol. Exp. Neurol. 2006, 65, 1012–1021. [Google Scholar] [CrossRef] [PubMed]
- Grimm, M.O.; Mett, J.; Stahlmann, C.P.; Haupenthal, V.J.; Zimmer, V.C.; Hartmann, T. Neprilysin and Aβ clearance: Impact of the APP intracellular domain in NEP regulation and implications in Alzheimer’s Disease. Front. Aging Neurosci. 2013, 5, 98. [Google Scholar] [CrossRef] [PubMed]
- Kakiya, N.; Saito, T.; Nilsson, P.; Matsuba, Y.; Tsubuki, S.; Takei, N.; Nawa, H.; Saido, T.C. Cell surface expression of the major amyloid-β peptide (Aβ)-degrading enzyme, neprilysin, depends on phosphorylation by mitogen-activated protein kinase/extracellular signal-regulated kinase kinase (MEK) and dephosphorylation by protein phosphatase 1a. J. Biol. Chem. 2012, 287, 29362–29372. [Google Scholar] [CrossRef] [PubMed]
- Morisaki, N.; Moriwaki, S.; Sugiyama-Nakagiri, Y.; Haketa, K.; Takema, Y.; Imokawa, G. Neprilysin is identical to skin fibroblast elastase—Its role in skin ageing and UV responses. J. Biol. Chem. 2010, 285, 39819–39827. [Google Scholar] [CrossRef] [PubMed]
- De Lombaert, S.; Erion, M.D.; Tan, J.; Blanchard, L.; el Chehabi, L.; Ghai, R.D.; Sakane, Y.; Berry, C.; Trapani, A.J. N-phosphonomethyl dipeptides and their phosphonate prodrugs, a new generation of neutral endopeptidase (NEP, EC 3.4.24.11) inhibitors. J. Med. Chem. 1994, 37, 498–511. [Google Scholar] [CrossRef] [PubMed]
- Suda, H.; Aoyagi, T.; Takeuchi, T.; Umezawa, H. Letter: A thermolysin inhibitor produced by Actinomycetes: Phosphoramidon. J. Antibiot. (Tokyo) 1973, 26, 621–623. [Google Scholar] [CrossRef]
- Oefner, C.; D’Arcy, A.; Hennig, M.; Winkler, F.K.; Dale, G.E. Structure of human neutral endopeptidase (Neprilysin) complexed with phosphoramidon. J. Mol. Biol. 2000, 296, 1–349. [Google Scholar] [CrossRef] [PubMed]
- Nadel, J.A. Neutral endopeptidase modulates neurogenic inflammation. Eur. Respir. J. 1991, 4, 745–754. [Google Scholar] [PubMed]
- Misery, L. [Neuro-immuno-cutaneous system (NICS)]. Pathol. Biol. 1996, 44, 867–874. [Google Scholar] [PubMed]
- Toyoda, M.; Nakamura, M.; Nakada, K.; Nakagawa, H.; Morohashi, M. Characteristic alterations of cutaneous neurogenic factors in photoaged skin. Br. J. Dermatol. 2005, 153, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, H.; Ezaki, Y.; Nagai, T.; Yoshioka, R.; Imokawa, G. Epithelial-mesenchymal interaction during UVB-induced up-regulation of neutral endopeptidase. Biochem. J. 2012, 443, 297–305. [Google Scholar] [CrossRef] [PubMed]
- Imokawa, G. Mechanism of UVB-induced wrinkling of the skin: Paracrine cytokine linkage between keratinocytes and fibroblasts leading to the stimulation of elastase. J. Investig. Dermatol. Symp. Proc. 2009, 14, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Tsukahara, K.; Nakagawa, H.; Moriwaki, S.; Shingo, K.; Ohuchi, A.; Takema, Y.; Imokawa, G. Ovariectomy is sufficient to accelerate spontaneous skin ageing and to stimulate ultraviolet irradiation-induced photoageing of murine skin. Br. J. Dermatol. 2004, 151, 984–994. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, N.; Moriwaki, S.; Suzuki, Y.; Takema, Y.; Imokawa, G. The role of elastases secreted by fibroblasts in wrinkle formation: Implication through selective inhibition of elastase activity. Photochem. Photobiol. 2001, 74, 283–290. [Google Scholar] [CrossRef] [PubMed]
- Brennan, M.1.; Bhatti, H.; Nerusu, K.C.; Bhagavathula, N.; Kang, S.; Fisher, G.J.; Varani, J.; Voorhees, J.J. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem. Photobiol. 2003, 78, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Kligman, L.H.; Kligman, A.M. The nature of photoaging: Its prevention and repair. Photodermatology 1986, 3, 215–227. [Google Scholar] [PubMed]
- Ma, W.; Wlaschek, M.; Tantcheva-Poór, I.; Schneider, L.A.; Naderi, L.; Razi-Wolf, Z.; Schüller, J.; Scharffetter-Kochanek, K. Chronological ageing and photoageing of the fibroblasts and the dermal connective tissue. Clin. Exp. Dermatol. 2001, 26, 592–599. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, G. Molecular mechanisms of skin ageing. Mech. Ageing Dev. 2002, 123, 801–810. [Google Scholar] [CrossRef] [PubMed]
- Rittié, L.; Fisher, G.J. UV-light-induced signal cascades and skin aging. Ageing Res. Rev. 2002, 1, 705–720. [Google Scholar] [CrossRef] [PubMed]
- Quan, T.; He, T.; Kang, S.; Voorhees, J.J.; Fisher, G.J. Solar ultraviolet irradiation reduces collagen in photoaged human skin by blocking transforming growth factor-β type II receptor/Smad signaling. Am. J. Pathol. 2004, 165, 741–751. [Google Scholar] [CrossRef] [PubMed]
- Fisher, G.J.; Wang, Z.Q.; Datta, S.C.; Varani, J.; Kang, S.; Voorhees, J.J. Pathophysiology of premature skin aging induced by ultraviolet light. N. Engl. J. Med. 1997, 337, 1419–1428. [Google Scholar] [CrossRef] [PubMed]
- Suganuma, K.; Nakajima, H.; Otsuki, M.; Imokawa, G. Astaxanthin attenuates the UVA-induced up-regulation of matrix-metalloproteinase-1 and skin fibroblast elastase in human dermal fibroblasts. J. Dermatol. Sci. 2010, 58, 136–142. [Google Scholar] [CrossRef] [PubMed]
- Imokawa, G.; Yada, Y.; Kimura, M.; Morisaki, N. Granulocyte/macrophage colony-stimulating factor is an intrinsic keratinocyte-derived growth factor for human melanocytes in UVA-induced melanosis. Biochem. J. 1996, 313, 625–631. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Imokawa, G.; Nakajima, H.; Ishida, K. Biological Mechanisms Underlying the Ultraviolet Radiation-Induced Formation of Skin Wrinkling and Sagging II: Over-Expression of Neprilysin Plays an Essential Role. Int. J. Mol. Sci. 2015, 16, 7776-7795. https://doi.org/10.3390/ijms16047776
Imokawa G, Nakajima H, Ishida K. Biological Mechanisms Underlying the Ultraviolet Radiation-Induced Formation of Skin Wrinkling and Sagging II: Over-Expression of Neprilysin Plays an Essential Role. International Journal of Molecular Sciences. 2015; 16(4):7776-7795. https://doi.org/10.3390/ijms16047776
Chicago/Turabian StyleImokawa, Genji, Hiroaki Nakajima, and Koichi Ishida. 2015. "Biological Mechanisms Underlying the Ultraviolet Radiation-Induced Formation of Skin Wrinkling and Sagging II: Over-Expression of Neprilysin Plays an Essential Role" International Journal of Molecular Sciences 16, no. 4: 7776-7795. https://doi.org/10.3390/ijms16047776
APA StyleImokawa, G., Nakajima, H., & Ishida, K. (2015). Biological Mechanisms Underlying the Ultraviolet Radiation-Induced Formation of Skin Wrinkling and Sagging II: Over-Expression of Neprilysin Plays an Essential Role. International Journal of Molecular Sciences, 16(4), 7776-7795. https://doi.org/10.3390/ijms16047776





