Genome-Wide Expression in Visceral Adipose Tissue from Obese Prepubertal Children
Abstract
:1. Introduction
2. Results and Discussion
2.1. Anthropometric and Biochemical Characteristics of the Study Population
| Microarray | qPCR | |||
|---|---|---|---|---|
| Characteristics | Normal-Weight (n = 6) | Obese (n = 5) | Normal-Weight (n = 10) | Obese (n = 10) |
| Sex (M:F) | 5:1 | 5:0 | 6:4 | 8:2 |
| Age (years) | 8.2 ± 0.6 | 10.2 ± 0.6 | 9.3 ± 0.8 | 9.5 ± 0.5 |
| Weight (kg) | 28.8 ± 1.1 | 55.8 ± 2.9 * | 32.9 ± 2.7 | 50.9 ± 3.6 * |
| Height (m) | 131.4 ± 3.4 | 141.0 ± 4.0 | 136.2 ± 4.2 | 138.9 ± 4.3 |
| BMI (kg/m2) | 16.8 ± 0.6 | 28.1 ± 0.7 * | 17.5 ± 0.5 | 26.1 ± 0.7 * |
| BMI z score | −0.61 ± 0.36 | 3.12 ± 0.5 * | −0.6 ± 0.2 | 2.8 ± 0.3 * |
| Systolic BP (mm·Hg) | 111.3 ± 3.20 | 123.0 ± 7.1 | 109.1 ± 2.6 | 119.9 ± 4.5 |
| Diastolic BP (mm·Hg) | 61.3 ± 3.0 | 71.2 ± 5.6 | 60.3 ± 3.2 | 67.8 ± 3.9 |
| Glucose (mmol/L) | 4.73 ± 0.30 | 4.95 ± 0.19 | 4.9 ± 0.2 | 5.1 ± 0.3 |
| Insulin (pmol/L) | 9.0 ± 1.6 | 19.3 ± 7.2 | 10.4 ± 1.6 | 11.1 ± 1.4 |
| HOMA-IR | 1.8 ± 0.32 | 4.3 ± 1.7 | 2.3 ± 0.33 | 2.4 ± 0.31 |
| TAG (mmol/L) | 0.77 ± 0.19 | 0.67 ± 0.09 | 0.88 ± 0.18 | 0.80 ± 0.09 |
| TC (mmol/L) | 4.19 ± 3.44 | 4.21 ± 0.57 | 3.86 ± 0.25 | 4.22 ± 0.42 |
| HDL-C (mmol/L) | 1.54 ± 0.09 | 1.78 ± 0.23 | 1.42 ± 0.15 | 1.40 ± 0.11 |
| LDL-C (mmol/L) | 2.29 ± 0.26 | 2.11 ± 0.43 | 2.02 ± 0.21 | 2.44 ± 0.34 |
| Apo A-1 (g/L) | 0.12 ± 0.08 | 1.44 ± 0.05 | 1.23 ± 0.10 | 1.19 ± 0.09 |
| Apo B (g/L) | 0.74 ± 0.09 | 0.61 ± 0.10 | 0.67 ± 0.07 | 0.73 ± 0.09 |
| Adiponectin (mg/L) | 14.8 ± 0.3 | 5.9 ± 3.6 | 16.7 ± 1.9 | 11.8 ± 1.9 |
| Leptin (μg/L) | 27.9 ± 2.3 | 33.1 ± 4.8 | 27.3 ± 1.7 | 34.8 ± 3.4 |
| CRP (g/L) | 0.29 ± 0.09 | 0.32 ± 0.14 | 0.29 ± 0.08 | 0.46 ± 0.09 |
2.2. Gene Expression Profile of Visceral Adipose Tissue in Obese Children
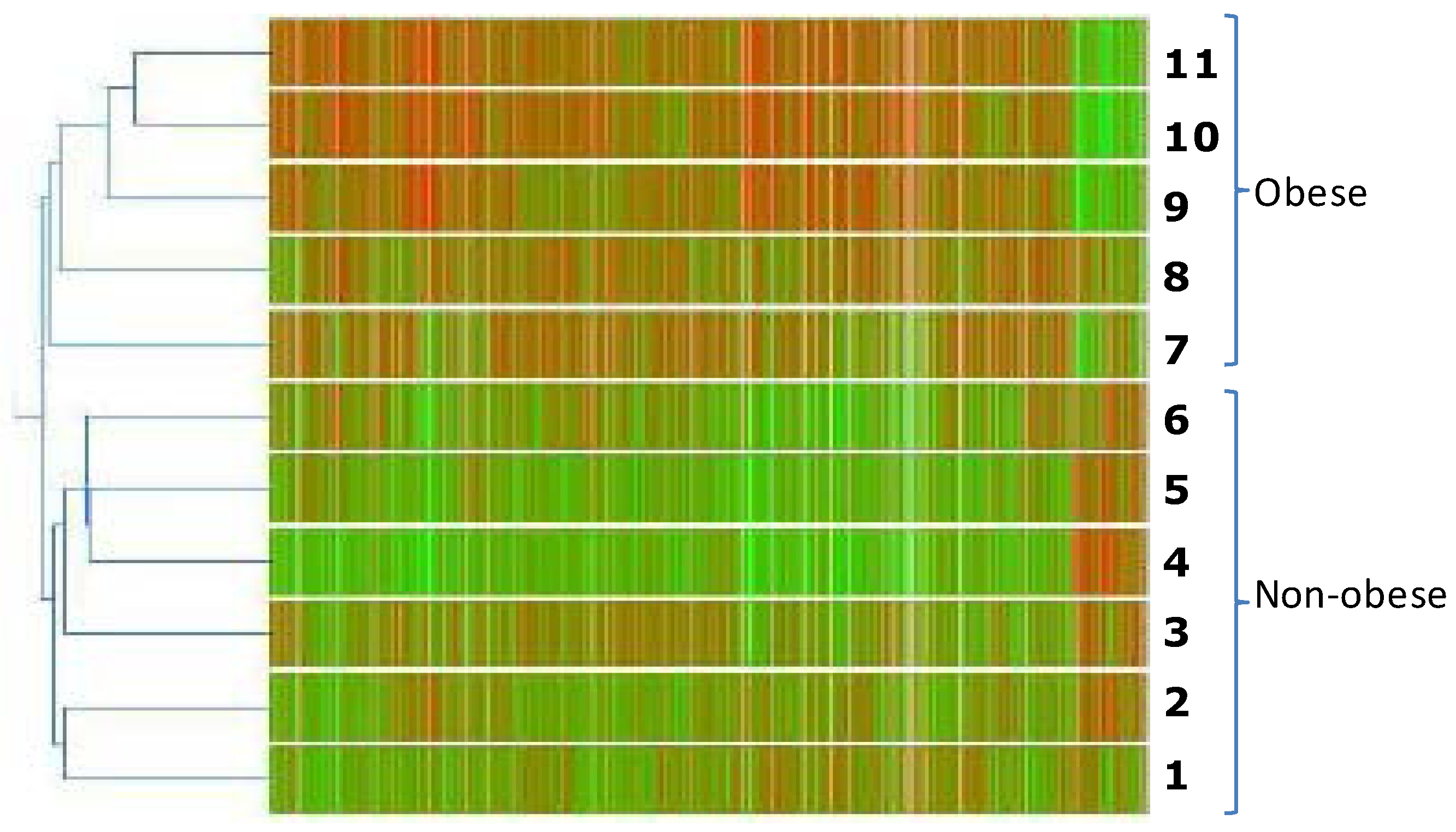
2.3. Validation of Gene Expression Data by qPCR
| Symbol | Name | Microarrays | qPCR | ||
|---|---|---|---|---|---|
| Fold Change | p Value | Fold Change | * p Value | ||
| CES1 | carboxylesterase 1 | 11.74 | 0.001 | 3.0 | 0.050 |
| TNMD | tenomodulin protein | 7.19 | 0.028 | 5.5 | 0.028 |
| LEP | leptin (obesity homolog, mouse) | 4.44 | 0.005 | 3.5 | 0.050 |
| NPR3 | natriuretic peptide receptor C | 4.31 | 0.005 | 5.1 | 0.006 |
| NQO1 | NAD(P)H dehydrogenase, quinone 1 | 3.99 | 0.006 | 2.8 | 0.041 |
| BHMT2 | betaine-homocysteine methyltransferase 2 | 3.14 | 0.005 | 2.3 | 0.049 |
| CRYAB | crystallin, alpha B | 2.25 | 0.005 | 2.6 | 0.034 |
| AFF1 | AF4/FMR2 family, member 1 | 2.20 | 0.004 | 2.9 | 0.041 |
| ANXA1 | annexin A1 | 2.14 | 0.037 | 1.9 | 0.049 |
| ITGB5 | integrin, beta 5 | 2.12 | 0.000 | 1.3 | 0.050 |
| CALCRL | calcitonin receptor-like | -2.64 | 0.006 | -2.4 | 0.034 |
2.3.1. Lipid and Amino Acid Metabolism
2.3.2. Oxidative Stress and Extracellular Matrix Related Genes
2.3.3. Adipogenesis Related Genes
2.3.4. Inflammation-Related Gene
3. Experimental Section
3.1. Study Population
3.2. Anthropometric and Biochemical Measurements
3.3. Adipose Tissue Biopsy and Total RNA Preparation
3.4. Microarray Analysis
3.5. qPCR Analysis
3.6. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- De Bruyne, R.M.; Fitzpatrick, E.; Dhawan, A. Fatty liver disease in children: Eat now pay later. Hepatol. Int. 2010, 4, 375–385. [Google Scholar]
- Alligier, M.; Meugnier, E.; Debard, C.; Lambert-Porcheron, S.; Chanseaume, E.; Sothier, M.; Loizon, E.; Hssain, A.A.; Brozek, J.; Scoazec, J.Y.; et al. Subcutaneous adipose tissue remodeling during the initial phase of weight gain induced by overfeeding in humans. J. Clin. Endocrinol. Metab. 2012, 97, E183–E192. [Google Scholar]
- Kershaw, EE; Flier, JS. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar]
- Tchernof, A.; Despres, J.P. Pathophysiology of human visceral obesity: An update. Physiol. Rev. 2013, 93, 359–404. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M. Subcutaneous and visceral adipose tissue: Structural and functional differences. Obes. Rev. 2010, 11, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Insenser, M.; Montes-Nieto, R.; Vilarrasa, N.; Lecube, A.; Simó, R.; Vendrell, J.; Escobar-Morreale, H.F. A nontargeted proteomic approach to the study of visceral and subcutaneous adipose tissue in human obesity. Mol. Cell. Endocrinol. 2012, 363, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Gil, A.; Olza, J.; Gil-Campos, M.; Gomez-Llorente, C.; Aguilera, C.M. Is adipose tissue metabolically different at different sites? Int. J. Pediatr. Obes. 2011, 6, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Gerhard, G.S.; Styer, A.M.; Strodel, W.E.; Roesch, S.L.; Yavorek, A.; Carey, D.J.; Wood, G.C.; Petrick, A.T.; Gabrielsen, J.; Ibele, A.; et al. Gene expression profiling in subcutaneous, visceral and epigastric adipose tissue of patients with extreme obesity. Int. J. Obes. 2014, 38, 371–378. [Google Scholar]
- Linder, K.; Arner, P.; Flores-Morales, A.; Tollet-Egnell, P.; Norstedt, G. Differentially expressed genes in visceral or subcutaneous adipose tissue of obese men and women. J. Lipid Res. 2004, 45, 148–154. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Nair, S.; Rousseau, E.; Allison, D.B.; Page, G.P.; Tatarami, P.A.; Bogardus, C.; Permana, P.A. Microarray profiling of isolated abdominal subcutaneous adipocytes from obese vs. non-obese Pima Indians: Increased expression of inflammation-related genes. Diabetologia 2005, 48, 1776–1783. [Google Scholar]
- Gomez-Ambrosi, J.; Catalán, V.; Diez-Caballero, A.; Martinez-Cruz, L.A.; Gil, M.J.; García-Foncillas, J.; Cienfuegos, J.A.; Salvador, J.; Mato, J.M.; Frühbeck, G. Gene expression profile of omental adipose tissue in human obesity. FASEB J. 2004, 18, 215–217. [Google Scholar] [PubMed]
- Baranova, A.; Collantes, R.; Gowder, S.J.; Elariny, H.; Schlauch, K.; Younoszai, A.; King, S.; Randhawa, M.; Pusulury, S.; Alsheddi, T.; et al. Obesity-related differential gene expression in the visceral adipose tissue. Obes. Surg. 2005, 15, 758–765. [Google Scholar]
- Tam, C.S.; Heilbronn, L.K.; Henegar, C.; Wong, M.; Cowell, C.T.; Cowley, M.J.; Kaplan, W.; Clément, K.; Baur, L.A. An early inflammatory gene profile in visceral adipose tissue in children. Int. J. Pediatr. Obes. 2011, 6, e360–e363. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, L.; de Zegher, F. Puberty and prenatal growth. Mol. Cell. Endocrinol. 2006, 255, 22–25. [Google Scholar] [CrossRef]
- Olza, J.; Aguilera, C.M.; Gil-Campos, M.; Leis, R.; Bueno, G.; Martínez-Jiménez, M.D.; Valle, M.; Cañete, R.; Tojo, R.; Moreno, L.A.; et al. Myeloperoxidase is an early biomarker of inflammation and cardiovascular risk in prepubertal obese children. Diabetes Care 2012, 35, 2373–2376. [Google Scholar]
- Olza, J.; Aguilera, C.M.; Gil-Campos, M.; Leis, R.; Bueno, G.; Valle, M.; Cañete, R.; Tojo, R.; Moreno, L.A.; Gil, A. Waist-to-height ratio, inflammation and CVD risk in obese children. Public Health Nutr. 2014, 2, 1–8. [Google Scholar]
- Lönngvist, F.; Arne, P.; Nordfors, L.; Schalling, M. Overexpression of the obese (ob) gene in adipose tissue of human obese subjects. Nat. Med. 1995, 1, 950–953. [Google Scholar] [CrossRef] [PubMed]
- Arner, P.; Langin, D. The role of neutral lipases in human adipose tissue lipolysis. Curr. Opin. Lipidol. 2007, 18, 246–250. [Google Scholar] [CrossRef] [PubMed]
- Blais, D.R.; Lyn, R.K.; Joyce, M.A.; Rouleau, Y.; Steenbergen, R. Activity based protein profiling identifies a host enzyme, carboxylesterase 1, which is differentially active during hepatitis C virus replication. J. Biol. Chem. 2010, 285, 25602–25612. [Google Scholar] [CrossRef] [PubMed]
- Jernas, M.; Olsson, B.; Arner, P.; Jacobson, P.; Sjöström, W.A; Froguel, P.; McTernan, P.G.; Hoffstedt, J.; Carlsson, L.M.S. Regulation of carboxylesterase 1 (CES1) in human adipose tissue. Biochem. Biophys. Res. Commun. 2009, 383, 63–67. [Google Scholar]
- Nagashima, S.; Yagyu, H.; Takahashi, N.; Kurashina, T.; Takahashi, M.; Tsuchita, T.; Tazoe, F.; Wang, X.L.; Bayasgalan, T.; Sato, N.; et al. Depot-specific expression of lipolytic genes in human adipose tissues: Association among CES1 expression, triglyceride lipase activity and adiposity. J. Atheroscler. Thromb. 2011, 18, 190–199. [Google Scholar]
- Friedrichsen, M.; Poulsen, P.; Wojtaszewski, J.; Hansen, P.R.; Vaag, A.; Rasmussen, H.B. Carboxylesterase 1 gene duplication and mRNA expression in adipose tissue are linked to obesity and metabolic function. PLoS ONE 2013, 8, e56861. [Google Scholar] [CrossRef] [PubMed]
- Lafontan, M.; Moro, C.; Berlan, M.; Crampes, F.; Sengenes, C.; Galitzky, J. Control of lipolysis by natriuretic peptides and cyclic GMP. Trends Endocrinol. Metab. 2008, 19, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Pivovarova, O.; Gögebakan, Ö.; Kloöting, N.; Sparwasser, A.; Weickert, M.O.; Haddad, I.; Nikiforova, V.J.; Bergmann, A.; Kruse, M.; Seltmann, A.C.; et al. Insulin up-regulates natriuretic peptide clearance receptor expression in the subcutaneous fat depot in obese sunjects: A missing link between CVD risk and obesity? J. Clin. Endocrinol. Metab. 2012, 97, E731–E739. [Google Scholar]
- Selleti, D.F.; Perrella, G.; Doi, S.Q.; Curcio, F. Natriuretic peptides increase cAMP production in human thyrocytes via the natriuretic peptide clearance (NPR-C). Regul. Pept. 2001, 2, 103–109. [Google Scholar] [CrossRef]
- Teng, Y.W.; Ellis, J.M.; Coleman, R.A.; Zeisel, S.H. Mouse betaine-homocysteine S-methyltransferase deficiency reduces body fat via increasing energy expenditure and impairing lipid synthesis and enhancing glucose oxidation in white adipose tissue. J. Biol. Chem. 2012, 287, 16187–16198. [Google Scholar] [CrossRef] [PubMed]
- Chun, T.H. Peri-adipocyte ECM remodeling in obesity and adipose tissue fibrosis. Adipocyte 2012, 1, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Henegar, C.; Tordjman, J.; Achard, V.; Lacasa, D.; Cremer, I.; Guerre-Millo, M.; Poitou, C.; Basdevant, A.; Stich, V.; Viguerrie, N.; et al. Adipose tissue transcriptomic signature highlights the pathological relevance of extracelular matrix in human obesity. Genome Biol. 2008, 21. [Google Scholar] [CrossRef]
- Kolehmainen, M.; Salopuro, T.; Schwab, U.S.; Kekalainen, J.; Kallio, P.; Laaksonen, D.E.; Pulkkinen, L.; Lindi, V.I.; Sivenius, K.; Mager, U. Weight reduction modulates expression of genes involved in extracellular matrix and cell death: The GENOBIN study. Int. J. Obes. 2008, 32, 292–303. [Google Scholar] [CrossRef]
- Saiki, A.; Olsson, M.; Jernas, M.; Gummesson, A.; Mcternan, PG.; Andersson, J.; Jacobson, P.; Sjöholm, K.; Olsson, P.; Yamamura, S.; et al. Tenomodulin is highly expressed in adipose tissue, increased in obesity, and down-regulated during diet-induced weight loss. J. Clin. Endocrinol. Metab. 2009, 94, 3987–3994. [Google Scholar]
- Palming, J.; Sjöholm, K.; Jernas, M.; Lustig, T.C.; Gummesson, A.; Romeo, S.; Lönn, L.; Lönn, M.; Carlsson, B.; Carlsson, L.M. The expression of NAD(P)H: Quinine oxidoreductase 1 is high in human adipose tissue, reduced by weight loss, and correlates with adipostity, insulin sensitivity, and markers of liver dysfunction. J. Clin. Endocrinol. Metab. 2007, 92, 2346–2352. [Google Scholar] [CrossRef] [PubMed]
- Lehr, S.; Hartwig, S.; Lamers, D.; Famulla, S.; Müller, S.; Hanisch, F.G.; Cuvelier, C.; Ruige, J.; Eckardt, K.; Ouwens, M.; et al. Identification and validation of novel adipokines released from primary human adipocytes. Mol. Cell. Proteomics 2012, 11, 1–13. [Google Scholar]
- Li, Y.; Jiang, C.; Wang, X.; Zhang, Y.; Shibahara, S.; Takahashi, K. Adrenomedullin is a novel adipokine: Adrenomedullin in adipocytes and adipose tissues. Peptides 2007, 28, 1129–1143. [Google Scholar]
- Harmancey, R.; Senard, J.M.; Rouet, P.; Pathak, A.; Smih, F. Adrenomedullin inhibits adipogenesis under transcriptional control of insulin. Diabetes 2007, 56, 553–563. [Google Scholar] [CrossRef] [PubMed]
- Waterworth, D.M.; Ricketts, S.L.; Song, K.; Chen, L.; Zhao, J.H.; Ripatti, S.; Aulchenko, Y.S.; Zhang, W.; Yuan, X.; Lim, N.; et al. Genetic variants influencing circulating lipids levels and risk of coronary artery disease. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2264–2276. [Google Scholar]
- Xia, Z.B.; Popovic, R.; Chen, J.; Theisler, C.; Stuart, T.; Santillan, D.A.; Erfurth, F.; Diaz, M.O.; Zeleznik-Le, N.J. The MLL fusion gene, MLL-AF4, regulates cyclin-dependent kinase inhibitor CDKN1B (p27kip1) expression. Proc. Natl. Acad. Sci. USA 2005, 102, 14028–14033. [Google Scholar] [CrossRef] [PubMed]
- Naaz, A.; Holsberger, D.R.; Iwamoto, G.A.; Nelson, A.; Kiyokawa, H.; Cooke, P.S. Loss of cyclin-dependent kinase inhibitors produces adipocyte hyperplasia and obesity. FASEB J. 2004, 18, 1925–1927. [Google Scholar] [PubMed]
- Kosicka, A.; Cunliffe, A.D; Mackenzie, R.; Zariwala, M.G.; Parretti, M.; Flower, R.J.; Renshaw, D. Attenuation of plasma annexin A1 in human obesity. FASEB J. 2013, 27, 368–378. [Google Scholar]
- Cole, T.J.; Bellizi, M.C.; FLegal, K.M.; Dietz, W.H. Establishing a standard definition for child overweight and obesity worldwide: International survey. BMJ 2000, 320, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Database for Annotation, Visualization and Integrated Discovery (DAVID). Available online: http://http://david.abcc.ncifcrf.gov/ (accessed on 15 February 2015).
- QIAGEN’s Ingenuity® Pathway Analysis. Available online: www.qiagen.com/ingenuity (accessed on 15 February 2015).
- National Center for Biotechnology Information Gene Expression Omnibus web site. Available online: http://www.ncbi.nlm.nih.gov/projects/geo/ (accessed on 15 February 2015).
- Yuan, J.S.; Reed, A.; Chen, F.L.; Stewart, N.C. Statistical analysis of real-time PCR data. BMC Bioinform. 2006, 7. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aguilera, C.M.; Gomez-Llorente, C.; Tofe, I.; Gil-Campos, M.; Cañete, R.; Gil, Á. Genome-Wide Expression in Visceral Adipose Tissue from Obese Prepubertal Children. Int. J. Mol. Sci. 2015, 16, 7723-7737. https://doi.org/10.3390/ijms16047723
Aguilera CM, Gomez-Llorente C, Tofe I, Gil-Campos M, Cañete R, Gil Á. Genome-Wide Expression in Visceral Adipose Tissue from Obese Prepubertal Children. International Journal of Molecular Sciences. 2015; 16(4):7723-7737. https://doi.org/10.3390/ijms16047723
Chicago/Turabian StyleAguilera, Concepción M., Carolina Gomez-Llorente, Inés Tofe, Mercedes Gil-Campos, Ramón Cañete, and Ángel Gil. 2015. "Genome-Wide Expression in Visceral Adipose Tissue from Obese Prepubertal Children" International Journal of Molecular Sciences 16, no. 4: 7723-7737. https://doi.org/10.3390/ijms16047723
APA StyleAguilera, C. M., Gomez-Llorente, C., Tofe, I., Gil-Campos, M., Cañete, R., & Gil, Á. (2015). Genome-Wide Expression in Visceral Adipose Tissue from Obese Prepubertal Children. International Journal of Molecular Sciences, 16(4), 7723-7737. https://doi.org/10.3390/ijms16047723






