Male Reproductive Cancers and Infertility: A Mutual Relationship
Abstract
:1. Introduction
2. Reproductive Cancers
2.1. Testicular Cancer
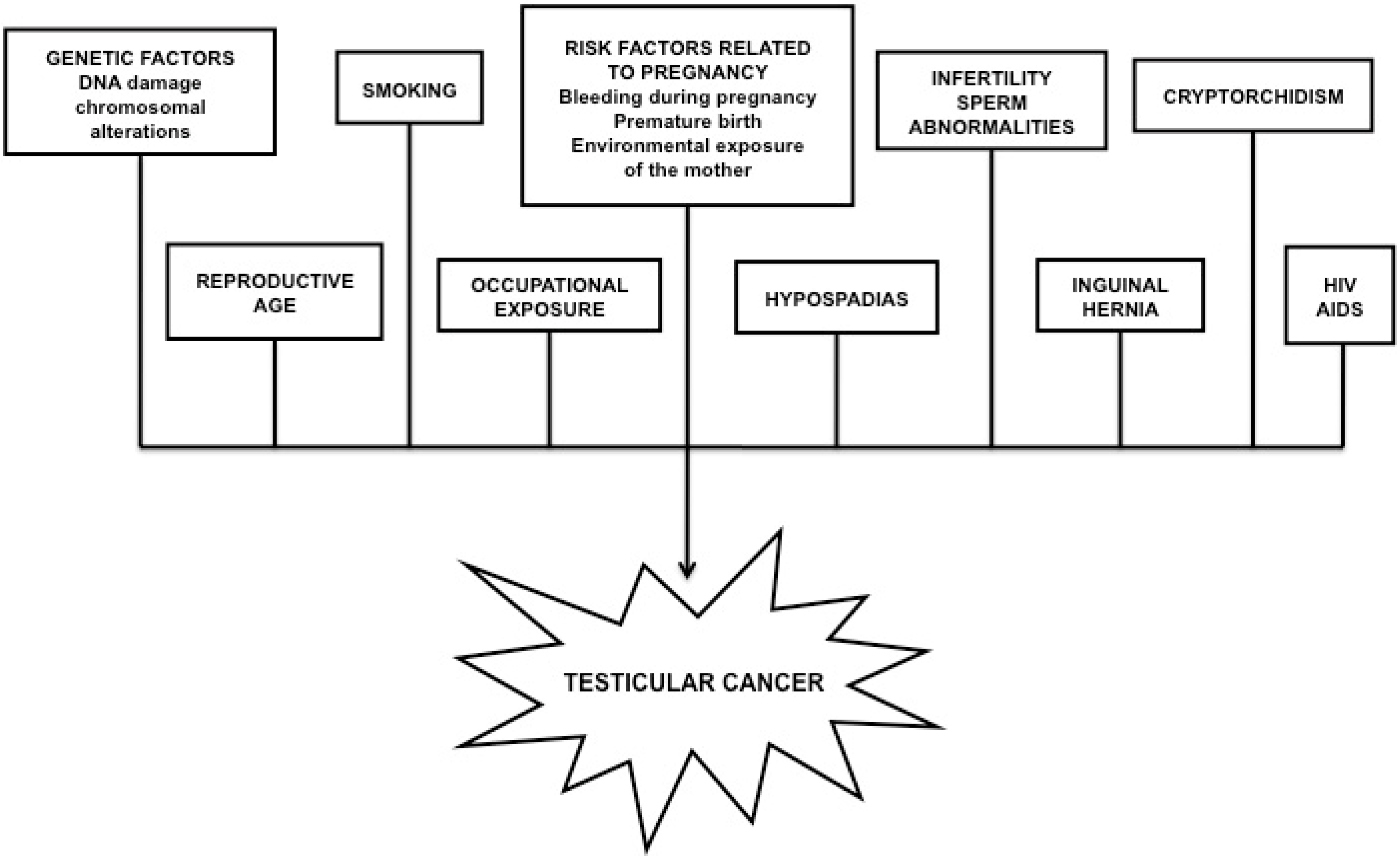
2.2. Prostate Cancer
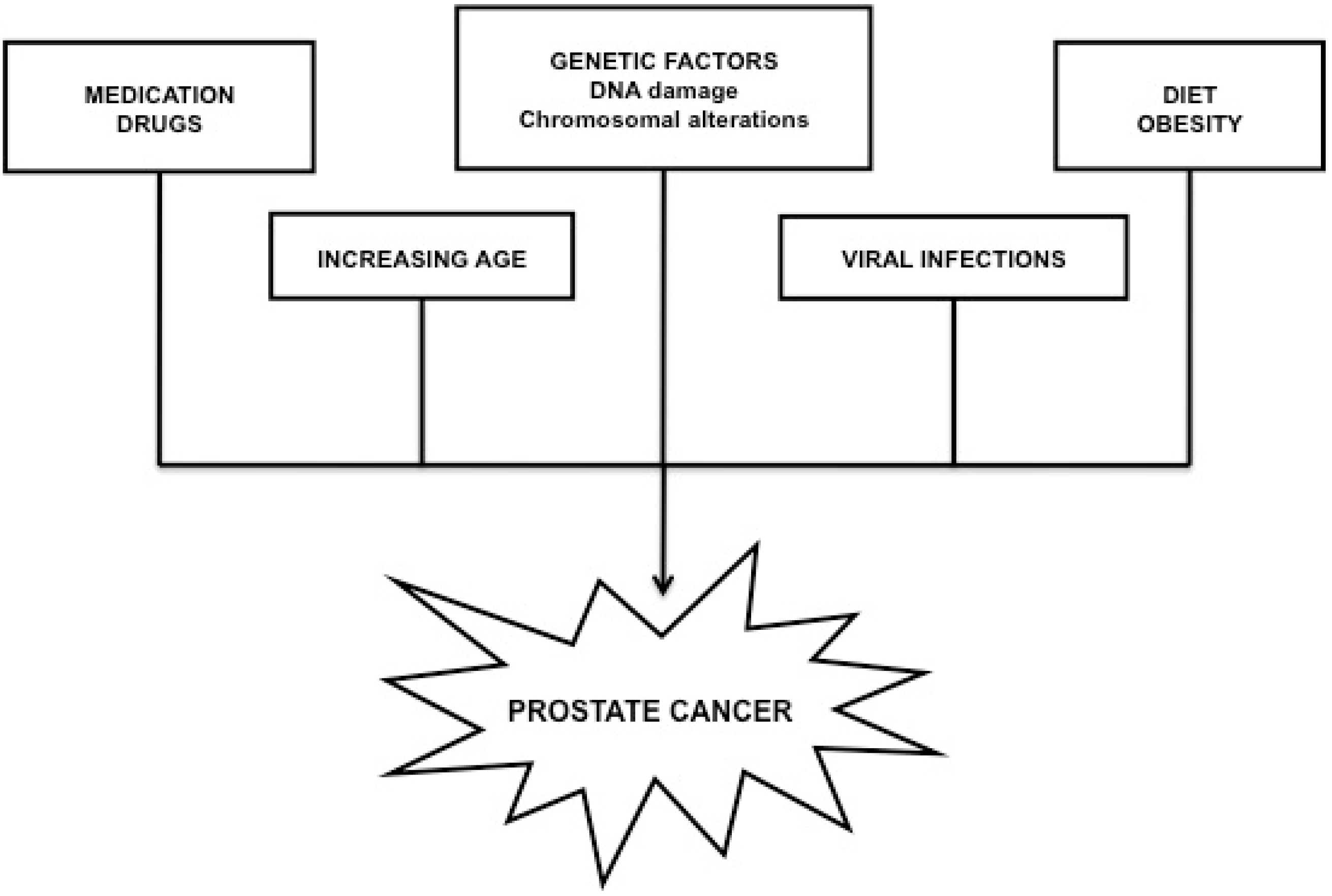
3. Causes and Factors Governing the Association between Male Infertility and Reproductive Cancers
3.1. Genetic Factors
3.2. Epigenetic Factors
3.3. Environmental Factors
4. Infertility: A Precedent of Male Reproductive Cancers?
| Author(s) | Country and Year | Design | Subjects | Finding(s) | Conclusions |
|---|---|---|---|---|---|
| Pryor et al. [129] | UK 1983 | Case study | 2043 males from infertile couples who underwent testicular biopsy from 1955 to 1982. |
| The findings are applicable to the selection of patients for biopsy and appropriate treatment of CIS when diagnosed. |
| Strader et al. [136] | Western Washington State, USA 1988 | Population based case-control study | Patients diagnosed with TC between 1977 and 1983 (n = 333) and 675 healthy controls. |
| The study supports the hypothesis that one or more local factors may account for the increased risk of germ cell testicular tumors in cryptorchidic men. |
| Møller and Skakkebæk [137] | Denmark 1999 | Population based case-control study | 514 patients diagnosed with TC identified in the Danish Cancer Registry and 720 controls randomly selected from the Danish population. |
| Data supporting the hypothesis that compromised male fertility and TC share important etiologies. |
| Jacobsen et al. [132] | Denmark 2000 | Cohort study | 3530 Danish men, born between 1945–1980 and diagnosed with TC in the period of 1960–1993. Control: the total population of Danish men born between 1945–1980 (n = 1,488,957) and their biological children (n = 1,250,989). |
| The study confirms earlier results from less conclusive studies, and indicates that TC, subfertility and a female-biased sex ratio among newborns are interrelated by biological mechanisms. |
| Jacobsen et al. [134] | Denmark 2000 | Cohort study | 32,442 men who had a semen analysis done during 1963–1995. |
| The results emphasize on the existence of common etiologies for low semen quality and TC. Low semen quality may be associated with increased incidence of germ cell tumors. |
| Pasqualotto et al. [138] | Cleveland, USA 2003 | Case study | Seven patients presenting with infertility, followed by eventual TC diagnosis over a 15-year period. |
| Most of the men who have TC and male infertility will most likely present with a seminona. Men diagnosed with infertility should be thoroughly investigated to rule out diseases associated with their infertility. |
| Richiardi et al. [139] | Sweden 2004 | Population based case-control study | 4592 patients with TC and 12,254 control subjects. |
| The report provides evidence of an association between subfertility and the subsequent risk for TC. |
| Doria-Rose et al. [135] | Western Washington State, USA 2005 | Case-control study | 329 TC patients diagnosed from 1977 to 1983, and 672 cancer-free controls. |
| The results are consistent with an increased risk of TC among men with reduced fertility, going beyond the effects of cryptorchidism. |
| Walsh et al. [2] | State of California, USA 2009 | Cohort study | A total of 51,461 couples evaluated for infertility from 1967 to 1998 linked with 22,562 TC patients. |
| Men with male factor infertility have an increased risk of subsequently developing TC, suggesting common etiologic factors for infertility and TC. |
| Author(s) | Country and Year | Design | Subjects | Finding(s) | Conclusions |
|---|---|---|---|---|---|
| Giwercman et al. [150] | Sweden 2005 | Population-based case-control | 48,850 cases of PC between 1958–1998. For each case, one control was matched by year of birth. |
| A dysfunctional reproductive system supporting the prostatic growth to a lesser extent could be a feasible underlying cause of this association. |
| Negri et al. [151] | Italy 2006 | Case-control study | 1294 patients diagnosed with PC between 1991 and 2002, and 1451 controls as cases for a wide spectrum of acute and non-neoplastic conditions. |
| The report concludes that the relation between the number of children and PC risk remains controversial. |
| Haralp et al. [152] | Israel 2007 | Cohort study | 15,268 fathers followed for 28–41 years from the birth of a live offspring. |
| The study suggests that stillbirth and PC may have shared environmental causes. Genetic susceptibility to PC might increase the risk of a stillbirth in offspring. |
| Jørgensen et al. [149] | Denmark 2008 | Cohort study | All men born in Denmark between 1935 and 1988, among whom 3400 developed PC during follow-ups between 1968 and 2003. |
| Men without children are at a moderately reduced risk of PC. Among men with children, there appears to be a linear decline in PC occurrence with an increasing number of children, independent of the sex of the offspring. |
| Ruhayel et al. [46] | Sweden 2010 | Case-control study | 445 PC cases and 446 controls. 841 men were biological fathers and 50 men were infertile. | Infertile men were at a significantly lower risk of being diagnosed with PC than fertile men (odds ratio of 0.45). | Enduring male infertility may be associated with a reduced PC risk, validating the theory that normal testicular function and steroidogenesis are important factors to the later development of PC. |
| Walsh et al. [140] | State of California, USA 2010 | Population-based case-control | A total of 22,562 patients being evaluated for infertility from 1967 to 1998, and linked to the cancer registry. The incidence of PC was compared with the incidence in an age- and geography-matched sample of men from the general population. |
| Male infertility may be an early and identifiable risk factor for the development of clinically significant PC. |
| Wirén et al. [153] | Sweden 2013 | Population-based case-control | 117,328 PC cases and 562,644 controls, matched on birth year and residence. |
| The report claims that associations between the fatherhood status and PC are predominantly due to socioeconomic factors influencing health care-seeking behavior. |
5. Clinical Potential of MicroRNAs in the Diagnosis and Treatment of Male Infertility and Reproductive Cancers
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Honig, S.C.; Lipshultz, L.I.; Jarow, J. Significant medical pathology uncovered by a comprehensive male infertility evaluation. Fertil. Steril. 1994, 62, 1028–1034. [Google Scholar] [PubMed]
- Walsh, T.J.; Croughan, M.S.; Schembri, M.; Chan, J.M.; Turek, P.J. Increased risk of testicular germ cell cancer among infertile men. Arch. Intern. Med. 2009, 169, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.J. Male reproductive health and prostate cancer risk. Curr. Opin. Urol. 2011, 21, 506–513. [Google Scholar] [CrossRef] [PubMed]
- McGlynn, K.A.; Cook, M.B. The epidemiology of testicular cancer. In Male Reproductive Cancers: 51 Epidemiology, Pathology and Genetics, 1st ed.; Foulkes, W.D., Cooney, K.A., Eds.; Springer Science + Business Media, LLC: New York, NY, USA, 2010; pp. 51–83. [Google Scholar]
- Shanmugalingam, T.; Soultati, A.; Chowdhury, S.; Rudman, S.; van Hemelrijck, M. Global incidence and outcome of testicular cancer. Clin. Epidemiol. 2013, 5, 417–427. [Google Scholar] [PubMed]
- Rajpert-De Meyts, E.; Jorgensen, N.; Brondum-Nielsen, K.; Muller, J.; Skakkebaek, N.E. Developmental arrest of germ cells in the pathogenesis of germ cell neoplasia. APMIS 1998, 106, 198–204. [Google Scholar]
- Umeh, K.; Chadwick, R. Early detection of testicular cancer: revisiting the role of self-efficacy in testicular self-examination among young asymptomatic males. J. Behav. Med. 2010. [Google Scholar] [CrossRef]
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics, 2013. CA Cancer J. Clin. 2013, 63, 11–30. [Google Scholar] [CrossRef] [PubMed]
- Humphrey, P.A. Histological variants of prostatic carcinoma and their significance. Histopathology 2012, 60, 59–74. [Google Scholar] [CrossRef] [PubMed]
- Grignon, D.J. Unusual subtypes of prostate cancer. Modern Pathol. 2004, 17, 316–327. [Google Scholar] [CrossRef]
- Bubendorf, L.; Schöpfer, A.; Wagner, U.; Sauter, G.; Moch, H.; Willi, N.; Gasser, T.C.; Mihatsch, M.J. Metastatic patterns of prostate cancer: An autopsy study of 1589 patients. Hum. Pathol. 2000, 31, 578–583. [Google Scholar] [CrossRef] [PubMed]
- Gronberg, H. Prostate cancer epidemiology. Lancet 2003, 361, 859–864. [Google Scholar] [CrossRef] [PubMed]
- Turnbull, C.; Rahman, N. Genome-wide association studies provide new insights into the genetic basis of testicular germ-cell tumour. Int. J. Androl. 2011, 34, e86–e96. [Google Scholar] [CrossRef] [PubMed]
- Uehara, S.; Hashiyada, M.; Sato, K.; Nata, M.; Funato, T.; Okamura, K. Complete XY gonadal dysgenesis and aspects of the SRY genotype and gonadal tumor formation. J. Hum. Genet. 2002, 47, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Rajpert-De Meyts, E. Developmental model for the pathogenesis of testicular carcinoma in situ: Genetic and environmental aspects. Hum. Reprod. Update 2006, 12, 303–323. [Google Scholar] [CrossRef] [PubMed]
- Jiang, L.I.; Nadeau, J.H. 129/Sv mice-a model system for studying germ cell biology and testicular cancer. Mamm. Genome 2001, 12, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Burns, W.R.; Dada, R.; Sabanegh, E.S., Jr. Male infertility and testicular cancer—Points of common causality. Eur. Urol. Rev. 2010, 5, 56–59. [Google Scholar]
- Fedier, A.; Fink, D. Mutations in DNA mismatch repair genes: implications for DNA damage signaling and drug sensitivity (review). Int. J. Oncol. 2004, 24, 1039–1047. [Google Scholar] [PubMed]
- Leongamornlert, D.; Saunders, E.; Dadaev, T.; Tymrakiewicz, M.; Goh, C.; Jugurnauth-Little, S.; Kozarewa, I.; Fenwick, K.; Assiotis, I.; Barrowdale, D.; et al. Frequent germline deleterious mutations in DNA repair genes in familial prostate cancer cases are associated with advanced disease. Br. J. Cancer 2014, 110, 1663–1672. [Google Scholar] [CrossRef] [PubMed]
- Vogelstein, B.; Sur, S.; Prives, C. p53: The most frequently altered gene in human cancers. Nat. Educ. 2010, 3, 6. [Google Scholar]
- Burns, W.R.; Sabanegh, E.; Dada, R.; Rein, B.; Agarwal, A. Is male infertility a forerunner to cancer? Int. Braz. J. Urol. 2010, 36, 527–536. [Google Scholar] [CrossRef] [PubMed]
- Looijenga, L.H.; Zafarana, G.; Grygalewicz, B.; Summersgill, B.; Debiec-Rychter, M.; Veltman, J.; Oosterhuis, J.W. Role of gain of 12p in germ cell tumour development. APMIS 2003, 111, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez, S.; Jafer, O.; Goker, H.; Summersgill, B.M.; Zafarana, G.; Gillis, A.J.; van Gurp, R.J.; Oosterhuis, J.W.; Lu, Y.J.; Huddart, R.; et al. Expression profile of genes from 12p in testicular germ cell tumors of adolescents and adults associated with i(12p) and amplification at 12p11.2-p12.1. Oncogene 2003, 22, 1880–1891. [Google Scholar] [CrossRef] [PubMed]
- Von Eyben, F.E. Chromosomes, genes, and development of testicular germ cell tumors. Cancer Genet. Cytogenet. 2004, 151, 93–138. [Google Scholar] [CrossRef] [PubMed]
- Perinchery, G.; Sasaki, M.; Angan, A.; Kumar, V.; Carroll, P.; Dahiya, R. Deletion of Y-chromosome specific genes in human prostate cancer. J. Urol. 2000, 163, 1339–1342. [Google Scholar] [CrossRef] [PubMed]
- McMullin, R.P.; Mutton, L.N.; Bieberich, C.J. Hoxb13 regulatory elements mediate transgene expression during prostate organogenesis and carcinogenesis. Dev. Dyn. 2009, 238, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Reijo, R.; Alagappan, R.K.; Patrizio, P.; Page, D.C. Severe oligozoospermia resulting from deletions of azoospermia factor gene on Y chromosome. Lancet 1996, 347, 1290–1293. [Google Scholar] [CrossRef] [PubMed]
- Kleiman, S.E.; Bar-ShiraMaymon, B.; Yogev, L.; Paz, G.; Yavetz, H. The prognostic role of the extent of Y microdeletion on spermatogenesis and maturity of Sertoli cells. Hum. Reprod. 2001, 16, 399–402. [Google Scholar] [CrossRef] [PubMed]
- Lau, Y.F.; Zhang, J. Expression analysis of thirty one Y chromosome genes in human prostate cancer. Mol. Carcinog. 2000, 27, 308–321. [Google Scholar] [CrossRef] [PubMed]
- Mosaad, Y.M.; Shahin, D.; Elkholy, A.A.; Mosbah, A.; Badawy, W. CAG repeat length in androgen receptor gene and male infertility in Egyptian patients. Andrologia 2012, 44, 26–33. [Google Scholar] [CrossRef] [PubMed]
- Krausz, C.; Forti, G.; McElreavey, K. The Y chromosome and male fertility and infertility. Int. J. Androl. 2003, 26, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Krausz, C.; Quintana-Murci, L.; Forti, G. Y chromosome polymorphisms in medicine. Ann. Med. 2004, 36, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Silva Neto, B.; Koff, W.J.; Biolchi, V.; Brenner, C.; Biolo, K.D.; Spritzer, P.M.; Brum, I.S. Polymorphic CAG and GGC repeat lengths in the androgen receptor gene and prostate cancer risk: Analysis of a Brazilian population. Cancer Investig. 2008, 26, 74–80. [Google Scholar] [CrossRef]
- Mittal, R.D.; Mishra, D.; Mandhani, A.K. Role of an androgen receptor gene polymorphism in development of hormone refractory prostate cancer in Indian population. Asian Pac. J. Cancer Prev. 2007, 8, 275–278. [Google Scholar] [PubMed]
- Giwercman, C.; Giwercman, A.; Pedersen, H.S.; Toft, G.; Lundin, K.; Bonde, J.P.; Lundberg Giwercman, Y. Polymorphisms in genes regulating androgen activity among prostate cancer low-risk Inuit men and high-risk Scandinavians. Int. J. Androl. 2008, 31, 25–30. [Google Scholar] [PubMed]
- Veveris-Lowe, T.L.; Lawrence, M.G.; Collard, R.L.; Bui, L.; Herington, A.C.; Nicol, D.L.; Clements, J.A. Kallikrein 4 (hK4) and prostate-specific antigen (PSA) are associated with the loss of E-cadherin and an epithelial-mesenchymal transition (EMT)-like effect in prostate cancer cells. Endocr. Relat. Cancer 2005, 12, 631–643. [Google Scholar] [CrossRef] [PubMed]
- Nudell, D.; Castillo, M.; Turek, P.J.; Pera, R.R. Increased frequency of mutations in DNA from infertile men with meiotic arrest. Hum. Reprod. 2000, 15, 1289–1294. [Google Scholar] [CrossRef] [PubMed]
- Judis, L.; Chan, E.R.; Schwartz, S.; Seftel, A.; Hassold, T. Meiosis I arrest and azoospermia in an infertile male explained by failure of formation of a component of the synaptonemal complex. Fertil. Steril. 2004, 81, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Wei, K.; Clark, A.B.; Wong, E.; Kane, M.F.; Mazur, D.J.; Parris, T.; Kolas, N.K.; Russell, R.; Hou, H., Jr.; Kneitz, B.; et al. Inactivation of Exonuclease 1 in mice results in DNA mismatch repair defects, increased cancer susceptibility, and male and female sterility. Genes Dev. 2003, 17, 603–614. [Google Scholar] [CrossRef] [PubMed]
- Norris, A.M.; Woodruff, R.D.; D’Agostino, R.B., Jr.; Clodfelter, J.E.; Scarpinato, K.D. Elevated levels of the mismatch repair protein PMS2 are associated with prostate cancer. Prostate 2007, 67, 214–225. [Google Scholar] [CrossRef] [PubMed]
- Baskin, L.S. Abnormalities of Sexual Determination & Differentiation. In Smith’s General Urology, 17th ed.; Tanagho, E.A., McAninch, J.W., Eds.; McGraw Hill: New York, NY, USA, 2008; p. 649. [Google Scholar]
- Hafeez, S.; Sharma, R.A.; Huddart, R.A.; Dearnaley, D.P.; Horwich, A. Challenges in treating patients with Down’s syndrome and testicular cancer with chemotherapy and radiotherapy: The Royal Marsden experience. Clin. Oncol. 2007, 19, 135–142. [Google Scholar] [CrossRef]
- Patja, K.; Sund, R.; Kaski, M.; Pukkala, E. Cancer incidence among persons with Prader-Willi syndrome in Finland. Int. J. Disabil. Hum. Dev. 2008, 7, 69–72. [Google Scholar] [CrossRef]
- Nielsen, J.; Wohlert, M. Chromosome abnormalities found among 34,910 newborn children: Results from a 13-year incidence study in Arhus, Denmark. Hum. Genet. 1991, 87, 81–83. [Google Scholar] [CrossRef] [PubMed]
- Hasle, H.; Mellemgaard, A.; Nielsen, J.; Hansen, J. Cancer incidence in men with Klinefelter syndrome. Br. J. Cancer 1995, 71, 416–420. [Google Scholar] [CrossRef] [PubMed]
- Ruhayel, Y.; Giwercman, A.; Ulmert, D.; Rylander, L.; Bjartell, A.; Manjer, J.; Giwercman, Y.L. Male infertility and prostate cancer risk: a nested case-control study. Cancer Causes Control 2010, 21, 1635–1643. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, A.J.; Schoemaker, M.J.; Higgins, C.D.; Wright, A.F.; Jacobs, P.A. Cancer incidence and mortality in men with Klinefelter syndrome: a cohort study. J. Natl. Cancer Inst. 2005, 97, 1204–1210. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.J.; Dharmawardana, P.G.; Uchio, E.M.; Wynberg, J.; Phillips, J.L. Prostate cancer in Klinefelter syndrome during hormonal replacement therapy. Urology 2003, 62, 941. [Google Scholar] [CrossRef] [PubMed]
- Bydder, S.A.; Joseph, D.J.; Weinstein, S.; Stuckey, B.G. Prostate cancer following testosterone replacement in Klinefelter syndrome. ANZ J. Surg. 2007, 77, 93–94. [Google Scholar] [CrossRef] [PubMed]
- Dada, R.; Kumar, M.; Jesudasan, R.; Fernández, J.L.; Gosálvez, J.; Agarwal, A. Epigenetics and its role in male infertility. J. Assist. Reprod. Genet. 2012, 29, 213–223. [Google Scholar] [CrossRef] [PubMed]
- Ooi, S.L.; Henikoff, S. Germline histone dynamics and epigenetics. Curr. Opin. Cell. Biol. 2007, 19, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Ito, T. Role of histone modification in chromatin dynamics. J. Biochem. 2007, 141, 609–614. [Google Scholar] [CrossRef] [PubMed]
- Houshdaran, S.; Cortessis, V.K.; Siegmund, K.; Yang, A.; Laird, P.W.; Sokol, R.Z. Widespread epigenetic abnormalities suggest a broad DNA methylation erasure defect in abnormal human sperm. PLoS ONE 2007, 2, e1289. [Google Scholar] [CrossRef] [PubMed]
- Platts, A.E.; Dix, D.J.; Chemes, H.E.; Thompson, K.E.; Goodrich, R.; Rockett, J.C.; Rawe, V.Y.; Quintana, S.; Diamond, M.P.; Strader, L.F.; et al. Success and failure in human spermatogenesis as revealed by teratozoospermic RNAs. Hum. Mol. Genet. 2007, 16, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Ostermeier, G.C.; Dix, D.J.; Miller, D.; Khatri, P.; Krawetz, S.A. Spermatozoal RNA profiles of normal fertile men. Lancet 2002, 360, 772–777. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, S.E.; Houseman, E.A.; Christensen, B.C.; Marsit, C.J.; Kelsey, K.T.; Sigman, M.; Boekelheide, K. Integrative DNA methylation and gene expression analyses identify DNA packaging and epigenetic regulatory genes associated with low motility sperm. PLoS ONE 2011, 6, e20280. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Zhou, Z.; Xu, M.; Li, J.; Xiao, J.; Xu, Z.Y.; Sha, J. A spermatogenesis-related gene expression profile in human spermatozoa and its potential clinical applications. J. Mol. Med. 2004, 82, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Carrell, D.T. Epigenetics of the male gamete. Fertil. Steril. 2012, 97, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Suter, C.M.; Martin, D.I.; Ward, R.L. Germline epimutation of MLH1 in individuals with multiple cancers. Nat. Genet. 2004, 36, 497–501. [Google Scholar] [CrossRef] [PubMed]
- Rajender, S.; Avery, K.; Agarwal, A. Epigenetics, spermatogenesis and male infertility. Mutat. Res. 2011, 727, 62–71. [Google Scholar] [CrossRef] [PubMed]
- Albany, C.; Alva, A.S.; Aparicio, A.M.; Singal, R.; Yellapragada, S.; Sonpavde, G.; Hahn, N.M. Epigenetics in prostate cancer. Prostate Cancer 2011. [Google Scholar] [CrossRef]
- Godmann, M.; Lambrot, R.; Kimmins, S. The dynamic epigenetic program in male germ cells: Its role in spermatogenesis, testis cancer, and its response to the environment. Microsc. Res. Tech. 2009, 72, 603–619. [Google Scholar] [CrossRef] [PubMed]
- Alagaratnam, S.; Lind, G.E.; Kraggerud, S.M.; Lothe, R.A.; Skotheim, R.I. The testicular germ cell tumour transcriptome. Int. J. Androl. 2011, 34, e133–e150. [Google Scholar] [CrossRef] [PubMed]
- Le Bouc, Y.; Rossignol, S.; Azzi, S.; Brioude, F.; Cabrol, S.; Gicquel, C.; Netchine, I. Epigenetics, genomic imprinting and developmental disorders. Bull. Acad. Natl. Med. 2010, 194, 287–300. [Google Scholar] [PubMed]
- Vega, A.; Baptissart, M.; Caira, F.; Brugnon, F.; Lobaccaro, J.M.A.; Volle, D.H. Epigenetic: A molecular link between testicular cancer and environmental exposures. Front. Endocrinol. 2012, 3, 150. [Google Scholar] [CrossRef]
- Nettersheim, D.; Biermann, K.; Gillis, A.J.M.; Steger, K.; Looijenga, L.H.J.; Schorle, H. NANOG promoter methylation and expression correlation during normal and malignant human germ cell development. Epigenetics 2011, 6, 114–122. [Google Scholar] [CrossRef] [PubMed]
- De Jong, J.; Weeda, S.; Gillis, A.J.M.; Oosterhuis, J.W.; Looijenga, L.H.J. Differential methylation of the OCT3/4 upstream region in primary human testicular germ cell tumors. Oncol. Rep. 2007, 18, 127–132. [Google Scholar] [PubMed]
- Krausz, C.; Guarducci, E.; Becherini, L.; Degl’Innocenti, S.; Gerace, L.; Balercia, G.; Forti, G. The clinical significance of the POLG gene polymorphism in male infertility. J. Clin. Endocrinol. MeTable 2004, 89, 4292–4297. [Google Scholar] [CrossRef]
- Park, J.Y. Promoter hypermethylation in prostate cancer. Cancer Control. 2010, 17, 245–255. [Google Scholar] [PubMed]
- Horsthemke, B.; Buiting, K. Imprinting defects on human chromosome 15. Cytogenet. Genome Res. 2006, 113, 292–299. [Google Scholar] [CrossRef] [PubMed]
- Spira, A.; Multigner, L. The effect of industrial and agricultural pollution on human spermatogenesis. Hum. Reprod. 1998, 13, 2041–2042. [Google Scholar] [CrossRef] [PubMed]
- Béranger, R.; le Cornet, C.; Schüz, J.; Fervers, B. Occupational and environmental exposures associated with testicular germ cell tumours: Systematic review of prenatal and life-long exposures. PLoS ONE 2013, 8, e77130. [Google Scholar] [CrossRef] [PubMed]
- Doolan, G.; Benke, G.; Giles, G. An update on occupation and prostate cancer. Asian Pac. J. Cancer Prev. 2014, 15, 501–516. [Google Scholar] [CrossRef] [PubMed]
- Chacko, J.K.; Barthold, J.S. Genetic and environmental contributors to cryptorchidism. Pediatr. Endocrinol. Rev. 2009, 6, 476–480. [Google Scholar] [PubMed]
- Carmichael, S.L.; Shaw, G.M.; Lammer, E.J. Environmental and genetic contributors to hypospadias: A review of the epidemiologic evidence. Birth Defects Res. A. Clin. Mol. Teratol. 2012, 94, 499–510. [Google Scholar] [CrossRef] [PubMed]
- Oliva, A.; Spira, A.; Multigner, L. Contribution of environmental factors to the risk of male infertility. Hum. Reprod. 2001, 16, 1768–1776. [Google Scholar] [CrossRef] [PubMed]
- Adams, A.; Galloway, T.S.; Mondal, D.; Esteves, S.C.; Mathews, F. Effect of mobile telephones on sperm quality: A systematic review and meta-analysis. Environ. Int. 2014, 70, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Mendiola, J.; Torres-Cantero, A.M.; Moreno-Grau, J.M.; Ten, J.; Roca, M.; Moreno-Grau, S.; Bernabeu, R. Exposure to environmental toxins in males seeking infertility treatment: A case-controlled study. Reprod. Biomed. Online 2008, 16, 842–850. [Google Scholar] [CrossRef] [PubMed]
- Sharpe, R.N.; Skakkebaek, N.E. Are oestrogens involved in falling sperm counts and disorders of the male reproductive tract? Lancet 1993, 343, 1392–1395. [Google Scholar] [CrossRef]
- Zoeller, R.T.; Brown, T.R.; Doan, L.L.; Gore, A.C.; Skakkebaek, N.E.; Soto, A.M.; Woodruff, T.J.; vom Saal, F.S. Endocrine-disrupting chemicals and public health protection: A statement of principles from The Endocrine Society. Endocrinology 2012, 153, 4097–4110. [Google Scholar] [CrossRef] [PubMed]
- De Coster, S.; van Larebeke, N. Endocrine-disrupting chemicals: Associated disorders and mechanisms of action. J. Environ. Pub. Health 2012. [Google Scholar] [CrossRef]
- Yoshioka, W.; Peterson, R.E.; Tohyama, C. Molecular targets that link dioxin exposure to toxicity phenotypes. J. Steroid Biochem. Mol. Biol. 2011, 127, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Ohtake, F.; Fujii-Kuriyama, Y.; Kawajiri, K.; Kato, S. Cross-talk of dioxin and estrogen receptor signals through the ubiquitin system. J. Steroid Biochem. Mol. Biol. 2011, 127, 102–107. [Google Scholar] [CrossRef] [PubMed]
- Phillips, K.P.; Foster, W.G. Key developments in endocrine disrupter research and human health. J. Toxicol. Environ. Health B 2008, 11, 322–344. [Google Scholar] [CrossRef]
- Whitehead, S.A.; Rice, S. Endocrine-disrupting chemicals as modulators of sex steroid synthesis. Best Pract. Res. Clin. Endocrinol. MeTable 2006, 20, 45–61. [Google Scholar] [CrossRef]
- Marques-Pinto, A.; Carvalho, A. Human infertility: Are endocrine disruptors to blame? Endocr. Connect. 2013, 2, R15–R29. [Google Scholar] [CrossRef] [PubMed]
- Cederroth, C.R.; Zimmermann, C.; Nef, S. Soy phytoestrogens and their impact on reproductive health. Mol. Cell. Endocrinol. 2012, 355, 192–200. [Google Scholar] [CrossRef] [PubMed]
- Iavicoli, I.; Fontana, L.; Bergamaschi, A. The effects of metals as endocrine disruptors. J. Toxicol. Environ. Health B 2009, 12, 206–223. [Google Scholar] [CrossRef]
- Aitken, R.J.; Skakkebaek, N.E.; Roman, S.D. Male reproductive health and the environment. Med. J. Aust. 2006, 185, 414–415. [Google Scholar] [PubMed]
- Skakkebaek, N.E.; De Meyts, E.R.; Main, K.M. Testicular dysgenesis syndrome: An increasingly common developmental disorder with environmental aspects. Hum. Reprod. 2001, 16, 972–978. [Google Scholar] [CrossRef] [PubMed]
- Docimo, S.G.; Silver, R.; Cromie, W. The undescended testicle: diagnosis and management. Am. Fam. Physician 2000, 62, 2037–2048. [Google Scholar] [PubMed]
- Lu, C.C.; Steinberger, A. Effects of estrogen on human seminiferous tubules: Light and electron microscopic analysis. Am. J. Anat. 1978, 153, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Fowler, K.A.; Gill, K.; Kirma, N.; Dillehay, D.L.; Tekmal, R.R. Overexpression of aromatase leads to development of testicular Leydig cell tumors: An in vivo model for hormone-mediated testicular cancer. Am. J. Pathol. 2000, 156, 347–353. [Google Scholar] [CrossRef] [PubMed]
- Weir, H.K.; Marrett, L.D.; Kreiger, N.; Darlington, G.A.; Sugar, L. Pre-natal and peri-natal exposures and risk of testicular germ-cell cancer. Int. J. Cancer 2000, 87, 438–443. [Google Scholar] [CrossRef] [PubMed]
- Joensen, U.N.; Jorgensen, N.; Rajpert-De Meyts, E.; Skakkebaek, N.E. Testicular dysgenesis syndrome and Leydig cell function. Basic Clin. Pharmacol. Toxicol. 2008, 102, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Andersson, A.M.; Jorgensen, N.; Frydelund-Larsen, L.; Rajpert-De Meyts, E.; Skakkebaek, N.E. Impaired Leydig cell function in infertile men: A study of 357 idiopathic infertile men and 318 proven fertile controls. J. Clin. Endocrinol. MeTable 2004, 89, 3161–3167. [Google Scholar] [CrossRef]
- Olesen, I.A.; Sonne, S.B.; Hoei-Hansen, C.E.; De Meyts, E.R.; Skakkebaek, N.E. Environment, testicular dysgenesis and carcinoma in situ testis. Best Pract. Res. Clin. Endocrinol. MeTable 2007, 21, 462–478. [Google Scholar] [CrossRef]
- Del-Mazo, J.; Brieño-Enríquez, M.A.; García-López, J.; López-Fernández, L.A.; De-Felici, M. Endocrine disruptors, gene deregulation and male germ cell tumors. Int. J. Dev. Biol. 2013, 57, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Andersen, H.R.; Schmidt, I.M.; Grandjean, P.; Jensen, T.K.; Budtz-Jorgensen, E.; Kjaerstad, M.B.; Baelum, J.; Nielsen, J.B.; Skakkebaek, N.E.; Main, K.M. Impaired reproductive development in sons of women occupationally exposed to pesticides during pregnancy. Environ. Health Perspect. 2008, 116, 566–572. [Google Scholar] [CrossRef] [PubMed]
- Brucker-Davis, F.; Wagner-Mahler, K.; Delattre, I.; Ducot, B.; Ferrari, P.; Bongain, A.; Kurzenne, J.Y.; Mas, J.C.; Fenichel, P. Cryptorchidism study group from Nice. Cryptorchidism at birth in Nice area (France) is associated with higher prenatal exposure to PCBs and DDE, as assessed by colostrum concentrations. Hum. Reprod. 2008, 23, 1708–1718. [Google Scholar] [CrossRef] [PubMed]
- Fernandez, M.F.; Olmos, B.; Granada, A.; Lopez-Espinosa, M.J.; Molina-Molina, J.M.; Fernandez, J.M.; Cruz, M.; Olea-Serrano, F.; Olea, N. Human exposure to endocrine-disrupting chemicals and prenatal risk factors for cryptorchidism and hypospadias: A nested case-control study. Environ. Health Perspect. 2007, 115, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.; Tang, R.; Fu, G.; Xu, B.; Zhu, P.; Qiao, S.; Chen, X.; Xu, B.; Qin, Y.; Lu, C.; et al. Association of exposure to phenols and idiopathic male infertility. J. Hazard. Mater. 2013, 250–251, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, N.; Vierula, M.; Jacobsen, R.; Pukkala, E.; Perheentupa, A.; Virtanen, H.E.; Skakkebaek, N.E.; Toppari, J. Recent adverse trends in semen quality and testis cancer incidence among Finnish men. Int. J. Androl. 2011, 34, e37–e48. [Google Scholar] [CrossRef] [PubMed]
- Hardell, L.; Bavel, B.; Lindstrom, G.; Eriksson, M.; Carlberg, M. In utero exposure to persistent organic pollutants in relation to testicular cancer risk. Int. J. Androl. 2006, 29, 228–234. [Google Scholar] [CrossRef] [PubMed]
- Richiardi, L.; Bellocco, R.; Adami, H.O.; Torrang, A.; Barlow, L.; Hakulinen, T.; Akre, O. Testicular cancer incidence in eight northern European countries: secular and recent trends. Cancer Epidemiol. Biomark. Prev. 2004, 13, 2157–2166. [Google Scholar]
- Walsh, T.J.; Dall’Era, M.A.; Croughan, M.S.; Carroll, P.R.; Turek, P.J. Prepubertal orchiopexy for cryptorchidism may be associated with lower risk of testicular cancer. J. Urol. 2007, 178, 1440–1446. [Google Scholar] [CrossRef] [PubMed]
- Alavanja, M.C.; Samanic, C.; Dosemeci, M.; Lubin, J.; Tarone, R.; Lynch, C.F.; Knott, C.; Thomas, K.; Hoppin, J.A.; Barker, J.; et al. Use of agricultural pesticides and prostate cancer risk in the Agricultural Health Study cohort. Am. J. Epidemiol. 2003, 157, 800–814. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, R.; Bonner, M.R.; Hoppin, J.A.; Alavanja, M.C. Phorate exposure and incidence of cancer in the agricultural health study. Environ. Health Perspect. 2006, 114, 1205–1209. [Google Scholar] [CrossRef] [PubMed]
- Finnström, N.; Bjelfman, C.; Söderström, T.; Smith, G.; Egevad, L.; Norlén, B.; Wolf, C.R.; Rane, A. Detection of cytochrome P450 mRNA transcripts in prostate samples by RT-PCR. Eur. J. Clin. Investig. 2001, 31, 880–886. [Google Scholar] [CrossRef]
- Sterling, K.M.; Cutrineo, K.R. Constitutive and inducible expression of cytochromes P4501A (CYP1A1 and CYP1A2) in normal prostate and prostate cancer cells. J. Cell Biol. 2004, 91, 423–429. [Google Scholar]
- Ho, S.M.; Tang, W.Y.; Belmonte, J.; Prins, G.S. Developmental exposure estradiol and bisphenol A (BPA) increases susceptibility to prostate carcinogenesis and epigenetically regulates phosphodisesterase type 4 variant (PDE4D4) in the rat prostate. Cancer Res. 2006, 66, 5624–5632. [Google Scholar] [CrossRef] [PubMed]
- Cillo, F.; de Eguileor, M.; Gandolfi, F.; Brevini, T. Aroclor-1254 affects mRNA polyadenylation, translational activation, cell morphology, and DNA integrity of rat primary prostate cells. Endocr. Relat. Cancer 2007, 14, 257–266. [Google Scholar] [CrossRef] [PubMed]
- Anway, M.; Skinner, M. Transgenerational effects of the endocrine disruptor vinclozolin on the prostate transcriptome and adult onset disease. Prostate 2008, 68, 515–529. [Google Scholar] [CrossRef]
- Nelson, W.G.; DeWeese, T.L.; DeMarzo, A.M. The diet, prostate inflammation, and the development of prostate cancer. Cancer Metastasis Rev. 2002, 21, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Masters, J.R.; Koberle, B. Curing metastatic cancer: Lessons from testicular germ-cell tumours. Nat. Rev. Cancer 2003, 3, 517–525. [Google Scholar] [CrossRef] [PubMed]
- Fraga, M.F.; Ballestar, E.; Paz, M.F.; Ropero, S.; Setien, F.; Ballestar, M.L.; Heine-Suñer, D.; Cigudosa, J.C.; Urioste, M.; Benitez, J.; et al. Epigenetic differences arise during the lifetime of monozygotic twins. Proc. Natl. Acad. Sci. USA 2005, 102, 10604–10609. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; Huang, R.; Ran, X.R.; Liu, H.Y.; Zhang, Y.; Dai, L.J.; Li, B. Three-generation experiment showed female C57BL/6J mice drink drainage canal water containing low level of TCDD-like activity causing high pup mortality. J. Toxicol. Sci. 2011, 36, 713–724. [Google Scholar] [CrossRef] [PubMed]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469. [Google Scholar] [CrossRef] [PubMed]
- Brannigan, R.E. Fertility preservation in adult male cancer patients. Cancer Treat. Res. 2007, 138, 28–49. [Google Scholar] [PubMed]
- Meirow, D.; Schenker, J.G. Cancer and male infertility. Hum. Reprod. 1995, 10, 2017–2022. [Google Scholar] [PubMed]
- Carroll, P.R.; Whitmore, W.F., Jr.; Herr, H.W.; Morse, M.J.; Sogani, P.C.; Bajorunas, D.; Fair, W.R.; Chaganti, R.S. Endocrine and exocrine profiles of men with testicular tumors before orchiectomy. J. Urol. 1987, 137, 420–423. [Google Scholar] [PubMed]
- Morrish, D.W.; Venner, P.M.; Siy, O.; Barron, G.; Bhardwaj, D.; Outhet, D. Mechanisms of endocrine dysfunction in patients with testicular cancer. J. Natl. Cancer Inst. 1990, 82, 412–418. [Google Scholar] [CrossRef] [PubMed]
- Parker, C.; Milosevic, M.; Panzarella, T.; Banerjee, D.; Jewett, M.; Catton, C.; Tew-George, B.; Gospodarowicz, M.; Warde, P. The prognostic significance of the tumour infiltrating lymphocyte count in stage I testicular seminoma managed by surveillance. Eur. J. Cancer 2002, 38, 2014–2019. [Google Scholar] [CrossRef] [PubMed]
- Ramos, E.J.; Suzuki, S.; Marks, D.; Inui, A.; Asakawa, A.; Meguid, M.M. Cancer anorexia-cachexia syndrome: Cytokines and neuropeptides. Curr. Opin. Clin. Nutr. Metab. Care 2004, 7, 427–434. [Google Scholar] [CrossRef] [PubMed]
- Foster, R.S.; Rubin, L.R.; McNulty, A.; Bihrle, R.; Donohue, J.P. Detection of antisperm-antibodies in patients with primary testicular cancer. Int. J. Androl. 1991, 14, 179–185. [Google Scholar] [CrossRef] [PubMed]
- Inui, A. Cancer anorexia-cachexia syndrome: Current issues in research and management. CA Cancer J. Clin. 2002, 52, 72–91. [Google Scholar] [CrossRef] [PubMed]
- Viviani, S.; Ragni, G.; Santoro, A.; Perotti, L.; Caccamo, E.; Negretti, E.; Valagussa, P.; Bonadonna, G. Testicular dysfunction in Hodgkin's disease before and after treatment. Eur. J. Cancer 1991, 27, 1389–1392. [Google Scholar] [CrossRef] [PubMed]
- Arai, Y.; Kawakita, M.; Okada, Y.; Yoshida, O. Sexuality and fertility in long-term survivors of testicular cancer. J. Clin. Oncol. 1997, 15, 1444–1448. [Google Scholar] [PubMed]
- Pryor, J.P.; Cameron, K.M.; Chilton, C.P.; Ford, T.F.; Parkinson, M.C.; Sinokrot, J.; Westwood, C.A. Carcinoma in situ in testicular biopsies from men presenting with infertility. Br. J. Urol. 1983, 55, 780–784. [Google Scholar] [CrossRef] [PubMed]
- Ezeh, U.I.; Turek, P.J.; Reijo, R.A.; Clark, A.T. Human embryonic stem cell genes OCT4, NANOG, STELLAR, and GDF3 are expressed in both seminoma and breast carcinoma. Cancer 2005, 104, 2255–2565. [Google Scholar] [CrossRef] [PubMed]
- Møller, H. Clues to the aetiology of testicular germ cell tumours from descriptive epidemiology. Eur. Urol. 1993, 23, 8–15. [Google Scholar] [PubMed]
- Jacobsen, R.; Bostofte, E.; Engholm, G.; Hansen, J.; Skakkebaek, N.E.; Møller, H. Fertility and offspring sex ratio of men who develop testicular cancer: A record linkage study. Hum. Reprod. 2000, 15, 1958–1961. [Google Scholar] [CrossRef] [PubMed]
- Petersen, P.M.; Giwercman, A.; Hansen, S.W.; Berthelsen, J.G.; Daugaard, G.; Rørth, M.; Skakkebaek, N.E. Impaired testicular function in patients with carcinoma-in-situ of the testis. J. Clin. Oncol. 1999, 17, 173–179. [Google Scholar] [PubMed]
- Jacobsen, R.; Bostofte, E.; Engholm, G.; Hansen, J.; Olsen, J.H.; Skakkebaek, N.E.; Moller, H. Risk of testicular cancer in men with abnormal semen characteristics: cohort study. BMJ 2000, 321, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Doria-Rose, V.P.; Biggs, M.L.; Weiss, N.S. Subfertility and the risk of testicular germ cell tumors (United States). Cancer Causes Control. 2005, 16, 651–656. [Google Scholar] [CrossRef] [PubMed]
- Strader, C.H.; Weiss, N.S.; Daling, J.R.; Karagas, M.R.; McKnight, B. Detection of testicular cancer in men presenting with infertility. Am. J. Epidemiol. 1988, 127, 1013–1018. [Google Scholar] [PubMed]
- Møller, H.; Skakkebaek, N.E. Risk of testicular cancer in subfertile men: Case-control study. BMJ 1999, 318, 559–562. [Google Scholar] [CrossRef] [PubMed]
- Pasqualotto, F.F.; Pasqualotto, E.B.; Agarwal, A.; Thomas, A.J., Jr. Fecundity and twinning rates as measures of fertility before diagnosis of germ-cell testicular cancer. Rev. Hosp. Clin. Fac. Med. Sao Paulo 2003, 58, 75–80. [Google Scholar] [PubMed]
- Richiardi, L.; Akre, O.; Montgomery, S.M.; Lambe, M.; Kvist, U.; Ekbom, A. Fecundity and twinning rates as measures of fertility before diagnosis of germ-cell testicular cancer. J. Natl. Cancer Inst. 2004, 96, 145–147. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.J.; Schembri, M.; Turek, P.J.; Chan, J.M.; Carroll, P.R.; Smith, J.F.; Eisenberg, M.L.; Van Den Eeden, S.K.; Croughan, M.S. Increased risk of high-grade prostate cancer among infertile men. Cancer 2010, 116, 2140–2147. [Google Scholar] [PubMed]
- Sonnenschein, C.; Olea, N.; Pasanen, M.E.; Soto, A.M. Negative controls of cell proliferation: Human prostate cancer cells and androgens. Cancer Res. 1989, 49, 3474–3481. [Google Scholar] [PubMed]
- Chen, H.Z.; Kirschenbaum, A.; Mandeli, J.; Hollander, V.P. The effect of dihydrotestosterone and culture conditions on proliferation of the human prostatic cancer cell line LNCaP. Steroids 1992, 57, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Pollard, M.; Luckert, P.H. Prevention and treatment of primary intestinal tumors in rats by piroxicam. Cancer Res. 1989, 49, 6471–6473. [Google Scholar] [PubMed]
- Roddam, A.W.; Allen, N.E.; Appleby, P.; Key, T.J. Endogenous sex hormones and prostate cancer: A collaborative analysis of 18 prospective studies. J. Natl. Cancer Inst. 2008, 100, 170–183. [Google Scholar] [CrossRef] [PubMed]
- Morgentaler, A.; Traish, A.M. Shifting the paradigm of testosterone and prostate cancer: The saturation model and the limits of androgen-dependent growth. Eur. Urol. 2009, 55, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Pavlovich, C.P.; King, P.; Goldstein, M.; Schlegel, P.N. Evidence of a treatable endocrinopathy in infertile men. J. Urol. 2001, 165, 837–841. [Google Scholar] [CrossRef] [PubMed]
- Thompson, I.M.; Goodman, P.J.; Tangen, C.M.; Lucia, M.S.; Miller, G.J.; Ford, L.G.; Lieber, M.M.; Cespedes, R.D.; Atkins, J.N.; Lippman, S.M.; et al. The influence of finasteride on the development of prostate cancer. N. Engl. J. Med. 2003, 349, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Andriole, G.L.; Bostwick, D.; Brawley, O.W.; Gomella, L.; Marberger, M.; Montorsi, F.; Pettaway, C.; Tammela, T.L.J.; Teloken, C.; Tindall, D.; et al. The effect of dutasteride on the usefulness of prostate specific antigen for the diagnosis of high grade and clinically relevant prostate cancer in men with a previous negative biopsy: Results from the REDUCE study. J. Urol. 2011, 185, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Jørgensen, K.T.; Pedersen, B.V.; Johansen, C.; Frisch, M. Fatherhood status and prostate cancer risk. Cancer 2008, 112, 919–923. [Google Scholar] [CrossRef] [PubMed]
- Giwercman, A.; Richiardi, L.; Kaijser, M.; Ekbom, A.; Akre, O. Reduced risk of prostate cancer in men who are childless as compared to those who have fathered a child: A population based case-control study. Int. J. Cancer 2005, 115, 994–997. [Google Scholar] [CrossRef] [PubMed]
- Negri, E.; Talamini, R.; Bosetti, C.; Montella, M.; Franceschi, S.; La Vecchia, C. Risk of prostate cancer in men who are childless. Int. J. Cancer 2006, 118, 786–787. [Google Scholar] [CrossRef] [PubMed]
- Harlap, S.; Friedlander, Y.; Barchana, M.; Calderon, R.; Deutsch, L.; Kleinhaus, K.R.; Perrin, M.C.; Tiram, E.; Yanetz, R.; Paltiel, O. Late fetal death in offspring and subsequent incidence of prostate cancer in fathers: the Jerusalem Perinatal Study cohort. Prostate 2007, 67, 989–998. [Google Scholar] [CrossRef] [PubMed]
- Wirén, S.M.; Drevin, L.I.; Carlsson, S.V.; Akre, O.; Holmberg, E.C.; Robinson, D.E.; Garmo, H.G.; Stattin, P.E. Fatherhood status and risk of prostate cancer: Nationwide, population-based case-control study. Int. J. Cancer 2013, 133, 937–943. [Google Scholar] [CrossRef] [PubMed]
- Drabovich, A.P.; Saraon, P.; Jarvi, K.; Diamandis, E.P. Seminal plasma as a diagnostic fluid for male reproductive system disorders. Nat. Rev. Urol. 2014, 11, 278–288. [Google Scholar] [CrossRef] [PubMed]
- Thomas, M.; Lieberman, J.; Lal, A. Desperately seeking microRNA targets. Nat. Struct. Mol. Biol. 2010, 17, 1169–1174. [Google Scholar] [CrossRef] [PubMed]
- Stark, A.; Bushati, N.; Jan, C.H.; Kheradpour, P.; Hodges, E.; Brennecke, J.; Bartel, D.P.; Cohen, S.M.; Kellis, M. A single Hox locus in Drosophila produces functional microRNAs from opposite DNA strands. Genes Dev. 2008, 22, 8–13. [Google Scholar] [CrossRef] [PubMed]
- Rajender, S.; Meador, C.; Agarwal, A. Small RNA in spermatogenesis and male infertility. Front. Biosci. 2012, 4, 1266–1274. [Google Scholar] [CrossRef]
- McIver, S.C.; Roman, S.D.; Nixon, B.; McLaughlin, E.A. miRNA and mammalian male germ cells. Hum. Reprod. Update 2012, 18, 44–59. [Google Scholar] [CrossRef] [PubMed]
- Bjork, J.K.; Sandqvist, A.; Elsing, A.N.; Kotaja, N.; Sistonen, L. miR-18, a member of Oncomir-1, targets heat shock transcription factor 2 in spermatogenesis. Development 2010, 137, 3177–3184. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Raabe, T.; Hecht, N.B. MicroRNA Mirn122a reduces expression of the posttranscriptionally regulated germ cell transition protein 2 (Tnp2) messenger RNA (mRNA) by mRNA cleavage. Biol. Reprod. 2005, 73, 427–433. [Google Scholar] [CrossRef] [PubMed]
- Bouhallier, F.; Allioli, N.; Lavial, F.; Chalmel, F.; Perrard, M.H.; Durand, P.; Samarut, J.; Pain, B.; Rouault, J.P. Role of miR-34c microRNA in the late steps of spermatogenesis. RNA 2010, 16, 720–731. [Google Scholar] [CrossRef] [PubMed]
- Voorhoeve, P.M.; le Sage, C.; Schrier, M.; Gillis, A.J.; Stoop, H.; Nagel, R.; Liu, Y.P.; van Duijse, J.; Drost, J.; Griekspoor, A.; et al. A genetic screen implicates miRNA-372 and miRNA-373 as oncogenes in testicular germ cell tumors. Cell 2006, 124, 1169–1181. [Google Scholar] [CrossRef] [PubMed]
- Gillis, A.J.; Stoop, H.J.; Hersmus, R.; Oosterhuis, J.W.; Sun, Y.; Chen, C.; Guenther, S.; Sherlock, J.; Veltman, I.; Baeten, J.; et al. High-throughput microRNAome analysis in human germ cell tumours. J. Pathol. 2007, 213, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Bezan, A.; Gerger, A.; Pichler, M. MicroRNAs in testicular cancer: Implications for pathogenesis, diagnosis, prognosis and therapy. Anticancer Res. 2014, 34, 2709–2713. [Google Scholar] [PubMed]
- Lian, J.; Tian, H.; Liu, L.; Zhang, X.S.; Li, W.Q.; Deng, Y.M.; Yao, G.D.; Yin, M.M.; Sun, F. Down-regulation of microRNA-383 is associated with male infertility and promotes testicular embryonal carcinoma cell proliferation by targeting IRF1. Cell Death Dis. 2010, 1, e94. [Google Scholar] [CrossRef] [PubMed]
- Sita-Lumsden, A.; Dart, D.A.; Waxman, J.; Bevan, C.L. Circulating microRNAs as potential new biomarkers for prostate cancer. Br. J. Cancer 2013, 108, 1925–1930. [Google Scholar] [CrossRef] [PubMed]
- Jackson, B.L.; Grabowska, A.; Ratan, H.L. MicroRNA in prostate cancer: functional importance and potential as circulating biomarkers. BMC Cancer 2014, 14, 930. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.B.; Xue, L.; Ma, A.H.; Tepper, C.G.; Kung, H.J.; White, R.W. miR-125b promotes growth of prostate cancer xenograft tumor through targeting Pro-apoptotic genes. Prostate 2011, 71, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.Y.; Hwang, C.I.; Corney, D.C.; Flesken-Nikitin, A.; Jiang, L.; Oner, G.M.; Munroe, R.J.; Schimenti, J.C.; Hermeking, H.; Nikitin, A.Y. miR-34 cooperates with p53 in suppression of prostate cancer by joint regulation of stem cell compartment. Cell Rep. 2014, 6, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Hsu, T.I.; Hsu, C.H.; Lee, K.H.; Lin, J.T.; Chen, C.H.; Chang, K.C.; Su, C.Y.J.; Hsiao, M.; Lu, P.J. MicroRNA-18a is elevated in prostate cancer and promotes tumorigenesis through suppressing STK4 in vitro and in vivo. Oncogenesis 2014, 3, e99. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Feng, Y.; Coukos, G.; Zhang, L. Therapeutic microRNA strategies in human cancer. AAPS J. 2009, 11, 747–757. [Google Scholar] [CrossRef] [PubMed]
- Bader, A.G. miR-34—A microRNA replacement therapy is headed to the clinic. Front. Genet. 2012, 3, 120. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tvrda, E.; Agarwal, A.; Alkuhaimi, N. Male Reproductive Cancers and Infertility: A Mutual Relationship. Int. J. Mol. Sci. 2015, 16, 7230-7260. https://doi.org/10.3390/ijms16047230
Tvrda E, Agarwal A, Alkuhaimi N. Male Reproductive Cancers and Infertility: A Mutual Relationship. International Journal of Molecular Sciences. 2015; 16(4):7230-7260. https://doi.org/10.3390/ijms16047230
Chicago/Turabian StyleTvrda, Eva, Ashok Agarwal, and Nawaf Alkuhaimi. 2015. "Male Reproductive Cancers and Infertility: A Mutual Relationship" International Journal of Molecular Sciences 16, no. 4: 7230-7260. https://doi.org/10.3390/ijms16047230
APA StyleTvrda, E., Agarwal, A., & Alkuhaimi, N. (2015). Male Reproductive Cancers and Infertility: A Mutual Relationship. International Journal of Molecular Sciences, 16(4), 7230-7260. https://doi.org/10.3390/ijms16047230








