Sex-Specific Diurnal Immobility Induced by Forced Swim Test in Wild Type and Clock Gene Deficient Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Results
2.1.1. Sex-Specific Circadian Rhythms in the Duration of Immobility in Wild Type Mice
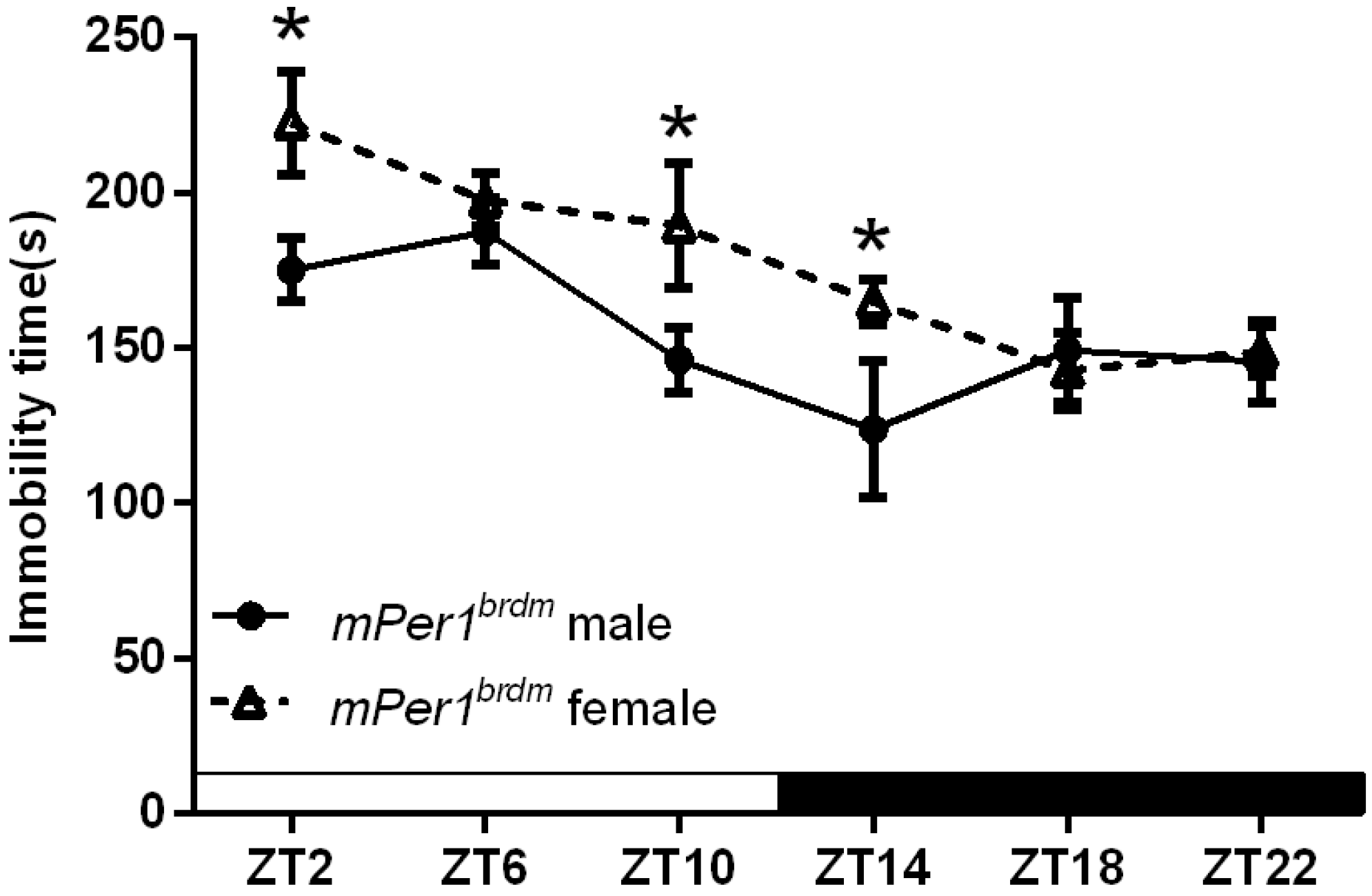
2.1.2. Circadian Rhythms in the Duration of Immobility Were Changed in Female, but not Male Per1Brdm1 Mice
| Parameter | Type of Mice | |||
|---|---|---|---|---|
| Wild Type | Per1Brdm1 | |||
| Male | Female | Male | Female | |
| Mesor | 167.19 ± 5.66 | 169.88 ± 12.46 | 154.42 ± 10.29 | 177.55 ± 11.73 |
| Amplitude | 40.33 ± 6.56 | 29.59 ± 1.88 | 27.36 ± 5.39 | 35.91 ± 2.58 |
| Peak time | 10.08 ± 1.43 | 15.17 ± 0.36 # | 6.22 ± 2.55 | 5.33 ± 0.24 |
| Nadir time | 18.42 ± 2.08 | 3.17 ± 0.36 # | 15.22 ± 0.46 | 17.33 ± 0.24 |
| Cosinor p value | F = 5.24 * | F = 11.39 * | F = 7.35 * | F = 7.82 * |
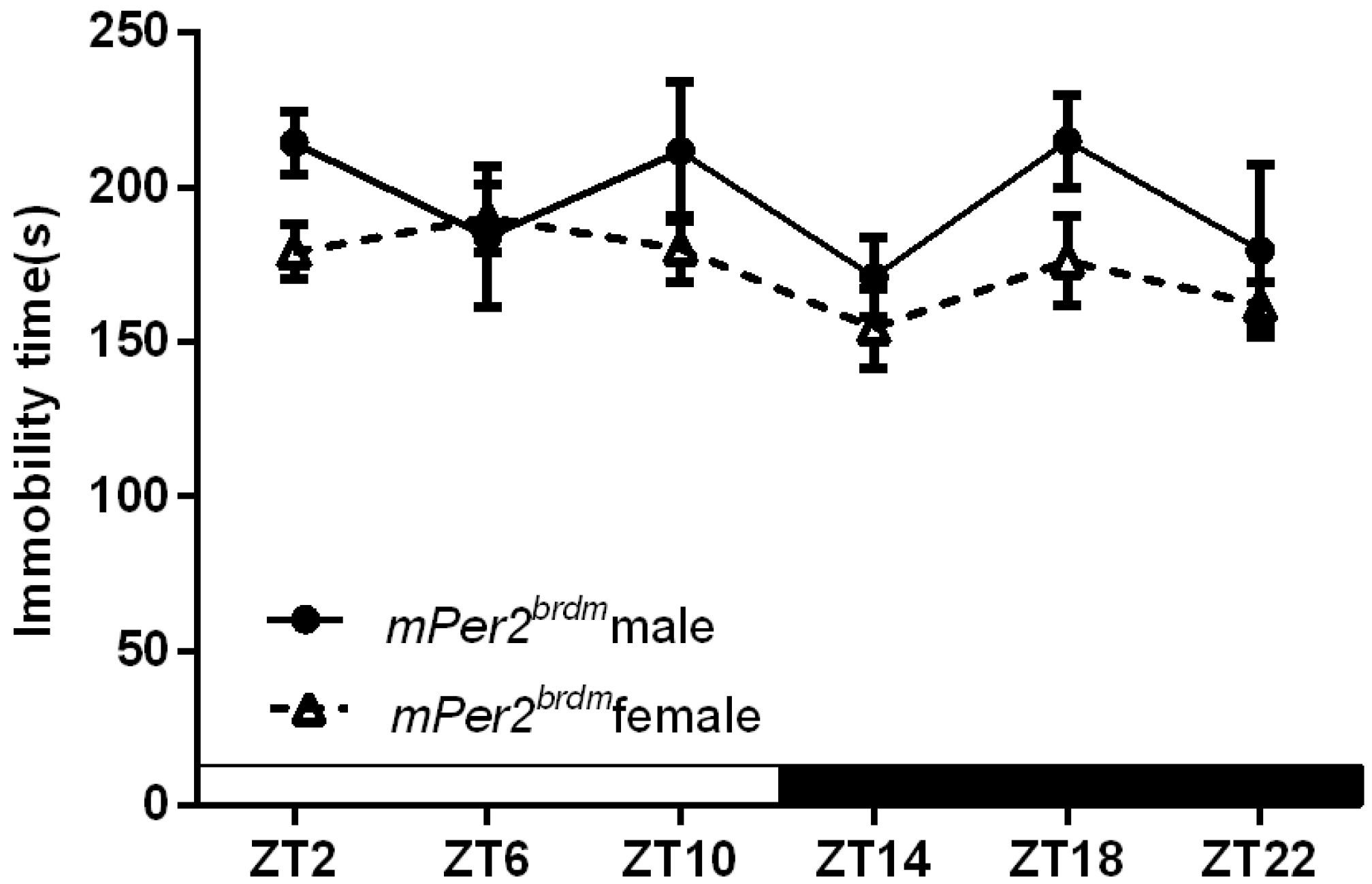
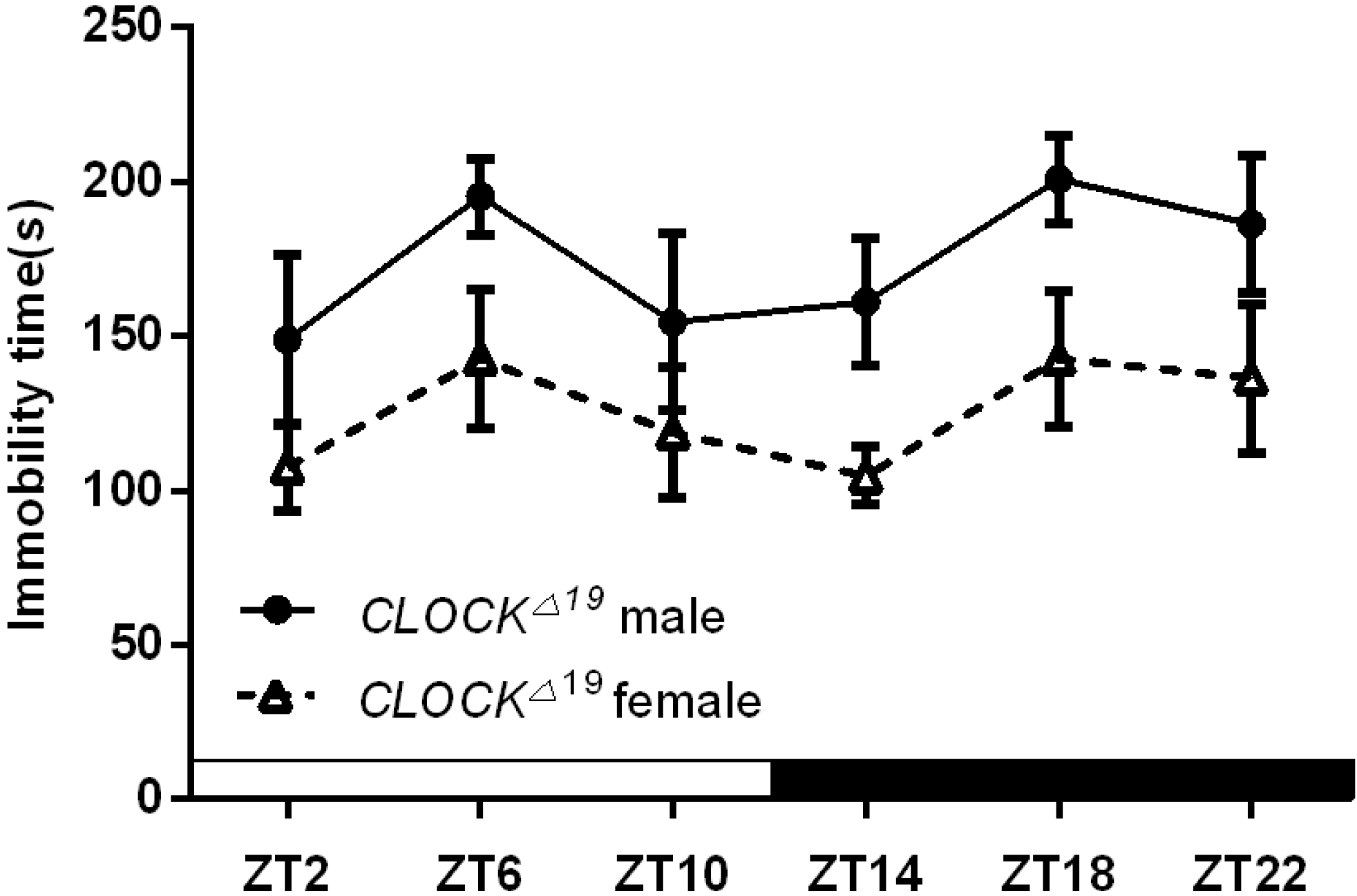
2.1.3. Per2Brdm1 and ClockΔ19 Mice Lost the Circadian Rhythm in Their Duration of Immobility
| Parameter | Type of Mice | |||
|---|---|---|---|---|
| Per2Brdm1 | ClockΔ19 | |||
| Male | Female | Male | Female | |
| Mesor | 195.89 ± 10.42 | 175.21 ± 5.07 | 174.36 ± 8.65 | 125.35 ± 5.88 |
| Amplitude | 30.26 ± 5.41 | 25.18 ± 3.62 | 59.53 ± 5.21 | 28.86 ± 5.71 |
| Cosinor p value | F = 0.06 | F = 2.26 | F = 0.63 | F = 0.66 |
2.1.4. Sex-Specific Differences in the Duration of Immobility within Each Genotype of Mice

2.2. Discussion
3. Materials and Methods
3.1. Subjects
3.2. Forced Swim Test
3.3. Statistical Analyses
Acknowledgments
Author Contributions
Conflicts of Interest
References
- McClung, C.A. How might circadian rhythms control mood? Let me count the ways. Biol. Psychiatry 2013, 74, 242–249. [Google Scholar] [CrossRef]
- Cornelissen, G.; Watson, D.; Mitsutake, G.; Fišer, B.; Siegelová, J.; Dušek, J.; Vohlídalová, I.; Svaèinová, H.; Halberg, F. Mapping of circaseptan and circadian changes in mood. Scr. Med. 2005, 78, 89–98. [Google Scholar]
- Benedetti, F.; Terman, M. Much ado about…a moody clock. Biol. Psychiatry 2013, 74, 236–237. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, M.J.; Welsh, D.K. Cellular circadian clocks in mood disorders. J. Biol. Rhythm. 2012, 27, 339–352. [Google Scholar] [CrossRef]
- King, D.P.; Takahashi, J.S. Molecular genetics of circadian rhythms in mammals. Annu. Rev. Neurosci. 2000, 23, 713–742. [Google Scholar] [CrossRef] [PubMed]
- Landgraf, D.; McCarthy, M.J.; Welsh, D.K. The role of the circadian clock in animal models of mood disorders. Behav. Neurosci. 2014, 128, 344–359. [Google Scholar] [CrossRef] [PubMed]
- Johansson, C.; Willeit, M.; Smedh, C.; Ekholm, J.; Paunio, T.; Kieseppä, T.; Lichtermann, D.; Praschak-Rieder, N.; Neumeister, A.; Nilsson, L.G.; et al. Circadian clock-related polymorphisms in seasonal affective disorder and their relevance to diurnal preference. Neuropsychopharmacology 2003, 28, 734–739. [Google Scholar]
- Partonen, T.; Treutlein, J.; Alpman, A.; Frank, J.; Johansson, C.; Depner, M.; Aron, L.; Rietschel, M.; Wellek, S.; Soronen, P.; et al. Three circadian clock genes Per2, Arntl, and Npas2 contribute to winter depression. Ann. Med. 2007, 39, 229–238. [Google Scholar]
- Roybal, K.; Theobold, D.; Graham, A.; DiNieri, J.A.; Russo, S.J.; Krishnan, V.; Chakravarty, S.; Peevey, J.; Oehrlein, N.; Birnbaum, S.; et al. Mania-like behavior induced by disruption of CLOCK. Proc. Natl. Acad. Sci. USA 2007, 104, 6406–6411. [Google Scholar] [CrossRef]
- Hampp, G.; Ripperger, J.A.; Houben, T.; Schmutz, I.; Blex, C.; Perreau-Lenz, S.; Brunk, I.; Spanagel, R.; Ahnert-Hilger, G.; Meijer, J.H.; et al. Regulation of monoamine oxidase a by circadian-clock components implies clock influence on mood. Curr. Biol. 2008, 18, 678–683. [Google Scholar]
- Spencer, S.; Falcon, E.; Kumar, J.; Krishnan, V.; Mukherjee, S.; Birnbaum, S.G.; McClung, C.A. Circadian genes Period 1 and Period 2 in the nucleus accumbens regulate anxiety-related behavior. Eur. J. Neurosci. 2013, 37, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Bogdanova, O.V.; Kanekar, S.; D'Anci, K.E.; Renshaw, P.F. Factors influencing behavior in the forced swim test. Physiol. Behav. 2013, 118, 227–239. [Google Scholar] [CrossRef] [PubMed]
- Pitychoutis, P.M.; Sanoudou, D.; Papandreou, M.; Nasias, D.; Kouskou, M.; Tomlinson, C.R.; Tsonis, P.A.; Papadopoulou-Daifoti, Z. Forced swim test induces divergent global transcriptomic alterations in the hippocampus of high vs. low novelty-seeker rats. Hum. Genomics 2014, 8, 4. [Google Scholar] [CrossRef]
- Kessler, R.C. Epidemiology of women and depression. J. Affect. Disord. 2003, 74, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Stone, A.A.; Schwartz, J.E.; Schkade, D.; Schwarz, N.; Krueger, A.; Kahneman, D. A population approach to the study of emotion: Diurnal rhythms of a working day examined with the day reconstruction method. Emotion 2006, 6, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Uz, T.; Ahmed, R.; Akhisaroglu, M.; Kurtuncu, M.; Imbesi, M.; Dirim Arslan, A.; Manev, H. Effect of fluoxetine and cocaine on the expression of clock genes in the mouse hippocampus and striatum. Neuroscience 2005, 134, 1309–1316. [Google Scholar] [CrossRef] [PubMed]
- Serretti, A.; Cusin, C.; Benedetti, F.; Mandelli, L.; Pirovano, A.; Zanardi, R.; Colombo, C.; Smeraldi, E. Insomnia improvement during antidepressant treatment and CLOCK gene polymorphism. Am. J. Med. Genet. B 2005, 137B, 36–39. [Google Scholar] [CrossRef]
- Benedetti, F.; Serretti, A.; Colombo, C.; Barbini, B.; Lorenzi, C.; Campori, E.; Smeraldi, E. Influence of CLOCK gene polymorphism on circadian mood fluctuation and illness recurrence in bipolar depression. Am. J. Med. Genet. Part B 2003, 123, 23–26. [Google Scholar] [CrossRef]
- Bae, K.; Jin, X.; Maywood, E.S.; Hastings, M.H.; Reppert, S.M.; Weaver, D.R. Differential functions of mPer1, mPer2, and mPer3 in the SCN circadian clock. Neuron 2001, 30, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Nader, N.; Chrousos, G.P.; Kino, T. Interactions of the circadian CLOCK system and the HPA axis. Trends Endocrinol. Metab. 2010, 21, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Kino, T. Circadian rhythms of glucocorticoid hormone actions in target tissues: Potential clinical implications. Sci. Signal. 2012, 5, pt4. [Google Scholar] [PubMed]
- Palanza, P. Animal models of anxiety and depression: How are females different. Neurosci. Biobehav. Rev. 2001, 25, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Iwahana, E.; Karatsoreos, I.; Shibata, S.; Silver, R. Gonadectomy reveals sex differences in circadian rhythms and suprachiasmatic nucleus androgen receptors in mice. Horm. Behav. 2008, 53, 422–430. [Google Scholar] [CrossRef] [PubMed]
- Verma, P.; Hellemans, K.G.C.; Choi, F.Y.; Yu, W.; Weinberg, J. Circadian phase and sex effects on depressive/anxiety-like behaviors and HPA axis responses to acute stress. Physiol. Behav. 2010, 99, 276–285. [Google Scholar] [CrossRef] [PubMed]
- Miller, B.H.; Olson, S.L.; Turek, F.W.; Levine, J.E.; Horton, T.H.; Takahashi, J.S. Circadian clock mutation disrupts estrous cyclicity and maintenance of pregnancy. Curr. Biol. 2004, 14, 1367–1373. [Google Scholar] [CrossRef] [PubMed]
- Pilorz, V.; Steinlechner, S. Low reproductive success in Per1 and Per2 mutant mouse females due to accelerated ageing. Reproduction 2008, 135, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Perrin, J.S.; Segall, L.A.; Harbour, V.L.; Woodside, B.; Amir, S. The expression of the clock protein PER2 in the limbic forebrain is modulated by the estrous cycle. Proc. Natl. Acad. Sci. USA 2006, 103, 5591–5596. [Google Scholar] [CrossRef] [PubMed]
- Consoli, D.; Fedotova, J.; Micale, V.; Sapronov, N.S.; Drago, F. Stressors affect the response of male and female rats to clomipramine in a model of behavioral despair (forced swim test). Eur. J. Pharmacol. 2005, 520, 100–107. [Google Scholar] [CrossRef] [PubMed]
- Kokras, N.; Dalla, C.; Sideris, A.C.; Dendi, A.; Mikail, H.G.; Antoniou, K.; Papadopoulou-Daifoti, Z. Behavioral sexual dimorphism in models of anxiety and depression due to changes in HPA axis activity. Neuropharmacology 2012, 62, 436–445. [Google Scholar] [CrossRef] [PubMed]
- Porsolt, R.D.; Le Pichon, M.; Jalfre, M. Depression: A new animal model sensitive to antidepressant treatments. Nature 1977, 266, 730–732. [Google Scholar] [CrossRef] [PubMed]
- Hirani, K.; Khisti, R.T.; Chopde, C.T. Behavioral action of ethanol in Porsolt’s forced swim test: Modulation by 3 alpha-hydroxy-5 alpha-pregnan-20-one. Neuropharmacology 2002, 43, 1339–1350. [Google Scholar] [CrossRef] [PubMed]
- Mojon, A.; Fernandez, J.R.; Hermida, R.C. Chronolab: An interactive software package for chronobiologic time series analysis written for the Macintosh computer. Chronobiol. Int. 1992, 9, 403–412. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, N.; Xu, Y.; Chen, X.; Duan, Q.; Zhao, M. Sex-Specific Diurnal Immobility Induced by Forced Swim Test in Wild Type and Clock Gene Deficient Mice. Int. J. Mol. Sci. 2015, 16, 6831-6841. https://doi.org/10.3390/ijms16046831
Li N, Xu Y, Chen X, Duan Q, Zhao M. Sex-Specific Diurnal Immobility Induced by Forced Swim Test in Wild Type and Clock Gene Deficient Mice. International Journal of Molecular Sciences. 2015; 16(4):6831-6841. https://doi.org/10.3390/ijms16046831
Chicago/Turabian StyleLi, Ningyue, Yanhua Xu, Xiaojuan Chen, Qing Duan, and Mei Zhao. 2015. "Sex-Specific Diurnal Immobility Induced by Forced Swim Test in Wild Type and Clock Gene Deficient Mice" International Journal of Molecular Sciences 16, no. 4: 6831-6841. https://doi.org/10.3390/ijms16046831
APA StyleLi, N., Xu, Y., Chen, X., Duan, Q., & Zhao, M. (2015). Sex-Specific Diurnal Immobility Induced by Forced Swim Test in Wild Type and Clock Gene Deficient Mice. International Journal of Molecular Sciences, 16(4), 6831-6841. https://doi.org/10.3390/ijms16046831





