Coronary Artery Calcium Screening: Does it Perform Better than Other Cardiovascular Risk Stratification Tools?
Abstract
:1. Introduction
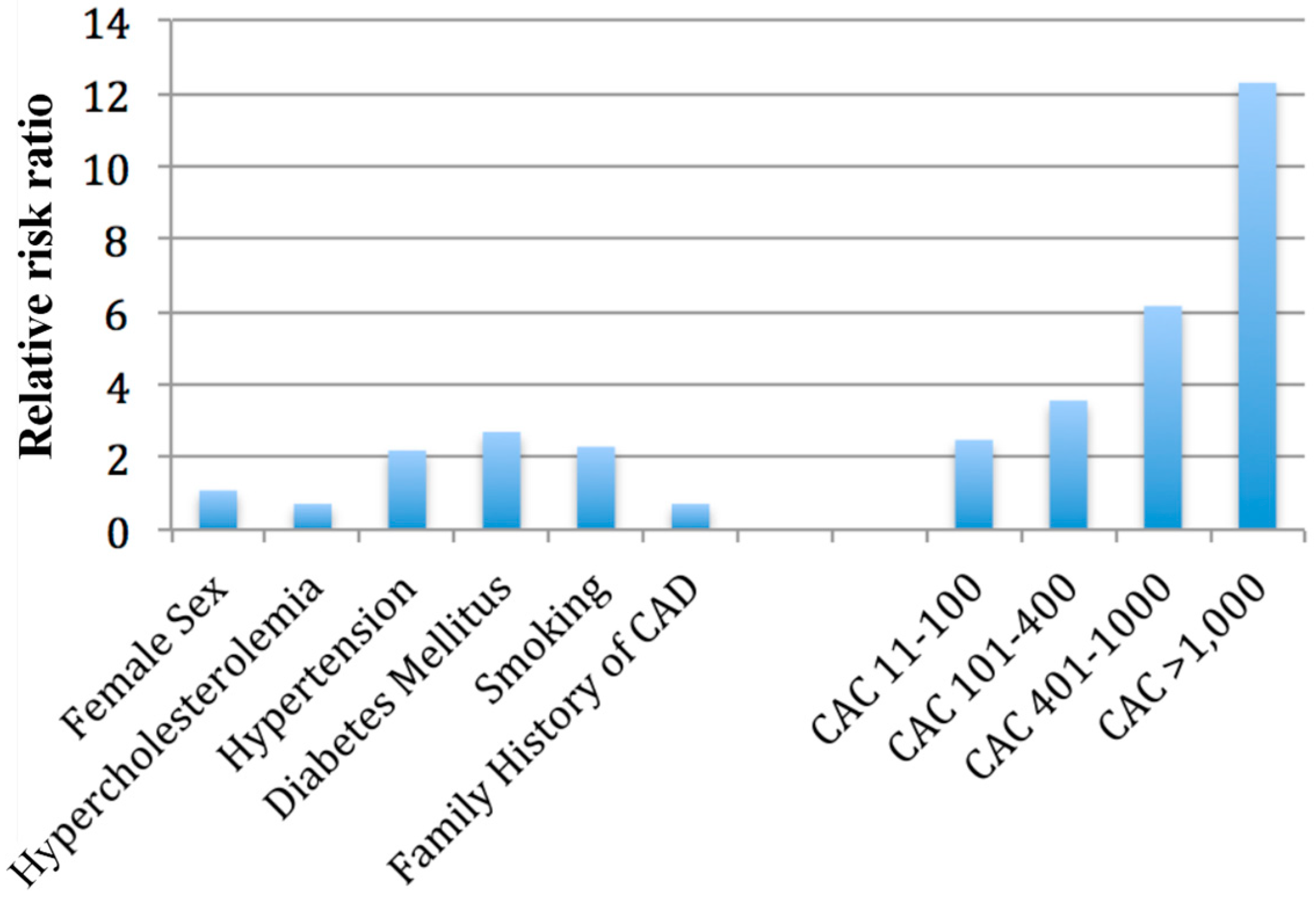
2. Framingham Risk Score and Coronary Artery Calcium
| Author | Year | Total No. of Participants | Participants with CAC Score 0 | Event Rate | Follow up Period |
|---|---|---|---|---|---|
| Blaha et al. [21] | 2009 | 44,052 | 19,898 | 0.87/1000 person-years | Mean follow-up of 5.6 ± 2.6 years |
| Arad et al. [22] | 2005 | 4613 | 1504 | 1/1000 person-years | 4.3 years |
| Taylor et al. [9] | 2005 | 2000 | 1263 | 0.6/1000 person-years | Mean follow-up of 3.0 ±1.4 years |
| Budoff et al. [23] | 2007 | 25,253 | 11,046 | 0.6/1000 person-years | Mean follow-up of 6.8 ± 3 years |
| Detrano et al. [20] | 2009 | 6722 | 3409 | 0.6/1000 person-years | 3.7 years |
| Shaw et al. [4] | 2003 | 10,377 | 5067 | 1.5 events/1000 person-years | Mean follow-up of 5.0 years |
| LaMonte et al. [24] | 2005 | 10,746 | 2692 | 1.6 events/1000 person-years | 3.5 years |
3. Reynolds Risk Score and Coronary Artery Calcium
4. Carotid Intima Media Thickness and Coronary Artery Calcium
5. C-Reactive Protein and Coronary Artery Calcium
6. Coronary Artery Calcium and Other Imaging Parameters for Risk Prediction
7. Conclusions
Author Contributions
Conflicts of Interest
References
- Lloyd-Jones, D.; Adams, R.; Carnethon, M.; de Simone, G.; Ferguson, T.B.; Flegal, K.; Ford, E.; Furie, K.; Go, A.; Greenlund, K.; et al. Heart disease and stroke statistics—2009 Update: A report from the American Heart Association Statistics Committee and Stroke Statistics Subcommittee. Circulation 2009, 119, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Go, A.S.; Mozaffarian, D.; Roger, V.L.; Benjamin, E.J.; Berry, J.D.; Blaha, M.J.; Dai, S.; Ford, E.S.; Fox, C.S.; Franco, S.; et al. Heart disease and stroke statistics—2014 Update: A report from the American Heart Association. Circulation 2014, 129, e28–e292. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M.; Pasternak, R.; Greenland, P.; Smith, S., Jr.; Fuster, V. Assessment of cardiovascular risk by use of multiple-risk-factor assessment equations: A statement for healthcare professionals from the American Heart Association and the American College of Cardiology. Circulation 1999, 100, 1481–1492. [Google Scholar] [CrossRef] [PubMed]
- Shaw, L.J.; Raggi, P.; Schisterman, E.; Berman, D.S.; Callister, T.Q. Prognostic value of cardiac risk factors and coronary artery calcium screening for all-cause mortality. Radiology 2003, 228, 826–833. [Google Scholar] [CrossRef] [PubMed]
- Greenland, P.; LaBree, L.; Azen, S.P.; Doherty, T.M.; Detrano, R.C. Coronary artery calcium score combined with Framingham score for risk prediction in asymptomatic individuals. JAMA 2004, 291, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Greenland, P.; Alpert, J.S.; Beller, G.A.; Benjamin, E.J.; Budoff, M.J.; Fayad, Z.A.; Foster, E.; Hlatky, M.A.; Hodgson, J.M.; Kushner, F.G.; et al. 2010 ACCF/AHA guideline for assessment of cardiovascular risk in asymptomatic adults: A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. J. Am. Coll. Cardiol. 2010, 56, e50–e103. [Google Scholar] [CrossRef] [PubMed]
- Okwuosa, T.M.; Greenland, P.; Ning, H.; Liu, K.; Bild, D.E.; Burke, G.L.; Eng, J.; Lloyd-Jones, D.M. Distribution of coronary artery calcium scores by Framingham 10-year risk strata in the MESA (Multi-Ethnic Study of Atherosclerosis) potential implications for coronary risk assessment. J. Am. Coll. Cardiol. 2011, 57, 1838–1845. [Google Scholar] [CrossRef] [PubMed]
- Okwuosa, T.M.; Greenland, P.; Burke, G.L.; Eng, J.; Cushman, M.; Michos, E.D.; Ning, H.; Lloyd-Jones, D.M. Prediction of coronary artery calcium progression in individuals with low Framingham Risk Score: The Multi-Ethnic Study of Atherosclerosis. JACC Cardiovasc. Imaging 2012, 5, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.J.; Bindeman, J.; Feuerstein, I.; Cao, F.; Brazaitis, M.; O’Malley, P.G. Coronary calcium independently predicts incident premature coronary heart disease over measured cardiovascular risk factors: Mean three-year outcomes in the Prospective Army Coronary Calcium (PACC) project. J. Am. Coll. Cardiol. 2005, 46, 807–814. [Google Scholar] [CrossRef] [PubMed]
- Ford, E.S.; Giles, W.H.; Mokdad, A.H. The distribution of 10-year risk for coronary heart disease among US adults: Findings from the National Health and Nutrition Examination Survey III. J. Am. Coll. Cardiol. 2004, 43, 1791–1796. [Google Scholar] [CrossRef] [PubMed]
- Berry, J.D.; Lloyd-Jones, D.M.; Garside, D.B.; Greenland, P. Framingham risk score and prediction of coronary heart disease death in young men. Am. Heart J. 2007, 154, 80–86. [Google Scholar] [CrossRef] [PubMed]
- Akosah, K.O.; Schaper, A.; Cogbill, C.; Schoenfeld, P. Preventing myocardial infarction in the young adult in the first place: How do the National Cholesterol Education Panel III guidelines perform? J. Am. Coll. Cardiol. 2003, 41, 1475–1479. [Google Scholar] [CrossRef] [PubMed]
- Lakoski, S.G.; Greenland, P.; Wong, N.D.; Schreiner, P.J.; Herrington, D.M.; Kronmal, R.A.; Liu, K.; Blumenthal, R.S. Coronary artery calcium scores and risk for cardiovascular events in women classified as “low risk” based on Framingham risk score: The Multi-Ethnic Study of Atherosclerosis (MESA). Arch. Intern. Med. 2007, 167, 2437–2442. [Google Scholar] [CrossRef] [PubMed]
- Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adult. Executive summary of the third report of the National Cholesterol Education Program (NCEP) expert panel on detection, evaluation, and treatment of high blood cholesterol in adults (Adult Treatment Panel III). JAMA 2001, 285, 2486–2497.
- Elias-Smale, S.E.; Proenca, R.V.; Koller, M.T.; Kavousi, M.; van Rooij, F.J.; Hunink, M.G.; Steyerberg, E.W.; Hofman, A.; Oudkerk, M.; Witteman, J.C. Coronary calcium score improves classification of coronary heart disease risk in the elderly: The Rotterdam study. J. Am. Coll. Cardiol. 2010, 56, 1407–1414. [Google Scholar] [CrossRef] [PubMed]
- Nasir, K.; Budoff, M.J.; Wong, N.D.; Scheuner, M.; Herrington, D.; Arnett, D.K.; Szklo, M.; Greenland, P.; Blumenthal, R.S. Family history of premature coronary heart disease and coronary artery calcification: Multi-Ethnic Study of Atherosclerosis (MESA). Circulation 2007, 116, 619–626. [Google Scholar] [CrossRef] [PubMed]
- Mulders, T.A.; Sivapalaratnam, S.; Stroes, E.S.; Kastelein, J.J.; Guerci, A.D.; Pinto-Sietsma, S.J. Asymptomatic individuals with a positive family history for premature coronary artery disease and elevated coronary calcium scores benefit from statin treatment: A post hoc analysis from the St. Francis Heart Study. JACC Cardiovasc. Imaging 2012, 5, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Blaha, M.J.; Sharma, K.; Rivera, J.; Budoff, M.J.; Blankstein, R.; Al-Mallah, M.; Wong, N.D.; Shaw, L.; Carr, J.; et al. Family history of coronary heart disease and the incidence and progression of coronary artery calcification: Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis 2014, 232, 369–376. [Google Scholar] [CrossRef] [PubMed]
- Polonsky, T.S.; McClelland, R.L.; Jorgensen, N.W.; Bild, D.E.; Burke, G.L.; Guerci, A.D.; Greenland, P. Coronary artery calcium score and risk classification for coronary heart disease prediction. JAMA 2010, 303, 1610–1016. [Google Scholar] [CrossRef] [PubMed]
- Detrano, R.; Guerci, A.D.; Carr, J.J.; Bild, D.E.; Burke, G.; Folsom, A.R.; Liu, K.; Shea, S.; Szklo, M.; Bluemke, D.A.; et al. Coronary calcium as a predictor of coronary events in four racial or ethnic groups. New Engl. J. Med. 2008, 358, 1336–1345. [Google Scholar] [CrossRef] [PubMed]
- Blaha, M.; Budoff, M.J.; Shaw, L.J.; Khosa, F.; Rumberger, J.A.; Berman, D.; Callister, T.; Raggi, P.; Blumenthal, R.S.; Nasir, K. Absence of coronary artery calcification and all-cause mortality. JACC Cardiovasc. Imaging 2009, 2, 692–700. [Google Scholar] [CrossRef] [PubMed]
- Arad, Y.; Goodman, K.J.; Roth, M.; Newstein, D.; Guerci, A.D. Coronary calcification, coronary disease risk factors, C-reactive protein, and atherosclerotic cardiovascular disease events: The St. Francis Heart Study. J. Am. Coll. Cardiol. 2005, 46, 158–165. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Shaw, L.J.; Liu, S.T.; Weinstein, S.R.; Mosler, T.P.; Tseng, P.H.; Flores, F.R.; Callister, T.Q.; Raggi, P.; Berman, D.S. Long-term prognosis associated with coronary calcification: Observations from a registry of 25,253 patients. J. Am. Coll. Cardiol. 2007, 49, 1860–1870. [Google Scholar] [CrossRef] [PubMed]
- LaMonte, M.J.; FitzGerald, S.J.; Church, T.S.; Barlow, C.E.; Radford, N.B.; Levine, B.D.; Pippin, J.J.; Gibbons, L.W.; Blair, S.N.; Nichaman, M.Z. Coronary artery calcium score and coronary heart disease events in a large cohort of asymptomatic men and women. Am. J. Epidemiol. 2005, 162, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Hermann, D.M.; Gronewold, J.; Lehmann, N.; Moebus, S.; Jockel, K.H.; Bauer, M.; Erbel, R. Coronary artery calcification is an independent stroke predictor in the general population. Stroke 2013, 44, 1008–1013. [Google Scholar] [CrossRef] [PubMed]
- O’Neal, W.T.; Efird, J.T.; Dawood, F.Z.; Yeboah, J.; Alonso, A.; Heckbert, S.R.; Soliman, E.Z. Coronary artery calcium and risk of atrial fibrillation (from the multi-ethnic study of atherosclerosis). Am. J. Cardiol. 2014, 114, 1707–1712. [Google Scholar] [CrossRef] [PubMed]
- Goff, D.C., Jr.; Lloyd-Jones, D.M.; Bennett, G.; Coady, S.; D’Agostino, R.B.; Gibbons, R.; Greenland, P.; Lackland, D.T.; Levy, D.; O’Donnell, C.J.; et al. 2013 ACC/AHA guideline on the assessment of cardiovascular risk: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 2014, 129, S49–S73. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Buring, J.E.; Rifai, N.; Cook, N.R. Development and validation of improved algorithms for the assessment of global cardiovascular risk in women: The Reynolds Risk Score. JAMA 2007, 297, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Paynter, N.P.; Rifai, N.; Gaziano, J.M.; Cook, N.R. C-reactive protein and parental history improve global cardiovascular risk prediction: The Reynolds Risk Score for men. Circulation 2008, 118, 2243–2251. [Google Scholar] [CrossRef] [PubMed]
- DeFilippis, A.P.; Blaha, M.J.; Ndumele, C.E.; Budoff, M.J.; Lloyd-Jones, D.M.; McClelland, R.L.; Lakoski, S.G.; Cushman, M.; Wong, N.D.; Blumenthal, R.S.; et al. The association of Framingham and Reynolds Risk Scores with incidence and progression of coronary artery calcification in MESA (Multi-Ethnic Study of Atherosclerosis). J. Am. Coll. Cardiol. 2011, 58, 2076–2083. [Google Scholar] [CrossRef] [PubMed]
- Desai, MY; Halliburton, S.; Masri, A.; Kottha, A.; Kuzmiak, S.; Flamm, S.; Schoenhagen, P. Reclassification of cardiovascular risk with coronary calcium scoring in subjects without documented coronary heart disease: Comparison with risk assessment based on Reynolds Risk Score. J. Am. Coll. Cardiol. 2012, 59, E1186. [Google Scholar] [CrossRef]
- Naqvi, T.Z.; Lee, M.S. Carotid Intima-media thickness and plaque in cardiovascular risk assessment. JACC Cardiovasc. Imaging 2014, 7, 1025–1038. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, M.W.; Markus, H.S.; Bots, M.L.; Rosvall, M.; Sitzer, M. Prediction of clinical cardiovascular events with carotid intima-media thickness: A systematic review and meta-analysis. Circulation 2007, 115, 459–467. [Google Scholar] [CrossRef] [PubMed]
- Folsom, A.R.; Kronmal, R.A.; Detrano, R.C.; O’Leary, D.H.; Bild, D.E.; Bluemke, D.A.; Budoff, M.J.; Liu, K.; Shea, S.; Szklo, M.; et al. Coronary artery calcification compared with carotid intima-media thickness in the prediction of cardiovascular disease incidence: The Multi-Ethnic Study of Atherosclerosis (MESA). Arch. Int. Med. 2008, 168, 1333–1339. [Google Scholar] [CrossRef]
- Terry, J.G.; Carr, J.J.; Tang, R.; Evans, G.W.; Kouba, E.O.; Shi, R.; Cook, D.R.; Vieira, J.L.; Espeland, M.A.; Mercuri, M.F.; et al. Coronary artery calcium outperforms carotid artery intima-media thickness as a noninvasive index of prevalent coronary artery stenosis. Arterioscler. Thromb. Vasc. Biol. 2005, 25, 1723–1728. [Google Scholar] [CrossRef] [PubMed]
- Danad, I.; Raijmakers, P.G.; Kamali, P.; Harms, H.J.; de Haan, S.; Lubberink, M.; van Kuijk, C.; Hoekstra, O.S.; Lammertsma, A.A.; Smulders, Y.M.; et al. Carotid artery intima-media thickness, but not coronary artery calcium, predicts coronary vascular resistance in patients evaluated for coronary artery disease. Eur. Heart J. Cardiovasc. Imaging 2012, 13, 317–323. [Google Scholar] [CrossRef] [PubMed]
- Knaapen, P.; Camici, P.G.; Marques, K.M.; Nijveldt, R.; Bax, J.J.; Westerhof, N.; Gotte, M.J.; Jerosch-Herold, M.; Schelbert, H.R.; Lammertsma, A.A.; et al. Coronary microvascular resistance: Methods for its quantification in humans. Basic Res. Cardiol. 2009, 104, 485–498. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, T.Z.; Mendoza, F.; Rafii, F.; Gransar, H.; Guerra, M.; Lepor, N.; Berman, D.S.; Shah, P.K. High prevalence of ultrasound detected carotid atherosclerosis in subjects with low Framingham Risk Score: Potential implications for screening for subclinical atherosclerosis. J. Am. Soc. Echocardiogr. 2010, 23, 809–815. [Google Scholar] [CrossRef] [PubMed]
- Davis, P.H.; Dawson, J.D.; Mahoney, L.T.; Lauer, R.M. Increased carotid intimal-medial thickness and coronary calcification are related in young and middle-aged adults: The Muscatine study. Circulation 1999, 100, 838–842. [Google Scholar] [CrossRef] [PubMed]
- Lester, S.J.; Eleid, M.F.; Khandheria, B.K.; Hurst, R.T. Carotid intima-media thickness and coronary artery calcium score as indications of subclinical atherosclerosis. Mayo Clin. Proc. 2009, 84, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Greenland, P.; Alpert, J.S.; Beller, G.A.; Benjamin, E.J.; Budoff, M.J.; Fayad, Z.A.; Foster, E.; Hlatky, M.A.; Hodgson, J.M.; Kushner, F.G.; et al. 2010 ACCF/AHA guideline for assessment of cardiovascular risk in asymptomatic adults: Executive summary: A report of the American College of Cardiology Foundation/American Heart Association task force on practice guidelines. Circulation 2010, 122, 2748–2764. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Danielson, E.; Fonseca, F.A.; Genest, J.; Gotto, A.M., Jr.; Kastelein, J.J.; Koenig, W.; Libby, P.; Lorenzatti, A.J.; MacFadyen, J.G.; et al. Rosuvastatin to prevent vascular events in men and women with elevated C-reactive protein. N. Engl. J. Med. 2008, 359, 2195–2207. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Cushman, M.; Stampfer, M.J.; Tracy, R.P.; Hennekens, C.H. Inflammation, aspirin, and the risk of cardiovascular disease in apparently healthy men. N. Engl. J. Med. 1997, 336, 973–979. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Hennekens, C.H.; Buring, J.E.; Rifai, N. C-reactive protein and other markers of inflammation in the prediction of cardiovascular disease in women. N. Engl. J. Med. 2000, 342, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Ridker, P.M.; Rifai, N.; Rose, L.; Buring, J.E.; Cook, N.R. Comparison of C-reactive protein and low-density lipoprotein cholesterol levels in the prediction of first cardiovascular events. N. Engl. J. Med. 2002, 347, 1557–1565. [Google Scholar] [CrossRef] [PubMed]
- Koenig, W.; Lowel, H.; Baumert, J.; Meisinger, C. C-reactive protein modulates risk prediction based on the Framingham Score: Implications for future risk assessment: Results from a large cohort study in southern Germany. Circulation 2004, 109, 1349–1353. [Google Scholar] [CrossRef] [PubMed]
- Pai, J.K.; Pischon, T.; Ma, J.; Manson, J.E.; Hankinson, S.E.; Joshipura, K.; Curhan, G.C.; Rifai, N.; Cannuscio, C.C.; Stampfer, M.J.; et al. Inflammatory markers and the risk of coronary heart disease in men and women. New Engl. J. Med. 2004, 351, 2599–2610. [Google Scholar] [CrossRef] [PubMed]
- Boekholdt, S.M.; Hack, C.E.; Sandhu, M.S.; Luben, R.; Bingham, S.A.; Wareham, N.J.; Peters, R.J.; Jukema, J.W.; Day, N.E.; Kastelein, J.J.; et al. C-reactive protein levels and coronary artery disease incidence and mortality in apparently healthy men and women: The EPIC-Norfolk prospective population study 1993–2003. Atherosclerosis 2006, 187, 415–422. [Google Scholar] [CrossRef] [PubMed]
- Ballantyne, C.M.; Hoogeveen, R.C.; Bang, H.; Coresh, J.; Folsom, A.R.; Heiss, G.; Sharrett, A.R. Lipoprotein-associated phospholipase A2, high-sensitivity C-reactive protein, and risk for incident coronary heart disease in middle-aged men and women in the Atherosclerosis Risk in Communities (ARIC) study. Circulation 2004, 109, 837–842. [Google Scholar] [CrossRef] [PubMed]
- Park, R.; Detrano, R.; Xiang, M.; Fu, P.; Ibrahim, Y.; LaBree, L.; Azen, S. Combined use of computed tomography coronary calcium scores and C-reactive protein levels in predicting cardiovascular events in nondiabetic individuals. Circulation 2002, 106, 2073–2077. [Google Scholar] [CrossRef] [PubMed]
- Lakoski, S.G.; Cushman, M.; Blumenthal, R.S.; Kronmal, R.; Arnett, D.; D’Agostino, R.B., Jr.; Detrano, R.C.; Herrington, D.M. Implications of C-reactive protein or coronary artery calcium score as an adjunct to global risk assessment for primary prevention of CHD. Atherosclerosis 2007, 193, 401–407. [Google Scholar] [CrossRef] [PubMed]
- Blaha, M.J.; Budoff, M.J.; DeFilippis, A.P.; Blankstein, R.; Rivera, J.J.; Agatston, A.; O’Leary, D.H.; Lima, J.; Blumenthal, R.S.; Nasir, K. Associations between C-reactive protein, coronary artery calcium, and cardiovascular events: Implications for the JUPITER population from MESA, a population-based cohort study. Lancet 2011, 378, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Mohlenkamp, S.; Lehmann, N.; Moebus, S.; Schmermund, A.; Dragano, N.; Stang, A.; Siegrist, J.; Mann, K.; Jockel, K.H.; Erbel, R.; et al. Quantification of coronary atherosclerosis and inflammation to predict coronary events and all-cause mortality. J. Am. Coll. Cardiol. 2011, 57, 1455–1464. [Google Scholar] [CrossRef] [PubMed]
- Reilly, M.P.; Wolfe, M.L.; Localio, A.R.; Rader, D.J. Study of inherited risk of coronary A. C-reactive protein and coronary artery calcification: The study of Inherited Risk of Coronary Atherosclerosis (SIRCA). Arterioscler. Thromb. Vasc. Biol. 2003, 23, 1851–1856. [Google Scholar] [CrossRef] [PubMed]
- Ding, J.; Hsu, F.C.; Harris, T.B.; Liu, Y.; Kritchevsky, S.B.; Szklo, M.; Ouyang, P.; Espeland, M.A.; Lohman, K.K.; Criqui, M.H.; et al. The association of pericardial fat with incident coronary heart disease: The Multi-Ethnic Study of Atherosclerosis (MESA). Am. J. Clin. Nutr. 2009, 90, 499–504. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Nasir, K.; Katz, R.; Takasu, J.; Carr, J.J.; Wong, N.D.; Allison, M.; Lima, J.A.; Detrano, R.; Blumenthal, R.S.; et al. Thoracic aortic calcification and coronary heart disease events: The Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis 2011, 215, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Zeb, I.; Budoff, M.J.; Katz, R.; Lloyd-Jones, D.; Agatston, A.; Blumenthal, R.S.; Blaha, M.; Blankstein, R.; Carr, J.J.; Nasir, K. Non-alcoholic fatty liver disease is an independent predictor of long-term incident coronary heart disease events—The Multi-Ethnic Study of Atherosclerosis. Circulation 2012, 126, A13688. [Google Scholar]
- Yeboah, J.; Carr, J.J.; Terry, J.G.; Ding, J.; Zeb, I.; Liu, S.; Nasir, K.; Post, W.; Blumenthal, R.S.; Budoff, M.J. Computed tomography-derived cardiovascular risk markers, incident cardiovascular events, and all-cause mortality in nondiabetics: The Multi-Ethnic Study of Atherosclerosis. Eur. J. Prev. Cardiol. 2014, 21, 1233–1241. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Nasir, K.; McClelland, R.L.; Detrano, R.; Wong, N.; Blumenthal, R.S.; Kondos, G.; Kronmal, R.A. Coronary calcium predicts events better with absolute calcium scores than age-sex-race/ethnicity percentiles: MESA (Multi-Ethnic Study of Atherosclerosis). J. Am. Coll. Cardiol. 2009, 53, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Kalia, N.K.; Miller, L.G.; Nasir, K.; Blumenthal, R.S.; Agrawal, N.; Budoff, M.J. Visualizing coronary calcium is associated with improvements in adherence to statin therapy. Atherosclerosis 2006, 185, 394–399. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.J.; Bindeman, J.; Feuerstein, I.; Le, T.; Bauer, K.; Byrd, C.; Wu, H.; O’Malley, P.G. Community-based provision of statin and aspirin after the detection of coronary artery calcium within a community-based screening cohort. J. Am. Coll. Cardiol. 2008, 51, 1337–1341. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Hokanson, J.E.; Nasir, K.; Shaw, L.J.; Kinney, G.L.; Chow, D.; Demoss, D.; Nuguri, V.; Nabavi, V.; Ratakonda, R.; et al. Progression of coronary artery calcium predicts all-cause mortality. JACC Cardiovasc. Imaging 2010, 3, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Berry, J.D.; Liu, K.; Folsom, A.R.; Lewis, C.E.; Carr, J.J.; Polak, J.F.; Shea, S.; Sidney, S.; O’Leary, D.H.; Chan, C.; et al. Prevalence and progression of subclinical atherosclerosis in younger adults with low short-term but high lifetime estimated risk for cardiovascular disease: The coronary artery risk development in young adults study and multi-ethnic study of atherosclerosis. Circulation 2009, 119, 382–389. [Google Scholar] [CrossRef] [PubMed]
- Raggi, P.; Cooil, B.; Shaw, L.J.; Aboulhson, J.; Takasu, J.; Budoff, M.; Callister, T.Q. Progression of coronary calcium on serial electron beam tomographic scanning is greater in patients with future myocardial infarction. Am. J. Cardiol. 2003, 92, 827–829. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Young, R.; Lopez, V.A.; Kronmal, R.A.; Nasir, K.; Blumenthal, R.S.; Detrano, R.C.; Bild, D.E.; Guerci, A.D.; Liu, K.; et al. Progression of coronary calcium and incident coronary heart disease events: MESA (Multi-Ethnic Study of Atherosclerosis). J. Am. Coll. Cardiol. 2013, 61, 1231–1239. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Achenbach, S.; Blumenthal, R.S.; Carr, J.J.; Goldin, J.G.; Greenland, P.; Guerci, A.D.; Lima, J.A.; Rader, D.J.; Rubin, G.D.; et al. Assessment of coronary artery disease by cardiac computed tomography: A scientific statement from the American Heart Association Committee on Cardiovascular Imaging and Intervention, Council on Cardiovascular Radiology and Intervention, and Committee on Cardiac Imaging, Council on Clinical Cardiology. Circulation 2006, 114, 1761–1791. [Google Scholar] [CrossRef] [PubMed]
- Parker, M.S.; Hui, F.K.; Camacho, M.A.; Chung, J.K.; Broga, D.W.; Sethi, N.N. Female breast radiation exposure during CT pulmonary angiography. AJR Am. J. Roentgenol. 2005, 185, 1228–1233. [Google Scholar] [CrossRef] [PubMed]
- Horton, K.M.; Post, W.S.; Blumenthal, R.S.; Fishman, E.K. Prevalence of significant noncardiac findings on electron-beam computed tomography coronary artery calcium screening examinations. Circulation 2002, 106, 532–534. [Google Scholar] [CrossRef] [PubMed]
- Schragin, J.G.; Weissfeld, J.L.; Edmundowicz, D.; Strollo, D.C.; Fuhrman, C.R. Non-cardiac findings on coronary electron beam computed tomography scanning. J. Thora. Imaging 2004, 19, 82–86. [Google Scholar] [CrossRef]
- Machaalany, J.; Yam, Y.; Ruddy, T.D.; Abraham, A.; Chen, L.; Beanlands, R.S.; Chow, B.J. Potential clinical and economic consequences of noncardiac incidental findings on cardiac computed tomography. J. Am. Coll. Cardiol. 2009, 54, 1533–1541. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zeb, I.; Budoff, M. Coronary Artery Calcium Screening: Does it Perform Better than Other Cardiovascular Risk Stratification Tools? Int. J. Mol. Sci. 2015, 16, 6606-6620. https://doi.org/10.3390/ijms16036606
Zeb I, Budoff M. Coronary Artery Calcium Screening: Does it Perform Better than Other Cardiovascular Risk Stratification Tools? International Journal of Molecular Sciences. 2015; 16(3):6606-6620. https://doi.org/10.3390/ijms16036606
Chicago/Turabian StyleZeb, Irfan, and Matthew Budoff. 2015. "Coronary Artery Calcium Screening: Does it Perform Better than Other Cardiovascular Risk Stratification Tools?" International Journal of Molecular Sciences 16, no. 3: 6606-6620. https://doi.org/10.3390/ijms16036606
APA StyleZeb, I., & Budoff, M. (2015). Coronary Artery Calcium Screening: Does it Perform Better than Other Cardiovascular Risk Stratification Tools? International Journal of Molecular Sciences, 16(3), 6606-6620. https://doi.org/10.3390/ijms16036606





