Phenolic Composition and Antioxidant Properties of Different Peach [Prunus persica (L.) Batsch] Cultivars in China
Abstract
:1. Introduction
2. Results and Discussion
2.1. Fruit Quality Evaluation
| Number | Cultivars | Abbreviation | Fruit Type | Flesh Colour | FW (g) | FSI | SSC (°Brix) |
|---|---|---|---|---|---|---|---|
| 1 | Annongshuimi | ANSM | Melting | White | 179.55 ± 18.55 c | 0.97 ± 0.05 d | 8.34 ± 1.06 f |
| 2 | Chunfeng | CF | Melting | White | 122.59 ± 12.30 g | 1.12 ± 0.06 a | 8.75 ± 0.85 e,f |
| 3 | Chiyue | CY | Melting | White | 181.47 ± 8.56 c | 0.97 ± 0.03 d | 11.27 ± 1.34 c,d |
| 4 | Danxia | DX | Melting | White | 181.85 ± 10.92 c | 0.96 ± 0.04 d | 10.92 ± 0.72 c,d |
| 5 | Dayubaifeng | DYBF | Melting | White | 166.63 ± 11.79 d,e | 0.96 ± 0.03 d | 11.75 ± 0.53 c |
| 6 | Hujingmilu | HJML | Melting | White | 200.47 ± 11.95 b | 0.95 ± 0.05 d | 13.92 ± 1.98 b |
| 7 | Jinhuadabaitao | JHDBT | Melting | White | 201.32 ± 26.57 b | 1.02 ± 0.04 c | 8.81 ± 0.92 e,f |
| 8 | Qingshuibaitao | QSBT | Melting | White | 201.55 ± 10.58 b | 0.89 ± 0.02 e | 15.30 ± 2.05 a |
| 9 | Shazizaosheng | SZZS | Melting | White | 233.94 ± 24.01 a | 0.98 ± 0.04 d | 10.61 ± 1.03 d |
| 10 | Wujingzaobaifeng | WJZBF | Melting | Red | 136.66 ± 7.70 f | 0.95 ± 0.03 d | 9.30 ± 1.23 e,f |
| 11 | Xinyu | XY | Melting | White | 209.98 ± 13.64 b | 1.02 ± 0.03 c | 14.10 ± 0.73 b |
| 12 | Yulu | YL | Melting | White | 198.84 ± 17.24 b | 0.95 ± 0.04 d | 15.48 ± 1.03 a |
| 13 | Zhaoxia | ZX | Melting | White | 172.05 ± 18.55 c,d | 0.90 ± 0.05 e | 8.45 ± 1.94 f |
| 14 | Huyou002 | HY002 | Nectarine | White | 83.61 ± 6.22 i | 1.14 ± 0.04 a | 9.45 ± 0.69 e,f |
| 15 | Huyou003 | HY003 | Nectarine | Yellow | 100.57 ± 11.39 h | 0.97 ± 0.03 d | 9.57 ± 0.78 e |
| 16 | Huyou004 | HY004 | Nectarine | Yellow | 135.85 ± 8.07 f | 1.02 ± 0.04 c | 8.42 ± 1.06 f |
| 17 | Huyou018 | HY018 | Nectarine | Yellow | 157.83 ± 13.62 e | 1.06 ± 0.05 b | 9.21 ± 1.12 e,f |
2.2. Identification of Individual Phenolic Compound
| Phenolic Groups | λmax (nm) | Molecular Weight | MS2 (m/z) | Tentative Identification | R Groups |
|---|---|---|---|---|---|
Hydroxycinnamates 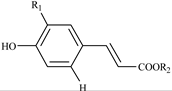
| 324.6, 240.4 | 354 | 353.1, 191.1, 179.0 | NCHA | R1 = OH; R2 = 5-quinic acid |
| 327.0, 241.6 | 354 | 353.1, 191.1, 179.0 | CHA | R1 = OH; R2 = 3-quinic acid | |
Flavan-3-ols 
| 279.4 | 578 | 577.1, 425.1, 407.1, 289.1 | B1 | R1 = H; R2 = OH; R3 = epicatechin |
| 279.4 | 290 | 289.0 | CAT | R1 = H; R2 = OH; R3 = H | |
Anthocyanins 
| 514.9, 279.4 | 449 | 447.0, 284.9 | C3G | R1 = glucoside |
Flavonols 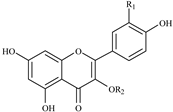
| 355.7, 254.6 | 464 | 463.1, 301.0, 300.0 | Q3GAL | R1 = OH; R2 = galactoside |
| 356.9, 255.7 | 464 | 463.1, 301.0, 300.0 | Q3GLU | R1 = OH; R2 = glucoside | |
| 355.7, 255.7 | 610 | 609.2, 301.0, 300.0 | Q3R | R1 = OH; R2 = rutinoside | |
| 347.3, 265.2 | 594 | 593.1, 285.0 | K3R | R1 = H; R2 = rutinoside |
2.3. Quantification of Phenolic Compounds
| Cultivars | Hydroxycinnamates | Flavan-3-ols | Anthocyanin | Flavonols | |||||
|---|---|---|---|---|---|---|---|---|---|
| NCHA | CHA | B1 | CAT | C3G | Q3GAL | Q3GLU | Q3R | K3R | |
| ANSM | 5.77 ± 0.51 j | 52.20 ± 0.76 k | nd | 60.14 ± 8.39 j | 9.33 ± 0.73 g | 15.52 ± 1.19 j | 16.37 ± 1.53 h–j | 120.16 ± 6.09 e,f | 51.17 ± 4.08 c |
| CF | 138.72 ± 5.89 g | 609.27 ± 25.74 h | 54.76 ± 2.10 h | 474.88 ± 29.90 f,g | 134.66 ± 11.72 e | 62.08 ± 2.56 d,e | 58.64 ± 3.04 e,f | 193.25 ± 5.94 a | 110.86 ± 4.43 a |
| CY | 193.02 ± 3.31 d | 1090.86 ± 19.57 d,e | 496.26 ± 16.13 a,b | 768.15 ± 45.85 c,d | 25.39 ± 1.91 g | 24.09 ± 4.71 h–j | 15.04 ± 0.43 i,j | 78.32 ± 0.79 j | 29.45 ± 0.60 g,h |
| DX | 219.13 ± 4.57 c | 1166.15 ± 15.73 c | 295.16 ± 33.03 e | 514.75 ± 38.27 e,f | 69.27 ± 7.76 f | 17.86 ± 0.26 i,j | 55.44 ± 11.94 e,f | 69.82 ± 1.74 k | 39.51 ± 0.36 e |
| DYBF | 195.74 ± 5.90 d | 1051.63 ± 23.82 d,e | 324.11 ± 9.70 d,e | 576.17 ± 15.80 e | 19.15 ± 2.32 g | 42.02 ± 1.12 f,g | 27.15 ± 0.41 h,i | 85.67 ± 0.26 i | 29.79 ± 0.49 g,h |
| HJML | 130.05 ± 1.57 g | 720.93 ± 10.47 g | 206.17 ± 36.62 f | 428.95 ± 18.97 g | 24.75 ± 1.63 g | 22.98 ± 0.47 h–j | 20.19 ± 1.28 h–j | 74.24 ± 1.17 j,k | 32.50 ± 0.53 f–h |
| JHDBT | 32.04 ± 1.61 i | 162.72 ± 8.64 j | nd | 149.69 ± 25.47 i | 11.65 ± 0.49 g | 23.98 ± 1.13 h–j | 23.08 ± 1.00 h–j | 115.82 ± 2.84 f,g | 33.02 ± 1.27 f,g |
| QSBT | 171.16 ± 6.74 e | 1110.77 ± 46.02 c,d | 452.63 ± 39.33 b | 806.87 ± 36.87 c | nd | 33.53 ± 1.04 g–i | 31.67 ± 1.14 g–i | 126.09 ± 7.63 d,e | 34.83 ± 0.86 f |
| SZZS | 87.61 ± 2.98 h | 329.71 ± 6.93 i | nd | 253.46 ± 18.26 h | 21.00 ± 0.58 g | 52.66 ± 2.36 e,f | 52.29 ± 2.34 e–g | 139.54 ± 3.51 c | 39.62 ± 1.60 e |
| WJZBF | 136.15 ± 4.68 g | 674.38 ± 25.83 g,h | 120.65 ± 16.94 g | 483.52 ± 56.84 f,g | 220.30 ± 11.40 d | 69.38 ± 2.77 d | 67.84 ± 2.86 e | 172.73 ± 6.07 b | 44.87 ± 1.63 d |
| XY | 342.75 ± 16.06 a | 1020.50 ± 51.80 e,f | 539.22 ± 45.95 a | 707.32 ± 82.98 d | 18.57 ± 1.59 g | 20.26 ± 3.89 i,j | 14.00 ± 2.63 i,j | 75.72 ± 4.30 j,k | 34.54 ± 4.43 f |
| YL | 166.62 ± 8.99 e,f | 955.83 ± 49.43 f | 368.75 ± 47.92 c,d | 573.79 ± 25.82 e | nd | 8.45 ± 0.57 j | 2.45 ± 0.21 j | 59.15 ± 0.65 l | 28.81 ± 0.73 h |
| ZX | 10.56 ± 0.43 j | 126.72 ± 6.50 j | nd | 196.62 ± 18.36 h,i | 28.55 ± 5.12 g | 36.61 ± 1.58 g,h | 39.29 ± 0.86 f–h | 90.80 ± 1.37 i | 55.64 ± 1.18 b |
| HY002 | 257.16 ± 9.73 b | 1631.25 ± 71.17 a | 153.71 ± 35.96 g | 748.86 ± 40.54 c,d | 304.78 ± 37.12 c | 396.49 ± 19.96 a | 581.21 ± 18.09 a | 131.76 ± 2.97 d | 29.85 ± 0.84 g,h |
| HY003 | 95.54 ± 1.43 h | 741.79 ± 18.35 g | 203.44 ± 8.24 f | 911.35 ± 21.10 b | 125.52 ± 17.51 e | 267.38 ± 5.19 c | 388.36 ± 8.11 d | 111.01 ± 2.18 g,h | 18.39 ± 0.29 i |
| HY004 | 135.63 ± 7.66 g | 1037.81 ± 62.36 d,e | 108.79 ± 14.09 g | 741.83 ± 54.10 c,d | 398.93 ± 47.39 b | 324.67 ± 17.09 b | 530.32 ± 26.04 c | 109.07 ± 3.17 h | 18.64 ± 0.95 i |
| HY018 | 157.48 ± 12.15 f | 1264.42 ± 99.20 b | 384.59 ± 20.18 c | 1030.06 ± 39.18 a | 670.59 ± 59.63 a | 335.07 ± 22.05 b | 555.83 ± 37.15 b | 106.32 ± 3.54 h | 16.91 ± 1.00 i |
| Cultivars | Hydroxycinnamates | Flavan-3-ols | Anthocyanin | Flavonols | |||||
|---|---|---|---|---|---|---|---|---|---|
| NCHA | CHA | B1 | CAT | C3G | Q3GAL | Q3GLU | Q3R | K3R | |
| ANSM | 25.77 ± 1.70 j | 41.28 ± 2.50 k | nd | nd | 35.44 ± 4.12 b | nd | nd | nd | nd |
| CF | 262.49 ± 13.09 a,b | 238.78 ± 11.17 e,f | 10.28 ± 3.97 g | 156.19 ± 11.85 e,f | nd | nd | nd | nd | nd |
| CY | 179.01 ± 6.54 d | 568.07 ± 20.91 a | 331.64 ± 19.49 b | 374.30 ± 11.38 a | nd | nd | nd | nd | nd |
| DX | 174.60 ± 7.09 d | 349.04 ± 14.23 c | 89.00 ± 8.89 e | 123.97 ± 5.18 g | 12.86 ± 1.33 c | nd | nd | nd | nd |
| DYBF | 157.44 ± 3.94 e | 354.06 ± 9.07 c | 142.58 ± 8.82 d | 206.65 ± 6.85 c,d | nd | nd | nd | nd | nd |
| HJML | 117.20 ± 7.73 f | 262.55 ± 15.95 e | 62.83 ± 5.18 f | 87.71 ± 8.42 h | 3.83 ± 0.67 d | nd | nd | nd | nd |
| JHDBT | 57.34 ± 5.02 i | 76.99 ± 6.34 j | nd | nd | 10.88 ± 1.00 c,d | 4.90 ± 0.87 a | 2.02 ± 0.38 b | nd | nd |
| QSBT | 112.00 ± 11.70 f | 291.22 ± 29.12 d | 141.27 ± 20.72 d | 174.48 ± 15.84 d–f | nd | nd | nd | nd | nd |
| SZZS | 119.13 ± 5.62 f | 109.42 ± 3.73 i | nd | nd | 18.08 ± 1.03 c | 2.93 ± 0.10 b | 0.71 ± 0.14 b | nd | nd |
| WJZBF | 110.15 ± 3.80 f | 175.89 ± 5.84 h | nd | 70.85 ± 1.34 h | 184.81 ± 10.14 a | 4.90 ± 0.02 a | 21.35 ± 1.10 a | nd | nd |
| XY | 267.78 ± 16.47 a | 189.14 ± 11.16 g,h | 403.75 ± 24.5 a | 293.32 ± 40.38 b | 32.24 ± 5.39 b | nd | nd | nd | nd |
| YL | 251.66 ± 14.99 b | 419.35 ± 26.37 b | 186.20 ± 9.15 c | 190.34 ± 11.65 c,d | 39.19 ± 2.74 b | nd | nd | nd | nd |
| ZX | 15.74 ± 0.53 j | 27.50 ± 0.79 k | nd | nd | nd | nd | nd | nd | nd |
| HY002 | 232.36 ± 7.27 c | 312.14 ± 15.78 d | nd | nd | nd | nd | nd | nd | nd |
| HY003 | 82.86 ± 2.60 g,h | 129.09 ± 11.28 i | 67.89 ± 4.38 e,f | 211.16 ± 17.14 c | 12.68 ± 0.87 c | nd | nd | nd | nd |
| HY004 | 95.92 ± 1.97 g | 201.65 ± 13.11 g | 13.01 ± 1.69 g | 144.36 ± 30.23 f,g | 4.74 ± 1.10 d | nd | nd | nd | nd |
| HY018 | 77.95 ± 5.01 h | 232.56 ± 11.76 f | 68.26 ± 16.09 e,f | 184.60 ± 10.00 c–e | 38.13 ± 6.27 b | nd | nd | nd | nd |
2.4. Variation Patterns and Principal Components Analysis (PCA)
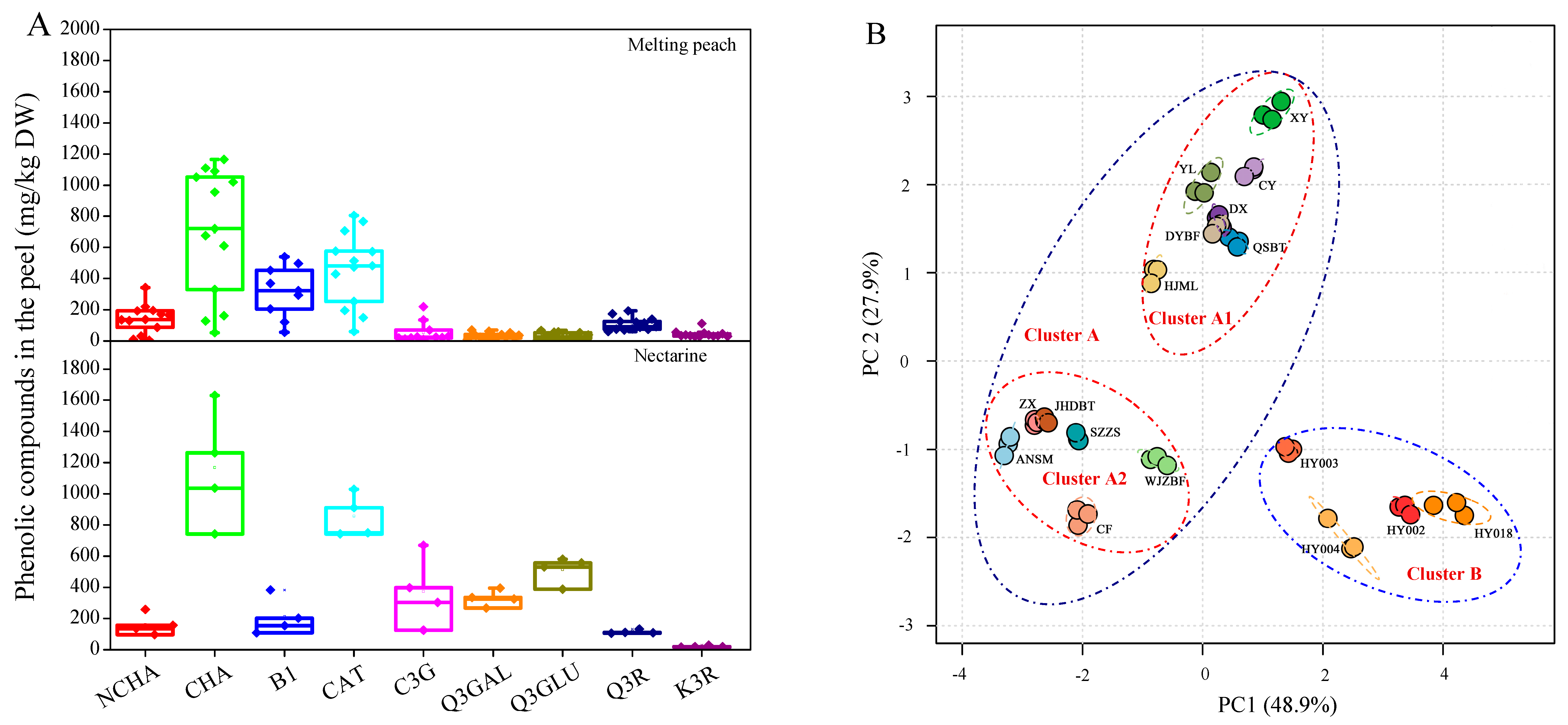
2.5. Total Phenolics and Antioxidant Capacities
| Cultivars | Peel | Pulp | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Total Phenolics mg GAE/g DW | DPPH mg TEAC/g DW | FRAP mg TEAC/g DW | APC | Rank | Total Phenolics mg GAE/g DW | DPPH mg TEAC/g DW | FRAP mg TEAC/g DW | APC | Rank | |
| ANSM | 4.58 ± 0.06 j | 6.66 ± 0.27 h | 3.24 ± 0.01 j | 28.36 | 16 | 2.82 ± 0.13 i | 4.42 ± 0.06 e | 1.96 ± 0.08 h | 28.92 | 14 |
| CF | 9.54 ± 0.11 g | 13.83 ± 0.34 d | 7.70 ± 0.21 g | 62.34 | 12 | 4.09 ± 0.12 d,e | 6.56 ± 0.24 c,d | 3.26 ± 0.01 e | 45.47 | 7 |
| CY | 11.19 ± 0.12 d,e | 17.15 ± 1.01 b | 12.41 ± 0.13 b | 87.72 | 4 | 5.89 ± 0.02 c | 9.75 ± 0.62 b | 6.89 ± 0.04 a | 82.02 | 2 |
| DX | 10.12 ± 0.12 f | 15.53 ± 0.10 c | 11.17 ± 0.13 d | 79.16 | 8 | 4.19 ± 0.08 d | 7.17 ± 0.18 c | 4.95 ± 0.13 c | 59.63 | 4 |
| DYBF | 9.60 ± 0.02 g | 15.13 ± 0.26 c | 10.98 ± 0.07 d | 77.47 | 9 | 3.65 ± 0.05 f | 5.96 ± 0.11 d | 4.06 ± 0.07 d | 49.11 | 6 |
| HJML | 9.72 ± 0.34 f,g | 15.03 ± 0.95 c | 8.69 ± 0.21 f | 69.01 | 11 | 3.33 ± 0.08 g | 6.28 ± 0.10 c,d | 3.09 ± 0.05 e | 43.34 | 8 |
| JHDBT | 6.20 ± 0.17 h | 9.45 ± 0.71 f | 4.70 ± 0.04 h | 40.63 | 14 | 2.35 ± 0.07 k | 4.28 ± 0.41 e | 1.97 ± 0.05 h | 28.67 | 15 |
| QSBT | 11.06 ± 0.2 1,e | 19.79 ± 0.88 a | 11.71 ± 0.25 c | 91.73 | 3 | 3.59 ± 0.29 f | 6.46 ± 0.70 c,d | 3.89 ± 0.29 d | 49.75 | 5 |
| SZZS | 6.32 ± 0.23 h | 8.20 ± 0.47 g | 4.19 ± 0.10 i | 35.64 | 15 | 2.13 ± 0.01 l | 3.00 ± 0.12 f | 1.45 ± 0.02 i | 20.55 | 16 |
| WJZBF | 9.82 ± 0.23 f,g | 12.79 ± 0.11 e | 7.59 ± 0.18 g | 59.41 | 13 | 3.93 ± 0.07 e | 5.73 ± 0.19 d | 3.23 ± 0.03 e | 42.43 | 9 |
| XY | 12.25 ± 0.76 a,b | 19.84 ± 0.48 a | 13.85 ± 0.66 a | 99.57 | 1 | 6.52 ± 0.15 a | 15.01 ± 2.36 a | 6.99 ± 0.18 a | 99.89 | 1 |
| YL | 10.78 ± 0.43 e | 16.98 ± 0.12 b | 8.78 ± 0.69 f | 74.22 | 10 | 6.08 ± 0.10 b | 9.58 ± 0.22 b | 5.17 ± 0.16 b | 69.22 | 3 |
| ZX | 5.15 ± 0.04 i | 6.35 ± 0.63 h | 3.44 ± 0.13 j | 28.29 | 17 | 0.82 ± 0.03 m | 1.05 ± 0.07 g | 0.61 ± 0.01 j | 7.92 | 17 |
| HY002 | 12.68 ± 0.14 a | 17.63 ± 0.14 b | 11.72 ± 0.15 c | 86.41 | 5 | 3.03 ± 0.07 h | 4.42 ± 0.15 e | 2.69 ± 0.04 f | 34.19 | 11 |
| HY003 | 11.54 ± 0.06 c,d | 17.43 ± 0.15 b | 10.49 ± 0.07 e | 81.47 | 7 | 2.87 ± 0.03 h,i | 4.26 ± 0.35 e | 2.61 ± 0.03 f | 32.94 | 12 |
| HY004 | 11.94 ± 0.04 b,c | 16.81 ± 0.50 b | 11.25 ± 0.32 c,d | 82.64 | 6 | 2.59 ± 0.02 j | 3.59 ± 0.14 e,f | 2.41 ± 0.04 g | 29.41 | 13 |
| HY018 | 12.61 ± 0.12 a | 19.29 ± 0.58 a | 12.49 ± 0.09 b | 93.32 | 2 | 3.35 ± 0.02 g | 5.59 ± 0.12 d | 3.13 ± 0.03 e | 41.29 | 10 |
2.6. Correlations between Fruit Bioactivity Traits
| Antioxidant Capacities/Phenolic Contents | Peel | Pulp | ||
|---|---|---|---|---|
| DPPH | FRAP | DPPH | FRAP | |
| FRAP | 0.952 ** | 1 | 0.918 ** | 1 |
| Total phenolics | 0.956 ** | 0.936 ** | 0.935 ** | 0.950 ** |
| TIP | 0.863 ** | 0.876 ** | 0.829 ** | 0.953 ** |
| Hydroxycinnamates | 0.881 ** | 0.919 ** | 0.642 ** | 0.802 ** |
| Flavan-3-ols | 0.942 ** | 0.938 ** | 0.827 ** | 0.907 ** |
| Anthocyanin | 0.473 ** | 0.408 ** | 0.062 | 0.071 |
| Flavonols | 0.338 * | 0.329 * | −0.097 | −0.124 |
| NCHA | 0.788 ** | 0.851 ** | 0.698 ** | 0.687 ** |
| CHA | 0.874 ** | 0.905 ** | 0.519 ** | 0.752 ** |
| B1 | 0.675 ** | 0.676 ** | 0.873 ** | 0.925 ** |
| CAT | 0.907 ** | 0.891 ** | 0.550 ** | 0.718 ** |
3. Experimental Section
3.1. Chemicals
3.2. Materials
3.3. Fruit Quality Analysis
3.4. Preparation of Fruit Peels and Pulp Extracts
3.5. Determination of Total Phenolics
3.6. HPLC-DAD and LC-ESI-MS/MS Analysis of Phenolic Compounds
3.7. Antioxidant Activity Assays
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Bazzano, L.A.; He, J.; Ogden, L.G.; Loria, C.M.; Vupputuri, S.; Myers, L.; Whelton, P.K. Fruit and vegetable intake and risk of cardiovascular disease in US adults: The first national health and nutrition examination survey epidemiologic follow-up study. Am. J. Clin. Nutr. 2002, 76, 93–99. [Google Scholar] [PubMed]
- Del Rio, D.; Rodriguez-Mateos, A.; Spencer, J.P.E.; Tognolini, M.; Borges, G.; Crozier, A. Dietary (poly)phenolics in human health: Structures, bioavailability, and evidence of protective effects against chronic diseases. Antioxid. Redox Signal. 2013, 18, 1818–1892. [Google Scholar]
- Eberhardt, M.V.; Lee, C.Y.; Liu, R.H. Antioxidant activity of fresh apples. Nature 2000, 405, 903–904. [Google Scholar] [PubMed]
- Crozier, A.; Jaganath, I.B.; Clifford, M.N. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep. 2009, 26, 1001–1043. [Google Scholar] [CrossRef] [PubMed]
- Noratto, G.; Porter, W.; Byrne, D.; Cisneros-Zevallos, L. Identifying peach and plum polyphenols with chemopreventive potential against estrogen-independent breast cancer cells. J. Agric. Food Chem. 2009, 57, 5219–5226. [Google Scholar] [CrossRef] [PubMed]
- Mouly, P.P.; Arzouyan, C.R.; Gaydou, E.M.; Estienne, J.M. Differentiation of citrus juices by factorial discriminant analysis using liquid chromatography of flavanone glycosides. J. Agric. Food Chem. 1994, 42, 70–79. [Google Scholar] [CrossRef]
- Peterson, J.J.; Dwyer, J.T.; Beecher, G.R.; Bhagwat, S.A.; Gebhardt, S.E.; Haytowitz, D.B.; Holden, J.M. Flavanones in oranges, tangerines (mandarins), tangors, and tangelos: A compilation and review of the data from the analytical literature. J. Food Compos. Anal. 2006, 19, S66–S73. [Google Scholar] [CrossRef]
- Faust, M.; Timon, B. Origin and dissemination of peach. Hortic. Rev. 1995, 17, 331–379. [Google Scholar]
- Cevallos-Casals, B.A.; Byrne, D.; Okie, W.R.; Cisneros-Zevallos, L. Selecting new peach and plum genotypes rich in phenolic compounds and enhanced functional properties. Food Chem. 2006, 96, 273–280. [Google Scholar] [CrossRef]
- Chang, S.; Tan, C.; Frankel, E.N.; Barrett, D.M. Low-density lipoprotein antioxidant activity of phenolic compounds and polyphenol oxidase activity in selected clingstone peach cultivars. J. Agric. Food Chem. 2000, 48, 147–151. [Google Scholar] [CrossRef] [PubMed]
- Gil, M.I.; Tomás-Barberán, F.A.; Hess-Pierce, B.; Kader, A.A. Antioxidant capacities, phenolic compounds, carotenoids, and vitamin C contents of nectarine, peach, and plum cultivars from California. J. Agric. Food Chem. 2002, 50, 4976–4982. [Google Scholar] [CrossRef] [PubMed]
- Di Vaio, C.; Graziani, G.; Marra, L.; Cascone, A.; Ritieni, A. Antioxidant capacities, carotenoids and polyphenols evaluation of fresh and refrigerated peach and nectarine cultivars from Italy. Eur. Food Res. Technol. 2008, 227, 1225–1231. [Google Scholar] [CrossRef]
- Cantín, C.M.; Moreno, M.A.; Gogorcena, Y. Evaluation of the antioxidant capacity, phenolic compounds, and vitamin C content of different peach and nectarine [Prunus persica (L.) Batsch] breeding progenies. J. Agric. Food Chem. 2009, 57, 4586–4592. [Google Scholar] [CrossRef] [PubMed]
- Reig, G.; Iglesias, I.; Gatius, F.; Alegre, S. Antioxidant capacity, quality, and anthocyanin and nutrient contents of several peach cultivars [Prunus persica (L.) Batsch] grown in Spain. J. Agric. Food Chem. 2013, 61, 6344–6357. [Google Scholar] [CrossRef] [PubMed]
- Rossato, S.B.; Haas, C.; Raseira, M.D.C.B.; Moreira, J.C.F.; Zuanazzi, J.Â.S. Antioxidant potential of peels and fleshes of peaches from different cultivars. J. Med. Food 2009, 12, 1119–1126. [Google Scholar] [CrossRef] [PubMed]
- Manzoor, M.; Anwar, F.; Mahmood, Z.; Rashid, U.; Ashraf, M. Variation in minerals, phenolics and antioxidant activity of peel and pulp of different cultivar of peach (Prunus persica L.) fruit from Pakistan. Molecules 2012, 17, 6491–6506. [Google Scholar] [CrossRef] [PubMed]
- Scordino, M.; Sabatino, L.; Muratore, A.; Belligno, A.; Gagliano, G. Phenolic characterization of Sicilian yellow flesh peach (Prunus persica L.) cultivars at different ripening stages. J. Food Qual. 2012, 35, 255–262. [Google Scholar] [CrossRef]
- Tomás-Barberán, F.A.; Gil, M.I.; Cremin, P.; Waterhouse, A.L.; Hess-Pierce, B.; Kader, A.A. HPLC-DAD-ESIMS analysis of phenolic compounds in nectarines, peaches, and plums. J. Agric. Food Chem. 2001, 49, 4748–4760. [Google Scholar] [CrossRef] [PubMed]
- FAOSTAT (Food and Agriculture Organization of the United Nations Statistics Division). Available online: http://faostat3.fao.org/download/Q/QC/E. page (accessed on 2 March 2015).
- Legua, P.; Hernández, F.; Díaz-Mula, H.M.; Valero, D.; Serrano, M. Quality, bioactive compounds, and antioxidant activity of new flat-type peach and nectarine cultivars: A comparative study. J. Food Sci. 2011, 76, 729–935. [Google Scholar] [CrossRef]
- Tavarini, S.; Degl’Innocenti, E.; Remorini, D.; Massai, R.; Guidi, L. Preliminary characterisation of peach cultivars for their antioxidant capacity. Int. J. Food Sci. Technol. 2008, 43, 810–815. [Google Scholar] [CrossRef]
- Clifford, M.N.; Johnston, K.L.; Knight, S.; Kuhnert, N. Hierarchical scheme for LC-MSn identification of chlorogenic acids. J. Agric. Food Chem. 2003, 51, 2900–2911. [Google Scholar] [CrossRef] [PubMed]
- Sanz, M.; Cadahía, E.; Esteruelas, E.; Muñoz, A.M.; Fernández de Simón, B.; Hernández, T.; Estrella, I. Phenolic compounds in cherry (Prunus avium) heartwood with a view to their use in cooperage. J. Agric. Food Chem. 2010, 58, 4907–4914. [Google Scholar] [CrossRef] [PubMed]
- Hvattum, E.; Ekeberg, D. Study of the collision-induced radical cleavage of flavonoid glycosides using negative electrospray ionization tandem quadrupole mass spectrometry. J. Mass Spectrom. 2003, 38, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Cheng, G.W.; Crisosto, C.H. Browning potential, phenolic composition, and polyphenoloxidase activity of buffer extracts of peach and nectarine skin tissue. J. Am. Soc. Hortic. Sci. 1995, 120, 835–838. [Google Scholar]
- Guo, C.J.; Yang, J.J.; Wei, J.Y.; Li, Y.F.; Xu, J.; Jiang, Y.G. Antioxidant activities of peel, pulp and seed fractions of common fruits as determined by FRAP assay. Nutr. Res. 2003, 23, 1719–1726. [Google Scholar] [CrossRef]
- Seeram, N.P.; Aviram, M.; Zhang, Y.J.; Henning, S.M.; Feng, L.; Dreher, M.; Heber, D. Comparison of antioxidant potency of commonly consumed polyphenol-rich beverages in the United States. J. Agric. Food Chem. 2008, 56, 1415–1422. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.Z.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef] [PubMed]
- Brand-williams, W.; Cuvelier, M.E.; Berset, C. Use of a free-radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.G.; Wishart, D.S. Web-based inference of biological patterns, functions and pathways from metabolomic data using MetaboAnalyst. Nat. Protoc. 2011, 6, 743–760. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, X.; Zhang, W.; Yin, X.; Su, M.; Sun, C.; Li, X.; Chen, K. Phenolic Composition and Antioxidant Properties of Different Peach [Prunus persica (L.) Batsch] Cultivars in China. Int. J. Mol. Sci. 2015, 16, 5762-5778. https://doi.org/10.3390/ijms16035762
Zhao X, Zhang W, Yin X, Su M, Sun C, Li X, Chen K. Phenolic Composition and Antioxidant Properties of Different Peach [Prunus persica (L.) Batsch] Cultivars in China. International Journal of Molecular Sciences. 2015; 16(3):5762-5778. https://doi.org/10.3390/ijms16035762
Chicago/Turabian StyleZhao, Xiaoyong, Wenna Zhang, Xueren Yin, Mingshen Su, Chongde Sun, Xian Li, and Kunsong Chen. 2015. "Phenolic Composition and Antioxidant Properties of Different Peach [Prunus persica (L.) Batsch] Cultivars in China" International Journal of Molecular Sciences 16, no. 3: 5762-5778. https://doi.org/10.3390/ijms16035762
APA StyleZhao, X., Zhang, W., Yin, X., Su, M., Sun, C., Li, X., & Chen, K. (2015). Phenolic Composition and Antioxidant Properties of Different Peach [Prunus persica (L.) Batsch] Cultivars in China. International Journal of Molecular Sciences, 16(3), 5762-5778. https://doi.org/10.3390/ijms16035762







