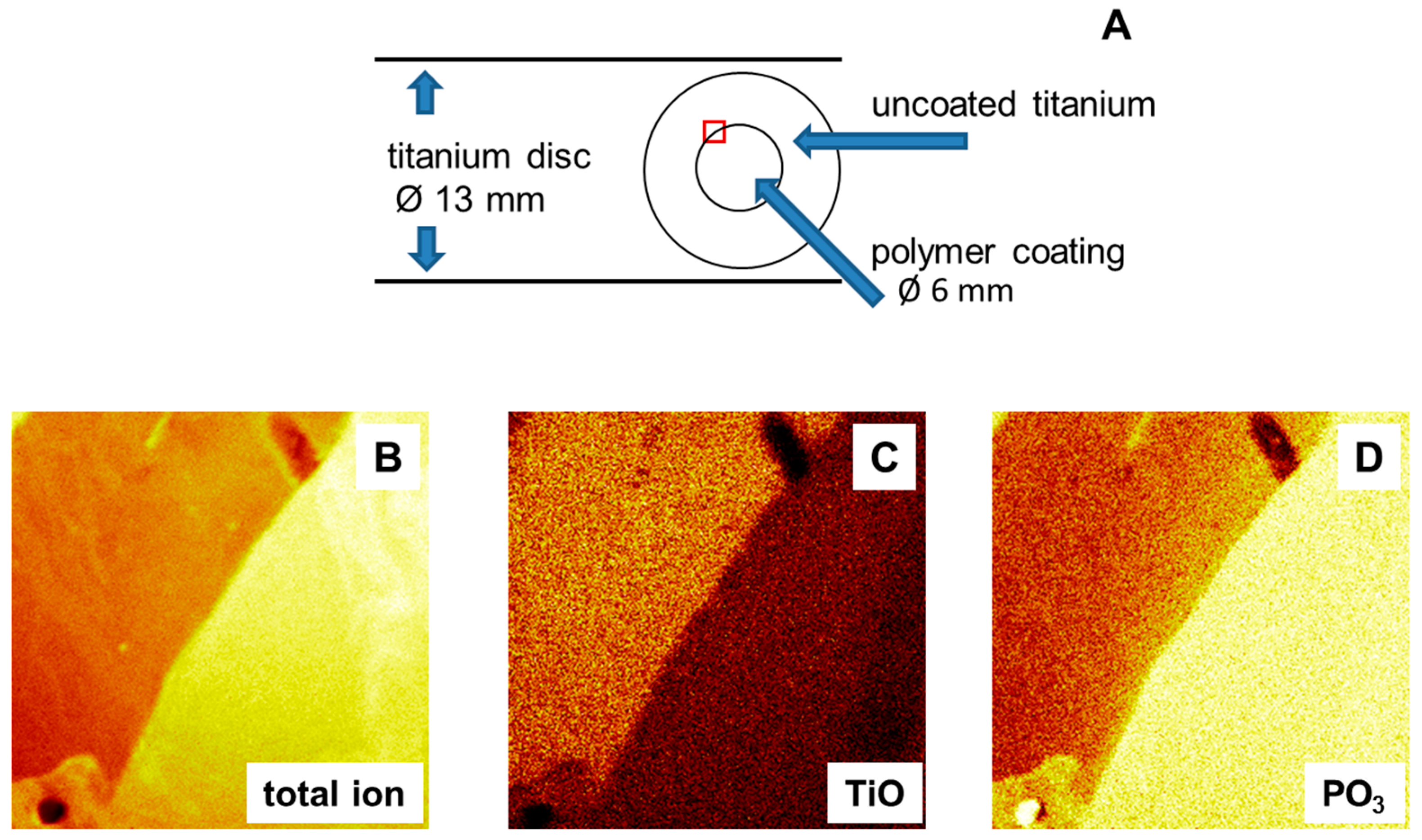
2.1. Implementation of a Semi-Coated Test Model
A well-established approach to determine the biocompatibility or antibacterial activity of a coated substance is to use two different discs per sample run. One disc is coated with the substance and the other is an uncoated disc as control. The limitation of this approach is that—even if there is an antibacterial effect—it remains unclear after microscopy whether this is caused by direct contact or by release of antibacterial substances from the surface coating into the medium. Additional tests would be necessary such as comparison of cfu (colony-forming units) from supernatants or washing solutions to clarify cause and effect. This would include labor-intensive and time-consuming plating and counting processes.
However, if the control experiment and the substance test could be conducted on the same disc, the medium as well as the preparation of bacteria would be the same and the reduction in bacterial adhesion on the coated parts could be directly related to the contact of bacteria with the surface. This is of interest in particular if working with putative microbe-repelling coatings and a broad variety of different surfaces simultaneously. Artifacts due to detached material or soluble substances can be excluded, if there is an unambiguous and defined border between the coated and uncoated areas, as shown in
Figure 1B. Moreover, the demonstration of such a border region is the most convincing argument for the target function of an antibacterial coating by just one picture. Thus, the development of an easy and fast partially coating strategy would save material, time and effort regarding preparation and performance of assays in comparison to the conventional approach with completely coated discs if presuming that no artefacts by this new technique occur and for completely coated discs additional microbiological (colony-forming units) or molecular biological analyses would be necessary to address the same mentioned limitation.
For this reason, a new method was designed: Semi-coated titanium discs were prepared with the help of an adhesive foil as template; see
Figure 1A. The sample disc was covered with the foil, except 6 mm of the internal diameter, which exposed the pure titanium oxide surface. During the spin coating process, copolymer solution was spread over the whole sample disc. The adhesive foil was removed before tempering, leaving a copolymer film on only the unlaminated part. Sample discs were then heated in an oven at 120 °C for 19 h to finish the binding of the copolymer to the titanium oxide surface. The result were semi-coated titanium discs, with a polymer coated center surrounded by a pure titanium oxide surface, see
Figure 1B.
The influence of the foil or the lamination itself was investigated to confirm that the properties of the primary foil laminated part and the pure titanium surface were the same. Contact angle measurements, ellipsometric measurements and tests of antibacterial activity were conducted on titanium surfaces before and after covering with an adhesive foil while passing through the normal coating cycle, e.g., annealing the sample at 120 °C for 19 h.
Figure 1.
(A) Sketch of titanium disc and coating pattern; (B–D) Secondary ion mass spectrometry images (image size: 500 µm × 500 µm); (B) Total ion image at the border between coated (bright yellow) and uncoated areas; and (C,D) secondary ion mass spectrometry (SIMS) images of the coated area for different ions.
Figure 1.
(A) Sketch of titanium disc and coating pattern; (B–D) Secondary ion mass spectrometry images (image size: 500 µm × 500 µm); (B) Total ion image at the border between coated (bright yellow) and uncoated areas; and (C,D) secondary ion mass spectrometry (SIMS) images of the coated area for different ions.
A layer with a thickness in the range of 2.0 ± 0.1 nm was observed after removing the adhesive foil. Contact angles were in the range of θ
adv = 66° ± 2° and θ
rec = 45° ± 4°, see
Table 1. These results indicated a change in the passivating oxide layer on titanium surfaces. The oxide forms within seconds on the bare metal surface and grows further as a function of time and temperature [
11]. Natural oxide layers reported in the literature are in the range of 3–7 nm [
12]. Therefore titanium substrates have contact angles of θ
adv = 33° and θ
rec = 22° directly after polishing and washing. The contact angles increased to θ
adv = 84° and θ
rec = 64° for aged titanium samples [
9]. The observed shift to higher contact angles and the increase in layer thickness after annealing the titanium samples can be explained with a temperature-accelerated aging process, resulting in an increased oxide layer. The adhesive foil itself did not modify the surface properties.
2.2. Copolymer Coating
In previous studies, the statistical copolymer (
i.e., a copolymer with a statistical sequence of the two monomers) consisting of 4-vinyl-
N-hexylpyridinium bromide (VP) and dimethyl(2-methacryloyloxyethyl)phosphonate (DMMEP) was intensively investigated with respect to the influence of copolymer composition on the balance between biocompatibility and antimicrobial activity [
9,
10]. A composition of 0.24 VP and 0.76 DMMEP (VP:DMMEP 30:70) was identified as optimal and chosen as a test substance to compare the new semi-coated sample testing method to the standard method using coated and uncoated samples.
Table 1.
Coating characteristics of VP:DMMEP 30:70.
Table 1.
Coating characteristics of VP:DMMEP 30:70.
| Coating Method | Sample | Layer Thickness (nm) | Contact Angle (°) |
|---|
| θadv | θrec |
|---|
| semi-coated | VP:DMMEP 30:70 | 5.0 ± 0.6 | 68 ± 2 | 48 ± 6 |
| titanium area of semi-coated sample | 2.0 ± 0.1 | 66 ± 2 | 45 ± 4 |
| completely coated | VP:DMMEP 30:70 | 5.1 ± 1.4 | 64 ± 3 | 43 ± 2 |
| uncoated | pure titanium | 3–7 c | 33 a | 22 a |
| 84 b | 64 b |
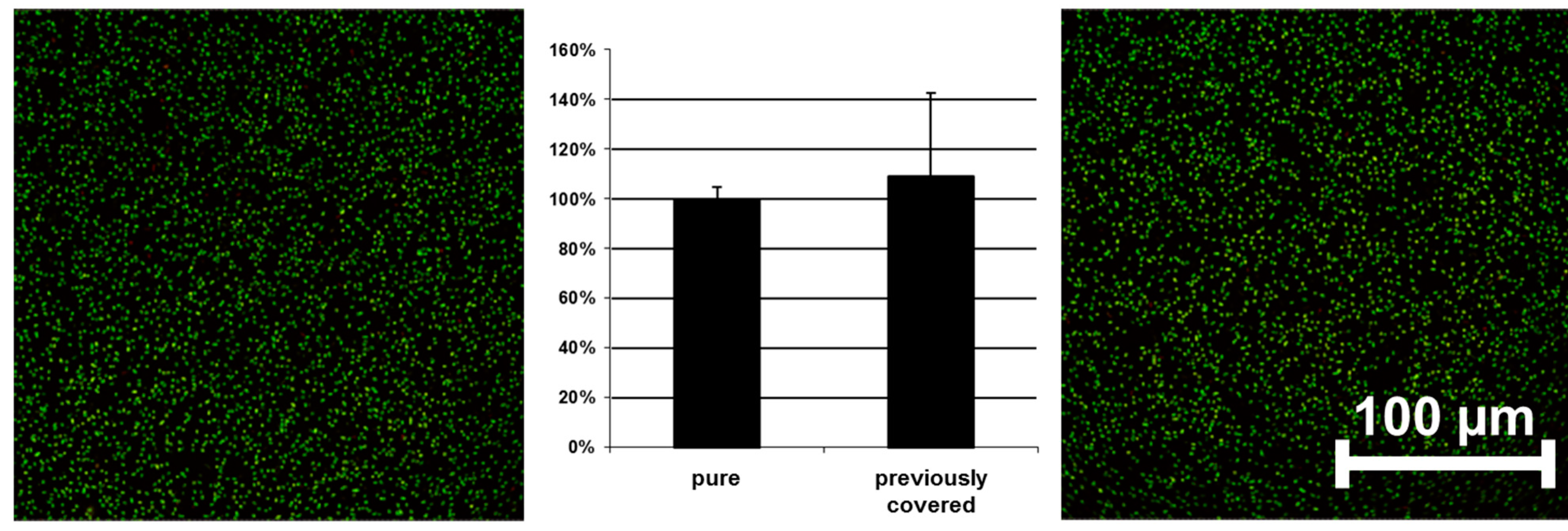
Tests on antibacterial activity were conducted on these surfaces, as shown in
Figure 2. No significant difference could be observed between a pure and a previously covered titanium surface. The results of the control experiments clearly indicate that the newly designed semi-coated test model can be used to study the influence of an innovative copolymer coating on different bacteria.
Figure 2.
Adhesion of bacteria on pure (left) and previously covered surfaces (right) without polymer coating (E. coli used as example)—confocal laser scanning microscopy (CLSM) pictures and quantified data. Scale bar = 100 μm.
Figure 2.
Adhesion of bacteria on pure (left) and previously covered surfaces (right) without polymer coating (E. coli used as example)—confocal laser scanning microscopy (CLSM) pictures and quantified data. Scale bar = 100 μm.
In
Table 1, coating characteristics are compiled from different experimental runs, including layer thickness and water contact angle. Comparison of the layer thicknesses of VP:DMMEP 30:70 on completely coated and semi-coated titanium substrates showed that neither coating method affected layer formation. Layer thicknesses of about 5 nm could be obtained, so that no polymer was removed from the surface during detachment of the template. Contact angles varied only slightly and within the error of the measurements, from θ
adv 64° ± 3° for complete coating to θ
adv 68° ± 2° for semi-coated samples and θ
rec 43° ± 2° to θ
rec 48° ± 6°, respectively. These specifications provide additional proof for the applicability of the semi-coating method.
In addition, imaging of the chemical surface composition was conducted by secondary ion mass spectrometry (TOF-SIMS); see
Figure 1B–D. An image representing the total ion flux of the polymer coated surface is shown in
Figure 1B. A distinct border between coated and uncoated areas could be identified, thus confirming the presence of a semi-coated surface. Binding of the copolymer is related to a reaction between the OH-groups of the metal oxide surface and the phosphonate groups of the DMMEP component. The phosphorous-containing ions can therefore be taken as an indicator of polymer coating (
Figure 1D). This was further confirmed using the CN ions (data not shown), as nitrogen atoms are present in the copolymer in the VP fraction. TOF-SIMS measurements are highly surface sensitive, so that the signal intensity of titanium ions detected beneath the coating was lower than in the uncoated part (
Figure 1C).
2.3. Antibacterial Activity
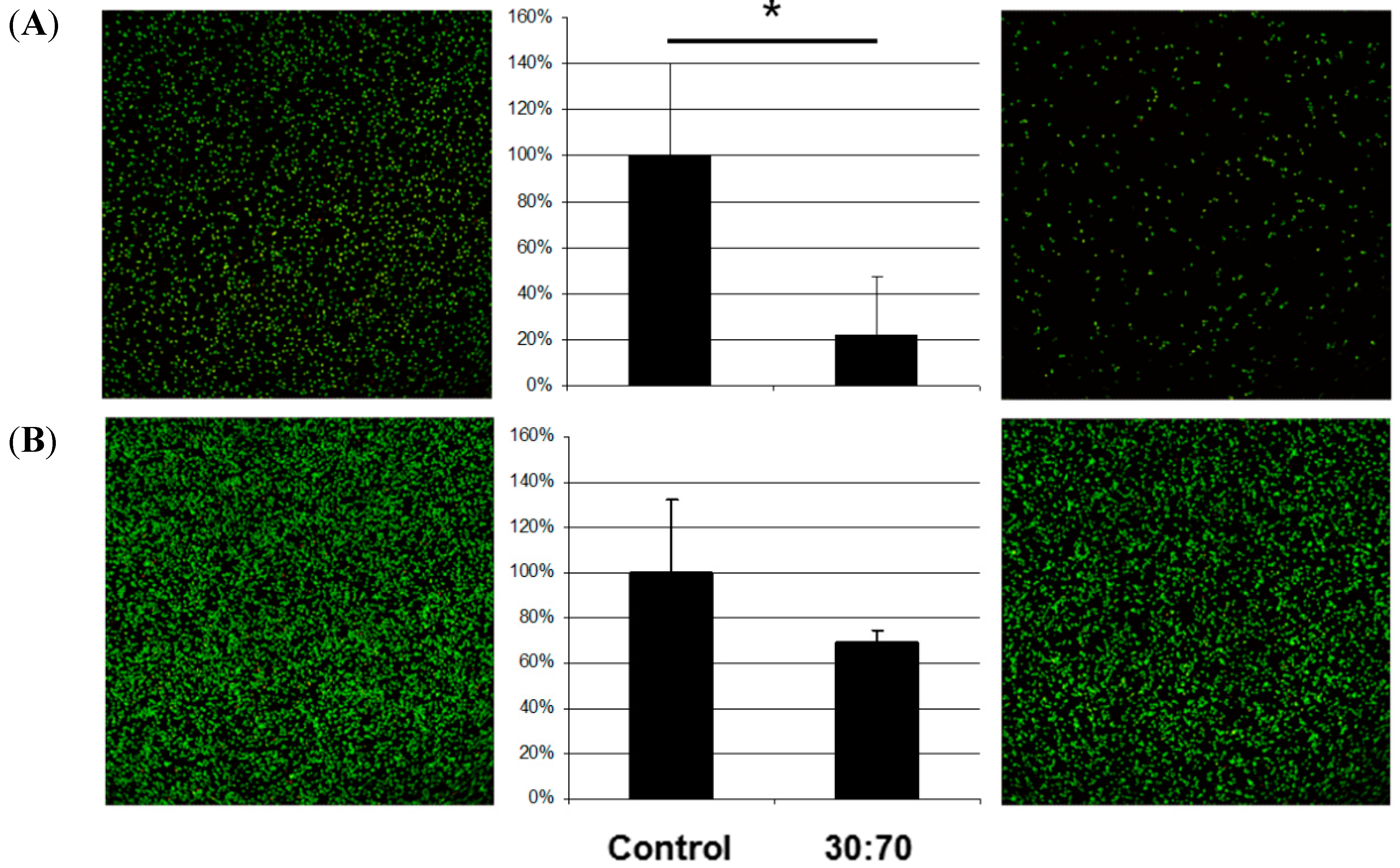
The effect on bacteria by the coating depended on the species (
Figure 3).
S. aureus and
S. epidermidis are members of the natural skin flora. When displaced inside the body they can nevertheless cause serious infections, especially in enervated or immune deficient persons; they are common in peri-implant infections after orthopedic or trauma surgery [
13]. In this study, coating with VP:DMMEP 30:70 led to a 90% decrease in the bacterial load of
S. aureus and
S. epidermidis after 5 h of incubation in comparison to uncoated samples. This confirmed results from a previous study with only 1 h incubation [
14]. Similar or even better results could be achieved for
S. sanguinis, a primary colonizer of human teeth and dental implants. Therefore, coating dental or orthopedic implants with VP:DMMEP is an option in the future worth additional study.
Figure 3.
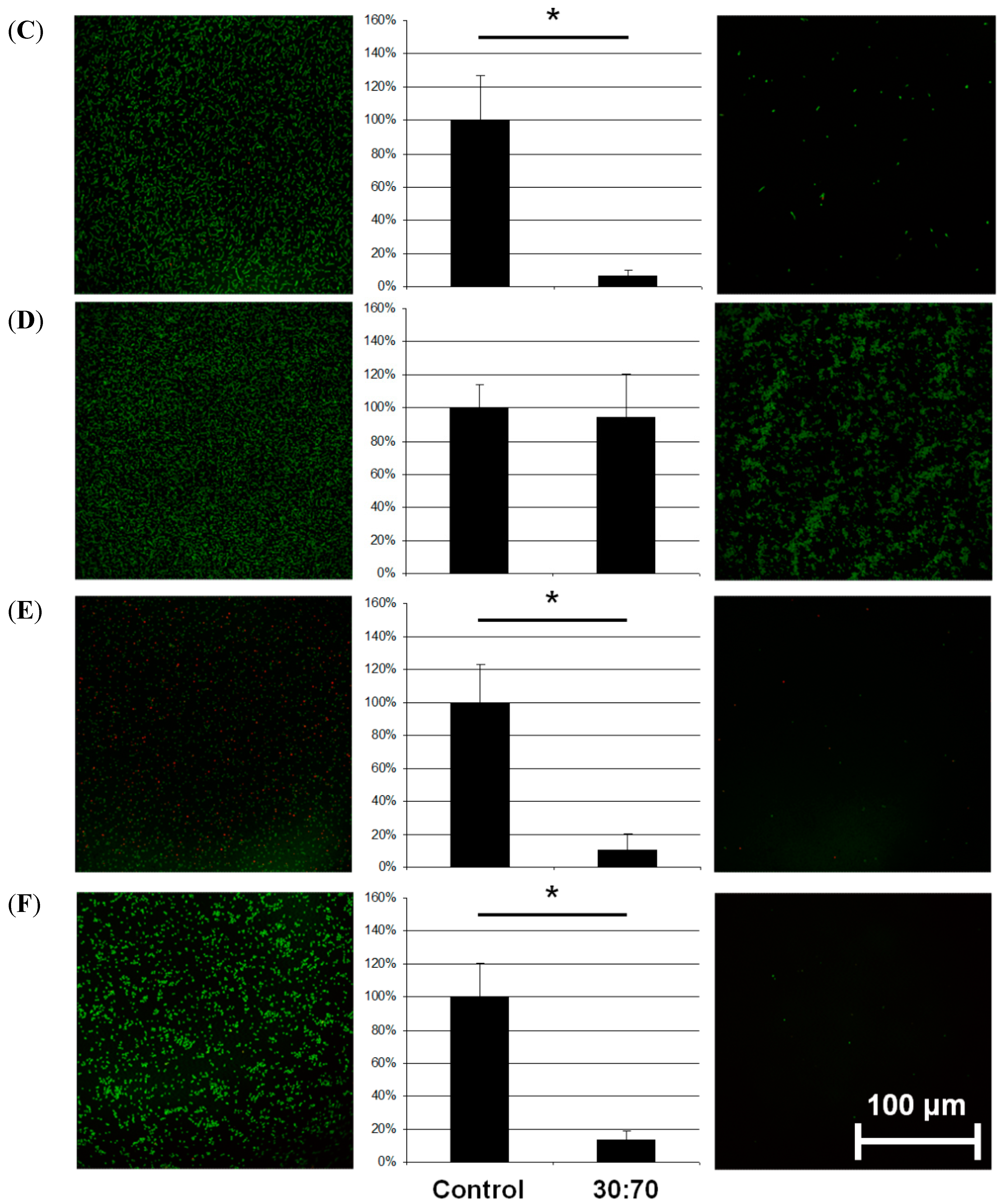
Adhesion of different bacterial species (A = E. coli; B = P. aeruginosa; C = S. sanguinis; D = S. mutans; E = S. aureus; F = S. epidermidis) on titanium discs coated with VP:DMMEP 30:70 (right) relative to the uncoated control (left)—CLSM pictures and quantified data (* p < 0.05). Scale bar = 100 μm.
Figure 3.
Adhesion of different bacterial species (A = E. coli; B = P. aeruginosa; C = S. sanguinis; D = S. mutans; E = S. aureus; F = S. epidermidis) on titanium discs coated with VP:DMMEP 30:70 (right) relative to the uncoated control (left)—CLSM pictures and quantified data (* p < 0.05). Scale bar = 100 μm.
Regarding the adhesion of E. coli, a typical pathogen in smear infections, the coating was slightly less effective than in the cases of S. aureus, S. epidermidis or S. sanguinis, but still exhibited distinct antibacterial activity. In comparison with streptococci and staphylococci, the gram-negative E. coli bacteria are mobile and less influenced by changes in adhesion. The same argument holds for P. aeruginosa, which forms biofilms on lens tubes (in cases of long-term artificial respiration) and devices in otolaryngology causing serious infections in these areas. However, the antibacterial effect for this species was less than 40% and not significant. Accordingly, a coating of devices in otolaryngology or on lens tubes might be less effective.
In contrast to previous results with
S. mutans [
9] the present experiments showed no clear reduction in bacterial adhesion. However, the bacteria did not adhere homogenously to the surface, but aggregated to some extent. Such aggregation has been interpreted as a stress reaction after bacteria are placed on an unfavorable surface [
15] and has been observed for other combinations of VP:DMMEP copolymers with higher VP proportions, as well as with other bacterial species [
9,
10]. In the current study we incubated the bacteria for 5 h, in contrast to 1 h in previous studies. The results indicate that
S. mutans was initially barely able to adhere to the coating, but grows nevertheless, although it is stressed by the surface.
Our approach aimed to the initial healing process directly after implant insertion, when bacteria and periimplant host tissue compete in adhesion to the implant surface. In this period a significant reduction of bacterial colonization might give cells the advantage in this “race to the surface” to establish a stable wound closure that prevents biofilm progression. Since the adhesion of some bacterial species is reduced, but the proportion of dead bacteria (detected by LIVE/DEAD staining) remains stable in this study (data not shown), VP:DMMEP 30:70 coatings could be regarded as repellent to bacteria [
16]. In recent decades, several coating designs for implant surfaces have been described that employ this approach [
17,
18,
19,
20]. However, most of these studies have focused on only one or two bacterial species. In the light of the wide range of the diversity and characteristics of bacteria in different biofilms, this limits their application
in vivo.
Other approaches used releasing mechanisms, provided killing on contact [
21,
22,
23], or a combination of both [
24,
25]. One unsolved problem is the depletion of the established pool of the antibacterial substance over time, along with the increasing risk of creating resistances within the bacterial species. Another problem is the gradually masking of a bactericide surface by the remaining dead bacteria, which might serve as a safe substrate for subsequent bacteria. However, the mode of action of a new coating and its usefulness and risks can only be reliably assessed when there is unambiguous evidence as to whether the effect is generated by killing on contact or by the release of the coating material.
In order to address the question of whether the observed antibacterial effects are caused just by contact or by soluble fragments of the coating or residues of the coating/washing processes, additional pictures of the border regions on semi-coated titanium discs were taken.
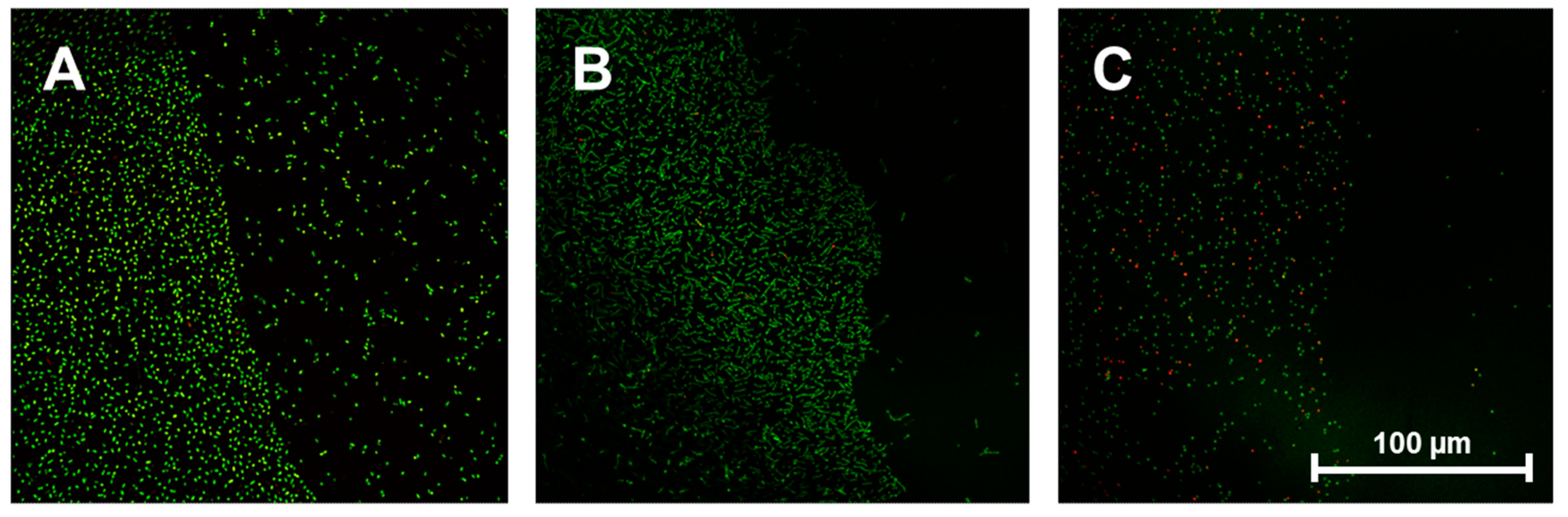
Figure 4A–C show typical results for
E. coli,
S. sanguinis and
S. aureus (which displayed markedly reduced adhesion on VP:DMMEP 30:70). These exhibit a defined border line between coated and uncoated areas of the discs, as bacterial colonization is potently reduced on the polymer coating. This experiment clearly demonstrated that the reported antibacterial effects are related to the direct contact between bacteria and the modified surface, whereas nearby areas free of VP:DMMEP 30:70 showed normal bacterial adhesion, without any signs of putative antibacterial residues or components dissolved during culturing. This confirms the first part of the hypothesis—that the innovative antibacterial copolymer coating is effective against a variety of clinically relevant bacterial strains.
Figure 4.
Border region between uncoated (left side of each picture) and coated areas (right side of each picture) of titanium discs after seeding with E. coli (A), S. sanguinis (B) and S. aureus (C). Scale bar = 100 μm.
Figure 4.
Border region between uncoated (left side of each picture) and coated areas (right side of each picture) of titanium discs after seeding with E. coli (A), S. sanguinis (B) and S. aureus (C). Scale bar = 100 μm.
2.4. Adhesion, Proliferation, and Morphology of Human Fibroblasts
Thus, this titanium surface modification demonstrated a dramatic antibacterial effect for several bacterial species, but might not be able to prevent microbial attachment completely, as has been reported for various polymer coatings [
18,
21,
25,
26]. It is therefore even more important that for certain applications in implant coating (like prosthetic dentistry or orthopedics) the polymer supports or at least permits strong tissue binding to suppress subsequent bacterial growth. We showed in previous studies that some polymer coatings cause a crucial decrease in adhered cells [
9]. It is important to investigate antibacterial effects at a very early stage as well as cytocompatibility, an aspect often overlooked.
Adhesion, proliferation and morphological changes in both gingival and dermis fibroblasts were tested in vitro, in order to verify the compatibility between VP:DMMEP 30:70 and relevant soft tissues. The number of attached cells was calculated from overall lactate dehydrogenase (LDH) activity. Microscopic evaluation was carried out after different incubation times. In all cases non-coated titanium discs served as a reference and related cell numbers were set at 100%.
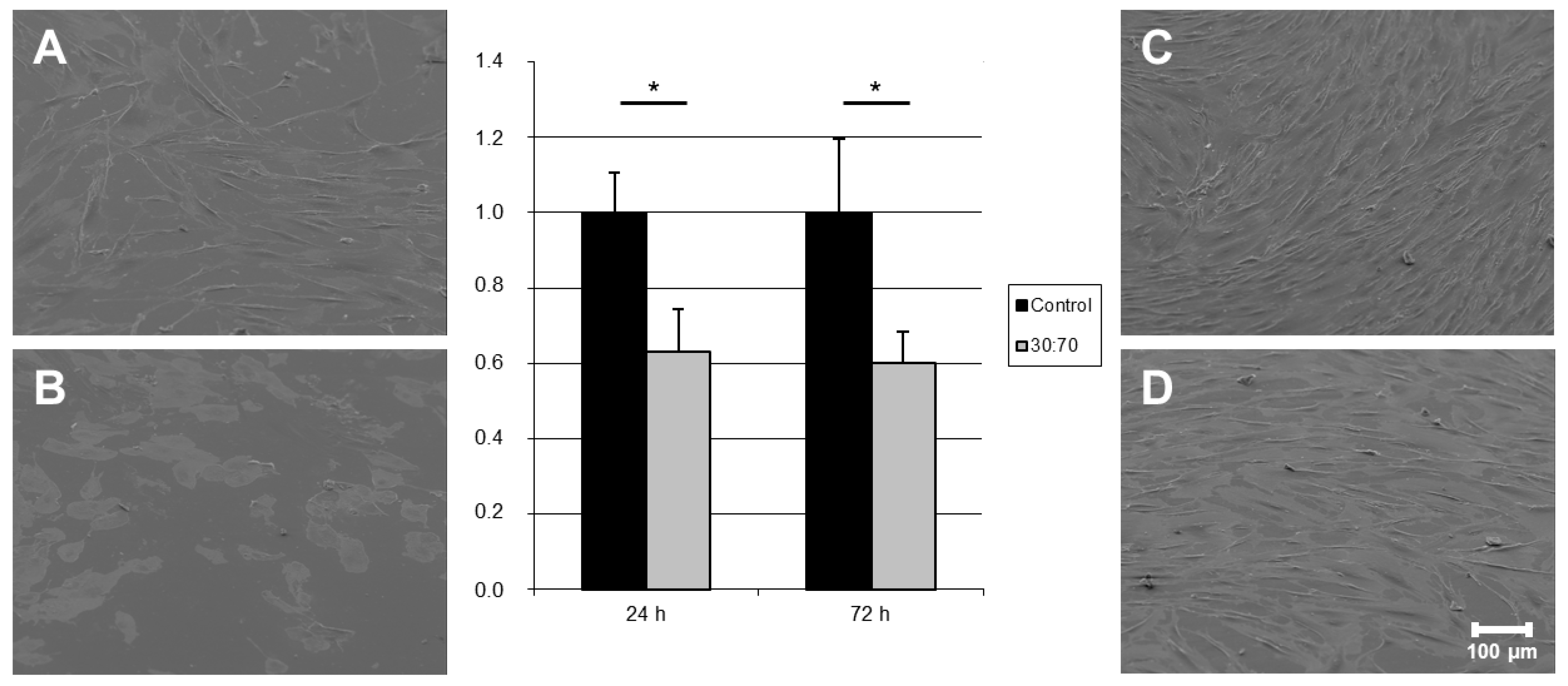
Gingival fibroblasts (HGFIBs) are the most important type of soft tissues surrounding dental implants and are crucial for effective wound closure in an area with constant high bacterial load [
27]. The antibacterial polymer coating VP:DMMEP 30:70 resulted in a reduced initial adhesion of HGFIB to the surface. This was reflected in the significant reduction of adherent cells by 40% compared to the control value (
Figure 5). Such a reduction could arise from an unsuitable chemical composition or topography of the surface, changes in wettability or surface co
ntaminations depending on the used cell type [
28]. Since topography and water contact angle varied only slightly, the inefficient focal adhesion of HGFIBs on the specific chemical composition of the statistical copolymer might be responsible for this effect. This would match also to the higher proportion of round fibroblastic cells in comparison to the uncoated control visualized in scanning electron microscope (SEM) after 24 h (
Figure 5A,B). However, these effects seem to apply just to the adhesion process itself. Once completely adhered, gingival fibroblast behaved adequately on both surfaces, with normal fibroblast morphology and comparable growth rates (
Figure 5C,D). No signs of cell toxicity in terms of a reduction in proliferation were detected, as these would have caused a significantly lowered cell number (in %) after 72 h. Instead we got a reduction of about 40% in relation to the uncoated titanium at both time-points without any significant difference indicating that on both surfaces (coated with VP:DMMEP 30:70 or uncoated titanium) the proliferation rate within 48 h is nearly the same. A detailed analysis of the reasons for the reduced initial adhesion despite an unaffected proliferation and viability was beyond the scope of the presented study but should be addressed in further investigations.
Adhesion of human dermis fibroblasts (HDFIBs) is an essential part of skin- and wound-related healing processes and is important for all transcutaneous prostheses, catheters or similar devices. Fibroblasts from dermis showed only a slight but in case of 24 h still significant reduction in adhered cell numbers on coated samples—in contrast to HGFIBs—with no morphological abnormalities (
Figure 6). Such a different behavior of divers cell types on the same surface was described already for topographies [
29] and chemical binding sites [
30] demonstrating specific dependence on a suitable distribution of adhesion points and composition of chemical receptors. The decrease of about 20% appeared stable between the first and third days of incubation. This observation indicates again a comparable proliferation rate of cells on uncoated material and was supported by SEM images.
Figure 5.
Quantification and visualization (SEM) of adhered human gingival fibroblasts on titanium discs coated with VP:DMMEP 30:70 (B = 24 h; D = 72 h) in relation to the uncoated control (A = 24 h; C = 72 h)—SEM pictures and quantified data (* p < 0.05). Scale bar = 100 μm.
Figure 5.
Quantification and visualization (SEM) of adhered human gingival fibroblasts on titanium discs coated with VP:DMMEP 30:70 (B = 24 h; D = 72 h) in relation to the uncoated control (A = 24 h; C = 72 h)—SEM pictures and quantified data (* p < 0.05). Scale bar = 100 μm.
In conclusion, coating titanium surfaces with the copolymer VP:DMMEP 30:70 has no negative impact on biocompatibility, as assessed by the proliferation and viability of cells isolated from peri-implant soft tissue, although initial adhesion might be affected. Therefore, the second part of the hypothesis—that the polymer coating exhibits sufficient adhesion of human cells—is confirmed. However, especially regarding human gingival fibroblasts further optimization of the copolymer composition should have the objective of improving initial cell adhesion onto the coating, as this would accelerate attachment of sealing tissue. The fast and severe adhesion of connective tissue remains one of the major differences between implants and native structures, which prevents epithelium downgrowth and bacteria proceeding [
28].
Figure 6.
Quantification and visualization (SEM) of adhered human dermis fibroblasts on titanium discs coated with VP:DMMEP 30:70 (B = 24 h; D = 72 h) in relation to the uncoated control (A = 24 h; C = 72 h)—SEM pictures and quantified data (* p < 0.05). Scale bar = 100 μm.
Figure 6.
Quantification and visualization (SEM) of adhered human dermis fibroblasts on titanium discs coated with VP:DMMEP 30:70 (B = 24 h; D = 72 h) in relation to the uncoated control (A = 24 h; C = 72 h)—SEM pictures and quantified data (* p < 0.05). Scale bar = 100 μm.