Antimicrobial Polymeric Materials with Quaternary Ammonium and Phosphonium Salts
Abstract
:1. Introduction
2. Polymeric Biocides with Pendant Quaternary Ammonium/Phosphonium Salts
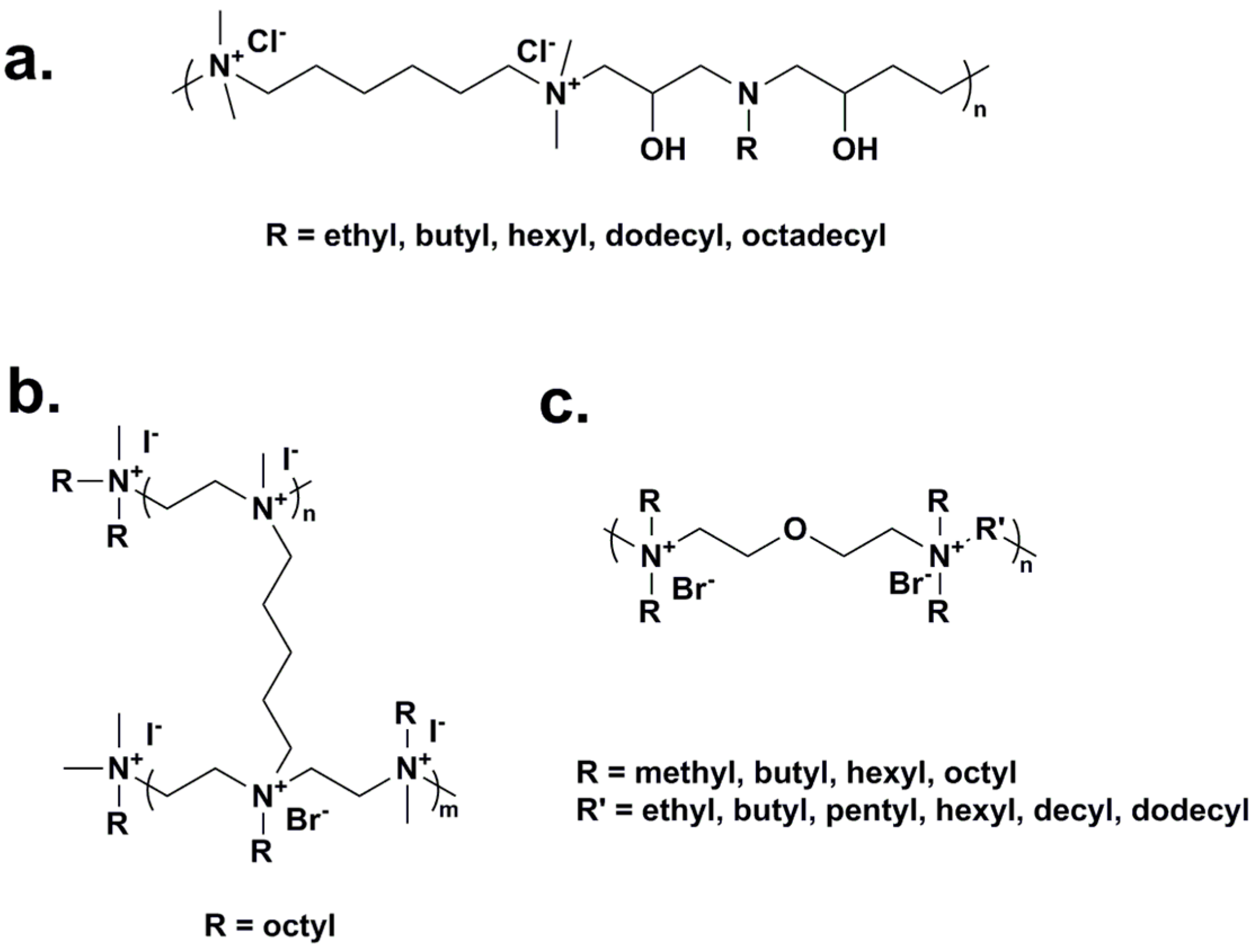
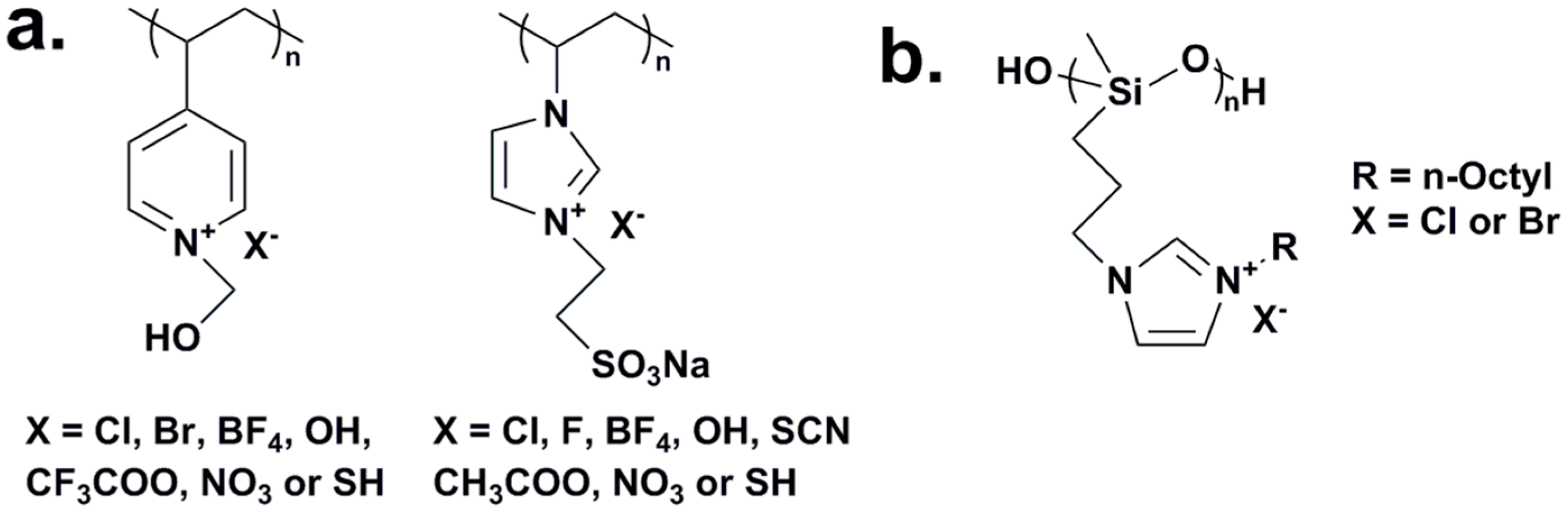
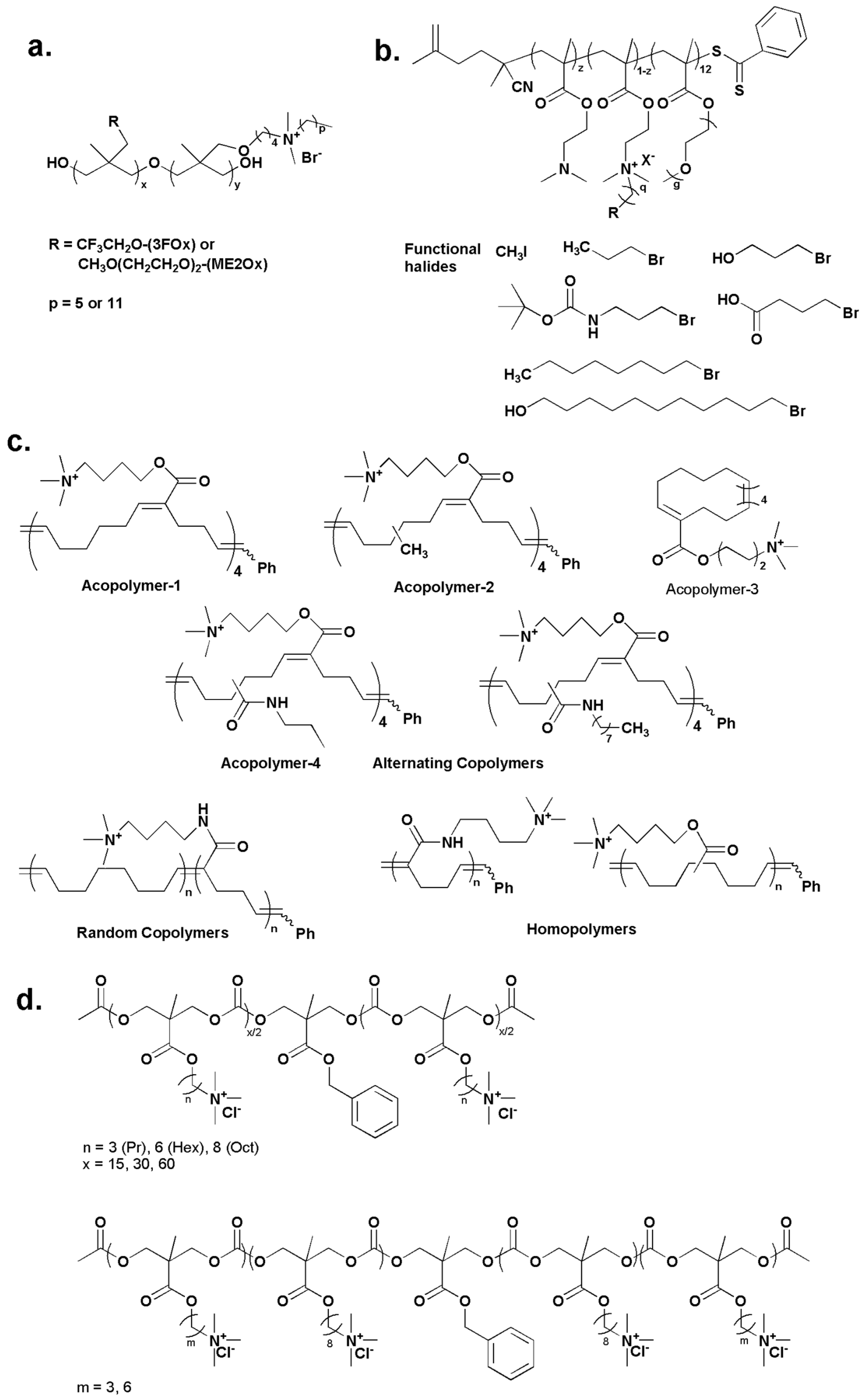
2.1. Water-Soluble Quaternary Ammonium/Phosphonium Polymers
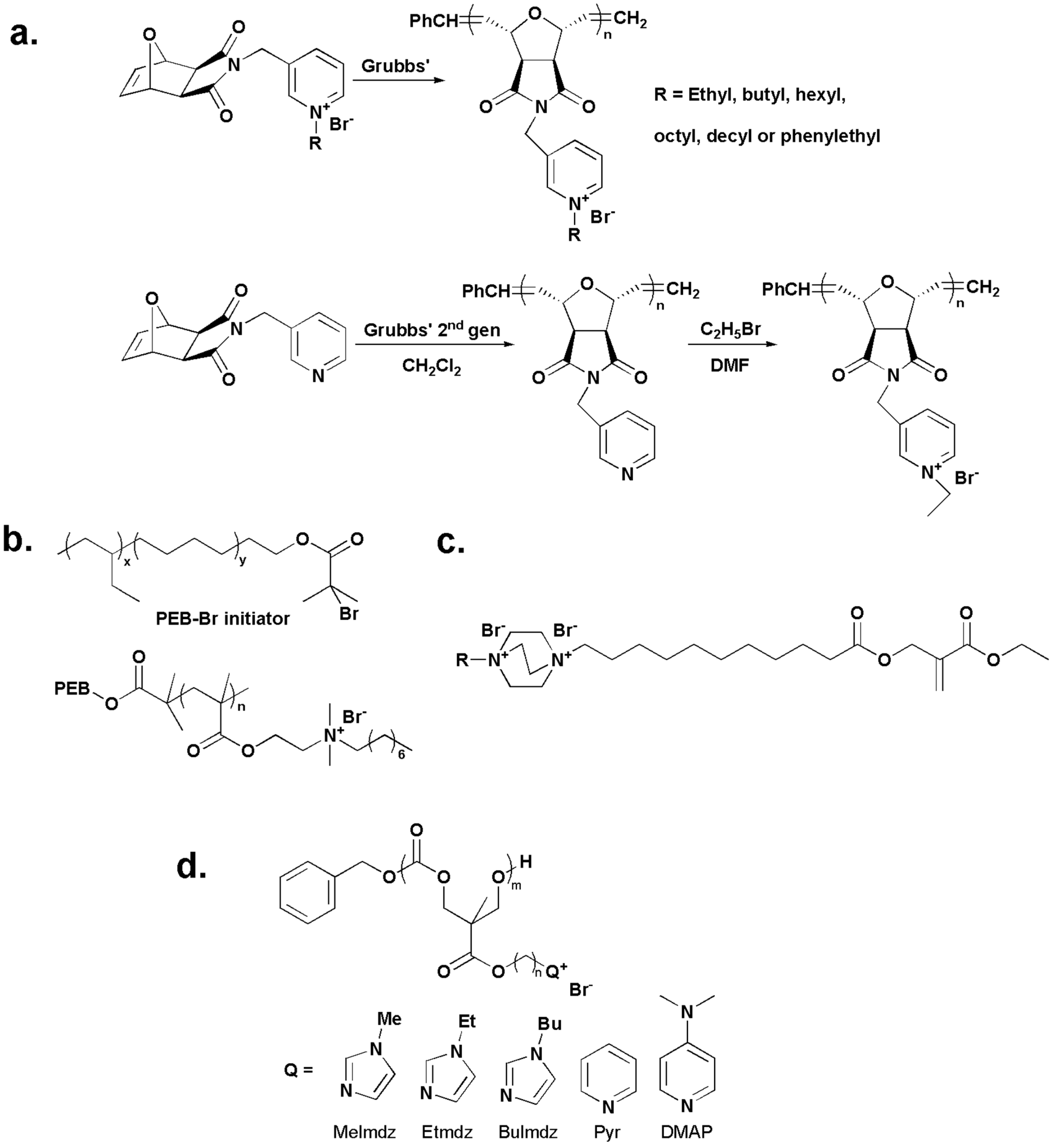
2.2. Water-Insoluble Quaternary Ammonium/Phosphonium Polymers
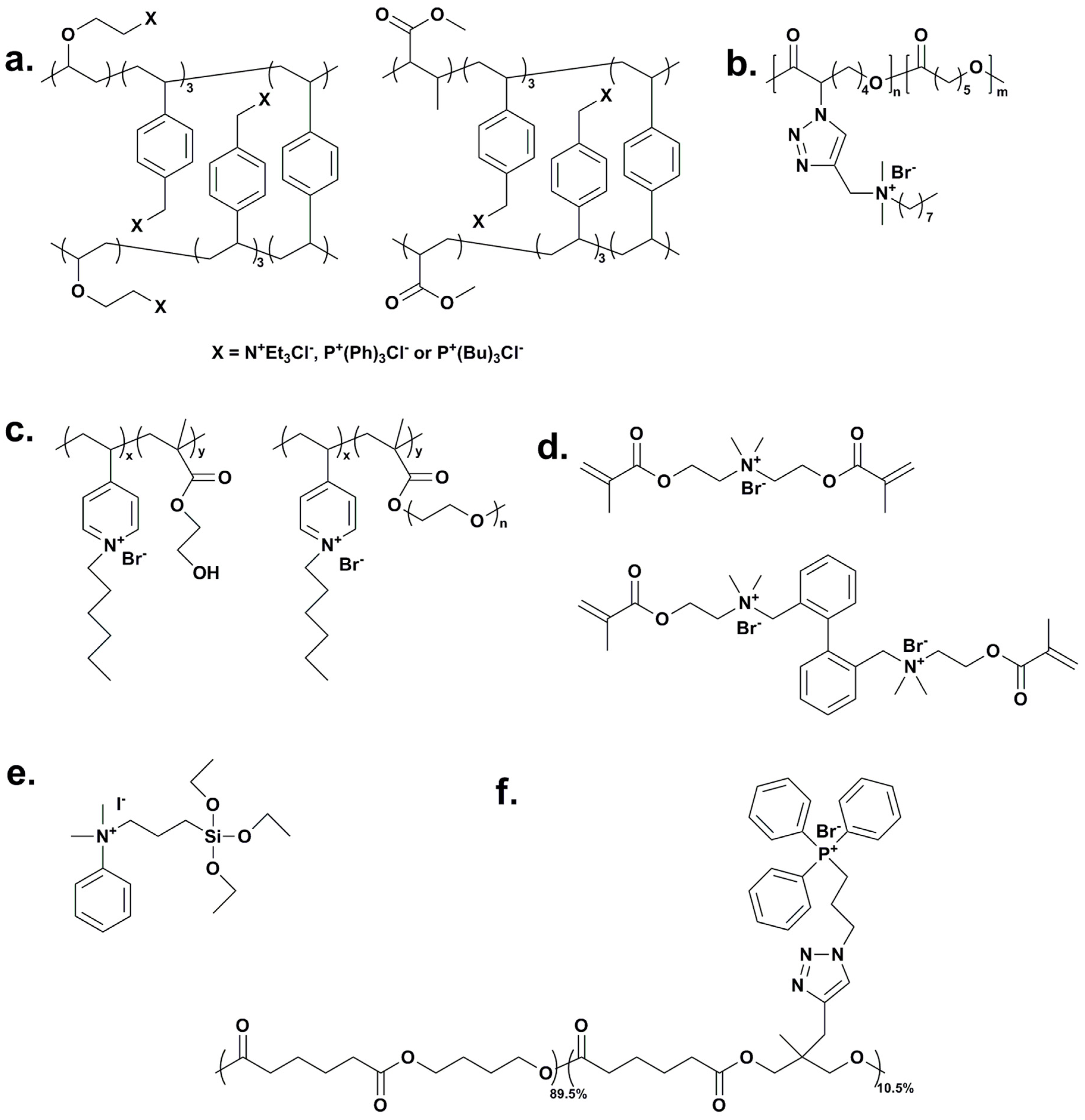
3. Polymers with Quaternary Ammonium/Phosphonium Salt within the Main Chain
4. Hyper-Branched and Dendritic Polymers

| Step | Low-Molecular-Weight Biocides | Polymeric Biocides | Dendritic Biocides |
|---|---|---|---|
| Initial adsorption | Low | High | High |
| Diffusion past the cell wall | High | Low | Medium |
| Binding to the membrane | Low | Medium | High |
| Disruption of the membrane | Low | Medium | High |
5. Immobilization of QAS/QPS on Material Surfaces
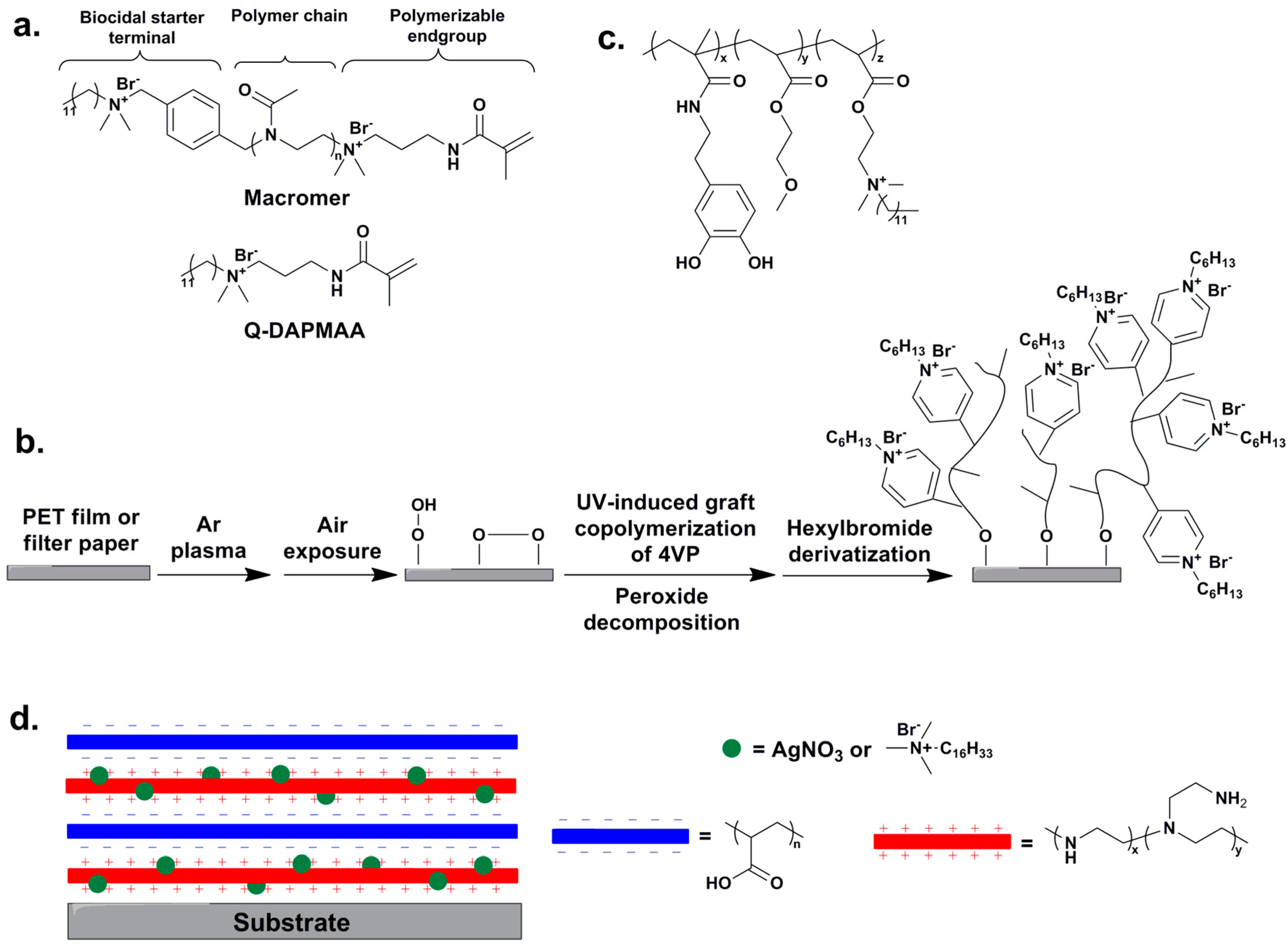
6. Effect of Counter Anion and Amphiphilic Balance on the Antimicrobial Activity
6.1. Counter Anion
6.2. Hydrophobicity-Hydrophilicity Balance
7. Comparison of Antimicrobial Activity between Polymeric QAS and Polymeric QPS
8. Conclusions and Future Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fauci, A.S.; Touchette, N.A.; Folkers, G.K. Emerging infectious diseases: A 10-year perspective from the National Institute of Allergy and Infectious Diseases. Emerg. Infect. Dis. 2005, 11, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, G.J.; Som, A.; Madkour, A.E.; Eren, T.; Tew, G.N. Infectious disease: Connecting innate immunity to biocidal polymers. Mater. Sci. Eng. R. Rep. 2007, 57, 28–64. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Senapati, D.; Khan, S.A.; Singh, A.K.; Hamme, A.; Yust, B.; Sardar, D.; Ray, P.C. Popcorn-shaped magnetic core-plasmonic shell multifunctional nanoparticles for the targeted magnetic separation and enrichment, label-free SERS imaging, and photothermal destruction of multidrug-resistant bacteria. Chem. Eur. J. 2013, 19, 2839–2847. [Google Scholar] [CrossRef] [PubMed]
- Gould, I.M. A review of the role of antibiotic policies in the control of antibiotic resistance. J. Antimicrob. Chemother. 1999, 43, 459–465. [Google Scholar] [CrossRef] [PubMed]
- Stone, A. Microbicides: A new approach to preventing HIV and other sexually transmitted infections. Nat. Rev. Drug Discov. 2002, 1, 977–985. [Google Scholar] [CrossRef] [PubMed]
- Boucher, H.W.; Talbot, G.H.; Bradley, J.S.; Edwards, J.E., Jr.; Gilbert, D.; Rice, L.B.; Scheld, M.; Spellberg, B.; Bartlett, J. Bad bugs, no drugs: No ESKAPE! An update from the Infectious Diseases Society of America. Clin. Infect. Dis. 2009, 48, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Paino, M.; Muñoz-Bonilla, A.; López-Fabal, F.; Gómez-Garcés, J.L.; Heuts, J.P.A.; Fernández-García, M. Effect of glycounits on the antimicrobial properties and toxicity behavior of polymers based on quaternized DMAEMA. Biomacromolecules 2015, 16, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Talbot, G.H.; Bradley, J.; Edwards, J.E., Jr.; Gilbert, D.; Scheid, M.; Bartlett, J.G. Bad bugs need drugs: An update on the development pipeline from the Antimicrobial Availability Task Force of the Infectious Diseases Society of America. Clin. Infect. Dis. 2006, 42, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.M. The role of healthcare strategies in controlling antibiotic resistance. Br. J. Nurs. 2013, 22, 1066–1074. [Google Scholar] [CrossRef] [PubMed]
- Burman, W.J. Rip van winkle wakes up: Development of tuberculosis treatment in the 21st century. Clin. Infect. Dis. 2010, 50, S165–S172. [Google Scholar] [CrossRef] [PubMed]
- Christian, K.A.; Ijaz, K.; Dowell, S.F.; Chow, C.C.; Chitale, R.A.; Bresee, J.S.; Mintz, E.; Pallansch, M.A.; Wassilak, S.; McCray, E.; et al. What we are watching-five top global infectious disease threats, 2012: A perspective from CDCʼs global disease detection operations center. Emerg. Health Threats J. 2013, 6, 1–8. [Google Scholar] [PubMed]
- Fuchs, A.D.; Tiller, J.C. Contact-active antimicrobial coatings derived from aqueous suspensions. Angew. Chem. Int. Ed. 2006, 45, 6759–6762. [Google Scholar] [CrossRef]
- Thomassin, J.-M.; Lenoir, S.; Riga, J.; Jérôme, R.; Detrembleur, C. Grafting of poly[2-(tert-butylamino)ethyl methacrylate] onto polypropylene by reactive blending and antibacterial activity of the copolymer. Biomacromolecules 2007, 8, 1171–1177. [Google Scholar] [CrossRef] [PubMed]
- Ilker, M.F.; Nüsslein, K.; Tew, G.N.; Coughlin, E.B. Tuning the hemolytic and antibacterial activities of amphiphilic polynorbornene derivatives. J. Am. Chem. Soc. 2004, 126, 15870–15875. [Google Scholar] [CrossRef] [PubMed]
- Dong, C.; Ye, Y.; Qian, L.; Zhao, G.; He, B.; Xiao, H. Antibacterial modification of cellulose fibers by grafting β-cyclodextrin and inclusion with ciprofloxacin. Cellulose 2014, 21, 1921–1932. [Google Scholar] [CrossRef]
- Majumdar, P.; Lee, E.; Gubbins, N.; Stafslien, S.J.; Daniels, J.; Thorson, C.J.; Chisholm, B.J. Synthesis and antimicrobial activity of quaternary ammonium-functionalized POSS (Q-POSS) and polysiloxane coatings containing Q-POSS. Polymer 2009, 50, 1124–1133. [Google Scholar] [CrossRef]
- Malmsten, M. Antimicrobial and antiviral hydrogels. Soft Matter 2011, 7, 8725–8736. [Google Scholar] [CrossRef]
- Zheng, A.; Xue, Y.; Wei, D.; Li, S.; Xiao, H.; Guan, Y. Synthesis and characterization of antimicrobial polyvinyl pyrrolidone hydrogel as wound dressing. Soft Mater. 2014, 12, 179–187. [Google Scholar] [CrossRef]
- Domagk, G. A new class of disinfectants. Dtsch. Med. Wochenschr. 1935, 61, 829–832. [Google Scholar] [CrossRef]
- Fu, E.; McCue, K.; Boesenberg, D. Chemical disinfection of hard surfaces-household, industrial and institutional settings. In Handbook for Cleaning/Decontamination of Surfaces, 1st ed.; Johansson, I., Somasundaran, P., Eds.; Elsevier: Oxford, UK, 2007; Volume 1, pp. 573–592. [Google Scholar]
- Tezel, U.; Pavlostathis, S.G. Role of quaternary ammonium compounds on antimicrobial resistance in the environment. In Antimicrobial Resistance in the Environment, 1st ed.; Keen, P.L., Montforts, M.H.M.M., Eds.; John Wiley and Sons: Hoboken, NJ, USA, 2011; pp. 349–387. [Google Scholar]
- Kourai, H.; Yabuhara, T.; Shirai, A.; Maeda, T.; Nagamune, H. Syntheses and antimicrobial activities of a series of new bis-quaternary ammonium compounds. Eur. J. Med. Chem. 2006, 41, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ding, M.; Zhou, L.; Tan, H.; Li, J.; Xiao, H.; Li, J.; Snow, J. Synthesis and antibacterial characterization of gemini surfactant monomers and copolymers. Polym. Chem. 2012, 3, 907–913. [Google Scholar] [CrossRef]
- Xiao, H.; Qian, L. Water-soluble antimicrobial polymers for functional cellulose fibres and hygiene paper products. In Polymeric Materials with Antimicrobial Activity: From Synthesis to Applications, 1st ed.; Muñoz-Bonilla, A., Cerrada, M.L., Fernández-García, M., Eds.; Royal Society of Chemistry: Cambridge, UK, 2014; pp. 75–96. [Google Scholar]
- Tan, H.; Xiao, H. Synthesis and antimicrobial characterization of novel l-lysine gemini surfactants pended with reactive groups. Tetrahedron Lett. 2008, 49, 1759–1761. [Google Scholar] [CrossRef]
- Arciola, C.R.; Campoccia, D.; Speziale, P.; Montanaro, L.; Costerton, J.W. Biofilm formation in Staphylococcus implant infections. A review of molecular mechanisms and implications for biofilm-resistant materials. Biomaterials 2012, 33, 5967–5982. [Google Scholar] [CrossRef] [PubMed]
- Buffet-Bataillon, S.; Tattevin, P.; Bonnaure-Mallet, M.; Jolivet-Gougeon, A. Emergence of resistance to antibacterial agents: The role of quaternary ammonium compounds—A critical review. Int. J. Antimicrob. Agents 2012, 39, 381–389. [Google Scholar] [CrossRef] [PubMed]
- Hook, A.L.; Chang, C.Y.; Yang, J.; Luckett, J.; Cockayne, A.; Atkinson, S.; Mei, Y.; Bayston, R.; Irvine, D.J.; Langer, R.; et al. Combinatorial discovery of polymers resistant to bacterial attachment. Nat. Biotechnol. 2012, 30, 868–875. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Ribeiro, A.M.; de Melo Carrasco, L.D. Cationic antimicrobial polymers and their assemblies. Int. J. Mol. Sci. 2013, 14, 9906–9946. [Google Scholar] [CrossRef] [PubMed]
- Thiyagarajan, D.; Goswami, S.; Kar, C.; Das, G.; Ramesh, A. A prospective antibacterial for drug-resistant pathogens: A dual warhead amphiphile designed to track interactions and kill pathogenic bacteria by membrane damage and cellular DNA cleavage. Chem. Commun. 2014, 50, 7434–7436. [Google Scholar] [CrossRef]
- Parent, J.S.; Penciu, A.; Guillén-Castellanos, S.A.; Liskova, A.; Whitney, R.A. Synthesis and characterization of isobutylene-based ammonium and phosphonium bromide ionomers. Macromolecules 2004, 37, 7477–7483. [Google Scholar] [CrossRef]
- Eren, T.; Som, A.; Rennie, J.R.; Nelson, C.F.; Urgina, Y.; Nüsslein, K.; Coughlin, E.B.; Tew, G.N. Antibacterial and hemolytic activities of quaternary pyridinium functionalized polynorbornenes. Macromol. Chem. Phys. 2008, 209, 516–524. [Google Scholar] [CrossRef]
- Lenoir, S.; Pagnoulle, C.; Detrembleur, C.; Galleni, M.; Jérôme, R. New antibacterial cationic surfactants prepared by atom transfer radical polymerization. J. Polym. Sci. Part A 2006, 44, 1214–1224. [Google Scholar] [CrossRef]
- Abel, T.; Cohen, J.I.; Engel, R.; Filshtinskaya, M.; Melkonian, A.; Melkonian, K. Preparation and investigation of antibacterial carbohydrate-based surfaces. Carbohydr. Res. 2002, 337, 2495–2499. [Google Scholar] [CrossRef] [PubMed]
- Dizman, B.; Elasri, M.O.; Mathias, L.J. Synthesis and antimicrobial activities of new water-soluble bis-quaternary ammonium methacrylate polymers. J. Appl. Polym. Sci. 2004, 94, 635–642. [Google Scholar] [CrossRef]
- Nonaka, T.; Hua, L.; Ogata, T.; Kurihara, S. Synthesis of water-soluble thermosensitive polymers having phosphonium groups from methacryloyloxyethyl trialkyl phosphonium chlorides-N-isopropylacrylamide copolymers and their functions. J. Appl. Polym. Sci. 2002, 87, 386–393. [Google Scholar] [CrossRef]
- Ng, V.W.L.; Tan, J.P.K.; Leong, J.; Voo, Z.X.; Hedrick, J.L.; Yang, Y.Y. Antimicrobial polycarbonates: Investigating the impact of nitrogen-containing heterocycles as quaternizing agents. Macromolecules 2014, 47, 1285–1291. [Google Scholar] [CrossRef]
- Codling, C.E.; Maillard, J.Y.; Russell, A.D. Aspects of the antimicrobial mechanisms of action of a polyquaternium and an amidoamine. J. Antimicrob. Chemother. 2003, 51, 1153–1158. [Google Scholar] [CrossRef] [PubMed]
- Guan, Y.; Qian, L.; Xiao, H. Novel anti-microbial host-guest complexes based on cationic β-cyclodextrin polymers and triclosan/butylparaben. Macromol. Rapid Commun. 2007, 28, 2244–2248. [Google Scholar] [CrossRef]
- Lambert, P.A. Mechanisms of action of microbicides. In Russell, Hugo & Ayliffe’s: Principles and Practice of Disinfection, Preservation and Sterilization, 5th ed.; Fraise, A.P., Maillard, J.Y., Sattar, S.A., Eds.; Wiley-Blackwell: Oxford, UK, 2013; pp. 95–107. [Google Scholar]
- Haldar, J.; Kondaiah, P.; Bhattacharya, S. Synthesis and antibacterial properties of novel hydrolyzable cationic amphiphiles. Incorporation of multiple head groups leads to impressive antibacterial activity. J. Med. Chem. 2005, 48, 3823–3831. [Google Scholar] [CrossRef] [PubMed]
- Rawlinson, L.A.B.; Ryan, S.M.; Mantovani, G.; Syrett, J.A.; Haddleton, D.M.; Brayden, D.J. Antibacterial effects of poly(2-(dimethylamino ethyl)methacrylate) against selected gram-positive and gram-negative bacteria. Biomacromolecules 2010, 11, 443–453. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Wu, D.; Fu, R. Studies on the synthesis and antibacterial activities of polymeric quaternary ammonium salts from dimethylaminoethyl methacrylate. React. Funct. Polym. 2007, 67, 355–366. [Google Scholar] [CrossRef]
- Campoccia, D.; Montanaro, L.; Arciola, C.R. A review of the biomaterials technologies for infection-resistant surfaces. Biomaterials 2013, 34, 8533–8554. [Google Scholar] [CrossRef] [PubMed]
- Fantner, G.E.; Barbero, R.J.; Gray, D.S.; Belcher, A.M. Kinetics of antimicrobial peptide activity measured on individual bacterial cells using high-speed atomic force microscopy. Nat. Nanotechnol. 2010, 5, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Qian, L.; Xiao, H.; Zhao, G.; He, B. Synthesis of modified guanidine-based polymers and their antimicrobial activities revealed by AFM and CLSM. ACS Appl. Mater. Interfaces 2011, 3, 1895–1901. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, G.J.; Madkour, A.E.; Dabkowski, J.M.; Nelson, C.F.; Nüsslein, K.; Tew, G.N. Synthetic mimic of antimicrobial peptide with nonmembrane-disrupting antibacterial properties. Biomacromolecules 2008, 9, 2980–2983. [Google Scholar] [CrossRef] [PubMed]
- Fadida, T.; Kroupitski, Y.; Peiper, U.M.; Bendikov, T.; Sela Saldinger, S.; Poverenov, E. Air-ozonolysis to generate contact active antimicrobial surfaces: Activation of polyethylene and polystyrene followed by covalent graft of quaternary ammonium salts. Colloids Surf. B Biointerfaces 2014, 122, 294–300. [Google Scholar] [CrossRef] [PubMed]
- Kenawy, E.-R.; Mahmoud, Y.A.G. Biologically active polymers, 6. Macromol. Biosci. 2003, 3, 107–116. [Google Scholar] [CrossRef]
- Oku, N.; Yamaguchi, N.; Yamaguchi, N.; Shibamoto, S.; Ito, F.; Nango, M. The fusogenic effect of synthetic polycations on negatively charged lipid bilayers. J. Biochem. 1986, 100, 935–944. [Google Scholar] [PubMed]
- Yaroslavov, A.A.; Efimova, A.A.; Lobyshev, V.I.; Kabanov, V.A. Reversibility of structural rearrangements in the negative vesicular membrane upon electrostatic adsorption/desorption of the polycation. BBA—Biomembranes 2002, 1560, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Yaroslavov, A.A.; Melik-Nubarov, N.S.; Menger, F.M. Polymer-induced flip-flop in biomembranes. Acc. Chem. Res. 2006, 39, 702–710. [Google Scholar] [CrossRef] [PubMed]
- Timofeeva, L.; Kleshcheva, N. Antimicrobial polymers: Mechanism of action, factors of activity, and applications. Appl. Microbiol. Biotechnol. 2011, 89, 475–492. [Google Scholar] [CrossRef] [PubMed]
- Lewis, K. Programmed death in bacteria. Microbiol. Mol. Biol. R. 2000, 64, 503–514. [Google Scholar] [CrossRef]
- Anselme, K.; Davidson, P.; Popa, A.M.; Giazzon, M.; Liley, M.; Ploux, L. The interaction of cells and bacteria with surfaces structured at the nanometre scale. Acta Biomater. 2010, 6, 3824–3846. [Google Scholar] [CrossRef] [PubMed]
- Uday, S.P.; Thiyagarajan, D.; Goswami, S.; Adhikari, M.D.; Das, G.; Ramesh, A. Amphiphile-mediated enhanced antibiotic efficacy and development of a payload nanocarrier for effective killing of pathogenic bacteria. J. Mater. Chem. B 2014, 2, 5818–5827. [Google Scholar] [CrossRef]
- Nikaido, H. Prevention of drug access to bacterial targets: Permeability barriers and active efflux. Science 1994, 264, 382–388. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, G.; Russell, A.D. Antiseptics and disinfectants: Activity, action, and resistance. Clin. Microbiol. Rev. 1999, 12, 147–179. [Google Scholar] [PubMed]
- Xue, Y.; Guan, Y.; Zheng, A.; Wang, H.; Xiao, H. Synthesis and characterization of ciprofloxacin pendant antibacterial cationic polymers. J. Biomater. Sci. Polym. Ed. 2012, 23, 1115–1128. [Google Scholar] [CrossRef]
- Thoma, L.M.; Boles, B.R.; Kuroda, K. Cationic methacrylate polymers as topical antimicrobial agents against Staphylococcus aureus nasal colonization. Biomacromolecules 2014, 15, 2933–2943. [Google Scholar] [CrossRef] [PubMed]
- Kenawy, E.R.; Abdel-Hay, F.I.; El-Magd, A.A.; Mahmoud, Y. Biologically active polymers: VII. Synthesis and antimicrobial activity of some crosslinked copolymers with quaternary ammonium and phosphonium groups. React. Funct. Polym. 2006, 66, 419–429. [Google Scholar] [CrossRef]
- Riva, R.; Lussis, P.; Lenoir, S.; Jérôme, C.; Jérôme, R.; Lecomte, P. Contribution of “click chemistry” to the synthesis of antimicrobial aliphatic copolyester. Polymer 2008, 49, 2023–2028. [Google Scholar] [CrossRef]
- Sellenet, P.H.; Allison, B.; Applegate, B.M.; Youngblood, J.P. Synergistic activity of hydrophilic modification in antibiotic polymers. Biomacromolecules 2007, 8, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Antonucci, J.M.; Zeiger, D.N.; Tang, K.; Lin-Gibson, S.; Fowler, B.O.; Lin, N.J. Synthesis and characterization of dimethacrylates containing quaternary ammonium functionalities for dental applications. Dent. Mater. 2012, 28, 219–228. [Google Scholar] [CrossRef] [PubMed]
- Marini, M.; Bondi, M.; Iseppi, R.; Toselli, M.; Pilati, F. Preparation and antibacterial activity of hybrid materials containing quaternary ammonium salts via sol–gel process. Eur. Polym. J. 2007, 43, 3621–3628. [Google Scholar] [CrossRef]
- Anthierens, T.; Billiet, L.; Devlieghere, F.; Du Prez, F. Poly(butylene adipate) functionalized with quaternary phosphonium groups as potential antimicrobial packaging material. Innov. Food Sci. Emerg. 2012, 15, 81–85. [Google Scholar] [CrossRef]
- Tiller, J.C.; Liao, C.J.; Lewis, K.; Klibanov, A.M. Designing surfaces that kill bacteria on contact. Proc. Natl. Acad. Sci. USA 2001, 98, 5981–5985. [Google Scholar] [CrossRef] [PubMed]
- Bieser, A.M.; Tiller, J.C. Mechanistic considerations on contact-active antimicrobial surfaces with controlled functional group densities. Macromol. Biosci. 2011, 11, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Fitzpatrick, R.; Klinger, J.D.; Shackett, K.K. Ionene Polymers and Their Use as Antimicrobial Agents. WO 2002080939, 2002. [Google Scholar]
- Tamami, M.; Salas-de la Cruz, D.; Winey, K.I.; Long, T.E. Structure–property relationships of water-soluble ammonium–ionene copolymers. Macromol. Chem. Phys. 2012, 213, 965–972. [Google Scholar] [CrossRef]
- Rodič, P.; Bratuša, M.; Lukšič, M.; Vlachy, V.; Hribar-Lee, B. Influence of the hydrophobic groups and the nature of counterions on ion-binding in aliphatic ionene solutions. Colloids Surf. A Physicochem. Eng. Asp. 2013, 424, 18–25. [Google Scholar] [CrossRef]
- Williams, S.R.; Long, T.E. Recent advances in the synthesis and structure-property relationships of ammonium ionenes. Prog. Polym. Sci. 2009, 34, 762–782. [Google Scholar] [CrossRef]
- Laschewsky, A. Recent trends in the synthesis of polyelectrolytes. Curr. Opin. Colloid 2012, 17, 56–63. [Google Scholar] [CrossRef]
- Bachl, J.; Zanuy, D.; López-Pérez, D.E.; Revilla-López, G.; Cativiela, C.; Alemán, C.; Díaz, D.D. synergistic computational-experimental approach to improve ionene polymer-based functional hydrogels. Adv. Funct. Mater. 2014, 24, 4893–4904. [Google Scholar] [CrossRef]
- Cakmak, I.; Ulukanli, Z.; Tuzcu, M.; Karabuga, S.; Genctav, K. Synthesis and characterization of novel antimicrobial cationic polyelectrolytes. Eur. Polym. J. 2004, 40, 2373–2379. [Google Scholar] [CrossRef]
- Zheng, A.; Xu, X.; Xiao, H.; Guan, Y.; Li, S.; Wei, D. Preparation of antistatic and antimicrobial polyethylene by incorporating of comb-like ionenes. J. Mater. Sci. 2012, 47, 7201–7209. [Google Scholar] [CrossRef]
- Xu, X.; Xiao, H.; Ziaee, Z.; Wang, H.; Guan, Y.; Zheng, A. Novel comb-like ionenes with aliphatic side chains: Synthesis and antimicrobial properties. J. Mater. Sci. 2013, 48, 1162–1171. [Google Scholar] [CrossRef]
- Beyth, N.; Yudovin-Farber, I.; Bahir, R.; Domb, A.J.; Weiss, E.I. Antibacterial activity of dental composites containing quaternary ammonium polyethylenimine nanoparticles against Streptococcus mutans. Biomaterials 2006, 27, 3995–4002. [Google Scholar] [CrossRef] [PubMed]
- Yudovin-Farber, I.; Beyth, N.; Nyska, A.; Weiss, E.I.; Golenser, J.; Domb, A.J. Surface characterization and biocompatibility of restorative resin containing nanoparticles. Biomacromolecules 2008, 9, 3044–3050. [Google Scholar] [CrossRef] [PubMed]
- Beyth, N.; Houri-Haddad, Y.; Baraness-Hadar, L.; Yudovin-Farber, I.; Domb, A.J.; Weiss, E.I. Surface antimicrobial activity and biocompatibility of incorporated polyethylenimine nanoparticles. Biomaterials 2008, 29, 4157–4163. [Google Scholar] [CrossRef] [PubMed]
- Rembaum, A. Biological activity of ionene polymers. Appl. Polym. Symp. 1973, 2, 299–317. [Google Scholar]
- Rembaum, A.; Senyei, A.E.; Rajaraman, R. Interaction of living cells with polyionenes and polyionene coated surfaces. J. Biomed. Mater. Res. 1977, 11, 101–110. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, T.; Yamaguchi, H.; Tazuke, S. Phase separation in phospholipid bilayers induced by biologically active polycations. BBA—Biomembranes 1990, 1026, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Narita, T.; Ohtakeyama, R.; Nishino, M.; Gong, J.P.; Osada, Y. Effects of charge density and hydrophobicity of ionene polymer on cell binding and viability. Colloid Polym. Sci. 2000, 278, 884–887. [Google Scholar] [CrossRef]
- Narita, T.; Ohtakeyama, R.; Matsukata, M.; Gong, J.P.; Osada, Y. Kinetic study of cell disruption by ionic polymers with varied charge density. Colloid Polym. Sci. 2001, 279, 178–183. [Google Scholar] [CrossRef]
- Mattheis, C.; Zheng, M.; Agarwal, S. Closing one of the last gaps in polyionene compositions: Alkyloxyethylammonium ionenes as fast-acting biocides. Macromol. Biosci. 2012, 12, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Meyers, S.R.; Juhn, F.S.; Griset, A.P.; Luman, N.R.; Grinstaff, M.W. Anionic amphiphilic dendrimers as antibacterial agents. J. Am. Chem. Soc. 2008, 130, 14444–14445. [Google Scholar] [CrossRef] [PubMed]
- Strydom, S.J.; Rose, W.E.; Otto, D.P.; Liebenberg, W.; de Villiers, M.M. Poly(amidoamine) dendrimer-mediated synthesis and stabilization of silver sulfonamide nanoparticles with increased antibacterial activity. Nanomed. Nanotechnol. 2013, 9, 85–93. [Google Scholar] [CrossRef]
- Zarena, A.S.; Gopal, S. Dendrimer a new dimension in targeting biofilms. Mini-Rev. Med. Chem. 2013, 13, 1448–1461. [Google Scholar] [CrossRef] [PubMed]
- Tomalia, D.A. Birth of a new macromolecular architecture: Dendrimers as quantized building blocks for nanoscale synthetic polymer chemistry. Prog. Polym. Sci. 2005, 30, 294–324. [Google Scholar] [CrossRef]
- Astruc, D.; Boisselier, E.; Ornelas, C. Dendrimers designed for functions: From physical, photophysical, and supramolecular properties to applications in sensing, catalysis, molecular electronics, photonics, and nanomedicine. Chem. Rev. 2010, 110, 1857–1959. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, W.; Bohrisch, J.; Laschewsky, A. Synthetic polymers with quaternary nitrogen atoms—Synthesis and structure of the most used type of cationic polyelectrolytes. Prog. Polym. Sci. 2010, 35, 511–577. [Google Scholar] [CrossRef]
- Chen, K.; Zhou, X.; Wang, X. Synthesis and application of a hyperbranched polyester quaternary ammonium surfactant. J. Surfactants Deterg. 2014, 17, 1081–1088. [Google Scholar] [CrossRef]
- Asri, L.A.T.W.; Crismaru, M.; Roest, S.; Chen, Y.; Ivashenko, O.; Rudolf, P.; Tiller, J.C.; van der Mei, H.C.; Loontjens, T.J.A.; Busscher, H.J. A shape-adaptive, antibacterial-coating of immobilized quaternary-ammonium compounds tethered on hyperbranched polyurea and its mechanism of action. Adv. Funct. Mater. 2014, 24, 346–355. [Google Scholar] [CrossRef]
- Worley, B.V.; Slomberg, D.L.; Schoenfisch, M.H. Nitric oxide-releasing quaternary ammonium-modified poly(amidoamine) dendrimers as dual action antibacterial agents. Bioconjug. Chem. 2014, 25, 918–927. [Google Scholar] [PubMed]
- Chen, C.Z.; Tan, N.C.B.; Cooper, S.L. Incorporation of dimethyldodecylammonium chloride functionalities onto poly(propylene imine) dendrimers significantly enhances their antibacterial properties. Chem. Commun. 1999, 1585–1586. [Google Scholar] [CrossRef]
- Chen, C.Z.; Beck-Tan, N.C.; Dhurjati, P.; van Dyk, T.K.; LaRossa, R.A.; Cooper, S.L. Quaternary ammonium functionalized poly(propylene imine) dendrimers as effective antimicrobials: Structure-activity studies. Biomacromolecules 2000, 1, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Charles, S.; Vasanthan, N.; Kwon, D.; Sekosan, G.; Ghosh, S. Surface modification of poly(amidoamine) (PAMAM) dendrimer as antimicrobial agents. Tetrahedron Lett. 2012, 53, 6670–6675. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.Z.; Cooper, S.L. Interactions between dendrimer biocides and bacterial membranes. Biomaterials 2002, 23, 3359–3368. [Google Scholar] [CrossRef] [PubMed]
- Kenawy, E.R.; Worley, S.D.; Broughton, R. The chemistry and applications of antimicrobial polymers: A state-of-the-art review. Biomacromolecules 2007, 8, 1359–1384. [Google Scholar] [CrossRef] [PubMed]
- Williams, J.F.; Worley, S.D. Infection-resistant nonleachable materials for urologic devices. J. Endourol. 2000, 14, 395–400. [Google Scholar] [CrossRef] [PubMed]
- Hugues, C.; Bressy, C.; Bartolomeo, P.; Margaillan, A. Complexation of an acrylic resin by tertiary amines: Synthesis and characterisation of new binders for antifouling paints. Eur. Polym. J. 2003, 39, 319–326. [Google Scholar] [CrossRef]
- Ho, C.H.; Tobis, J.; Sprich, C.; Thomann, R.; Tiller, J.C. Nanoseparated polymeric networks with multiple antimicrobial properties. Adv. Mater. 2004, 16, 957–961. [Google Scholar] [CrossRef]
- Tiller, J.C.; Sprich, C.; Hartmann, L. Amphiphilic conetworks as regenerative controlled releasing antimicrobial coatings. J. Control. Release 2005, 103, 355–367. [Google Scholar] [CrossRef] [PubMed]
- Sambhy, V.; MacBride, M.M.; Peterson, B.R.; Sen, A. Silver bromide nanoparticle/polymer composites: Dual action tunable antimicrobial materials. J. Am. Chem. Soc. 2006, 128, 9798–9808. [Google Scholar] [CrossRef] [PubMed]
- Zeiger, D.N.; Stafford, C.M.; Cheng, Y.; Leigh, S.D.; Lin-Gibson, S.; Lin, N.J. Effects of sample preparation on bacterial colonization of polymers. Langmuir 2009, 26, 2659–2664. [Google Scholar] [CrossRef]
- Bazaka, K.; Jacob, M.V.; Crawford, R.J.; Ivanova, E.P. Efficient surface modification of biomaterial to prevent biofilm formation and the attachment of microorganisms. Appl. Microbiol. Biotechnol. 2012, 95, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Förch, R.; Duque, L.; Lotz, A. Antimicrobial bioactive polymer coatings. In Comprehensive Materials Processing, 1st ed.; Hashmi, S., Ed.; Elsevier Science & Tech: Oxford, UK, 2014; Volume 4, pp. 449–461. [Google Scholar]
- Grapski, J.A.; Cooper, S.L. Synthesis and characterization of non-leaching biocidal polyurethanes. Biomaterials 2001, 22, 2239–2246. [Google Scholar] [CrossRef] [PubMed]
- Gottenbos, B.; van der Mei, H.C.; Klatter, F.; Nieuwenhuis, P.; Busscher, H.J. In vitro and in vivo antimicrobial activity of covalently coupled quaternary ammonium silane coatings on silicone rubber. Biomaterials 2002, 23, 1417–1423. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Murata, H.; Koepsel, R.R.; Russell, A.J.; Matyjaszewski, K. Antibacterial polypropylene via surface-initiated atom transfer radical polymerization. Biomacromolecules 2007, 8, 1396–1399. [Google Scholar] [CrossRef] [PubMed]
- Klibanov, A.M. Permanently microbicidal materials coatings. J. Mater. Chem. 2007, 17, 2479–2482. [Google Scholar] [CrossRef]
- Yao, F.; Fu, G.-D.; Zhao, J.; Kang, E.-T.; Neoh, K.G. Antibacterial effect of surface-functionalized polypropylene hollow fiber membrane from surface-initiated atom transfer radical polymerization. J. Membr. Sci. 2008, 319, 149–157. [Google Scholar] [CrossRef]
- Madkour, A.E.; Dabkowski, J.M.; Nüsslein, K.; Tew, G.N. Fast disinfecting antimicrobial surfaces. Langmuir 2009, 25, 1060–1067. [Google Scholar] [CrossRef] [PubMed]
- Schaer, T.P.; Stewart, S.; Hsu, B.B.; Klibanov, A.M. Hydrophobic polycationic coatings that inhibit biofilms and support bone healing during infection. Biomaterials 2012, 33, 1245–1254. [Google Scholar] [CrossRef] [PubMed]
- Sang, Y.; Xiao, H. Preparation and application of cationic cellulose fibers modified by in situ grafting of cationic PVA. Colloids Surf. A Physicochem. Eng. Asp. 2009, 335, 121–127. [Google Scholar] [CrossRef]
- Zhang, D.; Xiao, H. Dual-functional beeswaxes on enhancing antimicrobial activity and water vapor barrier property of paper. ACS Appl. Mater. Interfaces 2013, 5, 3464–3468. [Google Scholar] [CrossRef] [PubMed]
- Roy, D.; Knapp, J.S.; Guthrie, J.T.; Perrier, S. Antibacterial cellulose fiber via RAFT surface graft polymerization. Biomacromolecules 2008, 9, 91–99. [Google Scholar] [CrossRef] [PubMed]
- Bazaka, K.; Jacob, M.V.; Crawford, R.J.; Ivanova, E.P. Plasma-assisted surface modification of organic biopolymers to prevent bacterial attachment. Acta Biomater. 2011, 7, 2015–2028. [Google Scholar] [CrossRef] [PubMed]
- Charnley, M.; Textor, M.; Acikgoz, C. Designed polymer structures with antifouling-antimicrobial properties. React. Funct. Polym. 2011, 71, 329–334. [Google Scholar] [CrossRef]
- Wischerhoff, E.; Badi, N.; Laschewsky, A.; Lutz, J.-F. Smart polymer surfaces: Concepts and applications in biosciences. In Bioactive Surfaces; Börner, H.G., Lutz, J.-F., Eds.; Springer: Berlin/Heidelberg, Germany, 2011; Volume 240, pp. 1–33. [Google Scholar]
- Lin, J.; Qiu, S.; Lewis, K.; Klibanov, A.M. Mechanism of bactericidal and fungicidal activities of textiles covalently modified with alkylated polyethylenimine. Biotechnol. Bioeng. 2003, 83, 168–172. [Google Scholar] [CrossRef] [PubMed]
- Waschinski, C.J.; Zimmermann, J.; Salz, U.; Hutzier, R.; Sadowski, G.; Tiller, J.C. Design of contact-active antimicrobial acrylate-based materials using biocidal macromers. Adv. Mater. 2008, 20, 104–108. [Google Scholar] [CrossRef]
- Cen, L.; Neoh, K.G.; Kang, E.T. Surface functionalization technique for conferring antibacterial properties to polymeric and cellulosic surfaces. Langmuir 2003, 19, 10295–10303. [Google Scholar] [CrossRef]
- Jampala, S.N.; Sarmadi, M.; Somers, E.B.; Wong, A.C.L.; Denes, F.S. Plasma-enhanced synthesis of bactericidal quaternary ammonium thin layers on stainless steel and cellulose surfaces. Langmuir 2008, 24, 8583–8591. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Wu, J.; Avery, C.W.; Mizutani, M.; Jiang, X.; Kamigaito, M.; Chen, Z.; Xi, C.; Kuroda, K. Immobilization of amphiphilic polycations by catechol functionality for antimicrobial coatings. Langmuir 2011, 27, 4010–4019. [Google Scholar] [CrossRef] [PubMed]
- Sever, M.J.; Weisser, J.T.; Monahan, J.; Srinivasan, S.; Wilker, J.J. Metal-mediated cross-linking in the generation of a marine-mussel adhesive. Angew. Chem. Int. Ed. 2004, 43, 448–450. [Google Scholar] [CrossRef]
- Zürcher, S.; Wäckerlin, D.; Bethuel, Y.; Malisova, B.; Textor, M.; Tosatti, S.; Gademann, K. Biomimetic surface modifications based on the cyanobacterial iron chelator anachelin. J. Am. Chem. Soc. 2006, 128, 1064–1065. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-inspired surface chemistry for multifunctional coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Shi, Z.; Neoh, K.G.; Kang, E.T. Surface-grafted viologen for precipitation of silver nanoparticles and their combined bactericidal activities. Langmuir 2004, 20, 6847–6852. [Google Scholar] [CrossRef] [PubMed]
- Podsiadlo, P.; Paternel, S.; Rouillard, J.-M.; Zhang, Z.; Lee, J.; Lee, J.-W.; Gulari, E.; Kotov, N.A. Layer-by-layer assembly of nacre-like nanostructured composites with antimicrobial properties. Langmuir 2005, 21, 11915–11921. [Google Scholar] [CrossRef] [PubMed]
- Grunlan, J.C.; Choi, J.K.; Lin, A. Antimicrobial behavior of polyelectrolyte multilayer films containing cetrimide and silver. Biomacromolecules 2005, 6, 1149–1153. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Lee, D.; Sheng, X.; Cohen, R.E.; Rubner, M.F. Two-level antibacterial coating with both release-killing and contact-killing capabilities. Langmuir 2006, 22, 9820–9823. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.; Qiu, S.; Lewis, K.; Klibanov, A.M. Bactericidal properties of flat surfaces and nanoparticles derivatized with alkylated polyethylenimines. Biotechnol. Prog. 2002, 18, 1082–1086. [Google Scholar] [CrossRef] [PubMed]
- Imazato, S.; Kinomoto, Y.; Tarumi, H.; Ebisu, S.; Tay, F.R. Antibacterial activity and bonding characteristics of an adhesive resin containing antibacterial monomer MDPB. Dent. Mater. 2003, 19, 313–319. [Google Scholar] [CrossRef] [PubMed]
- Cao, Z.; Mi, L.; Mendiola, J.; Ella-Menye, J.-R.; Zhang, L.; Xue, H.; Jiang, S. Reversibly switching the function of a surface between attacking and defending against bacteria. Angew. Chem. Int. Ed. 2012, 51, 2602–2605. [Google Scholar] [CrossRef]
- Nigmatullin, R.; Gao, F. Onium-functionalised polymers in the design of non-leaching antimicrobial surfaces. Macromol. Mater. Eng. 2012, 297, 1038–1074. [Google Scholar] [CrossRef]
- Lewis, K.; Klibanov, A.M. Surpassing nature: Rational design of sterile-surface materials. Trends Biotechnol. 2005, 23, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Milović, N.M.; Wang, J.; Lewis, K.; Klibanov, A.M. Immobilized N-alkylated polyethylenimine avidly kills bacteria by rupturing cell membranes with no resistance developed. Biotechnol. Bioeng. 2005, 90, 715–722. [Google Scholar] [CrossRef] [PubMed]
- Murata, H.; Koepsel, R.R.; Matyjaszewski, K.; Russell, A.J. Permanent, non-leaching antibacterial surfaces-2: How high density cationic surfaces kill bacterial cells. Biomaterials 2007, 28, 4870–4879. [Google Scholar] [CrossRef] [PubMed]
- Siedenbiedel, F.; Tiller, J.C. Antimicrobial polymers in solution and on surfaces: Overview and functional principles. Polymers 2012, 4, 46–71. [Google Scholar] [CrossRef]
- Larson, A.M.; Klibanov, A.M. Biocidal packaging for pharmaceuticals, foods, and other perishables. Annu. Rev. Chem. Biomol. Eng. 2013, 4, 171–186. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.B.; Koepsel, R.R.; Morley, S.W.; Matyjaszewski, K.; Sun, Y.; Russell, A.J. Permanent, nonleaching antibacterial surfaces, 1. Synthesis by atom transfer radical polymerization. Biomacromolecules 2004, 5, 877–882. [Google Scholar] [CrossRef] [PubMed]
- Kügler, R.; Bouloussa, O.; Rondelez, F. Evidence of a charge-density threshold for optimum efficiency of biocidal cationic surfaces. Microbiology 2005, 151, 1341–1348. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Koepsel, R.R.; Murata, H.; Wu, W.; Lee, S.B.; Kowalewski, T.; Russell, A.J.; Matyjaszewski, K. Nonleaching antibacterial glass surfaces via “grafting onto”: The effect of the number of quaternary ammonium groups on biocidal activity. Langmuir 2008, 24, 6785–6795. [Google Scholar] [CrossRef] [PubMed]
- Vaara, M. Agents that increase the permeability of the outer membrane. Microbiol. Rev. 1992, 56, 395–411. [Google Scholar] [PubMed]
- Yatvin, J.; Gao, J.; Locklin, J. Durable defense: Robust and varied attachment of non-leaching poly“-onium” bactericidal coatings to reactive and inert surfaces. Chem. Commun. 2014, 50, 9433–9442. [Google Scholar] [CrossRef]
- Sharma, S.K.; Chauhan, G.S.; Gupta, R.; Ahn, J.H. Tuning anti-microbial activity of poly(4-vinyl 2-hydroxyethyl pyridinium) chloride by anion exchange reactions. J. Mater. Sci. Mater. Med. 2010, 21, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Garg, G.; Chauhan, G.S.; Gupta, R.; Ahn, J.H. Anion effects on anti-microbial activity of poly[1-vinyl-3-(2-sulfoethyl imidazolium betaine)]. J. Colloid Interface Sci. 2010, 344, 90–96. [Google Scholar] [CrossRef] [PubMed]
- Kanazawa, A.; Ikeda, T.; Endo, T. Polymeric phosphonium salts as a novel class of cationic biocides. II. Effects of counter anion and molecular weight on antibacterial activity of polymeric phosphonium salts. J. Polym. Sci. Part A Polym. Chem. 1993, 31, 1441–1447. [Google Scholar] [CrossRef]
- Panarin, E.F.; Solovskii, M.V.; Zaikina, N.A.; Afinogenov, G.E. Biological activity of cationic polyelectrolytes. Makromol. Chem. 1985, 9, 25–33. [Google Scholar] [CrossRef]
- Mizerska, U.; Fortuniak, W.; Chojnowski, J.; Hałasa, R.; Konopacka, A.; Werel, W. Polysiloxane cationic biocides with imidazolium salt (ImS) groups, synthesis and antibacterial properties. Eur. Polym. J. 2009, 45, 779–787. [Google Scholar] [CrossRef]
- Xie, D.; Weng, Y.; Guo, X.; Zhao, J.; Gregory, R.L.; Zheng, C. Preparation and evaluation of a novel glass-ionomer cement with antibacterial functions. Dent. Mater. 2011, 27, 487–496. [Google Scholar] [CrossRef] [PubMed]
- Ingalsbe, M.L.; Denis, J.D.S.; McGahan, M.E.; Steiner, W.W.; Priefer, R. Development of a novel expression, ZIMAX/KZI, for determination of the counter-anion effect on the antimicrobial activity of tetrabutylammonium salts. Bioorg. Med. Chem. Lett. 2009, 19, 4984–4987. [Google Scholar] [CrossRef] [PubMed]
- Waschinski, C.J.; Barnert, S.; Theobald, A.; Schubert, R.; Kleinschmidt, F.; Hoffmann, A.; Saalwächter, K.; Tiller, J.C. Insights in the antibacterial action of poly(methyloxazoline)s with a biocidal end group and varying satellite groups. Biomacromolecules 2008, 9, 1764–1771. [Google Scholar] [CrossRef] [PubMed]
- Colak, S.; Nelson, C.F.; Nusslein, K.; Tew, G.N. Hydrophilic modifications of an amphiphilic polynorbornene and the effects on its hemolytic and antibacterial activity. Biomacromolecules 2009, 10, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Palermo, E.F.; Kuroda, K. Structural determinants of antimicrobial activity in polymers which mimic host defense peptides. Appl. Microbiol. Biotechnol. 2010, 87, 1605–1615. [Google Scholar] [CrossRef] [PubMed]
- Ohta, S.; Misawa, Y.; Miyamoto, H.; Makino, M.; Nagai, K.; Shiraishi, T.; Nakagawa, Y.; Yamato, S.; Tachikawa, E.; Zenda, H. A comparative study of characteristics of current-type and conventional-type cationic bactericides. Biol. Pharm. Bull. 2001, 24, 1093–1096. [Google Scholar] [CrossRef] [PubMed]
- Sandt, C.; Barbeau, J.; Gagnon, M.A.; Lafleur, M. Role of the ammonium group in the diffusion of quaternary ammonium compounds in Streptococcus mutans biofilms. J. Antimicrob. Chemother. 2007, 60, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Bridier, A.; Briandet, R.; Thomas, V.; Dubois-Brissonnet, F. Resistance of bacterial biofilms to disinfectants: A review. Biofouling 2011, 27, 1017–1032. [Google Scholar] [CrossRef] [PubMed]
- Sambhy, V.; Peterson, B.R.; Sen, A. Antibacterial and hemolytic activities of pyridinium polymers as a function of the spatial relationship between the positive charge and the pendant alkyl tail. Angew. Chem. Int. Ed. 2008, 47, 1250–1254. [Google Scholar] [CrossRef]
- Kanazawa, A.; Ikeda, T. Multifunctional tetracoordinate phosphorus species with high self-organizing ability. Coord. Chem. Rev. 2000, 198, 117–131. [Google Scholar] [CrossRef]
- Palermo, E.F.; Kuroda, K. Chemical structure of cationic groups in amphiphilic polymethacrylates modulates the antimicrobial and hemolytic activities. Biomacromolecules 2009, 10, 1416–1428. [Google Scholar] [CrossRef] [PubMed]
- Oda, Y.; Kanaoka, S.; Sato, T.; Aoshima, S.; Kuroda, K. Block versus random amphiphilic copolymers as antibacterial agents. Biomacromolecules 2011, 12, 3581–3591. [Google Scholar] [CrossRef] [PubMed]
- Hasan, J.; Crawford, R.J.; Ivanova, E.P. Antibacterial surfaces: The quest for a new generation of biomaterials. Trends Biotechnol. 2013, 31, 295–304. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, K.; DeGrado, W.F. Amphiphilic polymethacrylate derivatives as antimicrobial agents. J. Am. Chem. Soc. 2005, 127, 4128–4129. [Google Scholar] [CrossRef] [PubMed]
- Stratton, T.R.; Rickus, J.L.; Youngblood, J.P. In vitro biocompatibility studies of antibacterial quaternary polymers. Biomacromolecules 2009, 10, 2550–2555. [Google Scholar] [CrossRef] [PubMed]
- King, A.; Chakrabarty, S.; Zhang, W.; Zeng, X.; Ohman, D.E.; Wood, L.F.; Abraham, S.; Rao, R.; Wynne, K.J. High antimicrobial effectiveness with low hemolytic and cytotoxic activity for PEG/quaternary copolyoxetanes. Biomacromolecules 2014, 15, 456–467. [Google Scholar] [CrossRef] [PubMed]
- Dizman, B.; Elasri, M.O.; Mathias, L.J. Synthesis and characterization of antibacterial and temperature responsive methacrylamide polymers. Macromolecules 2006, 39, 5738–5746. [Google Scholar] [CrossRef]
- Kurt, P.; Wood, L.; Ohman, D.E.; Wynne, K.J. Highly effective contact antimicrobial surfaces via polymer surface modifiers. Langmuir 2007, 23, 4719–4723. [Google Scholar] [CrossRef] [PubMed]
- Venkataraman, S.; Zhang, Y.; Liu, L.; Yang, Y.-Y. Design, syntheses and evaluation of hemocompatible pegylated-antimicrobial polymers with well-controlled molecular structures. Biomaterials 2010, 31, 1751–1756. [Google Scholar] [CrossRef] [PubMed]
- Song, A.; Walker, S.G.; Parker, K.A.; Sampson, N.S. Antibacterial studies of cationic polymers with alternating, random, and uniform backbones. ACS Chem. Biol. 2011, 6, 590–599. [Google Scholar] [CrossRef] [PubMed]
- Engler, A.C.; Tan, J.P.K.; Ong, Z.Y.; Coady, D.J.; Ng, V.W.L.; Yang, Y.Y.; Hedrick, J.L. Antimicrobial polycarbonates: Investigating the impact of balancing charge and hydrophobicity using a same-centered polymer approach. Biomacromolecules 2013, 14, 4331–4339. [Google Scholar] [CrossRef] [PubMed]
- Muñoz-Bonilla, A.; Fernández-García, M. Polymeric materials with antimicrobial activity. Prog. Polym. Sci. 2012, 37, 281–339. [Google Scholar] [CrossRef]
- Komen, C.M.D. Phosphonium salts, ylides and phosphoranes. In The Chemistry of Organophosphorus Compounds, 1st ed.; Hartley, F.R., Ed.; John Wiley & Sons, Ltd.: Chichester, UK, 1994; Volume 3, pp. 382–382. [Google Scholar]
- Popa, A.; Davidescu, C.M.; Trif, R.; Ilia, G.; Iliescu, S.; Dehelean, G. Study of quaternary “onium” salts grafted on polymers: Antibacterial activity of quaternary phosphonium salts grafted on “gel-type” styrene-divinylbenzene copolymers. React. Funct. Polym. 2003, 55, 151–158. [Google Scholar] [CrossRef]
- Cai, X.; Tan, S.; Lin, M.; Xie, A.; Mai, W.; Zhang, X.; Lin, Z.; Wu, T.; Liu, Y. Synergistic antibacterial brilliant blue/reduced graphene oxide/quaternary phosphonium salt composite with excellent water solubility and specific targeting capability. Langmuir 2011, 27, 7828–7835. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Pan, Y.; Xiao, H.; Zhao, Y. Novel quaternary phosphonium-type cationic polyacrylamide and elucidation of dual-functional antibacterial/antiviral activity. RSC Adv. 2014, 4, 46887–46895. [Google Scholar] [CrossRef]
- Jangu, C.; Long, T.E. Phosphonium cation-containing polymers: From ionic liquids to polyelectrolytes. Polymer 2014, 55, 3298–3304. [Google Scholar] [CrossRef]
- Kenawy, E.R.; Abdel-Hay, F.I.; El-Shanshoury, A.E.R.R.; El-Newehy, M.H. Biologically active polymers: Synthesis and antimicrobial activity of modified glycidyl methacrylate polymers having a quaternary ammonium and phosphonium groups. J. Control. Release 1998, 50, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Kenawy, E.-R.; Abdel-Hay, F.I.; El-Shanshoury, A.E.-R.R.; El-Newehy, M.H. Biologically active polymers. V. Synthesis and antimicrobial activity of modified poly(glycidyl methacrylate-co-2-hydroxyethyl methacrylate) derivatives with quaternary ammonium and phosphonium salts. J. Polym. Sci. Part A Polym. Chem. 2002, 40, 2384–2393. [Google Scholar] [CrossRef]
- Kanazawa, A.; Ikeda, T.; Endo, T. Polymeric phosphonium salts as a novel class of cationic biocides. VIII. Synergistic effect on antibacterial activity of polymeric phosphonium and ammonium salts. J. Appl. Polym. Sci. 1994, 53, 1245–1249. [Google Scholar] [CrossRef]
- El-Newehy, M.H.; Kenawy, E.R.; Al-Deyab, S.S. Biocidal polymers: Preparation and antimicrobial assessment of immobilized onium salts onto modified chitosan. Int. J. Polym. Mater. 2014, 63, 758–766. [Google Scholar] [CrossRef]
- Gao, B.; Liu, Q.; Li, Y. Preparation of water-insoluble antibacterial materials with surface-grafted material PSt/SiO2 and their antibacterial activity. J. Polym. Environ. 2010, 18, 474–483. [Google Scholar] [CrossRef]
- García-Argüelles, S.; Serrano, M.C.; Gutiérrez, M.C.; Ferrer, M.L.; Yuste, L.; Rojo, F.; del Monte, F. Deep Eutectic Solvent-Assisted Synthesis of Biodegradable Polyesters with Antibacterial Properties. Langmuir 2013, 29, 9525–9534. [Google Scholar] [CrossRef] [PubMed]
- Qiu, T.; Zeng, Q.; Ao, N. Preparation and characterization of chlorinated nature rubber (CNR) based polymeric quaternary phosphonium salt bactericide. Mater. Lett. 2014, 122, 13–16. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xue, Y.; Xiao, H.; Zhang, Y. Antimicrobial Polymeric Materials with Quaternary Ammonium and Phosphonium Salts. Int. J. Mol. Sci. 2015, 16, 3626-3655. https://doi.org/10.3390/ijms16023626
Xue Y, Xiao H, Zhang Y. Antimicrobial Polymeric Materials with Quaternary Ammonium and Phosphonium Salts. International Journal of Molecular Sciences. 2015; 16(2):3626-3655. https://doi.org/10.3390/ijms16023626
Chicago/Turabian StyleXue, Yan, Huining Xiao, and Yi Zhang. 2015. "Antimicrobial Polymeric Materials with Quaternary Ammonium and Phosphonium Salts" International Journal of Molecular Sciences 16, no. 2: 3626-3655. https://doi.org/10.3390/ijms16023626
APA StyleXue, Y., Xiao, H., & Zhang, Y. (2015). Antimicrobial Polymeric Materials with Quaternary Ammonium and Phosphonium Salts. International Journal of Molecular Sciences, 16(2), 3626-3655. https://doi.org/10.3390/ijms16023626