Coronary CT Angiography in Managing Atherosclerosis
Abstract
:1. Background
2. Coronary Plaque Morphology and Pathophysiology
3. Coronary Calcification
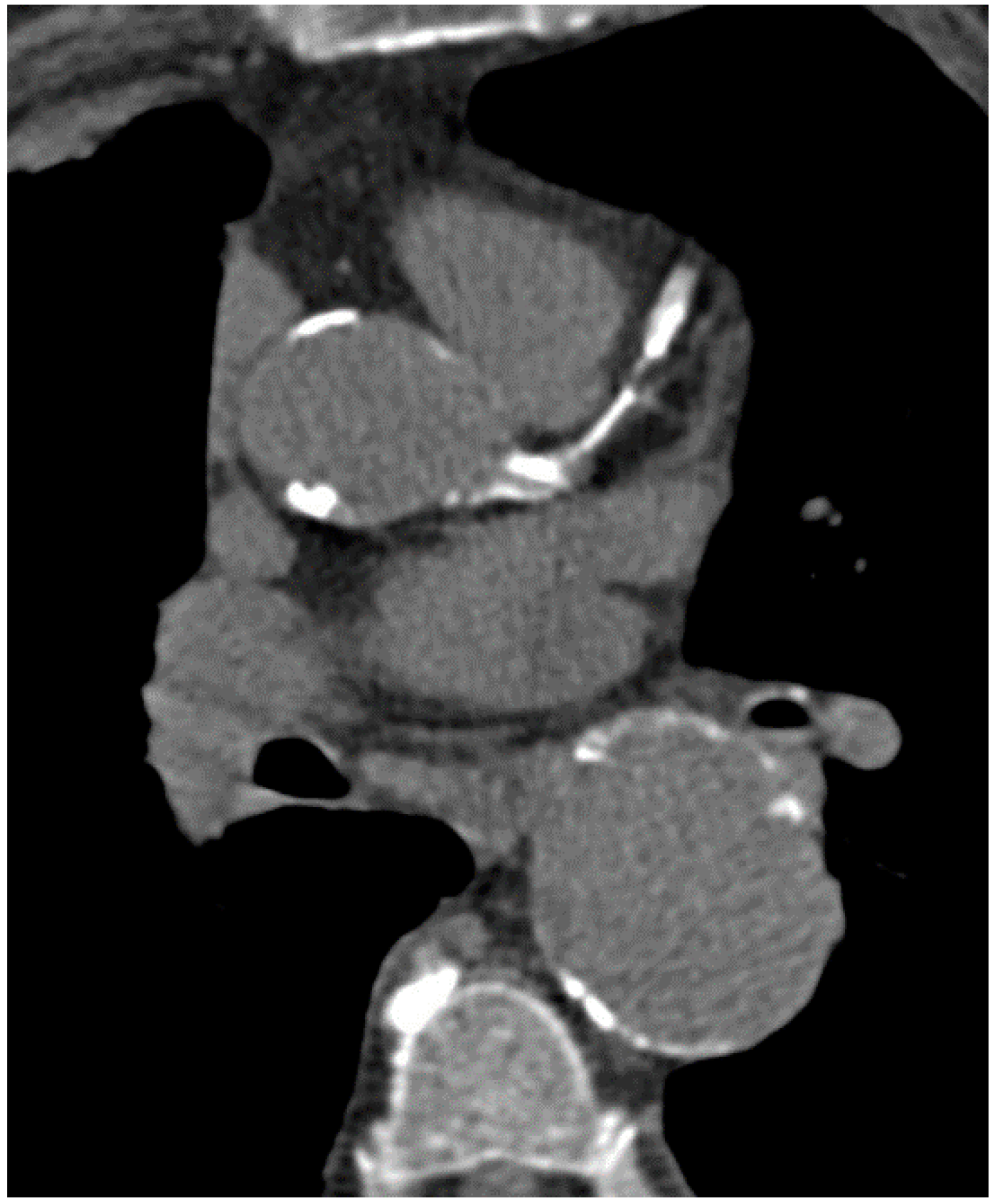
4. Clinical Implication and Prognosis of Coronary Artery Calcium
5. Coronary CT Angiography
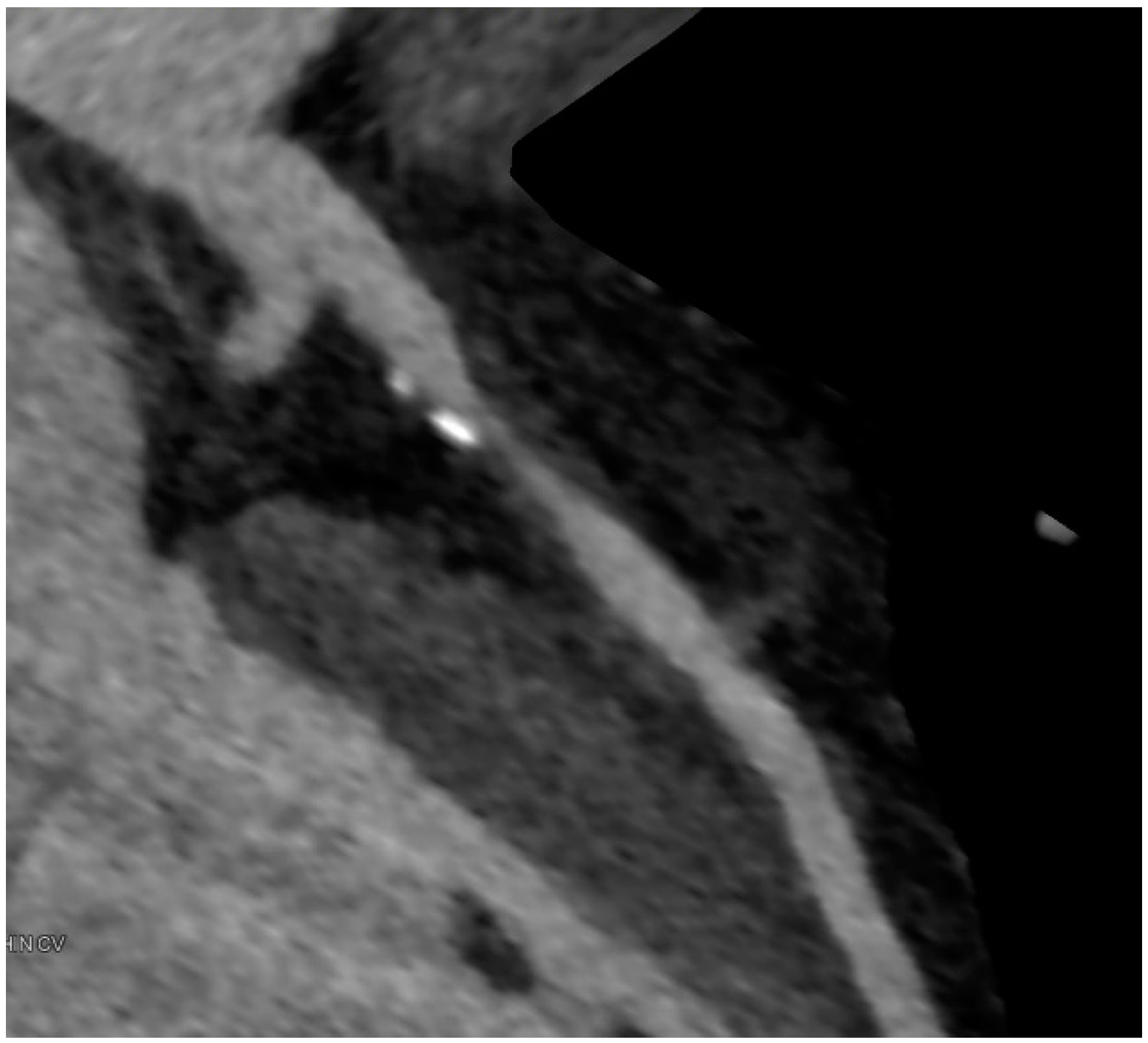
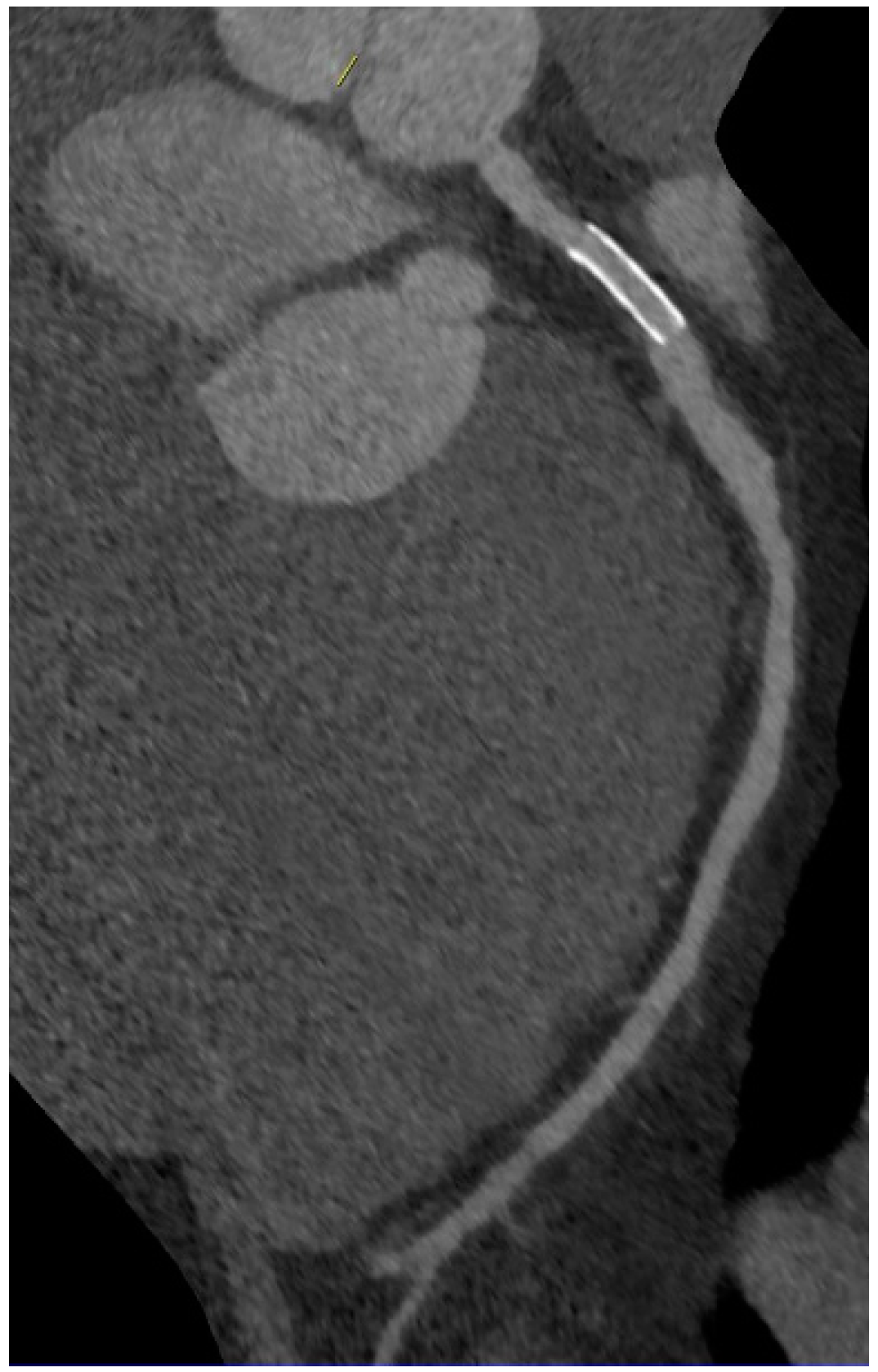
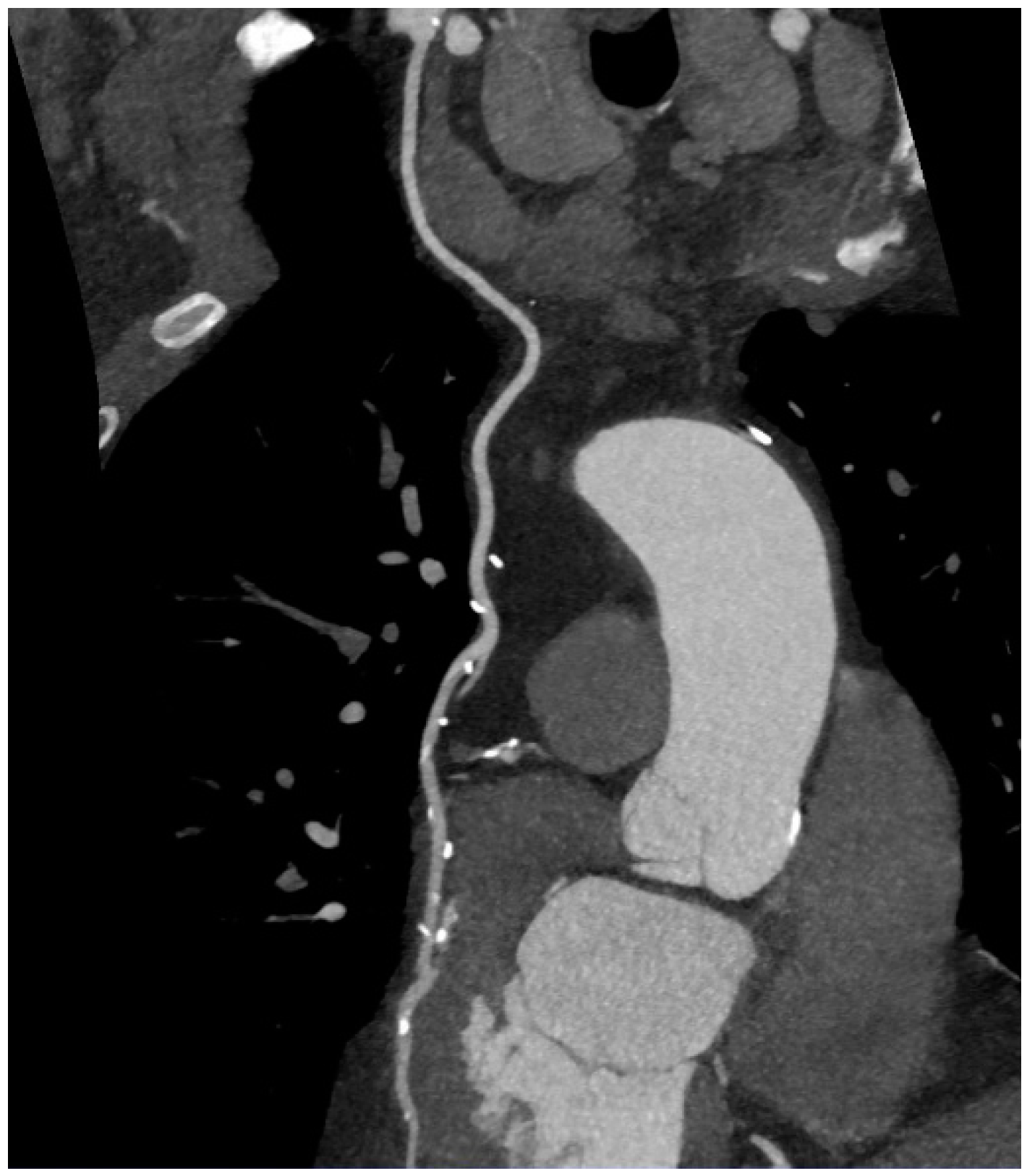
6. Imaging of Coronary Plaques and Stenoses
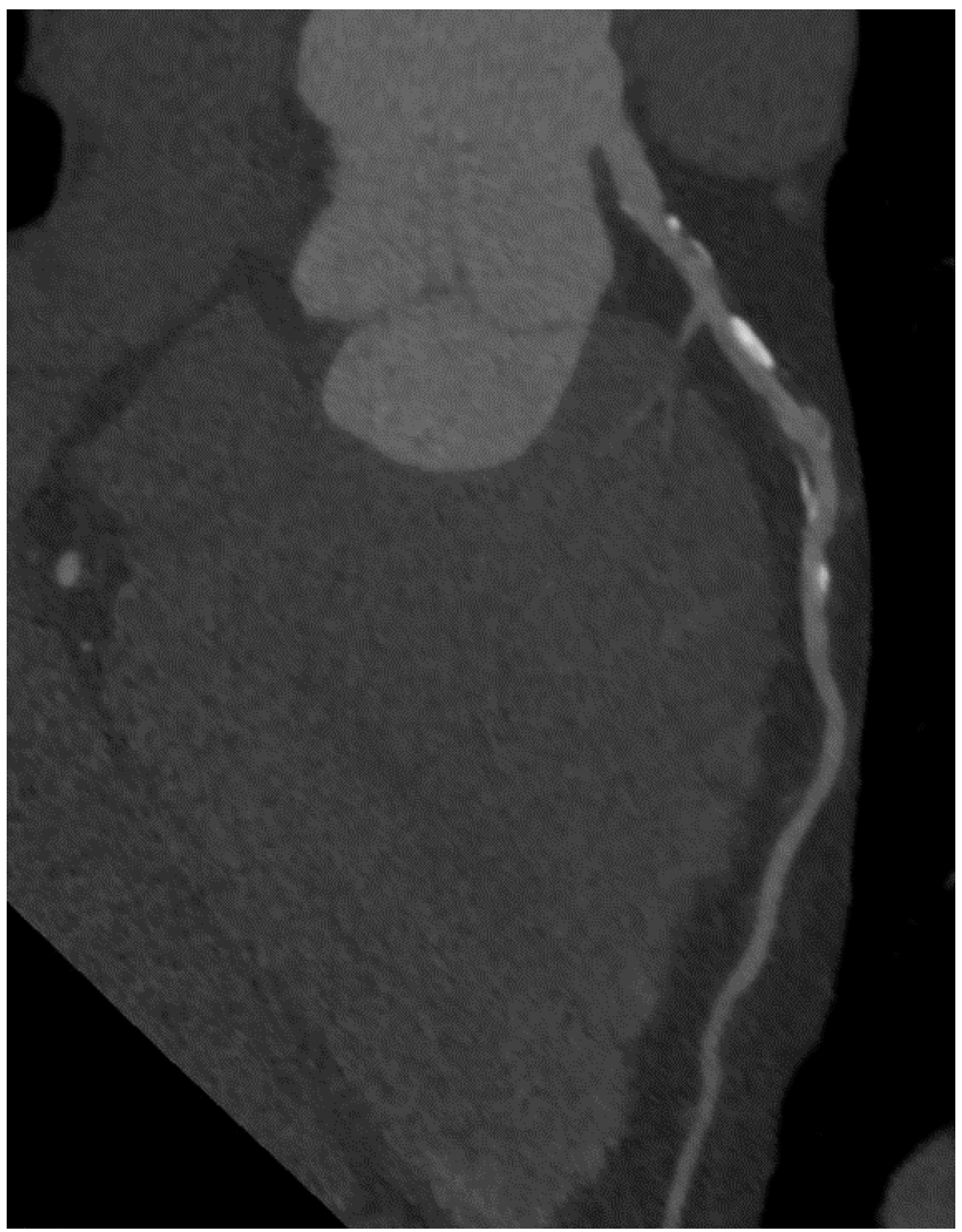
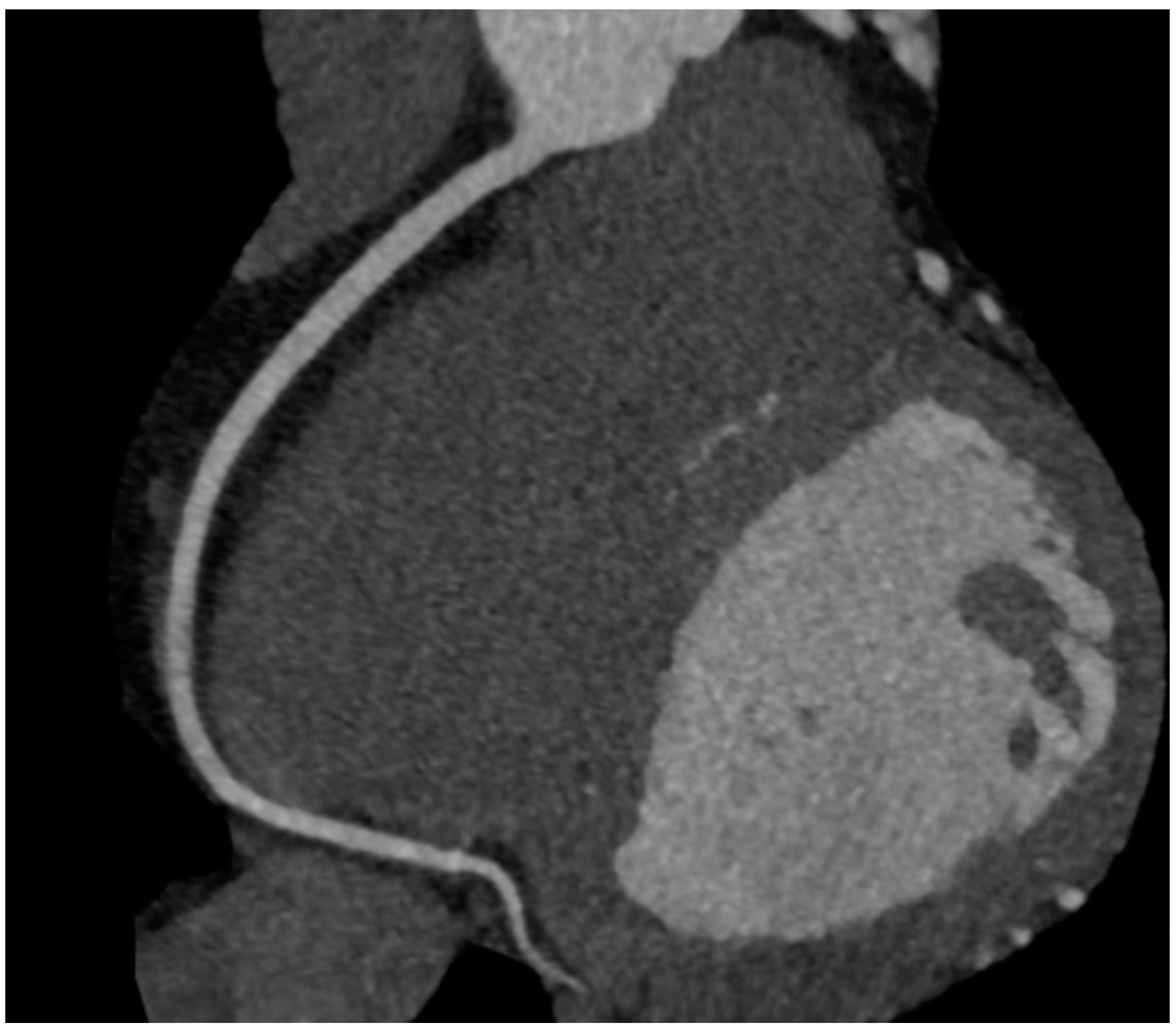
7. Qualitative Plaque Characterization
8. Hemodynamic Relevance of Angiographic Stenoses
9. CCTA in the Emergency Department
10. Prognostic Data of CCTA
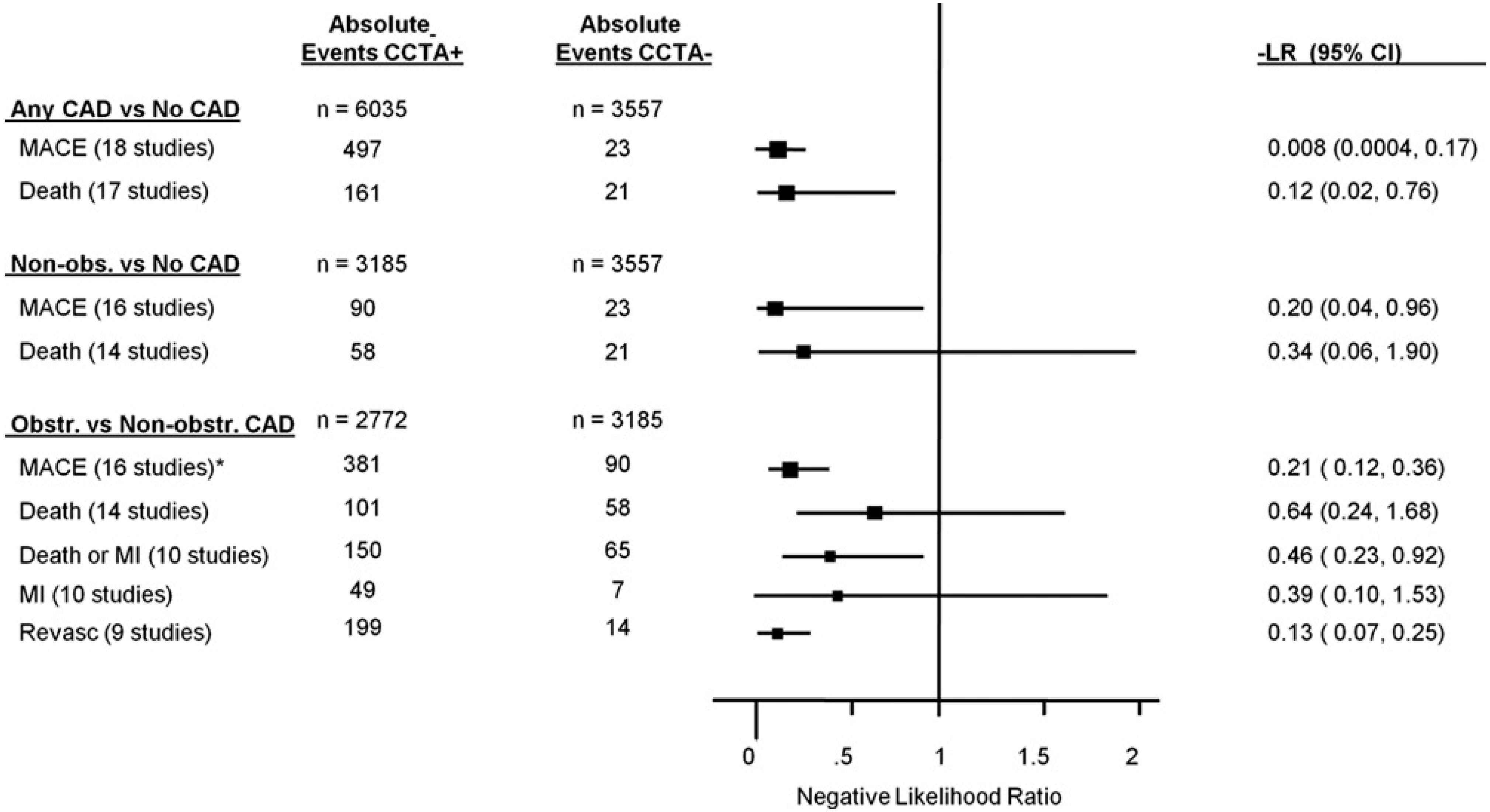
11. Summary and Future Directions
Conflicts of Interest
References
- Leipsic, J.; Abbara, S.; Achenbach, S.; Cury, R.; Earls, J.P.; Mancini, G.J.; Nieman, K.; Pontone, G.; Raff, G.L. SCCT guidelines for the interpretation and reporting of coronary CT angiography: A report of the Society of Cardiovascular Computed Tomography Guidelines Committee. J. Cardiovasc. Comput. Tomogr. 2014, 8, 342–358. [Google Scholar] [CrossRef] [PubMed]
- Tabas, I.; Williams, K.J.; Borén, J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: Update and therapeutic implications. Circulation 2007, 116, 1832–1844. [Google Scholar] [CrossRef] [PubMed]
- Stary, H.C.; Chandler, A.B.; Glagov, S.; Guyton, J.R.; Insull, W.; Rosenfeld, M.E.; Schaffer, S.A.; Schwartz, C.J.; Wagner, W.D.; Wissler, R.W. A definition of initial, fatty streak, and intermediate lesions of atherosclerosis: A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Arterioscler. Thromb. Vasc. Biol. 1994, 14, 840–856. [Google Scholar] [CrossRef]
- Stary, H.C.; Chandler, A.B.; Dinsmore, R.E.; Fuster, V.; Glagov, S.; Insull, W.; Rosenfeld, M.E.; Schwartz, C.J.; Wagner, W.D.; Wissler, R.W. A definition of advanced types of atherosclerotic lesions and a histological classification of atherosclerosis: A report from the Committee on Vascular Lesions of the Council on Arteriosclerosis, American Heart Association. Circulation 1995, 92, 1355–1374. [Google Scholar] [CrossRef] [PubMed]
- Glagov, S.; Weisenberg, E.; Zarins, C.K.; Stankunavicius, R.; Kolettis, G.J. Compensatory enlargement of human atherosclerotic coronary arteries. N. Engl. J. Med. 1987, 316, 1371–1375. [Google Scholar] [CrossRef] [PubMed]
- Virmani, R.; Burke, A.P.; Farb, A.; Kolodgie, F.D. Pathology of the vulnerable plaque. J. Am. Coll. Cardiol. 2006, 47, C13–C18. [Google Scholar] [CrossRef] [PubMed]
- Agatston, A.S.; Janowitz, W.R.; Hildner, F.J.; Zusmer, N.R.; Viamonte, M., Jr.; Detrano, R. Quantification of coronary artery calcium using ultrafast computed tomography. J. Am. Coll. Cardiol. 1990, 15, 827–832. [Google Scholar] [CrossRef]
- O’Rourke, R.A.; Brundage, B.H.; Froelicher, V.F.; Greenland, P.; Grundy, S.M.; Hachamovitch, R.; Pohost, G.M.; Shaw, L.J.; Weintraub, W.S.; Winters, W.L.; et al. American college of cardiology/American heart association expert consensus document on electron-beam computed tomography for the diagnosis and prognosis of coronary artery disease committee members. J. Am. Coll. Cardiol. 2000, 36, 326–340. [Google Scholar] [CrossRef] [PubMed]
- Pohle, K.; Ropers, D.; Mäffert, R.; Geitner, P.; Moshage, W.; Regenfus, M.; Kusus, M.; Daniel, W.G.; Achenbach, S. Coronary calcifications in young patients with first, unheralded myocardial infarction: A risk factor matched analysis by electron beam tomography. Heart 2003, 89, 625–628. [Google Scholar] [CrossRef] [PubMed]
- Schmermund, A.; Schwartz, R.S.; Adamzik, M.; Sangiorgi, G.; Pfeifer, E.A.; Rumberger, J.A.; Burke, A.P.; Farb, A.; Virmani, R. Coronary atherosclerosis in unheralded sudden coronary death under age 50: Histo-pathologic comparison with “healthy” subjects dying out of hospital. Atherosclerosis 2001, 155, 499–508. [Google Scholar] [CrossRef] [PubMed]
- Davies, M.J. The composition of coronary-artery plaques. N. Engl. J. Med. 1997, 336, 1312–1314. [Google Scholar] [CrossRef] [PubMed]
- Marwan, M.; Ropers, D.; Pflederer, T.; Daniel, W.G.; Achenbach, S. Clinical characteristics of patients with obstructive coronary lesions in the absence of coronary calcification: An evaluation by coronary CT angiography. Heart 2009, 95, 1056–1060. [Google Scholar] [CrossRef]
- Gottlieb, I.; Miller, J.M.; Arbab-Zadeh, A.; Dewey, M.; Clouse, M.E.; Sara, L.; Niinuma, H.; Bush, D.E.; Paul, N.; Vavere, A.L.; et al. The absence of coronary calcification does not exclude obstructive coronary artery disease or the need for revascularization in patients referred for conventional coronary angiography. J. Am. Coll. Cardiol. 2010, 55, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Boström, K.; Watson, K.E.; Horn, S.; Wortham, C.; Herman, I.M.; Demer, L.L. Bone morphogenetic protein expression in human atherosclerotic lesions. J. Clin. Investig. 1993, 91, 1800–1809. [Google Scholar] [CrossRef] [PubMed]
- Jeziorska, M.; McCollum, C.; Wooley, D.E. Observations on bone formation and remodelling in advanced atherosclerotic lesions of human carotid arteries. Virchows Arch. 1998, 433, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Stary, H.C. The sequence of cell and matrix changes in atherosclerotic lesions of coronary arteries in the first forty years of life. Eur. Heart J. 1990, 11 (Suppl. E), 3–19. [Google Scholar] [CrossRef]
- Greenland, P.; Bonow, R.O.; Brundage, B.H.; Budoff, M.J.; Eisenberg, M.J.; Grundy, S.M.; Lauer, M.S.; Post, W.S.; Raggi, P.; Redberg, R.F.; et al. ACCF/AHA 2007 clinical expert consensus document on coronary artery calcium scoring by computed tomography in global cardiovascular risk assessment and in evaluation of patients with chest pain. J. Am. Coll. Cardiol. 2007, 49, 378–402. [Google Scholar] [CrossRef] [PubMed]
- McClelland, R.L.; Chung, H.; Detrano, R.; Post, W.; Kronmal, R.A. Distribution of coronary artery calcium by race, gender, and age: Results from the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation 2006, 113, 30–37. [Google Scholar] [CrossRef] [PubMed]
- Raggi, P.; Cooil, B.; Ratti, C.; Callister, T.Q.; Budoff, M. Progression of coronary artery calcium and occurrence of myocardial infarction in patients with and without diabetes mellitus. Hypertension 2005, 46, 238–243. [Google Scholar] [CrossRef] [PubMed]
- Goh, V.K.; Lau, C.P.; Mohlenkamp, S.; Rumberger, J.A.; Achenbach, S.; Budoff, M.J. Outcome of coronary plaque burden: A 10-year follow-up of aggressive medical management. Cardiovasc. Ultrasound 2010, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Cassidy-Bushrow, A.E.; Bielak, L.F.; Sheedy, P.F.; Turner, S.T.; Kullo, I.J.; Lin, X.; Peyser, P.A. Coronary artery calcification progression is heritable. Circulation 2007, 116, 25–31. [Google Scholar] [CrossRef] [PubMed]
- Erbel, R.; Lehmann, N.; Churzidse, S.; Rauwolf, M.; Mahabadi, A.A.; Möhlenkamp, S.; Moebus, S.; Bauer, M.; Kälsch, H.; Budde, T.; et al. Heinz Nixdorf Recall study investigators progression of coronary artery calcification seems to be inevitable, but predictable—Results of the Heinz Nixdorf Recall (HNR) study. Eur. Heart J. 2014, 35, 2960–2971. [Google Scholar] [CrossRef] [PubMed]
- Sarwar, A.; Shaw, L.J.; Shapiro, M.D.; Blankstein, R.; Hoffmann, U.; Hoffman, U.; Cury, R.C.; Abbara, S.; Brady, T.J.; Budoff, M.J.; et al. Diagnostic and prognostic value of absence of coronary artery calcification. JACC Cardiovasc. Imaging 2009, 2, 675–688. [Google Scholar] [CrossRef] [PubMed]
- Greenland, P.; LaBree, L.; Azen, S.P.; Doherty, T.M.; Detrano, R.C. Coronary artery calcium score combined with Framingham score for risk prediction in asymptomatic individuals. JAMA 2004, 291, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Hokanson, J.E.; Nasir, K.; Shaw, L.J.; Kinney, G.L.; Chow, D.; Demoss, D.; Nuguri, V.; Nabavi, V.; Ratakonda, R.; et al. Progression of coronary artery calcium predicts all-cause mortality. JACC Cardiovasc. Imaging 2010, 3, 1229–1236. [Google Scholar] [CrossRef] [PubMed]
- Raggi, P.; Callister, T.Q.; Shaw, L.J. Progression of coronary artery calcium and risk of first myocardial infarction in patients receiving cholesterol-lowering therapy. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 1272–1277. [Google Scholar] [CrossRef] [PubMed]
- Meijboom, W.B.; van Mieghem, C.A.G.; Mollet, N.R.; Pugliese, F.; Weustink, A.C.; van Pelt, N.; Cademartiri, F.; Nieman, K.; Boersma, E.; de Jaegere, P.; et al. 64-Slice computed tomography coronary angiography in patients with high, intermediate, or low pretest probability of significant coronary artery disease. J. Am. Coll. Cardiol. 2007, 50, 1469–1475. [Google Scholar] [CrossRef] [PubMed]
- Achenbach, S.; Goroll, T.; Seltmann, M.; Pflederer, T.; Anders, K.; Ropers, D.; Daniel, W.G.; Uder, M.; Lell, M.; Marwan, M. Detection of coronary artery stenoses by low-dose, prospectively ECG-triggered, high-pitch spiral coronary CT angiography. JACC Cardiovasc. Imaging 2011, 4, 328–337. [Google Scholar] [CrossRef] [PubMed]
- Kröpil, P.; Rojas, C.A.; Ghoshhajra, B.; Lanzman, R.S.; Miese, F.R.; Scherer, A.; Kalra, M.; Abbara, S. Prospectively ECG-triggered high-pitch spiral acquisition for cardiac CT angiography in routine clinical practice: Initial results. J. Thorac. Imaging 2012, 27, 194–201. [Google Scholar] [CrossRef] [PubMed]
- Yin, W.H.; Lu, B.; Hou, Z.H.; Li, N.; Han, L.; Wu, Y.J.; Niu, H.X.; Silverman, J.R.; Nicola De Cecco, C.; Schoepf, U.J. Detection of coronary artery stenosis with sub-milliSievert radiation dose by prospectively ECG-triggered high-pitch spiral CT angiography and iterative reconstruction. Eur. Radiol. 2013, 23, 2927–2933. [Google Scholar] [CrossRef] [PubMed]
- Hausleiter, J.; Meyer, T.; Hadamitzky, M.; Kastrati, A.; Martinoff, S.; Schömig, A. Prevalence of noncalcified coronary plaques by 64-slice computed tomography in patients with an intermediate risk for significant coronary artery disease. J. Am. Coll. Cardiol. 2006, 48, 312–318. [Google Scholar] [CrossRef] [PubMed]
- Budoff, M.J.; Dowe, D.; Jollis, J.G.; Gitter, M.; Sutherland, J.; Halamert, E.; Scherer, M.; Bellinger, R.; Martin, A.; Benton, R.; et al. Diagnostic performance of 64-multidetector row coronary computed tomographic angiography for evaluation of coronary artery stenosis in individuals without known coronary artery disease: results from the prospective multicenter ACCURACY (Assessment by Coronary Computed Tomographic Angiography of Individuals Undergoing Invasive Coronary Angiography) trial. J. Am. Coll. Cardiol. 2008, 52, 1724–1732. [Google Scholar] [CrossRef] [PubMed]
- Choi, B.J.; Kang, D.K.; Tahk, S.J.; Choi, S.Y.; Yoon, M.H.; Lim, H.S.; Kang, S.J.; Yang, H.M.; Park, J.S.; Zheng, M.; et al. Comparison of 64-slice multidetector computed tomography with spectral analysis of intravascular ultrasound backscatter signals for characterizations of noncalcified coronary arterial plaques. Am. J. Cardiol. 2008, 102, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Fischer, C.; Hulten, E.; Belur, P.; Smith, R.; Voros, S.; Villines, T.C. Coronary CT angiography versus intravascular ultrasound for estimation of coronary stenosis and atherosclerotic plaque burden: A meta-analysis. J. Cardiovasc. Comput. Tomogr. 2013, 7, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Meijboom, W.B.; Meijs, M.F.L.; Schuijf, J.D.; Cramer, M.J.; Mollet, N.R.; van Mieghem, C.A.G.; Nieman, K.; van Werkhoven, J.M.; Pundziute, G.; Weustink, A.C.; et al. Diagnostic accuracy of 64-slice computed tomography coronary angiography: A prospective, multicenter, multivendor study. J. Am. Coll. Cardiol. 2008, 52, 2135–2144. [Google Scholar] [CrossRef] [PubMed]
- Leber, A.W.; Knez, A.; von Ziegler, F.; Becker, A.; Nikolaou, K.; Paul, S.; Wintersperger, B.; Reiser, M.; Becker, C.R.; Steinbeck, G.; et al. Quantification of obstructive and nonobstructive coronary lesions by 64-slice computed tomography: A comparative study with quantitative coronary angiography and intravascular ultrasound. J. Am. Coll. Cardiol. 2005, 46, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Alkadhi, H.; Scheffel, H.; Desbiolles, L.; Gaemperli, O.; Stolzmann, P.; Plass, A.; Goerres, G.W.; Luescher, T.F.; Genoni, M.; Marincek, B.; et al. Dual-source computed tomography coronary angiography: Influence of obesity, calcium load, and heart rate on diagnostic accuracy. Eur. Heart J. 2008, 29, 766–776. [Google Scholar] [CrossRef] [PubMed]
- Voros, S.; Rinehart, S.; Qian, Z.; Joshi, P.; Vazquez, G.; Fischer, C.; Belur, P.; Hulten, E.; Villines, T.C. Coronary atherosclerosis imaging by coronary CT angiography: Current status, correlation with intravascular interrogation and meta-analysis. JACC Cardiovasc. Imaging 2011, 4, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Cheng, V.Y.; Nakazato, R.; Dey, D.; Gurudevan, S.; Tabak, J.; Budoff, M.J.; Karlsberg, R.P.; Min, J.; Berman, D.S. Reproducibility of coronary artery plaque volume and composition quantification by 64-detector row coronary computed tomographic angiography: An intraobserver, interobserver, and interscan variability study. J. Cardiovasc. Comput. Tomogr. 2009, 3, 312–320. [Google Scholar] [CrossRef] [PubMed]
- Leber, A.W.; Becker, A.; Knez, A.; von Ziegler, F.; Sirol, M.; Nikolaou, K.; Ohnesorge, B.; Fayad, Z.A.; Becker, C.R.; Reiser, M.; et al. Accuracy of 64-slice computed tomography to classify and quantify plaque volumes in the proximal coronary system: A comparative study using intravascular ultrasound. J. Am. Coll. Cardiol. 2006, 47, 672–677. [Google Scholar] [CrossRef] [PubMed]
- Pflederer, T.; Schmid, M.; Ropers, D.; Ropers, U.; Komatsu, S.; Daniel, W. G.; Achenbach, S. Interobserver variability of 64-slice computed tomography for the quantification of non-calcified coronary atherosclerotic plaque. Rofo 2007, 179, 953–957. [Google Scholar] [CrossRef] [PubMed]
- Boogers, M.J.; Broersen, A.; van Velzen, J.E.; de Graaf, F.R.; El-Naggar, H.M.; Kitslaar, P.H.; Dijkstra, J.; Delgado, V.; Boersma, E.; de Roos, A.; et al. Automated quantification of coronary plaque with computed tomography: Comparison with intravascular ultrasound using a dedicated registration algorithm for fusion-based quantification. Eur. Heart J. 2012, 33, 1007–1016. [Google Scholar] [CrossRef] [PubMed]
- Rolf, A.; Werner, G.S.; Schuhbäck, A.; Rixe, J.; Möllmann, H.; Nef, H.M.; Gundermann, C.; Liebetrau, C.; Krombach, G.A.; Hamm, C.W.; et al. Preprocedural coronary CT angiography significantly improves success rates of PCI for chronic total occlusion. Int. J. Cardiovasc. Imaging 2013, 29, 1819–1827. [Google Scholar] [CrossRef] [PubMed]
- Ueno, K.; Kawamura, A.; Onizuka, T.; Kawakami, T.; Nagatomo, Y.; Hayashida, K.; Yuasa, S.; Maekawa, Y.; Anzai, T.; Jinzaki, M.; et al. Effect of preoperative evaluation by multidetector computed tomography in percutaneous coronary interventions of chronic total occlusions. Int. J. Cardiol. 2012, 156, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Soon, K.H.; Cox, N.; Wong, A.; Chaitowitz, I.; Macgregor, L.; Santos, P.T.; Selvanayagam, J.B.; Farouque, H.M.O.; Rametta, S.; Bell, K.W.; et al. CT coronary angiography predicts the outcome of percutaneous coronary intervention of chronic total occlusion. J. Interv. Cardiol. 2007, 20, 359–366. [Google Scholar] [CrossRef] [PubMed]
- Mintz, G.S.; Nissen, S.E.; Anderson, W.D.; Bailey, S.R.; Erbel, R.; Fitzgerald, P.J.; Pinto, F.J.; Rosenfield, K.; Siegel, R.J.; Tuzcu, E.M.; et al. American college of cardiology clinical expert consensus document on standards for acquisition, measurement and reporting of Intravascular Ultrasound Studies (IVUS): A report of the American college of cardiology task force on clinical expert consensus documents. J. Am. Coll. Cardiol. 2001, 37, 1478–1492. [Google Scholar] [CrossRef] [PubMed]
- Nissen, S.E.; Yock, P. Intravascular ultrasound: Novel pathophysiological insights and current clinical applications. Circulation 2001, 103, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Marwan, M.; Taher, M.A.; El Meniawy, K.; Awadallah, H.; Pflederer, T.; Schuhbäck, A.; Ropers, D.; Daniel, W.G.; Achenbach, S. In vivo CT detection of lipid-rich coronary artery atherosclerotic plaques using quantitative histogram analysis: A head to head comparison with IVUS. Atherosclerosis 2011, 215, 110–115. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, S.; Flohr, T.; Kopp, A.F.; Meisner, C.; Kuettner, A.; Herdeg, C.; Baumbach, A.; Ohnesorge, B. Accuracy of density measurements within plaques located in artificial coronary arteries by X-ray multislice CT: Results of a phantom study. J. Comput. Assist. Tomogr. 2001, 25, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Cademartiri, F.; Mollet, N.R.; Runza, G.; Bruining, N.; Hamers, R.; Somers, P.; Knaapen, M.; Verheye, S.; Midiri, M.; Krestin, G.P.; et al. Influence of intracoronary attenuation on coronary plaque measurements using multislice computed tomography: Observations in an ex vivo model of coronary computed tomography angiography. Eur. Radiol. 2005, 15, 1426–1431. [Google Scholar] [CrossRef] [PubMed]
- Obaid, D.R.; Calvert, P.A.; Gopalan, D.; Parker, R.A.; West, N.E.J.; Goddard, M.; Rudd, J.H.F.; Bennett, M.R. Dual-energy computed tomography imaging to determine atherosclerotic plaque composition: A prospective study with tissue validation. J. Cardiovasc. Comput. Tomogr. 2014, 8, 230–237. [Google Scholar] [CrossRef] [PubMed]
- Narula, J.; Finn, A.V.; Demaria, A.N. Picking plaques that pop…. J. Am. Coll. Cardiol. 2005, 45, 1970–1973. [Google Scholar] [CrossRef]
- Motoyama, S.; Kondo, T.; Sarai, M.; Sugiura, A.; Harigaya, H.; Sato, T.; Inoue, K.; Okumura, M.; Ishii, J.; Anno, H.; et al. Multislice computed tomographic characteristics of coronary lesions in acute coronary syndromes. J. Am. Coll. Cardiol. 2007, 50, 319–326. [Google Scholar] [CrossRef] [PubMed]
- Motoyama, S.; Sarai, M.; Harigaya, H.; Anno, H.; Inoue, K.; Hara, T.; Naruse, H.; Ishii, J.; Hishida, H.; Wong, N.D.; et al. Computed tomographic angiography characteristics of atherosclerotic plaques subsequently resulting in acute coronary syndrome. J. Am. Coll. Cardiol. 2009, 54, 49–57. [Google Scholar] [CrossRef]
- Hoffmann, U.; Moselewski, F.; Nieman, K.; Jang, I.K.; Ferencik, M.; Rahman, A.M.; Cury, R.C.; Abbara, S.; Joneidi-Jafari, H.; Achenbach, S.; et al. Noninvasive assessment of plaque morphology and composition in culprit and stable lesions in acute coronary syndrome and stable lesions in stable angina by multidetector computed tomography. J. Am. Coll. Cardiol. 2006, 47, 1655–1662. [Google Scholar] [CrossRef] [PubMed]
- Maurovich-Horvat, P.; Schlett, C.L.; Alkadhi, H.; Nakano, M.; Otsuka, F.; Stolzmann, P.; Scheffel, H.; Ferencik, M.; Kriegel, M.F.; Seifarth, H.; et al. The napkin-ring sign indicates advanced atherosclerotic lesions in coronary CT angiography. JACC Cardiovasc. Imaging 2012, 5, 1243–1252. [Google Scholar] [CrossRef] [PubMed]
- Otsuka, K.; Fukuda, S.; Tanaka, A.; Nakanishi, K.; Taguchi, H.; Yoshikawa, J.; Shimada, K.; Yoshiyama, M. Napkin-ring sign on coronary CT angiography for the prediction of acute coronary syndrome. JACC Cardiovasc. Imaging 2013, 6, 448–457. [Google Scholar] [CrossRef] [PubMed]
- Dweck, M.R.; Chow, M.W.L.; Joshi, N.V.; Williams, M.C.; Jones, C.; Fletcher, A.M.; Richardson, H.; White, A.; McKillop, G.; van Beek, E.J.R.; et al. Coronary arterial 18F-sodium fluoride uptake: A novel marker of plaque biology. J. Am. Coll. Cardiol. 2012, 59, 1539–1548. [Google Scholar] [CrossRef] [PubMed]
- Joshi, N.V.; Vesey, A.T.; Williams, M.C.; Shah, A.S.V.; Calvert, P.A.; Craighead, F.H.M.; Yeoh, S.E.; Wallace, W.; Salter, D.; Fletcher, A.M.; et al. 18F-fluoride positron emission tomography for identification of ruptured and high-risk coronary atherosclerotic plaques: A prospective clinical trial. Lancet 2014, 383, 705–713. [Google Scholar] [CrossRef] [PubMed]
- De Bruyne, B.; Fearon, W.F.; Pijls, N.H.J.; Barbato, E.; Tonino, P.; Piroth, Z.; Jagic, N.; Mobius-Winckler, S.; Rioufol, G.; Witt, N.; et al. Fractional flow reserve-guided PCI for stable coronary artery disease. N. Engl. J. Med. 2014, 371, 1208–1217. [Google Scholar] [CrossRef] [PubMed]
- Meijboom, W.B.; van Mieghem, C.A.G.; van Pelt, N.; Weustink, A.; Pugliese, F.; Mollet, N.R.; Boersma, E.; Regar, E.; van Geuns, R.J.; de Jaegere, P.J.; et al. Comprehensive assessment of coronary artery stenoses: Computed tomography coronary angiography versus conventional coronary angiography and correlation with fractional flow reserve in patients with stable angina. J. Am. Coll. Cardiol. 2008, 52, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Min, J.K.; Leipsic, J.; Pencina, M.J.; Berman, D.S.; Koo, B.K.; van Mieghem, C.; Erglis, A.; Lin, F.Y.; Dunning, A.M.; Apruzzese, P.; et al. Diagnostic accuracy of fractional flow reserve from anatomic CT angiography. JAMA 2012, 308, 1237–1245. [Google Scholar] [CrossRef] [PubMed]
- Gaur, S.; Bezerra, H.G.; Lassen, J.F.; Christiansen, E.H.; Tanaka, K.; Jensen, J.M.; Oldroyd, K.G.; Leipsic, J.; Achenbach, S.; Kaltoft, A.K.; et al. Fractional flow reserve derived from coronary CT angiography: Variation of repeated analyses. J. Cardiovasc. Comput. Tomogr. 2014, 8, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Taylor, C.A.; Fonte, T.A.; Min, J.K. Computational fluid dynamics applied to cardiac computed tomography for noninvasive quantification of fractional flow reserve: Scientific basis. J. Am. Coll. Cardiol. 2013, 61, 2233–2241. [Google Scholar] [CrossRef] [PubMed]
- Goldstein, J.A.; Gallagher, M.J.; O’Neill, W.W.; Ross, M.A.; O’Neil, B.J.; Raff, G.L. A randomized controlled trial of multi-slice coronary computed tomography for evaluation of acute chest pain. J. Am. Coll. Cardiol. 2007, 49, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, U.; Bamberg, F.; Chae, C.U.; Nichols, J.H.; Rogers, I.S.; Seneviratne, S.K.; Truong, Q.A.; Cury, R.C.; Abbara, S.; Shapiro, M.D.; et al. Coronary computed tomography angiography for early triage of patients with acute chest pain: The ROMICAT (Rule Out Myocardial Infarction using Computer Assisted Tomography) trial. J. Am. Coll. Cardiol. 2009, 53, 1642–1650. [Google Scholar] [CrossRef] [PubMed]
- Schlett, C.L.; Banerji, D.; Siegel, E.; Bamberg, F.; Lehman, S.J.; Ferencik, M.; Brady, T.J.; Nagurney, J.T.; Hoffmann, U.; Truong, Q.A. Prognostic value of CT angiography for major adverse cardiac events in patients with acute chest pain from the emergency department: 2-Year outcomes of the ROMICAT trial. JACC Cardiovasc. Imaging 2011, 4, 481–491. [Google Scholar] [CrossRef] [PubMed]
- Al-Mallah, M.H.; Qureshi, W.; Lin, F.Y.; Achenbach, S.; Berman, D.S.; Budoff, M.J.; Callister, T.Q.; Chang, H.J.; Cademartiri, F.; Chinnaiyan, K.; et al. Does coronary CT angiography improve risk stratification over coronary calcium scoring in symptomatic patients with suspected coronary artery disease? Results from the prospective multicenter international CONFIRM registry. Eur. Heart J. Cardiovasc. Imaging 2014, 15, 267–274. [Google Scholar] [CrossRef] [PubMed]
- Andreini, D.; Pontone, G.; Mushtaq, S.; Bartorelli, A.L.; Bertella, E.; Antonioli, L.; Formenti, A.; Cortinovis, S.; Veglia, F.; Annoni, A.; et al. A long-term prognostic value of coronary CT angiography in suspected coronary artery disease. JACC Cardiovasc. Imaging 2012, 5, 690–701. [Google Scholar] [CrossRef] [PubMed]
- Hadamitzky, M.; Täubert, S.; Deseive, S.; Byrne, R.A.; Martinoff, S.; Schömig, A.; Hausleiter, J. Prognostic value of coronary computed tomography angiography during 5 years of follow-up in patients with suspected coronary artery disease. Eur. Heart J. 2013, 34, 3277–3285. [Google Scholar] [CrossRef] [PubMed]
- Hadamitzky, M.; Freissmuth, B.; Meyer, T.; Hein, F.; Kastrati, A.; Martinoff, S.; Schömig, A.; Hausleiter, J. Prognostic value of coronary computed tomographic angiography for prediction of cardiac events in patients with suspected coronary artery disease. JACC Cardiovasc. Imaging 2009, 2, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Min, J.K.; Shaw, L.J.; Devereux, R.B.; Okin, P.M.; Weinsaft, J.W.; Russo, D.J.; Lippolis, N.J.; Berman, D.S.; Callister, T.Q. Prognostic value of multidetector coronary computed tomographic angiography for prediction of all-cause mortality. J. Am. Coll. Cardiol. 2007, 50, 1161–1170. [Google Scholar] [CrossRef] [PubMed]
- Pundziute, G.; Schuijf, J.D.; Jukema, J.W.; Boersma, E.; de Roos, A.; van der Wall, E.E.; Bax, J.J. Prognostic value of multislice computed tomography coronary angiography in patients with known or suspected coronary artery disease. J. Am. Coll. Cardiol. 2007, 49, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Ahmadi, N.; Nabavi, V.; Hajsadeghi, F.; Flores, F.; French, W.J.; Mao, S.S.; Shavelle, D.; Ebrahimi, R.; Budoff, M. Mortality incidence of patients with non-obstructive coronary artery disease diagnosed by computed tomography angiography. Am. J. Cardiol. 2011, 107, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Chow, B.J.W.; Wells, G.A.; Chen, L.; Yam, Y.; Galiwango, P.; Abraham, A.; Sheth, T.; Dennie, C.; Beanlands, R.S.; Ruddy, T.D. Prognostic value of 64-slice cardiac computed tomography severity of coronary artery disease, coronary atherosclerosis, and left ventricular ejection fraction. J. Am. Coll. Cardiol. 2010, 55, 1017–1028. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Lu, B.; Gao, Y.; Jiang, S.; Wang, Y.; Li, W.; Budoff, M.J. Prognostic value of coronary CT angiography and calcium score for major adverse cardiac events in outpatients. JACC Cardiovasc. Imaging 2012, 5, 990–999. [Google Scholar] [CrossRef] [PubMed]
- Hulten, E.A.; Carbonaro, S.; Petrillo, S.P.; Mitchell, J.D.; Villines, T.C. Prognostic value of cardiac computed tomography angiography: A systematic review and meta-analysis. J. Am. Coll. Cardiol. 2011, 57, 1237–1247. [Google Scholar] [CrossRef] [PubMed]
- Choi, E.K.; Choi, S.I.; Rivera, J.J.; Nasir, K.; Chang, S.A.; Chun, E.J.; Kim, H.K.; Choi, D.J.; Blumenthal, R.S.; Chang, H.J. Coronary computed tomography angiography as a screening tool for the detection of occult coronary artery disease in asymptomatic individuals. J. Am. Coll. Cardiol. 2008, 52, 357–365. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eckert, J.; Schmidt, M.; Magedanz, A.; Voigtländer, T.; Schmermund, A. Coronary CT Angiography in Managing Atherosclerosis. Int. J. Mol. Sci. 2015, 16, 3740-3756. https://doi.org/10.3390/ijms16023740
Eckert J, Schmidt M, Magedanz A, Voigtländer T, Schmermund A. Coronary CT Angiography in Managing Atherosclerosis. International Journal of Molecular Sciences. 2015; 16(2):3740-3756. https://doi.org/10.3390/ijms16023740
Chicago/Turabian StyleEckert, Joachim, Marco Schmidt, Annett Magedanz, Thomas Voigtländer, and Axel Schmermund. 2015. "Coronary CT Angiography in Managing Atherosclerosis" International Journal of Molecular Sciences 16, no. 2: 3740-3756. https://doi.org/10.3390/ijms16023740
APA StyleEckert, J., Schmidt, M., Magedanz, A., Voigtländer, T., & Schmermund, A. (2015). Coronary CT Angiography in Managing Atherosclerosis. International Journal of Molecular Sciences, 16(2), 3740-3756. https://doi.org/10.3390/ijms16023740




