Antiproliferative and Antioxidant Properties of Anthocyanin Rich Extracts from Blueberry and Blackcurrant Juice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Identification and Quantification of Anthocyanins

| Sample Name | Peak No | Rt (min) | Peak Assignment | Concentration (mg/100mL) * | [M + H] + Ion/Fragments |
|---|---|---|---|---|---|
| Blueberry | 1 | 8.1 | Delphinidin-3-O-galactoside | 11.46 ± 1.12 | 465/303 |
| 2 | 9.3 | Delphinidin-3-O-glucoside | 22.63 ± 1.45 | 465/303 | |
| 3 | 11.2 | Cyanidin-3-O-galactoside | 6.42 ± 1.60 | 449/287 | |
| 4 | 11.5 | Delphinidin-3-O-arabinoside | 11.23 ± 1.93 | 435/303 | |
| 5 | 12.9 | Cyanidin-3-O-glucoside | 14.2 ± 1.86 | 449/287 | |
| 6 | 13.6 | Petunidin-3-O-galactoside | 6.33 ± 1.67 | 479/317 | |
| 7 | 15.2 | Cyanidin-3-O-arabinoside | 22.3 ± 1.54 | 419/287 | |
| 8 | 18.0 | Paeonidin-3-O-galactoside | 2.65 ± 1.04 | 463/301 | |
| 9 | 20.5 | Petunidin-3-O-arabinoside | 5.42 ± 1.33 | 449/317 | |
| 10 | 23.4 | Malvidin-3-O-galactoside | 10.66 ± 1.21 | 493/331 | |
| 11 | 26.3 | Malvidin-3-O-glucoside | 26 ± 1.96 | 493/331 | |
| 12 | 26.3 | Malvidin-3-O-arabinoside | 4.6 ± 2.06 | 463/331 | |
| Total | 143.90 ± 1.56 | ||||
| Blackcurrant | 1 | 10.72 | Delphinidin-3-O-glucoside | 22.3 ± 1.36 | 465/303 |
| 2 | 11.77 | Delphinidin-3-O-rutinoside | 57.95 ± 2.31 | 300/283/252 | |
| 3 | 14.8 | Cyanidin-3-O-glucoside | 9.9 ± 1.79 | 449/287 | |
| 4 | 16.01 | Cyanidin-3-O- rutinoside | 50.6 ± 1.63 | 595/287 | |
| Total | 140.75 ± 1.77 | ||||
2.2. Total Monomeric Anthocyanins, Flavonoid and Total Polyphenol Content
| Sample | Total Polyphenol GAE mg/100 mL a | Total Flavonoid QE mg/100 g b | Total Anthocyanin C3GE mg/100 g c |
|---|---|---|---|
| Blueberry | 711.3 ± 0.82 # | 96.0 ± 0.96 # | 360.0 ± 0.76 # |
| Blackcurrant | 580.4 ± 0.86 | 84.6 ± 0.71 | 116.1 ± 0.59 |
2.3. Antioxidant Activity
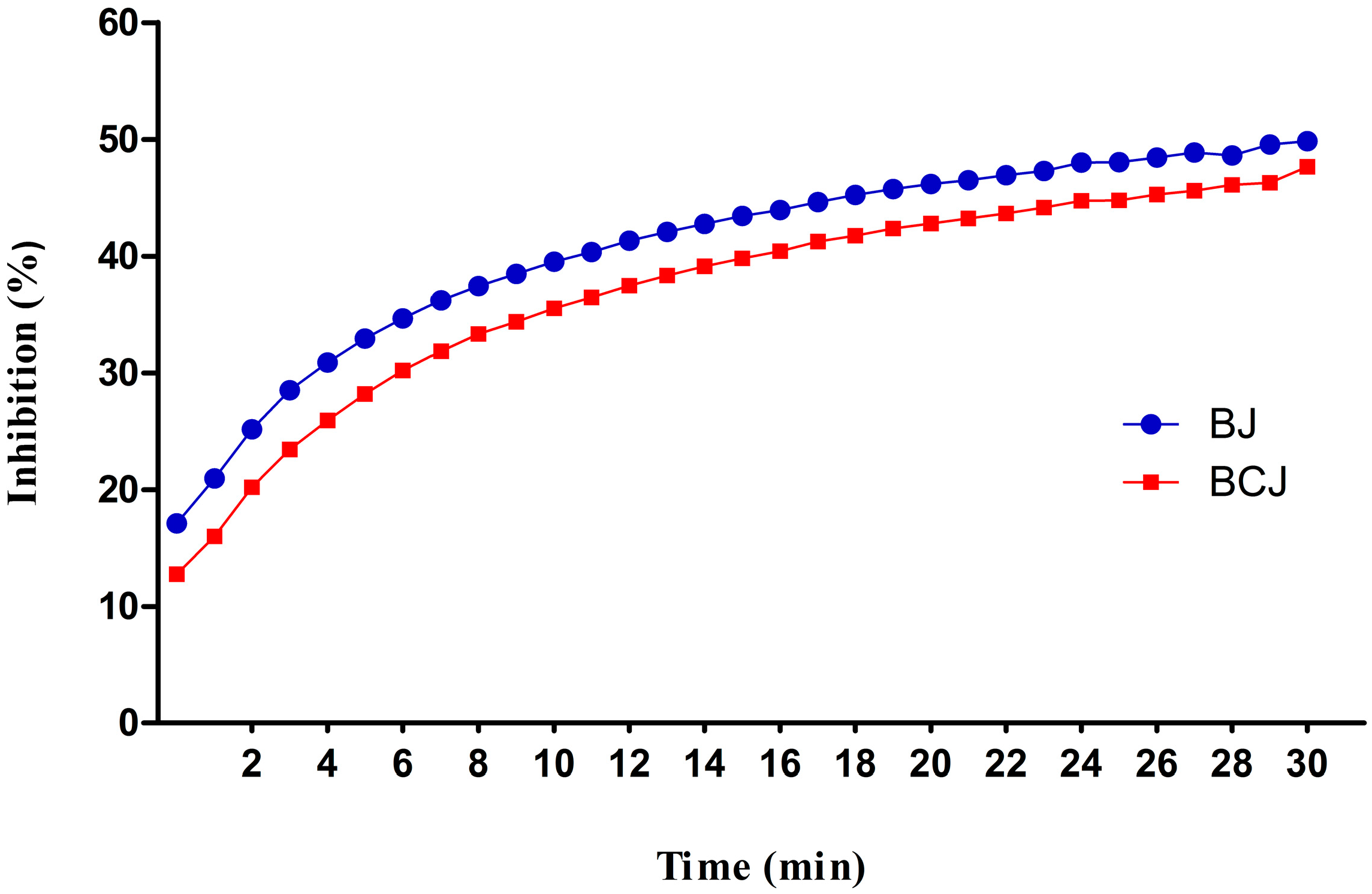
2.4. Analysis of Cell Proliferation
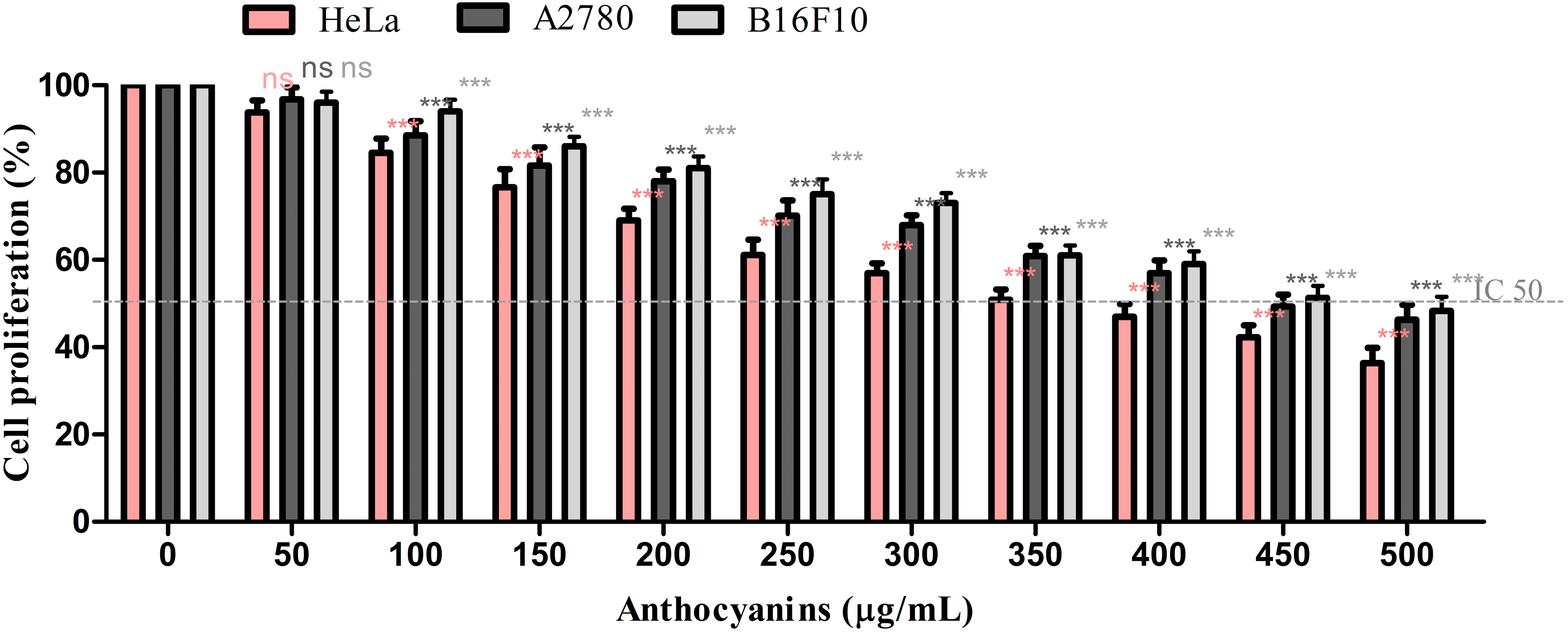
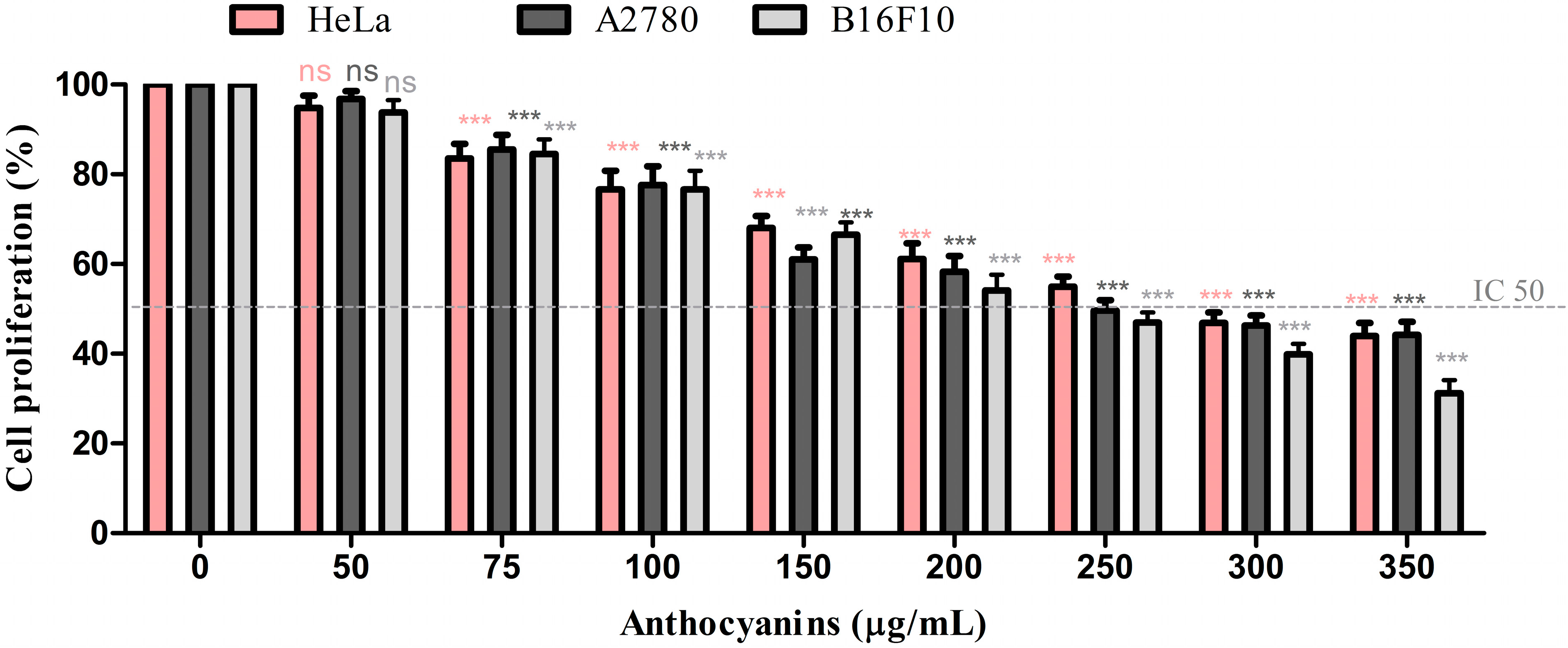
3. Experimental Section
3.1. Sample Preparation
3.2. Quantification of Total Anthocyanins
3.3. HPLC-PDA/-ESI-MS Identification and Quantification of Anthocyanins
3.4. Total Flavonoid Content
3.5. DPPH-Scavenging Activity
3.6. Oxygen Radical Absorbance Capacity (ORAC) Assay
3.7. Cell Culture
3.8. Analysis of Cell Proliferation
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Steinmetz, K.A.; Potter, J.D. Vegetables, fruit, and cancer prevention: A review. J. Am. Diet. Assoc. 1996, 96, 1027–1039. [Google Scholar] [PubMed]
- Philpott, M.; Lim, C.C.; Ferguson, L. Dietary protection against free radicals: A case for multiple testing to establish structure-activity relationships for antioxidant potential of anthocyanic plant species. Int. J. Mol. Sci. 2009, 10, 1081–1103. [Google Scholar] [CrossRef] [PubMed]
- Muselík, J.; García-Alonso, M.; Martín-López, M.; Žemlička, M.; Rivas-Gonzalo, J. Measurement of antioxidant activity of wine catechins, procyanidins, anthocyanins and pyranoanthocyanins. Int. J. Mol. Sci. 2007, 8, 797–809. [Google Scholar] [CrossRef]
- Li, J.; Jiang, Y. Litchi flavonoids: Isolation, identification and biological activity. Molecules 2007, 12, 745–758. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.H. Dietary bioactive compounds and their health implications. J. Food Sci. 2013, 78, 1750–3841. [Google Scholar]
- Liu, R.H. Health-promoting components of fruits and vegetables in the diet. Adv. Nutr. 2013, 4, S384–S392. [Google Scholar] [CrossRef]
- Slavin, J.L.; Lloyd, B. Health benefits of fruits and vegetables. Adv. Nutr. 2012, 3, 506–516. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Cao, G.; Prior, R.L. Oxygen radical absorbing capacity of anthocyanins. J. Agric. Food Chem. 1997, 45, 304–309. [Google Scholar] [CrossRef]
- He, J.; Giusti, M.M. High-purity isolation of anthocyanins mixtures from fruits and vegetables—A novel solid-phase extraction method using mixed mode cation-exchange chromatography. J. Chromatogr. A 2011, 1218, 7914–7922. [Google Scholar] [CrossRef] [PubMed]
- Gehm, B.D.; McAndrews, J.M.; Chien, P.Y.; Jameson, J.L. Resveratrol, a polyphenolic compound found in grapes and wine, is an agonist for the estrogen receptor. Proc. Natl. Acad. Sci. USA 1997, 94, 14138–14143. [Google Scholar] [CrossRef] [PubMed]
- Welch, C.R.; Wu, Q.; Simon, J.E. Recent advances in anthocyanin analysis and characterization. Curr. Anal. Chem. 2008, 4, 75–101. [Google Scholar] [CrossRef]
- Brouillard, R. Chapter 1—chemical structure of anthocyanins. In Anthocyanins as Food Colors; Markakis, P., Ed.; Academic Press: New York, NY, USA, 1982; pp. 1–40. [Google Scholar]
- Bochi, V.C.; Barcia, M.T.; Rodrigues, D.; Speroni, C.S.; Giusti, M.M.; Godoy, H.T. Polyphenol extraction optimisation from ceylon gooseberry (dovyalis hebecarpa) pulp. Food Chem. 2014, 164, 347–354. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Mu, L.; Yan, G.L.; Liang, N.N.; Pan, Q.H.; Wang, J.; Reeves, M.J.; Duan, C.Q. Biosynthesis of anthocyanins and their regulation in colored grapes. Molecules 2010, 15, 9057–9091. [Google Scholar] [CrossRef] [PubMed]
- Adlercreutz, H. Epidemiology of phytoestrogens. Baillieres Clin. Endocrinol. Metab. 1998, 12, 605–623. [Google Scholar] [CrossRef] [PubMed]
- Fung, T.T.; Chiuve, S.E.; Willett, W.C.; Hankinson, S.E.; Hu, F.B.; Holmes, M.D. Intake of specific fruits and vegetables in relation to risk of estrogen receptor-negative breast cancer among postmenopausal women. Breast Cancer Res. Treat. 2013, 138, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Serafino, A.; Sinibaldi-Vallebona, P.; Lazzarino, G.; Tavazzi, B.; Rasi, G.; Pierimarchi, P.; Andreola, F.; Moroni, G.; Galvano, G.; Galvano, F.; et al. Differentiation of human melanoma cells induced by cyanidin-3-o-beta-glucopyranoside. FASEB J. 2004, 18, 1940–1942. [Google Scholar] [PubMed]
- Huang, H.P.; Shih, Y.W.; Chang, Y.C.; Hung, C.N.; Wang, C.J. Chemoinhibitory effect of mulberry anthocyanins on melanoma metastasis involved in the ras/pi3k pathway. J. Agric. Food Chem. 2008, 56, 9286–9293. [Google Scholar] [CrossRef] [PubMed]
- Akim, A.M.; Ling, L.C.; Rahmat, A.; Zakaria, Z.A. Antioxidant and anti-proliferative activities of roselle juice on caov-3, mcf-7, mda-mb-231 and hela cancer cell lines. Afr. J. Pharm. Pharmacol. 2011, 5, 957–965. [Google Scholar]
- Shaughnessy, K.S.; Boswall, I.A.; Scanlan, A.P.; Gottschall-Pass, K.T.; Sweeney, M.I. Diets containing blueberry extract lower blood pressure in spontaneously hypertensive stroke-prone rats. Nutr. Res. 2009, 29, 130–138. [Google Scholar] [CrossRef] [PubMed]
- Kalt, W.; Foote, K.; Fillmore, S.A.; Lyon, M.; Van Lunen, T.A.; McRae, K.B. Effect of blueberry feeding on plasma lipids in pigs. Br. J. Nutr. 2008, 100, 70–78. [Google Scholar] [CrossRef] [PubMed]
- De Pascual-Teresa, S.; Moreno, D.A.; García-Viguera, C. Flavanols and anthocyanins in cardiovascular health: A review of current evidence. Int. J. Mol. Sci. 2010, 11, 1679–1703. [Google Scholar]
- Matchett, M.D.; MacKinnon, S.L.; Sweeney, M.I.; Gottschall-Pass, K.T.; Hurta, R.A. Inhibition of matrix metalloproteinase activity in du145 human prostate cancer cells by flavonoids from lowbush blueberry (vaccinium angustifolium): Possible roles for protein kinase c and mitogen-activated protein-kinase-mediated events. J. Nutr. Biochem. 2006, 17, 117–125. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, W.; Jing, H.; Popovich, D.G. Bog bilberry (vaccinium uliginosum l.) extract reduces cultured hep-g2, caco-2, and 3t3-l1 cell viability, affects cell cycle progression, and has variable effects on membrane permeability. J. Food Sci. 2010, 75, H103–H107. [Google Scholar] [CrossRef] [PubMed]
- Yi, W.; Fischer, J.; Krewer, G.; Akoh, C.C. Phenolic compounds from blueberries can inhibit colon cancer cell proliferation and induce apoptosis. J. Agric. Food Chem. 2005, 53, 7320–7329. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Lu, X.; He, G.; Gao, X.; Xu, M.; Zhang, J.; Li, M.; Wang, L.; Li, Z.; Wang, L.; et al. Protective roles of gadd45 and mdm2 in blueberry anthocyanins mediated DNA repair of fragmented and non-fragmented DNA damage in uv-irradiated hepg2 cells. Int. J. Mol. Sci. 2013, 14, 21447–21462. [Google Scholar] [CrossRef] [PubMed]
- Stoner, G.D.; Wang, L.S.; Casto, B.C. Laboratory and clinical studies of cancer chemoprevention by antioxidants in berries. Carcinogenesis 2008, 29, 1665–1674. [Google Scholar] [CrossRef] [PubMed]
- Veberic, R.; Slatnar, A.; Bizjak, J.; Stampar, F.; Mikulic-Petkovsek, M. Anthocyanin composition of different wild and cultivated berry species. LWT Food Sci. Technol. 2015, 60, 509–517. [Google Scholar] [CrossRef]
- Heinonen, M. Antioxidant activity and antimicrobial effect of berry phenolics—A finnish perspective. Mol. Nutr. Food Res. 2007, 51, 684–691. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P. Berry fruits: Compositional elements, biochemical activities, and the impact of their intake on human health, performance, and disease. J. Agric. Food Chem. 2008, 56, 627–629. [Google Scholar] [CrossRef]
- Bonarska-Kujawa, D.; Cyboran, S.; Żyłka, R.; Oszmiański, J.; Kleszczyńska, H. Biological activity of blackcurrant extracts (ribes nigrum l.) in relation to erythrocyte membranes. Biomed Res. Int. 2014, 2014, 13. [Google Scholar] [CrossRef]
- Kapasakalidis, P.G.; Rastall, R.A.; Gordon, M.H. Extraction of polyphenols from processed black currant (ribes nigrum l.) residues. J. Agric. Food Chem. 2006, 54, 4016–4021. [Google Scholar] [CrossRef] [PubMed]
- Hogan, S.; Chung, H.; Zhang, L.; Li, J.; Lee, Y.; Dai, Y.; Zhou, K. Antiproliferative and antioxidant properties of anthocyanin-rich extract from açai. Food Chem. 2010, 118, 208–214. [Google Scholar] [CrossRef]
- Zhang, Z.; Knobloch, T.J.; Seamon, L.G.; Stoner, G.D.; Cohn, D.E.; Paskett, E.D.; Fowler, J.M.; Weghorst, C.M. A black raspberry extract inhibits proliferation and regulates apoptosis in cervical cancer cells. Gynecol. Oncol. 2011, 123, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Rubinskiene, M.; Jasutiene, I.; Venskutonis, P.R.; Viskelis, P. Hplc determination of the composition and stability of blackcurrant anthocyanins. J. Chromatogr. Sci. 2005, 43, 478–482. [Google Scholar] [CrossRef]
- Bordonaba, J.G.; Crespo, P.; Terry, L.A. A new acetonitrile-free mobile phase for hplc-dad determination of individual anthocyanins in blackcurrant and strawberry fruits: A comparison and validation study. Food Chem. 2011, 129, 1265–1273. [Google Scholar] [CrossRef] [PubMed]
- J. Bermúdez-Soto, M.; A. Tomás-Barberán, F. Evaluation of commercial red fruit juice concentrates as ingredients for antioxidant functional juices. Eur. Food Res. Technol. 2004, 219, 133–141. [Google Scholar]
- Goiffon, J.P.; Brun, M.; Bourrier, M.J. High-performance liquid chromatography of red fruit anthocyanins. J. Chromatogr. A 1991, 537, 101–121. [Google Scholar] [CrossRef]
- Jakobek, L.; Šeruga, M.; Medvidović-Kosanović, M.; Novak, I. Antioxidant activity and polyphenols of aronia in comparison to other berry species. ACS 2007, 72, 301–306. [Google Scholar]
- Bunea, A.; Rugina, D.; Sconta, Z.; Pop, R.M.; Pintea, A.; Socaciu, C.; Tabaran, F.; Grootaert, C.; Struijs, K.; VanCamp, J. Anthocyanin determination in blueberry extracts from various cultivars and their antiproliferative and apoptotic properties in b16-f10 metastatic murine melanoma cells. Phytochemistry 2013, 95, 436–444. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Beecher, G.R.; Holden, J.M.; Haytowitz, D.B.; Gebhardt, S.E.; Prior, R.L. Concentrations of anthocyanins in common foods in the united states and estimation of normal consumption. J. Agric. Food Chem. 2006, 54, 4069–4075. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, I.L.; Haren, G.R.; Magnussen, E.L.; Dragsted, L.O.; Rasmussen, S.E. Quantification of anthocyanins in commercial black currant juices by simple high-performance liquid chromatography. Investigation of their pH stability and antioxidative potency. J. Agric. Food Chem. 2003, 51, 5861–5866. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N. Anthocyanins—nature, occurrence and dietary burden. J. Sci. Food Agric. 2000, 80, 1063–1072. [Google Scholar] [CrossRef]
- Costantino, L.; Albasini, A.; Rastelli, G.; Benvenuti, S. Activity of polyphenolic crude extracts as scavengers of superoxide radicals and inhibitors of xanthine oxidase. Planta Med. 1992, 58, 342–344. [Google Scholar] [CrossRef] [PubMed]
- Rotundo, A.; Bounous, G.; Benvenuti, S.; Vampa, G.; Melegari, M.; Soragni, F. Quality and yield of ribes and rubus cultivars grown in southern italy hilly locations. Phytother. Res. 1998, 12, S135–S137. [Google Scholar] [CrossRef]
- Moyer, R.A.; Hummer, K.E.; Finn, C.E.; Frei, B.; Wrolstad, R.E. Anthocyanins, phenolics, and antioxidant capacity in diverse small fruits: Vaccinium, rubus, and ribes. J. Agric. Food Chem. 2002, 50, 519–525. [Google Scholar] [CrossRef] [PubMed]
- Konić-Ristić, A.; Šavikin, K.; Zdunić, G.; Janković, T.; Juranic, Z.; Menković, N.; Stanković, I. Biological activity and chemical composition of different berry juices. Food Chem. 2011, 125, 1412–1417. [Google Scholar] [CrossRef]
- Mattila, P.H.; Hellström, J.; McDougall, G.; Dobson, G.; Pihlava, J.M.; Tiirikka, T.; Stewart, D.; Karjalainen, R. Polyphenol and vitamin C contents in european commercial blackcurrant juice products. Food Chem. 2011, 127, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Benvenuti, S.; Pellati, F.; Melegari, M.; Bertelli, D. Polyphenols, anthocyanins, ascorbic acid, and radical scavenging activity of rubus, ribes, and aronia. J. Food Sci. 2004, 69, FCT164–FCT169. [Google Scholar]
- Piljac-Žegarac, J.; Valek, L.; Martinez, S.; Belščak, A. Fluctuations in the phenolic content and antioxidant capacity of dark fruit juices in refrigerated storage. Food Chem. 2009, 113, 394–400. [Google Scholar] [CrossRef]
- Kai, H.; Fuse, T.; Kunitake, H.; Morishita, K.; Matsuno, K. Comparison of cultivars and seasonal variation in blueberry (vaccinium species) leaf extract on adult t-cell leukemia cell line growth suppression. Medicines 2014, 1, 3–11. [Google Scholar] [CrossRef]
- Prior, R.L.; Cao, G.; Martin, A.; Sofic, E.; McEwen, J.; O’Brien, C.; Lischner, N.; Ehlenfeldt, M.; Kalt, W.; Krewer, G.; et al. Antioxidant capacity as influenced by total phenolic and anthocyanin content, maturity, and variety of vaccinium species. J. Agric. Food Chem. 1998, 46, 2686–2693. [Google Scholar] [CrossRef]
- Wang, C.Y.; Chen, C.T.; Wang, S.Y. Changes of flavonoid content and antioxidant capacity in blueberries after illumination with uv-c. Food Chem. 2009, 117, 426–431. [Google Scholar] [CrossRef]
- Hosseini-Beheshti, E.; Lund, S.T.; Kitts, D.D. Characterization of antioxidant capacity from fruits with distinct anthocyanin biosynthetic pathways. Nutr. Food Sci. 2012, 2, 122–132. [Google Scholar]
- Zhang, Y.; Seeram, N.P.; Lee, R.; Feng, L.; Heber, D. Isolation and identification of strawberry phenolics with antioxidant and human cancer cell antiproliferative properties. J. Agric. Food Chem. 2008, 56, 670–675. [Google Scholar] [CrossRef] [PubMed]
- Seeram, N.P.; Adams, L.S.; Zhang, Y.; Lee, R.; Sand, D.; Scheuller, H.S.; Heber, D. Blackberry, black raspberry, blueberry, cranberry, red raspberry, and strawberry extracts inhibit growth and stimulate apoptosis of human cancer cells in vitro. J. Agric. Food Chem. 2006, 54, 9329–9339. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.X.; Lei, J.T.; LV Shi, J. Bilberry extract anthocyanins on effect of cervical cancer hela cells. Matern Child Health J. 2010, 14, 733–737. [Google Scholar]
- Olsson, M.E.; Gustavsson, K.E.; Andersson, S.; Nilsson, A.; Duan, R.D. Inhibition of cancer cell proliferation in vitro by fruit and berry extracts and correlations with antioxidant levels. J. Agric. Food Chem. 2004, 52, 7264–7271. [Google Scholar] [CrossRef] [PubMed]
- Bishayee, A.; Haznagy-Radnai, E.; Mbimba, T.; Sipos, P.; Morazzoni, P.; Darvesh, A.S.; Bhatia, D.; Hohmann, J. Anthocyanin-rich black currant extract suppresses the growth of human hepatocellular carcinoma cells. Nat. Prod. Commun. 2010, 5, 1613–1618. [Google Scholar] [PubMed]
- Wu, L.C.; Hsu, H.W.; Chen, Y.C.; Chiu, C.C.; Lin, Y.I.; Ho, J.A.A. Antioxidant and antiproliferative activities of red pitaya. Food Chem. 2006, 95, 319–327. [Google Scholar] [CrossRef]
- Khoo, G.M.; Clausen, M.R.; Pedersen, H.L.; Larsen, E. Bioactivity and chemical composition of blackcurrant (ribes nigrum) cultivars with and without pesticide treatment. Food Chem. 2012, 132, 1214–1220. [Google Scholar] [CrossRef]
- Giusti, M.M.; Rodriguez-Saona, L.E.; Wrolstad, R.E. Molar absorptivity and color characteristics of acylated and non-acylated pelargonidin-based anthocyanins. J. Agric. Food Chem. 1999, 47, 4631–4637. [Google Scholar] [CrossRef] [PubMed]
- Giusti, M.M.; Wrolstad, R.E. Characterization and measurement of anthocyanins by uv-visible spectroscopy. In Current Protocols in Food Analytical Chemistry; John Wiley & Sons, Inc.: New York, NY, USA, 2001. [Google Scholar]
- Zhishen, J.; Mengcheng, T.; Jianming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Huang, D.; Ou, B.; Prior, R.L. The chemistry behind antioxidant capacity assays. J. Agric. Food Chem. 2005, 53, 1841–1856. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diaconeasa, Z.; Leopold, L.; Rugină, D.; Ayvaz, H.; Socaciu, C. Antiproliferative and Antioxidant Properties of Anthocyanin Rich Extracts from Blueberry and Blackcurrant Juice. Int. J. Mol. Sci. 2015, 16, 2352-2365. https://doi.org/10.3390/ijms16022352
Diaconeasa Z, Leopold L, Rugină D, Ayvaz H, Socaciu C. Antiproliferative and Antioxidant Properties of Anthocyanin Rich Extracts from Blueberry and Blackcurrant Juice. International Journal of Molecular Sciences. 2015; 16(2):2352-2365. https://doi.org/10.3390/ijms16022352
Chicago/Turabian StyleDiaconeasa, Zoriţa, Loredana Leopold, Dumitriţa Rugină, Huseyin Ayvaz, and Carmen Socaciu. 2015. "Antiproliferative and Antioxidant Properties of Anthocyanin Rich Extracts from Blueberry and Blackcurrant Juice" International Journal of Molecular Sciences 16, no. 2: 2352-2365. https://doi.org/10.3390/ijms16022352
APA StyleDiaconeasa, Z., Leopold, L., Rugină, D., Ayvaz, H., & Socaciu, C. (2015). Antiproliferative and Antioxidant Properties of Anthocyanin Rich Extracts from Blueberry and Blackcurrant Juice. International Journal of Molecular Sciences, 16(2), 2352-2365. https://doi.org/10.3390/ijms16022352







