Selective Analysis of Sulfur-Containing Species in a Heavy Crude Oil by Deuterium Labeling Reactions and Ultrahigh Resolution Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
2.1. Ethylation of Dibenzothiophene (DBT)
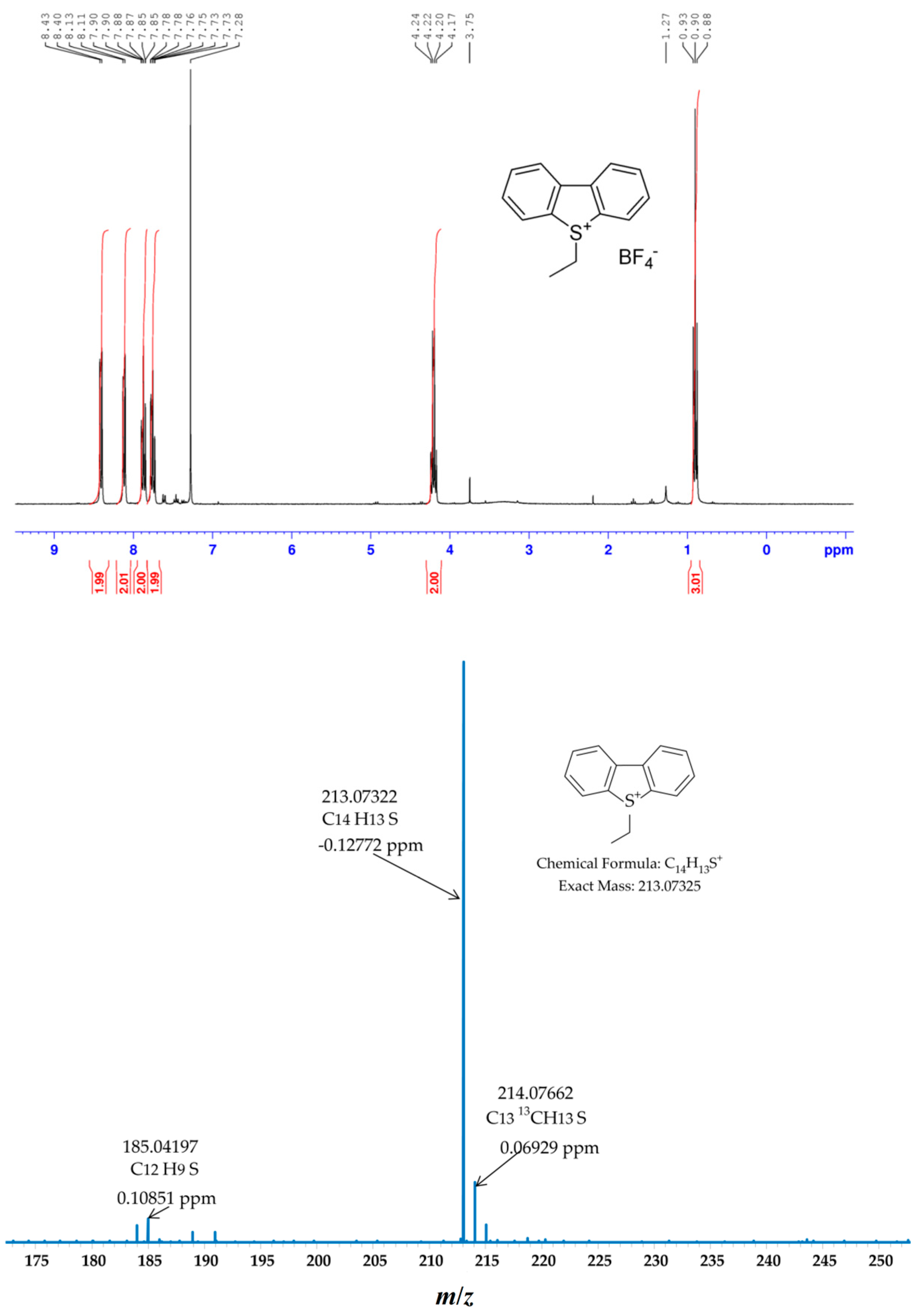
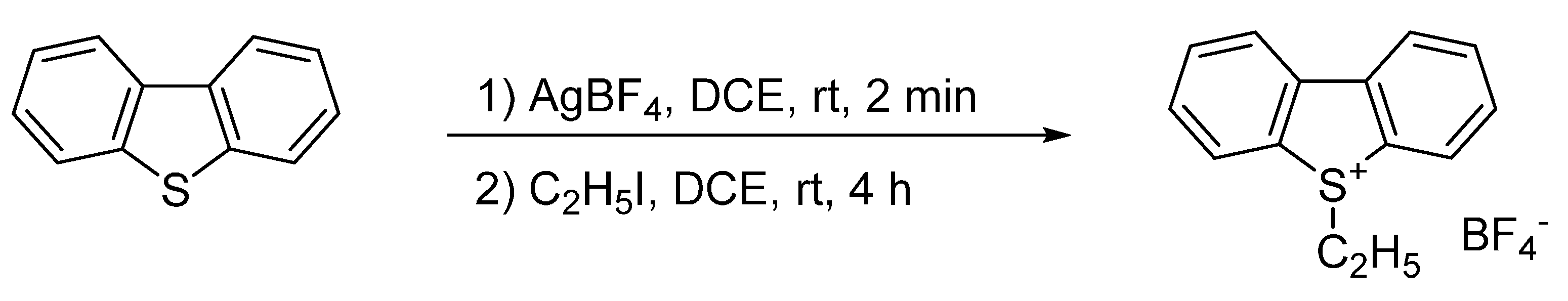
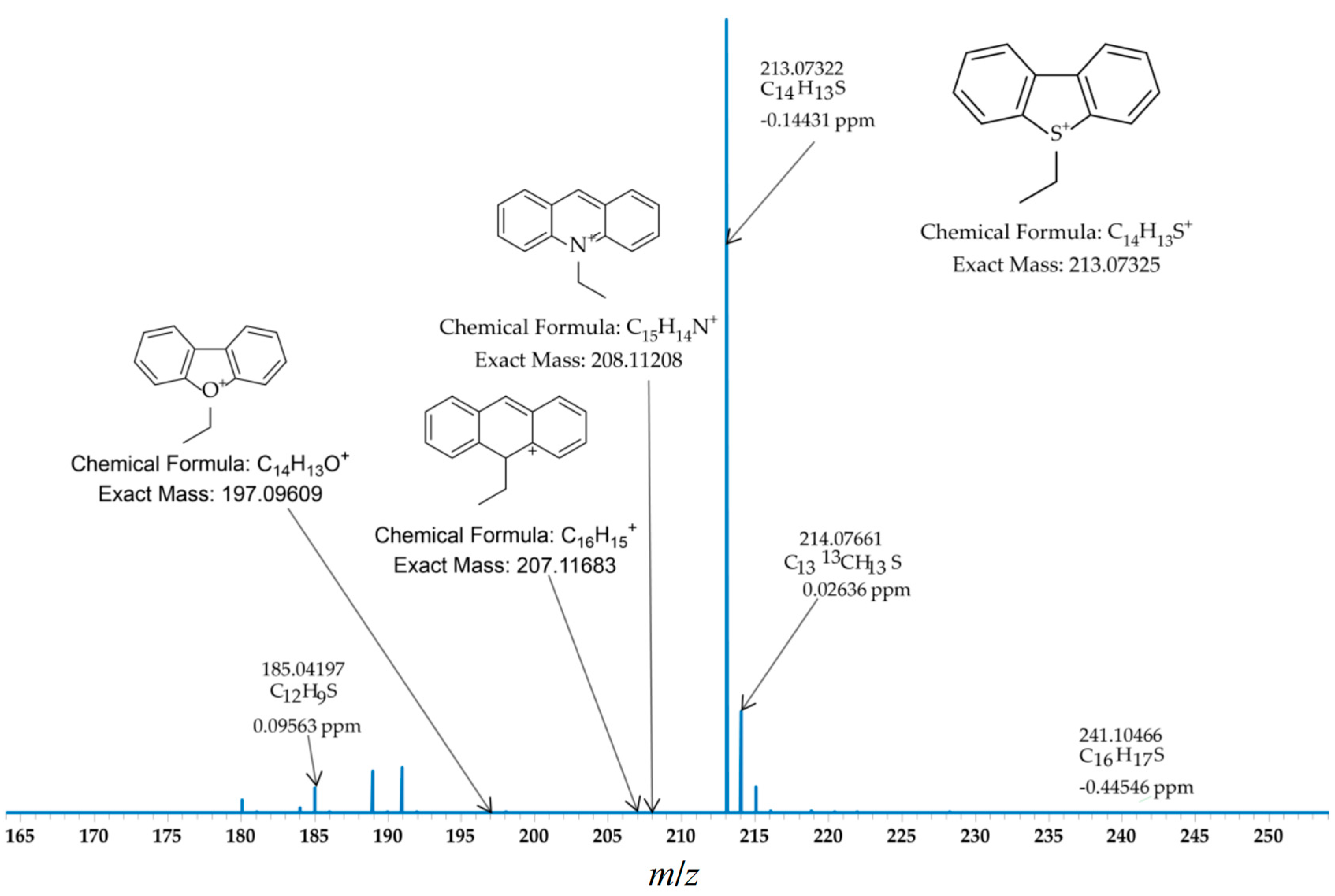
2.2. Ethylation of a Mixture of Standards (ANTH, DBT, ACR and DBF)
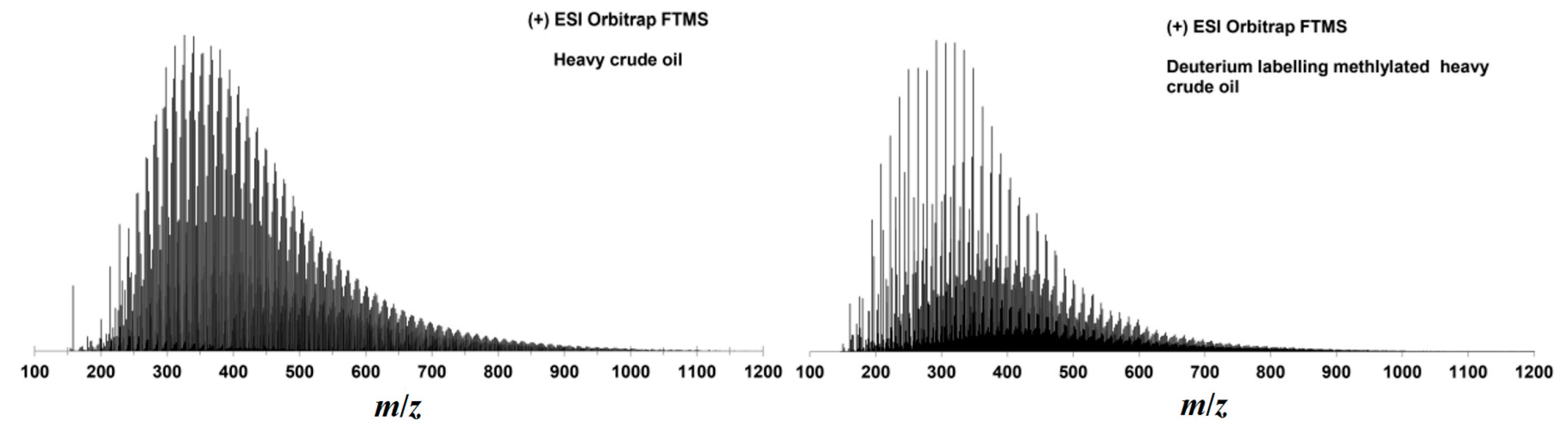
2.3. Methylation and Ethylation of a Heavy Crude Oil
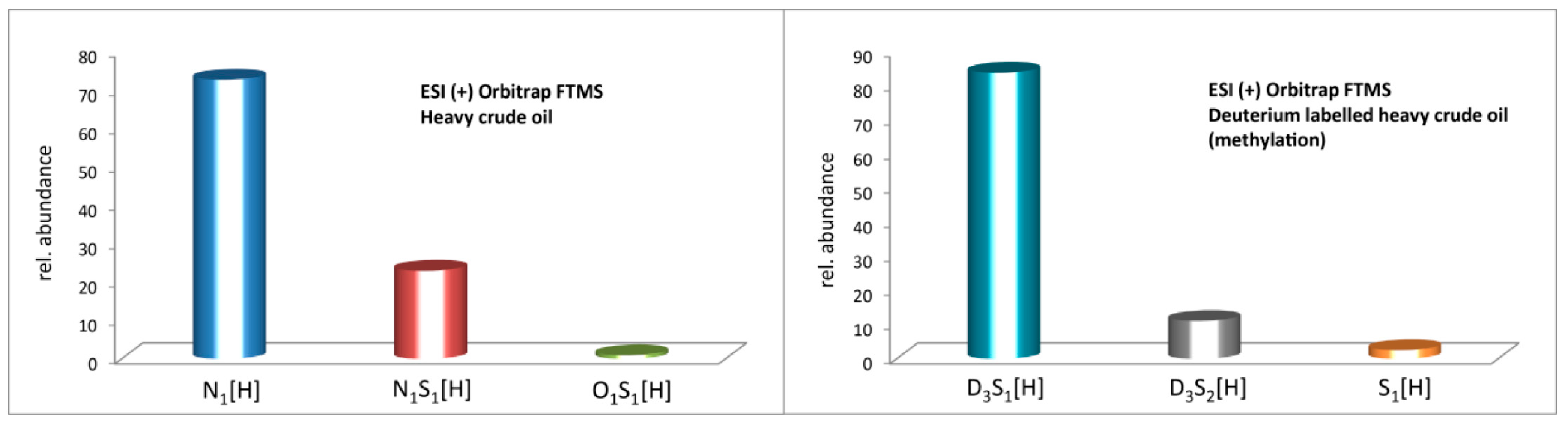
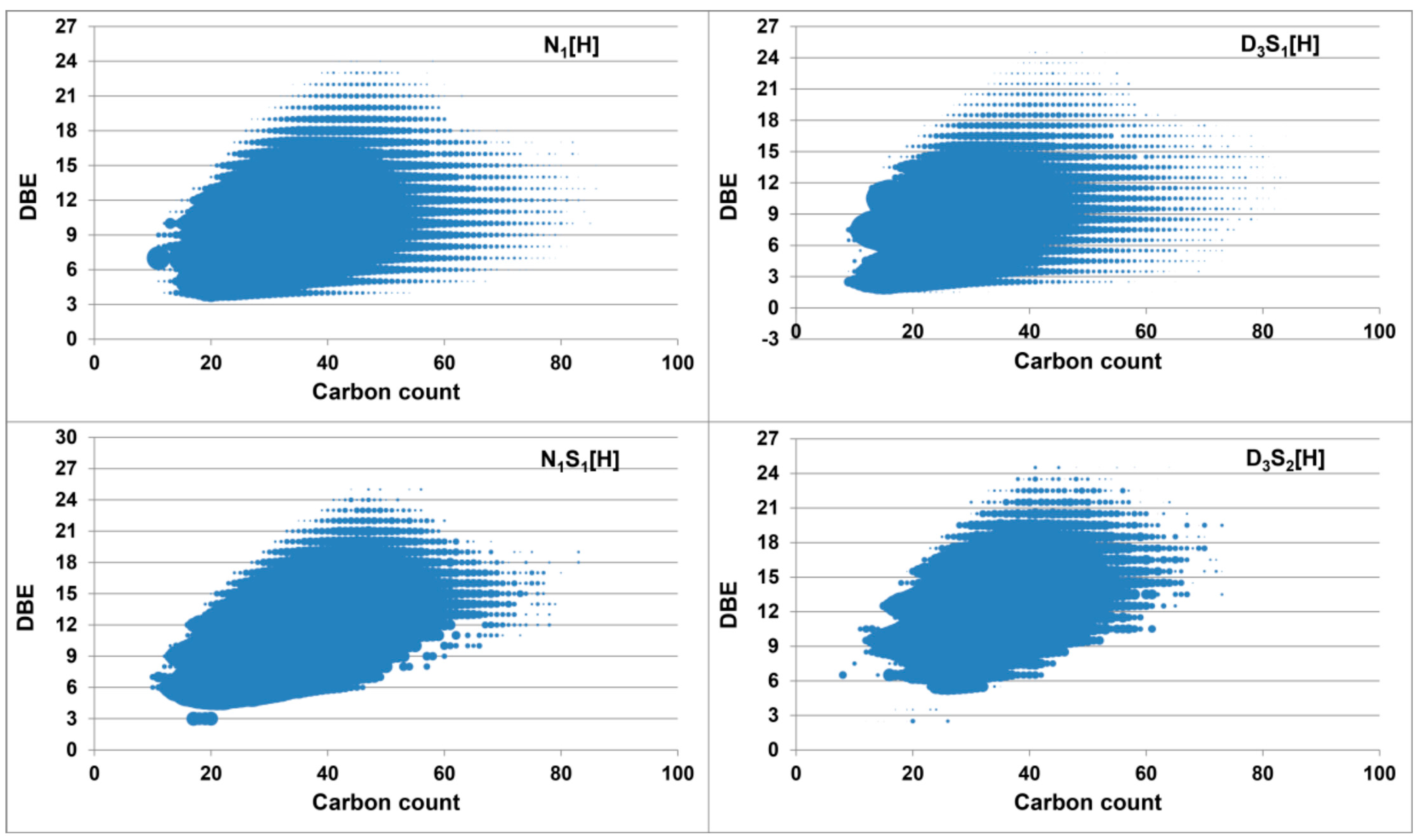
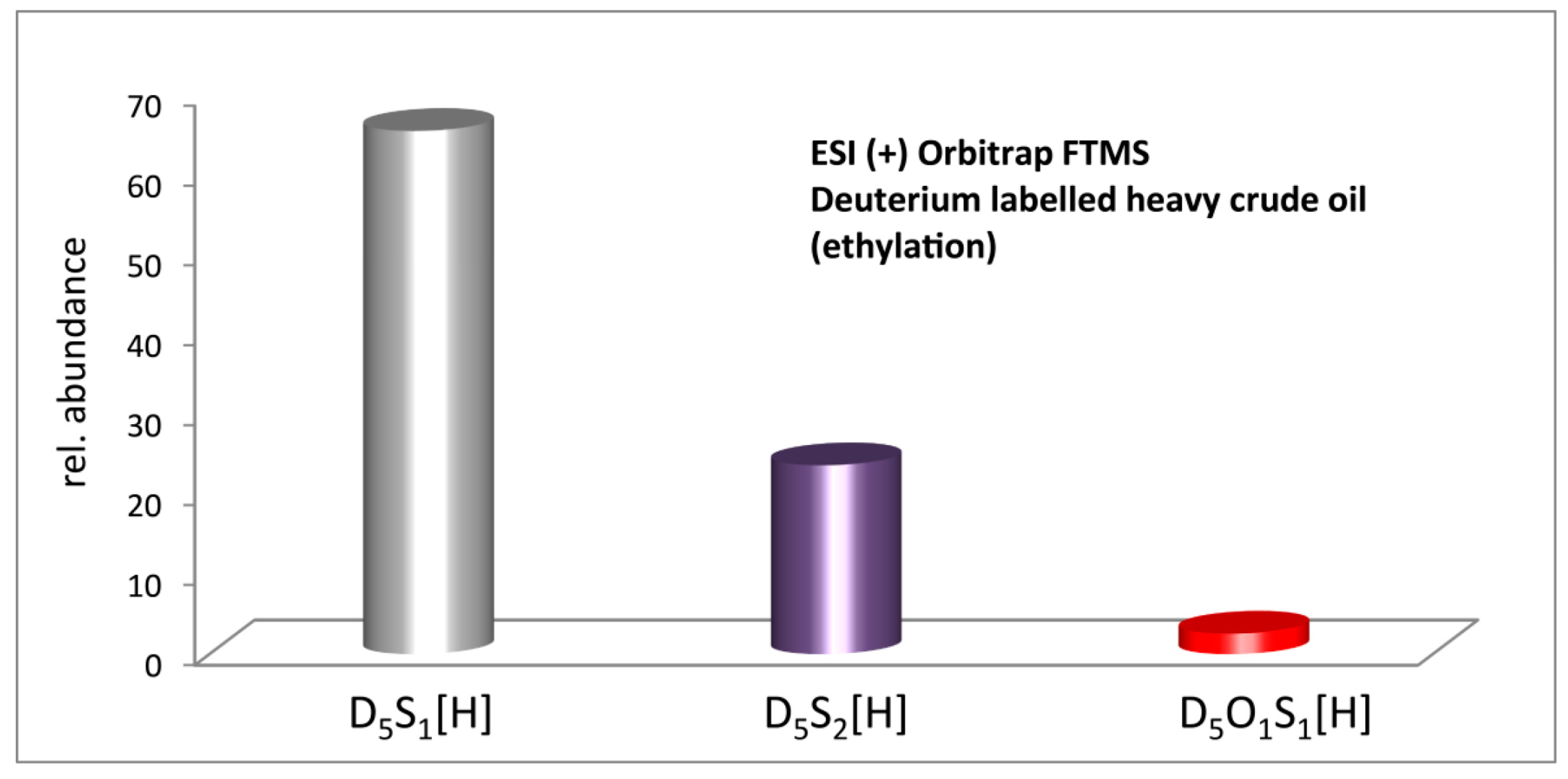
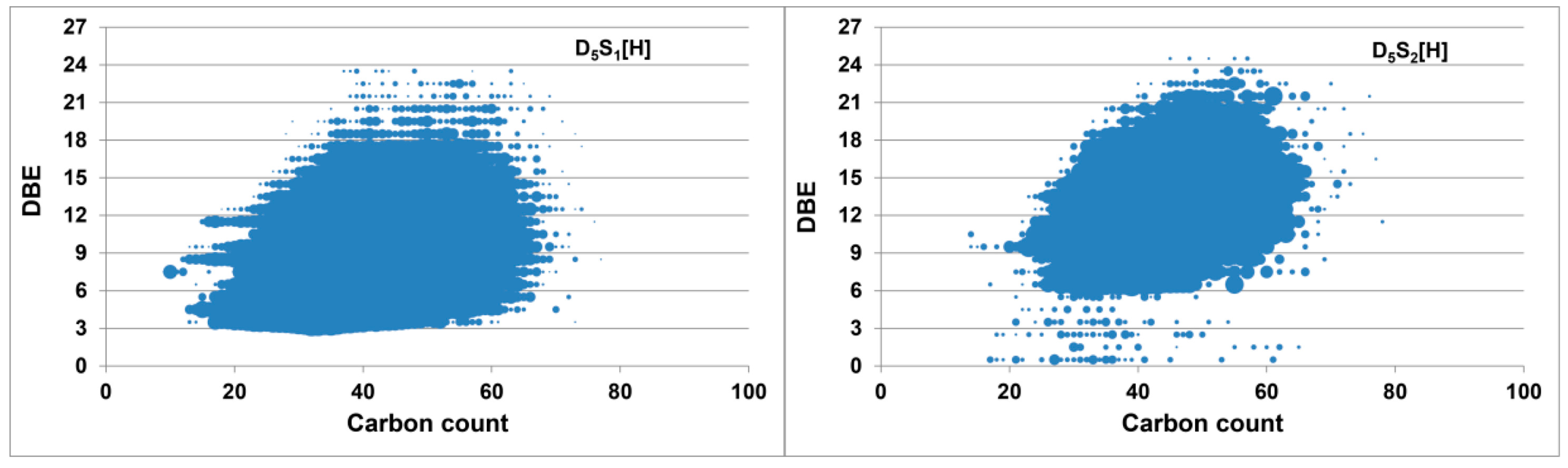
3. Materials and Methods
3.1. Alkylation
3.2. Nuclear Magnetic Resonance Spectroscopy
3.3. ESI Orbitrap FTMS Analysis
3.4. ESI FT-ICR MS Analysis
3.5. Data Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Panda, S.K.; Andersson, J.T.; Schrader, W. Characterization of supercomplex crude oil mixtures: What is really in there? Angew. Chemie Int. Ed. 2009, 48, 1788–1791. [Google Scholar] [CrossRef] [PubMed]
- Marshall, A.G.; Rodgers, R.P. Petroleomics: The next grand challenge for chemical analysis. Acc. Chem. Res. 2004, 37, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.; Ahmed, A.; Islam, A.; Kim, S. Developments in FT-ICR MS instrumentation, ionization techniques, and data interpretation methods for petroleomics. Mass Spectrom. Rev. 2015, 34, 248–263. [Google Scholar]
- Panda, S.K.; Andersson, J.T.; Schrader, W. Mass-spectrometric analysis of complex volatile and nonvolatile crude oil components: a challenge. Anal. Bioanal. Chem. 2007, 389, 1329–1339. [Google Scholar] [CrossRef] [PubMed]
- Marshall, A.G.; Hendrickson, C.L.; Jackson, G.S. Fourier transform ion cyclotron resonance mass spectrometry: A primer. Mass Spectrom. Rev. 1998, 17, 1–35. [Google Scholar] [CrossRef]
- Makarov, A.; Denisov, E.; Kholomeev, A.; Baischun, W.; Lange, O.; Strupat, K.; Horning, S. Performance evaluation of a hybrid linear ion trap/orbitrap mass spectrometer. Anal. Chem. 2006, 78, 2113–2120. [Google Scholar] [CrossRef] [PubMed]
- Makarov, A.; Denisov, E.; Lange, O. Performance evaluation of a high-field orbitrap mass analyzer. J. Am. Soc. Mass. Spectrom. 2009, 20, 1391–1396. [Google Scholar] [CrossRef] [PubMed]
- Denisov, E.; Damoc, E.; Lange, O.; Makarov, A. Orbitrap mass spectrometry with resolving powers above 1,000,000. Int. J. Mass spectrom. 2012, 325, 80–85. [Google Scholar] [CrossRef]
- Zhurov, K.O.; Kozhinov, A.N.; Tsybin, Y.O. Evaluation of high-field orbitrap fourier transform mass spectrometer for petroleomics. Energ. Fuel 2013, 27, 2974–2983. [Google Scholar] [CrossRef]
- Vetere, A.; Schrader, W. 1-and 2-Photon ionization for online FAIMS-FTMS coupling allows new insights into the constitution of crude oils. Anal. Chem. 2015, 87, 8874–8879. [Google Scholar] [CrossRef] [PubMed]
- Fenn, J.B.; Mann, M.; Meng, C.K.; Wong, S.F.; Whitehouse, C.M. Electrospray ionization-principles and practice. Mass Spectrom. Rev. 1990, 9, 37–70. [Google Scholar] [CrossRef]
- Qian, K.; Rodgers, R.P.; Hendrickson, C.L.; Emmett, M.R.; Marshall, A.G. Reading chemical fine print: Resolution and identification of 3000 nitrogen-containing aromatic compounds from a single electrospray ionization Fourier transform ion cyclotron resonance mass spectrum of heavy petroleum crude oil. Energy. Fuels 2001, 15, 492–498. [Google Scholar] [CrossRef]
- Schrader, W.; Xuan, Y.; Gaspar, A. Studying ultra-complex crude oil mixtures by using high-field asymmetric waveform ion mobility spectrometry (FAIMS) coupled to an electrospray ionisation-LTQ-Orbitrap mass spectrometer. Eur. J. Mass Spectrom. 2014, 20, 43–49. [Google Scholar] [CrossRef]
- Qian, K.N.; Robbins, W.K.; Hughey, C.A.; Cooper, H.J.; Rodgers, R.P.; Marshall, A.G. Resolution and identification of elemental compositions for more than 3000 crude acids in heavy petroleum by negative-ion microelectrospray high-field Fourier transform ion cyclotron resonance mass spectrometry. Energ. Fuel 2001, 15, 1505–1511. [Google Scholar] [CrossRef]
- Guricza, L.M.; Schrader, W. Electrospray ionization for determination of non-polar polyaromatic hydrocarbons and polyaromatic heterocycles in heavy crude oil asphaltenes. J. Mass Spectrom. 2015, 50, 549–557. [Google Scholar] [CrossRef] [PubMed]
- Muller, H.; Andersson, J.T.; Schrader, W. Characterization of high-molecular-weight sulfur-containing aromatics in vacuum residues using Fourier transform ion cyclotron resonance mass spectrometry. Anal. Chem. 2005, 77, 2536–2543. [Google Scholar] [CrossRef] [PubMed]
- Purcell, J.M.; Juyal, P.; Kim, D.G.; Rodgers, R.P.; Hendrickson, C.L.; Marshall, A.G. Sulfur speciation in petroleum: Atmospheric pressure photoionization or chemical derivatization and electrospray ionization fourier transform ion cyclotron resonance mass spectrometry. Energ. Fuel 2007, 21, 2869–2874. [Google Scholar] [CrossRef]
- Rudzinski, W.E.; Zhou, K.; Luo, X.M. Mass spectrometric characterization of organosulfur compounds using palladium(II) as a sensitivity-enhancing reagent. Energ Fuel 2004, 18, 16–21. [Google Scholar] [CrossRef]
- Roussis, S.G.; Proulx, R. Molecular weight distributions of heavy aromatic petroleum fractions by Ag+ electrospray ionization mass spectrometry. Anal. Chem. 2002, 74, 1408–1414. [Google Scholar] [CrossRef] [PubMed]
- Panda, S.K.; Schrader, W.; Andersson, J.T. β-cyclodextrin as a stationary phase for the group separation of polycyclic aromatic compounds in normal-phase liquid chromatography. J. Chromatogr. 2006, 1122, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Fox, B.S.; Beyer, M.K.; Bondybey, V.E. Coordination chemistry of silver cations. J. Am. Chem. Soc. 2002, 124, 13613–13623. [Google Scholar] [CrossRef] [PubMed]
- Abul Haj, M.; Aakeroy, C.B.; Desper, J. Silver(I) coordination chemistry: From 1-D chains to molecular rectangles. New J. Chem. 2013, 37, 204–211. [Google Scholar] [CrossRef]
- Kharrat, A.M.; Zacharia, J.; Cherian, V.J.; Anyatonwu, A. Issues with comparing SARA methodologies. Energ. Fuel 2007, 21, 3618–3621. [Google Scholar] [CrossRef]
- Panda, S.K.; Schrader, W.; Andersson, J.T. Fourier transform ion cyclotron resonance mass spectrometry in the speciation of high molecular weight sulfur heterocycles in vacuum gas oils of different boiling ranges. Anal. Bioanal. Chem. 2008, 392, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Swiegers, G.F.; Malefetse, T.J. New self-assembled structural motifs in coordination chemistry. Chem. Rev. 2000, 100, 3483–3537. [Google Scholar] [CrossRef] [PubMed]
- Lobodin, V.V.; Juyal, P.; McKenna, A.M.; Rodgers, R.P.; Marshall, A.G. Silver cationization for rapid speciation of sulfur-containing species in crude oils by positive electrospray ionization fourier transform ion cyclotron resonance mass spectrometry. Energ. Fuels 2014, 28, 447–452. [Google Scholar] [CrossRef]
- Hutchinson, D.J.; Cameron, S.A.; Hanton, L.R.; Moratti, S.C. Sensitivity of Silver(I) complexes of a pyrimidine-hydrazone ligand to solvent, counteranion, and metal-to-ligand ratio changes. Inorg. Chem. 2012, 51, 5070–5081. [Google Scholar] [CrossRef] [PubMed]
- Acheson, R.M.; Harrison, D.R. Synthesis, spectra, and reactions of some S-alkylthiophenium salts. J. Chem. Soc. C 1970, 13, 1764–1784. [Google Scholar] [CrossRef]
- Ansell, G.B. Crystal-structure of a 1:1 complex between silver-iodide and morpholine. J. Chem. Soc. 1976, 1, 104–106. [Google Scholar] [CrossRef]
- Schrader, W.; Panda, S.K.; Brockmann, K.J.; Benter, T. Characterization of non-polar aromatic hydrocarbons in crude oil using atmospheric pressure laser ionization and Fourier transform ion cyclotron resonance mass spectrometry (APLI FT-ICR MS). Analyst 2008, 133, 867–869. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.P.; Qian, K.N.; Nefliu, M.; Cooks, R.G. Ambient analysis of saturated hydrocarbons using discharge-induced oxidation in desorption electrospray ionization. J. Am. Soc. Mass. Spectrom. 2010, 21, 261–267. [Google Scholar] [CrossRef] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.; Schrader, W. Selective Analysis of Sulfur-Containing Species in a Heavy Crude Oil by Deuterium Labeling Reactions and Ultrahigh Resolution Mass Spectrometry. Int. J. Mol. Sci. 2015, 16, 30133-30143. https://doi.org/10.3390/ijms161226205
Wang X, Schrader W. Selective Analysis of Sulfur-Containing Species in a Heavy Crude Oil by Deuterium Labeling Reactions and Ultrahigh Resolution Mass Spectrometry. International Journal of Molecular Sciences. 2015; 16(12):30133-30143. https://doi.org/10.3390/ijms161226205
Chicago/Turabian StyleWang, Xuxiao, and Wolfgang Schrader. 2015. "Selective Analysis of Sulfur-Containing Species in a Heavy Crude Oil by Deuterium Labeling Reactions and Ultrahigh Resolution Mass Spectrometry" International Journal of Molecular Sciences 16, no. 12: 30133-30143. https://doi.org/10.3390/ijms161226205
APA StyleWang, X., & Schrader, W. (2015). Selective Analysis of Sulfur-Containing Species in a Heavy Crude Oil by Deuterium Labeling Reactions and Ultrahigh Resolution Mass Spectrometry. International Journal of Molecular Sciences, 16(12), 30133-30143. https://doi.org/10.3390/ijms161226205




