Advances in Intracranial Pressure Monitoring and Its Significance in Managing Traumatic Brain Injury
Abstract
:1. Introduction
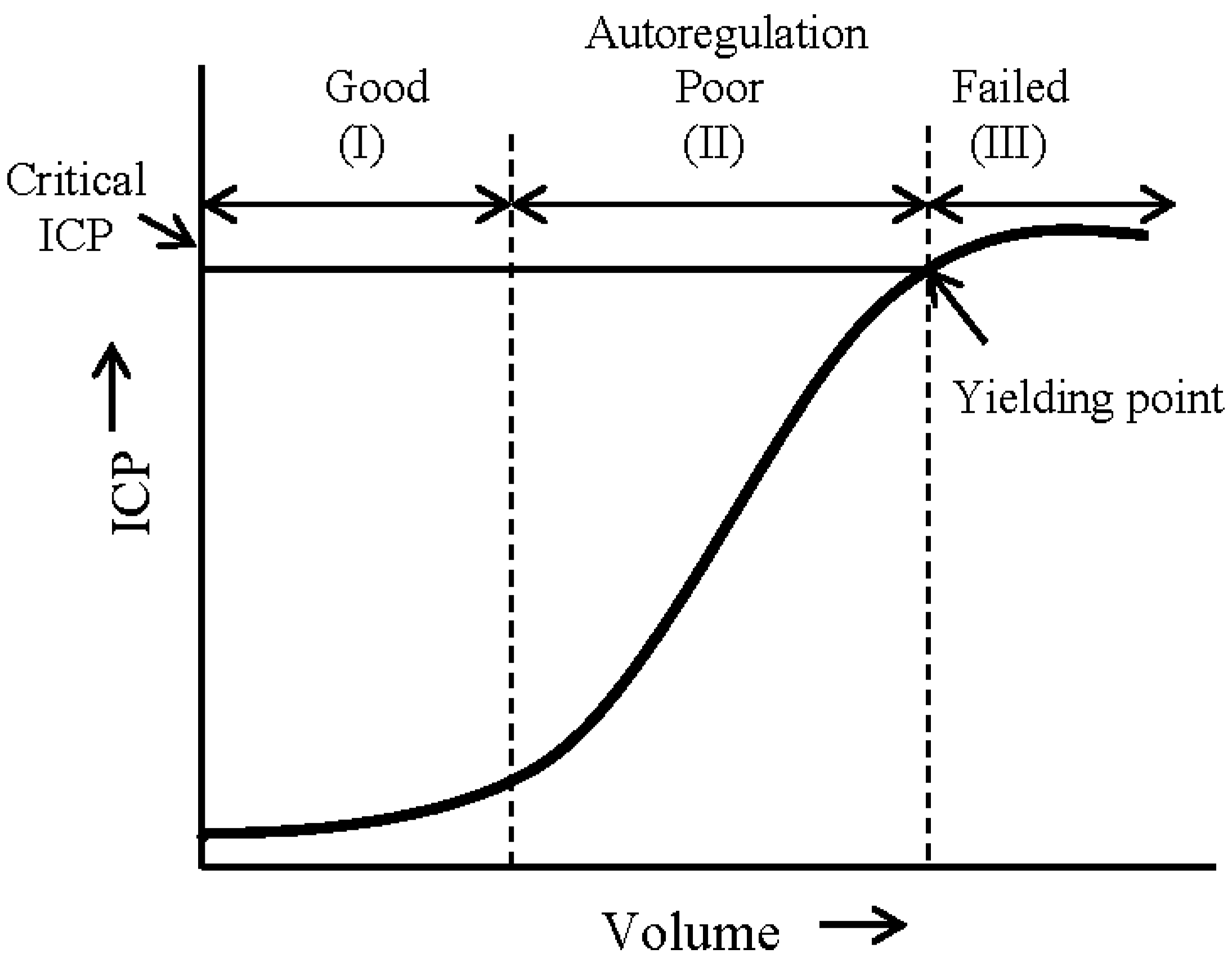
1.1. Significance of ICP Monitoring
1.2. ICP Technology
2. Key Factors to Be Considered in ICP Monitoring Technology
2.1. Device Accuracy
2.2. Transducer Position for ICP Measurements
2.3. Pressure Gradients
2.4. Complications
3. Methods of ICP Monitoring
3.1. Invasive
3.1.1. Fluid-Filled Systems
3.1.2. Implantable Transducer Catheter Systems
3.1.3. Telemetric Systems
3.2. Non-Invasive
3.2.1. Impedance Mismatch
3.2.2. Tympanic Membrane Displacement (TMD)
3.2.3. Transcranial Doppler (TCD)
3.2.4. Near Infrared Spectroscopy (NIRS)
3.2.5. Optic Nerve Sheath Diameter (ONSD)
3.2.6. Fontanometery
3.2.7. Pulsed Phase Lock Loop (PPLL) Technique
4. ICP Waveform Analysis
4.1. Waveform Analysis
4.2. Histogram Analysis
4.3. Frequency Analysis
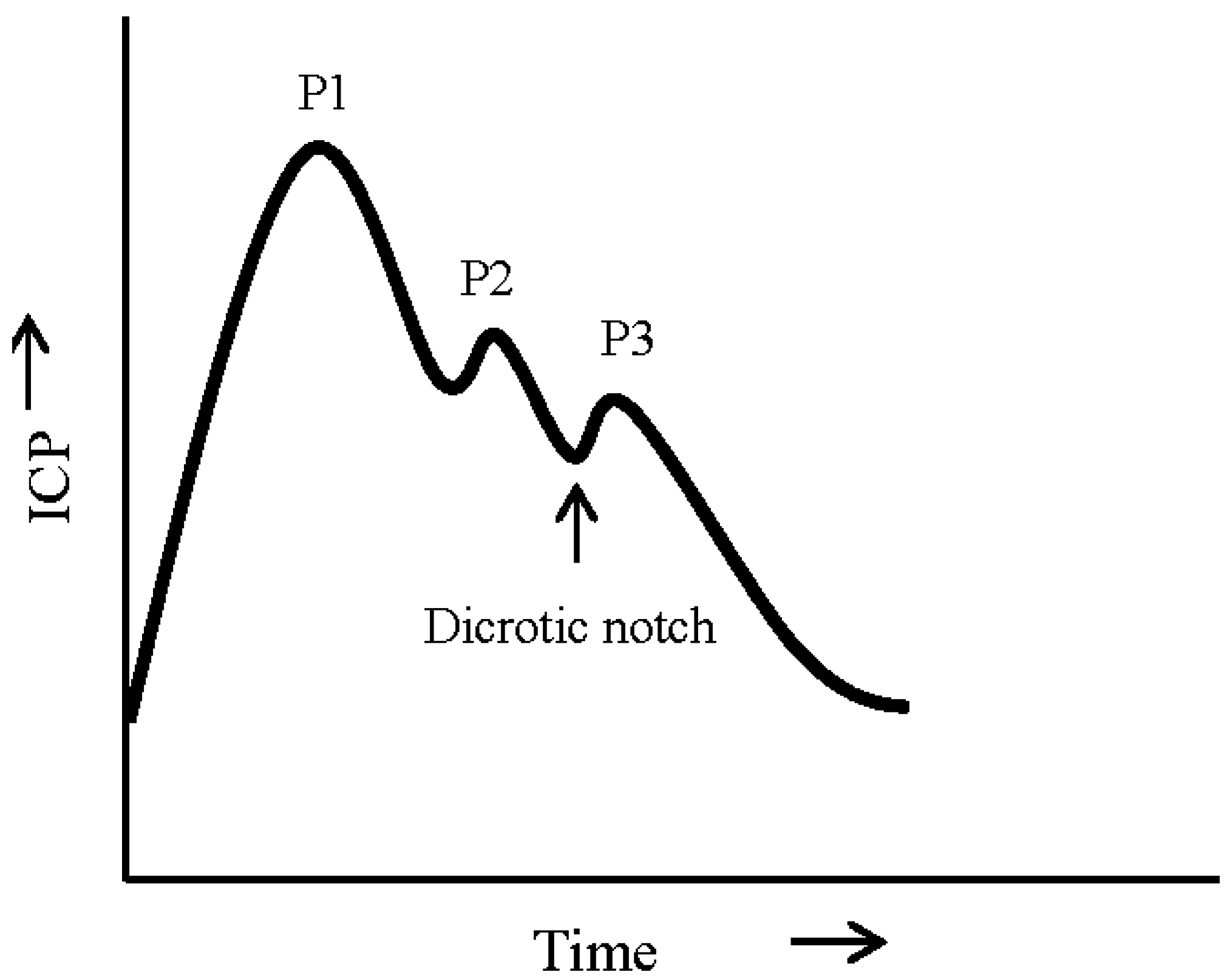
5. Role of ICP Monitoring in Traumatic Brain Injury
Advanced ICP Analysis for TBI
6. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Baral, B.; Agrawal, A.; Cincu, R. Intracranial pressure monitoring: Concepts in evaluation and measurement. Pak. J. Med. Sci. 2007, 23, 798–804. [Google Scholar]
- Oestern, H.J.; Trentz, O.; Uranues, S. Head, Thoracic, Abdominal, and Vascular Injuries: Trauma Surgery I; Springer: Berlin, Germany, 2011. [Google Scholar]
- Smith, M. Monitoring intracranial pressure in traumatic brain injury. Anesth. Analg. 2008, 106, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Doyle, D.J.; Mark, P.W. Analysis of intracranial pressure. J. Clin. Monit. 1992, 8, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Campbell, W.W.; DeJong, R.N. Dejong's the Neurologic Examination; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2005. [Google Scholar]
- Carlson, K.K. Aacn Advanced Critical Care Nursing; Saunders: Philadelphia, PA, USA, 2008. [Google Scholar]
- Wiegand, D.J.L.-M. Aacn Procedure Manual for Critical Care; Elsevier Health Sciences: Philadelphia, PA, USA, 2013. [Google Scholar]
- Brain Trauma Foundation; American Association of Neurological Surgeons; Congress of Neurological Surgeons. Guidelines for the management of severe traumatic brain injury. J. Neurotrauma 2007, 24, S1–S106. [Google Scholar]
- Muizelaar, J.P.; Ward, J.D.; Marmarou, A.; Newlon, P.G.; Wachi, A. Cerebral blood flow and metabolism in severely head-injured children. Part 2: Autoregulation. J. Neurosurg. 1989, 71, 72–76. [Google Scholar] [CrossRef] [PubMed]
- Zweifel, C.; Lavinio, A.; Steiner, L.A.; Radolovich, D.; Smielewski, P.; Timofeev, I.; Hiler, M.; Balestreri, M.; Kirkpatrick, P.J.; Pickard, J.D.; et al. Continuous monitoring of cerebrovascular pressure reactivity in patients with head injury. Neurosurg. Focus 2008, 25, E2. [Google Scholar] [CrossRef] [PubMed]
- Czosnyka, M.; Pickard, J.D. Monitoring and interpretation of intracranial pressure. J. Neurol. Neurosurg. Psychiatry 2004, 75, 813–821. [Google Scholar] [CrossRef] [PubMed]
- Piper, I.; Spiegelberg, A.; Whittle, I.; Signorini, D.; Mascia, L. A comparative study of the spiegelberg compliance device with a manual volume-injection method: A clinical evaluation in patients with hydrocephalus. Br. J. Neurosurg. 1999, 13, 581–586. [Google Scholar] [PubMed]
- Gowers, W.R.; Taylor, J. A Manual of Diseases of the Nervous System; J&A Churchill: London, UK, 1892. [Google Scholar]
- Geocadin, R.G.; Varelas, P.N.; Rigamonti, D.; Williams, M.A. Continuous intracranial pressure monitoring via the shunt reservoir to assess suspected shunt malfunction in adults with hydrocephalus. Neurosurg. Focus 2007, 22, E10. [Google Scholar] [CrossRef] [PubMed]
- Bohn, D.J.; Biggar, W.D.; Smith, C.R.; Conn, A.W.; Barker, G.A. Influence of hypothermia, barbiturate therapy, and intracranial pressure monitoring on morbidity and mortality after near-drowning. Crit. Care Med. 1986, 14, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Chi, C.S.; Law, K.L.; Wong, T.T.; Su, G.Y.; Lin, N. Continuous monitoring of intracranial pressure in reye’s syndrome—5 Years experience. Acta Paediatr. Jpn. Overseas Ed. 1990, 32, 426–434. [Google Scholar] [CrossRef]
- Hamani, C.; Zanetti, M.V.; Pinto, F.C.; Andrade, A.F.; Ciquini, O., Jr.; Marino, R., Jr. Intraventricular pressure monitoring in patients with thalamic and ganglionic hemorrhages. Arq. Neuro-Psiquiatr. 2003, 61, 376–380. [Google Scholar] [CrossRef]
- Nabeta, H.W.; Bahr, N.C.; Rhein, J.; Fossland, N.; Kiragga, A.N.; Meya, D.B.; Dunlop, S.J.; Boulware, D.R. Accuracy of noninvasive intraocular pressure or optic nerve sheath diameter measurements for predicting elevated intracranial pressure in cryptococcal meningitis. Open Forum Infect. Dis. 2014, 1. [Google Scholar] [CrossRef] [PubMed]
- Petersen, K.D.; Landsfeldt, U.; Cold, G.E.; Petersen, C.B.; Mau, S.; Hauerberg, J.; Holst, P.; Olsen, K.S. Intracranial pressure and cerebral hemodynamic in patients with cerebral tumors: A randomized prospective study of patients subjected to craniotomy in propofol-fentanyl, isoflurane-fentanyl, or sevoflurane-fentanyl anesthesia. Anesthesiology 2003, 98, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Belli, A.; Sen, J.; Petzold, A.; Russo, S.; Kitchen, N.; Smith, M. Metabolic failure precedes intracranial pressure rises in traumatic brain injury: A microdialysis study. Acta Neurochir. 2008, 150, 461–469. [Google Scholar] [CrossRef] [PubMed]
- Marin, J.R.; Weaver, M.D.; Yealy, D.M.; Mannix, R.C. Trends in visits for traumatic brain injury to emergency departments in the united states. JAMA 2014, 311, 1917–1919. [Google Scholar] [CrossRef] [PubMed]
- Ling, G.S.; Ecklund, J.M. Traumatic brain injury in modern war. Curr. Opin. Anaesthesiol. 2011, 24, 124–130. [Google Scholar] [CrossRef] [PubMed]
- Warden, D. Military tbi during the iraq and afghanistan wars. J. Head Trauma Rehabil. 2006, 21, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Guillaume, J.; Janny, P. Continuous intracranial manometry; importance of the method and first results. Rev. Neurol. 1951, 84, 131–142. [Google Scholar] [PubMed]
- Lundberg, N. Continuous recording and control of ventricular fluid pressure in neurosurgical practice. Acta Psychiatr. Scand. Suppl. 1960, 36, 1–193. [Google Scholar] [CrossRef] [PubMed]
- Bering, E.A., Jr. Choroid plexus and arterial pulsation of cerebrospinal fluid; demonstration of the choroid plexuses as a cerebrospinal fluid pump. AMA Arch. Neurol. Psychiatry 1955, 73, 165–172. [Google Scholar] [CrossRef] [PubMed]
- Goldensohn, E.S.; Whitehead, R.W.; Parry, T.M.; Spencer, J.N.; Grover, R.F.; Draper, W.B. Studies on diffusion respiration. Ix. Effect of diffusion respiration and high concentrations of CO2 on cerebrospinal fluid pressure of anesthetized dogs. Am. J. Physiol. 1951, 165, 334–340. [Google Scholar] [PubMed]
- Fernandes, H.M.; Bingham, K.; Chambers, I.R.; Mendelow, A.D. Clinical evaluation of the codman microsensor intracranial pressure monitoring system. Acta Neurochir. Suppl. 1998, 71, 44–46. [Google Scholar] [PubMed]
- Gray, W.P.; Palmer, J.D.; Gill, J.; Gardner, M.; Iannotti, F. A clinical study of parenchymal and subdural miniature strain-gauge transducers for monitoring intracranial pressure. Neurosurgery 1996, 39, 927–931. [Google Scholar] [PubMed]
- Hollingsworth-Fridlund, P.; Vos, H.; Daily, E.K. Use of fiber-optic pressure transducer for intracranial pressure measurements: A preliminary report. Heart Lung 1988, 17, 111–120. [Google Scholar] [PubMed]
- Murtha, L.; McLeod, D.; Spratt, N. Epidural intracranial pressure measurement in rats using a fiber-optic pressure transducer. J. Vis. Exp. 2012, 62, 3689. [Google Scholar] [CrossRef] [PubMed]
- Chibbaro, S.; Marsella, M.; Romano, A.; Ippolito, S.; Benericetti, E. Combined internal uncusectomy and decompressive craniectomy for the treatment of severe closed head injury: Experience with 80 cases. J. Neurosurg. 2008, 108, 74–79. [Google Scholar] [CrossRef] [PubMed]
- Gelabert-Gonzalez, M.; Ginesta-Galan, V.; Sernamito-Garcia, R.; Allut, A.G.; Bandin-Dieguez, J.; Rumbo, R.M. The camino intracranial pressure device in clinical practice. Assessment in a 1000 cases. Acta Neurochir. 2006, 148, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Sadaka, F.; Kasal, J.; Lakshmanan, R.; Palagiri, A. Placement of intracranial pressure monitors by neurointensivists: Case series and a systematic review. Brain Inj. 2013, 27, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Chambers, K.R.; Kane, P.J.; Choksey, M.S.; Mendelow, A.D. An evaluation of the camino ventricular bolt system in clinical practice. Neurosurgery 1993, 33, 866–868. [Google Scholar] [CrossRef] [PubMed]
- Munch, E.; Weigel, R.; Schmiedek, P.; Schurer, L. The camino intracranial pressure device in clinical practice: Reliability, handling characteristics and complications. Acta Neurochir. 1998, 140, 1113–1119. [Google Scholar] [PubMed]
- Schurer, L.; Munch, E.; Piepgras, A.; Weigel, R.; Schilling, L.; Schmiedek, P. Assessment of the camino intracranial pressure device in clinical practice. Acta Neurochir. Suppl. 1997, 70, 296–298. [Google Scholar] [PubMed]
- Weinstabl, C.; Richling, B.; Plainer, B.; Czech, T.; Spiss, C.K. Comparative analysis between epidural (gaeltec) and subdural (camino) intracranial pressure probes. J. Clin. Monit. 1992, 8, 116–120. [Google Scholar] [CrossRef] [PubMed]
- Becker, D.P.; Miller, J.D.; Ward, J.D.; Greenberg, R.P.; Young, H.F.; Sakalas, R. The outcome from severe head injury with early diagnosis and intensive management. J. Neurosurg. 1977, 47, 491–502. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, R.P.; Mayer, D.J.; Becker, D.P.; Miller, J.D. Evaluation of brain function in severe human head trauma with multimodality evoked potentials. Part 1: Evoked brain-injury potentials, methods, and analysis. J. Neurosurg. 1977, 47, 150–162. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, R.P.; Sakalas, R.; Miller, J.D.; Becker, D.P. Multimodality evoked potentials and cbf in patients with severe head injury. Acta Neurol. Scand. Suppl. 1977, 64, 498–499. [Google Scholar] [PubMed]
- Marik, P.; Chen, K.; Varon, J.; Fromm, R., Jr.; Sternbach, G.L. Management of increased intracranial pressure: A review for clinicians. J. Emerg. Med. 1999, 17, 711–719. [Google Scholar] [CrossRef]
- Miller, J.D.; Becker, D.P.; Ward, J.D.; Sullivan, H.G.; Adams, W.E.; Rosner, M.J. Significance of intracranial hypertension in severe head injury. J. Neurosurg. 1977, 47, 503–516. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.D.; Butterworth, J.F.; Gudeman, S.K.; Faulkner, J.E.; Choi, S.C.; Selhorst, J.B.; Harbison, J.W.; Lutz, H.A.; Young, H.F.; Becker, D.P. Further experience in the management of severe head injury. J. Neurosurg. 1981, 54, 289–299. [Google Scholar] [CrossRef] [PubMed]
- Association for the Advancement of Medical Instrumentation Imperial County Planning & Development Services; American National Standards Institute. Intracranial Pressure Monitoring Devices; The Association: Arlington, VA, USA, 1993. [Google Scholar]
- Gopinath, S.P.; Robertson, C.S.; Contant, C.F.; Narayan, R.K.; Grossman, R.G. Clinical evaluation of a miniature strain-gauge transducer for monitoring intracranial pressure. Neurosurgery 1995, 36, 1137–1140. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, G.; Zaccone, C.; Cardia, E.; Tomasello, F. Intracranial pressure monitoring in children: Comparison of external ventricular device with the fiberoptic system. Child Nerv. Syst. 1993, 9, 470–473. [Google Scholar] [CrossRef]
- Kosteljanetz, M.; Borgesen, S.E.; Stjernholm, P.; Christensen, L.; Osgaard, O.; Gjerris, F.; Rosenorn, J. Clinical evaluation of a simple epidural pressure sensor. Acta Neurochir. 1986, 83, 108–111. [Google Scholar] [CrossRef] [PubMed]
- Mendelow, A.D.; Rowan, J.O.; Murray, L.; Kerr, A.E. A clinical comparison of subdural screw pressure measurements with ventricular pressure. J. Neurosurg. 1983, 58, 45–50. [Google Scholar] [CrossRef] [PubMed]
- Mollman, H.D.; Rockswold, G.L.; Ford, S.E. A clinical comparison of subarachnoid catheters to ventriculostomy and subarachnoid bolts: A prospective study. J. Neurosurg. 1988, 68, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Piek, J.; Kosub, B.; Kuch, F.; Bock, W.J. A practical technique for continuous monitoring of cerebral tissue pressure in neurosurgical patients. Preliminary results. Acta Neurochir. 1987, 87, 144–149. [Google Scholar] [CrossRef] [PubMed]
- Piek, J.; Bock, W.J. Continuous monitoring of cerebral tissue pressure in neurosurgical practice—Experiences with 100 patients. Intensive Care Med. 1990, 16, 184–188. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, S.; Bowman, R.; Callahan, J.; Wolfla, C. The fiberoptic intraparenchymal cerebral pressure monitor in 244 patients. Surg. Neurol. 1996, 45, 278–282. [Google Scholar] [CrossRef]
- Chambers, I.R.; Mendelow, A.D.; Sinar, E.J.; Modha, P. A clinical evaluation of the camino subdural screw and ventricular monitoring kits. Neurosurgery 1990, 26, 421–423. [Google Scholar] [CrossRef] [PubMed]
- Gambardella, G.; D’Avella, D.; Tomasello, F. Monitoring of brain tissue pressure with a fiberoptic device. Neurosurgery 1992, 31, 918–922. [Google Scholar] [CrossRef] [PubMed]
- Schickner, D.J.; Young, R.F. Intracranial pressure monitoring: Fiberoptic monitor compared with the ventricular catheter. Surg. Neurol. 1992, 37, 251–254. [Google Scholar] [CrossRef]
- Dearden, N.M.; McDowall, D.G.; Gibson, R.M. Assessment of leeds device for monitoring intracranial pressure. J. Neurosurg. 1984, 60, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Weaver, D.D.; Winn, H.R.; Jane, J.A. Differential intracranial pressure in patients with unilateral mass lesions. J. Neurosurg. 1982, 56, 660–665. [Google Scholar] [CrossRef] [PubMed]
- Barlow, P.; Mendelow, A.D.; Lawrence, A.E.; Barlow, M.; Rowan, J.O. Clinical evaluation of two methods of subdural pressure monitoring. J. Neurosurg. 1985, 63, 578–582. [Google Scholar] [CrossRef] [PubMed]
- Czech, T.; Korn, A.; Reinprecht, A.; Schramm, W.; Kimla, T.; Spiss, C.K. Clinical evaluation of a new epidural pressure monitor. Acta Neurochir. 1993, 125, 169–172. [Google Scholar] [CrossRef] [PubMed]
- Powell, M.P.; Crockard, H.A. Behavior of an extradural pressure monitor in clinical use. Comparison of extradural with intraventricular pressure in patients with acute and chronically raised intracranial pressure. J. Neurosurg. 1985, 63, 745–749. [Google Scholar] [CrossRef] [PubMed]
- Schwarz, N.; Matuschka, H.; Meznik, A. The spiegelberg device for epidural registration of the icp. Unfallchirurg 1992, 95, 113–117. [Google Scholar] [PubMed]
- Mindermann, T.; Gratzl, O. Interhemispheric pressure gradients in severe head trauma in humans. Acta Neurochir. Suppl. 1998, 71, 56–58. [Google Scholar] [PubMed]
- Sahuquillo, J.; Poca, M.A.; Arribas, M.; Garnacho, A.; Rubio, E. Interhemispheric supratentorial intracranial pressure gradients in head-injured patients: Are they clinically important? J. Neurosurg. 1999, 90, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Brean, A.; Eide, P.K.; Stubhaug, A. Comparison of intracranial pressure measured simultaneously within the brain parenchyma and cerebral ventricles. J. Clin. Monit. Comput. 2006, 20, 411–414. [Google Scholar] [CrossRef] [PubMed]
- Stephensen, H.; Tisell, M.; Wikkelso, C. There is no transmantle pressure gradient in communicating or noncommunicating hydrocephalus. Neurosurgery 2002, 50, 763–771. [Google Scholar] [CrossRef] [PubMed]
- Raboel, P.H.; Bartek, J., Jr.; Andresen, M.; Bellander, B.M.; Romner, B. Intracranial pressure monitoring: Invasive versus non-invasive methods-a review. Crit. Care Res. Pract. 2012, 2012, 950393. [Google Scholar] [CrossRef] [PubMed]
- Eide, P.K.; Brean, A. Lumbar cerebrospinal fluid pressure waves versus intracranial pressure waves in idiopathic normal pressure hydrocephalus. Br. J. Neurosurg. 2006, 20, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, F.N.; Jha, A.N. Simultaneous lumbar and intraventricular manometry to evaluate the role and safety of lumbar puncture in raised intracranial pressure following subarachnoid haemorrhage. Br. J. Neurosurg. 1996, 10, 585–587. [Google Scholar] [CrossRef] [PubMed]
- Lenfeldt, N.; Koskinen, L.O.; Bergenheim, A.T.; Malm, J.; Eklund, A. Csf pressure assessed by lumbar puncture agrees with intracranial pressure. Neurology 2007, 68, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, A.; Gupta, A.K. Neuromonitoring in the intensive care unit. I. Intracranial pressure and cerebral blood flow monitoring. Intensive Care Med. 2007, 33, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Zhong, J.; Dujovny, M.; Park, H.K.; Perez, E.; Perlin, A.R.; Diaz, F.G. Advances in icp monitoring techniques. Neurol. Res. 2003, 25, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Ghajar, J. Intracranial pressure monitoring techniques. New Horiz. 1995, 3, 395–399. [Google Scholar] [PubMed]
- Ravi, R.; Morgan, R.J. Intracranial pressure monitoring. Curr. Anaesth. Crit. Care 2013, 14, 229–235. [Google Scholar] [CrossRef]
- Czosnyka, M.; Czosnyka, Z.; Pickard, J.D. Laboratory testing of the spiegelberg brain pressure monitor: A technical report. J. Neurol. Neurosurg. Psychiatry 1997, 63, 732–735. [Google Scholar] [CrossRef] [PubMed]
- Yau, Y.H.; Piper, I.R.; Contant, C.; Dunn, L.; Whittle, I.R. Assessment of different data representations and averaging methods on the spiegelberg compliance device. Acta Neurochir. Suppl. 2005, 95, 289–292. [Google Scholar] [PubMed]
- Yau, Y.H.; Piper, I.R.; Contant, C.F.; Dunn, L.T.; Whittle, I.R. Clinical experience in the use of the spiegelberg automated compliance device in the assessment of patients with hydrocephalus. Acta Neurochir. Suppl. 2002, 81, 171–172. [Google Scholar] [PubMed]
- Kawoos, U.; Meng, X.; Huang, S.M.; Rosen, A.; McCarron, R.M.; Chavko, M. Telemetric intracranial pressure monitoring in blast-induced traumatic brain injury. IEEE Trans. Bio-Med. Eng. 2014, 61, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Kawoos, U.; Xu, M.; Tofighi, M.R.; Rosen, A. Too much pressure: Wireless intracranial pressure monitoring and its application in traumatic brain injuries. IEEE Microw. Mag. 2015, 16, 39–53. [Google Scholar] [CrossRef]
- Bruder, N.; N’Zoghe, P.; Graziani, N.; Pelissier, D.; Grisoli, F.; Francois, G. A comparison of extradural and intraparenchymatous intracranial pressures in head injured patients. Intensive Care Med. 1995, 21, 850–852. [Google Scholar] [CrossRef] [PubMed]
- Kawoos, U.; Tofighi, M.R.; Warty, R.; Kralick, F.A.; Rosen, A. In-vitro and in vivo trans-scalp evaluation of an intracranial pressure implant at 2.4 ghz. IEEE Trans. Microw. Theory Tech. 2008, 56, 2356–2365. [Google Scholar] [CrossRef]
- Leidorf, A.; Mader, M.M.; Hecker, A.; Heimann, A.; Alessandri, B.; Mayr, P.; Kempski, O.; Wobker, G. Description of the response of a new multi-parametric brain sensor to physiological and pathophysiological challenges in the cortex of juvenile pigs. Turk. Neurosurg. 2014, 24, 913–922. [Google Scholar] [CrossRef] [PubMed]
- Kiefer, M.; Antes, S.; Leonhardt, S.; Schmitt, M.; Orakcioglu, B.; Sakowitz, O.W.; Eymann, R. Telemetric icp measurement with the first ce-approved device: Data from animal experiments and initial clinical experiences. Acta Neurochir. Suppl. 2012, 114, 111–116. [Google Scholar] [PubMed]
- Kroin, J.S.; McCarthy, R.J.; Stylos, L.; Miesel, K.; Ivankovich, A.D.; Penn, R.D. Long-term testing of an intracranial pressure monitoring device. J. Neurosurg. 2000, 93, 852–858. [Google Scholar] [CrossRef] [PubMed]
- Silasi, G.; MacLellan, C.L.; Colbourne, F. Use of telemetry blood pressure transmitters to measure intracranial pressure (icp) in freely moving rats. Curr. Neurovasc. Res. 2009, 6, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Zervas, N.T.; Cosman, E.R.; Cosman, B.J. A pressure-balanced radio-telemetry system for the measurement of intracranial pressure. A preliminary design report. J. Neurosurg. 1977, 47, 899–911. [Google Scholar] [CrossRef] [PubMed]
- Swoboda, M.; Hochman, M.G.; Fritz, F.J. Non-Invasive Intracranial Pressure Sensor. Google Patents US20130289422 A1, 31 October 2010. [Google Scholar]
- Reid, A.; Marchbanks, R.J.; Burge, D.M.; Martin, A.M.; Bateman, D.E.; Pickard, J.D.; Brightwell, A.P. The relationship between intracranial pressure and tympanic membrane displacement. Br. J. Audiol. 1990, 24, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Gwer, S.; Sheward, V.; Birch, A.; Marchbanks, R.; Idro, R.; Newton, C.R.; Kirkham, F.J.; Lin, J.P.; Lim, M. The tympanic membrane displacement analyser for monitoring intracranial pressure in children. Child Nerv. Syst. 2013, 29, 927–933. [Google Scholar] [CrossRef] [PubMed]
- Samuel, M.; Burge, D.M.; Marchbanks, R.J. Quantitative assessment of intracranial pressure by the tympanic membrane displacement audiometric technique in children with shunted hydrocephalus. Eur. J. Pediatr. Surg. 1998, 8, 200–207. [Google Scholar] [CrossRef] [PubMed]
- Samuel, M.; Burge, D.M.; Marchbanks, R.J. Tympanic membrane displacement testing in regular assessment of intracranial pressure in eight children with shunted hydrocephalus. J. Neurosurg. 1998, 88, 983–995. [Google Scholar] [CrossRef] [PubMed]
- Wunderlich, K.; Brinker, T.; Ernst, A. The correlation of tympanic membrane displacement measurements to direct intracranial pressure recordings in cat. In Intracranial and Intralabyrinthine Fluids: Basic Aspect and Clinical Applications; Ernst, A., Marchbanks, R., Samii, M., Eds.; Springer: Berlin, Germany, 1996. [Google Scholar]
- Behrens, A.; Lenfeldt, N.; Ambarki, K.; Malm, J.; Eklund, A.; Koskinen, L.O. Transcranial doppler pulsatility index: Not an accurate method to assess intracranial pressure. Neurosurgery 2010, 66, 1050–1057. [Google Scholar] [CrossRef] [PubMed]
- Bellner, J.; Romner, B.; Reinstrup, P.; Kristiansson, K.A.; Ryding, E.; Brandt, L. Transcranial doppler sonography pulsatility index (PI) reflects intracranial pressure (ICP). Surg. Neurol. 2004, 62, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Moreno, J.A.; Mesalles, E.; Gener, J.; Tomasa, A.; Ley, A.; Roca, J.; Fernandez-Llamazares, J. Evaluating the outcome of severe head injury with transcranial doppler ultrasonography. Neurosurg. Focus 2000, 8, e8. [Google Scholar] [CrossRef] [PubMed]
- Voulgaris, S.G.; Partheni, M.; Kaliora, H.; Haftouras, N.; Pessach, I.S.; Polyzoidis, K.S. Early cerebral monitoring using the transcranial doppler pulsatility index in patients with severe brain trauma. Med. Sci. Monit. 2005, 11, CR49–CR52. [Google Scholar] [PubMed]
- Weerakkody, R.A.; Czosnyka, M.; Zweifel, C.; Castellani, G.; Smielewski, P.; Brady, K.; Pickard, J.D.; Czosnyka, Z. Near infrared spectroscopy as possible non-invasive monitor of slow vasogenic icp waves. Acta Neurochir. Suppl. 2012, 114, 181–185. [Google Scholar] [PubMed]
- Weerakkody, R.A.; Czosnyka, M.; Zweifel, C.; Castellani, G.; Smielewski, P.; Keong, N.; Haubrich, C.; Pickard, J.; Czosnyka, Z. Slow vasogenic fluctuations of intracranial pressure and cerebral near infrared spectroscopy—An observational study. Acta Neurochir. 2010, 152, 1763–1769. [Google Scholar] [CrossRef] [PubMed]
- Diedler, J.; Zweifel, C.; Budohoski, K.P.; Kasprowicz, M.; Sorrentino, E.; Haubrich, C.; Brady, K.M.; Czosnyka, M.; Pickard, J.D.; Smielewski, P. The limitations of near-infrared spectroscopy to assess cerebrovascular reactivity: The role of slow frequency oscillations. Anesth. Analg. 2011, 113, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Tayal, V.S.; Neulander, M.; Norton, H.J.; Foster, T.; Saunders, T.; Blaivas, M. Emergency department sonographic measurement of optic nerve sheath diameter to detect findings of increased intracranial pressure in adult head injury patients. Ann. Emerg. Med. 2007, 49, 508–514. [Google Scholar] [CrossRef] [PubMed]
- Geeraerts, T.; Merceron, S.; Benhamou, D.; Vigue, B.; Duranteau, J. Non-invasive assessment of intracranial pressure using ocular sonography in neurocritical care patients. Intensive Care Med. 2008, 34, 2062–2067. [Google Scholar] [CrossRef] [PubMed]
- Geeraerts, T.; Launey, Y.; Martin, L.; Pottecher, J.; Vigue, B.; Duranteau, J.; Benhamou, D. Ultrasonography of the optic nerve sheath may be useful for detecting raised intracranial pressure after severe brain injury. Intensive Care Med. 2007, 33, 1704–1711. [Google Scholar] [CrossRef] [PubMed]
- Le, A.; Hoehn, M.E.; Smith, M.E.; Spentzas, T.; Schlappy, D.; Pershad, J. Bedside sonographic measurement of optic nerve sheath diameter as a predictor of increased intracranial pressure in children. Ann. Emerg. Med. 2009, 53, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Purin, V.R. Measurement of intracranial pressure in children without puncture (new method). Pediatriia 1964, 43, 82–85. [Google Scholar] [PubMed]
- Wealthall, S.R.; Smallwood, R. Methods of measuring intracranial pressure via the fontanelle without puncture. J. Neurol. Neurosurg. Psychiatry 1974, 37, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Rochefort, M.J.; Rolfe, P.; Wilkinson, A.R. New fontanometer for continuous estimation of intracranial pressure in the newborn. Arch. Dis. Child. 1987, 62, 152–155. [Google Scholar] [CrossRef] [PubMed]
- Pairaudeau, P.W.; Smith, S.L.; Hames, T.K.; Hall, M.A. Strain-gauge fontanometry—An advance in non-invasive intracranial pressure measurement. Clin. Phys. Physiol. Meas. 1990, 11, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Ueno, T.; Macias, B.R.; Hargens, A.R.; Yost, W.T. Pulsed Phase Lock Loop Technique to Measure Intracranial Pressure Non-Invasively. In Proceedings of the 2003 IEEE Symposium on Ultrasonics, Honolulu, HI, USA, 5–8 October 2003.
- Eide, P.K.; Bakken, A. The baseline pressure of intracranial pressure (icp) sensors can be altered by electrostatic discharges. Biomed. Eng. Online 2011, 10, 75. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, E.R.; Rowan, J.O.; Galbraith, S. Analysis of the cerebrospinal fluid pulse wave in intracranial pressure. J. Neurosurg. 1983, 59, 817–821. [Google Scholar] [CrossRef] [PubMed]
- Martin, G. Lundberg’s b waves as a feature of normal intracranial pressure. Surg. Neurol. 1978, 9, 347–348. [Google Scholar] [PubMed]
- Janny, P.; Fontaine, J.; Maille, M.; Colnet, G.; Hullo, G.; Janny, L. A module of statistical analysis of the intracranial pressure: The s.A.I.C.P. System. Neuro-Chirurgie 1977, 23, 187–194. [Google Scholar] [PubMed]
- Sharar, S.R. The ongoing and worldwide challenge of pediatric trauma. Int. J. Crit. Illn. Inj. Sci. 2012, 2, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Guiza, F.; Depreitere, B.; Piper, I.; Citerio, G.; Chambers, I.; Jones, P.A.; Lo, T.Y.; Enblad, P.; Nillson, P.; Feyen, B.; et al. Visualizing the pressure and time burden of intracranial hypertension in adult and paediatric traumatic brain injury. Intensive Care Med. 2015, 41, 1067–1076. [Google Scholar] [CrossRef] [PubMed]
- Badri, S.; Chen, J.; Barber, J.; Temkin, N.R.; Dikmen, S.S.; Chesnut, R.M.; Deem, S.; Yanez, N.D.; Treggiari, M.M. Mortality and long-term functional outcome associated with intracranial pressure after traumatic brain injury. Intensive Care Med. 2012, 38, 1800–1809. [Google Scholar] [CrossRef] [PubMed]
- Balestreri, M.; Czosnyka, M.; Steiner, L.A.; Schmidt, E.; Smielewski, P.; Matta, B.; Pickard, J.D. Intracranial hypertension: What additional information can be derived from icp waveform after head injury? Acta Neurochir. 2004, 146, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Balestreri, M.; Czosnyka, M.; Hutchinson, P.; Steiner, L.A.; Hiler, M.; Smielewski, P.; Pickard, J.D. Impact of intracranial pressure and cerebral perfusion pressure on severe disability and mortality after head injury. Neurocrit. Care 2006, 4, 8–13. [Google Scholar] [CrossRef]
- Feyen, B.F.; Sener, S.; Jorens, P.G.; Menovsky, T.; Maas, A.I. Neuromonitoring in traumatic brain injury. Minerva Anestesiol. 2012, 78, 949–958. [Google Scholar] [PubMed]
- Kochanek, P.M.; Carney, N.; Adelson, P.D.; Ashwal, S.; Bell, M.J.; Bratton, S.; Carson, S.; Chesnut, R.M.; Ghajar, J.; Goldstein, B.; et al. Guidelines for the acute medical management of severe traumatic brain injury in infants, children, and adolescents—Second edition. Pediatr. Crit. Care Med. 2012, 13, S1–S82. [Google Scholar] [CrossRef] [PubMed]
- Kosteljanetz, M. Acute head injury: Pressure-volume relations and cerebrospinal fluid dynamics. Neurosurgery 1986, 18, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, E.; Diedler, J.; Kasprowicz, M.; Budohoski, K.P.; Haubrich, C.; Smielewski, P.; Outtrim, J.G.; Manktelow, A.; Hutchinson, P.J.; Pickard, J.D.; et al. Critical thresholds for cerebrovascular reactivity after traumatic brain injury. Neurocrit. Care 2012, 16, 258–266. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Q.; Wu, X.; Sun, Y.; Yu, J.; Li, Z.; Du, Z.; Mao, Y.; Zhou, L.; Hu, J. Impact of intracranial pressure monitoring on mortality in patients with traumatic brain injury: A systematic review and meta-analysis. J. Neurosurg. 2015, 122, 574–587. [Google Scholar] [CrossRef] [PubMed]
- Alali, A.S.; Fowler, R.A.; Mainprize, T.G.; Scales, D.C.; Kiss, A.; de Mestral, C.; Ray, J.G.; Nathens, A.B. Intracranial pressure monitoring in severe traumatic brain injury: Results from the american college of surgeons trauma quality improvement program. J. Neurotrauma 2013, 30, 1737–1746. [Google Scholar] [CrossRef] [PubMed]
- Chesnut, R.M.; Temkin, N.; Carney, N.; Dikmen, S.; Rondina, C.; Videtta, W.; Petroni, G.; Lujan, S.; Pridgeon, J.; Barber, J.; et al. A trial of intracranial-pressure monitoring in traumatic brain injury. N. Engl. J. Med. 2012, 367, 2471–2481. [Google Scholar] [CrossRef] [PubMed]
- Stover, J.F. Contemporary view on neuromonitoring following severe traumatic brain injury. World J. Crit. Care Med. 2012, 1, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Hawthorne, C.; Piper, I. Monitoring of intracranial pressure in patients with traumatic brain injury. Front. Neurol. 2014, 5, 121. [Google Scholar] [CrossRef] [PubMed]
- Stocchetti, N.; Picetti, E.; Berardino, M.; Buki, A.; Chesnut, R.M.; Fountas, K.N.; Horn, P.; Hutchinson, P.J.; Iaccarino, C.; Kolias, A.G.; et al. Clinical applications of intracranial pressure monitoring in traumatic brain injury: Report of the milan consensus conference. Acta Neurochir. 2014, 156, 1615–1622. [Google Scholar] [CrossRef] [PubMed]
- Kukreti, V.; Mohseni-Bod, H.; Drake, J. Management of raised intracranial pressure in children with traumatic brain injury. J. Pediatr. Neurosci. 2014, 9, 207–215. [Google Scholar] [PubMed]
- Chesnut, R.; Videtta, W.; Vespa, P.; le Roux, P.; Participants in the International Multidisciplinary Consensus Conference on Multimodality Monitoring. Intracranial pressure monitoring: Fundamental considerations and rationale for monitoring. Neurocrit. Care 2014, 21, S64–S84. [Google Scholar] [CrossRef] [PubMed]
- Kirkman, M.A.; Smith, M. Intracranial pressure monitoring, cerebral perfusion pressure estimation, and icp/cpp-guided therapy: A standard of care or optional extra after brain injury? Br. J. Anaesth. 2014, 112, 35–46. [Google Scholar] [CrossRef] [PubMed]
- Rosner, M.J. Introduction to cerebral perfusion pressure management. Neurosurg. Clin. N. Am. 1995, 6, 761–773. [Google Scholar] [PubMed]
- Rosner, M.J.; Rosner, S.D.; Johnson, A.H. Cerebral perfusion pressure: Management protocol and clinical results. J. Neurosurg. 1995, 83, 949–962. [Google Scholar] [CrossRef] [PubMed]
- Robertson, C.S. Management of cerebral perfusion pressure after traumatic brain injury. Anesthesiology 2001, 95, 1513–1517. [Google Scholar] [CrossRef] [PubMed]
- Nordstrom, C.H. Physiological and biochemical principles underlying volume-targeted therapy—The “lund concept”. Neurocrit. Care 2005, 2, 83–95. [Google Scholar] [CrossRef]
- Contant, C.F.; Valadka, A.B.; Gopinath, S.P.; Hannay, H.J.; Robertson, C.S. Adult respiratory distress syndrome: A complication of induced hypertension after severe head injury. J. Neurosurg. 2001, 95, 560–568. [Google Scholar] [CrossRef] [PubMed]
- Juul, N.; Morris, G.F.; Marshall, S.B.; Marshall, L.F. Intracranial hypertension and cerebral perfusion pressure: Influence on neurological deterioration and outcome in severe head injury. The executive committee of the international selfotel trial. J. Neurosurg. 2000, 92, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.D.; Piper, I.R.; Dearden, N.M. Management of intracranial hypertension in head injury: Matching treatment with cause. Acta Neurochir. Suppl. 1993, 57, 152–159. [Google Scholar] [PubMed]
- Bohman, L.E.; Heuer, G.G.; Macyszyn, L.; Maloney-Wilensky, E.; Frangos, S.; le Roux, P.D.; Kofke, A.; Levine, J.M.; Stiefel, M.F. Medical management of compromised brain oxygen in patients with severe traumatic brain injury. Neurocrit. Care 2011, 14, 361–369. [Google Scholar] [CrossRef] [PubMed]
- Nates, J.L.; Niggemeyer, L.E.; Anderson, M.B.; Tuxen, D.V. Cerebral perfusion pressure monitoring alert! Crit. Care Med. 1997, 25, 895–896. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Xu, P.; Asgari, S.; Vespa, P.; Bergsneider, M. Forecasting icp elevation based on prescient changes of intracranial pressure waveform morphoogy. IEEE Trans. Bio-Med. Eng. 2010, 57, 1070–1078. [Google Scholar]
- Majdan, M.; Mauritz, W.; Wilbacher, I.; Brazinova, A.; Rusnak, M.; Leitgeb, J. Timing and duration of intracranial hypertension versus outcomes after severe traumatic brain injury. Minerva Anestesiol. 2014, 80, 1261–1272. [Google Scholar] [PubMed]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kawoos, U.; McCarron, R.M.; Auker, C.R.; Chavko, M. Advances in Intracranial Pressure Monitoring and Its Significance in Managing Traumatic Brain Injury. Int. J. Mol. Sci. 2015, 16, 28979-28997. https://doi.org/10.3390/ijms161226146
Kawoos U, McCarron RM, Auker CR, Chavko M. Advances in Intracranial Pressure Monitoring and Its Significance in Managing Traumatic Brain Injury. International Journal of Molecular Sciences. 2015; 16(12):28979-28997. https://doi.org/10.3390/ijms161226146
Chicago/Turabian StyleKawoos, Usmah, Richard M. McCarron, Charles R. Auker, and Mikulas Chavko. 2015. "Advances in Intracranial Pressure Monitoring and Its Significance in Managing Traumatic Brain Injury" International Journal of Molecular Sciences 16, no. 12: 28979-28997. https://doi.org/10.3390/ijms161226146
APA StyleKawoos, U., McCarron, R. M., Auker, C. R., & Chavko, M. (2015). Advances in Intracranial Pressure Monitoring and Its Significance in Managing Traumatic Brain Injury. International Journal of Molecular Sciences, 16(12), 28979-28997. https://doi.org/10.3390/ijms161226146




