Protective Effects of Cardamom in Isoproterenol-Induced Myocardial Infarction in Rats
Abstract
:1. Introduction
2. Results
2.1. Per Se Effect of the Cardamom
2.2. Effect of Cardamom Treatment on Hemodynamic Parameters
| Treatments | SAP (mm of Hg) | DAP (mm of Hg) | MAP (mm of Hg) | HR (beats/min) |
|---|---|---|---|---|
| Normal | 146 ± 21 | 138 ± 20 | 141 ± 23 | 390 ± 42 |
| ISO | 91 ± 18 a | 76 ± 18 a | 81 ± 19 a | 234 ± 28 a |
| C (100) | 150 ± 25 | 136 ± 24 | 142 ± 26 | 402 ± 37 |
| C (200) | 153 ± 22 | 141 ± 28 | 144 ± 24 | 396 ± 40 |
| C (100) + ISO | 100 ± 14 | 119 ± 15 b | 125 ± 18 b | 344 ± 52 b |
| C (200) + ISO | 102 ± 15 | 126 ± 24 b | 135 ± 22 b | 356 ± 46 b |
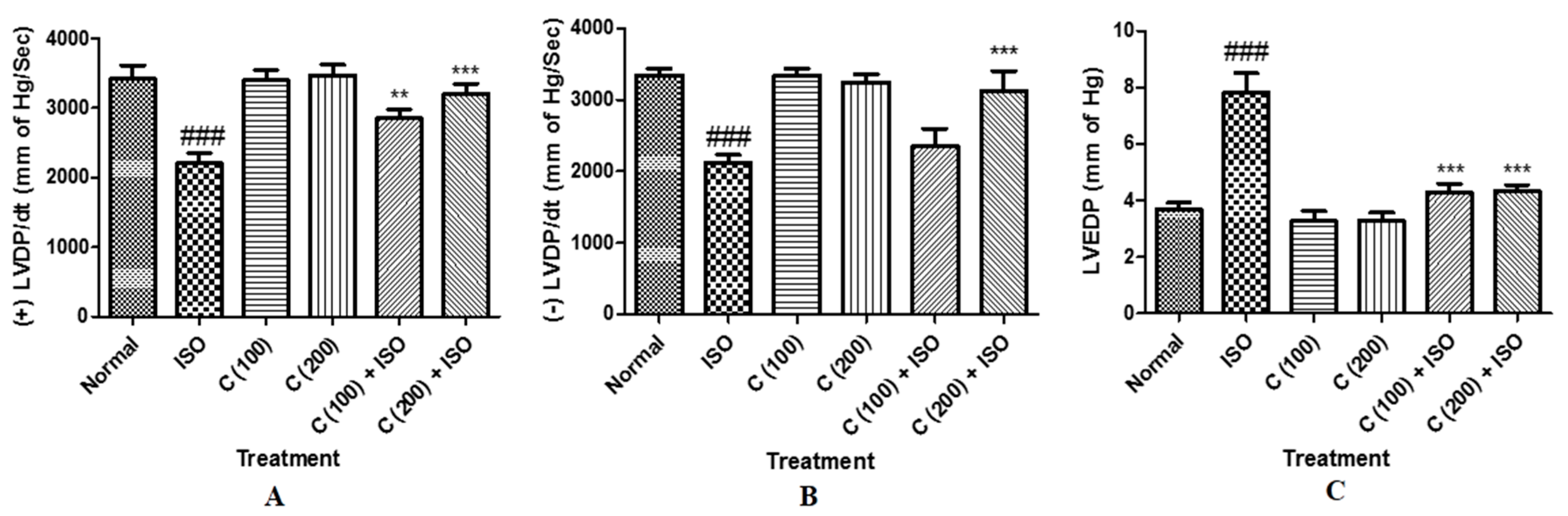
2.3. Effect of Cardamom Treatment on Left Ventricular Function
2.4. Effect of Cardamom Treatment on Myocardial Injury Markers
| Treatments | CK-MB (IU/mg Protein) | LDH (IU/mg Protein) |
|---|---|---|
| Normal | 176.32 ± 16.25 | 242.50 ± 18.21 |
| ISO | 76.50 ± 8.32 a | 134.26 ± 12.92 a |
| C (100) | 181.22 ± 14.86 | 241.62 ± 14.48 |
| C (200) | 179.53 ± 15.16 | 251.84 ± 17.33 |
| C (100) + ISO | 150.25 ± 13.72 b | 196.67 ± 20.12 b |
| C (200) + ISO | 163.00 ± 12.55 b | 229.11 ± 16.76 b |
2.5. Effect of Cardamom Treatment on Reduced Glutathione and Lipid Peroxidation
| Treatments | GSH (µmol/g Tissue) | MDA (nmol/g Tissue) |
|---|---|---|
| Normal | 2.42 ± 0.68 | 90.85 ± 10.25 |
| ISO | 1.16 ± 0.32 a | 312.36 ± 11.48 a |
| C (100) | 2.49 ± 0.61 | 76.45 ± 13.32 |
| C (200) | 2.71 ± 0.70 | 78.46 ± 11.68 |
| C (100) + ISO | 1.98 ± 0.27 b | 123.46 ± 14.23 b |
| C (200) + ISO | 2.10 ± 0.75 b | 93.88 ± 15.74 b |
| Treatments | SOD (U/mg Protein) | CAT (U/mg Protein) | GSHPx (U/mg Protein) |
|---|---|---|---|
| Normal | 10.35 ± 2.02 | 24.28 ± 2.12 | 1.96 ± 0.21 |
| ISO | 5.80 ± 1.44 a | 16.76 ± 1.66 a | 0.64 ± 0.13 a |
| C (100) | 11.20 ± 1.65 | 23.83 ± 3.24 | 1.79 ± 0.30 |
| C (200) | 10.98 ± 1.78 | 25.74 ± 2.95 | 1.88 ± 0.20 |
| C (100) + ISO | 8.48 ± 2.43 b | 20.64 ± 3.12 b | 1.35 ± 0.37 b |
| C (200) + ISO | 9.56 ± 2.68 b | 22.78 ± 2.43 b | 1.86 ± 0.28 b |
2.6. Effect of Cardamom Treatment on the Activities of SOD, CAT and GSHPx Enzymes
2.7. Effect of Cardamom Treatment on Light Microscopic Changes (Histopathology) of the Myocardium
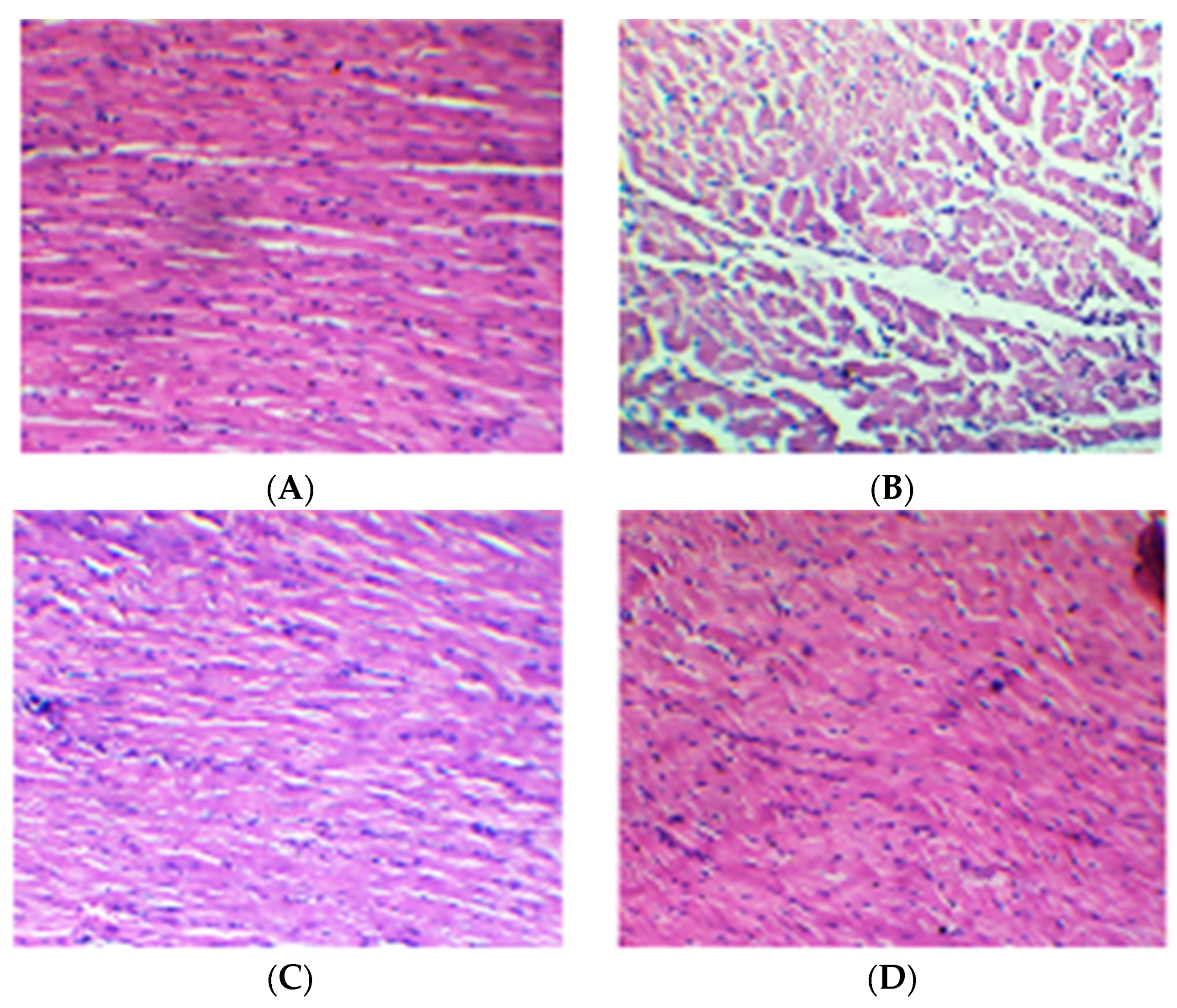
| Treatments | Myonecrosis | Inflammation | Edema |
|---|---|---|---|
| Normal | − | − | − |
| ISO | +++ | +++ | +++ |
| C (100) | − | − | − |
| C (200) | − | − | − |
| C (100) + ISO | ++ | ++ | + |
| C (200) + ISO | + | + | − |
2.8. Ultra Structural Changes by Electron Microscopy

3. Discussion
4. Experimental
4.1. Chemicals
4.2. Plant Extract and Composition
4.3. Experimental Animals
Induction of Experimental Myocardial Infarction
4.4. Experimental Design
4.4.1. Group 1 (Vehicle Treated/Normal)
4.4.2. Group II (ISO/Diseased Control)
4.4.3. Groups III–IV (Cardamom 100 or 200 mg/kg only)
4.4.4. Groups V–VI (Cardamom 100 or 200 mg/kg + ISO)
4.5. Assessment of Hemodynamic and Left Ventricular Function
4.6. Assessment of Biochemical Parameters in Heart
Processing of Heart Tissue
4.7. Assessment of Histopathological Studies
4.8. Ultra Structural Studies by Electron Microscopy
4.9. Statistical Analysis
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kloner, R.A. New observations regarding post-ischemia/reperfusion myocardial swelling. J. Am. Coll. Cardiol. 2015, 65, 324–326. [Google Scholar] [CrossRef] [PubMed]
- Prince, P.S.M.; Dhanasekar, K.; Rajakumar, S. Vanillic acid prevents altered ion pumps, ions, inhibits FAS-receptor and caspase mediated apoptosis-signaling pathway and cardiomyocyte death in myocardial infarcted rats. Chem. Biol. Interact. 2015, 232, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Hemalatha, K.; Prince, P. Preventive effects of zingerone on altered lipid peroxides and nonenzymatic antioxidants in the circulation of isoproterenol-induced myocardial infarcted rats. J. Biochem. Mol. Toxicol. 2015, 29, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Liu, J.; Shi, J.; Zhou, Q.; Liu, J.; Zhang, X.; Du, Z.; Liu, Q.; Guo, Y. Melatonin protects ADSCS from ROS and enhances their therapeutic potency in a rat model of myocardial infarction. J. Cell. Mol. Med. 2015, 19, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.Q.; Akhtar, M.S.; Akhtar, M.; Ali, J.; Haque, S.E.; Najmi, A.K. Edaravone protects rats against oxidative stress and apoptosis in experimentally induced myocardial infarction: Biochemical and ultrastructural evidence. Redox Rep. 2015. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, D.; Chattopadhyay, A.; Ghosh, G.; Datta, A.G. Oxidative stress-induced ischemic heart disease: Protection by antioxidants. Curr. Med. Chem. 2004, 11, 369–387. [Google Scholar] [CrossRef] [PubMed]
- Rathore, N.; John, S.; Kale, M.; Bhatnagar, D. Lipid peroxidation and antioxidant enzymes in isoproterenol induced oxidative stress in rat tissues. Pharmacol. Res. 1998, 38, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Ojha, S.K.; Nandave, M.; Arora, S.; Narang, R.; Dinda, A.K.; Arya, D.S. Chronic administration of Tribulus terrestrislinn extract improves cardiac function and attenuates myocardial infarction in rats. Int. J. Pharmacol. 2008, 4, 1–10. [Google Scholar] [CrossRef]
- Visioli, F.; Borsani, L.; Galli, C. Diet and prevention of coronary heart disease: The potential role of phytochemicals. Cardiovasc. Res. 2000, 47, 419–425. [Google Scholar] [CrossRef]
- Sandhu, D.S.; Heinrich, M. The use of health foods, spices and other botanicals in the Sikh community in London. Phytother. Res. 2005, 19, 633–642. [Google Scholar] [CrossRef] [PubMed]
- Vijayan, K.; Madhusoodanan, K.; Radhakrishnan, V.; Ravindran, P. 12 properties and end-uses of cardamom. In Cardamom; CRC Press: Boca Raton, FL, USA, 2002; pp. 269–283. [Google Scholar]
- Amma, K.P.; Sasidharan, I.; Sreekumar, M.; Sumathykutty, M.; Arumughan, C. Total antioxidant capacity and change in phytochemicals of four major varieties of cardamom oils during decortication. Int. J. Food Prop. 2015, 18, 1317–1325. [Google Scholar] [CrossRef]
- Bhattacharjee, S.; Rana, T.; Sengupta, A. Inhibition of lipid peroxidation and enhancement of GST activity by cardamom and cinnamon during chemically induced colon carcinogenesis in Swiss albino mice. Asian Pac. J. Cancer Prev. 2007, 8, 578–582. [Google Scholar] [PubMed]
- Jamal, A.; Javed, K.; Aslam, M.; Jafri, M. Gastroprotective effect of cardamom, elettaria cardamomummaton. Fruits in rats. J. Ethnopharmacol. 2006, 103, 149–153. [Google Scholar] [CrossRef] [PubMed]
- Gilani, A.H.; Jabeen, Q.; Khan, A.-u.; Shah, A.J. Gut modulatory, blood pressure lowering, diuretic and sedative activities of cardamom. J. Ethnopharmacol. 2008, 115, 463–472. [Google Scholar] [CrossRef] [PubMed]
- Verma, S.; Jain, V.; Katewa, S. Blood pressure lowering, fibrinolysis enhancing and antioxidant activities of cardamom (Elettaria.cardamomum). Ind. J. Biochem. Biophys. 2009, 46, 503–506. [Google Scholar]
- Al-Zuhair, H.; El-Sayeh, B.; Ameen, H.; Al-Shoora, H. Pharmacological studies of cardamom oil in animals. Pharmacol. Res. 1996, 34, 79–82. [Google Scholar] [CrossRef] [PubMed]
- Majdalawieh, A.F.; Carr, R.I. In vitro investigation of the potential immunomodulatory and anti-cancer activities of black pepper (Piper nigrum) and cardamom (Elettaria cardamomum). J. Med. Food 2010, 13, 371–381. [Google Scholar] [CrossRef] [PubMed]
- Qiblawi, S.; Dhanarasu, S. Chemopreventive effect of cardamom (Elettaria cardamomum L.) against benzo (a) pyrene-induced forestomach papillomagenesis in swiss albino mice. J. Environ. Pathol. Toxicol. Oncol. 2015, 34, 95–104. [Google Scholar] [CrossRef] [PubMed]
- Yadav, A.S.; Bhatnagar, D. Free radical scavenging activity, metal chelation and antioxidant power of some of the indianspices. Biofactors 2007, 31, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Suneetha, W.J.; Krishnakantha, T. Cardamom extract as inhibitor of human platelet aggregation. Phytother. Res. 2005, 19, 437–440. [Google Scholar] [CrossRef] [PubMed]
- Goyal, S.; Arora, S.; Bhatt, T.K.; Das, P.; Sharma, A.; Kumari, S.; Arya, D.S. Modulation of PPAR-γ by telmisartan protects the heart against myocardial infarction in experimental diabetes. Chem. Biol. Interact. 2010, 185, 271–280. [Google Scholar] [CrossRef] [PubMed]
- Lobo Filho, H.G.; Ferreira, N.L.; Sousa, R.B.d.; Carvalho, E.R.d.; Lobo, P.L.D.; Lobo Filho, J.G. Experimental model of myocardial infarction induced by isoproterenol in rats. Rev. Bras. Cir. Cardiovasc. 2011, 26, 469–476. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Babu, V.; Nagarajan, K.; Machawal, L.; Bajaj, U. Protective effects of centella asiatica against isoproterenol-induced myocardial infarction in rats: Biochemical, mitochondrial and histological findings. J. Phytopharmacol. 2015, 4, 80–86. [Google Scholar]
- Neri, M.; Fineschi, V.; di Paolo, M.; Pomara, C.; Riezzo, I.; Turillazzi, E.; Cerretani, D. Cardiac oxidative stress and inflammatory cytokines response after myocardial infarction. Curr. Vasc. Pharmacol. 2015, 13, 26–36. [Google Scholar] [CrossRef] [PubMed]
- Mnafgui, K.; Hajji, R.; Derbali, F.; Khlif, I.; Kraiem, F.; Ellefi, H.; Elfeki, A.; Allouche, N.; Gharsallah, N. Protective effect of hydroxytyrosol against cardiac remodeling after isoproterenol-induced myocardial infarction in rat. Cardiovasc. Toxicol. 2015. [Google Scholar] [CrossRef] [PubMed]
- Govindarajan, V.; Narasimhan, S.; Raghuveer, K.; Lewis, Y.; Stahl, W.H. Cardamom—Production, technology, chemistry, and quality. Crit. Rev. Food Sci. Nutr. 1982, 16, 229–326. [Google Scholar] [CrossRef] [PubMed]
- Ohkawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal. Biochem. 1979, 95, 351–358. [Google Scholar] [CrossRef]
- Moron, M.S.; Depierre, J.W.; Mannervik, B. Levels of glutathione, glutathione reductase and glutathione s-transferase activities in rat lung and liver. Biochim. Biophys. Acta 1979, 582, 67–78. [Google Scholar] [CrossRef]
- Aebi, H. Catalase in vitro. Meth. Enzymol. 1984, 105, 121–126. [Google Scholar] [PubMed]
- Marklund, S.; Marklund, G. Involvement of the superoxide anion radical in the autoxidation of pyrogallol and a convenient assay for superoxide dismutase. Eur. J. Biochem. 1974, 47, 469–474. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goyal, S.N.; Sharma, C.; Mahajan, U.B.; Patil, C.R.; Agrawal, Y.O.; Kumari, S.; Arya, D.S.; Ojha, S. Protective Effects of Cardamom in Isoproterenol-Induced Myocardial Infarction in Rats. Int. J. Mol. Sci. 2015, 16, 27457-27469. https://doi.org/10.3390/ijms161126040
Goyal SN, Sharma C, Mahajan UB, Patil CR, Agrawal YO, Kumari S, Arya DS, Ojha S. Protective Effects of Cardamom in Isoproterenol-Induced Myocardial Infarction in Rats. International Journal of Molecular Sciences. 2015; 16(11):27457-27469. https://doi.org/10.3390/ijms161126040
Chicago/Turabian StyleGoyal, Sameer N., Charu Sharma, Umesh B. Mahajan, Chandragouda R. Patil, Yogeeta O. Agrawal, Santosh Kumari, Dharamvir Singh Arya, and Shreesh Ojha. 2015. "Protective Effects of Cardamom in Isoproterenol-Induced Myocardial Infarction in Rats" International Journal of Molecular Sciences 16, no. 11: 27457-27469. https://doi.org/10.3390/ijms161126040
APA StyleGoyal, S. N., Sharma, C., Mahajan, U. B., Patil, C. R., Agrawal, Y. O., Kumari, S., Arya, D. S., & Ojha, S. (2015). Protective Effects of Cardamom in Isoproterenol-Induced Myocardial Infarction in Rats. International Journal of Molecular Sciences, 16(11), 27457-27469. https://doi.org/10.3390/ijms161126040








