The Use of the Schizonticidal Agent Quinine Sulfate to Prevent Pond Crashes for Algal-Biofuel Production
Abstract
:1. Introduction
2. Results and Discussion
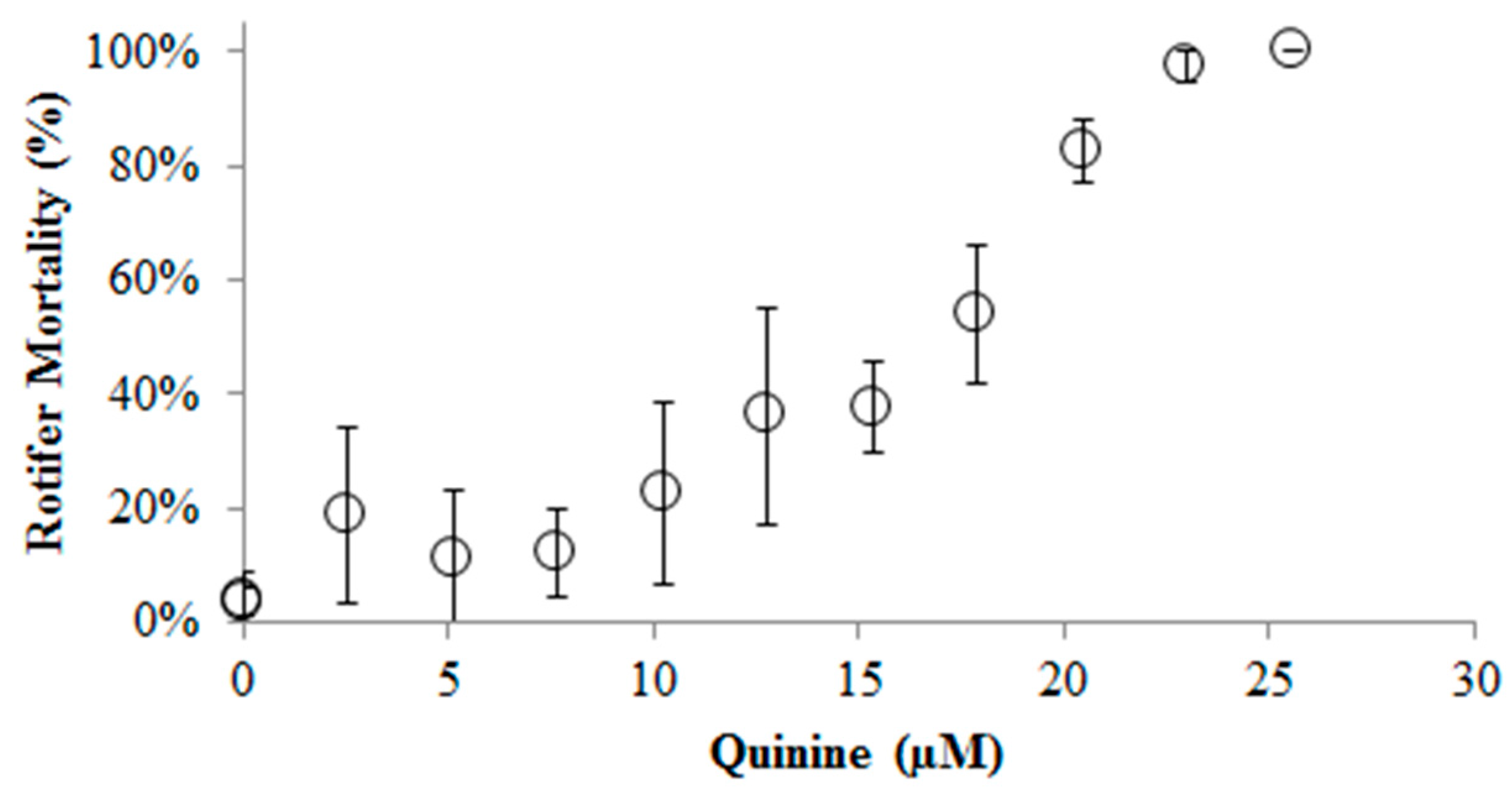
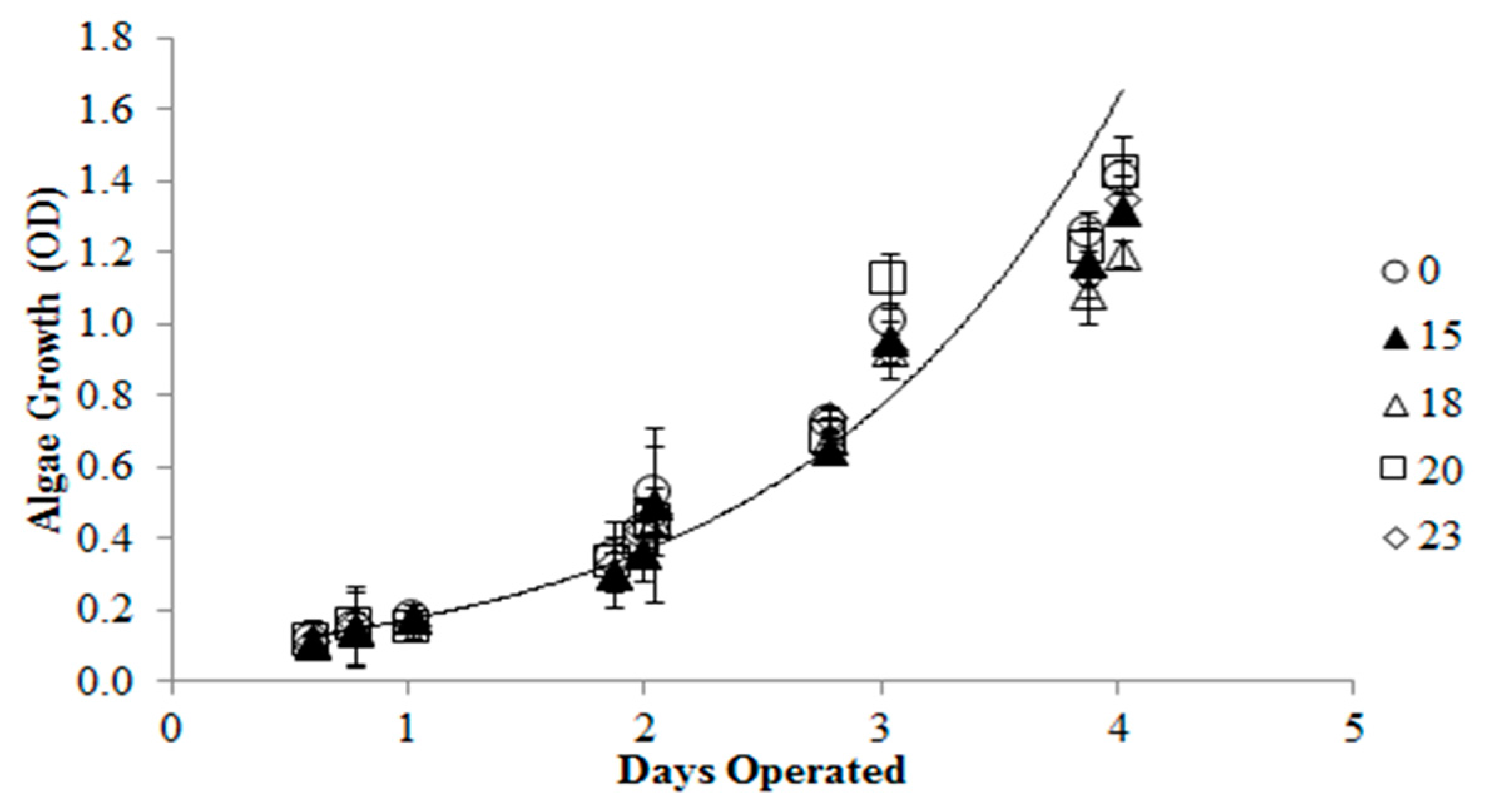
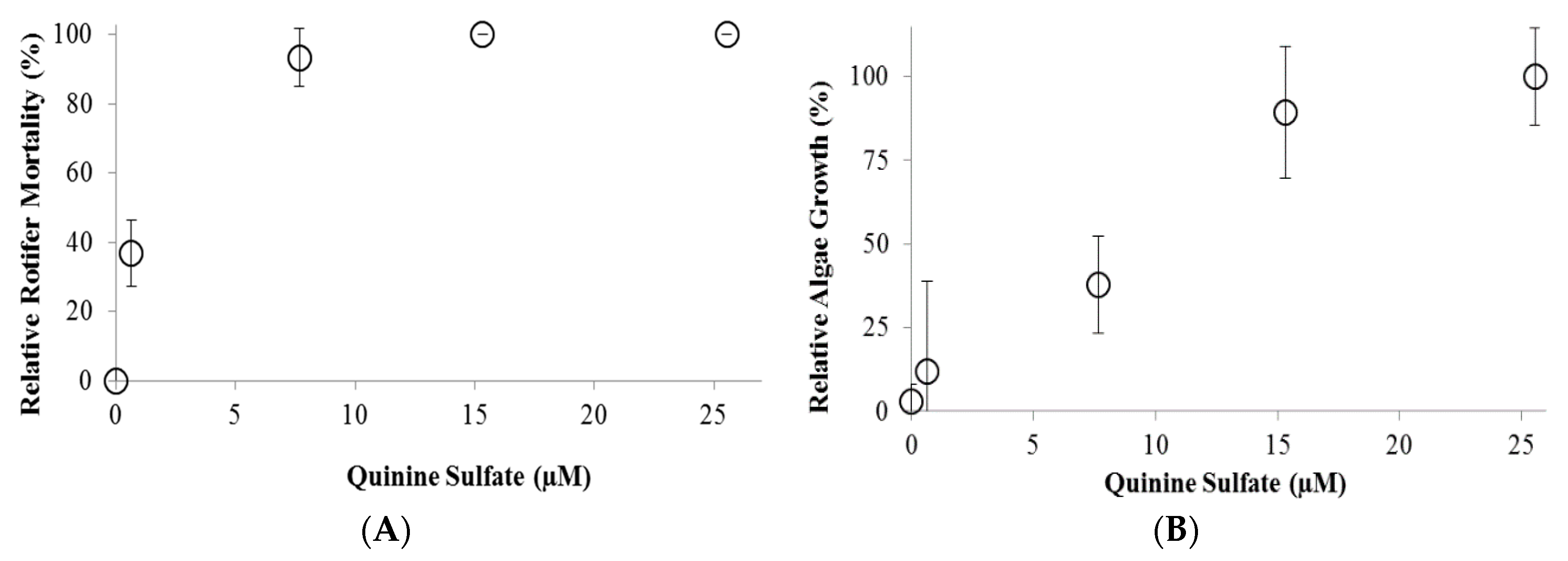

3. Experimental Section
3.1. Acute QS (Quinine Sulfate) Toxicity Tests on B. calyciflorus
3.2. QS Toxicity Tests on C. kessleri
3.3. QS Toxicity Tests on a Co-Culture of B. calyciflorus and C. kessleri
3.4. Testing the Persistence of QS
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Efroymson, R.A.; Dale, V.H. Environmental indicators for sustainable production of algal biofuels. Ecol. Indic. 2015, 49, 1–13. [Google Scholar] [CrossRef]
- Gao, Y.; Chapin, G.; Liang, Y.; Tang, D.; Tweed, C. Algae biodiesel—A feasibility report. Chem. Cent. J. 2012, 6 (Suppl. 1). [Google Scholar] [CrossRef] [PubMed]
- Committee on the Sustainable Development of Algal Biofuels; Board on Agriculture and Natural Resources; Board on Energy and Environmental Systems; Division on Earth and Life Studies; Division on Engineering and Physical Sciences; National Research Council. Sustainable Development of Algal Biofuels; National Research Council of the National Academies: Washington, DC, USA, 2012. [Google Scholar]
- Clarens, A.F.; Resurreccion, E.P.; White, M.A.; Colosi, L.M. Environmental life cycle comparison of algae to other bioenergy feedstocks. Environ. Sci. Technol. 2010, 44, 1813–1819. [Google Scholar] [CrossRef] [PubMed]
- Sander, K.; Murthy, G.S. Life cycle analysis of algae biodiesel. Int. J. Life Cycle Assess. 2010, 15, 704–714. [Google Scholar] [CrossRef]
- Woertz, I.; Feffer, A.; Lundquist, T.; Nelson, Y. Algae grown on dairy and municipal wastewater for simultaneous nutrient removal and lipid production for biofuel feedstock. J. Environ. Eng. 2009, 135, 1115–1122. [Google Scholar] [CrossRef]
- Craggs, R.; Sutherland, D.; Campbell, H. Hectare-scale demonstration of high rate algal ponds for enhanced wastewater treatment and biofuel production. J. Appl. Phycol. 2012, 24, 329–337. [Google Scholar] [CrossRef]
- Wallace, R.L. Rotifers: Exquisite metazoans. Integr. Comp. Biol. 2002, 42, 660–667. [Google Scholar] [CrossRef] [PubMed]
- Montemezzani, V.; Duggan, I.C.; Hogg, I.D.; Craggs, R.J. A review of potential methods for zooplankton control in wastewater treatment High Rate Algal Ponds and algal production raceways. Algal Res. 2015, 11, 211–226. [Google Scholar] [CrossRef]
- Xu, X.P.; Xi, Y.L.; Chu, Z.X.; Xiang, X.L. Effects of DDT and dicofol on population growth of Brachionus calyciflorus under different algal (Scenedesmus obliquus) densities. J. Environ. Biol. 2014, 35, 907–916. [Google Scholar] [PubMed]
- Van Ginkel, S.W.; Igou, T.; Hu, Z.; Narode, A.; Cheruvu, S.; Doi, S.; Johnston, R.; Snell, T.; Chen, Y. Taking advantage of rotifer sensitivity to rotenone to prevent pond crashes for algal-biofuel production. Algal Res. 2015, 10, 100–103. [Google Scholar] [CrossRef]
- Achan, J.; Talisuna, A.O.; Erhart, A.; Yeka, A.; Tibenderana, J.K.; Baliraine, F.N.; Rosenthal, P.J.; D’Alessandro, U. Quinine, an old anti-malarial drug in a modern world: Role in the treatment of malaria. Malar. J. 2011, 10, 1475–2875. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.O. A Comprehensive Review of Malaria with an Emphasis on Plasmodium Resistance. Master’s Thesis, University of Mississippi, Oxford, MS, USA, 2014. [Google Scholar]
- Wallace, R.L.; Snell, T.W. Rotifera. In Ecology and Systematics of North. American Freshwater Invertebrates, 3rd ed.; Thorp, J.H., Covich, A.P., Eds.; Academic Press: New York, NY, USA, 2010. [Google Scholar]
- Snell, T.W.; Moffat, B.D.; Janssen, C.; Persoone, G. Acute toxicity tests using rotifers: IV. Effects of cyst age, temperature, and salinity on the sensitivity of Brachionus calyciflorus. Ecotoxicol. Environ. Saf. 1991, 21, 308–317. [Google Scholar] [CrossRef]
- Preston, B.L.; Snell, T.W.; Fields, D.M.; Weissburg, M.J. The effects of fluid motion on toxicant sensitivity of the rotifer Brachionus calyciflorus. Aquat. Toxicol. 2001, 52, 117–131. [Google Scholar] [CrossRef]
- Moreno-Garrido, I.; Canavate, J.P. Assessing chemical compounds for controlling predator ciliates in outdoor mass cultures of the green algae Dunaliella salina. Aquac. Eng. 2001, 24, 107–114. [Google Scholar] [CrossRef]
- Kirst, G.O. Salinity tolerance of Eukaryotic Marine Algae. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 21–53. [Google Scholar] [CrossRef]
- Quinine Sulfate—Buy Quinine Sulfate, Chondroitine, Cs Product on Alibaba.com. Available online: http://www.alibaba.com/product-detail/Quinine-Sulfate_844495153.html (accessed on 19 June 2015).
- Ochi, K.; Eggermont, J.J. Effects of quinine on neural activity in cat primary auditory cortex. Hear. Res. 1997, 105, 105–118. [Google Scholar] [CrossRef]
- Stanier, R.Y.; Kunisawa, R.; Mandel, M.; CohenBazire, G. Purification and properties of unicellular blue green algae (order chroococcales). Bacteriol. Rev. 1971, 35, 171–205. [Google Scholar] [PubMed]
- Snell, T.W.; Moffat, B.D. A two-day life cycle test with the rotifer Brachionus calyciflorus. Environ. Toxicol. Chem. 1992, 11, 1249–1257. [Google Scholar] [CrossRef]
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xu, C.; Wu, K.; Van Ginkel, S.W.; Igou, T.; Lee, H.J.; Bhargava, A.; Johnston, R.; Snell, T.; Chen, Y. The Use of the Schizonticidal Agent Quinine Sulfate to Prevent Pond Crashes for Algal-Biofuel Production. Int. J. Mol. Sci. 2015, 16, 27450-27456. https://doi.org/10.3390/ijms161126035
Xu C, Wu K, Van Ginkel SW, Igou T, Lee HJ, Bhargava A, Johnston R, Snell T, Chen Y. The Use of the Schizonticidal Agent Quinine Sulfate to Prevent Pond Crashes for Algal-Biofuel Production. International Journal of Molecular Sciences. 2015; 16(11):27450-27456. https://doi.org/10.3390/ijms161126035
Chicago/Turabian StyleXu, Chunyan, Kangyan Wu, Steve W. Van Ginkel, Thomas Igou, Hwa Jong Lee, Aditya Bhargava, Rachel Johnston, Terry Snell, and Yongsheng Chen. 2015. "The Use of the Schizonticidal Agent Quinine Sulfate to Prevent Pond Crashes for Algal-Biofuel Production" International Journal of Molecular Sciences 16, no. 11: 27450-27456. https://doi.org/10.3390/ijms161126035
APA StyleXu, C., Wu, K., Van Ginkel, S. W., Igou, T., Lee, H. J., Bhargava, A., Johnston, R., Snell, T., & Chen, Y. (2015). The Use of the Schizonticidal Agent Quinine Sulfate to Prevent Pond Crashes for Algal-Biofuel Production. International Journal of Molecular Sciences, 16(11), 27450-27456. https://doi.org/10.3390/ijms161126035





