Neoadjuvant Down-Sizing of Hilar Cholangiocarcinoma with Photodynamic Therapy—Long-Term Outcome of a Phase II Pilot Study †
Abstract
:1. Introduction
2. Results
2.1. Clinical Protocol and Short-Term Results
| Patient | Age/Gender | Bismuth Stage 1 | UICC Stage 2 |
|---|---|---|---|
| 1 | 71/male | II (L 0.5 cm, C 3 cm) | II |
| 2 | 56/male | IV (L 3 cm, R 1.5 cm, C 3 cm) | III |
| 3 | 52/male | IV (L 4.5 cm, R 1.5 cm, C 2 cm) | IVA |
| 4 | 72/male | IIIa (L 0.5 cm, R 1.5 cm, C 2 cm) | IVA |
| 5 | 43/female | IIIa (L 2 cm, R 4 cm, C 1 cm) | III 3 |
| 6 | 61/male | IIIb (L 3.8 cm, R 0.5 cm, C 2.2 cm) | IVA |
| 7 | 59/male | IV (L 1.5 cm, R 3.5 cm, C 2 cm) | IVA |
2.2. Long-Term Results
| Patient | Surgery | Resection | Pathological TNM Stage | Grading | Complications | Tumor Recurrence | Survival (Months) | |
|---|---|---|---|---|---|---|---|---|
| Overall 1 | Disease-Free | |||||||
| 1 | Hilar resection, PD | R0 | pT2N0M0V0L1 | G2 | Local | 35 | 19 | |
| 2 | OLT, PD | R0 | pT2N1M0V0L1 2 | G2 | Insufficiency of the pancreatic anastomosis | (None) | >180 2 | >180 2 |
| 3 | Left hemi-hepatectomy (S1–4) | R0 | pT3N2M0V1L1 | G2 | Liver | 11 | 6 | |
| 4 | Right hemi-hepatectomy (S5–8) | R0 | pT3N1M0V0L0 | G2 | Subdiaphragmatic hematoma | Peritoneum | 85 | 81 |
| 5 | Right hemi-hepatectomy (S5–8) 3 | R0 | pT1bN0M0V0L0 | G1 | Liver, right Pleura | 75 | 57 | |
| 6 | Left hemi-hepatectomy (S1–4) | R0 | pT3N0M0V1L0 | G2 | Bile leakage | Peritoneum | 16 | 10 |
| 7 | Right trisegment-ectomy (S1, 4–8) | R0 | pT3N0M0V0L1 | G2 | Bile leakage | Local, Lung | 41 | 37 |
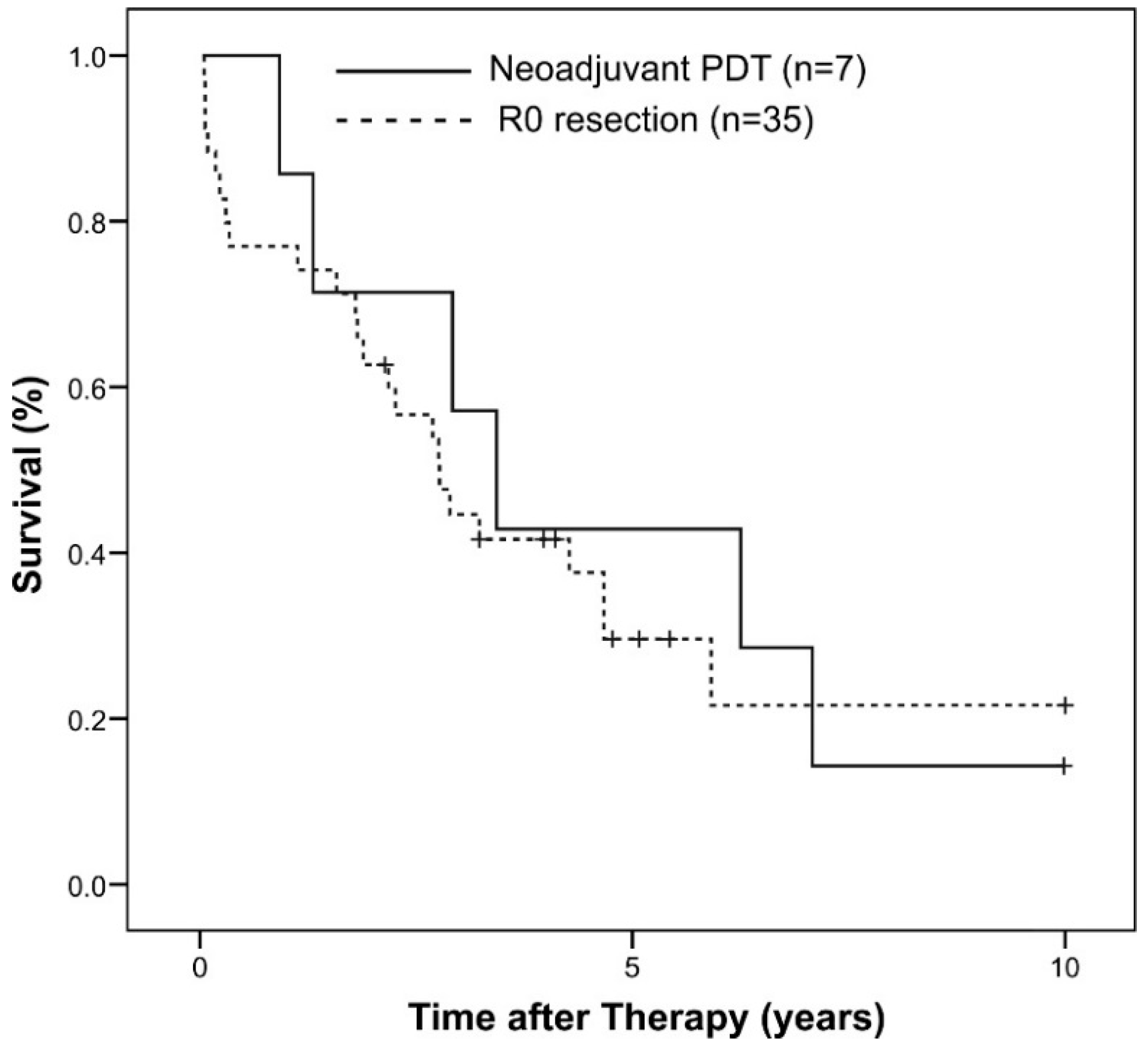
2.3. Historical Group Comparison
| Baseline Characteristics | Neoadjuvant PDT | No Pretreatment 1 | p |
|---|---|---|---|
| Patients (n) | 7 | 35 | - |
| Age (years) 2 | 59 (43–72) | 63 (38–78) | 0.41 |
| Serum Bilirubin (mg/dL) 3,4 | 1.6 ± 0.5 | 4.7 ± 1.3 | 0.47 |
| Ca 19-9 (U/L) 3 | 1347 ± 871 | 216 ± 66 | 0.17 |
| Staging | |||
| UICC Stages I & II | 29% | 26% | 0.92 |
| UICC Stage III | 14% | 3% | 0.64 |
| UICC Stage IVA | 57% | 71% | 0.57 |
| Bismuth Type I & II | 14% | 9% | 0.82 |
| Bismuth Type III | 43% | 49% | 0.82 |
| Bismuth Type IV | 43% | 43% | 1.00 |
| Lymph Node-Positive | 43% | 14% | 0.22 |
| Follow-Up | |||
| Overall Survival (years) 2 | 3.2 (0.9–7.1) | 1.8 (0.0–10.6) | 0.34 |
| 1-Year Survival Rate | 86% | 74% | - |
| 3-Year Survival Rate | 57% | 41% | - |
| 5-Year Survival Rate | 43% | 29% | - |
2.4. Discussion
3. Experimental Section
Statistics
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Blechacz, B.; Gores, G.J. Cholangiocarcinoma: Advances in pathogenesis, diagnosis, and treatment. Hepatology 2008, 48, 308–321. [Google Scholar] [CrossRef] [PubMed]
- De Groen, P.C.; Gores, G.J.; LaRusso, N.F.; Gunderson, L.L.; Nagorney, D.M. Biliary tract cancers. N. Engl. J. Med. 1999, 341, 1368–1378. [Google Scholar] [CrossRef] [PubMed]
- Shaib, Y.; El-Serag, H.B. The epidemiology of cholangiocarcinoma. Semin. Liver Dis. 2004, 24, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Mansour, J.C.; Aloia, T.A.; Crane, C.H.; Heimbach, J.K.; Nagino, M.; Vauthey, J.N. Hilar cholangiocarcinoma: Expert consensus statement. HPB (Oxford) 2015, 17, 691–699. [Google Scholar] [CrossRef] [PubMed]
- Seehofer, D.; Kamphues, C.; Neuhaus, P. Resection of klatskin tumors. Chirurg 2012, 83, 221–228. [Google Scholar] [CrossRef] [PubMed]
- Seehofer, D.; Thelen, A.; Neumann, U.P.; Veltzke-Schlieker, W.; Denecke, T.; Kamphues, C.; Pratschke, J.; Jonas, S.; Neuhaus, P. Extended bile duct resection and [corrected] liver and transplantation in patients with hilar cholangiocarcinoma: Long-term results. Liver Transpl. 2009, 15, 1499–1507. [Google Scholar] [CrossRef] [PubMed]
- Ito, F.; Agni, R.; Rettammel, R.J.; Been, M.J.; Cho, C.S.; Mahvi, D.M.; Rikkers, L.F.; Weber, S.M. Resection of hilar cholangiocarcinoma: Concomitant liver resection decreases hepatic recurrence. Ann. Surg. 2008, 248, 273–279. [Google Scholar] [CrossRef] [PubMed]
- Jarnagin, W.R.; Fong, Y.; DeMatteo, R.P.; Gonen, M.; Burke, E.C.; Bodniewicz, B.J.; Youssef, B.M.; Klimstra, D.; Blumgart, L.H. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann. Surg. 2001, 234, 507–519. [Google Scholar] [CrossRef] [PubMed]
- Nishio, H.; Nagino, M.; Nimura, Y. Surgical management of hilar cholangiocarcinoma: The nagoya experience. HPB (Oxford) 2005, 7, 259–262. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, A.; Miwa, S.; Nakata, T.; Miyagawa, S. Disease recurrence patterns after R0 resection of hilar cholangiocarcinoma. Br. J. Surg. 2010, 97, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Jarnagin, W.R.; Ruo, L.; Little, S.A.; Klimstra, D.; D’Angelica, M.; DeMatteo, R.P.; Wagman, R.; Blumgart, L.H.; Fong, Y. Patterns of initial disease recurrence after resection of gallbladder carcinoma and hilar cholangiocarcinoma: Implications for adjuvant therapeutic strategies. Cancer 2003, 98, 1689–1700. [Google Scholar] [CrossRef] [PubMed]
- Madariaga, J.R.; Iwatsuki, S.; Todo, S.; Lee, R.G.; Irish, W.; Starzl, T.E. Liver resection for hilar and peripheral cholangiocarcinomas: A study of 62 cases. Ann. Surg. 1998, 227, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Berr, F. Photodynamic therapy for cholangiocarcinoma. Semin. Liver Dis. 2004, 24, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Berr, F.; Wiedmann, M.; Tannapfel, A.; Halm, U.; Kohlhaw, K.R.; Schmidt, F.; Wittekind, C.; Hauss, J.; Mossner, J. Photodynamic therapy for advanced bile duct cancer: Evidence for improved palliation and extended survival. Hepatology 2000, 31, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Dumoulin, F.L.; Gerhardt, T.; Fuchs, S.; Scheurlen, C.; Neubrand, M.; Layer, G.; Sauerbruch, T. Phase II study of photodynamic therapy and metal stent as palliative treatment for nonresectable hilar cholangiocarcinoma. Gastrointest. Endosc. 2003, 57, 860–867. [Google Scholar] [CrossRef]
- Kahaleh, M.; Mishra, R.; Shami, V.M.; Northup, P.G.; Berg, C.L.; Bashlor, P.; Jones, P.; Ellen, K.; Weiss, G.R.; Brenin, C.M.; et al. Unresectable cholangiocarcinoma: Comparison of survival in biliary stenting alone versus stenting with photodynamic therapy. Clin. Gastroenterol. Hepatol. 2008, 6, 290–297. [Google Scholar] [CrossRef] [PubMed]
- McCaughan, J.S., Jr.; Mertens, B.F.; Cho, C.; Barabash, R.D.; Payton, H.W. Photodynamic therapy to treat tumors of the extrahepatic biliary ducts. A case report. Arch. Surg. 1991, 126, 111–113. [Google Scholar] [CrossRef] [PubMed]
- Ortner, M.A.; Liebetruth, J.; Schreiber, S.; Hanft, M.; Wruck, U.; Fusco, V.; Muller, J.M.; Hortnagl, H.; Lochs, H. Photodynamic therapy of nonresectable cholangiocarcinoma. Gastroenterology 1998, 114, 536–542. [Google Scholar] [CrossRef]
- Wiedmann, M.; Berr, F.; Schiefke, I.; Witzigmann, H.; Kohlhaw, K.; Mossner, J.; Caca, K. Photodynamic therapy in patients with non-resectable hilar cholangiocarcinoma: 5-year follow-up of a prospective phase II study. Gastrointest. Endosc. 2004, 60, 68–75. [Google Scholar] [CrossRef]
- Witzigmann, H.; Berr, F.; Ringel, U.; Caca, K.; Uhlmann, D.; Schoppmeyer, K.; Tannapfel, A.; Wittekind, C.; Mossner, J.; Hauss, J.; et al. Surgical and palliative management and outcome in 184 patients with hilar cholangiocarcinoma: Palliative photodynamic therapy plus stenting is comparable to r1/r2 resection. Ann. Surg. 2006, 244, 230–239. [Google Scholar] [CrossRef] [PubMed]
- Zoepf, T.; Jakobs, R.; Arnold, J.C.; Apel, D.; Riemann, J.F. Palliation of nonresectable bile duct cancer: Improved survival after photodynamic therapy. Am. J. Gastroenterol. 2005, 100, 2426–2430. [Google Scholar] [CrossRef] [PubMed]
- Ortner, M.E.; Caca, K.; Berr, F.; Liebetruth, J.; Mansmann, U.; Huster, D.; Voderholzer, W.; Schachschal, G.; Mossner, J.; Lochs, H. Successful photodynamic therapy for nonresectable cholangiocarcinoma: A randomized prospective study. Gastroenterology 2003, 125, 1355–1363. [Google Scholar] [CrossRef] [PubMed]
- Nanashima, A.; Abo, T.; Nonaka, T.; Nonaka, Y.; Morisaki, T.; Uehara, R.; Ohnita, K.; Fukuda, D.; Murakami, G.; Tou, K.; et al. Photodynamic therapy using talaporfin sodium (Laserphyrin®) for bile duct carcinoma: A preliminary clinical trial. Anticancer Res. 2012, 32, 4931–4938. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.; Denzer, U.W.; Neureiter, D.; Kiesslich, T.; Puespoeck, A.; Rauws, E.A.; Emmanuel, K.; Degenhardt, N.; Frick, U.; Beuers, U.; et al. Temoporfin improves efficacy of photodynamic therapy in advanced biliary tract carcinoma: A multicenter prospective phase II study. Hepatology 2015, 62, 1456–1465. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.; Kiesslich, T.; Neureiter, D.; Friesenbichler, P.; Puespoek, A.; Denzer, U.W.; Wolkersdorfer, G.W.; Emmanuel, K.; Lohse, A.W.; Berr, F. Photodynamic therapy for hilar bile duct cancer: Clinical evidence for improved tumoricidal tissue penetration by temoporfin. Photochem. Photobiol. Sci. 2013, 12, 1065–1073. [Google Scholar] [CrossRef] [PubMed]
- Nanashima, A.; Yamaguchi, H.; Shibasaki, S.; Ide, N.; Sawai, T.; Tsuji, T.; Hidaka, S.; Sumida, Y.; Nakagoe, T.; Nagayasu, T. Adjuvant photodynamic therapy for bile duct carcinoma after surgery: A preliminary study. J. Gastroenterol. 2004, 39, 1095–1101. [Google Scholar] [CrossRef] [PubMed]
- Wiedmann, M.; Caca, K.; Berr, F.; Schiefke, I.; Tannapfel, A.; Wittekind, C.; Mossner, J.; Hauss, J.; Witzigmann, H. Neoadjuvant photodynamic therapy as a new approach to treating hilar cholangiocarcinoma: A phase II pilot study. Cancer 2003, 97, 2783–2790. [Google Scholar] [CrossRef] [PubMed]
- Berr, F.; Tannapfel, A.; Lamesch, P.; Pahernik, S.; Wiedmann, M.; Halm, U.; Goetz, A.E.; Mossner, J.; Hauss, J. Neoadjuvant photodynamic therapy before curative resection of proximal bile duct carcinoma. J. Hepatol. 2000, 32, 352–357. [Google Scholar] [CrossRef]
- Sobin, L.H.; Gospodarowicz, M.K.; Wittekind, C. (Eds.) TNM Classification of Malignant Tumours, 7th ed.; Wiley-Blackwell: Hoboken, NJ, USA, 2009.
- Grendar, J.; Grendarova, P.; Sinha, R.; Dixon, E. Neoadjuvant therapy for downstaging of locally advanced hilar cholangiocarcinoma: A systematic review. HPB (Oxford) 2014, 16, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Skipworth, J.R.; Olde Damink, S.W.; Imber, C.; Bridgewater, J.; Pereira, S.P.; Malago, M. Review article: Surgical, neo-adjuvant and adjuvant management strategies in biliary tract cancer. Aliment. Pharmacol. Ther. 2011, 34, 1063–1078. [Google Scholar] [CrossRef] [PubMed]
- Darwish Murad, S.; Kim, W.R.; Harnois, D.M.; Douglas, D.D.; Burton, J.; Kulik, L.M.; Botha, J.F.; Mezrich, J.D.; Chapman, W.C.; Schwartz, J.J.; et al. Efficacy of neoadjuvant chemoradiation, followed by liver transplantation, for perihilar cholangiocarcinoma at 12 US centers. Gastroenterology 2012, 143, 88.e3–98.e3. [Google Scholar] [CrossRef] [PubMed]
- Soares, K.C.; Kamel, I.; Cosgrove, D.P.; Herman, J.M.; Pawlik, T.M. Hilar cholangiocarcinoma: Diagnosis, treatment options, and management. Hepatobiliary Surg. Nutr. 2014, 3, 18–34. [Google Scholar] [PubMed]
- McMasters, K.M.; Tuttle, T.M.; Leach, S.D.; Rich, T.; Cleary, K.R.; Evans, D.B.; Curley, S.A. Neoadjuvant chemoradiation for extrahepatic cholangiocarcinoma. Am. J. Surg. 1997, 174, 605–609. [Google Scholar] [CrossRef]
- Nelson, J.W.; Ghafoori, A.P.; Willett, C.G.; Tyler, D.S.; Pappas, T.N.; Clary, B.M.; Hurwitz, H.I.; Bendell, J.C.; Morse, M.A.; Clough, R.W.; et al. Concurrent chemoradiotherapy in resected extrahepatic cholangiocarcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2009, 73, 148–153. [Google Scholar] [CrossRef] [PubMed]
- Nonaka, Y.; Nanashima, A.; Nonaka, T.; Uehara, M.; Isomoto, H.; Abo, T.; Nagayasu, T. Synergic effect of photodynamic therapy using talaporfin sodium with conventional anticancer chemotherapy for the treatment of bile duct carcinoma. J. Surg. Res. 2013, 181, 234–241. [Google Scholar] [CrossRef] [PubMed]
- Park do, H.; Lee, S.S.; Park, S.E.; Lee, J.L.; Choi, J.H.; Choi, H.J.; Jang, J.W.; Kim, H.J.; Eum, J.B.; Seo, D.W.; et al. Randomised phase II trial of photodynamic therapy plus oral fluoropyrimidine, S-1, versus photodynamic therapy alone for unresectable hilar cholangiocarcinoma. Eur. J. Cancer 2014, 50, 1259–1268. [Google Scholar] [CrossRef] [PubMed]
- ClinicalTrials.gov. Available online: https://clinicaltrials.gov/ (accessed on 21 October 2015).
© 2015 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wagner, A.; Wiedmann, M.; Tannapfel, A.; Mayr, C.; Kiesslich, T.; Wolkersdörfer, G.W.; Berr, F.; Hauss, J.; Witzigmann, H. Neoadjuvant Down-Sizing of Hilar Cholangiocarcinoma with Photodynamic Therapy—Long-Term Outcome of a Phase II Pilot Study. Int. J. Mol. Sci. 2015, 16, 26619-26628. https://doi.org/10.3390/ijms161125978
Wagner A, Wiedmann M, Tannapfel A, Mayr C, Kiesslich T, Wolkersdörfer GW, Berr F, Hauss J, Witzigmann H. Neoadjuvant Down-Sizing of Hilar Cholangiocarcinoma with Photodynamic Therapy—Long-Term Outcome of a Phase II Pilot Study. International Journal of Molecular Sciences. 2015; 16(11):26619-26628. https://doi.org/10.3390/ijms161125978
Chicago/Turabian StyleWagner, Andrej, Marcus Wiedmann, Andrea Tannapfel, Christian Mayr, Tobias Kiesslich, Gernot W. Wolkersdörfer, Frieder Berr, Johann Hauss, and Helmut Witzigmann. 2015. "Neoadjuvant Down-Sizing of Hilar Cholangiocarcinoma with Photodynamic Therapy—Long-Term Outcome of a Phase II Pilot Study" International Journal of Molecular Sciences 16, no. 11: 26619-26628. https://doi.org/10.3390/ijms161125978
APA StyleWagner, A., Wiedmann, M., Tannapfel, A., Mayr, C., Kiesslich, T., Wolkersdörfer, G. W., Berr, F., Hauss, J., & Witzigmann, H. (2015). Neoadjuvant Down-Sizing of Hilar Cholangiocarcinoma with Photodynamic Therapy—Long-Term Outcome of a Phase II Pilot Study. International Journal of Molecular Sciences, 16(11), 26619-26628. https://doi.org/10.3390/ijms161125978