Formins: Linking Cytoskeleton and Endomembranes in Plant Cells
Abstract
:1. Introduction: Cytoskeleton in the Organization of Plant Endomembranes
2. FH2 Proteins as Versatile Cytoskeletal Regulators
3. Formins Can Associate with Cellular Membranes
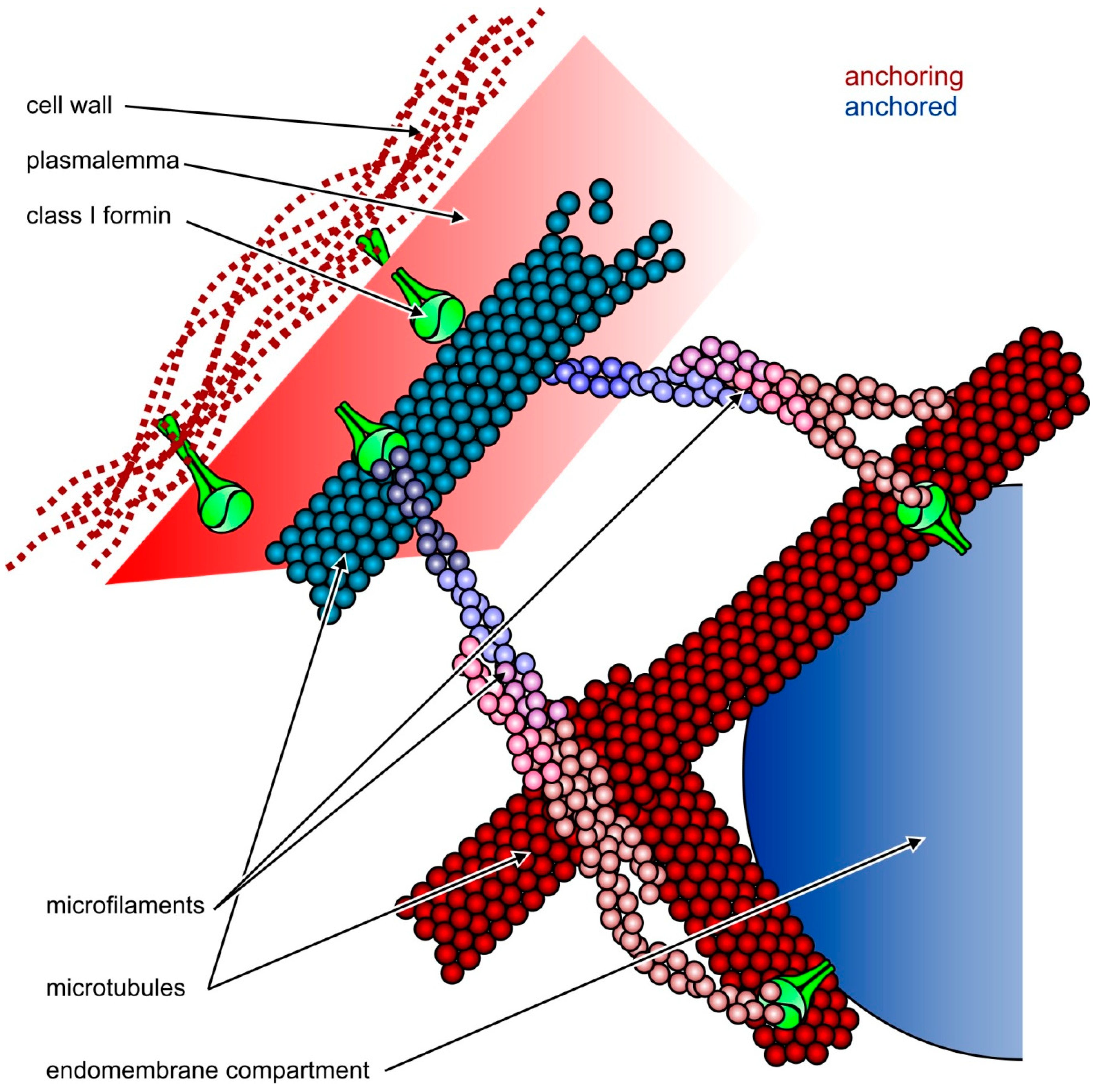
4. Fungal Formins Participate in Endomembrane Organization
5. Formins and Endomembranes: Evidence from Metazoans
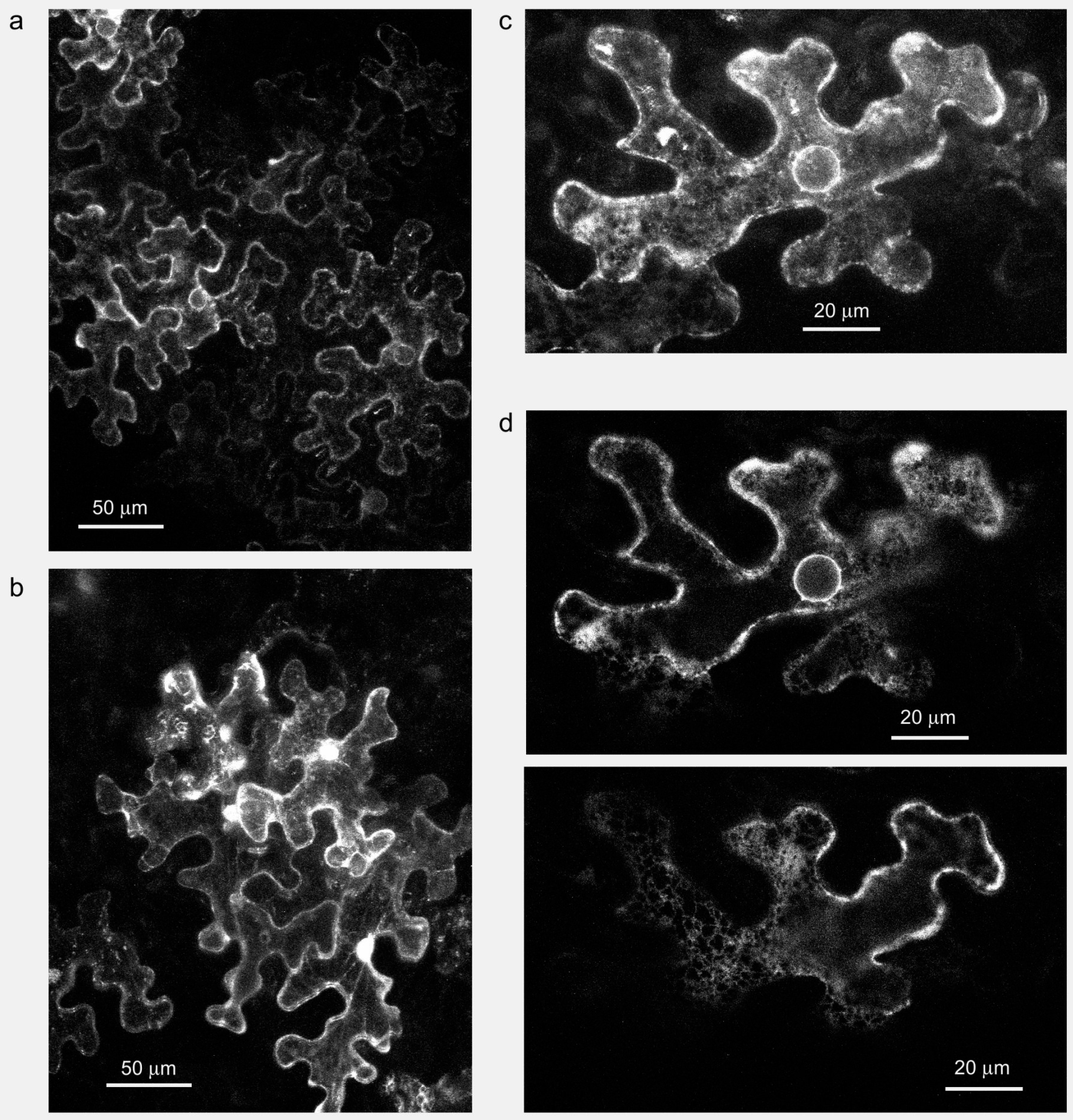
6. Membrane-Associated Plant Formins: No Longer Only at the Plasmalemma

7. Conclusion: Time to Look for Formin Functions in Plant Membrane Trafficking
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Brandizzi, F.; Barlowe, C. Organization of the ER-Golgi interface for membrane traffic control. Nat. Rev. Mol. Cell Biol. 2013, 14, 382–392. [Google Scholar] [CrossRef] [PubMed]
- Stefano, G.; Hawes, C.; Brandizzi, F. ER—the key to the highway. Curr. Opin. Plant Biol. 2014, 26, 30–38. [Google Scholar] [CrossRef]
- Sparkes, I.A.; Teanby, N.A.; Hawes, C. Truncated myosin XI tail fusions inhibit peroxisome, Golgi, and mitochondrial movement in tobacco leaf epidermal cells: A genetic tool for the next generation. J. Exp. Bot. 2008, 59, 2499–2512. [Google Scholar] [CrossRef] [PubMed]
- Avisar, D.; Abu-Abied, M.; Belausov, E.; Sadot, E.; Hawes, C.; Sparkes, I.A. A comparative study of the involvement of 17 Arabidopsis myosin family members on the motility of Golgi and other organelles. Plant Physiol. 2009, 150, 700–709. [Google Scholar] [CrossRef] [PubMed]
- Peremyslov, V.V.; Prokhnevsky, A.I.; Dolja, V.V. Class XI myosins are required for development, cell expansion, and F-Actin organization in Arabidopsis. Plant Cell 2010, 22, 1883–1897. [Google Scholar] [CrossRef]
- Avisar, D.; Abu-Abied, M.; Belausov, E.; Sadot, E. Myosin XIK is a major player in cytoplasm dynamics and is regulated by two amino acids in its tail. J. Exp. Bot. 2012, 63, 241–249. [Google Scholar] [CrossRef] [PubMed]
- Griffing, L.R.; Gao, H.T.; Sparkes, I. ER network dynamics are differentially controlled by myosins XI-K, XI-C, XI-E, XI-I, XI-1, and XI-2. Front. Plant Sci. 2014, 5. [Google Scholar] [CrossRef] [PubMed]
- Akkerman, M.; Overdijk, E.J.; Schel, J.H.; Emons, A.M.; Ketelaar, T. Golgi body motility in the plant cell cortex correlates with actin cytoskeleton organization. Plant Cell Physiol. 2011, 52, 1844–1855. [Google Scholar] [CrossRef] [PubMed]
- Miller, P.M.; Folkmann, A.W.; Maia, A.R.; Efimova, N.; Efimov, A.; Kaverina, I. Golgi-derived CLASP-dependent microtubules control Golgi organization and polarized trafficking in motile cells. Nat. Cell Biol. 2009, 11, 1069–1080. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.R.; Webster, B.M.; Mastronarde, D.N.; Verhey, K.J.; Voeltz, G.K. ER sliding dynamics and ER-mitochondrial contacts occur on acetylated microtubules. J. Cell Biol. 2010, 190, 363–375. [Google Scholar] [CrossRef] [PubMed]
- Hamada, T.; Tominaga, M.; Fukaya, T.; Nakamura, M.; Nakano, A.; Watanabe, Y.; Hashimoto, T.; Baskin, T.I. RNA processing bodies, peroxisomes, Golgi bodies, mitochondria, and endoplasmic reticulum tubule junctions frequently pause at cortical microtubules. Plant Cell Physiol. 2012, 53, 699–708. [Google Scholar] [CrossRef] [PubMed]
- Peña, E.J.; Heinlein, M. Cortical microtubule-associated ER sites: Organization centers of cell polarity and communication. Curr. Opin. Plant Biol. 2013, 16, 764–773. [Google Scholar] [CrossRef] [PubMed]
- Hamada, T.; Ueda, H.; Kawase, T.; Hara-Nishimura, I. Microtubules contribute to tubule elongation and anchoring of endoplasmic reticulum, resulting in high network complexity in Arabidopsis thaliana. Plant Physiol. 2014, 166, 1869–1876. [Google Scholar] [CrossRef] [PubMed]
- Foissner, I.; Menzel, D.; Wasteneys, G.O. Microtubule-dependent motility and orientation of the cortical endoplasmic reticulum in elongating characean internodal cells. Cell Motil. Cytoskelet. 2009, 66, 142–155. [Google Scholar] [CrossRef]
- Deeks, M.J.; Calcutt, J.R.; Ingle, E.K.; Hawkins, T.J.; Chapman, S.; Richardson, A.C.; Mentlak, D.A.; Dixon, M.R.; Cartwright, F.; Smertenko, A.P.; et al. A superfamily of actin-binding proteins at the actin-membrane nexus of higher plants. Curr. Biol. 2012, 22, 1595–1600. [Google Scholar] [CrossRef] [PubMed]
- Wang, P.; Hawkins, T.J.; Richardson, C.; Cummins, I.; Deeks, M.J.; Sparkes, I.; Hawes, C.; Hussey, P.J. The plant cytoskeleton, NET3C, and VAP27 mediate the link between the plasma membrane and endoplasmic reticulum. Curr. Biol. 2014, 24, 1397–1405. [Google Scholar] [CrossRef] [PubMed]
- Schmidt von Braun, S.; Schleiff, E. The chloroplast outer membrane protein CHUP1 interacts with actin and profilin. Planta 2008, 227, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Suetsugu, N.; Yamada, N.; Kagawa, T.; Yonekura, H.; Uyeda, T.Q.; Kadota, A.; Wada, M. Two kinesin-like proteins mediate actin-based chloroplast movement in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 2010, 107, 8860–8865. [Google Scholar] [CrossRef] [PubMed]
- Breitsprecher, D.; Goode, B.L. Formins at a glance. J. Cell Sci. 2013, 126, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Deeks, M.J.; Hussey, P.J.; Davies, B. Formins: Intermediates in signal-transduction cascades that affect cytoskeletal reorganization. Trends Plant Sci. 2002, 7, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Cvrčková, F.; Novotný, M.; Pícková, D.; Žárský, V. Formin homology 2 domains occur in multiple contexts in angiosperms. BMC Genomics 2004, 5. [Google Scholar] [CrossRef] [PubMed]
- Grunt, M.; Žárský, V.; Cvrčková, F. Roots of angiosperm formins: The evolutionary history of plant FH2 domain-containing proteins. BMC Evol. Biol. 2008, 8. [Google Scholar] [CrossRef]
- Pruyne, D.; Evangelista, M.; Yang, C.; Bi, E.; Zigmond, S.; Bretscher, A.; Boone, C. Role of formins in actin assembly: Nucleation and barbed-end association. Science 2002, 297, 612–615. [Google Scholar] [CrossRef] [PubMed]
- Sagot, I.; Rodal, A.A.; Moseley, J.; Goode, B.L.; Pellman, D. An actin nucleation mechanism mediated by Bni1 and profilin. Nat. Cell Biol. 2002, 4, 626–631. [Google Scholar] [PubMed]
- Bartolini, F.; Gundersen, G.G. Formins and microtubules. Biochim. Biophys. Acta 2010, 1803, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Chesarone, M.A.; DuPage, A.G.; Goode, B.L. Unleashing formins to remodel the actin and microtubule cytoskeletons. Nat. Rev. Mol. Cell Biol. 2010, 11, 62–74. [Google Scholar] [CrossRef]
- Wang, J.; Xue, X.; Ren, H. New insights into the role of plant formins: Regulating the organization of the actin and microtubule cytoskeleton. Protoplasma 2012, 249, S101–S107. [Google Scholar] [CrossRef] [PubMed]
- Rivero, F.; Muramoto, T.; Meyer, A.K.; Urushihara, H.; Uyeda, T.Q.; Kitayama, C. A comparative sequence analysis reveals a common GBD/FH3-FH1-FH2-DAD architecture in formins from Dictyostelium, fungi and metazoa. BMC Genomics 2005, 6. [Google Scholar] [CrossRef]
- Chalkia, D.; Nikolaidis, N.; Makalowski, W.; Klein, J.; Nei, M. Origins and evolution of the formin multigene family that is involved in the formation of actin filaments. Mol. Biol. Evol. 2008, 25, 2717–2733. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Moseley, J.B.; Sagot, I.; Poy, F.; Pellman, D.; Goode, B.L.; Eck, M.J. Crystal structures of a formin homology-2 domain reveal a tethered dimer architecture. Cell 2004, 116, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Kovar, D.R.; Harris, E.S.; Mahaffy, R.; Higgs, H.N.; Pollard, T.D. Control of the assembly of ATP- and ADP-actin by formins and profilin. Cell 2006, 124, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Scott, B.J.; Neidt, E.M.; Kovar, D.R. The functionally distinct fission yeast formins have specific actin-assembly properties. Mol. Biol. Cell 2011, 22, 3826–3839. [Google Scholar] [CrossRef] [PubMed]
- Michelot, A.; Guérin, C.; Huang, S.; Ingouff, M.; Richard, S.; Rodiuc, N.; Staiger, C.J.; Blanchoin, L. The formin homology 1 domain modulates the actin nucleation and bundling activity of Arabidopsis FORMIN1. Plant Cell 2005, 17, 2296–2313. [Google Scholar] [CrossRef] [PubMed]
- Deeks, M.J.; Cvrčková, F.; Machesky, L.M.; Mikitová, V.; Ketelaar, T.; Žárský, V.; Davies, B.; Hussey, P.J. Arabidopsis group Ie formins localize to specific cell membrane domains, interact with actin-binding proteins and cause defects in cell expansion upon aberrant expression. New Phytol. 2005, 168, 529–540. [Google Scholar] [CrossRef] [PubMed]
- Ingouff, M.; Fitz Gerald, J.N.; Guérin, C.; Robert, H.; Sørensen, M.B.; van Damme, D.; Geelen, D.; Blanchoin, L.; Berger, F. Plant formin AtFH5 is an evolutionarily conserved actin nucleator involved in cytokinesis. Nat. Cell Biol. 2005, 7, 374–380. [Google Scholar] [CrossRef] [PubMed]
- Yi, K.; Guo, C.; Chen, D.; Zhao, B.; Yang, B.; Ren, H. Cloning and functional characterization of a formin-like protein (AtFH8) from Arabidopsis. Plant Physiol. 2005, 138, 1071–1082. [Google Scholar] [CrossRef] [PubMed]
- Ye, J.; Zheng, Y.; Yan, A.; Chen, N.; Wang, Z.; Huang, S.; Yang, Z. Arabidopsis formin3 directs the formation of actin cables and polarized growth in pollen tubes. Plant Cell 2009, 21, 3868–3884. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Ren, S.; Zhang, X.; Gao, M.; Ye, S.; Qi, Y.; Zheng, Y.; Wang, J.; Zeng, L.; Li, Q.; et al. BENT UPPERMOST INTERNODE1 encodes the class II formin FH5 crucial for actin organization and rice development. Plant Cell 2011, 23, 661–680. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Xin, H.; Lin, J.; Liu, C.M.; Huang, S. An Arabidopsis class II formin, AtFH19, nucleates actin assembly, binds to the barbed end of actin filaments, and antagonizes the effect of AtFH1 on actin dynamics. J. Integr. Plant Biol. 2012, 54, 800–813. [Google Scholar] [CrossRef] [PubMed]
- Martinière, A.; Gayral, P.; Hawes, C.; Runions, J. Building bridges: Formin1 of Arabidopsis forms a connection between the cell wall and the actin cytoskeleton. Plant J. 2011, 66, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, Y.; Wu, J.; Meng, L.; Ren, H. AtFH16, an Arabidopsis type II formin, binds and bundles both microfilaments and microtubules, and preferentially binds to microtubules. J. Integr. Plant Biol. 2013, 55, 1002–1015. [Google Scholar] [CrossRef] [PubMed]
- Blanchoin, L.; Staiger, C.J. Plant formins: Diverse isoforms and unique molecular mechanism. Biochim. Biophys. Acta 2010, 1803, 201–206. [Google Scholar] [CrossRef] [PubMed]
- Schönichen, A.; Geyer, M. Fifteen formins for an actin filament: A molecular view on the regulation of human formins. Biochim. Biophys. Acta 2010, 1803, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Krainer, E.C.; Ouderkirk, J.L.; Miller, E.W.; Miller, M.R.; Mersich, A.T.; Blystone, S.D. The multiplicity of human formins: Expression patterns in cells and tissues. Cytoskeleton 2013, 70, 424–438. [Google Scholar] [CrossRef] [PubMed]
- Deeks, M.J.; Fendrych, M.; Smertenko, A.; Bell, K.S.; Oparka, K.; Cvrčková, F.; Žárský, V.; Hussey, P.J. The plant formin AtFH4 interacts with both actin and microtubules, and contains a newly identified microtubule-binding domain. J. Cell Sci. 2010, 123, 1209–1215. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Shen, Y.; Cai, C.; Zhong, C.; Zhu, L.; Yuan, M.; Ren, H. The type II Arabidopsis formin14 interacts with microtubules and microfilaments to regulate cell division. Plant Cell 2010, 22, 2710–2726. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.H.; Guo, C.Q.; Du, F.; Lu, Q.L.; Zhang, C.M.; Ren, H.Y. AtFH8 is involved in root development under effect of low-dose latrunculin B in dividing cells. Mol. Plant 2011, 4, 264–278. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhang, Y.; Tan, H.; Wang, Y.; Li, G.; Liang, W.; Yuan, Z.; Hu, J.; Ren, H.; Zhang, D. RICE MORPHOLOGY DETERMINANT encodes the type II formin FH5 and regulates rice morphogenesis. Plant Cell 2011, 23, 681–700. [Google Scholar] [CrossRef] [PubMed]
- Cvrčková, F. Formins and membranes: Anchoring cortical actin to the cell wall and beyond. Front. Plant Sci. 2013, 4. [Google Scholar] [CrossRef] [PubMed]
- Alberts, A.S. Diaphanous-related Formin homology proteins. Curr. Biol. 2002, 12, R796–R796. [Google Scholar] [CrossRef] [PubMed]
- Rousso, T.; Shewan, A.M.; Mostov, K.E.; Schejter, E.D.; Shilo, B.-Z. Apical targeting of the formin Diaphanous in Drosophila tubular epithelia. eLife 2013, 2. [Google Scholar] [CrossRef]
- Van Gisbergen, P.A.; Bezanilla, M. Plant formins: Membrane anchors for actin polymerization. Trends Cell Biol. 2013, 23, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Banno, H.; Chua, N.-H. Characterization of the arabidopsis formin-like protein AFH1 and its interacting protein. Plant Cell Physiol. 2000, 41, 617–626. [Google Scholar] [CrossRef] [PubMed]
- Cvrčková, F. Are plant formins integral membrane proteins? Genome Biol. 2000, 1. [Google Scholar] [CrossRef]
- Van Gisbergen, P.A.; Li, M.; Wu, S.Z.; Bezanilla, M. Class II formin targeting to the cell cortex by binding PI(3,5)P2 is essential for polarized growth. J. Cell Biol. 2012, 198, 235–250. [Google Scholar]
- GO:0000133: Polarisome. Available online: http://amigo.geneontology.org/amigo/term/GO:0000133 (accessed on 9 November 2014).
- Ozaki-Kuroda, K.; Yamamoto, Y.; Nohara, H.; Kinoshita, M.; Fujiwara, T.; Irie, K.; Takai, Y. Dynamic localization and function of Bni1p at the sites of directed growth in Saccharomyces cerevisiae. Mol. Cell Biol. 2001, 21, 827–839. [Google Scholar] [CrossRef] [PubMed]
- Buttery, S.M.; Yoshida, S.; Pellman, D. Yeast formins Bni1 and Bnr1 utilize different modes of cortical interaction during the assembly of actin cables. Mol. Biol. Cell 2007, 18, 1826–1838. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ferro-Novick, S.; Novick, P. Different polarisome components play distinct roles in Slt2p-regulated cortical ER inheritance in Saccharomyces cerevisiae. Mol. Biol. Cell 2013, 24, 3145–3154. [Google Scholar] [CrossRef] [PubMed]
- Chapa-Y-Lazo, B.; Lee, S.; Regan, H.; Sudbery, P. The mating projections of Saccharomyces cerevisiae and Candida albicans show key characteristics of hyphal growth. Fungal Biol. 2011, 115, 547–556. [Google Scholar] [CrossRef] [PubMed]
- Harris, S.D.; Read, N.D.; Roberson, R.W.; Shaw, B.; Seiler, S.; Plamann, M.; Momany, M. Polarisome meets spitzenkörper: Microscopy, genetics, and genomics converge. Eukaryot. Cell 2005, 4, 225–229. [Google Scholar] [CrossRef] [PubMed]
- Crampin, H.; Finley, K.; Gerami-Nejad, M.; Court, H.; Gale, C.; Berman, J.; Sudbery, P. Candida albicans hyphae have a Spitzenkörper that is distinct from the polarisome found in yeast and pseudohyphae. J. Cell Sci. 2005, 118, 2935–2947. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.A.; Sudbery, P.E. Spitzenkorper, exocyst, and polarisome components in Candida albicans hyphae show different patterns of localization and have distinct dynamic properties. Eukaryot. Cell 2010, 9, 1455–1465. [Google Scholar] [CrossRef] [PubMed]
- Sharpless, K.E.; Harris, S.D. Functional characterization and localization of the Aspergillus nidulans formin SEPA. Mol. Biol. Cell 2002, 13, 469–479. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.; Walther, A.; Wendland, J. Ras1-induced hyphal development in Candida albicans requires the formin Bni1. Eukaryot. Cell 2005, 4, 1712–1724. [Google Scholar] [CrossRef] [PubMed]
- Rida, P.C.; Nishikawa, A.; Won, G.Y.; Dean, N. Yeast-to-hyphal transition triggers formin-dependent Golgi localization to the growing tip in Candida albicans. Mol. Biol. Cell 2006, 17, 4364–4378. [Google Scholar] [CrossRef] [PubMed]
- Jourdain, I.; Dooley, H.C.; Toda, T. Fission yeast sec3 bridges the exocyst complex to the actin cytoskeleton. Traffic 2012, 13, 1481–1495. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Imai, J.; Arai, R.; Toh-E, A.; Matsui, Y.; Mabuchi, I. The small GTPase Rho3 and the diaphanous/formin For3 function in polarized cell growth in fission yeast. J. Cell Sci. 2002, 115, 4629–4639. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Novick, P. Bem1p contributes to secretory pathway polarization through a direct interaction with Exo70p. J. Cell Biol. 2014, 207, 59–72. [Google Scholar] [CrossRef] [PubMed]
- Colón-Franco, J.M.; Gomez, T.S.; Billadeau, D.D. Dynamic remodeling of the actin cytoskeleton by FMNL1γ is required for structural maintenance of the Golgi complex. J. Cell Sci. 2011, 124, 3118–3126. [Google Scholar] [CrossRef] [PubMed]
- Zilberman, Y.; Alieva, N.O.; Miserey-Lenkei, S.; Lichtenstein, A.; Kam, Z.; Sabanay, H.; Bershadsky, A. Involvement of the Rho-mDia1 pathway in the regulation of Golgi complex architecture and dynamics. Mol. Biol. Cell 2011, 22, 2900–2911. [Google Scholar] [CrossRef] [PubMed]
- Geron, E.; Schejter, E.D.; Shilo, B.Z. Directing exocrine secretory vesicles to the apical membrane by actin cables generated by the formin mDia1. Proc. Natl. Acad. Sci. USA 2013, 110, 10652–10657. [Google Scholar] [CrossRef] [PubMed]
- Chhabra, E.S.; Ramabhadran, V.; Gerber, S.A.; Higgs, H.N. INF2 is an endoplasmic reticulum-associated formin protein. J. Cell Sci. 2009, 122, 1430–1440. [Google Scholar] [CrossRef] [PubMed]
- Ramabhadran, V.; Korobova, F.; Rahme, G.J.; Higgs, H.N. Splice variant-specific cellular function of the formin INF2 in maintenance of Golgi architecture. Mol. Biol. Cell 2011, 22, 4822–4833. [Google Scholar] [CrossRef] [PubMed]
- Korobova, F.; Ramabhadran, V.; Higgs, H.N. An actin-dependent step in mitochondrial fission mediated by the ER-associated formin INF2. Science 2013, 339, 464–467. [Google Scholar] [CrossRef] [PubMed]
- Hatch, A.L.; Gurel, P.S.; Higgs, H.N. Novel roles for actin in mitochondrial fission. J. Cell Sci. 2014, 127, 4549–4560. [Google Scholar] [CrossRef] [PubMed]
- Young, K.G.; Thurston, S.F.; Copeland, S.; Smallwood, C.; Copeland, J.W. INF1 is a novel microtubule-associated formin. Mol. Biol. Cell 2008, 19, 5168–5180. [Google Scholar] [CrossRef] [PubMed]
- Liao, G.; Ma, X.; Liu, G. An RNA-zipcode-independent mechanism that localizes Dia1 mRNA to the perinuclear ER through interactions between Dia1 nascent peptide and Rho-GTP. J. Cell Sci. 2011, 124, 589–599. [Google Scholar] [CrossRef] [PubMed]
- Liao, G.; Liu, G. Immediate translation of formin DIAPH1 mRNA after its exiting the nucleus is required for its perinuclear localization in fibroblasts. PLoS One 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Ramalingam, N.; Zhao, H.; Breitsprecher, D.; Lappalainen, P.; Faix, J.; Schleicher, M. Phospholipids regulate localization and activity of mDia1 formin. Eur. J. Cell Biol. 2010, 89, 723–732. [Google Scholar] [CrossRef] [PubMed]
- Kerkhoff, E. Actin dynamics at intracellular membranes: The Spir/formin nucleator complex. Eur. J. Cell Biol. 2011, 90, 922–925. [Google Scholar] [CrossRef] [PubMed]
- Favery, B.; Chelysheva, L.A.; Lebris, M.; Jammes, F.; Marmagne, A.; de Almeida-Engler, J.; Lecomte, P.; Vaury, C.; Arkowitz, R.A.; Abad, P. Arabidopsis formin AtFH6 is a plasma membrane-associated protein upregulated in giant cells induced by parasitic nematodes. Plant Cell 2004, 16, 2529–2540. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.Y.; Wu, H.M. Overexpression of an Arabidopsis formin stimulates supernumerary actin cable formation from pollen tube cell membrane. Plant Cell 2004, 16, 257–269. [Google Scholar] [CrossRef] [PubMed]
- Rosero, A.; Stillerová, L.; Schiebertová, P.; Grunt, N.; Žárský, V.; Cvrčková, F. Arabidopsis FH1 and FH2 formins affect pavement cell shape by modulating cytoskeleton and membrane dynamics. Manuscript in preparation. 2014. [Google Scholar]
- Cheung, A.Y.; Niroomand, S.; Zou, Y.; Wu, H.M. A transmembrane formin nucleates subapical actin assembly and controls tip-focused growth in pollen tubes. Proc. Natl. Acad. Sci. USA 2010, 107, 16390–16395. [Google Scholar] [CrossRef] [PubMed]
- Voinnet, O.; Rivas, S.; Mestre, P.; Baulcombe, D. An enhanced transient expression system in plants based on suppression of gene silencing by the p19 protein of tomato bushy stunt virus. Plant J. 2003, 33, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Žárský, V.; Institute of Experimental Botany, Academy of Sciences of the Czech Republic, and Faculty of Sciences, Charles University, Prague, Czech Republic. Unpublished experimental data obtained by one of the authors. 2003.
- Oulehlová, D. Functional studies of selected members of the Arabidopsis formin family. Ph.D. Thesis, Charles University, Prague, Czech Republic, 2010. [Google Scholar]
- Rizvi, S.A.; Neidt, E.M.; Cui, J.; Feiger, Z.; Skau, C.T.; Gardel, M.L.; Kozmin, S.A.; Kovar, D.R. Identification and characterization of a small molecule inhibitor of formin-mediated actin assembly. Chem. Biol. 2009, 16, 1158–1168. [Google Scholar] [CrossRef] [PubMed]
- Rosero, A.; Žárský, V.; Cvrčková, F. AtFH1 formin mutation affects actin filament and microtubule dynamics in Arabidopsis thaliana. J. Exp. Bot. 2013, 64, 585–597. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Friml, J. Rho-GTPase-regulated vesicle trafficking in plant cell polarity. Biochem. Soc. Trans. 2014, 42, 212–218. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cvrčková, F.; Oulehlová, D.; Žárský, V. Formins: Linking Cytoskeleton and Endomembranes in Plant Cells. Int. J. Mol. Sci. 2015, 16, 1-18. https://doi.org/10.3390/ijms16010001
Cvrčková F, Oulehlová D, Žárský V. Formins: Linking Cytoskeleton and Endomembranes in Plant Cells. International Journal of Molecular Sciences. 2015; 16(1):1-18. https://doi.org/10.3390/ijms16010001
Chicago/Turabian StyleCvrčková, Fatima, Denisa Oulehlová, and Viktor Žárský. 2015. "Formins: Linking Cytoskeleton and Endomembranes in Plant Cells" International Journal of Molecular Sciences 16, no. 1: 1-18. https://doi.org/10.3390/ijms16010001
APA StyleCvrčková, F., Oulehlová, D., & Žárský, V. (2015). Formins: Linking Cytoskeleton and Endomembranes in Plant Cells. International Journal of Molecular Sciences, 16(1), 1-18. https://doi.org/10.3390/ijms16010001





