Diversity in TAF Proteomics: Consequences for Cellular Differentiation and Migration
Abstract
:1. Introduction
2. The Diversity of Transcription Factor Complex IID (TFIID) Complex Subunits in Organism Development
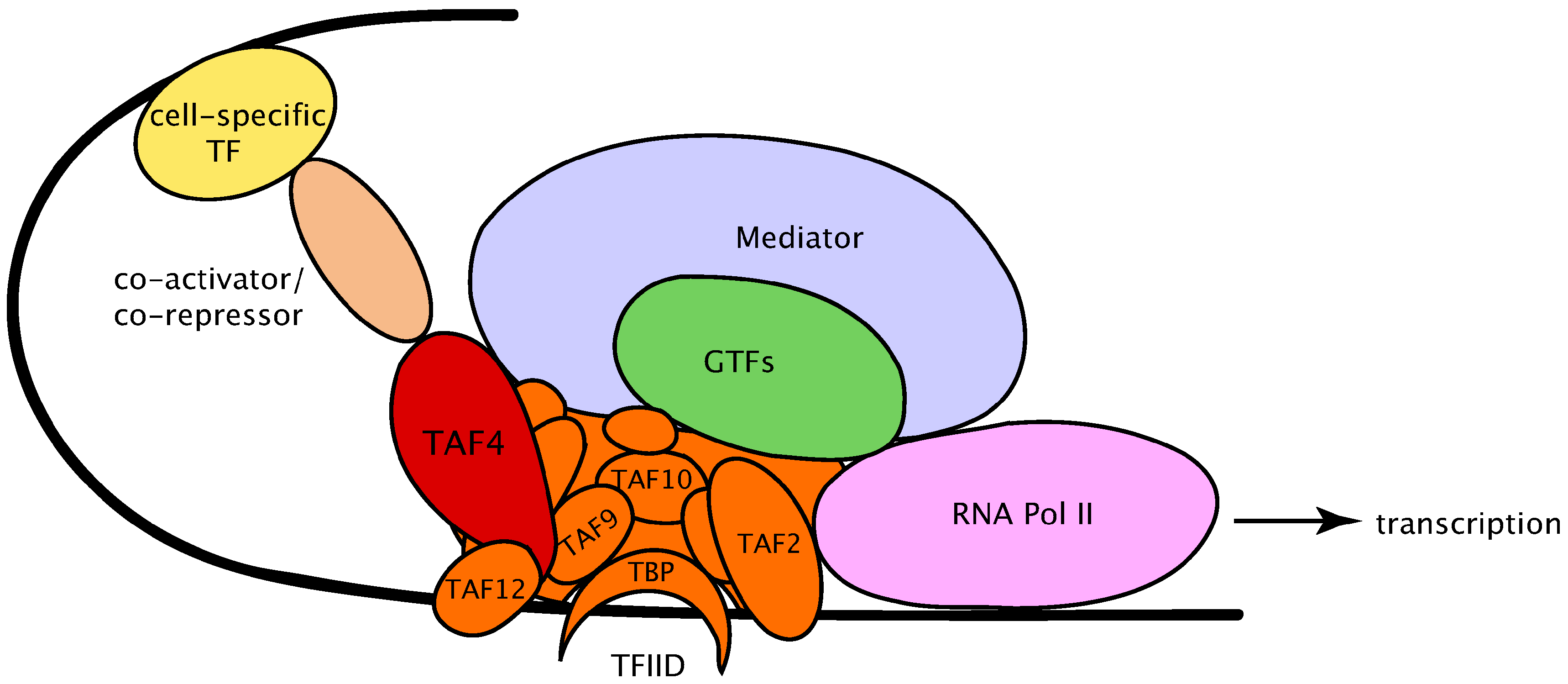
3. The Dynamics of TFIID Complex Components in Development and Differentiation
4. TATA Box-Binding Protein (TBP)-Associated Factor (TAF4): Structure, Function and Regulation
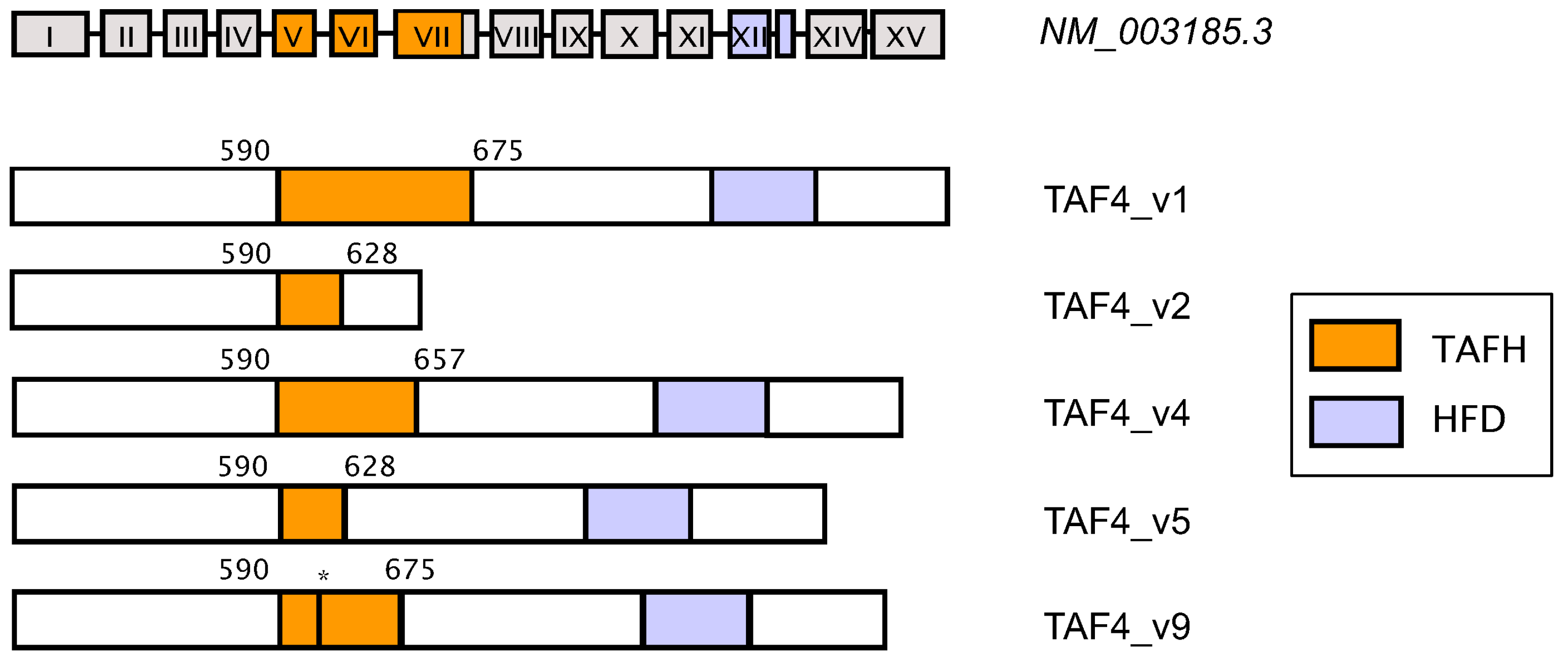
5. Coordinated Switching of TAF4 Alternative Splice Variants Controls Differentiation and Migration of Normal Progenitors and Cancer Stem Cells
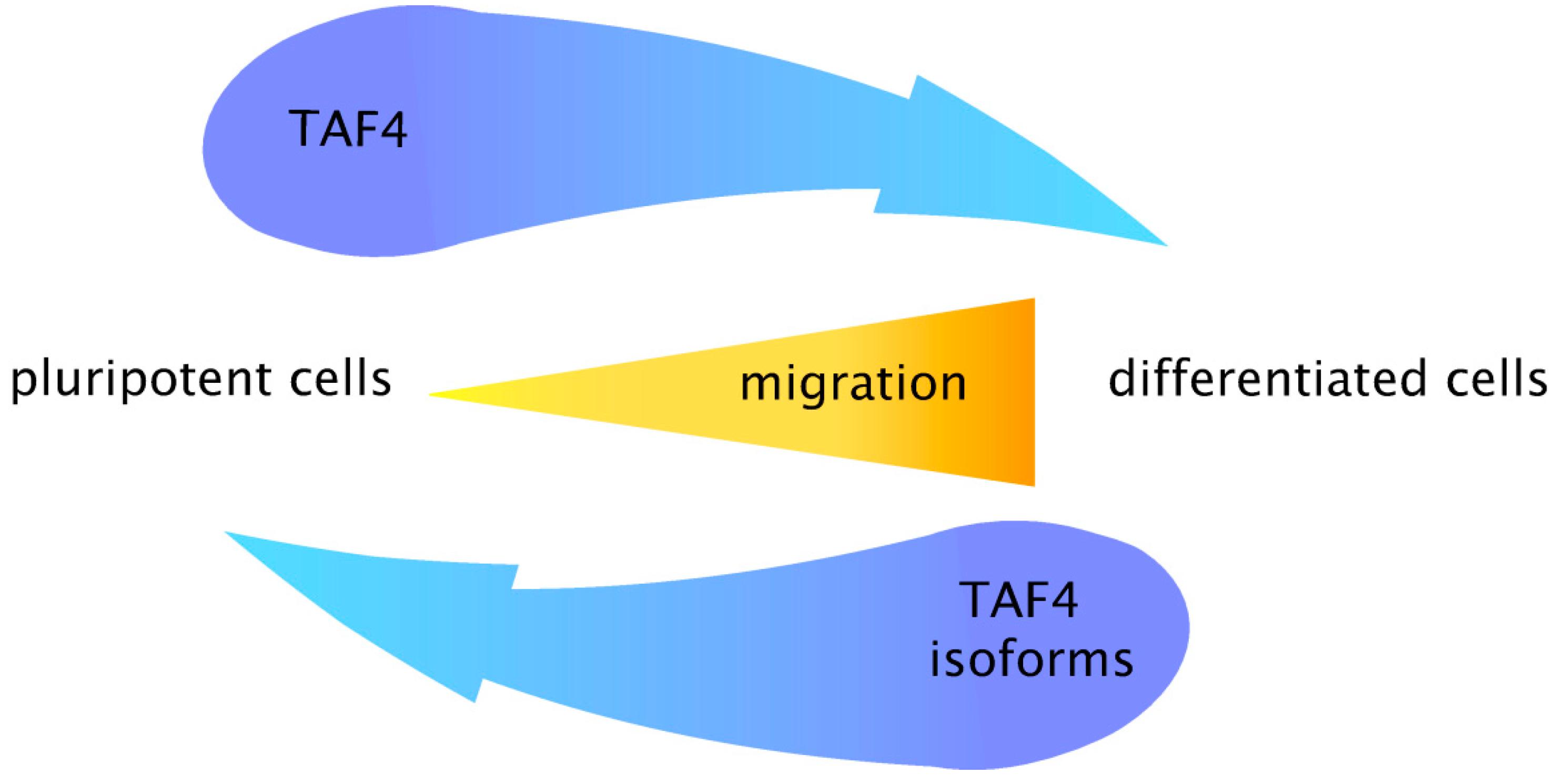
6. Conclusions
Abbreviations
| ATRA | all-trans retinoic acid |
| BAF45/BAF53 | BRG1-Associated Factor 45/53 |
| CA150 | co-activator of 150 kDa |
| ch-ERG | chicken Ets-related transcription factor |
| CREB | cAMP response element-binding protein |
| CTGF | connective tissue growth factor |
| c-Jun | V-jun avian sarcoma virus 17 oncogene homolog |
| EGF | epidermal growth factor |
| ETO domain | 8;21 translocation domain |
| GTFs | general transcription factors |
| HCF-1 | host cell factor 1 |
| HP | heterochromatin protein |
| KLF4 | Krüppel-like factor 4 |
| KRAB | Krüppel associated box |
| lncRNA | long non-coding RNA |
| LZIP | leucine zipper protein |
| MEFs | mouse embryonic fibroblasts |
| MIA/CD-RAP | melanoma inhibiting activity/cartilage-derived retinoic acid-sensitive protein |
| MITF | microphthalmia-associated transcription factor |
| MYC | myelocytomatosis viral oncogene |
| NCoR | nuclear receptor co-repressor |
| OCT4 | octamer-binding transcription factor 4 |
| OMA-1/2 | overlapping activity with M-AAA protease 1/2 |
| OPN | osteopontin |
| PBAF | polybromo-associated BAF |
| PBX | pre-B cell leukemia transcription factor |
| PGC-1 | peroxisome proliferator-activated receptor-gamma coactivator |
| PPARG | peroxisome proliferator-activated receptors |
| RARα | retinoic acid receptor-alpha |
| RXR | retinoid X receptor |
| ShcA | Src homology 2 domain containing transforming protein A |
| SKIP | Ski-interacting protein |
| SMAD | Sma and Mad Related Family |
| Sox9 | SRY (sex determining region Y)-box 9 |
| Sp1 | specificity protein 1 |
| SWI/SNF | SWItch/Sucrose NonFermentable |
| TAF | TATA box-binding protein (TBP)-associated factor |
| TAT-SF1 | cofactor required for Tat activation of HIV-1 transcription |
| TBP | TATA-box protein |
| TGFβ | transforming growth factor beta |
| WG/WNT | wingless |
| ZF | zinc finger |
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tang, H.; Sun, X.; Reinberg, D.; Ebright, R.H. Protein–protein interactions in eukaryotic transcription initiation: Structure of the preinitiation complex. Proc. Natl. Acad. Sci. USA 1996, 93, 1119–1124. [Google Scholar] [CrossRef] [PubMed]
- Kuras, L.; Struhl, K. Binding of TBP to promoters in vivo is stimulated by activators and requires Pol II holoenzyme. Nature 1999, 399, 609–613. [Google Scholar] [CrossRef] [PubMed]
- Burley, S.K.; Roeder, R.G. Biochemistry and structural biology of transcription factor IID (TFIID). Annu. Rev. Biochem. 1996, 65, 769–799. [Google Scholar] [CrossRef] [PubMed]
- Dynlacht, B.D.; Hoey, T.; Tjian, R. Isolation of coactivators associated with the TATA-binding protein that mediate transcriptional activation. Cell 1991, 66, 563–576. [Google Scholar] [CrossRef] [PubMed]
- Cianfrocco, M.A.; Kassavetis, G.A.; Grob, P.; Fang, J.; Juven-Gershon, T.; Kadonaga, J.T.; Nogales, E. Human TFIID binds to core promoter DNA in a reorganized structural state. Cell 2013, 152, 120–131. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, A.; Oelgeschläger, T.; Roeder, R.G. Considerations of transcriptional control mechanisms: Do TFIID-core promoter complexes recapitulate nucleosome-like functions? Proc. Natl. Acad. Sci. USA 1997, 94, 8928–8935. [Google Scholar] [CrossRef]
- Wassarman, D.A.; Sauer, F. TAF(II)250: A transcription toolbox. J. Cell Sci. 2001, 114, 2895–2902. [Google Scholar] [PubMed]
- Ohtsuki, S.; Levine, M.; Cai, H.N. Different core promoters possess distinct regulatory activities in the Drosophila embryo. Genes Dev. 1998, 12, 547–556. [Google Scholar] [CrossRef] [PubMed]
- D’Alessio, J.A.; Wright, K.J.; Tjian, R. Shifting players and paradigms in cell-specific transcription. Mol. Cell 2009, 36, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Crowley, T.E.; Hoey, T.; Liu, J.K.; Jan, Y.N.; Jan, L.Y.; Tjian, R. A new factor related to TATA-binding protein has highly restricted expression patterns in Drosophila. Nature 1993, 361, 557–561. [Google Scholar] [CrossRef] [PubMed]
- Hansen, S.K.; Takada, S.; Jacobson, R.H.; Lis, J.T.; Tjian, R. Transcription properties of a cell type-specific TATA-binding protein, TRF. Cell 1997, 91, 71–83. [Google Scholar] [CrossRef] [PubMed]
- Rabenstein, M.D.; Zhou, S.; Lis, J.T.; Tjian, R. TATA box-binding protein (TBP)-related factor 2 (TRF2), a third member of the TBP family. Proc. Natl. Acad. Sci. USA 1999, 96, 4791–4796. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, E. Transcriptional functions of a new mammalian TATA-binding protein-related factor. J. Biol. Chem. 1999, 274, 12963–12966. [Google Scholar] [CrossRef] [PubMed]
- Dantonel, J.C.; Quintin, S.; Lakatos, L.; Labouesse, M.; Tora, L. TBP-like factor is required for embryonic RNA polymerase II transcription in C. elegans. Mol. Cell 2000, 6, 715–722. [Google Scholar] [CrossRef]
- Sugiura, S.; Kashiwabara, S.; Iwase, S.; Baba, T. Expression of a testis-specific form of TBP-related factor 2 (TRF2) mRNA during mouse spermatogenesis. J. Reprod. Dev. 2003, 49, 107–111. [Google Scholar] [CrossRef] [PubMed]
- Gazdag, E.; Rajkovic, A.; Torres-Padilla, M.E.; Tora, L. Analysis of TATA-binding protein 2 (TBP2) and TBP expression suggests different roles for the two proteins in regulation of gene expression during oogenesis and early mouse development. Reproduction 2007, 134, 51–62. [Google Scholar] [CrossRef] [PubMed]
- Jallow, Z.; Jacobi, U.G.; Weeks, D.L.; Dawid, I.B.; Veenstra, G.J.C. Specialized and redundant roles of TBP and a vertebrate-specific TBP paralog in embryonic gene regulation in Xenopus. Proc. Natl. Acad. Sci. USA 2004, 101, 13525–13530. [Google Scholar] [CrossRef] [PubMed]
- Hart, D.O.; Santra, M.K.; Raha, T.; Green, M.R. Selective interaction between Trf3 and Taf3 required for early development and hematopoiesis. Dev. Dyn. 2009, 238, 2540–2549. [Google Scholar] [CrossRef] [PubMed]
- Deato, M.D.E.; Marr., M.T.; Sottero, T.; Inouye, C.; Hu, P.; Tjian, R. MyoD targets TAF3/TRF3 to activate myogenin transcription. Mol. Cell 2008, 32, 96–105. [Google Scholar] [CrossRef] [PubMed]
- Hochheimer, A.; Tjian, R. Diversified transcription initiation complexes expand promoter selectivity and tissue-specific gene expression. Genes Dev. 2003, 17, 1309–1320. [Google Scholar] [CrossRef] [PubMed]
- Falender, A.E.; Freiman, R.N.; Geles, K.G.; Lo, K.C.; Hwang, K.; Lamb, D.J.; Morris, P.L.; Tjian, R,; Richards, J.S. Maintenance of spermatogenesis requires TAF4b, a gonad-specific subunit of TFIID. Genes Dev. 2005, 19, 794–803. [Google Scholar]
- Pointud, J.; Mengus, G.; Brancorsini, S.; Monaco, L.; Parvinen, M.; Sassone-Corsi, P.; Davidson, I. The intracellular localisation of TAF7L, a paralogue of transcription factor TFIID subunit TAF7, is developmentally regulated during male germ-cell differentiation. J. Cell Sci. 2003, 116, 1847–1858. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Kaplan, T.; Li, Y.; Grubisic, I.; Zhang, Z.; Wang, P.J.; Eisen, M.B.; Tjian, R. Dual functions of TAF7L in adipocyte differentiation. Elife 2013, 2, e00170. [Google Scholar] [PubMed]
- Freiman, R.N. Specific variants of general transcription factors regulate germ cell development in diverse organisms. Biochim. Biophys. Acta 2009, 1789, 161–166. [Google Scholar] [CrossRef] [PubMed]
- Ferg, M.; Sanges, R.; Gehrig, J.; Kiss, J.; Bauer, M.; Lovas, A.; Szabo, M.; Yang, L.; Straehle, U.; Pankratz, M.J.; et al. The TATA-binding protein regulates maternal mRNA degradation and differential zygotic transcription in zebrafish. EMBO J. 2007, 26, 3945–3956. [Google Scholar] [CrossRef] [PubMed]
- Walker, A.K.; Rothman, J.H.; Shi, Y.; Blackwell, T.K. Distinct requirements for C. elegans TAF(II)s in early embryonic transcription. EMBO J. 2001, 20, 5269–5279. [Google Scholar]
- Tadros, W.; Lipshitz, H.D. The maternal-to-zygotic transition: A play in two acts. Development 2009, 136, 3033–3042. [Google Scholar] [CrossRef] [PubMed]
- Tatarakis, A.; Margaritis, T.; Martinez-Jimenez, C.P.; Kouskouti, A.; Mohan, W.S., 2nd; Haroniti, A.; Kafetzopoulos, D.; Tora, L.; Talianidis, I. Dominant and redundant functions of TFIID involved in the regulation of hepatic genes. Mol. Cell 2008, 31, 531–543. [Google Scholar]
- Hart, D.O.; Raha, T.; Lawson, N.D.; Green, M.R. Initiation of zebrafish haematopoiesis by the TATA-box-binding protein-related factor Trf3. Nature 2007, 450, 1082–1085. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Scannell, D.R.; Eisen, M.B.; Tjian, R. Control of embryonic stem cell lineage commitment by core promoter factor, TAF3. Cell 2011, 146, 720–731. [Google Scholar] [CrossRef] [PubMed]
- Fadloun, A.; Kobi, D.; Delacroix, L.; Dembélé, D.; Michel, I.; Lardenois, A.; Tisserand, J.; Losson, R.; Mengus, G.; Davidson, I.; et al. Retinoic acid induces TGFβ-dependent autocrine fibroblast growth. Oncogene 2008, 27, 477–489. [Google Scholar] [CrossRef] [PubMed]
- Guermah, M.; Ge, K.; Chiang, C.M.; Roeder, R.G. The TBN protein, which is essential for early embryonic mouse development, is an inducible TAFII implicated in adipogenesis. Mol. Cell 2003, 12, 991–1001. [Google Scholar] [CrossRef] [PubMed]
- Goodrich, J.A.; Tjian, R. Unexpected roles for core promoter recognition factors in cell-type-specific transcription and gene regulation. Nat. Rev. Genet. 2010, 11, 549–558. [Google Scholar] [PubMed]
- Wata, J.; Hosokawa, R.; Sanchez-Lara, P.A.; Urata, M.; Slavkin, H.; Chai, Y. Transforming growth factor-beta regulates basal transcriptional regulatory machinery to control cell proliferation and differentiation in cranial neural crest-derived osteoprogenitor cells. J. Biol. Chem. 2010, 285, 4975–4982. [Google Scholar] [CrossRef] [PubMed]
- Hiller, M.A.; Lin, T.Y.; Wood, C.; Fuller, M.T. Developmental regulation of transcription by a tissue-specific TAF homolog. Genes Dev. 2001, 15, 1021–1030. [Google Scholar] [CrossRef] [PubMed]
- Hiller, M.; Chen, X.; Pringle, M.J.; Suchorolski, M.; Sancak, Y.; Viswanathan, S.; Bolival, B.; Lin, T.Y.; Marino, S.; Fuller, M.T.; et al. Testis-specific TAF homologs collaborate to control a tissue-specific transcription program. Development 2004, 131, 5297–5308. [Google Scholar] [CrossRef] [PubMed]
- Dikstein, R.; Zhou, S.; Tjian, R. Human TAFII 105 is a cell type-specific TFIID subunit related to hTAFII130. Cell 1996, 87, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Yang, F.; Wang, P.J. Regulation of male fertility by X-linked genes. J. Androl. 2010, 31, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Siderakis, M.; Tarsounas, M. Telomere regulation and function during meiosis. Chromosome Res. 2007, 15, 667–679. [Google Scholar] [CrossRef] [PubMed]
- Müller, F.; Tora, L. TBP2 is a general transcription factor specialized for female germ cells. J. Biol. 2009, 8, 97. [Google Scholar] [CrossRef]
- Müller, F.; Zaucker, A.; Tora, L. Developmental regulation of transcription initiation: More than just changing the actors. Curr. Opin. Genet. Dev. 2010, 20, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Chambers, I.; Tomlinson, S.R. The transcriptional foundation of pluripotency. Development 2009, 136, 2311–2322. [Google Scholar] [CrossRef] [PubMed]
- Baumann, K. Stem cells: TFIID promotes pluripotency. Nat. Rev. Mol. Cell Biol. 2013, 14, 264. [Google Scholar] [CrossRef] [PubMed]
- Van Nuland, R.; Schram, A.W.; van Schaik, F.M.A.; Jansen, P.W.T.C.; Vermeulen, M.; Marc Timmers, H.T. Multivalent engagement of TFIID to nucleosomes. PLoS One 2013, 8, e73495. [Google Scholar] [CrossRef] [PubMed]
- Ku, M.; Jaffe, J.D.; Koche, R.P.; Rheinbay, E.; Endoh, M.; Koseki, H.; Carr, S.A.; Bernstein, B.E. H2A. Z landscapes and dual modifications in pluripotent and multipotent stem cells underlie complex genome regulatory functions. Genome Biol. 2012, 13, R85. [Google Scholar]
- Pijnappel, W.W.M.P.; Esch, D.; Baltissen, M.P.A.; Wu, G.; Mischerikow, N.; Bergsma, A.J.; van der Wal, E.; Han, D.W.; Bruch, H.V.; Moritz, S.; et al. A central role for TFIID in the pluripotent transcription circuitry. Nature 2013, 495, 516–519. [Google Scholar] [CrossRef] [PubMed]
- Hoey, T.; Weinzierl, R.O.; Gill, G.; Chen, J.L.; Dynlacht, B.D.; Tjian, R. Molecular cloning and functional analysis of Drosophila TAF110 reveal properties expected of coactivators. Cell 1993, 72, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Ferreri, K.; Gill, G.; Montminy, M. The cAMP-regulated transcription factor CREB interacts with a component of the TFIID complex. Proc. Natl. Acad. Sci. USA 1994, 91, 1210–1213. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Liu, S.; Lausen, J.; Woodrell, C.; Cho, S.; Biris, N.; Kobayashi, N.; Wei, Y.; Yokoyama, S.; Werner, M.H.; et al. A TAF4-homology domain from the corepressor ETO is a docking platform for positive and negative regulators of transcription. Nat. Struct. Mol. Biol. 2007, 14, 653–661. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zhang, J.; Geng, H.; Du, Z.; Nakadai, T.; Roeder, R.G. A TAF4 coactivator function for E proteins that involves enhanced TFIID binding. Genes Dev. 2013, 27, 1596–1609. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Coleman, R.A.; Grob, P.; King, D.S.; Florens, L.; Washburn, M.P.; Geles, K.G.; Yang, J.L.; Ramey, V.; Nogales, E.; et al. Structural changes in TAF4b-TFIID correlate with promoter selectivity. Mol. Cell 2008, 29, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Mengus, G.; May, M.; Carré, L.; Chambon, P.; Davidson, I. Human TAF(II)135 potentiates transcriptional activation by the AF-2s of the retinoic acid, vitamin D3, and thyroid hormone receptors in mammalian cells. Genes Dev. 1997, 11, 1381–1395. [Google Scholar] [PubMed]
- Vassallo, M.F.; Tanese, N. Isoform-specific interaction of HP1 with human TAFII130. Proc. Natl. Acad. Sci. USA 2002, 99, 5919–5924. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Truckses, D.M.; Takada, S.; Matsumura, T.; Tanese, N.; Jacobson, R.H. Conserved region I of human coactivator TAF4 binds to a short hydrophobic motif present in transcriptional regulators. Proc. Natl. Acad. Sci. USA 2007, 104, 7839–7844. [Google Scholar] [CrossRef] [PubMed]
- Gazit, K.; Moshonov, S.; Elfakess, R.; Sharon, M.; Mengus, G.; Davidson, I.; Dikstein, R. TAF4/4b × TAF12 displays a unique mode of DNA binding and is required for core promoter function of a subset of genes. J. Biol. Chem. 2009, 284, 26286–26296. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.J.; Marr, M.T., 2nd; Tjian, R. TAF4 nucleates a core subcomplex of TFIID and mediates activated transcription from a TATA-less promoter. Proc. Natl. Acad. Sci. USA 2006, 103, 12347–12352. [Google Scholar]
- Guven-Ozkan, T.; Nishi, Y.; Robertson, S.M.; Lin, R. Global transcriptional repression in C. elegans germline precursors by regulated sequestration of TAF-4. Cell 2008, 135, 149–160. [Google Scholar]
- Mengus, G.; Fadloun, A.; Kobi, D.; Thibault, C.; Perletti, L.; Michel, I.; Davidson, I. TAF4 inactivation in embryonic fibroblasts activates TGFβ signalling and autocrine growth. EMBO J. 2005, 24, 2753–2767. [Google Scholar] [CrossRef] [PubMed]
- Fadloun, A.; Kobi, D.; Pointud, J.; Indra, A.K.; Teletin, M.; Bole-Feysot, C.; Testoni, B.; Mantovani, R.; Metzger, D.; Mengus, G.; et al. The TFIID subunit TAF4 regulates keratinocyte proliferation and has cell-autonomous and non-cell-autonomous tumour suppressor activity in mouse epidermis. Development 2007, 134, 2947–2958. [Google Scholar] [CrossRef] [PubMed]
- Perletti, L.; Kopf, E.; Carré, L.; Davidson, I. Coordinate regulation of RARγ2, TBP, and TAFII135 by targeted proteolysis during retinoic acid-induced differentiation of F9 embryonal carcinoma cells. BMC Mol. Biol. 2001, 2, 4. [Google Scholar] [CrossRef] [PubMed]
- Harris, T.M.; Childs, G. Global gene expression patterns during differentiation of F9 embryonal carcinoma cells into parietal endoderm. Funct. Integr. Genomics 2002, 2, 105–119. [Google Scholar] [CrossRef] [PubMed]
- Perletti, L.; Dantonel, J.C.; Davidson, I. The TATA-binding protein and its associated factors are differentially expressed in adult mouse tissues. J. Biol. Chem. 1999, 274, 15301–15304. [Google Scholar] [CrossRef] [PubMed]
- Deato, M.D.E.; Tjian, R. An unexpected role of TAFs and TRFs in skeletal muscle differentiation: Switching core promoter complexes. Cold Spring Harb. Symp. Quant. Biol. 2008, 73, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Wright, K.J.; Tjian, R. Wnt signaling targets ETO coactivation domain of TAF4/TFIID in vivo. Proc. Natl. Acad. Sci. USA 2009, 106, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Marr, M.T., 2nd. TAF4 takes flight. Proc. Natl. Acad. Sci. USA 2009, 106, 1295–1296. [Google Scholar] [CrossRef] [PubMed]
- Martianov, I.; Cler, E.; Duluc, I.; Vicaire, S.; Philipps, M.; Freund, J.; Davidson, I. TAF4 inactivation reveals the 3 dimensional growth promoting activities of collagen 6A3. PLoS One 2014, 9, e87365. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Xu, H.; Yuan, P.; Fang, F.; Huss, M.; Vega, V.B.; Wong, E.; Orlov, Y.L.; Zhang, W.; Jiang, J.; et al. Integration of external signaling pathways with the core transcriptional network in embryonic stem cells. Cell 2008, 133, 1106–1117. [Google Scholar] [PubMed]
- International Human Genome Sequencing Consortium. Finishing the euchromatic sequence of the human genome. Nature 2004, 431, 931–945. [Google Scholar]
- Castle, J.C.; Zhang, C.; Shah, J.K.; Kulkarni, A.V.; Kalsotra, A.; Cooper, T.A.; Johnson, J.M. Expression of 24,426 human alternative splicing events and predicted cis regulation in 48 tissues and cell lines. Nat. Genet. 2008, 40, 1416–1425. [Google Scholar] [CrossRef] [PubMed]
- Wang, E.T.; Sandberg, R.; Luo, S.; Khrebtukova, I.; Zhang, L.; Mayr, C.; Kingsmore, S.F.; Schroth, G.P.; Burge, C.B. Alternative isoform regulation in human tissue transcriptomes. Nature 2008, 456, 470–476. [Google Scholar] [CrossRef] [PubMed]
- Romero, P.R.; Zaidi, S.; Fang, Y.Y.; Uversky, V.N.; Radivojac, P.; Oldfield, C.J.; Cortese, M.S.; Sickmeier, M.; LeGall, T.; Obradovic, Z.; et al. Alternative splicing in concert with protein intrinsic disorder enables increased functional diversity in multicellular organisms. Proc. Natl. Acad. Sci. USA 2006, 103, 8390–8395. [Google Scholar] [CrossRef] [PubMed]
- Hegyi, H.; Kalmar, L.; Horvath, T.; Tompa, P. Verification of alternative splicing variants based on domain integrity, truncation length and intrinsic protein disorder. Nucleic Acids Res. 2011, 39, 1208–1219. [Google Scholar] [CrossRef] [PubMed]
- Taneri, B.; Snyder, B.; Novoradovsky, A.; Gaasterland, T. Alternative splicing of mouse transcription factors affects their DNA-binding domain architecture and is tissue specific. Genome Biol. 2004, 5, R75. [Google Scholar] [CrossRef] [PubMed]
- Talavera, D.; Orozco, M.; de la Cruz, X. Alternative splicing of transcription factors’ genes: Beyond the increase of proteome diversity. Comp. Funct. Genomics 2009, 2009, 905894. [Google Scholar] [CrossRef]
- Resch, A.; Xing, Y.; Modrek, B.; Gorlick, M.; Riley, R.; Lee, C. Assessing the impact of alternative splicing on domain interactions in the human proteome. J. Proteome Res. 2004, 3, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Kornblihtt, A.R.; de la Mata, M.; Fededa, J.P.; Munoz, M.J.; Nogues, G. Multiple links between transcription and splicing. RNA 2004, 10, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- Proudfoot, N.J.; Furger, A.; Dye, M.J. Integrating mRNA processing with transcription. Cell 2002, 108, 501–512. [Google Scholar] [CrossRef] [PubMed]
- Zeng, C.; Berget, S.M. Participation of the C-terminal domain of RNA polymerase II in exon definition during pre-mRNA splicing. Mol. Cell. Biol. 2000, 20, 8290–8301. [Google Scholar] [CrossRef] [PubMed]
- Cramer, P.; Pesce, C.G.; Baralle, F.E.; Kornblihtt, A.R. Functional association between promoter structure and transcript alternative splicing. Proc. Natl. Acad. Sci. USA 1997, 94, 11456–11460. [Google Scholar] [CrossRef] [PubMed]
- Nogues, G.; Kadener, S.; Cramer, P.; Bentley, D.; Kornblihtt, A.R. Transcriptional activators differ in their abilities to control alternative splicing. J. Biol. Chem. 2002, 277, 43110–43114. [Google Scholar] [CrossRef] [PubMed]
- Auboeuf, D.; Hönig, A.; Berget, S.M.; O’Malley, B.W. Coordinate regulation of transcription and splicing by steroid receptor coregulators. Science 2002, 298, 416–419. [Google Scholar] [CrossRef] [PubMed]
- Alló, M.; Buggiano, V.; Fededa, J.P.; Petrillo, E.; Schor, I.; de la Mata, M.; Agirre, E.; Plass, M.; Eyras, E.; Elela, S.A.; et al. Control of alternative splicing through siRNA-mediated transcriptional gene silencing. Nat. Struct. Mol. Biol. 2009, 16, 717–724. [Google Scholar] [CrossRef] [PubMed]
- Kisielow, M.; Kleiner, S.; Nagasawa, M.; Faisal, A.; Nagamine, Y. Isoform-specific knockdown and expression of adaptor protein ShcA using small interfering RNA. Biochem. J. 2002, 363, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Prinos, P.; Garneau, D.; Lucier, J.; Gendron, D.; Couture, S.; Boivin, M.; Brosseau, J.-P.; Lapointe, E.; Thibault, P.; Durand, M.; et al. Alternative splicing of SYK regulates mitosis and cell survival. Nat. Struct. Mol. Biol. 2011, 18, 673–679. [Google Scholar] [CrossRef] [PubMed]
- Goldberg, M.S.; Sharp, P.A. Pyruvate kinase M2-specific siRNA induces apoptosis and tumor regression. J. Exp. Med. 2012, 209, 217–224. [Google Scholar] [CrossRef] [PubMed]
- Brunkhorst, A.; Neuman, T.; Hall, A.; Arenas, E.; Bartfai, T.; Hermanson, O.; Metsis, M. Novel isoforms of the TFIID subunit TAF4 modulate nuclear receptor-mediated transcriptional activity. Biochem. Biophys. Res. Commun. 2004, 325, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Kazantseva, J.; Kivil, A.; Tints, K.; Kazantseva, A.; Neuman, T.; Palm, K. Alternative splicing targeting the hTAF4–TAFH domain of TAF4 represses proliferation and accelerates chondrogenic differentiation of human mesenchymal stem cells. PLoS One 2013, 8, e74799. [Google Scholar] [CrossRef] [PubMed]
- Kazantseva, J.; Tints, K.; Neuman, T.; Palm, K. TAF4 controls differentiation of human neural progenitor cells through hTAF4–TAFH activity. J. Mol. Neurosci. 2014, in press. [Google Scholar]
- Topol, L.; Jiang, X.; Choi, H.; Garrett-Beal, L.; Carolan, P.J.; Yang, Y. Wnt-5a inhibits the canonical Wnt pathway by promoting GSK-3-independent β-catenin degradation. J. Cell. Biol. 2003, 162, 899–908. [Google Scholar] [CrossRef] [PubMed]
- He, F.; Xiong, W.; Yu, X.; Espinoza-Lewis, R.; Liu, C.; Gu, S.; Nishita, M.; Suzuki, K.; Yamada, G.; Minami, Y.; et al. Wnt5a regulates directional cell migration and cell proliferation via Ror2-mediated noncanonical pathway in mammalian palate development. Development 2008, 135, 3871–3879. [Google Scholar] [CrossRef] [PubMed]
- Niehrs, C. Function and biological roles of the Dickkopf family of Wnt modulators. Oncogene 2006, 25, 7469–7481. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, J.F.; Kaufman, M.H.; Harrison, D.J.; Clarke, A.R. High-frequency developmental abnormalities in p53-deficient mice. Curr. Biol. 1995, 5, 931–936. [Google Scholar] [CrossRef] [PubMed]
- Kazantseva, J.; Sadam, H.; Neuman, T.; Palm, K. Migration and multipotency in melanoma under control of TAF4 alternative splicing. 2014; submitted. [Google Scholar]
- Mani, S.A.; Guo, W.; Liao, M.; Eaton, E.N.; Ayyanan, A.; Zhou, A.Y.; Brooks, M.; Reinhard, F.; Zhang, C.C.; Shipitsin, M.; et al. The epithelial-mesenchymal transition generates cells with properties of stem cells. Cell 2008, 133, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Quante, M.; Tu, S.P.; Tomita, H.; Gonda, T.; Wang, S.S.W.; Takashi, S.; Baik, G.H.; Shibata, W.; Diprete, B.; Betz, K.S.; et al. Bone marrow-derived myofibroblasts contribute to the mesenchymal stem cell niche and promote tumor growth. Cancer Cell 2011, 19, 257–272. [Google Scholar] [CrossRef] [PubMed]
- Hendrix, M.J.C.; Seftor, E.A.; Hess, A.R.; Seftor, R.E.B. Molecular plasticity of human melanoma cells. Oncogene 2003, 22, 3070–3075. [Google Scholar] [CrossRef] [PubMed]
- Fang, D.; Nguyen, T.K.; Leishear, K.; Finko, R.; Kulp, A.N.; Hotz, S.; van Belle, P.A.; Xu, X.; Elder, D.E.; Herlyn, M.; et al. A tumorigenic subpopulation with stem cell properties in melanomas. Cancer Res. 2005, 65, 9328–9337. [Google Scholar] [CrossRef] [PubMed]
- Na, Y.R.; Seok, S.H.; Kim, D.J.; Han, J.H.; Kim, T.H.; Jung, H.; Lee, B.H.; Park, J.H. Isolation and characterization of spheroid cells from human malignant melanoma cell line WM-266-4. Tumour Biol. 2009, 30, 300–309. [Google Scholar] [CrossRef] [PubMed]
- Pittenger, M.F.; Martin, B.J. Mesenchymal stem cells and their potential as cardiac therapeutics. Circ. Res. 2004, 95, 9–20. [Google Scholar] [CrossRef] [PubMed]
- Balkwill, F. Cancer and the chemokine network. Nat. Rev. Cancer 2004, 4, 540–550. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kazantseva, J.; Palm, K. Diversity in TAF Proteomics: Consequences for Cellular Differentiation and Migration. Int. J. Mol. Sci. 2014, 15, 16680-16697. https://doi.org/10.3390/ijms150916680
Kazantseva J, Palm K. Diversity in TAF Proteomics: Consequences for Cellular Differentiation and Migration. International Journal of Molecular Sciences. 2014; 15(9):16680-16697. https://doi.org/10.3390/ijms150916680
Chicago/Turabian StyleKazantseva, Jekaterina, and Kaia Palm. 2014. "Diversity in TAF Proteomics: Consequences for Cellular Differentiation and Migration" International Journal of Molecular Sciences 15, no. 9: 16680-16697. https://doi.org/10.3390/ijms150916680
APA StyleKazantseva, J., & Palm, K. (2014). Diversity in TAF Proteomics: Consequences for Cellular Differentiation and Migration. International Journal of Molecular Sciences, 15(9), 16680-16697. https://doi.org/10.3390/ijms150916680



