Development of Laser Ionization Techniques for Evaluation of the Effect of Cancer Drugs Using Imaging Mass Spectrometry
Abstract
:1. Introduction
2. Results and Discussion
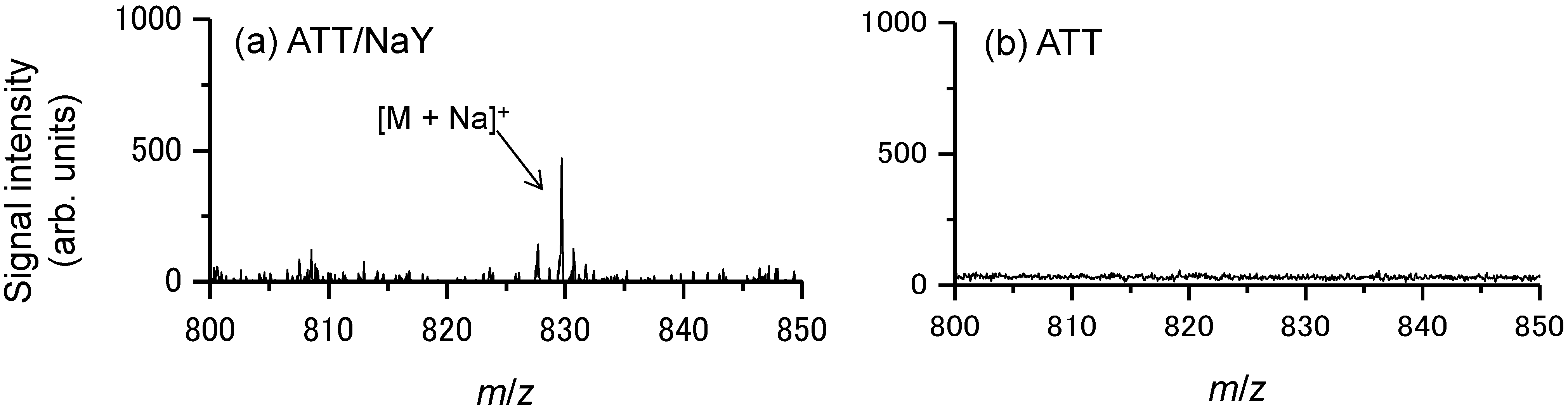
2.1. Ionization of Docetaxel Using Zeolite Matrix

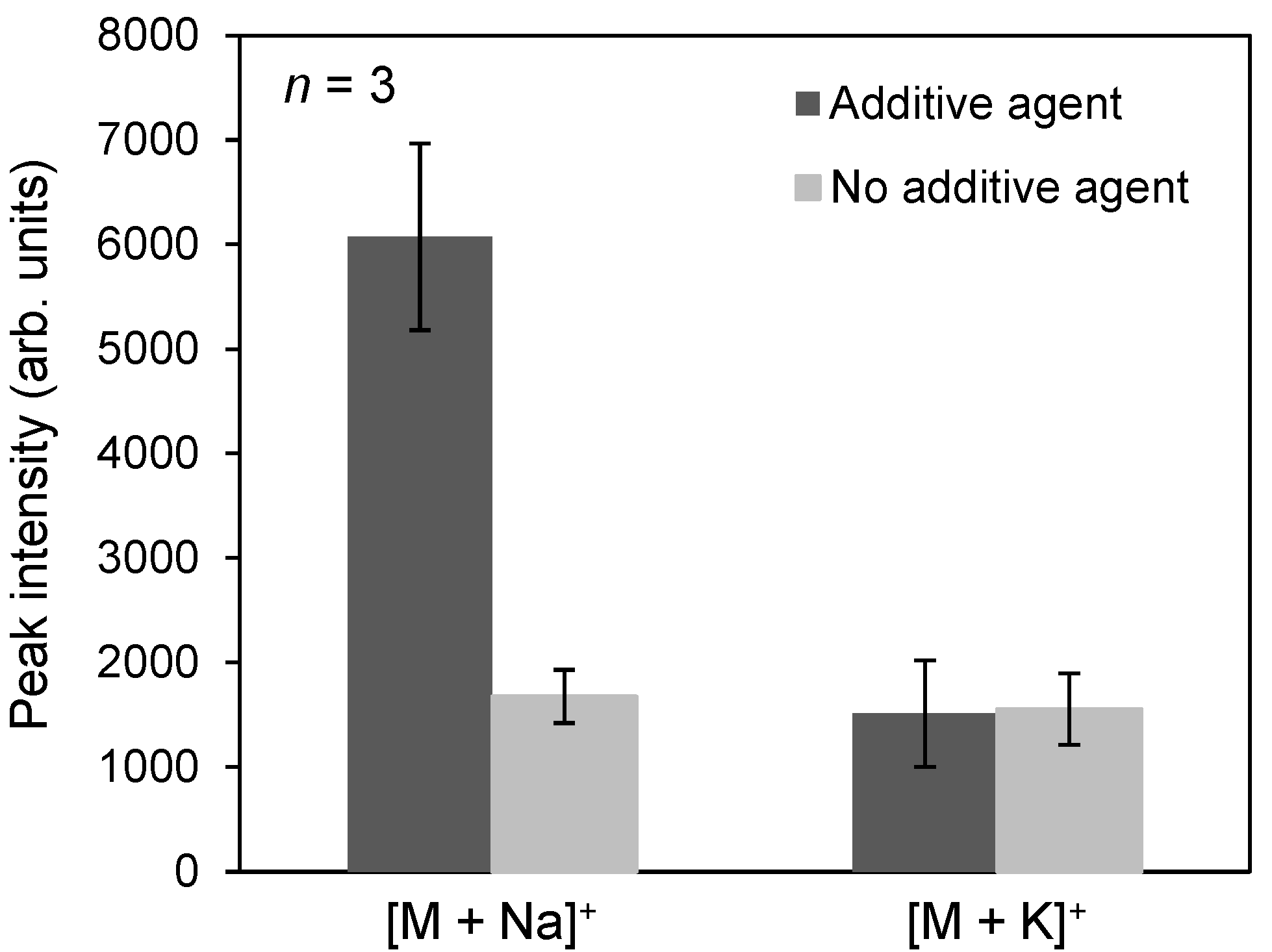

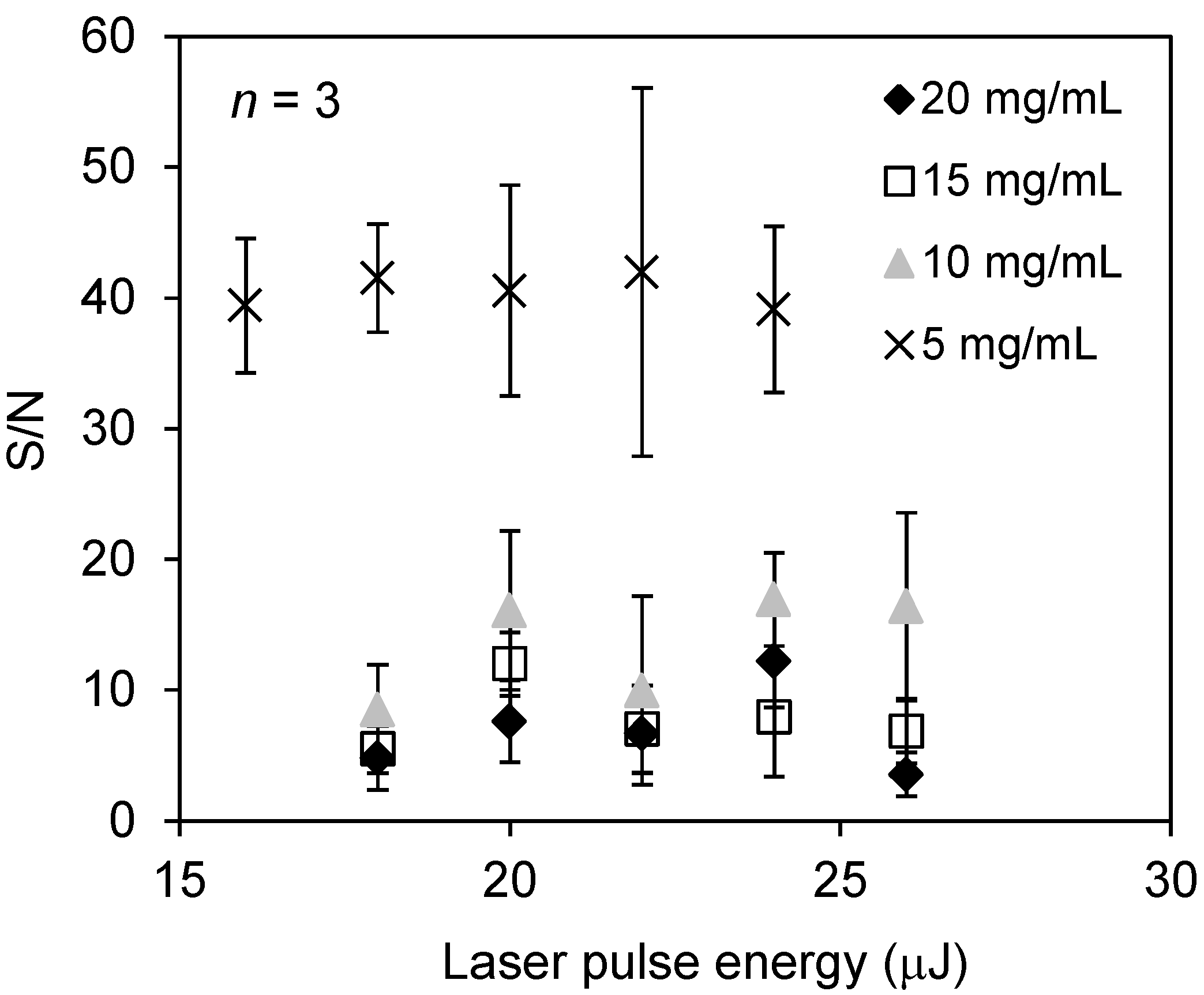

2.2. Detection of Docetaxel Administered to PC-3 Cells

2.3. Imaging of Cancer Drugs Administered to PC-3 Cells

2.4. Imaging of PpIX Administered to PC-3 Cells

3. Experimental Section
3.1. Chemical and Reagents
3.2. Cell Culture
3.3. Mass Spectrometer
3.4. Sample Preparation
3.5. Drug Administrations into Cells
3.6. Microscopy
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kaufmann, S.; Vaux, D. Alterations in the apoptotic machinery and their potential role in anticancer drug resistance. Oncogene 2003, 23, 7414–7430. [Google Scholar] [CrossRef]
- Patterson, S.G.; WEI, S.; Chen, X.; Sallman, D.A.; Gilvary, D.L.; Zhong, B.; Pow-Sang, J.; Yeatman, T.; Djeu, J.Y. Novel role of Stat1 in development of docetaxel resistance in prostate tumor cells. Oncogene 2006, 25, 6112–6122. [Google Scholar]
- The Interview Form of Taxotere® (Docetaxel for Injec-tion, Sanofi-Aventis). Available online: http://www.sec.gov/Archives/edgar/data/1121404/000119312511050947/d20f.htm (accessed on 16 June 2014).
- Thompson, M.S.; Jonansson, T.; Andersson-Engels, S.; Svanberg, S.; Bendsoe, N.; Svanberg, K. Clinical system for interstitial photodynamic therapy with combined on-line dosimetry measurements. Appl. Opt. 2005, 44, 4023–4031. [Google Scholar] [CrossRef]
- Yamauchi, M.; Honda, N.; Hazama, H.; Tachikawa, S.; Nakamura, H.; Kaneda, Y.; Awazu, K. A novel photodynamic therapy for drug-resistant prostate cancer cells using porphyrus envelope as a novel photosensitizer. Photodiagn. Photodyn. Ther. 2014, 11, 48–54. [Google Scholar] [CrossRef]
- Zuluaga, M.F. Combination of photodynamic therapy with anti-cancer agents. Curr. Med. Chem. 2008, 15, 1655–1673. [Google Scholar] [CrossRef]
- Sinha, A.; Anand, S.; Ortel, B.; Chang, Y.; Mai, Z.; Hasan, T.; Maytin, E. Methotrexate used in combination with aminolaevulinic acid for photodynamic killing of prostate cancer cells. Br. J. Cancer 2006, 95, 485–495. [Google Scholar] [CrossRef]
- Hsieh, Y.; Casale, R.; Fukuda, E.; Chen, J.; Knemeyer, I.; Wingate, J.; Morrison, R.; Korfmacher, W. Matrix-assisted laser desorption/ionization imaging mass spectrometry for direct measurement of clozapine in rat brain tissue. Rapid Commun. Mass Spectrom. 2006, 20, 965–972. [Google Scholar] [CrossRef]
- Russell, D.H.; Edmondson, R.D. High-resolution mass spectrometry and accurate mass measurements with emphasis on the characterization of peptides and proteins by matrix-assisted laser desorption/ionization time-of-flight mass spectrometry. J. Mass Spectrom. 1997, 32, 263–276. [Google Scholar] [CrossRef]
- Gusev, A.I.; Wilkinson, W.R.; Proctor, A.; Hercules, D.M. Improvement of signal reproducibility and matrix/comatrix effects in MALDI analysis. Anal. Chem. 1995, 67, 1034–1041. [Google Scholar] [CrossRef]
- Billeci, T.M.; Stults, J.T. Tryptic mapping of recombinant proteins by matrix-assisted laser desorption/ionization mass spectrometry. Anal. Chem. 1993, 65, 1709–1716. [Google Scholar] [CrossRef]
- Cohen, S.L.; Chait, B.T. Influence of matrix solution conditions on the MALDI-MS analysis of peptides and proteins. Anal. Chem. 1996, 68, 31–37. [Google Scholar] [CrossRef]
- Duncan, M.W.; Matanovic, G.; Cerpa-Poljak, A. Quantitative analysis of low molecular weight compounds of biological interest by matrix-assisted laser desorption ionization. Rapid Commun. Mass Spectrom. 1993, 7, 1090–1094. [Google Scholar] [CrossRef]
- Komori, Y.; Shima, H.; Fujino, T.; Kondo, J.N.; Hashimoto, K.; Korenaga, T. Pronounced selectivity in matrix-assisted laser desorption-ionization mass spectrometry with 2,4,6-trihydroxyacetophenone on a zeolite surface: Intensity enhancement of protonated peptides and suppression of matrix-related ions. J. Phys. Chem. 2010, 114, 1593–1600. [Google Scholar]
- Hazama, H.; Aoki, J.; Nagao, H.; Suzuki, R.; Tashima, T.; Fujii, K.; Masuda, K.; Awazu, K.; Toyoda, M.; Naito, Y. Construction of a novel stigmatic MALDI imaging mass spectrometer. Appl. Surf. Sci. 2008, 255, 1257–1263. [Google Scholar] [CrossRef]
- Aoki, J.; Ikeda, S.; Toyoda, M. Observation of accumulated metal cation distribution in fish by novel stigmatic imaging time-of-flight mass spectrometer. J. Phys. Soc. Jpn. 2014, 83. [Google Scholar] [CrossRef]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kannen, H.; Hazama, H.; Kaneda, Y.; Fujino, T.; Awazu, K. Development of Laser Ionization Techniques for Evaluation of the Effect of Cancer Drugs Using Imaging Mass Spectrometry. Int. J. Mol. Sci. 2014, 15, 11234-11244. https://doi.org/10.3390/ijms150711234
Kannen H, Hazama H, Kaneda Y, Fujino T, Awazu K. Development of Laser Ionization Techniques for Evaluation of the Effect of Cancer Drugs Using Imaging Mass Spectrometry. International Journal of Molecular Sciences. 2014; 15(7):11234-11244. https://doi.org/10.3390/ijms150711234
Chicago/Turabian StyleKannen, Hiroki, Hisanao Hazama, Yasufumi Kaneda, Tatsuya Fujino, and Kunio Awazu. 2014. "Development of Laser Ionization Techniques for Evaluation of the Effect of Cancer Drugs Using Imaging Mass Spectrometry" International Journal of Molecular Sciences 15, no. 7: 11234-11244. https://doi.org/10.3390/ijms150711234
APA StyleKannen, H., Hazama, H., Kaneda, Y., Fujino, T., & Awazu, K. (2014). Development of Laser Ionization Techniques for Evaluation of the Effect of Cancer Drugs Using Imaging Mass Spectrometry. International Journal of Molecular Sciences, 15(7), 11234-11244. https://doi.org/10.3390/ijms150711234



