The Cytoprotective Effect of Sulfuretin against tert-Butyl Hydroperoxide-Induced Hepatotoxicity through Nrf2/ARE and JNK/ERK MAPK-Mediated Heme Oxygenase-1 Expression
Abstract
:1. Introduction
2. Results and Discussion
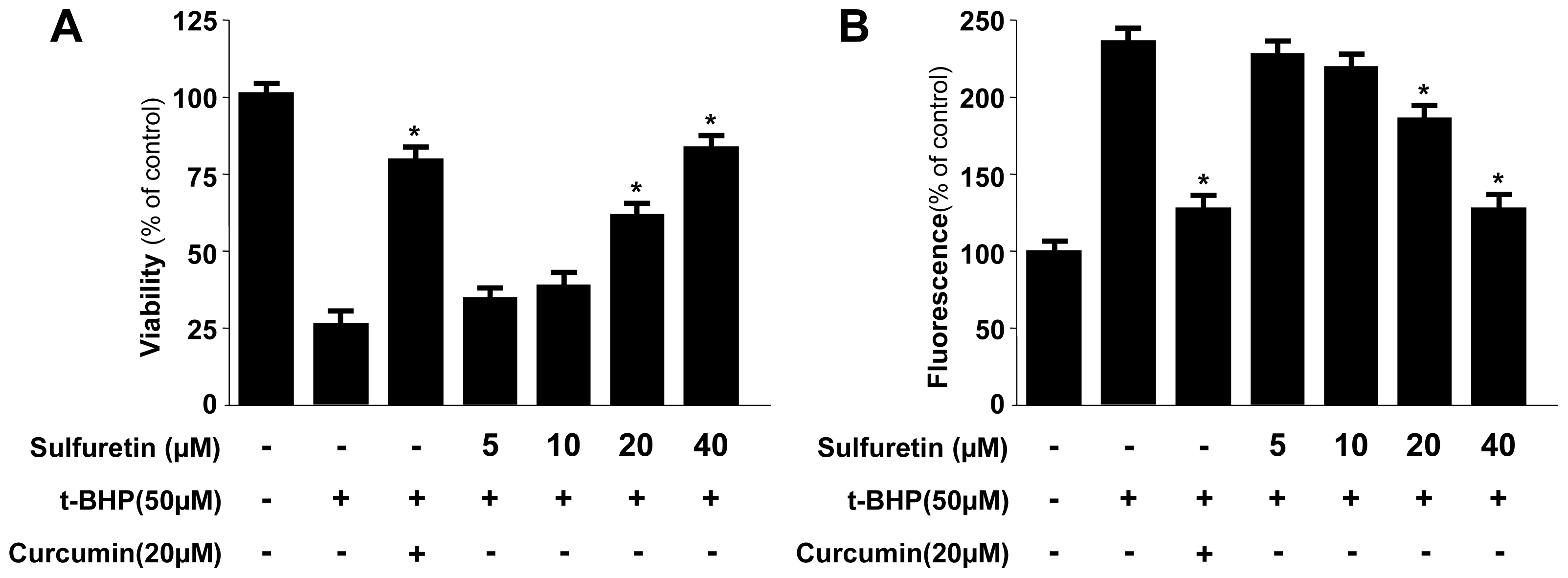
2.1. Effects of Sulfuretin on the Viability of Human Liver-Derived HepG2 Cells
2.2. Protective Effects of Sulfuretin on t-BHP-Induced Oxidative Injury and ROS Generation in Human Liver-Derived HepG2 Cells
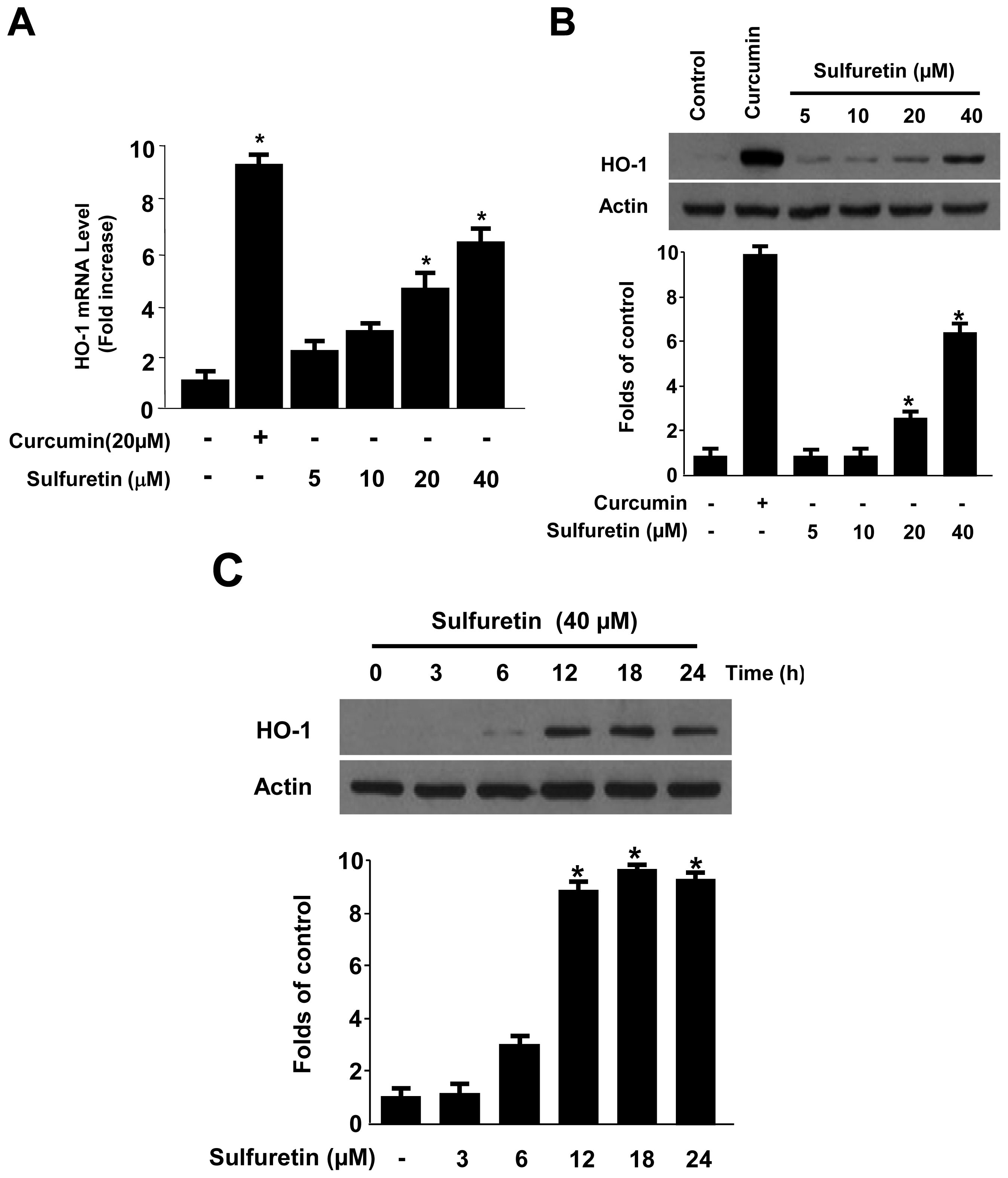
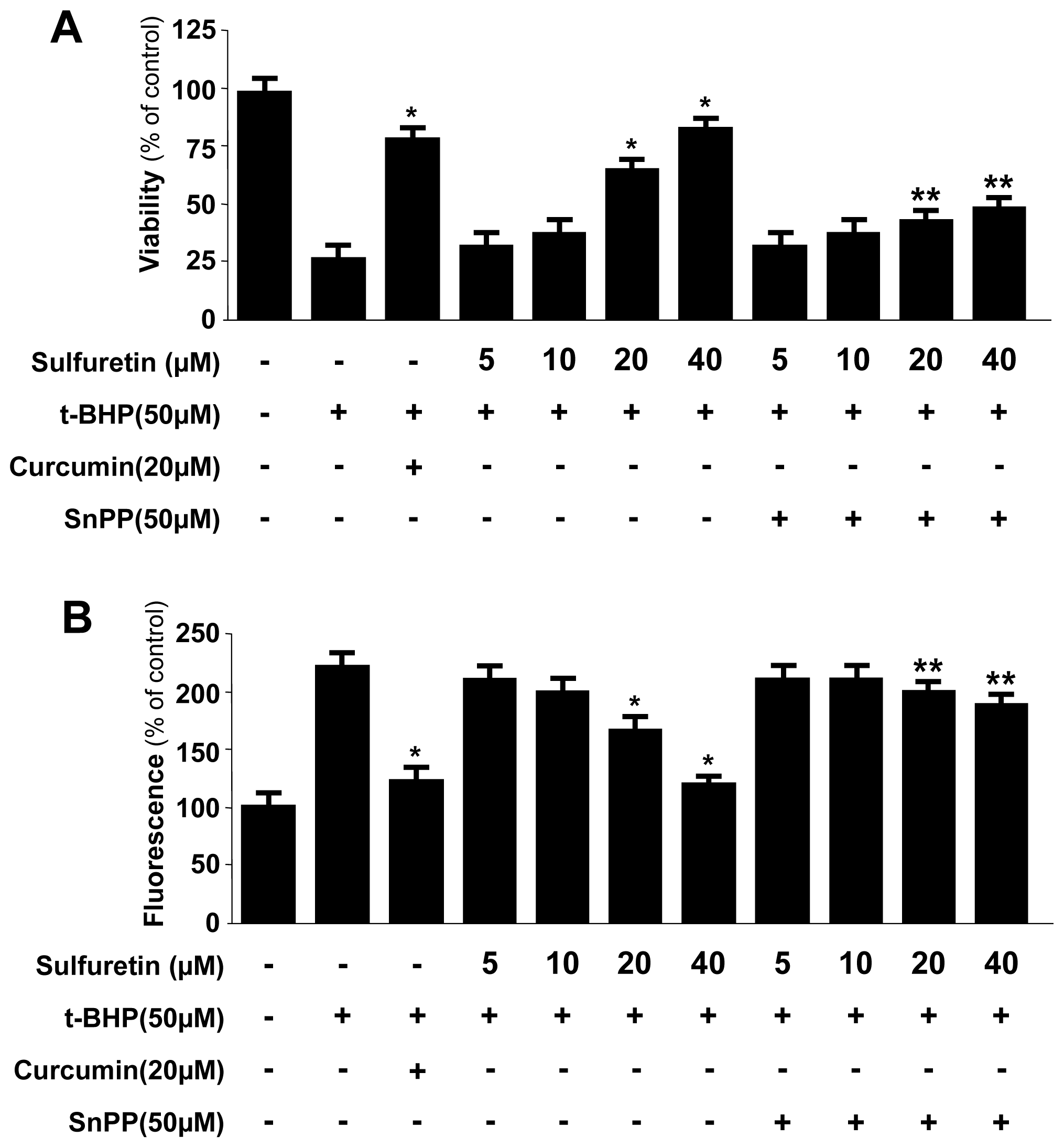
2.3. Effects of Sulfuretin-Induced HO-1 Expression on t-BHP-Induced Oxidative Injury and ROS Generation in Human Liver-Derived HepG2 Cells
2.4. Effects of Sulfuretin on HO-1 Expression through Nuclear Translocation of Nrf2 in Human Liver-Derived HepG2 Cells
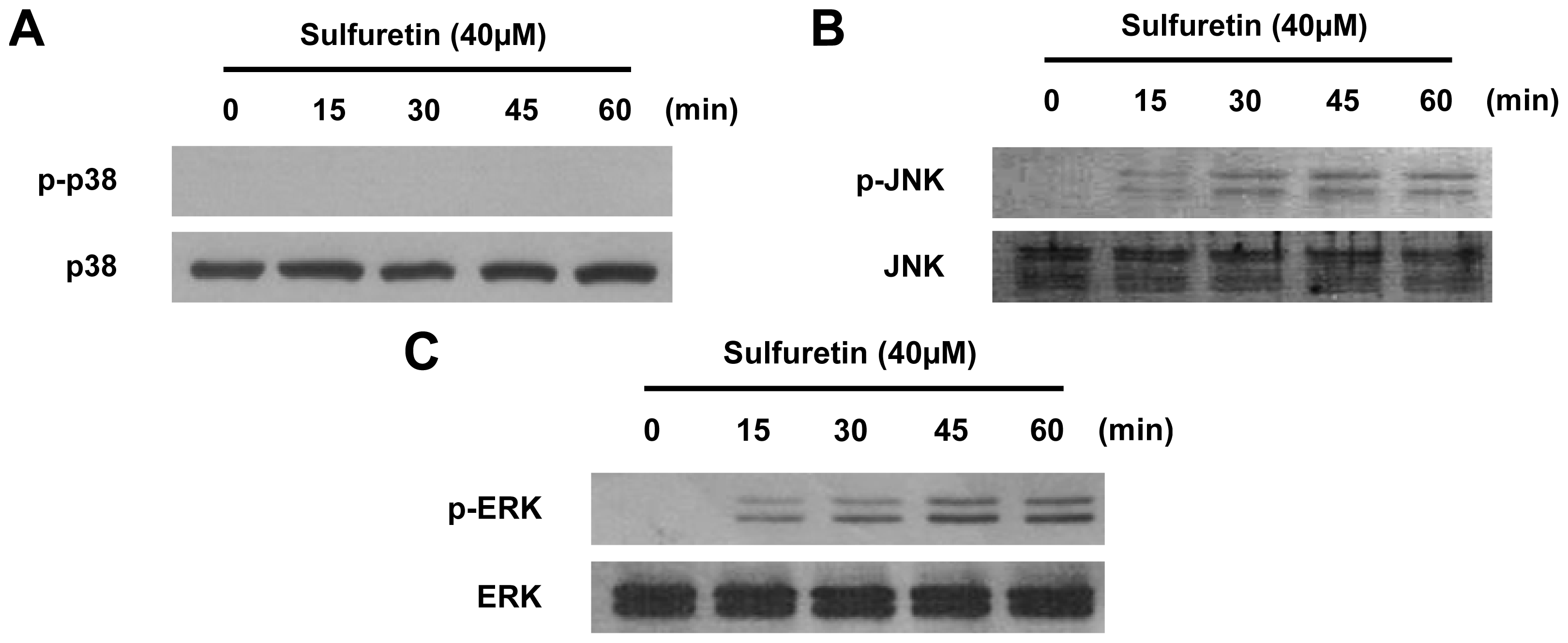
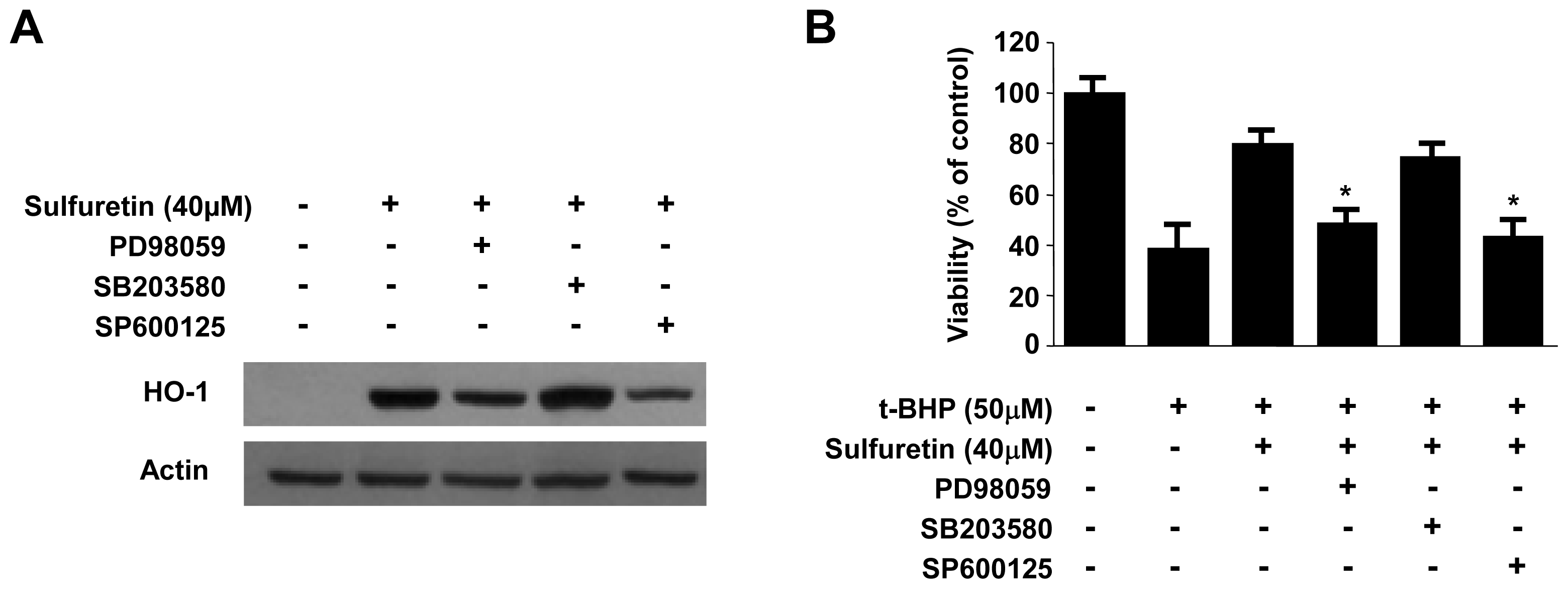
2.5. Involvement of the MAPK Pathways in Sulfuretin-Induced HO-1 Expression
3. Experimental Section
3.1. Materials
3.2. Cell Viability Assay
3.3. Preparation of Nuclear and Cytosolic Fractions
3.4. Western Blot Analysis
3.5. ROS Measurement
3.6. ARE Promoter Activity
3.7. Real-Time PCR
3.8. Transfection
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Loguercio, C.; Federico, A. Oxidative stress in viral and alcoholic hepatitis. Free Radic. Biol. Med 2003, 34, 1–10. [Google Scholar]
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Ciustarini, D.; Milzani, A. Biomarkers of oxidative damage in human disease. Clin. Chem 2006, 52, 601–623. [Google Scholar]
- Vitaglione, P.; Morisco, F.; Caporaso, N.; Fogliano, V. Dietary antioxidant compounds and liver health. Crit. Rev. Food Sci. Nutr 2004, 44, 575–586. [Google Scholar]
- Montellano, P.R. The mechanism of heme oxygenase. Curr. Opin. Chem. Biol 2000, 4, 221–227. [Google Scholar]
- Ryter, S.W.; Otterbein, L.E.; Morse, D.; Choi, A.M.K. Heme oxygenase/carbon monoxide signaling pathways: Regulation and functional significance. Mol. Cell Biochem 2002, 37, 249–263. [Google Scholar]
- Baird, L.; Dinkova-Kostova, A.T. The cytoprotective role of the Keap1–Nrf2 pathway. Arch. Toxicol 2011, 85, 241–272. [Google Scholar]
- Tkachev, V.O.; Menshchikova, E.B.; Zenkov, N.K. Mechanism of the Nrf2/Keap1/ARE signaling system. Biochemistry 2011, 76, 407–422. [Google Scholar]
- Kensler, T.W.; Wakabayashi, N.; Biswal, S. Cell survival responses to environmental stresses via the Keap1–Nrf2–ARE pathway. Annu. Rev. Pharmacol. Toxicol 2007, 47, 89–116. [Google Scholar]
- Kaspar, J.W.; Niture, S.K.; Jaiswal, A.K. Nrf2:INrf2 (Keap1) signaling in oxidative stress. Free Radic. Biol. Med 2009, 47, 1304–1309. [Google Scholar]
- Numazawa, S.; Ishikawa, M.; Yoshida, A.; Tanaka, S.; Yoshida, T. Atypical protein kinase C mediates activation of NF-E2-related factor 2 in response to oxidative stress. Am. J. Physiol. Cell Physiol 2003, 285, C334–C342. [Google Scholar]
- Liu, Y.; Shepherd, E.G.; Nelin, L.D. MAPK phosphatases—Regulating the immune response. Nat. Rev. Immunol 2007, 7, 202–212. [Google Scholar]
- Immenschuh, S.; Ramadori, G. Gene regulation of heme oxygenase-1 as a therapeutic target. Biochem. Pharmacol 2000, 60, 1121–1128. [Google Scholar]
- Zhang, X.; Bedard, E.L.; Potter, R.; Zhong, R.; Alam, J.; Choi, A.M.; Lee, P.J. Mitogen-activated protein kinases regulate HO-1 gene transcription after ischemia-reperfusion lung injury. Am. J. Physiol. Lung Cell Mol. Physiol 2002, 283, 815–829. [Google Scholar]
- Knasmuller, S.; Parzefall, W.; Sanyal, R.; Ecker, S.; Schwab, C.; Uhl, M.; Mersch-Sundermann, V.; Williamson, G.; Hietsch, G.; Langer, T.; et al. Use of metabolically competent human hepatoma cells for the detection of mutagens and antimutagens. Mutat. Res 1998, 402, 185–202. [Google Scholar]
- Alia, M.; Ramos, S.; Mateos, R.; Granado-Serrano, A.B.; Bravo, L.; Goya, L. Quercetin protects human hepatoma HepG2 against oxidative stress induced by tert-butyl hydroperoxide. Toxicol. Appl. Pharmacol 2006, 212, 110–118. [Google Scholar]
- Lee, H.U.; Bae, E.A.; Han, M.J.; Kim, N.J.; Kim, D.H. Hepatoprotective effect of ginsenoside Rb1 and compound K on tert-butyl hydroperoxide induced liver injury. Liver Int 2005, 25, 1069–1073. [Google Scholar]
- Martin, C.; Martinez, R.; Navarro, R.; Ruiz-Sanz, J.I.; Lacort, M.; Ruiz-Larrea, M.B. tert-Butyl hydroperoxide-induced lipid signaling in hepatocytes: Involvement of glutathione and free radicals. Biochem. Pharmacol 2001, 62, 705–712. [Google Scholar]
- Hix, S.; Kadiiska, M.B.; Mason, R.P.; Augusto, O. In vivo metabolism of tert-butyl hydroperoxide to methyl radicals: EPR spin-trapping and DNA methylation studies. Chem. Res. Toxicol 2000, 13, 1056–1064. [Google Scholar]
- Lee, J.C.; Lee, K.Y.; Kim, J.; Na, C.S.; Jung, N.C.; Chung, G.H.; Jang, Y.S. Extract from Rhus verniciflua Stokes is capable of inhibiting the growth of human lymphoma cells. Food Chem. Toxicol 2004, 42, 1383–1388. [Google Scholar]
- Lee, J.C.; Kim, J.; Lim, K.T.; Yang, M.S.; Jang, Y.S. Ethanol eluted extract of Rhus verniciflua Stokes showed both antioxidant and cytotoxic effects on mouse thymocytes depending on the dose and time of the treatment. Biochem. Mol. Biol 2001, 34, 250–258. [Google Scholar]
- Lim, K.T.; Hu, C.; Kitts, D.D. Antioxidant activity of a Rhus verniciflua Stokes ethanol extract. Food Chem. Toxicol 2001, 39, 229–237. [Google Scholar]
- Jeon, W.K.; Kim, J.H.; Lee, A.Y.; Kim, H.K. Inhibition of whole blood platelet aggregation from traditional medicines. Korean J. Orient. Med 2003, 9, 55–67. [Google Scholar]
- Kim, I.T.; Park, Y.M.; Shin, K.M.; Ha, J.; Choi, J.; Jung, H.J.; Park, H.J.; Lee, K.T. Anti-inflammatory and anti-nociceptive effects of the extract from Kalopanax pictus, Pueraria thunbergiana and Rhus verniciflua. J. Ethnopharmacol 2004, 94, 165–173. [Google Scholar]
- Park, K.Y.; Jung, G.O.; Lee, K.T.; Choi, J.; Choi, M.Y.; Kim, G.T.; Jung, H.J.; Park, H.J. Antimutagenic activity of flavonoids from the heartwood of Rhus verniciflua. J. Ethnopharmacol 2004, 90, 73–79. [Google Scholar]
- Lee, J.C.; Lim, K.T.; Jang, Y.S. Identification of Rhus verniciflua Stokes compounds that exhibit free radical scavenging and anti-apoptotic properties. Biochim. Biophys. Acta 2002, 1570, 181–191. [Google Scholar]
- Son, Y.O.; Lee, K.Y.; Lee, J.C.; Jang, H.S.; Kim, J.G.; Jeon, Y.M.; Jang, Y.S. Selective antiproliferative and apoptotic effects of flavonoids purified from Rhus verniciflua Stokes on normal versus transformed hepatic cell lines. Toxicol. Lett 2005, 155, 115–125. [Google Scholar]
- Ko, J.H.; Lee, S.J.; Lim, K.T. 36-kDa glycoprotein isolated from Rhus verniciflua Stokes fruit has a protective activity to glucose/glucose oxidase-induced apoptosis in NIH/3T3 cells. Toxicol. in Vitro 2005, 19, 353–363. [Google Scholar]
- Lee, K.W.; Chung, K.S.; Seo, J.H.; Yim, S.V.; Park, H.J.; Choi, J.H.; Lee, K.T. Sulfuretin from heartwood of Rhus verniciflua triggers apoptosis through activation of Fas, Caspase-8, and the mitochondrial death pathway in HL-60 human leukemia cells. J. Cell Biochem 2012, 113, 2835–2844. [Google Scholar]
- Shin, J.S.; Park, Y.M.; Choi, J.H.; Park, H.J.; Shin, M.C.; Lee, Y.S.; Lee, K.T. Sulfuretin isolated from heartwood of Rhus verniciflua inhibits LPS-induced inducible nitric oxide synthase, cyclooxygenase-2, and pro-inflammatory cytokines expression via the down-regulation of NF-κB in RAW 264.7 murine macrophage cells. Int. Immunopharmacol 2010, 10, 943–950. [Google Scholar]
- Jeon, W.K.; Lee, J.H.; Kim, H.K.; Lee, A.Y.; Lee, S.O.; Kim, Y.S.; Ryu, S.Y.; Kim, S.Y.; Lee, Y.J.; Ko, B.S. Anti-platelet effects of bioactive compounds isolated from the bark of Rhus verniciflua Stokes. J. Ethnopharmacol 2006, 106, 62–69. [Google Scholar]
- Choi, J.; Yoon, B.J.; Han, Y.N.; Lee, K.T.; Ha, J.; Jung, H.J.; Park, H.J. Antirheumatoid arthritis effect of Rhus verniciflua and of the active component, sulfuretin. Planta Med 2003, 69, 899–904. [Google Scholar]
- Lee, D.S.; Jeong, G.S.; Li, B.; Park, H.; Kim, Y.C. Anti-inflammatory effects of sulfuretin from Rhus verniciflua Stokes via the induction of heme oxygenase-1 expression in murine macrophages. Int. Immunopharmacol 2010, 10, 850–858. [Google Scholar]
- Thabrew, M.I.; Hughes, R.D.; McFarlane, I.G. Screening of hepatoprotective plant components using a HepG2 cell cytotoxicity assay. J. Pharm. Pharmacol 1997, 49, 1132–1135. [Google Scholar]
- Morse, D.; Choi, A.M. Heme oxygenase-1: The “emerging molecule” has arrived. Am. J. Respir. Cell Mol. Biol 2002, 27, 8–16. [Google Scholar]
- Ghattas, M.H.; Chuang, L.T.; Kappas, A.; Abraham, N.G. Protective effect of HO-1 against oxidative stress in human hepatoma cell line (HepG2) is independent of telomerase enzyme activity. Int. J. Biochem. Cell Biol 2002, 34, 1619–1628. [Google Scholar]
- Jeong, G.S.; Pae, H.O.; Jeong, S.O.; Kim, Y.C.; Kwon, T.O.; Lee, H.S.; Kim, N.S.; Park, S.D.; Chung, H.T. The α-methylene-γ-butyrolactone moiety in dehydrocostus lactone is responsible for cytoprotective heme oxygenase-1 expression through activation of the nuclear factor E2-related factor 2 in HepG2 cells. Eur. J. Pharmacol 2007, 565, 37–44. [Google Scholar]
- Byun, E.; Jeong, G.S.; An, R.B.; Min, T.S.; Kim, Y.C. Tribuli fructus constituents protect against tacrine-induced cytotoxicity in HepG2 cells. Arch. Pharm. Res 2010, 33, 67–70. [Google Scholar]
- Li, B.; Lee, D.S.; Kang, Y.; Yao, N.Q.; An, R.B.; Kim, Y.C. Protective effect of ganodermanondiol isolated from the Lingzhi mushroom against tert-butyl hydroperoxide-induced hepatotoxicity through Nrf2-mediated antioxidant enzymes. Food Chem. Toxicol 2013, 53, 317–324. [Google Scholar]
- Copple, I.M.; Goldring, C.E.; Kitteringham, N.R.; Park, B.K. The Nrf2-Keap1 defence pathway: Role in protection against drug-induced toxicity. Toxicology 2008, 246, 24–33. [Google Scholar]
- Choi, A.M.; Alam, J. Heme oxygenase-1: Function, regulation, and implication of a novel stress-inducible protein in oxidant-induced lung injury. Am. J. Respir. Cell Mol. Biol 1996, 15, 9–19. [Google Scholar]
- Hwang, Y.P.; Choi, J.H.; Choi, J.M.; Chung, Y.C.; Jeong, H.G. Protective mechanisms of anthocyanins from purple sweet potato against tert-butyl hydroperoxide-induced hepatotoxicity. Food Chem. Toxicol 2011, 49, 2081–2089. [Google Scholar]
- Jeong, G.S.; Lee, D.S.; Li, B.; Byun, E.; Kwon, D.Y.; Park, H.; Kim, Y.C. Protective effect of sauchinone by upregulating heme oxygenase-1 via the P38 MAPK and Nrf2/ARE pathways in HepG2 cells. Planta Med 2010, 76, 41–47. [Google Scholar]
- Surh, Y.J. Cancer chemoprevention with dietary phytochemicals. Nat. Rev. Cancer 2003, 3, 768–778. [Google Scholar]
- Farombi, E.O.; Shrotriya, S.; Na, H.K.; Kim, S.H.; Surh, Y.J. Curcumin attenuates dimethylnitrosamine-induced liver injury in rats through Nrf-2-mediated induction of heme oxygenase-1. Food Chem. Toxicol 2008, 46, 1279–1287. [Google Scholar]
- Yao, P.; Nussler, A.; Liu, L.; Hao, L.; Song, F.; Schirmeier, A.; Nussler, N. Quercetin protects human Hepatoma cells from ethanol-derived oxidative stress by inducing heme oxygenase-1 via the MAPK/Nrf-2 pathway. J. Hepatol 2007, 47, 253–261. [Google Scholar]
- Kietzmann, T.; Samoylenko, A.; Immenschuh, S. Transcriptional regulation of heme oxygenase-1 gene expression by MAP kinases of the JNK and p38 pathways in primary cultures of rat hepatocytes. J. Biol. Chem 2003, 278, 17927–17936. [Google Scholar]
- Choi, B.M.; Kim, Y.M.; Jeong, Y.R.; Pae, H.O.; Song, C.E.; Park, J.E.; Ahn, Y.K.; Chung, H.T. Induction of heme oxygenase-1 is involved in anti-proliferative effects of paclitaxel on rat vascular smooth muscle cells. Biochem. Biophys. Res. Commun 2004, 321, 132–137. [Google Scholar]
- Yu, R.; Lei, W.; Mandlekar, S.; Weber, M.J.; Der, C.J.; Wu, J.; Kong, A.N. Role of a mitogen-activated protein kinase pathway in the induction of phase II detoxifying enzymes by chemicals. J. Biol. Chem 1999, 274, 27545–27552. [Google Scholar]
- Koehn, F.E.; Carter, G.T. The evolving role of natural products in drug discovery. Nat. Rev. Drug Discov 2005, 4, 206–220. [Google Scholar]
- Paterson, I.; Anderson, E.A. The renaissance of natural products as drug candidates. Science 2005, 310, 451–453. [Google Scholar]


© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lee, D.-S.; Kim, K.-S.; Ko, W.; Li, B.; Jeong, G.-S.; Jang, J.-H.; Oh, H.; Kim, Y.-C. The Cytoprotective Effect of Sulfuretin against tert-Butyl Hydroperoxide-Induced Hepatotoxicity through Nrf2/ARE and JNK/ERK MAPK-Mediated Heme Oxygenase-1 Expression. Int. J. Mol. Sci. 2014, 15, 8863-8877. https://doi.org/10.3390/ijms15058863
Lee D-S, Kim K-S, Ko W, Li B, Jeong G-S, Jang J-H, Oh H, Kim Y-C. The Cytoprotective Effect of Sulfuretin against tert-Butyl Hydroperoxide-Induced Hepatotoxicity through Nrf2/ARE and JNK/ERK MAPK-Mediated Heme Oxygenase-1 Expression. International Journal of Molecular Sciences. 2014; 15(5):8863-8877. https://doi.org/10.3390/ijms15058863
Chicago/Turabian StyleLee, Dong-Sung, Kyoung-Su Kim, Wonmin Ko, Bin Li, Gil-Saeng Jeong, Jun-Hyeog Jang, Hyuncheol Oh, and Youn-Chul Kim. 2014. "The Cytoprotective Effect of Sulfuretin against tert-Butyl Hydroperoxide-Induced Hepatotoxicity through Nrf2/ARE and JNK/ERK MAPK-Mediated Heme Oxygenase-1 Expression" International Journal of Molecular Sciences 15, no. 5: 8863-8877. https://doi.org/10.3390/ijms15058863
APA StyleLee, D.-S., Kim, K.-S., Ko, W., Li, B., Jeong, G.-S., Jang, J.-H., Oh, H., & Kim, Y.-C. (2014). The Cytoprotective Effect of Sulfuretin against tert-Butyl Hydroperoxide-Induced Hepatotoxicity through Nrf2/ARE and JNK/ERK MAPK-Mediated Heme Oxygenase-1 Expression. International Journal of Molecular Sciences, 15(5), 8863-8877. https://doi.org/10.3390/ijms15058863





