Study on Biopharmaceutics Classification and Oral Bioavailability of a Novel Multikinase Inhibitor NCE for Cancer Therapy
Abstract
:1. Introduction
2. Results and Discussion
2.1. Solubility and Intrinsic Dissolution Ratio of NCE
2.2. Stability of NCE in Perfusion Buffers
2.3. The Effect of Intestinal Site and NCE Concentration on Intestinal Permeability
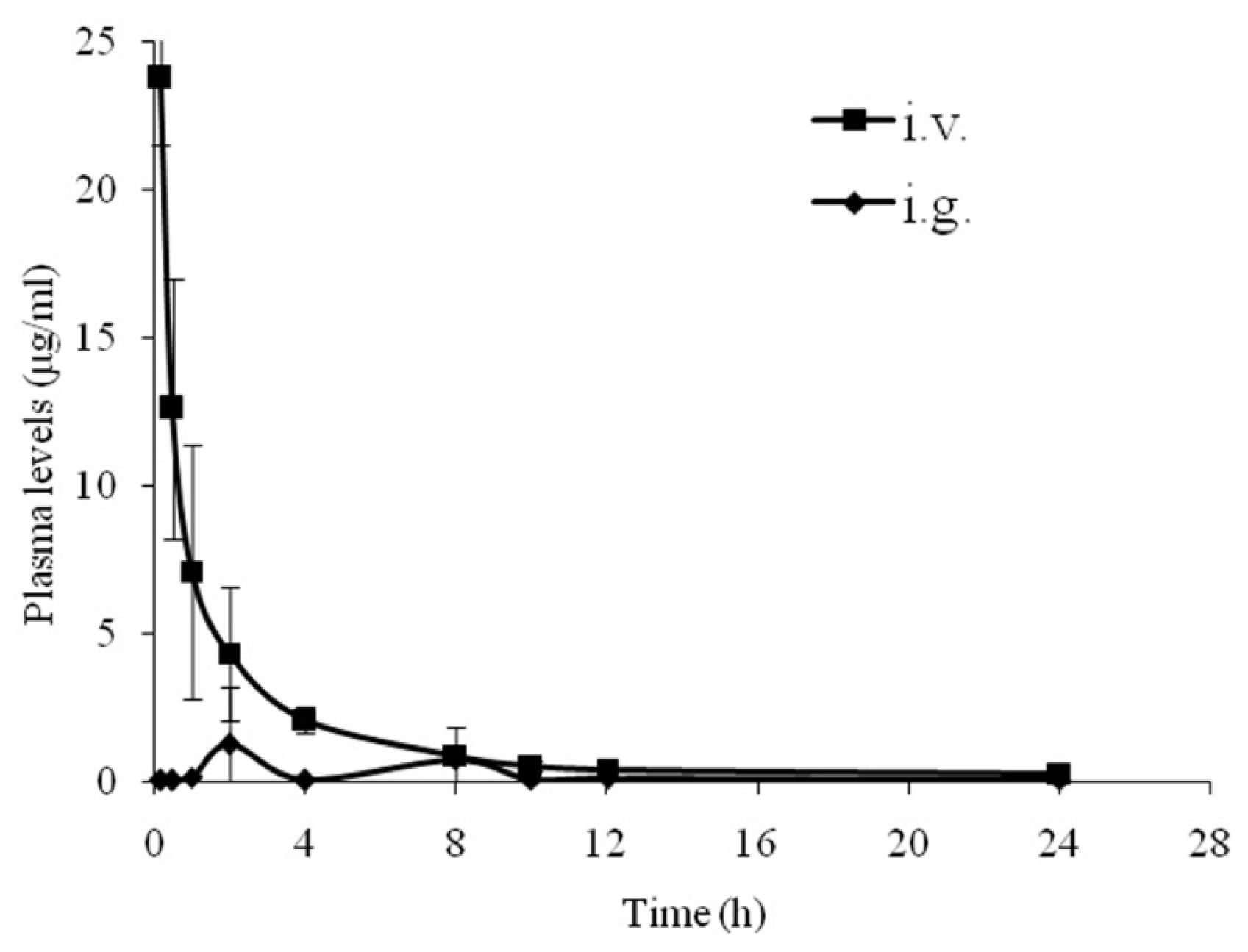
2.4. Pharmacokinetic Parameters of NCE in Beagle Dogs
3. Experimental Section
3.1. Materials
3.2. Solubility Determination
3.3. Dissolution-Based Classification Intrinsic Dissolution Ratio (IDR) Measurement
3.4. Stability Study
3.5. In Situ Single-Pass Perfusion (SPIP) Experiments
3.6. Pharmacokinetic Studies in Beagle Dogs
3.7. HPLC Analytical Method
3.8. IDR Calculation
3.9. Effective Permeability Coefficient (Peff) Calculation
3.10. Pharmacokinetic Parameters Calculation
3.11. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsThe authors Yang Yang and Chun-Mei Fan were in charge of the whole project design, experiments and paper written collectively. Xuan-He and Jin-Kun Zhang were responsible partly for the rat intestine permeability experiment sections. Ke Ren and Ying-Ju He had guided the IDR assays. Luo-Ting Yu and Ying-Lan Zhao helped to design the pharmacokinetic experiments. Chang-Yang Gong and Yu Zheng were in charge of the statistical analysis. Xiang-Rong Song and Jun Zeng had guided the conception and revision of this paper collectively.
References
- Luo, X.; Li, S.; Xie, Y.; He, J.; Li, J.; Lin, H.; Wang, N.; Yang, S.; Zhao, Y.; Yu, L.; et al. Pharmacokinetic studies of a novel multikinase inhibitor for treating cancer by HPLC-UV. J. Chromatogr. Sci 2013, 51, 17–20. [Google Scholar]
- Amidon, G.L.; Lennernas, H.; Shah, V.P.; Crison, J.R. A theoretical basis for a biopharmaceutic drug classification: The correlation of in vitro drug product dissolution and in vivo bioavailability. Pharm. Res 1995, 12, 413–420. [Google Scholar]
- Peternel, L.; Kristan, K.; Petrusevska, M.; Rizner, T.L.; Legen, I. Suitability of isolated rat jejunum model for demonstration of complete absorption in humans for BCS-based biowaiver request. J. Pharm. Sci 2012, 101, 1436–1449. [Google Scholar]
- Brouwers, J.; Mols, R.; Annaert, P.; Augustijns, P. Validation of a differential in situ perfusion method with mesenteric blood sampling in rats for intestinal drug interaction profiling. Biopharm. Drug Dispos 2010, 31, 278–285. [Google Scholar]
- Butler, J.M.; Dressman, J.B. The developability classification system: Application of biopharmaceutics concepts to formulation development. J. Pharm. Sci 2010, 99, 4940–4954. [Google Scholar]
- Li, H.; Jin, H.E.; Shim, W.S.; Shim, C.K. An improved prediction of the human in vivo intestinal permeability and BCS class of drugs using the in vitro permeability ratio obtained for rat intestine using an Ussing chamber system. Drug Dev. Ind. Pharm 2013, 39, 1515–1522. [Google Scholar]
- US Food and Drug Administration, Guidance for Industry, Waiver of in vivo Bioavailability and Bioequivalence Studies for Immediate-Release Solid Oral Dosage Forms Based on a Biopharmaceutics Classification System; Center for Drug Evaluation and Research: Rockville, MD, USA, 2000.
- Lindenberg, M.; Kopp, S.; Dressman, J.B. Classification of orally administered drugs on the world health organization model list of essential medicines according to the biopharmaceutics classification system. Eur. J. Pharm. Biopharm 2004, 58, 265–278. [Google Scholar]
- Rinaki, E.; Valsami, G.; Macheras, P. Quantitative biopharmaceutics classification system: The central role of dose/solubility ratio. Pharm. Res 2003, 20, 1917–1925. [Google Scholar]
- Volpe, D.A. Application of method suitability for drug permeability classification. AAPS J 2010, 12, 670–678. [Google Scholar]
- Ruan, L.P.; Chen, S.; Yu, B.Y.; Zhu, D.N.; Cordell, G.A.; Qiu, S.X. Prediction of human absorption of natural compounds by the non-everted rat intestinal sac model. Eur. J. Med. Chem 2006, 41, 605–610. [Google Scholar]
- Cao, Z.X.; Zheng, R.L.; Lin, H.J.; Luo, S.D.; Zhou, Y.; Xu, Y.Z.; Zeng, X.X.; Wang, Z.; Zhou, L.N.; Mao, Y.Q.; et al. SKLB610: A novel potential inhibitor of vascular endothelial growth factor receptor tyrosine kinases inhibits angiogenesis and tumor growth in vivo. Cell Physiol. Biochem. 2011, 27, 565–574. [Google Scholar]
- Linnell, R. Dissociation constants of 2-substituted pyridines. J. Org. Chem 1960, 25, 290–290. [Google Scholar]
- Wu, Z.; Razzak, M.; Tucker, I.G.; Medlicott, N.J. Physicochemical characterization of ricobendazole: I. Solubility, lipophilicity, and ionization characteristics. J. Pharm. Sci 2005, 94, 983–993. [Google Scholar]
- Yu, L.X.; Carlin, A.S.; Amidon, G.L.; Hussain, A.S. Feasibility studies of utilizing disk intrinsic dissolution rate to classify drugs. Int. J. Pharm 2004, 270, 221–227. [Google Scholar]
- Zakeri-Milani, P.; Barzegar-Jalali, M.; Azimi, M.; Valizadeh, H. Biopharmaceutical classification of drugs using intrinsic dissolution rate (IDR) and rat intestinal permeability. Eur. J. Pharm. Biopharm 2009, 73, 102–106. [Google Scholar]
- Schurgers, N.; Bijdendijk, J.; Tukker, J.J.; Crommelin, D.J. Comparison of four experimental techniques for studying drug absorption kinetics in the anesthetized rat in situ. J. Pharm. Sci. 1986, 75, 117–119. [Google Scholar]
- Berggren, S.; Hoogstraate, J.; Fagerholm, U.; Lennernas, H. Characterization of jejunal absorption and apical efflux of ropivacaine, lidocaine and bupivacaine in the rat using in situ and in vitro absorption models. Eur. J. Pharm. Sci 2004, 21, 553–560. [Google Scholar]
- Lennernas, H.; Nylander, S.; Ungell, A.L. Jejunal permeability: A comparison between the ussing chamber technique and the single-pass perfusion in humans. Pharm. Res 1997, 14, 667–671. [Google Scholar]
- Sun, D.; Yu, L.X.; Hussain, M.A.; Wall, D.A.; Smith, R.L.; Amidon, G.L. In vitro testing of drug absorption for drug “developability” assessment: Forming an interface between in vitro preclinical data and clinical outcome. Curr. Opin. Drug Discov. Devel 2004, 7, 75–85. [Google Scholar]
- Kesisoglou, F. Use of preclinical dog studies and absorption modeling to facilitate late stage formulation bridging for a BCS II drug candidate. AAPS PharmSciTech 2013, 15, 20–28. [Google Scholar]
- Salphati, L.; Childers, K.; Pan, L.; Tsutsui, K.; Takahashi, L. Evaluation of a single-pass intestinal-perfusion method in rat for the prediction of absorption in man. J. Pharm. Pharmacol 2001, 53, 1007–1013. [Google Scholar]
- You, J.; Li, Q.P.; Yu, Y.W.; Cui, F.D. Absorption of zedoary oil in rat intestine using in situ single pass perfusion model. Yao Xue Xue Bao 2004, 39, 849–853. (In Chinese) [Google Scholar]
- Ma, N.; Wang, J.; Xu, F.; Li, J.; Li, Y.; Xiao, H.; Xiang, D. Studies on absorptive characters of resveratrol derivative (e)-3,5,4′-trimethoxystilbene in rat intestine. Chin. J. Pharm. Anal 2010, 30, 586–591. [Google Scholar]
- Fagerholm, U.; Johansson, M.; Lennernas, H. Comparison between permeability coefficients in rat and human jejunum. Pharm. Res 1996, 13, 1336–1342. [Google Scholar]
- Escribano, E.; Sala, X.G.; Salamanca, J.; Navarro, C.R.; Regue, J.Q. Single-pass intestinal perfusion to establish the intestinal permeability of model drugs in mouse. Int. J. Pharm 2012, 436, 472–477. [Google Scholar]
- Zakeri-Milani, P.; Valizadeh, H.; Tajerzadeh, H.; Azarmi, Y.; Islambolchilar, Z.; Barzegar, S.; Barzegar-Jalali, M. Predicting human intestinal permeability using single-pass intestinal perfusion in rat. J. Pharm. Pharm. Sci 2007, 10, 368–379. [Google Scholar]
- Nagare, N.; Damre, A.; Singh, K.S.; Mallurwar, S.R.; Iyer, S.; Naik, A.; Chintamaneni, M. Determination of site of absorption of propranolol in rat gut using in situ single-pass intestinal perfusion. Indian J. Pharm. Sci 2010, 72, 625–629. [Google Scholar]
- Cook, T.J.; Shenoy, S.S. Intestinal permeability of chlorpyrifos using the single-pass intestinal perfusion method in the rat. Toxicology 2003, 184, 125–133. [Google Scholar]
- Ma, Y.M.; Sun, R.Y. Second peak of plasma diazepam concentration and enterogastric circulation. Zhongguo Yao Li Xue Bao 1993, 14, 218–221. (In Chinese) [Google Scholar]
- Yang, Y.; Fan, C.; Zhang, J.; He, X.; Yu, Y.; Fang, D.; He, Y.; Song, X. Intestinal absorption characteristics and pharmacokinetics of the novel targeted small-molecular NCE in rats. Chin. J. New Drugs 2013, 21, 2542–2546. (In Chinese) [Google Scholar]
- Wang, Z.; Hu, H.; Chen, F.; Lan, K.; Wang, A. Reduced system exposures of total rhein and baicalin after combinatory oral administration of rhein, baicalin and berberine to beagle dogs and rats. J. Ethnopharm 2013, 145, 442–449. [Google Scholar]
- Wood, J.; Syarto, J.; Letterman, H. Improved holder for intrinsic dissolution rate studies. J. Pharm. Sci 1965, 54, 1068. [Google Scholar]
- Zhang, J.; Liu, D.; Huang, Y.; Gao, Y.; Qian, S. Biopharmaceutics classification and intestinal absorption study of apigenin. Int. J. Pharm 2012, 436, 311–317. [Google Scholar]
- Incecayir, T.; Tsume, Y.; Amidon, G.L. Comparison of the permeability of metoprolol and labetalol in rat, mouse, and Caco-2 cells: Use as a reference standard for BCS classification. Mol. Pharm 2013, 10, 958–966. [Google Scholar]
- Li, M.; Si, L.; Pan, H.; Rabba, A.K.; Yan, F.; Qiu, J.; Li, G. Excipients enhance intestinal absorption of ganciclovir by P-gp inhibition: Assessed in vitro by everted gut sac and in situ by improved intestinal perfusion. Int. J. Pharm 2011, 403, 37–45. [Google Scholar]
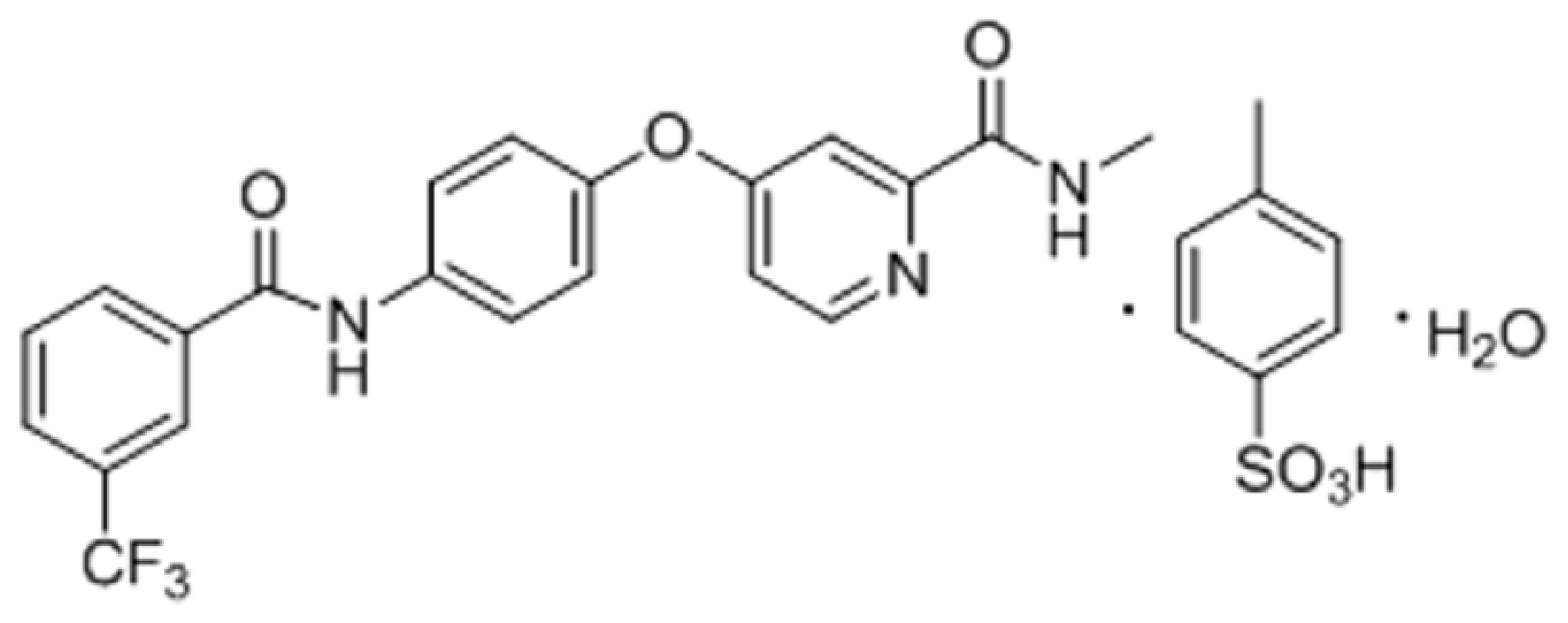

| Buffers pH | Solubility (μg/mL) | SD |
|---|---|---|
| 1.0 | 81.73 | 0.13 |
| 5.0 | 08.50 | 0.04 |
| 6.8 | 09.30 | 0.06 |
| 7.4 | 10.00 | 0.09 |
| Temperature | 20 °C | 4 °C | ||||
|---|---|---|---|---|---|---|
| 0 h | 2 h | 4 h | 6 h | 24 h | 48 h | |
| Detected (μg/mL) | 19.83 | 19.66 | 19.49 | 19.32 | 19.16 | 18.99 |
| Rate of change (%) | - | 0.86 | 1.70 | 2.55 | 3.38 | 4.21 |
| Perfusate (μg/mL) | Peff (10−4 cm/s) | ||
|---|---|---|---|
| Duodeum | Jejunum | Ileum | |
| NCE | 0.26 ± 0.29 | 0.25 ± 0.24 N.S. | 0.23 ± 0.18 N.S. |
| 0.4% SDS + NCE | 0.26 ± 0.25 N.S. | 0.25 ± 0.22 N.S. | 0.22 ± 0.15 N.S. |
| Perfusate (40 μg/mL) | Peff (10−4 cm/s) | ||
|---|---|---|---|
| Duodeum | Jejunum | Ileum | |
| NCE | 0.49 ± 0.29 * | 0.56 ± 0.26 N.S. * | 0.52 ± 0.28 N.S. * |
| Theophylline | 0.74 ± 0.02 * | 0.64 ± 0.01 N.S. * | 0.63 ± 0.04 N.S. * |
| Ranitidine | 0.15 ± 0.07 * | 0.19 ± 0.07 N.S. * | 0.17 ± 0.02 N.S * |
| Perfusate (μg/mL) | Peff (10−4 cm/s) | ||
|---|---|---|---|
| Duodeum | Jejunum | Ileum | |
| 2 | 0.21 ± 0.18 | 0.20 ± 0.30 N.S. | 0.23 ± 0.25 N.S. |
| 20 | 0.34 ± 0.12 | 0.30 ± 0.15 N.S. | 0.29 ± 0.10 N.S. |
| 40 | 0.49 ± 0.29 | 0.56 ± 0.26 N.S. | 0.52 ± 0.28 N.S. |
| Route | Beagles | Cmax (μg/L) | Tmax (h) | T1/2 (h) | AUC0−t (mg/L·h) | MRT (h) | F (%) |
|---|---|---|---|---|---|---|---|
| i.v. | Beagle 1 | 26.01 | 0.23 | 6.39 | 56.25 | 1.84 | - |
| Beagle 2 | 23.67 | 0.30 | 3.56 | 34.41 | 1.68 | - | |
| Beagle3 | 21.56 | 0.22 | 9.23 | 25.11 | 1.35 | - | |
| mean ± SD | 23.75 ± 2.23 | 0.25 ± 0.04 | 6.39 ± 2.83 | 38.59 ± 15.99 | 1.62 ± 0.25 | - | |
| i.g. | Beagle 4 | 12.19 | 2.00 | 12.28 | 6.86 | 10.79 | 12.19 |
| Beagle 5 | 0.86 | 1.73 | 17.34 | 13.61 | 10.57 | 39.56 | |
| Beagle 6 | 2.06 | 2.00 | 14.92 | 7.16 | 11.19 | 28.51 | |
| mean ± SD | 2.14 ±1.32 | 1.91 ± 0.15 | 14.85 ± 2.53 | 9.21 ± 3.82 | 10.85 ± 0.31 | 26.75 ± 13.77 | |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, Y.; Fan, C.-M.; He, X.; Ren, K.; Zhang, J.-K.; He, Y.-J.; Yu, L.-T.; Zhao, Y.-L.; Gong, C.-Y.; Zheng, Y.; et al. Study on Biopharmaceutics Classification and Oral Bioavailability of a Novel Multikinase Inhibitor NCE for Cancer Therapy. Int. J. Mol. Sci. 2014, 15, 7199-7212. https://doi.org/10.3390/ijms15057199
Yang Y, Fan C-M, He X, Ren K, Zhang J-K, He Y-J, Yu L-T, Zhao Y-L, Gong C-Y, Zheng Y, et al. Study on Biopharmaceutics Classification and Oral Bioavailability of a Novel Multikinase Inhibitor NCE for Cancer Therapy. International Journal of Molecular Sciences. 2014; 15(5):7199-7212. https://doi.org/10.3390/ijms15057199
Chicago/Turabian StyleYang, Yang, Chun-Mei Fan, Xuan He, Ke Ren, Jin-Kun Zhang, Ying-Ju He, Luo-Ting Yu, Ying-Lan Zhao, Chang-Yang Gong, Yu Zheng, and et al. 2014. "Study on Biopharmaceutics Classification and Oral Bioavailability of a Novel Multikinase Inhibitor NCE for Cancer Therapy" International Journal of Molecular Sciences 15, no. 5: 7199-7212. https://doi.org/10.3390/ijms15057199
APA StyleYang, Y., Fan, C.-M., He, X., Ren, K., Zhang, J.-K., He, Y.-J., Yu, L.-T., Zhao, Y.-L., Gong, C.-Y., Zheng, Y., Song, X.-R., & Zeng, J. (2014). Study on Biopharmaceutics Classification and Oral Bioavailability of a Novel Multikinase Inhibitor NCE for Cancer Therapy. International Journal of Molecular Sciences, 15(5), 7199-7212. https://doi.org/10.3390/ijms15057199




