The Reliability and Predictive Ability of a Biomarker of Oxidative DNA Damage on Functional Outcomes after Stroke Rehabilitation
Abstract
:1. Introduction
2. Results
2.1. Characteristics of the Stroke Patients
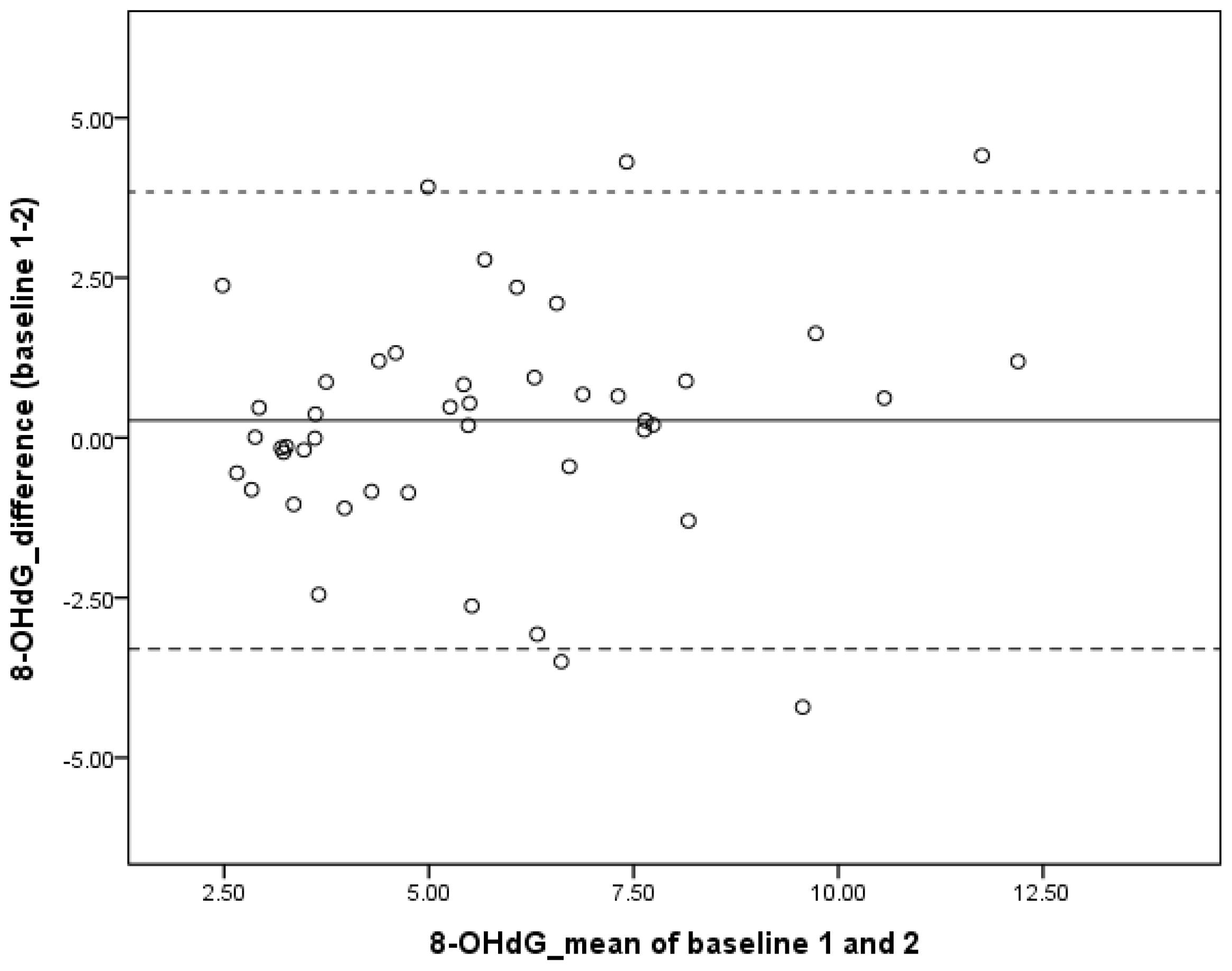
2.2. Test-Retest Reliability of 8-OHdG
2.3. Correlations between 8-OHdG and Functional Outcomes
2.4. Predictive Ability of Baseline 8-OHdG on Post-Stroke Functional Outcomes
2.5. Effect of Rehabilitation on 8-OHdG and Functional Outcomes
3. Discussion
Limitations and Further Suggestions
4. Experimental Section
4.1. Participants
4.2. Rehabilitation Intervention
4.3. Urine Sample Collection
4.4. Determination of 8-OHdG Levels by LC-MS/MS
4.5. Functional Outcomes
4.5.1. Fugl-Meyer Assessment (FMA)
4.5.2. Medical Research Council (MRC) Scale
4.5.3. Assessment of Pain and Fatigue
4.6. Statistical Analysis
5. Conclusions
Acknowledgments
Conflicts of Interest
- Author ContributionsY.W.H., K.C.L. and C.Y.W. contributed for the study design, grant and project management. Y.W.H. and M.K. analyses the data, interpretation, and drafted the manuscript. Y.W.H. involved in data collection. T.H.L. provided medical consultation. K.Y.W. involved in biomarker analysis and provided consultation. All authors have approved the final version of the article.
References
- Offord, E.; van Poppel, G.; Tyrrell, R. Markers of oxidative damage and antioxidant protection: Current status and relevance to disease. Free Radic. Res 2000, 33, S5–S19. [Google Scholar]
- Cherubini, A.; Ruggiero, C.; Polidori, M.C.; Mecocci, P. Potential markers of oxidative stress in stroke. Free Radic. Biol. Med 2005, 39, 841–852. [Google Scholar]
- Brewer, L.; Horgan, F.; Hickey, A.; Williams, D. Stroke rehabilitation: Recent advances and future therapies. QJM 2013, 106, 11–25. [Google Scholar]
- Chen, S.; Li, N.; Deb-Chatterji, M.; Dong, Q.; Kielstein, J.; Weissenborn, K.; Worthmann, H. Asymmetric dimethyarginine as marker and mediator in ischemic stroke. Int. J. Mol. Sci 2012, 13, 15983–16004. [Google Scholar]
- Cherubini, A.; Polidori, M.C.; Bregnocchi, M.; Pezzuto, S.; Cecchetti, R.; Ingegni, T.; di Iorio, A.; Senin, U.; Mecocci, P. Antioxidant profile and early outcome in stroke patients. Stroke 2000, 31, 2295–2300. [Google Scholar]
- Mizukoshi, G.; Katsura, K.; Katayama, Y. Urinary 8-hydroxy-2′-deoxyguanosine and serum S100beta in acute cardioembolic stroke patients. Neurol. Res 2005, 27, 644–646. [Google Scholar]
- Bémeur, C.; Ste-Marie, L.; Montgomery, J. Increased oxidative stress during hyperglycemic cerebral ischemia. Neurochem. Int 2007, 50, 890–904. [Google Scholar]
- Chuang, K.Y.; Wu, S.C.; Yeh, M.C.; Chen, Y.H.; Wu, C.L. Exploring the associations between long-term care and mortality rates among stroke patients. J. Nurs. Res 2005, 13, 66–74. [Google Scholar]
- Hsieh, Y.W.; Wu, C.Y.; Lin, K.C.; Chang, Y.F.; Chen, C.L.; Liu, J.S. Responsiveness and validity of three outcome measures of motor function after stroke rehabilitation. Stroke 2009, 40, 1386–1391. [Google Scholar]
- Stoller, O.; de Bruin, E.D.; Knols, R.H.; Hunt, K.J. Effects of cardiovascular exercise early after stroke: Systematic review and meta-analysis. BMC Neurol 2012, 12, 45. [Google Scholar]
- Lin, K.C.; Huang, P.C.; Chen, Y.T.; Wu, C.Y.; Huang, W.L. Combining afferent stimulation and mirror therapy for rehabilitating motor function, motor control, ambulation, and daily functions after stroke. Neurorehabil. Neural. Repair 2014, 28, 153–162. [Google Scholar]
- Nave, A.H.; Kröber, J.M.; Brunecker, P.; Fiebach, J.; List, J.; Grittner, U.; Endres, M.; Meisel, A.; Floel, A.; Ebinger, M. Biomarkers and perfusion-training-induced changes after stroke (BAPTISe): Protocol of an observational study accompanying a randomized controlled trial. BMC Neurol 2013, 13, 197. [Google Scholar]
- Banerjee, A.K.; Mandal, A.; Chanda, D.; Chakraborti, S. Oxidant, antioxidant and physical exercise. Mol. Cell Biochem 2003, 253, 307–312. [Google Scholar]
- Gordon, C.D.; Wilks, R.; McCaw-Binns, A. Effect of aerobic exercise (walking) training on functional status and health-related quality of life in chronic stroke survivors: A randomized controlled trial. Stroke 2013, 44, 1179–1181. [Google Scholar]
- Pinter, M.M.; Brainin, M. Rehabilitation after stroke in older people. Maturitas 2012, 71, 104–108. [Google Scholar]
- Nakajima, H.; Unoda, K.; Ito, T.; Kitaoka, H.; Kimura, F.; Hanafusa, T. The relation of urinary 8-OHdG, a marker of oxidative stress to DNA, and clinical outcomes for ischemic stroke. Open Neurol. J 2012, 6, 51–57. [Google Scholar]
- Hu, G.C.; Hsieh, S.F.; Chen, Y.M.; Hu, Y.N.; Kang, C.L.; Chien, K.L. The prognostic roles of initial glucose level and functional outcomes in patients with ischemic stroke: Difference between diabetic and nondiabetic patients. Disabil. Rehabil 2012, 34, 34–39. [Google Scholar]
- Palleschi, S.; de Angelis, S.; Diana, L.; Rossi, B.; Papa, V.; Severini, G.; Splendiani, G. Reliability of oxidative stress biomarkers in hemodialysis patients: A comparative study. Clin. Chem. Lab. Med 2007, 45, 1211–1218. [Google Scholar]
- Gedik, C.M.; Boyle, S.P.; Wood, S.G.; Vaughan, N.J.; Collins, A.R. Oxidative stress in humans: Validation of biomarkers of DNA damage. Carcinogenesis 2002, 23, 1441–1446. [Google Scholar]
- Portney, L.G.; Watkins, M.P. Foundations of Clinical Research: Applications to Practice; Pearson/Prentice Hall: Upper Saddle River, NJ, USA, 2009. [Google Scholar]
- Yong, M.; Kaste, M. Dynamic of hyperglycemia as a predictor of stroke outcome in the ECASS-II trial. Stroke 2008, 39, 2749–2755. [Google Scholar]
- Kwakkel, G.; Veerbeek, J.M.; van Wegen, E.E.; Nijland, R.; Harmeling-van der Wel, B.C.; Dippel, D.W. Predictive value of the NIHSS for ADL outcome after ischemic hemispheric stroke: Does timing of early assessment matter? J. Neurol. Sci 2010, 294, 57–61. [Google Scholar]
- Korivi, M.; Wu, C.Y.; Lin, K.C. Potential predictive values of inflammatory biomarkers for stroke rehabilitation outcomes. J. Formos. Med. Assoc 2013, 112, 735–737. [Google Scholar]
- Radák, Z.; Apor, P.; Pucsok, J.; Berkes, I.; Ogonovszky, H.; Pavlik, G.; Nakamoto, H.; Goto, S. Marathon running alters the DNA base excision repair in human skeletal muscle. Life Sci 2003, 72, 1627–1633. [Google Scholar]
- Parise, G.; Brose, A.; Tarnopolsky, M. Resistance exercise training decreases oxidative damage to DNA and increases cytochrome oxidase activity in older adults. Exp. Gerontol 2005, 40, 173–180. [Google Scholar]
- Parise, G.; Phillips, S.M.; Kaczor, J.J.; Tarnopolsky, M.A. Antioxidant enzyme activity is up-regulated after unilateral resistance exercise training in older adults. Free Radic. Biol. Med 2005, 39, 289–295. [Google Scholar]
- Hsieh, Y.W.; Wu, C.Y.; Liao, W.W.; Lin, K.C.; Wu, K.Y.; Lee, C.Y. Effects of treatment intensity in upper limb robot-assisted therapy for chronic stroke: A pilot randomized controlled trial. Neurorehabil. Neural. Repair 2011, 25, 503–511. [Google Scholar]
- Hu, C.W.; Wu, M.T.; Chao, M.R.; Pan, C.H.; Wang, C.J.; Swenberg, J.A.; Wu, K.Y. Comparison of analyses of urinary 8-hydroxy-2′-deoxyguanosine by isotope-dilution liquid chromatography with electrospray tandem mass spectrometry and by enzyme-linked immunosorbent assay. Rapid Commun. Mass Spectrom 2004, 18, 505–510. [Google Scholar]
- Fugl-Meyer, A.R.; Jääskö, L.; Leyman, I.; Olsson, S.; Steglind, S. The post-stroke hemiplegic patient. 1. A method for evaluation of physical performance. Scand. J. Rehabil. Med 1975, 7, 13–31. [Google Scholar]
- Platz, T.; Pinkowski, C.; van Wijck, F.; Kim, I.H.; di Bella, P.; Johnson, G. Reliability and validity of arm function assessment with standardized guidelines for the Fugl-Meyer Test, Action Research Arm Test and Box and Block Test: A multicentre study. Clin. Rehabil 2005, 19, 404–411. [Google Scholar]
- Gregson, J.M.; Leathley, M.J.; Moore, A.P.; Smith, T.L.; Sharma, A.K.; Watkins, C.L. Reliability of measurements of muscle tone and muscle power in stroke patients. Age Ageing 2000, 29, 223–228. [Google Scholar]
- Benaim, C.; Froger, J.; Cazottes, C.; Gueben, D.; Porte, M.; Desnuelle, C.; Pelissier, J.Y. Use of the Faces Pain Scale by left and right hemispheric stroke patients. Pain 2007, 128, 52–58. [Google Scholar]
- Christensen, D.; Johnsen, S.P.; Watt, T.; Harder, I.; Kirkevold, M.; Andersen, G. Dimensions of post-stroke fatigue: A two-year follow-up study. Cerebrovasc. Dis 2008, 26, 134–141. [Google Scholar]
- Underwood, J.; Clark, P.C.; Blanton, S.; Aycock, D.M.; Wolf, S.L. Pain, fatigue, and intensity of practice in people with stroke who are receiving constraint-induced movement therapy. Phys. Ther 2006, 86, 1241–1250. [Google Scholar]
- Fleiss, J.L. Statistical Methods for Rates and Proportions; Wiley: New York, NY, USA, 1981. [Google Scholar]
- Andresen, E. Criteria for assessing the tools of disability outcomes research. Arch. Phys. Med. Rehabil 2000, 81, S15–S20. [Google Scholar]
- Bland, J.M.; Altman, D.G. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986, 1, 307–310. [Google Scholar]
| Characteristics | Value |
|---|---|
| Mean ± SD or No. (%) | |
| Age, years | 54.60 ± 10.96 |
| Time after stroke, months | 23.46 ± 14.00 |
| Sex | |
| Male | 39 (63.9) |
| Female | 22 (36.1) |
| Side of stroke | |
| Right | 35 (57.4) |
| Left | 26 (42.6) |
| Stroke type | |
| Ischemic | 37 (60.7) |
| Hemorrhagic | 24 (39.3) |
| Smoker | 3 (4.9) |
| Drinker | 4 (6.6) |
| Antioxidants intake | 26 (42.6) |
| Comorbidities | |
| Hypertension | 41 |
| Diabetes mellitus | 23 |
| Heat disease | 18 |
| Osteoarthritis | 5 |
| Hyperlipidemia | 2 |
| Gout | 1 |
| Epilepsy | 1 |
| Thalassemia | 1 |
| Variable | 8-OHdG (ng/mg creatinine) (Mean ± SD) | Mean difference | ICC (95% CI) |
|---|---|---|---|
| Baseline 1 | 5.87 ± 2.77 | ||
| Baseline 2 | 5.60 ± 2.47 | 0.27 | 0.76 (0.60–0.86) |
| Functional outcome | Pearson r (95% CI) | |
|---|---|---|
| Pre-treatment | Post-treatment | |
| FMA | −0.38* (−0.58 to −0.14) | −0.34* (−0.55 to −0.10) |
| MRC | −0.30* (−0.51 to −0.05) | −0.19 (−0.42 to 0.06) |
| Fatigue | 0.21 (−0.04 to 0.44) | 0.18 (−0.08 to 0.41) |
| Pain | 0.13 (−0.13 to 0.37) | 0.26* (0.10 to 0.48) |
| Functional outcome | Pearson r (95% CI) |
|---|---|
| FMA | −0.34* (−0.55 to −0.10) |
| MRC | −0.31* (−0.52 to −0.06) |
| Fatigue | 0.21 (−0.04 to 0.44) |
| Pain | 0.25* (0 to 0.47) |
| Measurement (units) | Pre-treatment (Mean ± SD) | Post-treatment (Mean ± SD) | p value |
|---|---|---|---|
| 8-OHdG (ng/mg creatinine) | 5.02 ± 2.72 | 4.46 ± 2.37 | 0.04* |
| FMA (0–66) | 43.75 ± 8.96 | 47.70 ± 9.13 | <0.01* |
| MRC (0–5) | 3.57 ± 0.64 | 3.79 ± 0.64 | <0.01* |
| Fatigue (0–10) | 2.32 ± 2.47 | 2.16 ± 2.34 | 0.53 |
| Pain (0–10) | 1.82 ± 2.56 | 1.08 ± 1.83 | 0.02* |
© 2014 by the authors; licensee MDPI, Basel, Switzerland This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hsieh, Y.-W.; Lin, K.-C.; Korivi, M.; Lee, T.-H.; Wu, C.-Y.; Wu, K.-Y. The Reliability and Predictive Ability of a Biomarker of Oxidative DNA Damage on Functional Outcomes after Stroke Rehabilitation. Int. J. Mol. Sci. 2014, 15, 6504-6516. https://doi.org/10.3390/ijms15046504
Hsieh Y-W, Lin K-C, Korivi M, Lee T-H, Wu C-Y, Wu K-Y. The Reliability and Predictive Ability of a Biomarker of Oxidative DNA Damage on Functional Outcomes after Stroke Rehabilitation. International Journal of Molecular Sciences. 2014; 15(4):6504-6516. https://doi.org/10.3390/ijms15046504
Chicago/Turabian StyleHsieh, Yu-Wei, Keh-Chung Lin, Mallikarjuna Korivi, Tsong-Hai Lee, Ching-Yi Wu, and Kuen-Yuh Wu. 2014. "The Reliability and Predictive Ability of a Biomarker of Oxidative DNA Damage on Functional Outcomes after Stroke Rehabilitation" International Journal of Molecular Sciences 15, no. 4: 6504-6516. https://doi.org/10.3390/ijms15046504
APA StyleHsieh, Y.-W., Lin, K.-C., Korivi, M., Lee, T.-H., Wu, C.-Y., & Wu, K.-Y. (2014). The Reliability and Predictive Ability of a Biomarker of Oxidative DNA Damage on Functional Outcomes after Stroke Rehabilitation. International Journal of Molecular Sciences, 15(4), 6504-6516. https://doi.org/10.3390/ijms15046504